Do Microplastics Affect the Photodegradation of Duloxetine and Its Phototoxicity to Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microparticles
2.2. Chemicals
2.3. Photodegradation Experiments
2.4. Liquid Chromatography with Photodiode Array Detector
2.5. Liquid Chromatography with Mass Spectrometer Detector
2.6. Protozoan S. ambiguum
2.7. Spirotox Assay
2.8. Ingestion Studies
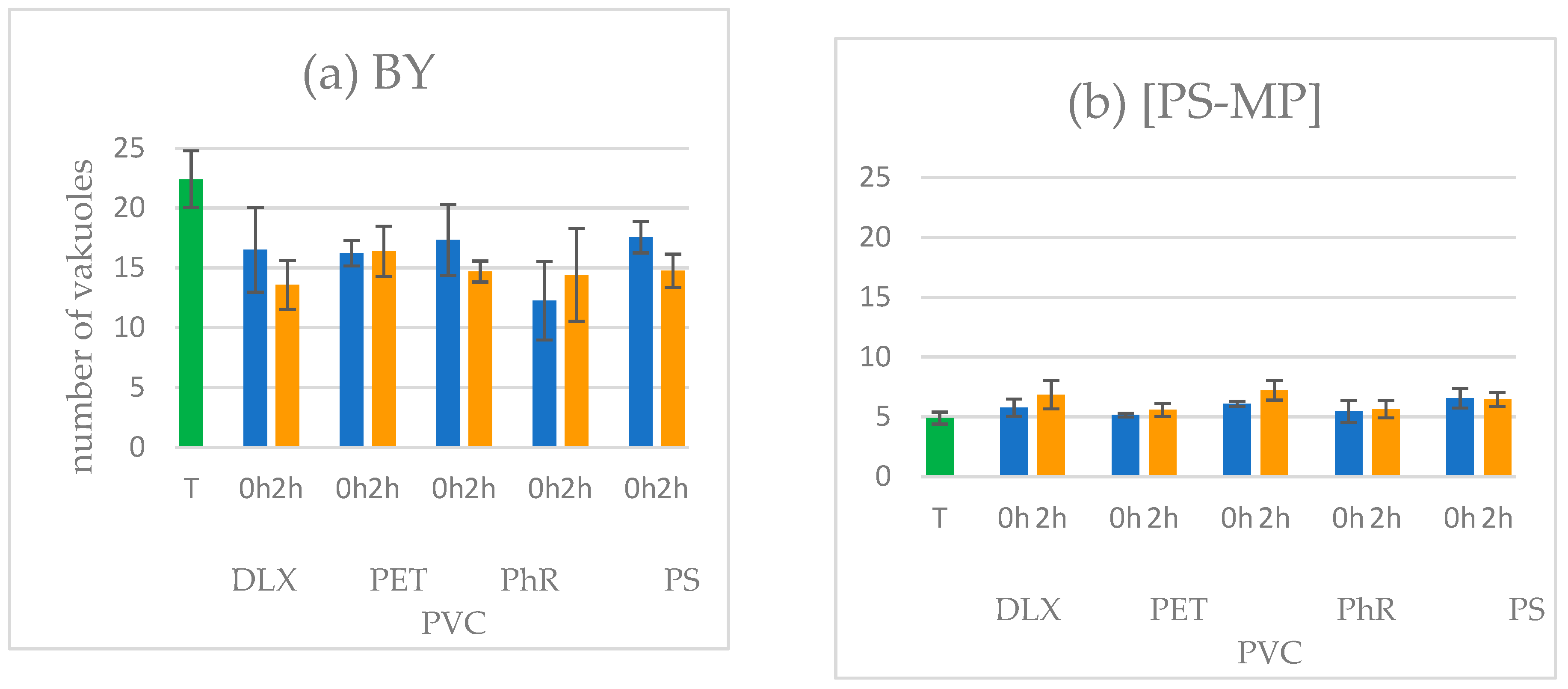
2.9. Counting of the Food Vacuoles in the Protozoa
2.10. In Silico Toxicity Assay
2.11. Data Treatment
