Pollution Source Identification and Suitability Assessment of Groundwater Quality for Drinking Purposes in Semi-Arid Regions of the Southern Part of India
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sample Collection and Analysis
2.3. Entropy Water Quality Index (EWQI)
2.4. Weighted Overlay Analysis (WOA)
2.5. Saturation Index (SI)
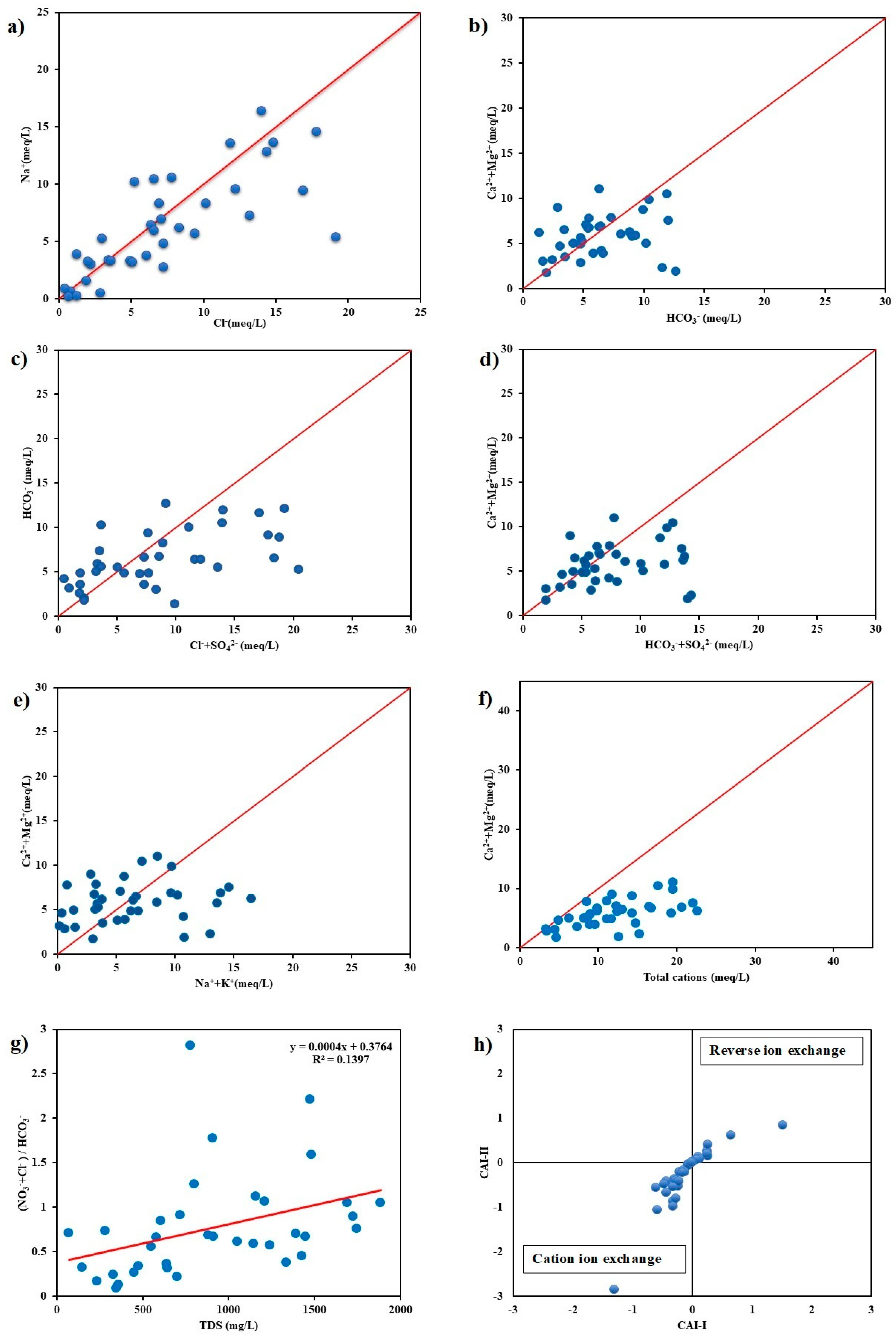
2.6. Chloro-Alkaline Indices (CAI)
3. Results and Discussion
3.1. Hydrochemistry Characteristics of Study Region
3.1.1. pH
3.1.2. Electrical Conductivity (EC)
3.1.3. Total Dissolved Solids (TDS)
3.1.4. Total Hardness (TH)
3.1.5. Calcium (Ca2+)
3.1.6. Magnesium (Mg2+)
3.1.7. Sodium (Na+)
3.1.8. Potassium (K+)
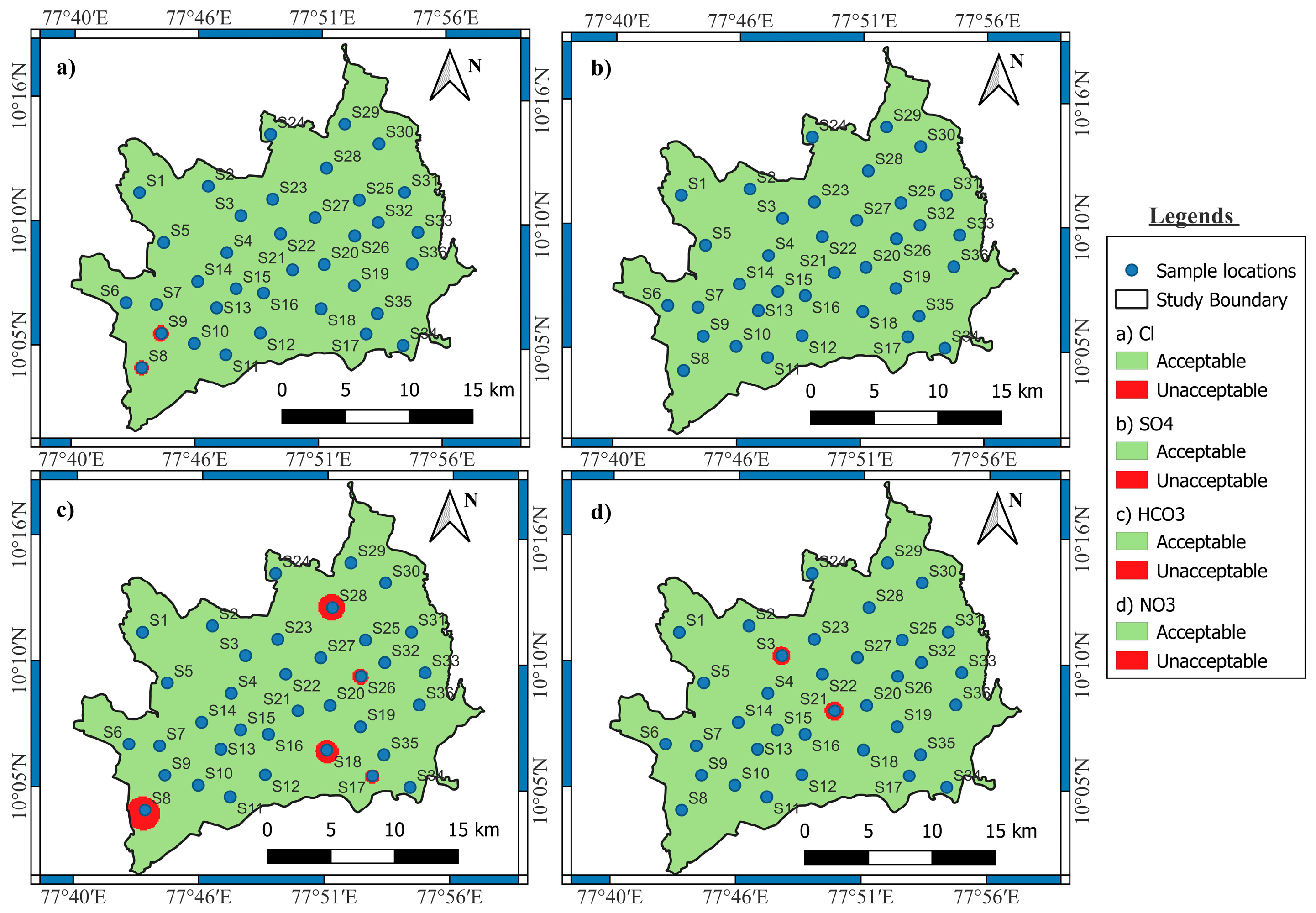
3.1.9. Chlorides (Cl−)
3.1.10. Sulphate (SO42−)
3.1.11. Bicarbonate (HCO3−)
3.1.12. Nitrate (NO3−)
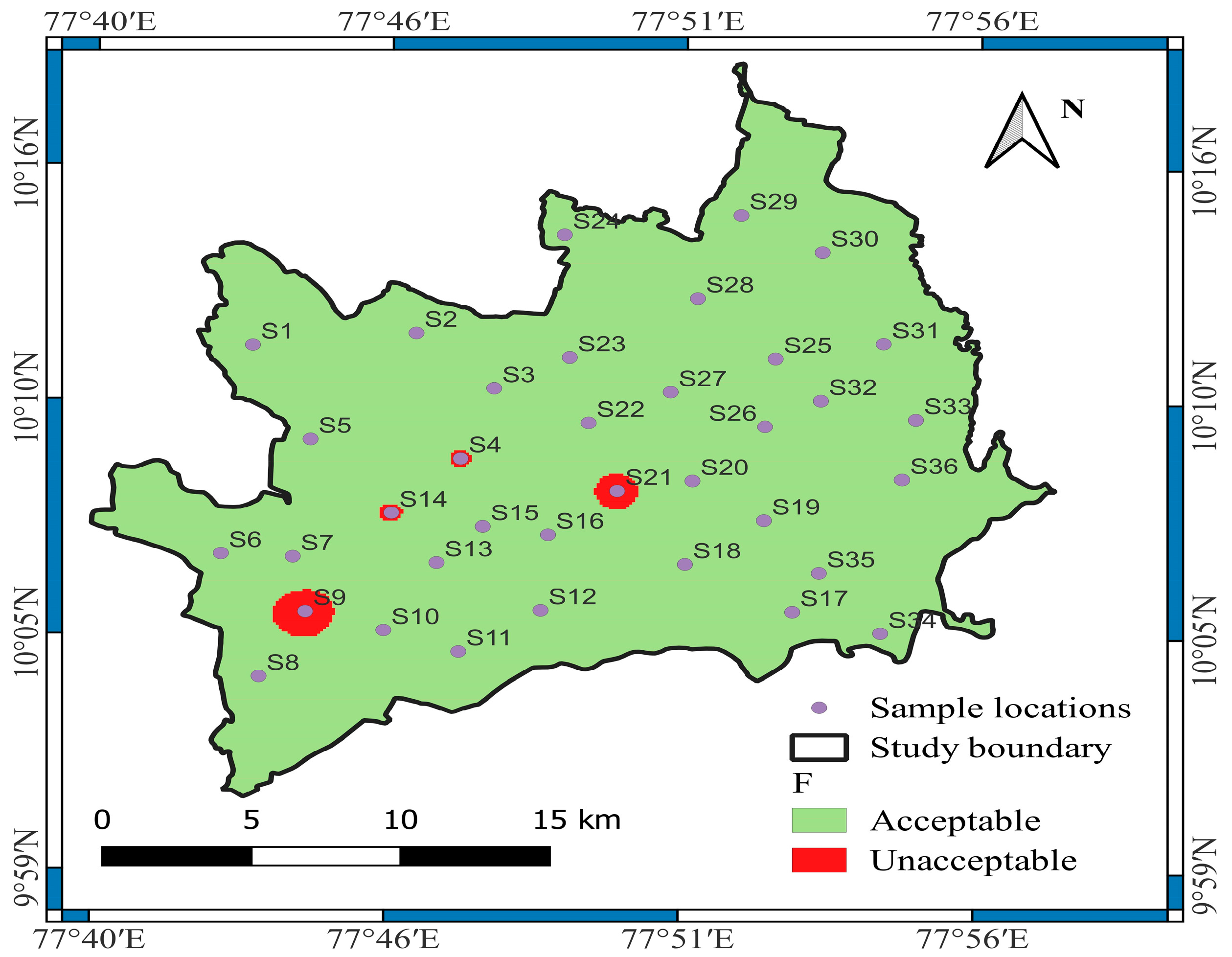
3.1.13. Fluoride (F−)
3.2. EWQI
3.3. Identification of Contamination Zone
3.4. Contamination Source Identification (CSI)
3.5. Effect of Evaporation Process on Groundwater Chemistry
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Panneerselvam, B.; Muniraj, K.; Pande, C.; Ravichandran, N. Prediction and evaluation of groundwater characteristics using the radial basic model in Semi-arid region, India. Int. J. Environ. Anal. Chem. 2021, 103, 1377–1393. [Google Scholar] [CrossRef]
- Maghrebi, M.; Noori, R.; Partani, S.; Araghi, A.; Barati, R.; Farnoush, H.; Torabi Haghighi, A. Iran’s Groundwater Hydrochemistry. Earth Space Sci. 2021, 8, e2021EA001793. [Google Scholar] [CrossRef]
- Shukla, S.; Saxena, A.; Khan, R.; Li, P. Spatial analysis of groundwater quality and human health risk assessment in parts of Raebareli district, India. Environ. Earth Sci. 2021, 80, 800. [Google Scholar] [CrossRef]
- Abendong, A.A.; Emmanuel, E.; Fongoh, E.J.; Akoachere, R.A. Assessment of groundwater quality in Bamenda–Cameroon for suitable applications. SN Appl. Sci. 2019, 1, 1389. [Google Scholar] [CrossRef]
- Baloch, M.Y.J.; Zhang, W.; Chai, J.; Li, S.; Alqurashi, M.; Rehman, G.; Tariq, A.; Talpur, S.A.; Iqbal, J.; Munir, M.; et al. Shallow Groundwater Quality Assessment and Its Suitability Analysis for Drinking and Irrigation Purposes. Water 2021, 13, 3361. [Google Scholar] [CrossRef]
- Chen, F.; Yao, L.; Mei, G.; Shang, Y.; Xiong, F.; Ding, Z. Groundwater Quality and Potential Human Health Risk Assessment for Drinking and Irrigation Purposes: A Case Study in the Semiarid Region of North China. Water 2021, 13, 783. [Google Scholar] [CrossRef]
