1. Introduction
With the dynamic development of industrialization and urbanization, the penetration of heavy metals from industrial discharges, domestic wastewater, and atmospheric deposition into natural water has caused a series of toxic effects on both aquatic ecosystems and human health, even at low concentrations [
1]). As an essential part of primary producers, algae show high sensitivity to heavy metals such as lead (Pb), mercury (Hg), cadmium (Cd), and nickel (Ni). Consequently, they can serve as a test model for freshwater quality assessment and pollutant toxicity estimation. To date, numerous studies have shown that most heavy metals, including Cd, Pb, Cu, and Ni, can interfere with the photosynthesis and enzymatic metabolism of algae, resulting in growth inhibition and even death. As previously found by Choudhary et al. [
2], Pb, Cu, and Zn could generate reactive oxygen species (ROS) and attack biomolecules in cells, further leading to irreversible adverse effects on spirulina. The toxic mechanism of copper in the microalga
Chlamydomonas reinhardtii was demonstrated as well. Thus, the addition of copper in a concentration of 5 mg/L in the microalga culture leads to growth inhibition, a phenomenon also observed in the case of the addition of chromium of the same concentration. However, compared to other heavy metals with higher half-maximal inhibitory concentrations (IC
50) such as Cd, As, Sb, or Pb, copper and chromium appeared to be less hazardous for heavy-metal sensitive aquatic organisms [
3]. Similarly, the algal growth inhibition test aimed at evaluating the toxicity of Zn, Cu, Cd, and Pb demonstrated that Zn showed the most toxic effects and Pb induced the lowest cell lethality in
Isochrysis galbana cultures [
4].
Although nickel is an essential microelement that plays an important role in the metabolism of primary producers, the risk of exposure to toxic doses of this metal has increased following the extension of its industrial uses. The response of microalgae to nickel toxicity depends on the alga species, its developmental stage, cultivation conditions, metal concentration in the culture media, and exposure time. It was shown that the toxic effect of nickel on cyanobacteria was due to its binding ability to sulphydryl-like groups present in the bacterial cell envelope, which resulted in the reduction of carbon assimilation and chlorophyll-A content [
5]. Microalgae grown in nickel-contaminated liquid medium show different responses and symptoms of toxicity, which include growth inhibition, a decrease of biomass production, chlorosis occurrence, disruption of the photosynthesis process, and inhibition of CO
2 assimilation. Many harmful effects of nickel are due to its interference with the metabolism of other essential metal ions, such as Fe (II), Mn (II), Cu (II), Zn (II), Cu (II), or Mg (II). In particular, nickel has the capacity to substitute other metallic ions from enzymes and proteins, or to bind the O-, S-, and N-functional groups of the cellular constituents and inhibit their functions [
6].
The toxic action of cadmium depends on the chemical species in which it is found. The toxicity of cadmium also depends on the route of exposure; as a consequence, the impact it exerts on ecosystems depends on its route of entry into the living organisms. Cadmium can be a catalyst of oxidation reactions, which disrupt and damage living tissues. Some studies have shown that cadmium can increase the oxidative capacity of living organisms, by acting as a catalyst in the generation of reactive oxygen species (ROS) when lipid peroxidation is enhanced and glutathione and protein-binding sulfhydryl groups are depleted. It is assumed that most of the survival mechanisms depend on the changes in Cd speciation, which influence its mobility [
7].
Lead is not known to be of biological importance and is toxic at very low concentrations [
8]. Lead toxicity depends on its bioavailability, so the bioavailable fraction of Pb (II) to which microalgae are exposed can be rather low [
9]. Hence, in laboratory experiments, the composition of the cell culture media must be attentively chosen to avoid precipitation and complexation of Pb (II), which may lead to an apparent decrease in its toxicity. Pb (II) toxicity is exerted as a result of changes in the conformation of nucleic acids and proteins, inhibition of enzyme activity, disruption of membrane functions and oxidative phosphorylation, as well as alteration of osmotic balance [
8]. Pb (II) also exhibits a stronger affinity for thiol and oxygen groups than other essential metals, such as calcium and zinc. Microorganisms, including microalgae, have developed several mechanisms that allow them to mitigate the high toxicity of Pb (II) and survive lead exposure.
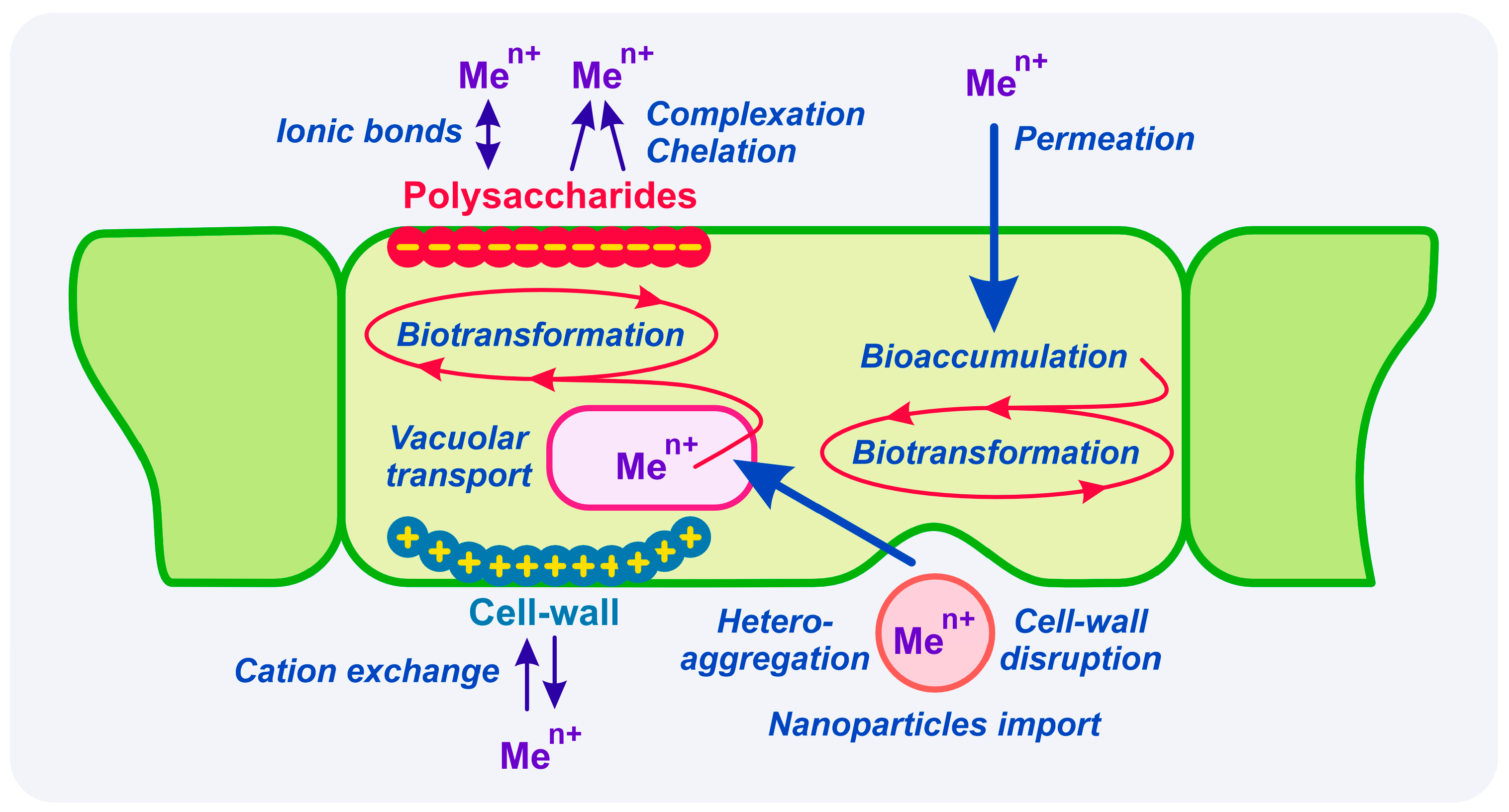
Many studies have shown that one mechanism responsible for heavy metal detoxification in algae is the metal accumulation in cells, in the form of metal-polysaccharide complexes; more precisely, the cell wall containing large amounts of polysaccharides can retain a great part of the heavy metals present in the aqueous media, thus preventing their entry into the cell and additional damage (
Figure 1). When the unicellular green alga
Chlamydomonas reinhardtii was exposed to Cd, Co, and Ni, more than half of the total heavy metals were aggregated on the cell surface [
10]). Research by Subramaniyam et al., 2016 [
11] also indicated that polysaccharides present in algal cell walls played a major role in the bioaccumulation of metal ions by algae.
It was found that the cell walls of the brown alga (
P. gymnospora) found in water contaminated with heavy metals produced a greater amount of polysaccharides than alga in clean water, precisely to avoid the intracellular accumulation of metals [
12]. Therefore, it can be inferred that the differences among heavy metals’ toxic effects on algae can be partially attributed to the cell wall adsorption capacity of heavy metals.
In general, algae with a high tolerance to one type of heavy metal can serve as a metal biosorbent in bioremediation treatments. Lately, several studies have been dedicated to the bioremediation potential of cyanobacteria spirulina towards heavy metals. Cepoi et al. [
13] showed a high ability of selective copper bioaccumulation by spirulina in the presence of iron, nickel, and zinc, when copper concentration was between 2.5 and 10 mg/L. A phytoremediation study conducted by Tabagari et al. [
14] showed that spirulina could remove up to 97% of non-radioactive cesium from solutions of 100 mg/L of this ion. Live spirulina biomass in lead solutions of low concentration (below 50 mg/L) was able to remove up to 74% of the metal in the first 12 min and 95% of the metal after 24 h [
9].