3. Results
3.1. Duloxetine Toxicity in the Spirotox Assay
3.2. Photodegradation of Duloxetine
3.3. Ingestion Assay
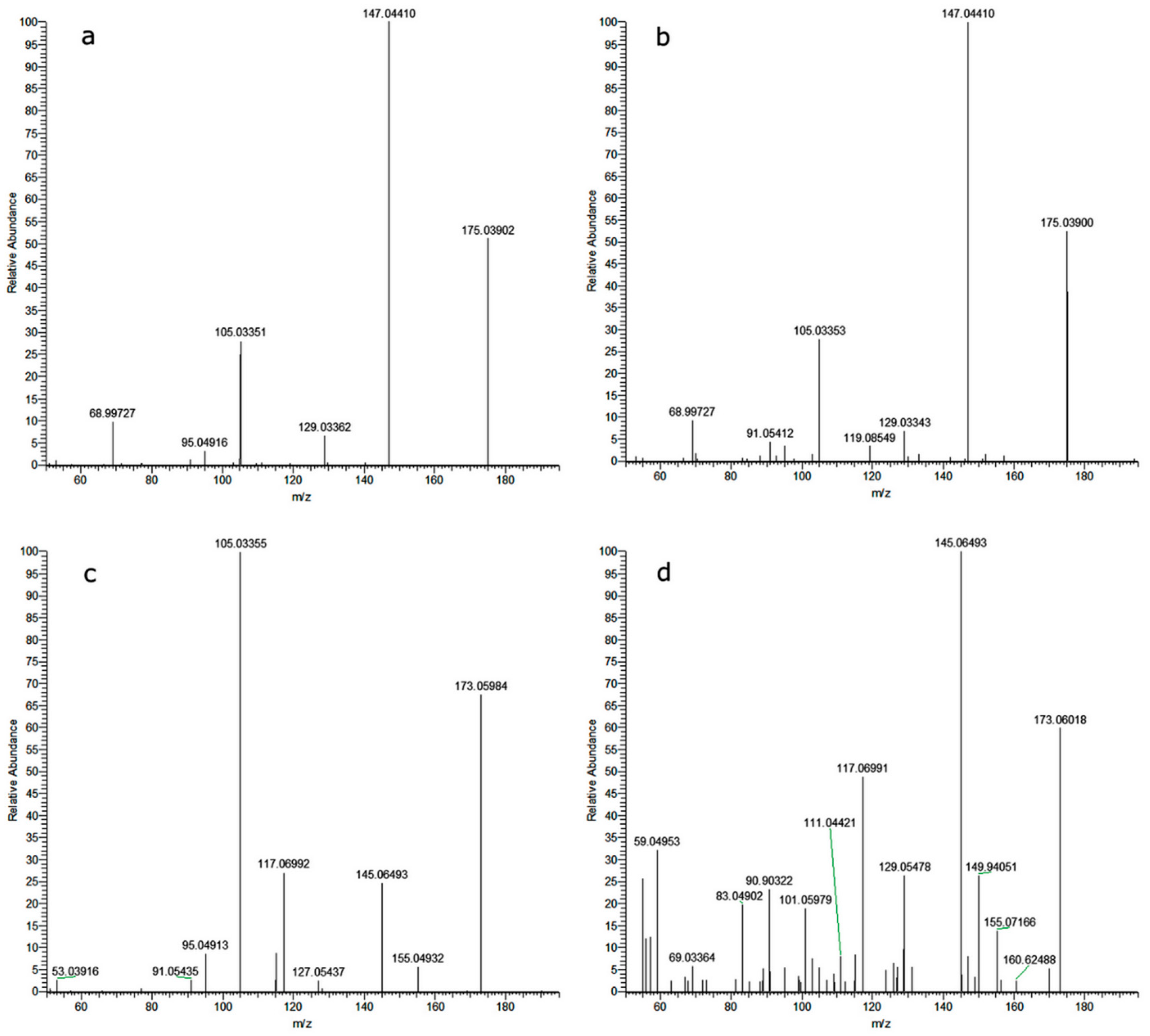
3.4. Analyses of Photoproducts with HPLC-MS/MS
3.5. Toxicity Assessment of DLX Photoproducts
4. Discussion
4.1. Irradiation of DLX
4.2. DLX Photodegradation Products
4.3. Toxicity of the DLX Derivatives
4.4. Ecological Consequences and Perspectives for Future Research
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Available online: www.clincalc.com (accessed on 31 May 2023).