- Ismail, A.H.; Hassan, G.; Sarhan, A.-H. Hydrochemistry of shallow groundwater and its assessment for drinking and irrigation purposes in Tarmiah district, Baghdad governorate, Iraq. Groundw. Sustain. Dev. 2019, 10, 100300. [Google Scholar] [CrossRef]
- Sarkar, M.; Pal, S.C.; Islam, A.R.M.T. Groundwater quality assessment for safe drinking water and irrigation purposes in Malda district, Eastern India. Environ. Earth Sci. 2022, 81, 52. [Google Scholar] [CrossRef]
- Jeihouni, M.; Toomanian, A.; Shahabi, M.; Alavipanah, S.K. Groundwater quality assessment for drinking purposes using GIS modelling (case study: City of Tabriz). Int. Arch. Photogramm. Remote. Sens. Spat. Inf. Sci.—ISPRS Arch. 2014, XL-2/W3, 163–168. [Google Scholar] [CrossRef]
- Water and Sanitation, United Nations International Children’s Emergency Fund. Available online: https://www.unicef.org/supply/water-and-sanitation (accessed on 3 April 2023).
- Lin, X.; Lu, K.; Hardison, A.K.; Liu, Z.; Xu, X.; Gao, D.; Gong, J.; Gardner, W.S. Membrane inlet mass spectrometry method (REOX/MIMS) to measure 15N-nitrate in isotope-enrichment experiments. Ecol. Indic. 2021, 126, 107639. [Google Scholar] [CrossRef]
- Liu, F.; Zhen, P.; Wang, S. Groundwater quality assessment and health risks from nitrate contamination in the Heilongdong Spring Basin, a typical headwater basin of the North China Plain. Environ. Sci. Pollut. Res. 2021, 29, 17655–17670. [Google Scholar] [CrossRef]
- Gugulothu, S.; Rao, N.S.; Das, R.; Duvva, L.K.; Dhakate, R. Judging the sources of inferior groundwater quality and health risk problems through intake of groundwater nitrate and fluoride from a rural part of Telangana, India. Environ. Sci. Pollut. Res. 2022, 29, 49070–49091. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Li, P.; Ji, Y.; Wang, Y.; Su, Z.; Elumalai, V. Groundwater Arsenic and Fluoride and Associated Arsenicosis and Fluorosis in China: Occurrence, Distribution and Management. Expo. Health 2020, 12, 355–368. [Google Scholar] [CrossRef]
- Yang, D.; Qiu, H.; Ye, B.; Liu, Y.; Zhang, J.; Zhu, Y. Distribution and Recurrence of Warming-Induced Retrogressive Thaw Slumps on the Central Qinghai-Tibet Plateau. J. Geophys. Res. Earth Surf. 2023, 128, e2022JF007047. [Google Scholar] [CrossRef]