Besides metal concentration, the pH of the aqueous media is a critical factor for heavy metals bioavailability because some salts of these metals precipitate like alkaline hydroxides when pH rises above 6.5.
The key factors for the growth of
Spirulina platensis are (i) sufficient light intensity, (ii) a pH of the culture medium around 9.5, (iii) a temperature of 30 °C to 35 °C, and (iv) sufficient availability of all required nutrients. Other aspects related to the possible interactions of heavy metals with microalgae and their bioremediation potential are presented in [
15].
The aim of this paper is to investigate the influence of nickel (II), cadmium (II), and lead (II) ions on the growth and some metabolic processes in the microalga Spirulina platensis cultivated in a standard culture media supplemented with these metal ions at different concentrations found in the sublethal concentrations range. The specific objective is to determine the time evolution of cell mass concentration, dry biomass resulted from the alga growth, protein concentration in the dry biomass, cell viability, and the pH of culture media during a 28-day cultivation period.
2. Materials and Methods
2.1. Inoculum Culture Conditions
A primary stock culture of Spirulina platensis (strain provided by Algae Research Supply, Carlsbad, CA, USA), was grown at room temperature (22° ± 2 °C), with a light/dark alternation (10 h under cold white light of 4500 lux, and 14 h dark), under continuous agitation on a low-speed orbital shaker (Biosan, Riga, Latvia), without mechanical air supply. To preserve the culture in an exponential growth phase, 10% of the stock culture was monthly passaged on a fresh culture medium. This culture represented the inoculum for the cultivation experiments of the present study.
Spirulina passage and cultivation experiments were carried out in a Zarrouk medium with an optimal formula for this cyanobacterium (pH = 9.5; content in g/L: NaHCO3—18, NaNO3—2.5, K2HPO4—0.5, K2SO4—1.0, NaCl—1.0, CaCl2 · 2H2O—0.04, MgSO4 · 7H2O—0.2, FeSO4 · 7H2O—0.01, Na2EDTA·2H2O—0.08; and micronutrient solution—1 mL). The composition of the micronutrient solution (in g/L): H3BO3—2.860; MnCl2·4H2O—1.810; ZnSO4·7H2O—0.22; CuSO4·5H2O—0.079; Na2MoO4·2H2O—0.390. Both the Zarrouk culture medium and the microelement solution were autoclaved at 121 °C for 20 min and kept refrigerated until use. All the chemicals used throughout the experimental study were of reagent grade (Sigma-Aldrich, St. Louis, MI, USA).
2.2. Experimental Set-Up
The heavy metal salts used in the experiments were nickel acetate, Ni (CH3COO)2, cadmium acetate, Cd (CH3COO)2, and lead acetate, Pb (CH3COO)2. Solutions with metal ion concentrations of 5 mg/L, 10 mg/L, and 15 mg/L were prepared from each metal salt.
The cultivation experiments were carried out in 250 mL Erlenmeyer flasks containing 40 mL of Zarrouk medium, 5 mL of S. platensis inoculum, and 5 mL of metal ion solution of 5 mg/L, 10 mg/L, and 15 mg/L for each metal. A 1:10 dilution of the metal solutions occurred when the cell cultures with a total volume of 50 mL were prepared. For each metal, three cell cultures resulted, with metal concentrations of 0.5 mg/L, 1.0 mg/L, and 1.5 mg/L, respectively. In addition, a control sample containing 40 mL of Zarrouk culture medium, 5 mL of S. platensis inoculum, and 5 mL of sterilized distilled water was prepared. The 10 experimental cell cultures were incubated under the same conditions used for the stock culture for a period of 28 days.
Aliquots of cell suspensions were withdrawn from all cultures at the starting point (T0) and 3 (T3), 7 (T7), 13 (T13), 18 (T18), 24 (T24), and 28 (T28) days, to evaluate the rate of cell growth, total protein content, dry biomass, the cell culture pH, and cell viability. Three parallel replicates were performed for each metal concentration level, with a standard deviation (SD) below 5%.
2.3. Evaluation of S. platensis Cell Growth in the Presence of Heavy Metals
Cell growth in the culture of microalgae S. platensis was assessed spectrophotometrically by measuring the optical density at a wavelength of 600 nm (OD600). For this, a volume of 2 mL of cell suspension was taken from each experimental sample (after vigorous stirring for homogenization), introduced into the measuring cell, and then the absorbance at λ = 600 nm was read on a Spectronic Helios Alpha UV-VIS spectrophotometer (ThermoFisher, Waltham, MA, USA). The absorbance of cell suspension is in direct correlation with the number of cells per volume unit (mL).
2.4. Total Protein Quantification (Lowry Method)
The total protein content in spirulina biomass was determined spectrophotometrically by the Lowry method, using the Folin–Ciocalteu reagent, and bovine serum albumin (BSA) as standard [
16]. BSA is the preferred standard in protein assays due to its low price, high purity, and feasible large-scale production. Briefly, an aliquot of 1 mL cell suspension was taken from each experimental sample, placed in a centrifuge tube, and subjected to cell wall lysis in an ultrasonic bath for 10 min, in cycles of 10 s of ultrasonication and 20 s of resting time, to prevent sample overheating. Then, in each test tube, 8.1 mL of Lowry reagent and 0.9 mL Folin–Ciocâlteu reagent (Sigma-Aldrich, St. Louis, MI, USA) were added. The control sample was prepared with 1 mL of distilled water instead of cell suspension. The reaction systems were incubated at room temperature in the dark for 2 h when the proteins in the cell suspension formed the characteristic blue complex with the Folin reagent. The supernatant was separated by centrifugation at 6000 rpm for 3 min, and the absorbance was measured at 660 nm on a Helyos UV-VIS spectrophotometer.
2.5. Quantification of Dry Spirulina Biomass
Dry organic spirulina biomass was determined gravimetrically. For this, 1 mL sample of microalga suspension was withdrawn from the culture medium; to remove the inorganic salts from the culture medium, the biomass was washed under vacuum three times with 25 mL portions of bidistilled water using a G5 filter crucible (porosity: 5 μm), previously brought to constant weight. The crucible with salt-free biomass was dried in an air oven at 105 °C to constant weight. The difference between the final and the initial weighing of the crucible represents the total amount of microbial biomass, according to the formula:
where G
f is the final weight of the crucible (g), and G
i is the initial weight of the crucible (g). Dry biomass was determined at the starting point (T
0) and after 3, 7, 13, 18, 24, and 28 days, on the control culture and the cultures treated with heavy metal solutions. Both protein and dry biomass were determined on 1 mL of culture medium so that reporting was consistent.
2.6. pH Measurement of Spirulina Cultures
The pH values of spirulina cultures were measured over the cultivation period, at T0, T3, T7, T13, T18, T24, and T28 with a Consort C3010 pH multimeter (Consort BVBA, Turnhout, Belgium).
2.7. Determination of the Sedimentation Index (IS) of Cell Cultures
The sedimentation index of cells (IS) was determined based on the difference between the optical density (OD) of the cell suspension, measured at 600 nm immediately after taking the sample and after 10 min of settling. The values of OD
600 were measured on a Helios UV-VIS spectrophotometer. The sedimentation index of cells was calculated by the formula:
where OD
600(t
0)—optical density of microalga culture, immediately after sample withdrawal; OD
600(t
10)—optical density of microalga culture after 10 min settling.
2.8. Microscopy Visualization of Cells
A small volume of the culture medium was transferred to the slide containing a drop of distilled water. The culture was teased properly to see the filaments. A cover slip was placed carefully without air bubbles. The cells were observed at 100× and 400× magnification under a light microscope model Motic BA 310 (Motic, Richmond, BC, Canada).
2.9. Statistical Analysis
Experimental data related to the growth dynamics of spirulina cells in the presence of metal cations, expressed by three parameters of the mature culture (the optical density, the total protein content of biomass, and the dry content of biomass,), were collected and analyzed for seven samples, on day 24 of spirulina cultivation. Sampling was performed from seven random points of the culture suspension at the end of the dark period. Samples from a control culture were also collected and analyzed. Data were then analyzed by using scripts written for R version 4.3.1 (16 June 2023) [
17], and the R packages easyanova v10.0 [
18], emmeans v1.8.9 [
19], mutoss v0.1-13 [
20], and ggstatsplot v0.12.1 [
21]. Bartlett test for equal variances, Shapiro–Wilk test for gaussianity, one-way ANOVA, and the post-hoc Tukey's HSD test for multiple comparisons were successively performed. For all of them, a value of 0.05 was considered for the level of statistical significance. The quality of ANOVA testing was assessed by means of the Welch F test. Before the graphical representation of the ANOVA results, the
p-values were adjusted by using the Dunnett T3 test. The final multiple comparisons were conducted by considering the
p-values correction calculated by using the Benjamini–Krieger–Yekutieli multiple-stage procedure for estimating the false discovery rate, according to which the decision was taken to reject or not the null hypothesis regarding the equality of the means of the compared variables.
