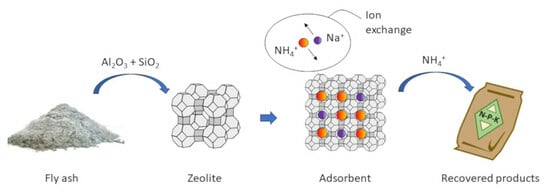
Municipal Solid Waste Fly Ash-Derived Zeolites as Adsorbents for the Recovery of Nutrients and Heavy Metals—A Review
Abstract
:1. Introduction
2. Zeolites
2.1. The Crystalline Structure of Zeolites
- X-ray diffraction (XRD) is used to study the crystal structure and size of zeolite, the approximate extent of heteroatom substitution, and the presence of defects in zeolites [26].
- Transmission electron microscopy (TEM) is used for the characterization of zeolite structures, for instance, structure determination of new zeolites, study of growth mechanisms of nano-sized zeolites and pore structures of hierarchical micro- and meso-porous zeolites, and analysis of metal sites in zeolites [27].
- Fourier transform infrared spectroscopy (FTIR) is used to specify the functional units of zeolites and to predict the reaction mechanisms in the zeolite framework. The FTIR spectrum is also used to indicate the secondary building units that were found in the zeolite structure [26].
- A scanning electron microscope (SEM) is used to study the surface of solids and give information about their morphology and textural properties [26].
2.2. Naturally and Synthesized Zeolites
2.3. Zeolite Synthesis
2.4. MSW-FA as Source to Silicate and Alumina in Zeolite Synthesis
| Element | Unit | Fly Ash/APC Residues | ||
|---|---|---|---|---|
| Min | Max | Median | ||
| Main elements | ||||
| Si | g/kg | 36 | 190 | - |
| Al | g/kg | 6.4 | 93 | - |
| Fe | g/kg | 0.76 | 71 | - |
| Ca | g/kg | 46 | 361 | - |
| Mg | g/kg | 1.1 | 19 | - |
| K | g/kg | 17 | 109 | - |
| Na | g/kg | 6.2 | 84 | - |
| Ti | g/kg | 0.7 | 12 | - |
| S | g/kg | 1.4 | 32 | - |
| Cl | g/kg | 45 | 380 | - |
| P | g/kg | 1.7 | 9.6 | - |
| Mn | g/kg | 0.2 | 1.7 | - |
| TOC | g/kg | 4.9 | 17 | - |
| LOI | g/kg | 11 | 120 | - |
| SiO2 | % | 11.5 | 41.4 | 19.1 |
| Al2O3 | % | 4.7 | 24.3 | 10.9 |
| CaO | % | 17 | 31.5 | 22.0 |
| SO3 | % | 3 | 10.2 | 6.4 |
| Na2O | % | 3.8 | 9.6 | 5.9 |
| K2O | % | 2 | 8.1 | 4.5 |
| Fe2O3 | % | 1.3 | 5.9 | 2.5 |
| MgO | % | 1.7 | 6.9 | 2.7 |
| Minor elements | ||||
| As | mg/kg | 18 | 960 | - |
| Cd | mg/kg | 16 | 1660 | - |
| Cr | mg/kg | 72 | 570 | - |
| Cu | mg/kg | 16 | 2220 | - |
| Hg | mg/kg | 0.1 | 51 | - |
| Ni | mg/kg | 19 | 710 | - |
| Pb | mg/kg | 254 | 27,000 | - |
| Zn | mg/kg | 4308 | 41,000 | - |
2.5. Producing Zeolite-Like Material from MSW Fly Ash
2.5.1. Specific Leaching of Salt as Pre-Treatment
2.5.2. Generating Al- and Si-Containing Zeolite Precursors
2.5.3. Hydrothermal Zeolite Synthesis from MSW-FA
2.5.4. Controlled Acid Leaching and Solidification of Heavy Metals
Controlled Acid Leaching of Heavy Metals
Solidification of Heavy Metals
2.5.5. Destruction of Dioxins and Furans
2.5.6. Production Efficiency and Waste Management Related to Production of Zeolites from MSW-FA
- Continuous-flow synthesis: The use of a tubular reactor in a continuous-flow synthesis of zeolites makes it possible to complete the crystallization in a matter of seconds or minutes due to the large heat transfer coefficient [128]. Liu et al. (2016) [129] synthesized ZSM-5 from a well-mixed and pre-heated precursor solution containing NaOH, pure colloidal silica, aluminium hydroxide (with gibbsite structure), and tetrapropylammonium hydroxide (TPAOH) as structural agents (50 NaOH:Al2O3:300 SiO2:20 TPAOH:2300 H2O). The precursor solution was continuously fed (1 mL/min) into a millimetre-sized (Di 2.18 mm) continuous flow reactor together with pre-heated (370 °C) pressurized water (1.6 mL/min), resulting in complete crystallization within tens of seconds. Because the actual reactor volume was quite small (15.6 mL), the continuous flow process generated a very high space–time yield (ca. 7000 kg/m3h) [129].