- Sajil Kumar, P.J. Hydrogeochemical and multivariate statistical appraisal of pollution sources in the groundwater of the lower Bhavani River basin in Tamil Nadu. Geol. Ecol. Landscapes 2019, 4, 40–51. [Google Scholar] [CrossRef]
- Panneerselvam, B.; Karuppannan, S.; Muniraj, K. Evaluation of drinking and irrigation suitability of groundwater with special emphasizing the health risk posed by nitrate contamination using nitrate pollution index (NPI) and human health risk assessment (HHRA). Hum. Ecol. Risk Assess. Int. J. 2020, 27, 1324–1348. [Google Scholar] [CrossRef]
- Vesali Naseh, M.R.; Noori, R.; Berndtsson, R.; Adamowski, J.; Sadatipour, E. Groundwater pollution sources apportionment in the Ghaen Plain, Iran. Int. J. Environ. Res. Public Health 2018, 15, 172. [Google Scholar] [CrossRef] [PubMed]
- Shunmugapriya, K.; Panneerselvam, B.; Muniraj, K.; Ravichandran, N.; Prasath, P.; Thomas, M.; Duraisamy, K. Integration of multi criteria decision analysis and GIS for evaluating the site suitability for aquaculture in southern coastal region, India. Mar. Pollut. Bull. 2021, 172, 112907. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; Water Environmental American Public Health Association (APHA): Washington, DC, USA; Federation: Washington, DC, USA, 2017. [Google Scholar]
- Adimalla, N. Application of the Entropy Weighted Water Quality Index (EWQI) and the Pollution Index of Groundwater (PIG) to Assess Groundwater Quality for Drinking Purposes: A Case Study in a Rural Area of Telangana State, India. Arch. Environ. Contam. Toxicol. 2021, 80, 31–40. [Google Scholar] [CrossRef]
- Panneerselvam, B.; Muniraj, K.; Duraisamy, K.; Pande, C.; Karuppannan, S.; Thomas, M. An integrated approach to explore the suitability of nitrate-contaminated groundwater for drinking purposes in a semiarid region of India. Environ. Geochem. Health 2022, 45, 647–663. [Google Scholar] [CrossRef]
- Panneerselvam, B.; Muniraj, K.; Pande, C.; Ravichandran, N.; Thomas, M.; Karuppannan, S. Geochemical evaluation and human health risk assessment of nitrate-contaminated groundwater in an industrial area of South India. Environ. Sci. Pollut. Res. 2021, 29, 86202–86219. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, J.; Liu, M.; Yin, Z.; Liu, X.; Yin, L.; Zheng, W. Remote sensing and geostatistics in urban water-resource monitoring: A review. Mar. Freshw. Res. 2023, 74, 747–765. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, H.; Fang, P.; Liang, B.; Zheng, K.; Sun, Q.; Wang, A. Life cycle assessment of integrated bioelectrochemical-constructed wetland system: Environmental sustainability and economic feasibility evaluation. Resour. Conserv. Recycl. 2023, 189, 106740. [Google Scholar] [CrossRef]