- Fernandez, J.P.; Almeida, C.M.R.; Salgado, M.A.; Carvalho, M.F. Pharmaceutical compounds in aquatic environments—Occurrence, fate and bioremediation prospective. Toxics 2021, 9, 257. [Google Scholar] [CrossRef] [PubMed]
- Osawa, R.A.; Carvalho, A.P.; Monteiro, O.C.; Florêncio, M.H. Degradation of duloxetine: Identification of transformation products by UHPLC-ESI(+)-HRMS/MS, in silico toxicity and wastewater analysis. J. Environ. Sci. 2019, 82, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.M.; Furlong, E.T. Trace analysis of antidepressant pharmaceuticals and their select degradates in aquatic matrixes by LC/ESI/MS/MS. Anal. Chem. 2008, 80, 1756–1762. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.M.; Furlong, E.T.; Kolpin, D.W.; Werner, S.L.; Schoenfuss, H.L.; Barber, L.B.; Blazer, V.S.; Norris, D.O.; Vaida, A.M. Antidepressant pharmaceuticals in two U.S. effluent-impacted streams: Occurrence and fate in water and sediment, and selective uptake in fish neural tissue. Environ. Sci. Technol. 2010, 44, 1918–1925. [Google Scholar] [CrossRef]
- Chadha, R.; Bali, A.; Bansal, G. Characterization of stress degradation products of duloxetine hydrochloride employing LC-UV/PDA and LC-MS/TOF studies. J. Pharm. Biomed. Anal. 2016, 121, 39–55. [Google Scholar] [CrossRef] [PubMed]
- Trawiński, J.; Skibiński, R. Studies on photodegradation process of psychotropic drugs: A review. Environ. Sci. Pollut. Res. 2017, 24, 1152–1199. [Google Scholar] [CrossRef]
- Akdogan, Z.; Guven, B. Microplastics in the environment: A critical review of current understanding and identification of future research needs. Environ. Pollut. 2019, 254, 113011. [Google Scholar] [CrossRef]
- Abeynayaka, A.; Werellagama, I.; Ngoc-Bao, P.; Hengesbaugh, M.; Gajanayake, P.; Nallaperuma, B.; Karkour, S.; Bui, X.T.; Itsubo, N. Microplastics in wastewater treatment plants. Curr. Dev. Biotechnol. Bioengin. 2022, 311–337. [Google Scholar] [CrossRef]
- Santos, L.H.M.L.M.; Rodríguez-Mozaz, S.; Barceló, D. Microplastics as vectors of pharmaceuticals in aquatic organisms—An overview of their environmental implications. CSCEE 2021, 3, 100079. [Google Scholar] [CrossRef]
- Li, C.; Jiang, B.; Guo, J.; Sun, C.; Shi, C.; Huang, S.; Liu, W.; Wu, C.; Zhang, Y. Aging Process of Microplastics in the Aquatic Environments: Aging Pathway, Characteristic Change, Compound Effect, and Environmentally Persistent Free Radicals Formation. Water 2022, 14, 3515. [Google Scholar] [CrossRef]
- Hartmann, N.B.; Hüffer, T.; Thompson, R.C.; Hassellöv, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsoon, T.; Brennholt, N.; Cole, M.; et al. Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris. Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar] [CrossRef]