3. Results and Discussion
Heavy metals play a key role in the metabolic processes of living organisms. Some heavy metals (like copper, nickel, zinc, iron, manganese, magnesium, etc.) are essential for living organisms—plants and animals—when they are present in low concentration and are known as oligo-elements or micronutrients. They are involved in oxidation-reduction processes, act as catalysts in enzymatic reactions, and support the osmotic balance as well. On the other hand, some metals have no biological role and are toxic to living organisms even at very low concentrations (cadmium, mercury, lead, chromium (VI), etc.). However, at high concentrations, both essential and non-essential metals become toxic to living organisms.
3.1. Growth of S. platensis Cellular Biomass under the Influence of Heavy Metal Ions
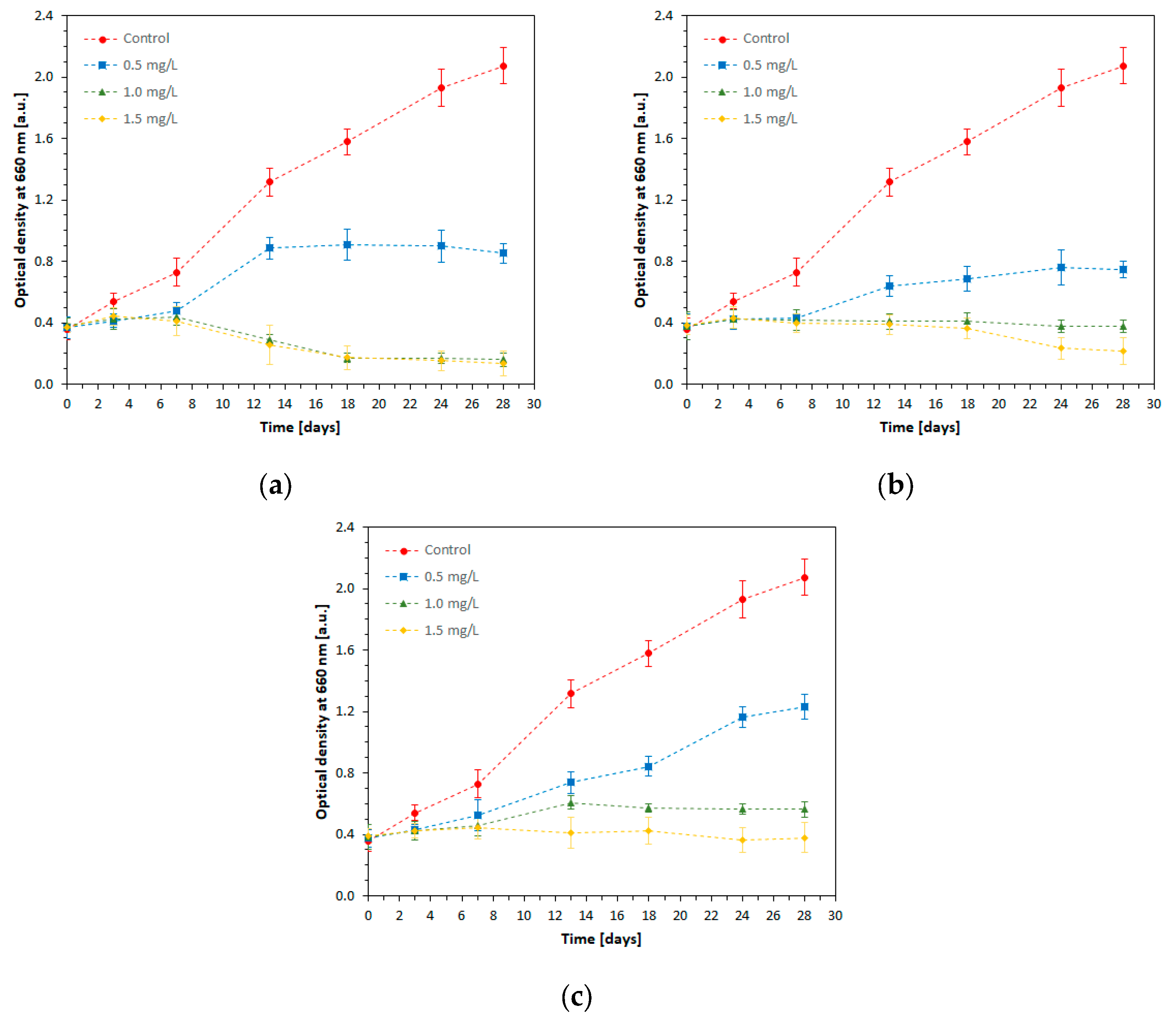
Based on the measurements of the optical density of the spirulina samples (OD
600), the growth curves for
S. platensis over a period of 28 days in the presence of Ni, Cd, and Pb ions were drawn, as given in
Figure 2.
In the control sample, spirulina cells underwent exponential growth, so the OD600 at the end of the cultivation time (28 days) was about 4.6 times higher compared with the OD600 at the starting point.
The influence of Ni (II) on cell growth depended on its concentration in the cultivation medium. At Ni concentration of 0.5 mg/L, the microalga culture developed relatively slowly in the first 7 days, after which an exponential growth until the 13th day took place, when the cell mass was about 2 times higher than the initial value. In the interval of 13–24 days, the stationary phase was installed, after which the growth rate entered the decline phase. At concentrations of 1.0 mg/L and 1.5 mg/L, nickel was particularly toxic for the spirulina culture: after a slight development in the first 7 days, the decline phase set in, and the cell mass decreased to lower values compared to the initial one. The toxic action of nickel is due to its penetration inside the algal cells, where it binds to cellular compounds essential to the cell metabolic activity, such as enzymes and nucleic acids, which are inactivated [
6]. Nickel can also cause oxidative stress, by producing reactive oxygen species (ROS) that cause damage to various macromolecules such as lipids, carbohydrates, and DNA, leading to cell death [
22].
Similar to nickel, the toxic action of cadmium ion, Cd (II) depends on its concentration in the culture media. Thus, at 0.5 mg/L Cd (II) in the growth medium, a significant increase in cell mass was observed till the 24th day of cultivation, but at much lower values compared to the control sample. Towards the end of the cultivation time, the stationary phase was established. When Cd (II) concentration was 1.0 mg/L, the spirulina cell number decreased slightly over the entire cultivation period. Conversely, Cd (II) in a concentration of 1.5 mg/L in the growth medium had a toxic effect, so that after a slight increase in cell number in the first 3 days, the decline phase set in and became very pronounced after 18 days of cultivation; the lysis of the cell mass took place, and the values determined towards the end of the experiment were below the initial threshold. The toxic effect of cadmium is due to its penetration into the spirulina cells and the blockage of functional groups in the plasma membrane and cell wall, which prevents the normal development of metabolic processes.
A concentration of 0.5 mg/L Pb (II) caused a slight inhibition of the spirulina development process, compared to the control. In this experimental variant, the culture was in an exponential growth phase throughout the experiment, so the cell number was approximately 3 times higher than the initial one and approximately 1.6 times lower than the final value determined in the control. At 1.0 mg/L lead, the spirulina culture recorded a slight increase in the period T0–T13, after which the decline phase started. Exposure of spirulina culture to a high concentration of lead (1.5 mg/L) had an inhibitory effect, and the decline phase started after three days of cultivation. However, lead was found to be less toxic to spirulina compared to nickel and cadmium at the same concentration.
3.2. The Influence of Metal Ions on Protein Accumulation in Spirulina Cultures
Spirulina is a well-known source of proteins, the content of which constitutes up to 70% of absolutely dry biomass. The amount of proteins in spirulina can vary widely by modifying nutrient medium chemical composition. A common response of cyanobacteria to stress, including that provoked by metal ions, is a change in the levels of proteins involved in central metabolic processes [
23].