- Zhang, N.; Lu, D.; Sheng, H.; Xia, J.; Kan, P.; Yao, Z.; Chen, H.; Li, G.; Zhu, D.Z.; Liu, H. Constructed wetlands as hotspots of antibiotic resistance genes and pathogens: Evidence from metagenomic analysis in Chinese rural areas. J. Hazard. Mater. 2023, 447, 130778. [Google Scholar] [CrossRef]
- Ghahremanzadeh, H.; Noori, R.; Baghvand, A.; Nasrabadi, T. Evaluating the main sources of groundwater pollution in the southern Tehran aquifer using principal component factor analysis. Environ. Geochem. Health 2017, 40, 1317–1328. [Google Scholar] [CrossRef] [PubMed]
- Panneerselvam, B.; Muniraj, K.; Thomas, M.; Ravichandran, N.; Bidorn, B. Identifying influencing groundwater parameter on human health associate with irrigation indices using the Automatic Linear Model (ALM) in a semi-arid region in India. Environ. Res. 2021, 202, 111778. [Google Scholar] [CrossRef] [PubMed]
- Nong, X.; Lai, C.; Chen, L.; Shao, D.; Zhang, C.; Liang, J. Prediction modelling framework comparative analysis of dissolved oxygen concentration variations using support vector regression coupled with multiple feature engineering and optimization methods: A case study in China. Ecol. Indic. 2023, 146, 109845. [Google Scholar] [CrossRef]
- Panneerselvam, B.; Paramasivam, S.K.; Karuppannan, S.; Ravichandran, N.; Selvaraj, P. A GIS-based evaluation of hydrochemical characterisation of groundwater in hard rock region, South Tamil Nadu, India. Arab. J. Geosci. 2020, 13, 837. [Google Scholar] [CrossRef]
- Ramalingam, S.; Panneerselvam, B.; Kaliappan, S.P. Effect of high nitrate contamination of groundwater on human health and water quality index in semi-arid region, South India. Arab. J. Geosci. 2022, 15, 242. [Google Scholar] [CrossRef]
- Panneerselvam, B.; Ravichandran, N.; Kaliyappan, S.P.; Karuppannan, S.; Bidorn, B. Quality and Health Risk Assessment of Groundwater for Drinking and Irrigation Purpose in Semi-Arid Region of India Using Entropy Water Quality and Statistical Techniques. Water 2023, 15, 601. [Google Scholar] [CrossRef]