- Collecting generated off-gases: Gases such as ammonia and hydrogen are often generated in generous amounts during hydrothermal synthesis. López-Delgado et al. (2020) [130] developed a conceptual design that included the recovery of 76 Nm3 NH3 (from aluminium nitride) and 106 Nm3 H2 per ton of aluminium waste (77% Al2O3 and 4 wt% SiO2) used in the one-step hydrothermal process (10 kg Al waste, 5.3 kg NaOH pellets, 22.9 kg waterglass, and 132 L tap water at 1 bar and 80 °C for 12 h). To avoid gas generation inside the reactor, the aluminium waste was partially hydrolyzed with water and NaOH in a separate compartment.
3. Targeted Sorption of Cations
3.1. Zeolites as Cation Exchange Resins
3.2. Sorption Mechanisms
3.2.1. Adsorption of Heavy Metals
3.2.2. Adsorption of Ammonium
3.3. Factors Affecting the Sorption of Cations
3.3.1. Framework Type vs. Size of the Cation
| Ion | Unhydrated Radius | Hydrated Radius | ΔhydG | Ion | Unhydrated Radius | Hydrated Radius | ΔhydG |
|---|---|---|---|---|---|---|---|
| Å | Å | kJ/mol | Å | Å | kJ/mol | ||
| Li+ | 0.60 | 3.82 | −475 | Cu2+ | 0.72 | 4.19 | −2010 |
| Na+ | 0.95 | 3.58 | −365 | Zn2+ | 0.74 | 4.30 | −1955 |
| K+ | 1.33 | 3.31 | −295 | Cd2+ | 0.97 | 4.26 | −1755 |
| Ca2+ | 0.99 | 4.12 | −1505 | Pb2+ | 1.32 | 4.01 | −1425 |
| NH4+ | 1.48 | 3.31 | −285 | Cr3+ | 0.64 | 4.61 | −4010 |
| NO3− | 2.64 | 3.35 | −300 | Ni2+ | 0.70 | 4.04 | −1980 |
| H2PO4− | - | 2.6 | - | ||||
| PO43− | - | 7.9 1 | −2765 |
| Zeolite | Origin | Si/Al | Selectivity | References |