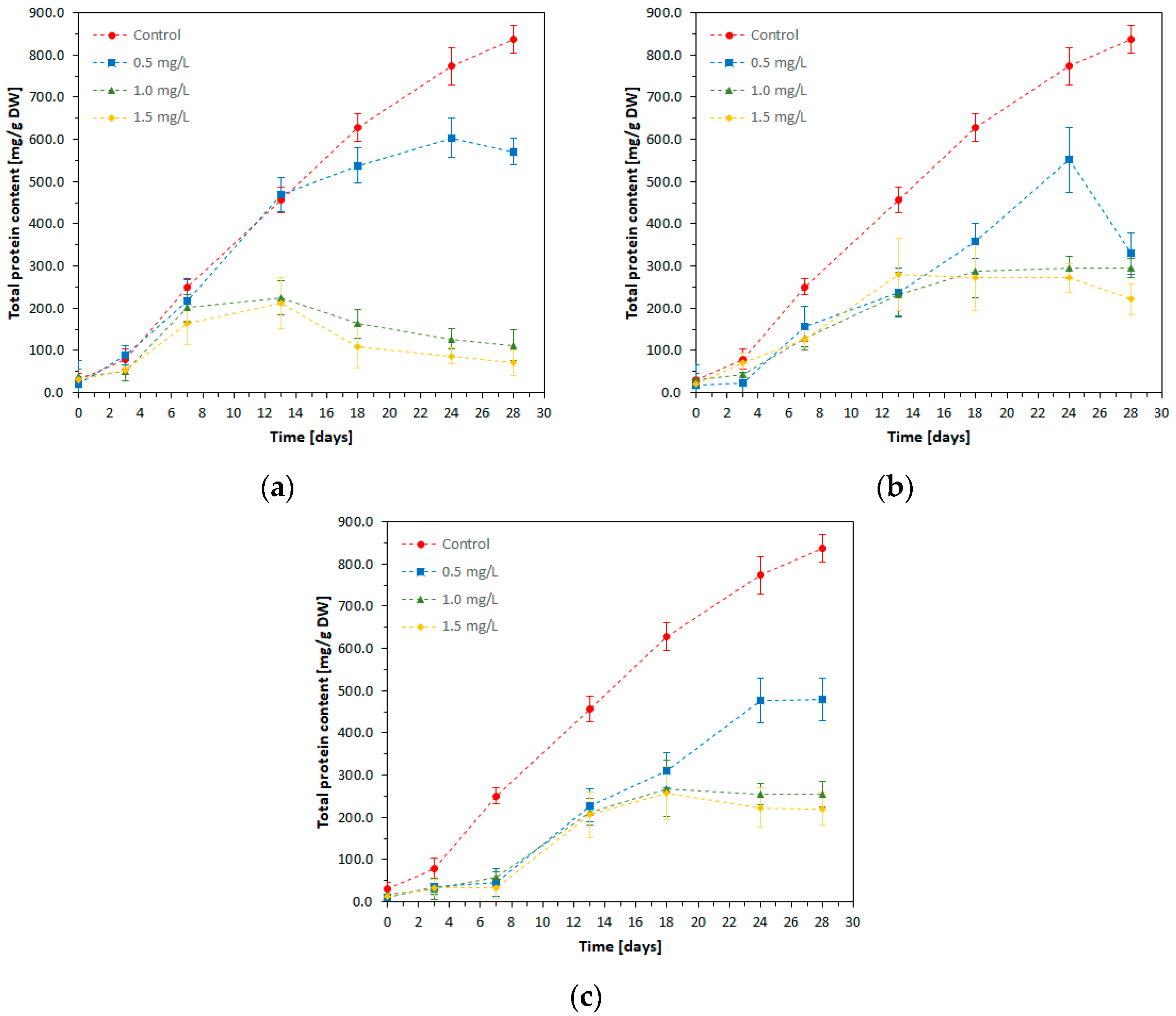
The amounts of total protein synthesized by
S. platensis and accumulated in the cell biomass over the cultivation period in the presence of heavy metals are presented in
Figure 3. The total protein content (TP) was reported per unit of dry weight (d.w.) of the biomass.
An overview of the experimental data shows that the amount of total protein differs from one experimental variant to another, depending both on the metal ion added to the spirulina growing medium, and on its concentration.
The control culture produced the highest amount of protein throughout the cultivation period, the protein concentration at the end of the experiment (T28) being about 16.8 times higher compared to that determined at the starting point (T0).
In the presence of metal ions (Ni, Cd, or Pb) at low concentration (0.5 mg/L), the protein synthesis increased progressively until the 24th day of the experiment. In the 24th-28th-day interval, the protein production decreased and was more pronounced in the presence of Cd ions. The maximum protein content in these experimental variants had values between 500 and 600 mg/g dry biomass, which represents approximately half of the value determined in the control culture.
Exposure to Ni (II) in concentrations of 1.0 mg/L and 1.5 mg/L caused severe inhibition of protein synthesis starting from the 13th day, so that the values of protein concentration at T28 were 120 mg/g dry biomass for the culture with 1.0 mg/L Ni (II), and 70 mg/g dry biomass for the culture with 1.5 mg/L Ni (II).
Cadmium at 1.0 mg/L and 1.5 mg/L determined a reduction of the total protein concentration by approximately 2.9 and 3.8 times, respectively, compared to the control culture. Exposure to this metal determined a slight increase of protein production in the interval T0–T18 (Cd—1.0 mg/L), and in the interval T0–T13 (Cd—1.5 mg/L), after which it remained constant or slightly decreased towards the end of the cultivation period.
In the presence of Pb ions at concentrations of 1.0 mg/L and 1.5 mg/L, the amount of protein remained almost constant during the first 7 days of microalga cultivation. Then, a gradual increase was observed in the T7–T18 interval, up to values comparable to those determined in the variants with Cd of the same concentrations. Under the action of lead at 1.0 mg/L and 1.5 mg/L, the maximum value of the protein amount was 260 mg/g dry biomass (T18), which is about 3 times lower than that of the control at T18.
Of the three metals, nickel at 0.5 mg/L was the least inhibitory for protein synthesis by spirulina, while at concentrations of 1.0 mg/L and 1.5 mg/L metal ions, nickel proved to be significantly more inhibitory for protein production than Cd and Pb, probably due to excessive intracellular penetration, with repercussions on metabolic processes.
3.3. Quantification of Dry Biomass Resulted from Spirulina Cultures
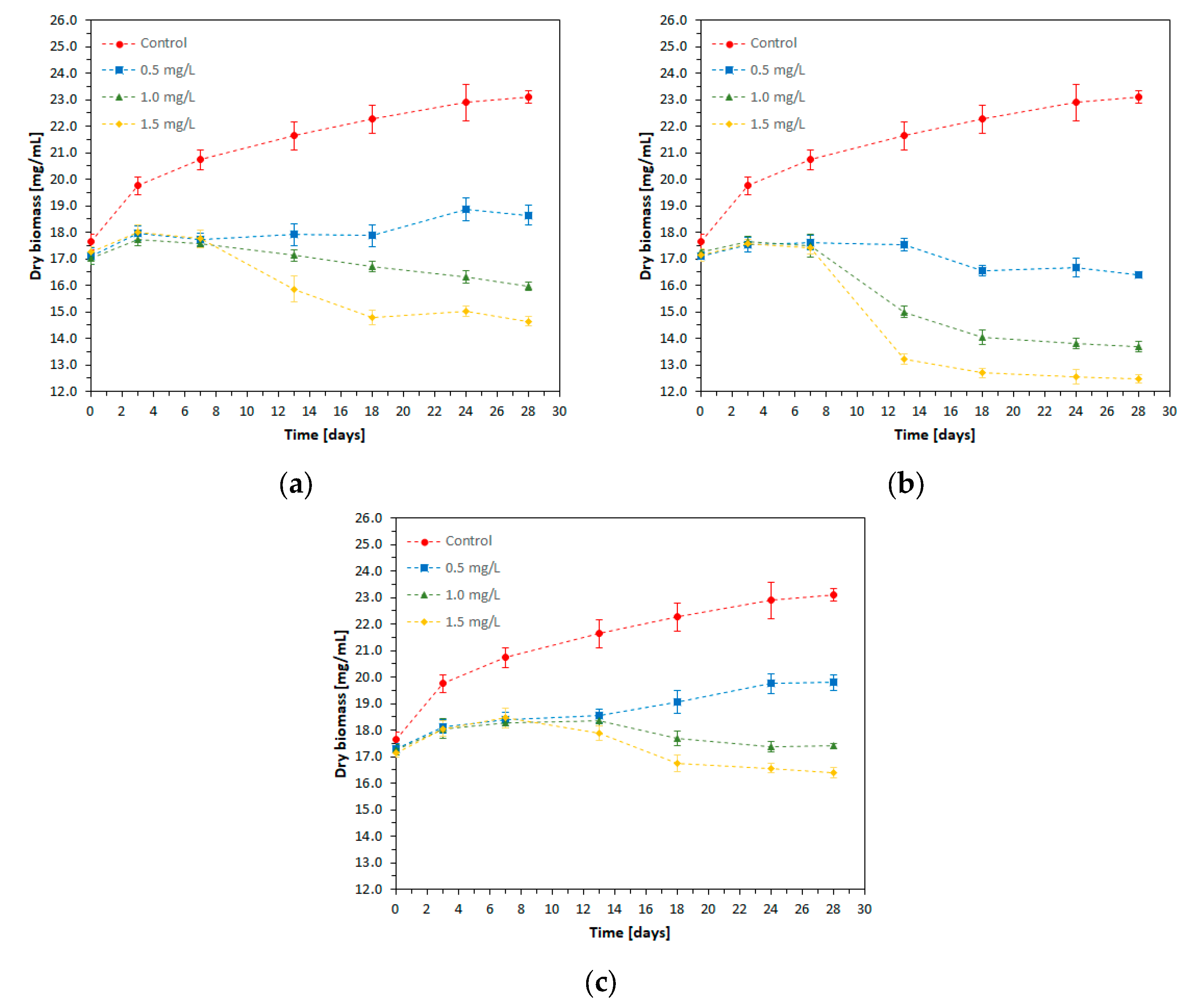
Biomass represents the total amount of cellular mass (viable and non-viable) existing in a culture at a given time. The experimental values of dry biomass from the cell cultures and the time evolution of dry biomass produced in the presence of the studied heavy metals are presented in
Figure 4. The results obtained in the experimental variants containing the toxic substance are compared with the control value, thus being able to estimate the effect of various toxic compounds on the microorganisms’ development process.
The amount of dry biomass produced during spirulina growth depends both on the type and concentration of the heavy metal ion added to the culture medium.
The highest dry biomass concentration and the best time evolution of biomass production were recorded for the control culture throughout the cultivation period, with the amount of dry biomass at the end of the experiment (T28) being approximately 1.3 times higher compared to that determined at T0.
Under the exposure to nickel and cadmium ions, regardless of their concentration, no new biomass production was recorded during the first 7 days of the cultivation period. Afterward, a slight increase in biomass content was observed in the presence of Ni in a concentration of 0.5 mg/L, while at Ni concentrations of 1.0 mg/L and 1.5 mg/L, a decrease in dry biomass was recorded until T28, down to values lower than the initial biomass concentration.