- Omeka, M.E. Evaluation and prediction of irrigation water quality of an agricultural district, SE Nigeria: An integrated heuristic GIS-based and machine learning approach. Environ. Sci. Pollut. Res. 2023, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhu, J.; Fu, L.; Zhu, Q.; Xie, Y.; Hu, Y. An augmented representation method of debris flow scenes to improve public perception. Int. J. Geogr. Inf. Sci. 2020, 35, 1521–1544. [Google Scholar] [CrossRef]
- Abdel Moneim, A.A.; Seleem, E.M.; Zeid, S.A.; Abdel Samie, S.G.; Zaki, S.; Abu El-Fotoh, A. Hydrogeochemical characteristics and age dating of groundwater in the Quaternary and Nubian aquifer systems in Wadi Qena, Eastern Desert, Egypt. Sustain. Water Resour. Manag. 2015, 1, 213–232. [Google Scholar] [CrossRef]
- Qiu, D.; Zhu, G.; Bhat, M.A.; Wang, L.; Liu, Y.; Sang, L.; Lin, X.; Zhang, W.; Sun, N. Water use strategy of nitraria tangutorum shrubs in ecological water delivery area of the lower inland river: Based on stable isotope data. J. Hydrol. 2023, 624, 129918. [Google Scholar] [CrossRef]
- APHA-AWWA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association/American Water Works Association/Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- Aragaw, T.T.; Gnanachandrasamy, G. Evaluation of groundwater quality for drinking and irrigation purposes using GIS-based water quality index in urban area of Abaya-Chemo sub-basin of Great Rift Valley, Ethiopia. Appl. Water Sci. 2021, 11, 148. [Google Scholar] [CrossRef]
- Zhu, G.; Liu, Y.; Shi, P.; Jia, W.; Zhou, J.; Liu, Y.; Ma, X.; Pan, H.; Zhang, Y.; Zhang, Z.; et al. Stable water isotope monitoring network of different water bodies in Shiyang River basin, a typical arid river in China. Earth Syst. Sci. Data 2022, 14, 3773–3789. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, L.; Zhong, X.; Yao, T.; Qi, J.; Wang, Y.; Xue, Y. Quantifying the major drivers for the expanding lakes in the interior Tibetan Plateau. Sci. Bull. 2021, 67, 474–478. [Google Scholar] [CrossRef] [PubMed]
- El Mountassir, O.; Bahir, M.; Ouazar, D.; Chehbouni, A.; Carreira, P.M. Temporal and spatial assessment of groundwater contamination with nitrate using nitrate pollution index (NPI), groundwater pollution index (GPI), and GIS (case study: Essaouira basin, Morocco). Environ. Sci. Pollut. Res. 2021, 29, 17132–17149. [Google Scholar] [CrossRef]
- Wu, B.; Quan, Q.; Yang, S.; Dong, Y. A social-ecological coupling model for evaluating the human-water relationship in basins within the Budyko framework. J. Hydrol. 2023, 619, 129361. [Google Scholar] [CrossRef]