- Martin, J.; Santos, J.L.; Aparicio, I.; Alonso, E. Microplastics and associated emerging contaminants in the environment: Analysis, sorption mechanisms and effects of co-exposure. Trends Environ. Anal. Chem. 2022, 35, e00170. [Google Scholar] [CrossRef]
- Atugoda, T.; Vithanage, M.; Wijesekara, H.; Balan, N.; Sarmah, A.K.; Bank, M.S.; You, S.; Ok, Y.S. Interactions between microplastics, pharmaceuticals and personal care products: Implications for vector transport. Environ. Int. 2021, 49, 106367. [Google Scholar] [CrossRef]
- Ma, J.; Zhao, J.; Zhu, Z.; Li, L.; Yu, F. Effect of microplastic size on the adsorption behavior and mechanism of triclosan on polyvinyl chloride. Environ. Pollut. 2019, 254, 113104. [Google Scholar] [CrossRef] [PubMed]
- Puckowski, A.; Ćwięk, W.; Mioduszewska, K.; Stepnowski, P.; Białk-Bielińska, A. Sorption of pharmaceuticals on the surface of microplastics. Chemosphere 2021, 263, 127976. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Wang, X.; Zhou, X.; Kong, X.; Tao, S.; Xing, B. Sorption of four hydrophobic organic compounds by three chemically distinct polymers: Role of chemical and physical composition. Environ. Sci. Technol. 2012, 46, 7252–7259. [Google Scholar] [CrossRef]
- Wang, X.; Zheng, H.; Zhao, J.; Luo, X.; Wang, Z.; Xing, B. Photodegradation elevated the toxicity of polystyrene microplastics to grouper (Epinephelus moara) through disrupting hepatic lipid homeostasis. Environ. Sci. Technol. 2020, 54, 6202–6212. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Pedriza, A.; Jaumot, J. Interaction of environmental pollutants with microplastics: A critical review of sorption factors, bioaccumulation and ecotoxicological effects. Toxics 2020, 8, 40. [Google Scholar] [CrossRef]
- Kong, F.; Xu, X.; Xue, Y.; Gao, Y.; Zhang, L.; Wang, L.; Jiang, S.; Zhang, Q. Investigation of the adsorption of sulfamethoxazole by degradable microplastics artificially aged by chemical oxidation. Arch. Environ. Contam. Toxicol. 2021, 81, 155–165. [Google Scholar] [CrossRef]
- Zhu, K.; Jia, H.; Zhao, S.; Xia, T.; Guo, X.; Wang, T.; Zhu, L. Formation of environmentally persistent free radicals on microplastics under light irradiation. Environ. Sci. Technol. 2019, 53, 8177–8186. [Google Scholar] [CrossRef]
- Minguez, L.; Pedelucq, J.; Farcy, E.; Ballandonne, C.; Budzinski, H.; Halm-Lemeille, M.P. Toxicities of 48 pharmaceuticals and their freshwater and marine environmental assessment in Northwestern France. Environ. Sci. Pollut. Res. 2016, 23, 4992–5001. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka, J.; Drobniewska, A.; Lenga, W.; Misztal, J.; Wawryniuk, M.; Nałęcz-Jawecki, G. The mutual effect of microparticles and antidepressants on the protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water 2023, 15, 552. [Google Scholar] [CrossRef]