Cadmium, regardless of concentration, caused a decrease in biomass production in the time interval T7–T28.
Regardless of lead concentration, the dry biomass remained almost constant during the first 10 days of spirulina cultivation. After this time interval, the presence of lead ions in a concentration of 0.5 mg/L led to a slight progressive increase in the amount of bio-mass until the end of the cultivation period; lead concentrations of 1.0 mg/L and 1.5 mg/L showed a toxic, inhibitory effect, which determined the reduction of the amount of bio-mass to lower values, or comparable to the initial ones.
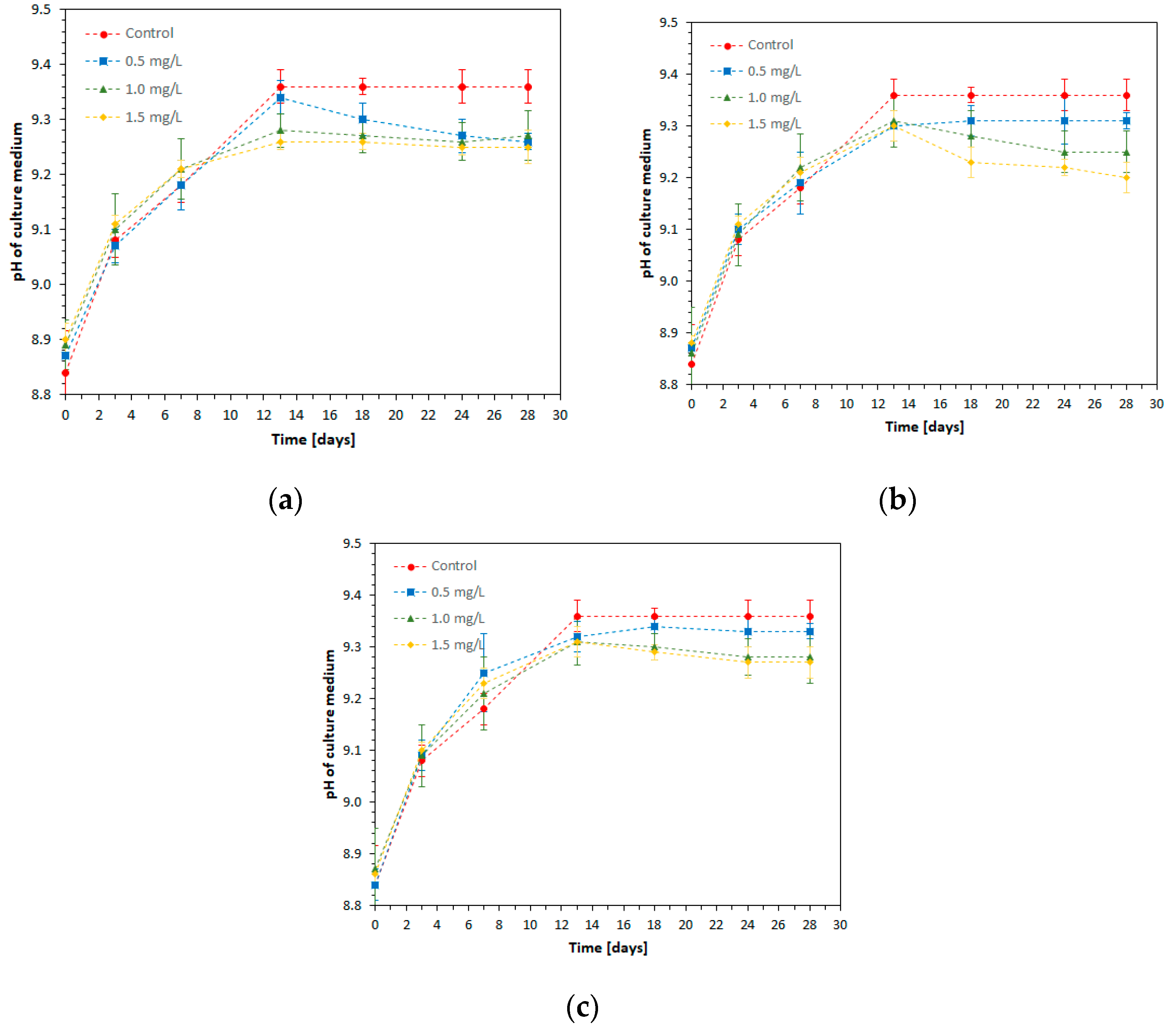
3.4. Time Evolution of pH in S. platensis Cultures under the Action of Metal Ions
One of the main parameters affecting the composition and concentration of biomass produced by
S. platensis is the pH of the culture medium. For optimal development, spirulina requires a relatively high pH (from 9.5 to 11.0 with an optimum of 10.5). In this respect, large amounts of sodium bicarbonate, Na
2CO
3 must always be present in the culture medium to keep pH at high values and inhibit the growth of other algae. The Zarrouk culture medium used in this study contains 18 g/L of Na
2CO
3, which provides a sufficient amount of carbon dioxide (through decomposition) for supporting metabolic processes and maintaining high pH values. The experimental values of the culture samples’ pH, measured over the cultivation period, are presented in
Figure 5.
The initial pH of the culture medium after the addition of spirulina inoculum and heavy metal solutions, in the control culture and the other experimental variants, had values between 8.83 and 8.89.
A pH increase to values above 9.3 was observed in all experimental variants up to the T13 of microalga cultivation, which demonstrates an efficient metabolization of the carbon source (sodium bicarbonate). After this time interval, the pH remained constant in the case of the control sample and a slight decrease in the experimental variants with the addition of metals was recorded, depending on the type of metal ion and its concentration. The fact that pH remained at values above 9.2 until the end of the experiment in all experimental variants proves that this parameter was not a limiting factor for the good development of the spirulina culture.
3.5. The Effect of Metal Ions on the Viability of S. platensis Cultures
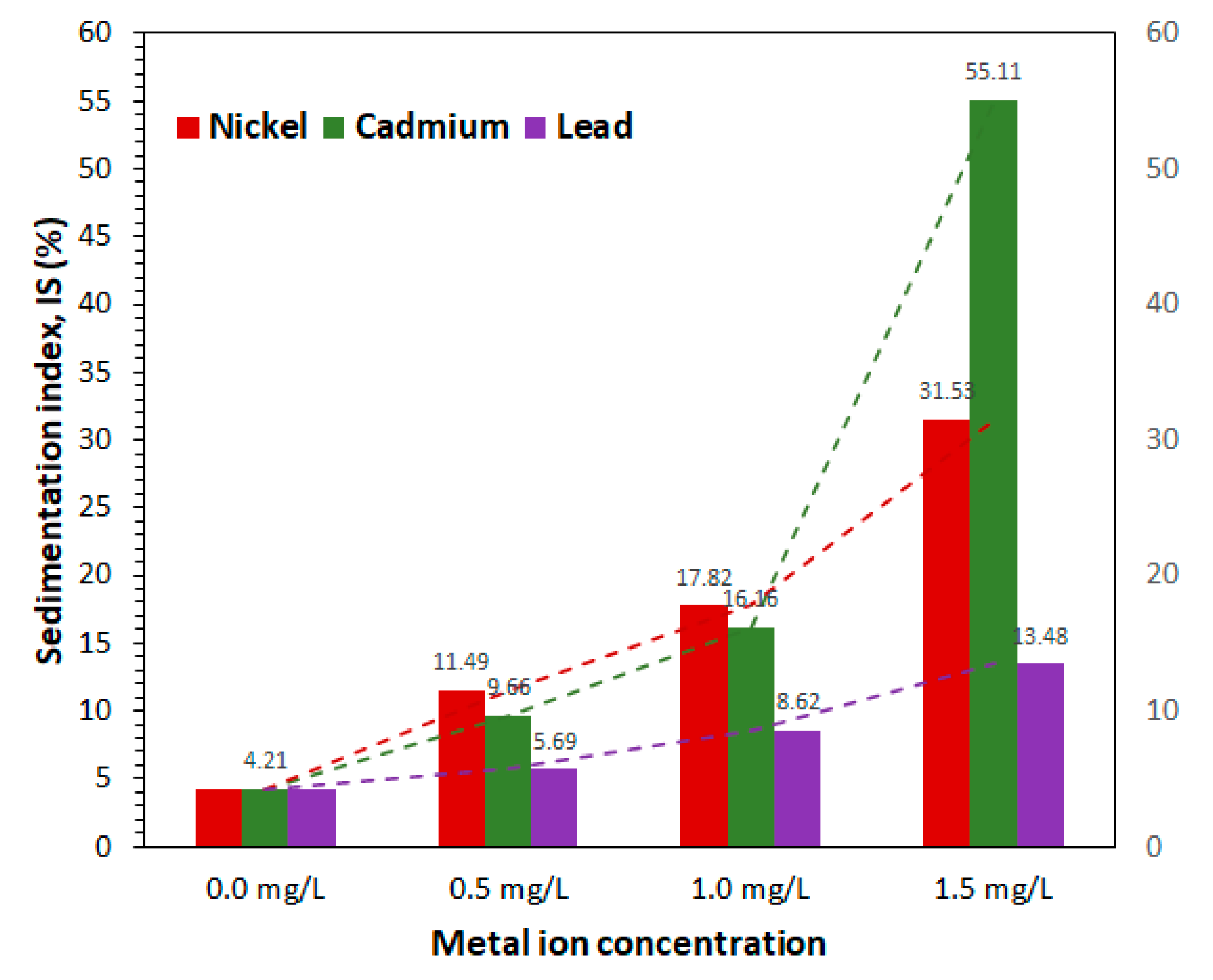
When spirulina is metabolically active and the cells are not altered by external factors, they tend to float due to the gas vacuole system present in large quantities in the cell cytoplasm and form a pellicle on the surface of the culture medium or water. As the microalga is damaged, the cells tend to settle. The functional integrity and viability of spirulina cells can be assessed through the sedimentation index (IS).