|---|---|---|---|---|
| Synthetic zeolites | ||||
| FAU-type | Coal FA | 2.5 | Pb2+ > Cu2+ > Cd2+ > Zn2+ > Co2+ | [159] |
| NaP1 | Coal FA | 1.7 | Cr3+ > Cu2+ > Zn2+ > Cd2+ > Ni2+ | [156] |
| 4A | Coal FA | 1.32 | Cu2+ > Cr3+ > Zn2+ > Co2+ > Ni2+ | [155] |
| X | Egyptian kaolin and Na2Si2O5 | 1.15 | Pb2+ > Cd2+ > Cu2+ > Zn2+ > Ni2+ | [160] |
| A | Egyptian kaolin and Na2Si2O5 | 1.04 | Pb2+ > Cd2+ > Cu2+ > Zn2+ > Ni2+ | [160] |
| Natural zeolites | ||||
| Mordenite | Natural | 4.4–5.5 | Cu2+ > Co2+≈Zn2+ > Ni2+ | [161] |
| Clinoptilolite | Natural | 4.9 | Pb2+ > Zn2+ > Cu2+ > Ni2+ | [162] |
| Clinoptilolite | Natural | 4.8 | Cu2+ > Cr3+ > Zn2+ > Cd2+ > Ni2+ | [156] |
| Clinoptilolite | Natural | 4.2 | Pb2+ > Cd2+ > Zn2+≈Cu2+ | [163] |
| Clinoptilolite | Natural | 2.7–5.3 | Pb2+ > Ag+ > Cd2+ ≈ Zn2+ > Cu2+ | [161] |
| Phillipsite | Natural | 2.4–2.7 | Pb2+ > Cd2+ > Zn2+ > Co2+ | [163] |
| Chabazite | Natural | 2.2–2.6 | Pb2+ > Cd2+ > Cu2+ > Zn2+ > Co2+ | [163] |
| Scolecite | Natural | 1.56 | Cu2+ > Zn2+ > Pb2+ > Ni2+ > Co2+ > Co2+ | [164] |
3.3.2. Cation Concentration and Competing Ions
3.3.3. Purity of the Zeolite
3.3.4. Hydrophilicity/Hydrophobicity
3.3.5. Compensation Cations
3.3.6. Available Adsorption Surface and Size of the Zeolite Particles
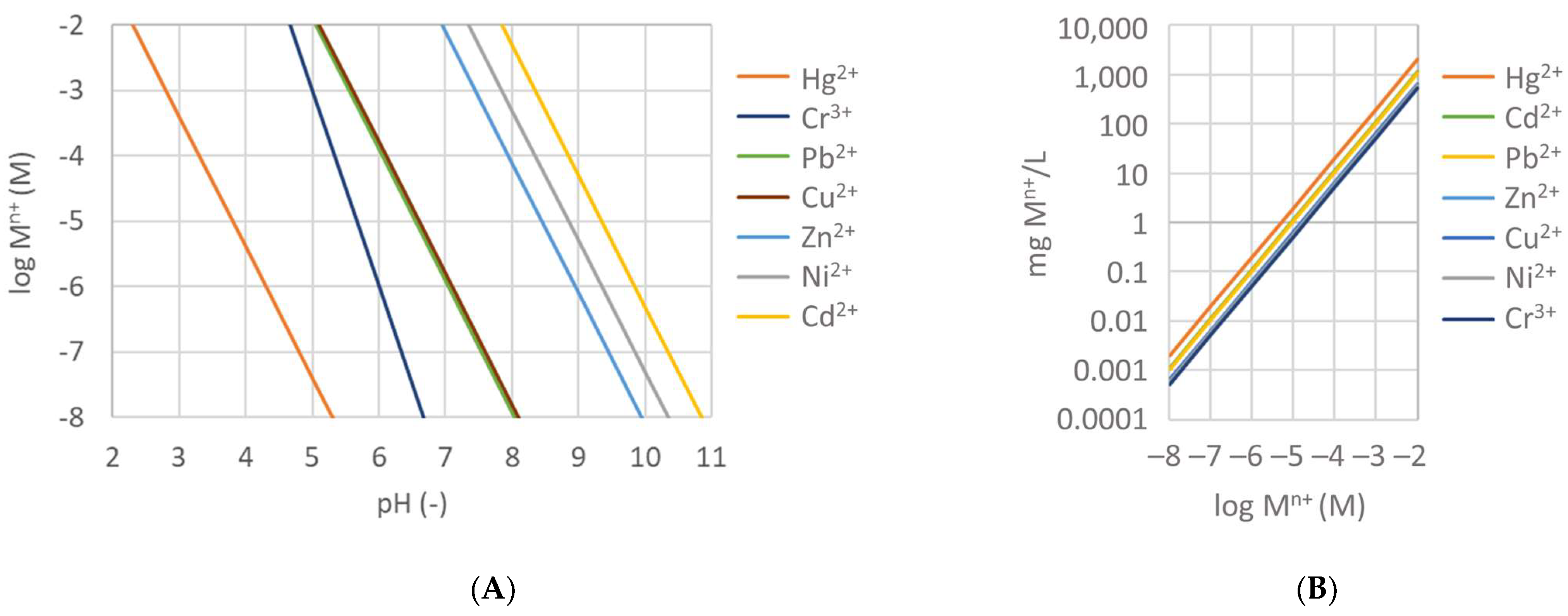
3.3.7. pH
3.3.8. Temperature
3.3.9. Contact Time
4. Sorption of Nitrate and Phosphate Using Zeolites
- Lowering the pH to make the zeolite cationic
- Modifying the surface of the zeolite by cationic metal-doping or using surfactants.