- Li, F.H.; Yao, K.; Lv, W.Y.; Liu, G.G.; Chen, P.; Huang, H.P.; Kang, Y.P. Photodegradation of ibuprofen under UV-Vis irradiation: Mechanism and toxicity of photolysis products. Bull. Environ. Contam. Toxicol. 2015, 94, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ma, L.-Y.; Li, L.-S.; Xu, L. Photodegradation kinetics, transformation, and toxicity prediction of ketoprofen, carprofen, and diclofenac acid in aqueous solutions. Environ. Toxicol. Chem. 2017, 36, 3232–3239. [Google Scholar] [CrossRef] [PubMed]
- Uzelac, M.M.; Armaković, S.J.; Armaković, S.; Cetojević-Simin, D.D.; Agbaba, J.; Binić, N.D. The role of environmental waters ionic composition and UV-LED radiation on photodegradation, mineralization and toxicity of commonly used beta-blockers. J. Mol. Struct. 2022, 1249, 131579. [Google Scholar] [CrossRef]
- Spina, M.; Venancio, W.; Rodrigues-Silva, C.; Pivetta, R.C.; Diniz, V.; Rath, S.; Guimaraes, J.R. Degradation of antidepressant pharmaceuticals by photoperoxidation in diverse water matrices: A highlight in the evaluation of acute and chronic toxicity. Environ. Sci. Pollut. Res. 2021, 28, 24034–24045. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G.; Chojnacka, J.; Wawryniuk, M.; Drobniewska, A. Influence of Nano and Small Microplastics on Ciliated Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water 2021, 13, 2857. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G.; Sawicki, J. Toxicity of inorganic compounds in the Spirotox test: A miniaturized version of the Spirostomum ambiguum test. Arch. Environ. Contam. Toxicol. 1998, 34, 1–5. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G. Spirotox Test—Spirostomum ambiguum Acute Toxicity Test. In Small-Scale Freshwater Toxicity Investigations: Toxicity Test Methods; Blaise, C., Férard, J.-F., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 299–322. [Google Scholar]
- Available online: https://www.epa.gov/chemical-research/toxicity-estimation-software-tool-test (accessed on 31 May 2023).
- Valimana-Traverso, J.; Amariei, G.; Boltes, K.; Garcia, M.A.; Marina, M.L. Stability and toxicity studies for duloxetine and econazole on Spirodela polyrhyza using chiral capillary electrophoresis. J. Hazard. Mater. 2019, 374, 203–210. [Google Scholar] [CrossRef]
- Valimana-Traverso, J.; Amariei, G.; Boltes, K.; Garcia, M.A.; Marina, M.L. Enantiomer stability and combined toxicity of duloxetine and econazole on Daphnia magma using real concentrations determined by capillary electrophoresis. Sci. Total Environ. 2019, 670, 770–778. [Google Scholar] [CrossRef]
- Datar, P.A.; Waghmare, R.U. Development and validation of an analytical method for the stability of duloxetine hydrochloride. J. Taibah Univ. Sci. 2014, 8, 357–363. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.; Su, F.; Wang, Y.; Peng, L.; Liu, D. Adsorption behaviour of microplastics on the heavy metal Cr(VI) before and after ageing. Chemosphere 2022, 302, 134865. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Zhu, K.; Ma, H.; Liu, J.; Zhang, C.; Dai, Y.; Jia, H. Sulfur-containing persistent free radicals and reactive species on photoaged microplastics: Identification and the formation mechanism. Environ. Sci. Technol. 2023, 57, 8680–8690. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Jiang, Y.; Gu, Y.; Ding, P.; Wang, C.; Pan, R.; Shi, C.; Zeng, L.; Chen, X.; Li, H. The generation of environmentally persistent free radicals on photoaged microbeads from cosmetics enhances the toxicity via oxidative stress. Environ. Int. 2023, 174, 107875. [Google Scholar] [CrossRef]
- Santoke, H.; Song, W.; Cooper, W.J.; Peake, B.M. Advanced oxidation treatment and photochemical fate of selected antidepressant pharmaceuticals in solutions of Suwannee River humic acid. J. Hazard. Mater. 2012, 217–218, 382–390. [Google Scholar] [CrossRef]
- Brussol, C.; Duane, M.; Carlier, P.; Kotzias, D. Photo-induced OH reactions of naphthalene and its oxidation products on SiO2. Environ. Sci. Pollut. Res. 1999, 6, 138–140. [Google Scholar] [CrossRef]
- Vialation, D.; Richard, C.; Baglio, D.; Paya-Perez, A.-B. Mechanism of the photochemical transformation of naphthalen in water. J. Photochem. Photobiol. A Chem. 1999, 123, 15–19. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G.; Sawicki, J. Spirotox—A new tool for testing the toxicity of volatile compounds. Chemosphere 1999, 38, 3211–3218. [Google Scholar] [CrossRef]
- Ding, F.; Guo, J.; Li, Z.; Li, L.Y.; Zhang, J.Y.; Zhang, J.H.; Lian, J.; Song, W.H.; Zhu, L. Evaluation and structure-activity relationship study of acute toxicity of naphthoquinones to Photobacterium phosphoreum, Photobacterium T3B. Bull. Environ. Contam. Toxicol. 2010, 85, 116–120. [Google Scholar] [CrossRef]
- Barata, C.; Varo, I.; Navarro, J.C.; Arun, S.; Porte, C. Antioxidant enzyme activities and lipid peroxidation in the freshwater cladoceran Daphnia magna exposed to redox cycling compounds. Comp. Biochem. Physiol. C 2005, 140, 175–186. [Google Scholar] [CrossRef]
- Klotz, L.-O.; Hou, X.; Jacob, C. 1,4-Naphthoquinones: From oxidative damage to cellular and inter-cellular signalling. Molecules 2014, 19, 14902–14918. [Google Scholar] [CrossRef] [PubMed]



| Name | Duloxetine |
|---|---|
| Formula | C18H19NOS |
| IUPAC name | (+)-(S)-N-Methyl -3-naphthalen-1-yloxy0-3-(thiophen-2-yl)propan-1-amine |
| Chemical structure |  |
| UV/Vis absorption spectrum |  |
| Sample | 24 h-EC50 | 24 h-EC20 | 48 h-EC50 | 48 h-EC20 |
|---|---|---|---|---|
| DLX 1 | 1.11 ± 0.26 | 0.98 ± 0.22 | 0.91 ± 0.04 | 0.75 ± 0.09 |
| DLX + PET | 1.03 ± 0.34 | 0.83 ± 0.23 | 0.96 ± 0.34 | 0.77 ± 0.23 |
| DLX + PhR | 0.87 ± 0.19 | 0.76 ± 0.16 | 0.83 ± 0.07 | 0.67 ± 0.10 |
| DLX + PS | 0.91 ± 0.18 | 0.80 ± 0.20 | 0.87 ± 0.17 | 0.74 ± 0.15 |
| DLX + PVC | 1.11 ± 0.25 | 0.91 ± 0.23 | 1.02 ± 0.20 | 0.82 ± 0.12 |
| Sample | Degradation Time [h] | TUmix 1 | TUDLX 2 | TUprod 3 |