The experimental values of the sedimentation index of the studied cultures are given in
Figure 6. The control culture had the lowest sedimentation index (4.21%), demonstrating the viability of this culture, which was still in the exponential growth phase after 28 days of cultivation.
The sedimentation index increased with the increase of metal concentration in the culture medium, which proves the detrimental effect of heavy metals on spirulina viability.
Nickel and cadmium exhibited the most pronounced effect on the viability of spirulina cells. At concentrations of 0.5 mg/L and 1.0 mg/L, Ni and Cd had comparable effects over the sedimentation index, with slightly higher values of IS for the nickel ion: 11.49% for Ni and 9.66% for Cd at 0.5 mg/L, and 17.82% for Ni and 16.16% for Cd at 1.0 mg/L.
Cadmium in a concentration of 1.5 mg/L had the most detrimental effect on spirulina viability, proved by the highest IS of all experimental cultures (55.11%), which show a strong nonlinear dependence between Ni concentration and IS value compared with the values at 0.5 mg/L and 1.0 mg/L. The second most detrimental metal was Ni at 1.5 mg/L, with an IS of 31.53%.
The viability and integrity of spirulina cells were least affected by lead, regardless of the concentration in which it was used; the values of the sedimentation index were 5.69% (Pb-0.5 mg/L), 8.62% (Pb-1.0 mg/L), and 13.48% (Pb-1.5 mg/L). For Pb, the dependence between metal concentration and IS value is almost linear (
Figure 6).
These findings are supported by the visual appearance of the spirulina cultures (
Figure 7) and the microscopic images of spirulina cells at the end of the cultivation period (T
28), in the control culture and the cultures exposed to the heavy metals (
Figure 8,
Figure 9,
Figure 10 and
Figure 11).
In
Figure 7, the control sample had a dark green color due to the density and viability of the spirulina cells, which produced chlorophyll in large amounts. Also, cultures containing lead ions preserved their intense green color, mainly when exposed to 0.5 mg/L Pb. Conversely, cultures with nickel and cadmium at concentrations of 1.0 and 1.5 mg/L, respectively, were strongly affected, the green color had almost disappeared because chlorophyll was destroyed, and the cell mass agglomerated in flocks and settled on the bottom of the Erlenmeyer flasks.
The microscopic image of the control spirulina culture (see
Figure 8) highlighted the abundance of cells and the preservation of the main morphological characteristic of this microalga, namely the helical twisting of cells, though, at the end of the cultivation period, some cells gained a filiform shape.
Microscopic images from
Figure 9 highlight the effect of nickel concentration on the viability of spirulina cells. At a concentration of 0.5 mg/L nickel, viable cells with characteristic helical shape were present, while at concentrations of 1.0 and 1.5 mg/L Ni, most of the cell mass was lysed and the remaining cells had lost spiral geometry.
The microscope images from
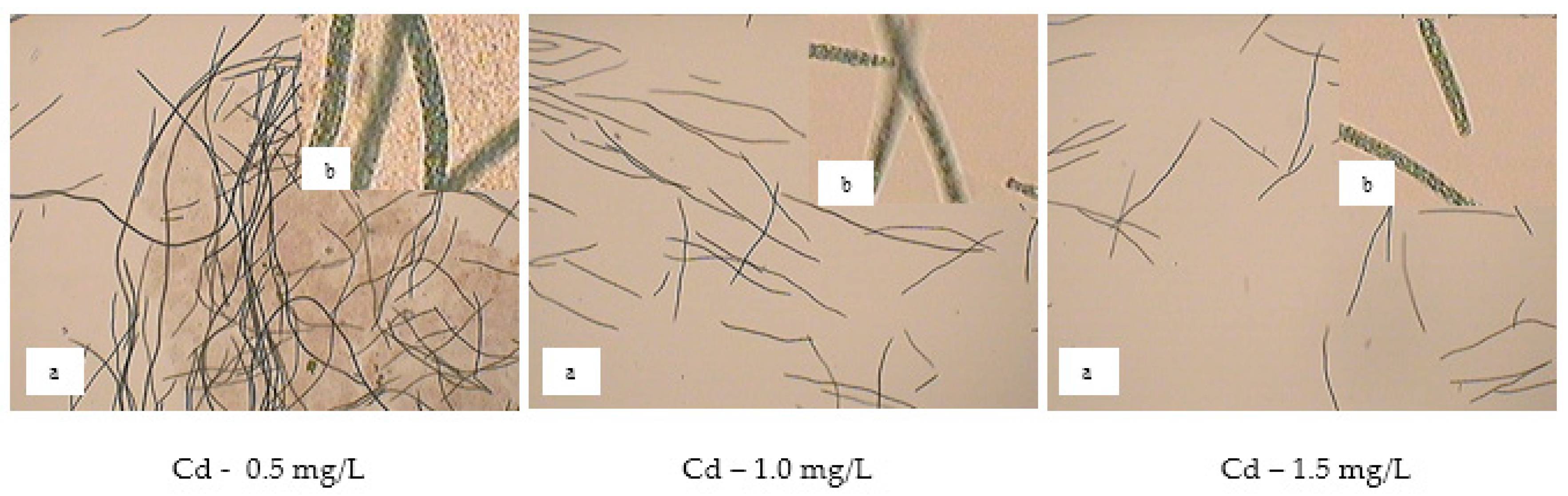
Figure 10 show the effect of cadmium concentration on the morphology of spirulina cells after 28 days of cultivation. The loss of the helical shape was apparent for most of the cultured cells even at 0.5 mg/L Cd. With the metal concentration increase, more cells lost their helical shape and underwent lysis, which determined the decrease of cell density of the microalga culture.
The influence of heavy metal ions on the growth of spirulina cultures depends both on the type of metal ion and its concentration in the culture media. In this respect, our study confirms and complements data in the literature [
24,
25,
26,
27,
28,
29]. The mechanisms of the interaction of heavy metal ions and
S. platensis are highly invasive in the case of cadmium, moderate for nickel, and less intense for lead, in accordance with previous data [
30].
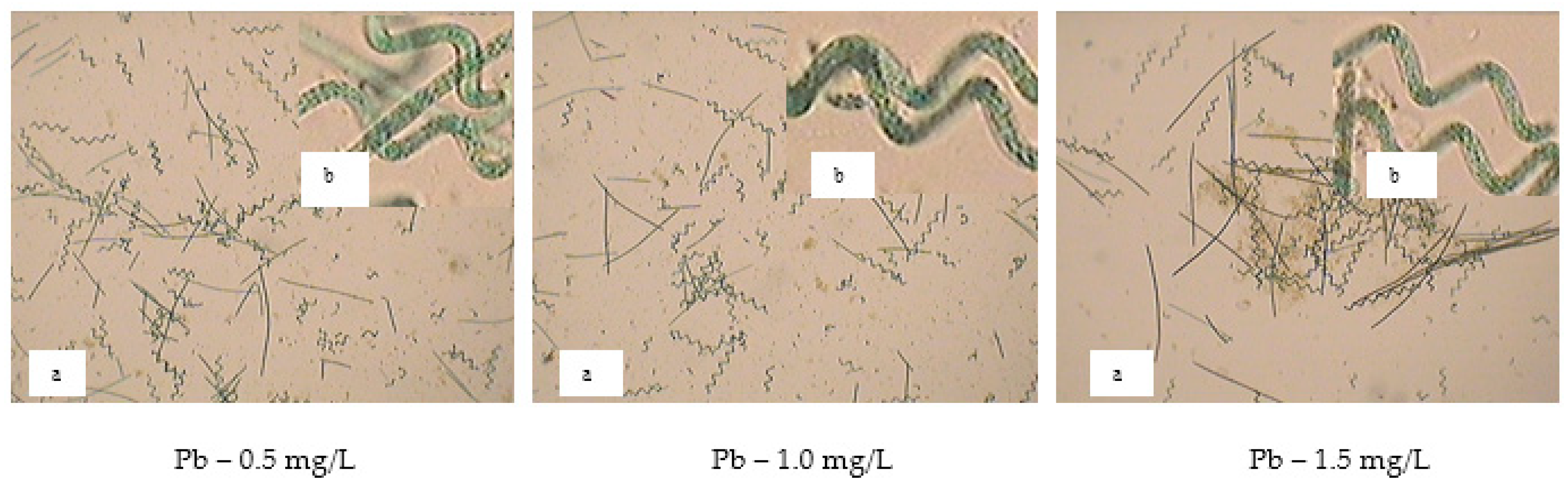
The microscopic images of spirulina cultures developed in the presence of 0.5 mg/L Pb showed cell abundance and morphological integrity. At 1.0 mg/L Pb, the number of cells decreased, but they preserved their spiral shape, while in the culture with 1.5 mg/L Pb the cell density was much reduced, with some cells lysed and flocculated (
Figure 11).