- Tamma Rao, G.; Srinivasa Rao, Y.; Mahesh, J.; Surinaidu, L.; Dhakate, R.; Gurunadha Rao, V.V.S.; Durga Prasad, M. Hydrochemical assessment of groundwater in alluvial aquifer region, Jalandhar District, Punjab, India. Environ. Earth Sci. 2014, 73, 8145–8153. [Google Scholar] [CrossRef]
- Jabbo, J.N.; Isa, N.M.; Aris, A.Z.; Ramli, M.F.; Abubakar, M.B. Geochemometric approach to groundwater quality and health risk assessment of heavy metals of Yankari Game Reserve and its environs, Northeast Nigeria. J. Clean. Prod. 2021, 330, 129916. [Google Scholar] [CrossRef]
- Zhu, X.; Xu, Z.; Liu, Z.; Liu, M.; Yin, Z.; Yin, L.; Zheng, W. Impact of dam construction on precipitation: A regional perspective. Mar. Freshw. Res. 2022, 74, 877–890. [Google Scholar] [CrossRef]
- Khan, R.; Jhariya, D.C. Groundwater quality assessment for drinking purpose in Raipur city, Chhattisgarh using water quality index and geographic information system. J. Geol. Soc. India 2017, 90, 69–76. [Google Scholar] [CrossRef]
- Khan, R.; Jhariya, D.C. Hydrogeochemistry and Groundwater Quality Assessment for Drinking and Irrigation Purpose of Raipur City, Chhattisgarh. J. Geol. Soc. India 2018, 91, 475–482. [Google Scholar] [CrossRef]
- Kom, K.P.; Gurugnanam, B.; Sunitha, V.; Reddy, Y.S.; Kadam, A.K. Hydrogeochemical assessment of groundwater quality for drinking and irrigation purposes in western Coimbatore, South India. Int. J. Energy Water Resour. 2021, 6, 475–494. [Google Scholar] [CrossRef]
- Li, P.; Tian, R.; Xue, C.; Wu, J. Progress, opportunities, and key fields for groundwater quality research under the impacts of human activities in China with a special focus on western China. Environ. Sci. Pollut. Res. 2017, 24, 13224–13234. [Google Scholar] [CrossRef]
- Yang, M.; Wang, H.; Hu, K.; Yin, G.; Wei, Z. IA-Net: An Inception–Attention-Module-Based Network for Classifying Underwater Images from Others. IEEE J. Ocean. Eng. 2022, 47, 704–717. [Google Scholar] [CrossRef]
- Zhou, G.; Yang, Z. Analysis for 3-D morphology structural changes for underwater topographical in Culebrita Island. Int. J. Remote. Sens. 2023, 44, 2458–2479. [Google Scholar] [CrossRef]
- Li, Y.; Li, P.; Cui, X.; He, S. Groundwater quality, health risk, and major influencing factors in the lower Beiluo River watershed of northwest China. Hum. Ecol. Risk Assess. Int. J. 2021, 27, 1987–2013. [Google Scholar] [CrossRef]
