Desalinating Real Shale Gas Wastewater by Membrane Distillation: Performance and Potentials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Superhydrophobic Membranes Based on Surface Modification
2.3. Characterization of the Membrane
2.4. Water Quality Monitoring
2.5. MD Experiment
2.6. Characterization of Shale Gas Wastewater
3. Results and Discussion
3.1. The Characteristics of the Membrane
3.2. Performance with Simulated Saline Oily Wastewater
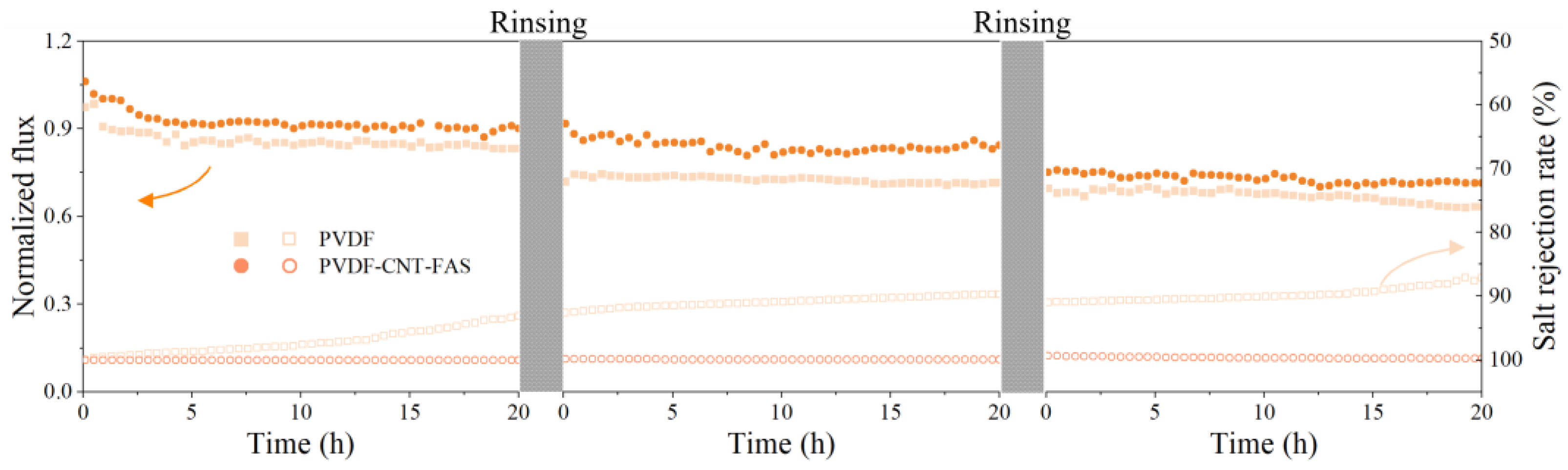
3.3. Flux and Concentration Factor with Real Shale Gas Wastewater
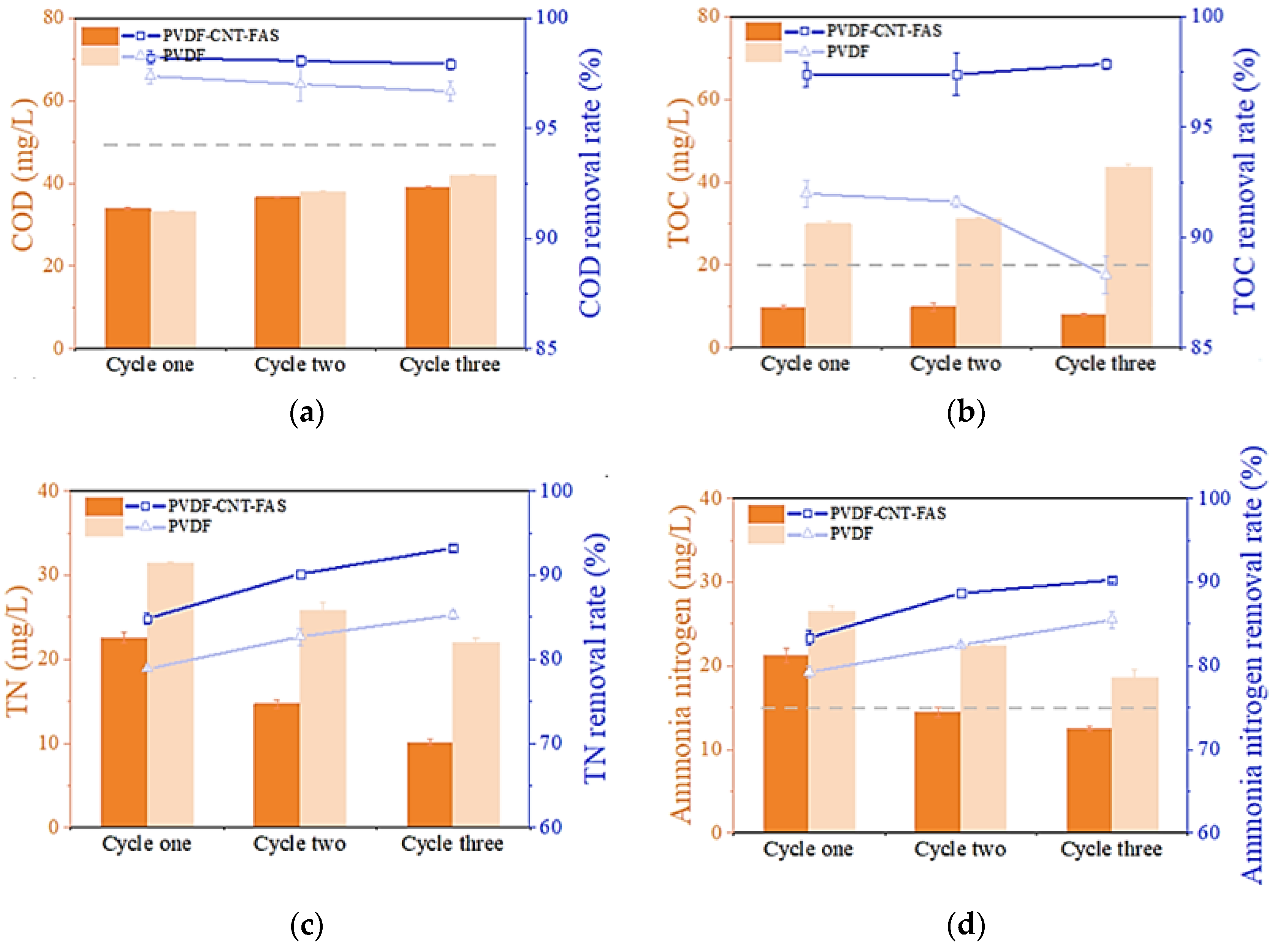
3.4. Permeate Quality with Real Shale Gas Wastewater
3.5. Membrane Fouling with Real Shale Gas Wastewater
3.6. Cost-Effectiveness Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tavakkoli, S.; Lokare, O.; Vidic, R.; Khanna, V. Shale gas produced water management using membrane distillation: An optimization-based approach. Resour. Conserv. Recycl. 2020, 158, 104803. [Google Scholar] [CrossRef]
- Boo, C.; Lee, J.; Elimelech, M. Omniphobic Polyvinylidene Fluoride (PVDF) Membrane for Desalination of Shale Gas Produced Water by Membrane Distillation. Environ. Sci. Technol. 2016, 50, 12275–12282. [Google Scholar] [CrossRef]
- Lan, D.; Chen, M.; Liu, Y.; Liang, Q.; Tu, W.; Chen, Y.; Liang, J. Development of Shale Gas in China and Treatment Options for Wastewater Produced from the Exploitation: Sustainability Lessons from the United States. J. Environ. Eng. 2020, 146, 04020103. [Google Scholar] [CrossRef]
- Wang, H.; Lu, L.; Chen, X.; Bian, Y.; Ren, Z.J. Geochemical and microbial characterizations of flowback and produced water in three shale oil and gas plays in the central and western United States. Water Res. 2019, 164, 114942. [Google Scholar] [CrossRef] [PubMed]
- Butkovskyi, A.; Bruning, H.; Kools, S.A.E.; Rijnaarts, H.H.M.; Van Wezel, A.P. Organic Pollutants in Shale Gas Flowback and Produced Waters: Identification, Potential Ecological Impact, and Implications for Treatment Strategies. Environ. Sci. Technol. 2017, 51, 4740–4754. [Google Scholar] [CrossRef]
- Khawaji, A.D.; Kutubkhanah, I.K.; Wie, J.M. Advances in seawater desalination technologies. Desalination 2008, 221, 47–69. [Google Scholar] [CrossRef]
- Li, Y.; Li, M.; Xiao, K.; Huang, X. Reverse osmosis membrane autopsy in coal chemical wastewater treatment: Evidences of spatially heterogeneous fouling and organic-inorganic synergistic effect. J. Clean Prod. 2020, 246, 118964. [Google Scholar] [CrossRef]