|---|---|---|---|---|
| DLX | 0 | 18.5 ± 3.8 | 18.5 ± 3.8 | - |
| 1 | 12.4 ± 1.0 | 1.8 ± 0.6 | 10.6 ± 1.1 | |
| 2 | 8.4 ± 1.9 | 0.8 ± 0.3 | 7.6 ± 1.8 | |
| DLX + PET | 0 | 21.2 ± 8.3 | 21.2 ± 8.3 | - |
| 1 | 12.3 ± 1.5 | 1.4 ± 0.4 | 10.9 ± 1.2 | |
| 2 | 9.2 ± 0.1 | 0.7 ± 0.1 | 8.5 ± 0.1 | |
| DLX + PhR | 0 | 23.9 ± 6.0 | 23.9 ± 6.0 | - |
| 1 | 10.0 ± 4.1 | 0.9 ± 0.4 | 9.1 ± 4.3 | |
| 2 | 6.0 ± 0.4 | 0.6 ± 0.1 | 5.4 ± 0.4 | |
| DLX + PS | 0 | 22.7 ± 5.1 | 22.7 ± 5.1 | - |
| 1 | 10.0 ± 1.2 | 1.2 ± 0.1 | 8.8 ± 1.3 | |
| 2 | 7.4 ± 1.7 | 0.7 ± 0.1 | 6.7 ± 1.6 | |
| DLX + PVC | 0 | 18.7 ± 4.7 | 18.7 ± 4.7 | - |
| 1 | 9.8 ± 0.5 | 1.5 ± 0.3 | 8.3 ± 0.6 | |
| 2 | 5.8 ± 0.8 | 0.6 ± 0.1 | 5.2 ± 0.8 |
| Name | Formula | Molecular Weight | Δ Mass [ppm] | Sfit a [%] | H/C b | RDBE c | MS2 |
|---|---|---|---|---|---|---|---|
| M329a | C18H19NO3S | 329.1084 | −0.43 | 78 | 1.1 | 10 | 312; 202; 169; 143 |
| M329b | C18H19NO3S | 329.1085 | −0.11 | 78 | 1.1 | 10 | 312; 299; 281; 175 |
| M325a | C18H15NO3S | 325.0771 | −2.56 | 77 | 0.8 | 12 | 132; 114; 105 |
| M325b | C18H15NO3S | 325.0772 | −0.28 | 79 | 0.8 | 12 | 290; 242; 213 |
| M323 | C18H13NO3S | 323.0615 | −0.31 | 81 | 0.7 | 13 | 212; 198; 111 |
| M321 | C18H11NO3S | 321.0458 | −0.38 | 62 | 0.6 | 14 | 294; 238; 182 |
| M313a | C18H19NO2S | 313.1134 | −0.81 | 77 | 1.1 | 10 | 296; 212; 161; 143 |
| M313b | C18H19NO2S | 313.1133 | −1.10 | 81 | 1.1 | 10 | 173; 97 |
| M307 | C18H13NO2S | 307.0664 | −0.91 | 81 | 0.7 | 13 | 293; 290; 275; 224 |
| M295 | C18H17NOS | 295.1030 | −0.14 | 83 | 0.9 | 11 | 186; 157; 145 |
| M293 | C18H15NOS | 293.0872 | −0.66 | 89 | 0.8 | 12 | 278; 251; 184 |
| M283 | C17H17NOS | 283.1029 | −0.54 | 65 | 1 | 10 | 266; 200; 182 |
| M279 | C17H13NOS | 279.0717 | −0.32 | 69 | 0.8 | 12 | 251; 246; 196 |
| M277a | C17H11NOS | 277.056 | −0.40 | 87 | 0.6 | 13 | 250; 194 |
| M277b | C17H11NOS | 277.0559 | −0.76 | 64 | 0.6 | 13 | 263; 220 |
| M266 | C16H10O2S | 266.0401 | −0.09 | 69 | 0.6 | 12 | 266; 250; 71 |
| M197a | C9H11NO2S | 197.0510 | −0.05 | 82 | 1.2 | 5 | 139; 127; 114; 72 |
| M197b | C9H11NO2S | 197.0510 | −0.41 | 72 | 1.2 | 5 | 137; 109; 96 |
| M193 | C9H7NO2S | 193.0198 | 0.04 | 93 | 0.8 | 7 | 166; 84 |
| M185 | C12H11NO | 185.0841 | 0.38 | 67 | 0.9 | 8 | 157; 145; 115; 103 |
| M181b | C9H11NOS | 181.0562 | 0.11 | 60 | 1.2 | 5 | 125; 123; 97; 85 |
| M181a | C9H11NOS | 181.0561 | 0.03 | 78 | 1.2 | 5 | 125; 123; 97 |
| M179 | C9H9NOS | 179.0404 | −0.41 | 89 | 1 | 6 | No |
| M174 | C10H6O3 | 174.03177 | 0.43 | 88 | 0.6 | 8 | 147;129;129;105 |
| M172 | C11H8O2 | 172.0526 | 0.76 | 89 | 0.7 | 8 | 145; 117; 91 |
| M171 | C8H13NOS | 171.0717 | −0.44 | 66 | 1.6 | 3 | No |
| M169b | C8H11NOS | 169.0562 | 0.07 | 52 | 1.4 | 4 | 123; 86; 58 |
| M169a | C8H11NOS | 169.0561 | −0.40 | 56 | 1.4 | 4 | 127; 92; 71 |
| M167 | C8H9NOS | 167.0406 | 0.85 | 89 | 1.1 | 5 | 152; 150; 140 |