- Liu, J.; Jin, D.; Wang, T.; Gao, M.; Yang, J.; Wang, Q. Hydrogeochemical processes and quality assessment of shallow groundwater in Chenqi coalfield, Inner Mongolia, China. Environ. Earth Sci. 2019, 78, 347. [Google Scholar] [CrossRef]
- Ponnusamy, D. Mapping of Potential Groundwater Recharge Zones: A Case Study of Maputaland Coastal Plain, South Africa. Res. Sq. 2020, 81, 418. [Google Scholar] [CrossRef]
- Yin, L.; Wang, L.; Keim, B.D.; Konsoer, K.; Yin, Z.; Liu, M.; Zheng, W. Spatial and wavelet analysis of precipitation and river discharge during operation of the Three Gorges Dam, China. Ecol. Indic. 2023, 154, 110837. [Google Scholar] [CrossRef]
- Pan, C.; Ng, K.T.W.; Fallah, B.; Richter, A. Evaluation of the bias and precision of regression techniques and machine learning approaches in total dissolved solids modeling of an urban aquifer. Environ. Sci. Pollut. Res. 2019, 26, 1821–1833. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Wu, G.; Zhou, X.; Xu, C.; Zhao, D.; Lin, J.; Liu, Z.; Zhang, H.; Wang, Q.; Xu, J.; et al. Adaptive model for the water depth bias correction of bathymetric LiDAR point cloud data. Int. J. Appl. Earth Obs. Geoinf. 2023, 118, 103253. [Google Scholar] [CrossRef]
- Sajil Kumar, P.J.; James, E.J. Assessing the Impact of Leather Industries on Groundwater Quality of Vellore District in South India Using a Geochemical Mixing Model. Environ. Claims J. 2019, 31, 335–348. [Google Scholar] [CrossRef]
- Wang, Z.-R.; Tian, X.; Wu, X. Hydrochemical characteristics and quality assessment of shallow groundwater and CBM co-produced water in the Shizhuangnan block, Qinshui Basin, China. Environ. Earth Sci. 2018, 77, 57. [Google Scholar] [CrossRef]
- Ma, S.; Qiu, H.; Yang, D.; Wang, J.; Zhu, Y.; Tang, B.; Sun, K.; Cao, M. Surface multi-hazard effect of underground coal mining. Landslides 2022, 20, 39–52. [Google Scholar] [CrossRef]
- Yin, L.; Wang, L.; Huang, W.; Tian, J.; Liu, S.; Yang, B.; Zheng, W. Haze Grading Using the Convolutional Neural Networks. Atmosphere 2022, 13, 522. [Google Scholar] [CrossRef]
- Derdour, A.; Abdo, H.G.; Almohamad, H.; Alodah, A.; Al Dughairi, A.A.; Ghoneim, S.S.M.; Ali, E. Prediction of Groundwater Quality Index Using Classification Techniques in Arid Environments. Sustainability 2023, 15, 9687. [Google Scholar] [CrossRef]