- Martinetti, C.R.; Childress, A.E.; Cath, T.Y. High recovery of concentrated RO brines using forward osmosis and membrane distillation. J. Membr. Sci. 2009, 331, 31–39. [Google Scholar] [CrossRef]
- Alkhudhiri, A.; Darwish, N.; Hilal, N. Membrane distillation: A comprehensive review. Desalination 2012, 287, 2–18. [Google Scholar] [CrossRef]
- Yadav, A.; Labhasetwar, P.K.; Shahi, V.K. Membrane distillation using low-grade energy for desalination: A review. J. Environ. Chem. Eng. 2021, 9, 105818. [Google Scholar] [CrossRef]
- Alklaibi, A.M.; Lior, N. Membrane-distillation desalination: Status and potential. Desalination 2005, 171, 111–131. [Google Scholar] [CrossRef]
- Han, L.; Tan, Y.Z.; Netke, T.; Fane, A.G.; Chew, J.W. Understanding oily wastewater treatment via membrane distillation. J. Membr. Sci. 2017, 539, 284–294. [Google Scholar] [CrossRef]
- Rezaei, M.; Warsinger, D.M.; Lienhard, J.H.; Duke, M.C.; Matsuura, T.; Samhaber, W.M. Wetting phenomena in membrane distillation: Mechanisms, reversal, and prevention. Water Res. 2018, 139, 329–352. [Google Scholar] [CrossRef] [PubMed]
- Horseman, T.; Yin, Y.M.; Christie, K.S.S.; Wang, Z.X.; Tong, T.Z.; Lin, S.H. Wetting, Scaling, and Fouling in Membrane Distillation: State-of-the-Art Insights on Fundamental Mechanisms and Mitigation Strategies. ACS EST Eng. 2021, 1, 117–140. [Google Scholar] [CrossRef]
- Warsinger, D.M.; Swarninathan, J.; Guillen-Burrieza, E.; Arafat, H.A.; Lienhard, J.H. Scaling and fouling in membrane distillation for desalination applications: A review. Desalination 2015, 356, 294–313. [Google Scholar] [CrossRef]
- Lin, S.H.; Nejati, S.; Boo, C.; Hu, Y.X.; Osuji, C.O.; Ehmelech, M. Omniphobic Membrane for Robust Membrane Distillation. Environ. Sci. Technol. Lett. 2014, 1, 443–447. [Google Scholar] [CrossRef]
- Eykens, L.; De Sitter, K.; Dotremont, C.; Pinoy, L.; Van der Bruggen, B. Membrane synthesis for membrane distillation: A review. Sep. Purif. Technol. 2017, 182, 36–51. [Google Scholar] [CrossRef]
- Son, M.; Park, H.; Liu, L.; Choi, H.; Kim, J.H.; Choi, H. Thin-film nanocomposite membrane with CNT positioning in support layer for energy harvesting from saline water. Chem. Eng. J. 2016, 284, 68–77. [Google Scholar] [CrossRef]
- Shan, H.; Liu, J.; Li, X.; Li, Y.; Tezel, F.H.; Li, B.; Wang, S. Nanocoated amphiphobic membrane for flux enhancement and comprehensive anti-fouling performance in direct contact membrane distillation. J. Membr. Sci. 2018, 567, 166–180. [Google Scholar] [CrossRef]