| M126b | C6H6OS | 126.0139 | −0.32 | 75 | 1 | 4 | 99; 85 |
| M126a | C6H6OS | 126.0139 | −0.32 | 77 | 1 | 4 | 101; 99; 85 |
| M122b | C7H6S | 122.0189 | −0.80 | 87 | 0.9 | 5 | 97; 79 |
| M122a | C7H6S | 122.0189 | −0.87 | 89 | 0.9 | 5 | 97; 79; 77 |
| M110 | C6H6S | 110.019 | −0.27 | 80 | 1 | 4 | 97; 85; 67 |
| Name | Formula | Molecular Weight | Predicted Toxicity [mg L−1] | |
|---|---|---|---|---|
| Consensus | Nearest Neighbor | |||
| DLX | C18H19NOS | 297.1260 | 2.26 | 9.19 |
| M329 | C18H19NO3S | 329.1084 | 1.14 | 6.73 |
| M325 | C18H15NO3S | 325.0771 | – | 6.49 |
| M323 | C18H13NO3S | 323.0615 | – | 4.78 |
| M321 | C18H11NO3S | 321.0458 | – | 4.75 |
| M313 | C18H19NO2S | 313.1134 | 1.64 | 8.24 |
| M307 | C18H13NO2S | 307.0664 | – | 4.55 |
| M295 | C18H17NOS | 295.1030 | 2.19 | 4.37 |
| M293 | C18H15NOS | 293.0872 | – | 4.34 |
| M283 | C17H17NOS | 283.1029 | 2.41 | 8.76 |
| M279 | C17H13NOS | 279.0717 | – | 4.13 |
| M277 | C17H11NOS | 277.056 | – | 4.10 |
| M266 | C16H10O2S | 266.0401 | – | 10.1 |
| M197 | C9H11NO2S | 197.0510 | 73.3 | 28.5 |
| M193 | C9H7NO2S | 193.0198 | 39.4 | 27.9 |
| M185 | C12H11NO | 185.0841 | – | 99.5 |
| M181 | C9H11NOS | 181.0561 | 51.1 | 26.2 |
| M179 | C9H9NOS | 179.0404 | 31.1 | 25.9 |
| M174 | C10H6O3 | 174.0317 | 5.14 | 0.76 |
| M172 | C11H8O2 | 172.0526 | 2.67 | 0.53 |
| M171 | C8H13NOS | 171.0717 | 55.4 | 24.7 |
| M169 | C8H11NOS | 169.0561 | 51.1 | 24.5 |
| M167 | C8H9NOS | 167.0406 | 52.9 | 24.2 |
| M126 | C6H6OS | 126.0139 | 163 | 135 |
| M122 | C7H6S | 122.0189 | 80.7 | 119 |
| M110 | C6H6S | 110.0190 | 124 | 409 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chojnacka, J.; Giebułtowicz, J.; Choińska, N.; Drobniewska, A.; Nałęcz-Jawecki, G. Do Microplastics Affect the Photodegradation of Duloxetine and Its Phototoxicity to Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835? Water 2023, 15, 3890. https://doi.org/10.3390/w15223890
Chojnacka J, Giebułtowicz J, Choińska N, Drobniewska A, Nałęcz-Jawecki G. Do Microplastics Affect the Photodegradation of Duloxetine and Its Phototoxicity to Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835? Water. 2023; 15(22):3890. https://doi.org/10.3390/w15223890
Chicago/Turabian StyleChojnacka, Justyna, Joanna Giebułtowicz, Natalia Choińska, Agata Drobniewska, and Grzegorz Nałęcz-Jawecki. 2023. "Do Microplastics Affect the Photodegradation of Duloxetine and Its Phototoxicity to Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835?" Water 15, no. 22: 3890. https://doi.org/10.3390/w15223890
APA StyleChojnacka, J., Giebułtowicz, J., Choińska, N., Drobniewska, A., & Nałęcz-Jawecki, G. (2023). Do Microplastics Affect the Photodegradation of Duloxetine and Its Phototoxicity to Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835? Water, 15(22), 3890. https://doi.org/10.3390/w15223890