3.6. ANOVA Analysis of the Effect of Metal Ions on the Development of S. platensis Cultures
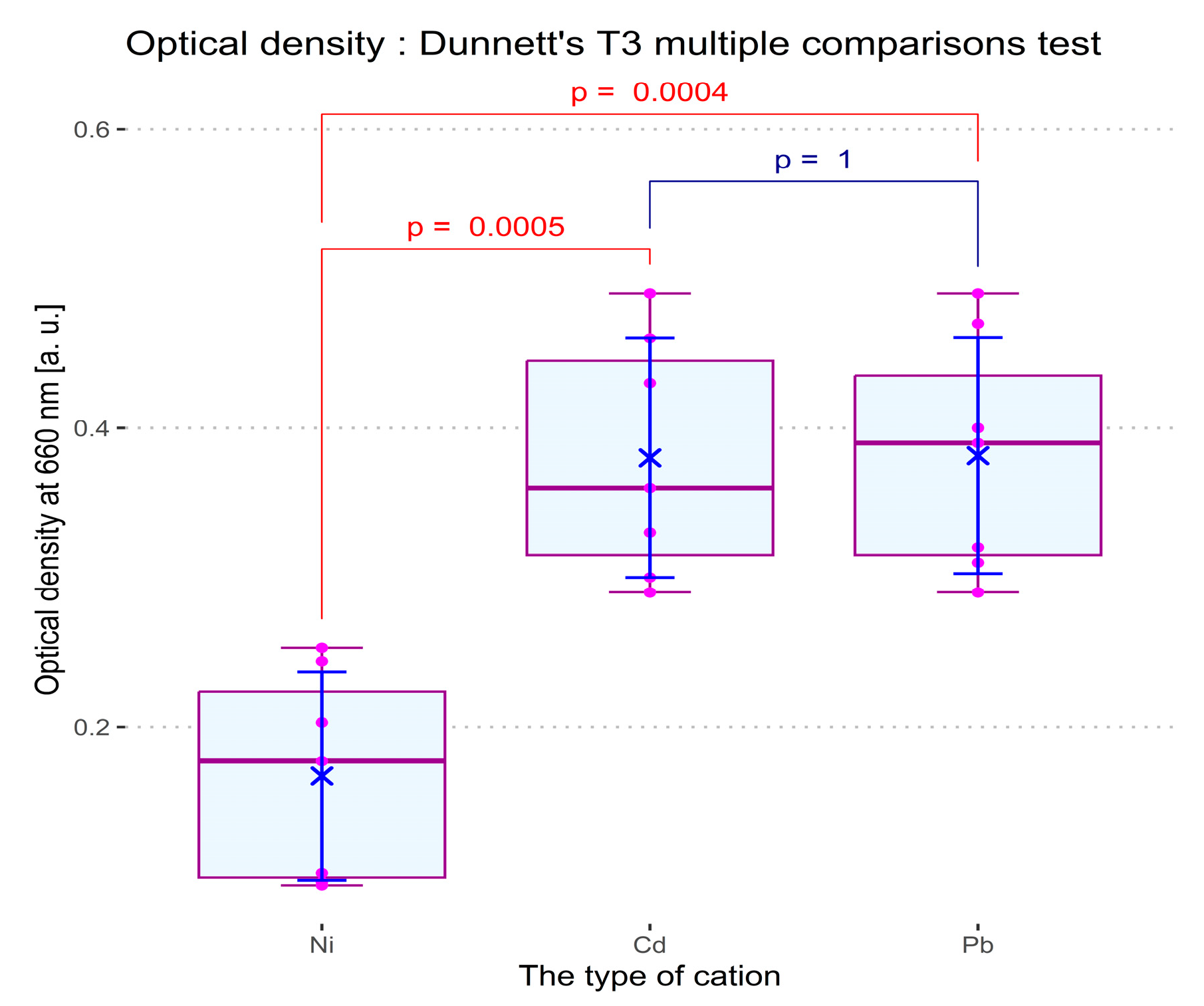
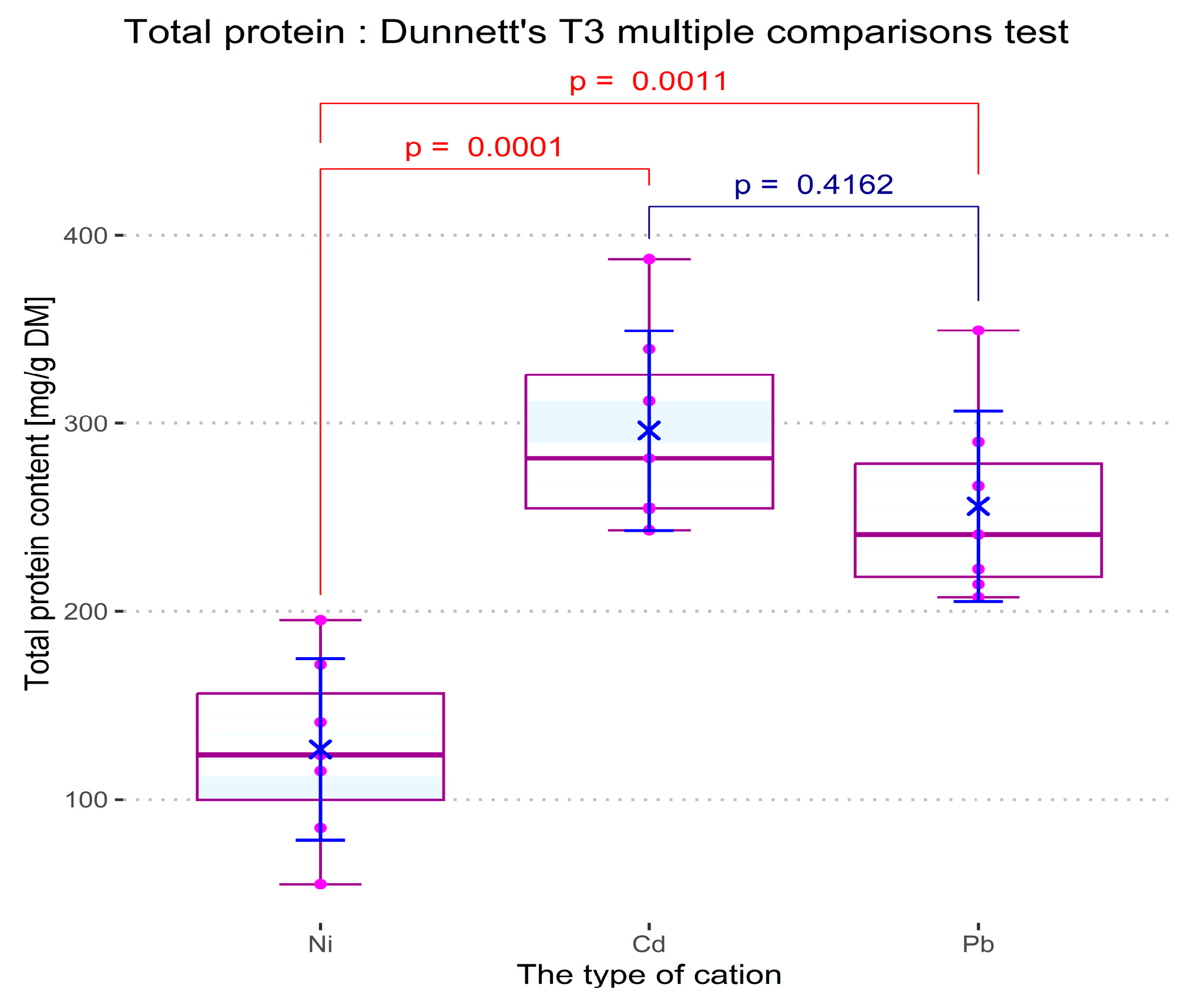
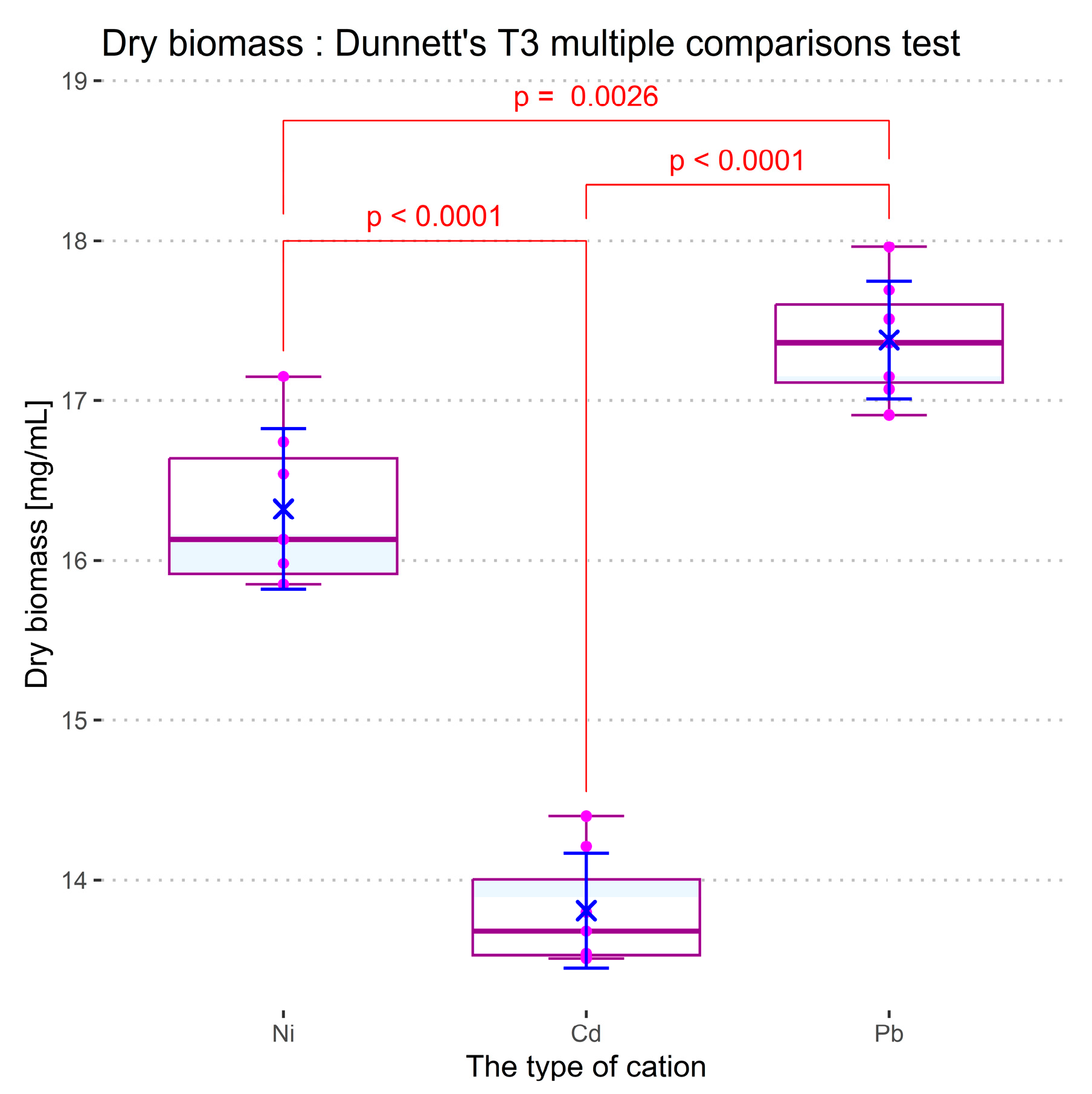
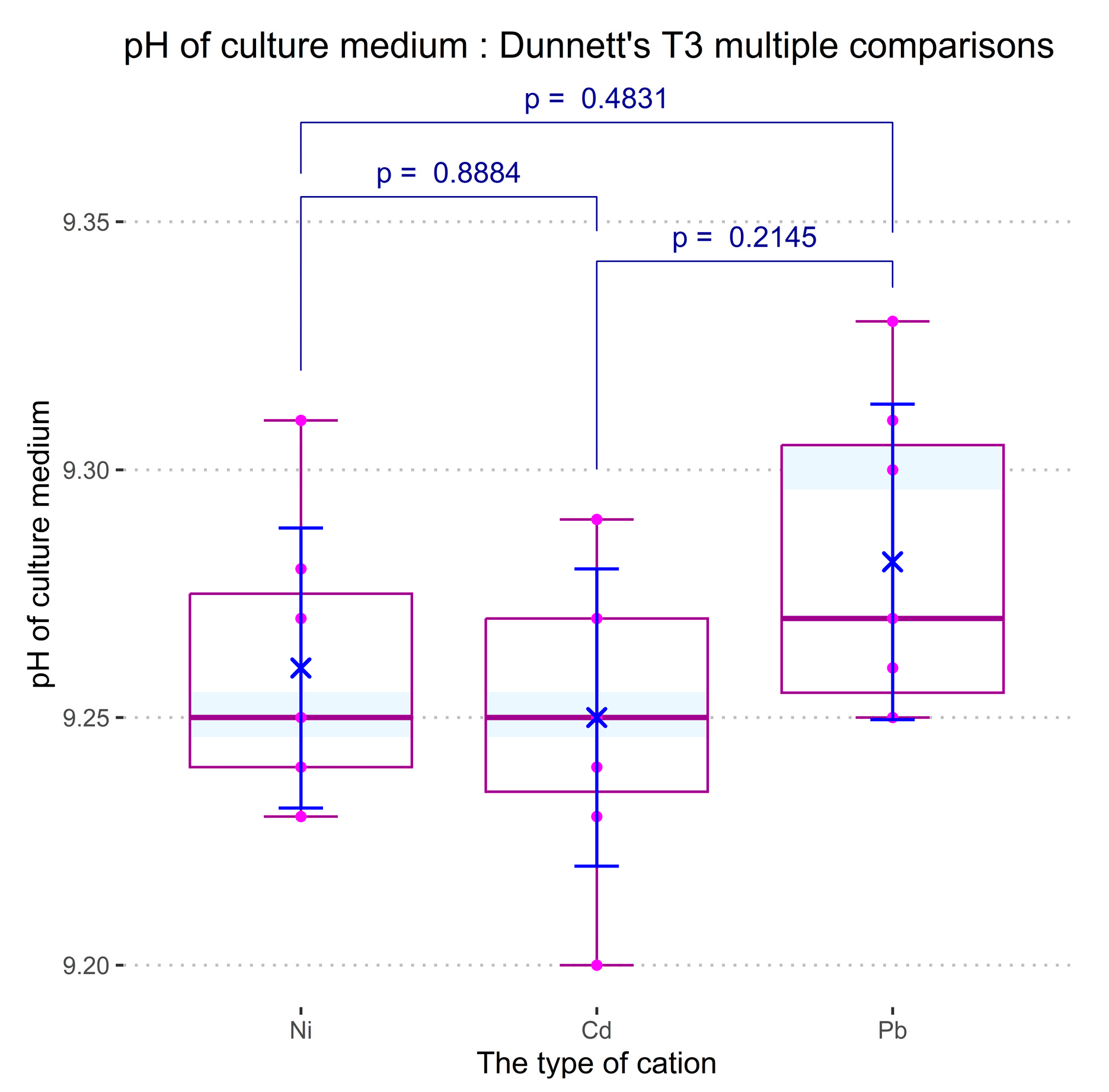
In order to investigate the effect of Ni (II), Cd (II), and Pb (II) concentration in the culture medium on the development of spirulina cyanobacteria, the ANOVA technique was applied. Two sets of comparisons were performed for each measured response: considering the concentration level of the cations as the independent variable, and considering the nature of cations (in a concentration of 1 mg/L) as an influential factor, respectively. For both cases, the optical density, the total protein content of biomass, and the dry biomass content were considered dependent variables. The pH value of the samples was also taken into consideration as a parameter of culture development in the presence of cations, but the recorded dependencies were statistically indiscernible for both comparison types.
Script run listings (obtained as specified in
Section 2.9) are presented in the sixteen log files included as an archive in the
Supplementary Materials. The graphical results of the one-way ANOVA performed for the second set of comparisons, which takes into account the nature of the cations as an influencing factor, are also presented in
Figure 12,
Figure 13,
Figure 14 and
Figure 15. As compared with the control culture (in which no heavy metal salts were added), all cations induce depletion of the cyanobacteria development, which increases proportionally with cation concentration. The influence of the heavy metal salts on the pH value of the culture media remains slightly below the limit of statistical significance in all the experiments. Based on the Benjamini –Krieger–Yekutieli test (see the ANOVA and post-hoc analysis logs in
Supplementary Material), when dosed at a concentration of 1 mg/L, Pb (II) cations appear to act minimally as a toxicant, while Ni (II) and Cd (II) have a detrimental effect on the spirulina biomass and total protein content.
4. Conclusions