- Wang, Y.; Han, M.; Liu, L.; Yao, J.; Han, L. Beneficial CNT Intermediate Layer for Membrane Fluorination toward Robust Superhydrophobicity and Wetting Resistance in Membrane Distillation. ACS Appl. Mater. Interfaces 2020, 12, 20942–20954. [Google Scholar] [CrossRef]
- Robbins, C.A.; Yin, Y.; Hanson, A.J.; Blotevogel, J.; Borch, T.; Tong, T. Mitigating membrane wetting in the treatment of unconventional oil and gas wastewater by membrane distillation: A comparison of pretreatment with omniphobic membrane. J. Membr. Sci. 2022, 645, 120198. [Google Scholar] [CrossRef]
- Du, X.; Zhang, Z.; Carlson, K.H.; Lee, J.; Tong, T. Membrane fouling and reusability in membrane distillation of shale oil and gas produced water: Effects of membrane surface wettability. J. Membr. Sci. 2018, 567, 199–208. [Google Scholar] [CrossRef]
- Gethard, K.; Sae-Khow, O.; Mitra, S. Water desalination using carbon-nanotube-enhanced membrane distillation. ACS Appl. Mater. Interfaces 2011, 3, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Dong, T.; Hou, D.; Yao, J.; Han, L. Carbon nanotube based Janus composite membrane of oil fouling resistance for direct contact membrane distillation. J. Membr. Sci. 2020, 607, 118078. [Google Scholar] [CrossRef]
- Han, M.; Wang, Y.; Yao, J.; Liu, C.; Chew, J.W.; Wang, Y.; Dong, Y.; Han, L. Electrically conductive hydrophobic membrane cathode for membrane distillation with super anti-oil-fouling capability: Performance and mechanism. Desalination 2021, 516, 115199. [Google Scholar] [CrossRef]
- Zheng, R.; Chen, Y.; Wang, J.; Song, J.; Li, X.-M.; He, T. Preparation of omniphobic PVDF membrane with hierarchical structure for treating saline oily wastewater using direct contact membrane distillation. J. Membr. Sci. 2018, 555, 197–205. [Google Scholar] [CrossRef]
- Chen, Y.; Lu, K.J.; Chung, T.-S. An omniphobic slippery membrane with simultaneous anti-wetting and anti-scaling properties for robust membrane distillation. J. Membr. Sci. 2020, 595, 117572. [Google Scholar] [CrossRef]
- Gu, J.; Xiao, P.; Huang, Y.; Zhang, J.; Chen, T. Controlled functionalization of carbon nanotubes as superhydrophobic material for adjustable oil/water separation. J. Mater. Chem. A 2015, 3, 4124–4128. [Google Scholar] [CrossRef]
- Wang, S.; Liang, S.; Liang, P.; Zhang, X.; Sun, J.; Wu, S.; Huang, X. In-situ combined dual-layer CNT/PVDF membrane for electrically-enhanced fouling resistance. J. Membr. Sci. 2015, 491, 37–44. [Google Scholar] [CrossRef]