4.1. pH-Derived Cationic Zeolites
| Zeolite | App. Sorption Capacity | Conc. Range | S/L Ratio | Contact Time | Temp. | pH | Ref. |
|---|---|---|---|---|---|---|---|
| mg/g | mg P/L | g/L | h | °C | - | ||
| Non-modified zeolites | |||||||
| NaP1 | 11.4 | 12.5–200 | 1 | 24 | 25 | 5.3 | [182] |
| NaA | 15.7 | ||||||
| Clinoptilolite | 20.2 | ||||||
| A | 52.9 | 50–1000 | 6.6 | 4 | 70 | 5.5 | [186] |
| Clinoptilolite | 1.3 | 10–100 | 48 | 2 | 25 | 2 | [188] |
| Zeolite from coal-FA | 11.7–42.4 | 1000 | 10 | 24 | room | 3.5–9 | [184] |
| Clinoptilolite | 0.77 | 0.03–3.1 | 8 | 24 | room | 3.0 | [189] |
| NaP1-zeolite from coal-FA | 34.7 | 0.5–1000 | 10 | 24 | 18–22 | - | [183] |
| Salt-modified zeolites | |||||||
| LaP1 | 58.2 | 12.5–200 | 1 | 24 | 25 | 5.3 | [182] |
| LaA | 48.9 | ||||||
| La-clinoptilolite | 25.5 | ||||||
| TiO2-modified clinoptilolite | 34.2 | 10–100 | 20 | 2 | 25 | 2 | [188] |
| Ca-bearing K-zeolite | 142–250 | 100–16,000 | 16.7 | 0.8–2.2 | 22 | 6–9 | [190] |
| Zr oxide merlinoite | 67.7 | 5–200 | 0.2–2 | 4 | 40 | <5 | [191] |
| CaP1-zeolite from coal-FA | 49.5 | 0.5–1000 | 10 | 24 | 18–22 | - | [183] |
| MgP1-zeolite from coal-FA | 31.3 | ||||||
| AlP1-zeolite from coal-FA | 29.9 | ||||||
| FeP1-zeolite from coal-FA | 30.9 | ||||||
| Cu-zeolite X | 87.7 | 10–200 | 1 | 24 | 25 | 5.0 | [192] |
| Surfactant-modified zeolites | |||||||
| HDTMA-Br clinoptilolite | 20.9 | 0.03–3.1 | 8 | 24 | room | 12.0 | [189] |
| HDP-Br clinoptilolite | 11.6 | ||||||
4.2. Modification of Zeolites
4.2.1. Metal-Doped Zeolites
4.2.2. Surfactant-Modified Zeolites (SMZs)
4.2.3. Adsorption of Phosphate by Modified Zeolites
Results with Metal-Doped Zeolites
Results with Surfactant-Modified Zeolites
4.2.4. Adsorption of Nitrate by Surfactant-Modified Zeolites
| Zeolite | Surfactant | Amount Adsorbed | Conc. Range | S/L Ratio | Contact Time | Temp. | pH | Ref. |
|---|---|---|---|---|---|---|---|---|
| mg NO3/g | mg NO3/L | g/L | h | °C | - | |||
| Clinoptilolite | polydopamine | 2.47 | 150 | - | 0.30 | 10 | 3 | [204] |
| ZSM-5 nanocrystals | HDTMA-Br | 50 | 50–2500 | 0.5 | 24 | room | 6 | [39] |
| ZSM-5 nanosheets | HDTMA-Br | 120 | ||||||
| ZSM-5 nanosponges | HDTMA-Br | 132 | ||||||
| clinoptilolite-rich turf | HDTMA-Br | 4.96 | 124–1240 | 100 | 24 | room | - | [202] |
| Natural zeolite | HDTMA-Br | 2.42 | 5 | 0.91 | 2 | room | 7 | [205] |