Spirulina platensis microalga is most suitable for laboratory ecotoxicity studies because it is easy to cultivate, has a fast growth rate, and is very sensitive to any change in the physical and chemical conditions of the cultivation process.
The influence of heavy metal ions on the growth of spirulina cultures depends both on the type of metal ion and its concentration in the culture media, which is in complete concordance with the previously published data.
The damaging interaction between S. platensis and the studied metals was the highest in the case of cadmium, moderate for nickel, and the lowest for lead. Nickel and cadmium proved to be the most toxic for cell growth and biomass accumulation at concentrations of 1.0 mg/L and 1.5 mg/L.
Looking at the values of the total protein content in the experimental variants, we can conclude that nickel is the least toxic at 0.5 mg/L and the most toxic at concentrations of 1.0 mg/L and 1.5 mg/L, probably due to excessive intracellular penetration, with repercussions/negative impact on metabolic processes.
Cadmium, regardless of concentration, caused a decrease in dry biomass throughout the microalgae cultivation period.
The pH increased to values above 9.3 in all experimental variants until the 13th day of microalga cultivation, which shows an effective decomposition of sodium bicarbonate, both under the action of light energy and as a metabolic requirement of spirulina cells, and remained at values above 9.2 until the end of the cultivation period.
The cultures treated with cadmium and nickel at 1.5 mg/L exhibited the highest values of the degree of sedimentation: 55.11% and 31.53%, respectively. Lead exerted the lowest effect on the viability and integrity of spirulina cells, regardless of the concentration in which it was used.
In our study, nickel proved to be more toxic than cadmium and lead. This may be due to nickel penetration inside the cells by taking up essential trace elements, with the impairment of the metabolic processes, while cadmium and lead bind to the active groups on the surface of the cell wall, preventing nutrients from entering the cells.