- Kharraz, J.A.; Farid, M.U.; Khanzada, N.K.; Deka, B.J.; Arafat, H.A.; An, A.K. Macro-corrugated and nano-patterned hierarchically structured superomniphobic membrane for treatment of low surface tension oily wastewater by membrane distillation. Water Res. 2020, 174, 115600. [Google Scholar] [CrossRef]
- Christie, K.S.S.; Yin, Y.; Lin, S.; Tong, T. Distinct Behaviors between Gypsum and Silica Scaling in Membrane Distillation. Environ. Sci. Technol. 2019, 54, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Zhu, Z.; Chang, H.; Fan, G.; Wang, Q.; Fu, X.; Qu, F.; Liang, H. Integrated membrane electrochemical reactor-membrane distillation process for enhanced landfill leachate treatment. Water Res. 2023, 230, 119559. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, N.; Chilkoor, G.; Wilder, J.; Ren, Z.J.; Gadhamshetty, V. Comparative performances of microbial capacitive deionization cell and microbial fuel cell fed with produced water from the Bakken shale. Bioelectrochemistry 2018, 121, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Qihang, X.; Zejun, Z.; Yong, H.; Yi, Z.; Shibin, X. Combined effect of ozone and PTFE membrane on treating shale gas produced wastewater in Fuling Shale Gas Field. Desalin. Water Treat. 2019, 140, 183–188. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Lee, D.W.; Cho, J. Application of direct contact membrane distillation process to treat anaerobic digestate. J. Membr. Sci. 2016, 511, 20–28. [Google Scholar] [CrossRef]
- Chen, C.; Dong, T.; Han, M.; Yao, J.; Han, L. Ammonium recovery from wastewater by Donnan Dialysis: A feasibility study. J. Clean Prod. 2020, 265, 121838. [Google Scholar] [CrossRef]
- Chen, C.; Dai, Z.; Li, Y.; Zeng, Q.; Yu, Y.; Wang, X.; Zhang, C.; Han, L. Fouling-free membrane stripping for ammonia recovery from real biogas slurry. Water Res. 2023, 229, 119453. [Google Scholar] [CrossRef]
- Li, F.; Huang, J.; Xia, Q.; Lou, M.; Yang, B.; Tian, Q.; Liu, Y. Direct contact membrane distillation for the treatment of industrial dyeing wastewater and characteristic pollutants. Sep. Purif. Technol. 2018, 195, 83–91. [Google Scholar] [CrossRef]
- Cho, H.; Choi, Y.; Lee, S. Effect of pretreatment and operating conditions on the performance of membrane distillation for the treatment of shale gas wastewater. Desalination 2018, 437, 195–209. [Google Scholar] [CrossRef]
- Han, L.; Xiao, T.; Tan, Y.Z.; Fane, A.G.; Chew, J.W. Contaminant rejection in the presence of humic acid by membrane distillation for surface water treatment. J. Membr. Sci. 2017, 541, 291–299. [Google Scholar] [CrossRef]