- Alao, J.O.; Abdo, H.G.; Ayejoto, D.A.; Mohammed, M.A.; Danladi, E.; Saqr, A.M.; Almohamad, H.; Fahad, A. Evaluation of Groundwater contamination and the Health Risk Due to Landfills using integrated geophysical methods and Physiochemical Water Analysis. Case Stud. Chem. Environ. Eng. 2023, 8, 100523. [Google Scholar] [CrossRef]
- Alao, J.O.; Fahad, A.; Abdo, H.G.; Ayejoto, D.A.; Almohamad, H.; Ahmad, M.S.; Nur, M.S.; Danjuma, T.T.; Yusuf, M.A.; Francis, O.T.; et al. Effects of dumpsite leachate plumes on surface and groundwater and the possible public health risks. Sci. Total Environ. 2023, 897, 165469. [Google Scholar] [CrossRef] [PubMed]





| EWQI | Rank | Number of Samples | % of Samples | Class of Water | Purpose of Use |
|---|---|---|---|---|---|
| <50 | 1 | 8 | 22.22 | Excellent | Fit for drinking purpose |
| 50–100 | 2 | 22 | 61.11 | Good | |
| 100–150 | 3 | 6 | 16.67 | Medium | Needs primary level of treatment before use |
| 150–200 | 4 | 0 | 0 | Poor | Needs advanced level of treatment before use |
| >200 | 5 | 0 | 0 | Extremely poor | Unsuitable for drinking |
| Minerals | Minimum | Maximum | Average |
|---|---|---|---|
| Anhydrite | −1.69 | 0.72 | −0.28 |
| Aragonite | 1.91 | 3.57 | 2.75 |
| Calcite | 2.05 | 3.71 | 2.90 |
| Dolomite | 3.21 | 6.88 | 5.54 |
| Fluorite | 0.63 | 2.92 | 1.67 |
| Gypsum | −1.39 | 1.01 | 0.01 |
| Halite | -6 | −2.67 | −3.70 |
| Sylvite | −5.56 | 0 | −3.77 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muthusamy, P.; Paneerselvam, B.; Kaliyappan, S.P.; Almohamad, H.; Aldosari, A.A.; Abdo, H.G. Pollution Source Identification and Suitability Assessment of Groundwater Quality for Drinking Purposes in Semi-Arid Regions of the Southern Part of India. Water 2023, 15, 3995. https://doi.org/10.3390/w15223995
Muthusamy P, Paneerselvam B, Kaliyappan SP, Almohamad H, Aldosari AA, Abdo HG. Pollution Source Identification and Suitability Assessment of Groundwater Quality for Drinking Purposes in Semi-Arid Regions of the Southern Part of India. Water. 2023; 15(22):3995. https://doi.org/10.3390/w15223995
Chicago/Turabian StyleMuthusamy, Periyasamy, Balamurugan Paneerselvam, Shunmuga Priya Kaliyappan, Hussein Almohamad, Ali Abdullah Aldosari, and Hazem Ghassan Abdo. 2023. "Pollution Source Identification and Suitability Assessment of Groundwater Quality for Drinking Purposes in Semi-Arid Regions of the Southern Part of India" Water 15, no. 22: 3995. https://doi.org/10.3390/w15223995