- Al-Obaidani, S.; Curcio, E.; Macedonio, F.; Di Profio, G.; Ai-Hinai, H.; Drioli, E. Potential of membrane distillation in seawater desalination: Thermal efficiency, sensitivity study and cost estimation. J. Membr. Sci. 2008, 323, 85–98. [Google Scholar] [CrossRef]
- Kesieme, U.K.; Milne, N.; Aral, H.; Cheng, C.Y.; Duke, M. Economic analysis of desalination technologies in the context of carbon pricing, and opportunities for membrane distillation. Desalination 2013, 323, 66–74. [Google Scholar] [CrossRef] [Green Version]
- Tavakkoli, S.; Lokare, O.R.; Vidic, R.D.; Khanna, V. A techno-economic assessment of membrane distillation for treatment of Marcellus shale produced water. Desalination 2017, 416, 24–34. [Google Scholar] [CrossRef]
- Khayet, M. Solar desalination by membrane distillation: Dispersion in energy consumption analysis and water production costs (a review). Desalination 2013, 308, 89–101. [Google Scholar] [CrossRef]




| Zeta Potential (mv) | Conductivity (mS/cm) | TDS (g/L) | TOC (mg/L) | TN (mg/L) | COD (mg/L) | NH4+ (mg/L) | pH | NTU |
| −4.7 | 41.4 | 21.5 | 372 | 153 | 1921 | 131 | 7.2 | 82 |
| Na (mg/L) | K (mg/L) | Ca (mg/L) | Mg (mg/L) | Ba (mg/L) | Fe (mg/L) | Mn (mg/L) | Cl (mg/L) | Si (mg/L) |
| 360,005 | 778 | 6749 | 30,850 | 4781 | 14,076 | 41 | 30,647 | 271,206 |
| Characteristic | PVDF | PVDF-CNT-FAS |
|---|---|---|
| Thickness δ (μm) | 125 ± 2 | 140 ± 6 |
| Pore diammater r (μm) | 0.22 ± 0.006 | 0.18 ± 0.003 |
| Slip angle θ0 (°) | >10° | <5° |
| Water contact angle θ1 (°) | 124 ± 3 | 180 |
| Mineral oil contact angle θ2 (°) | 42 ± 6 | 103 ± 2 |
| SEM image of the membrane’s plain surface |  |  |
| SEM image of the membrane’s cross-section |  |  |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhang, H.; Zhao, R.; Wang, D.; Zhou, L.; Han, L. Desalinating Real Shale Gas Wastewater by Membrane Distillation: Performance and Potentials. Water 2023, 15, 439. https://doi.org/10.3390/w15030439
Wang Y, Zhang H, Zhao R, Wang D, Zhou L, Han L. Desalinating Real Shale Gas Wastewater by Membrane Distillation: Performance and Potentials. Water. 2023; 15(3):439. https://doi.org/10.3390/w15030439
Chicago/Turabian StyleWang, Yuting, Haoquan Zhang, Ruixue Zhao, Die Wang, Lu Zhou, and Le Han. 2023. "Desalinating Real Shale Gas Wastewater by Membrane Distillation: Performance and Potentials" Water 15, no. 3: 439. https://doi.org/10.3390/w15030439