| *BEA-type zeolite nanosponge | HDTMA-Br | 83 | 50–1500 | 2 | 2 min | room | 5.5 | [41] |
| *BEA-type zeolite nanocrystals | HDTMA-Br | 19 | 25 | 5 min | ||||
| Clinoptilolite-rich tuf | HDTMA-Br | 6.07 | 1–113 | 20–200 | 24 | room | 5–6 | [207] |
| Natural zeolite | CPB | 9.68 | 89 | 2 | 0.5 | 15 | 6 | [206] |
4.2.5. Leaching of Surfactants—A Potential Setback
5. Practical Application of Zeolites as Adsorbent
5.1. Production and Use of Shaped and Structured Zeolites
5.2. Physical Separation of Powdered Zeolites—Magnetic Zeolites
6. Reuse of Adsorbed Compounds
- Use them as they are, embedded in zeolite, typically as slow-release compounds, for instance, in fertilizers.
- Recover them from the zeolite by controlled release.
6.1. Slow Release of Compounds from the Zeolite during Application
6.2. Controlled Release of Compounds of Interest
6.2.1. Methods Used to Release the Compounds from Zeolites
| Compound | Zeolite | Release Conditions | Important Factors | Released Compound | Desorption Efficiency | Ref. |
|---|---|---|---|---|---|---|
| Cu2+ | Synthetic from FA | 0.1–0.8 M H2SO4 | High conc. H2SO4 | CuSO4 | 96–102% (four cycles) | [233] |
| Ni2+ | Synthetic from FA | 0.1–0.8 M H2SO4 | High conc. H2SO4 | NiSO4 | 84–98% (four cycles) | [233] |
| Cd2+ | Natural zeolites | 0.1 M HCl (54–80 bed volumes) | - | CdCl2 | 90% first cycle | [234] |
| Zn2+ | Natural zeolites | 0.1 M HCl (6–30 bed volumes) | - | ZnCl2 | 90% first cycle | [234] |
| Cr6+ | HDTMA-modified clinoptilolite-rich tuff | 0.28 M Na2CO3 and 0.5 M NaOH (L/S: 3 mL/g); regeneration with 3 × 0.1 M HCl (L/S: 3 mL/g) | - | - | 90% first cycle (100% regeneration) | [213] |
| NH4+ | Alkali-treated clinoptilolite | 0.5 M HCl | - | NH4Cl | Adsorption unaffected after 12 cycles | [235] |
| NH4+ | Zeolite from FA | 1 M NaCl (3 × 25 mL/2 g zeolite) at 25 °C for 1.25 h | - | NH4Cl | Ca. 10% loss in adsorbent capacity after one cycle | [236] |
| NH4+ | Clinoptilolite | 20 g NaCl/L for 15 h | High NaCl conc. | NH4Cl | 100% (five cycles). Adsorption capacity increased from 9.2 mg/g to 10.9 mg/g (over first three cycles) | [237] |
| NH4+ | Clinoptilolite | 30 g NaCl/L (123–134 BV) | Low flow rate to get high conc. | NH4Cl | 88–95% | [231] |
| NH4+ | Synthetic NaA | 30 g NaCl/L (43–46 BV) | 92–95% | |||
| NH4+ | Clinoptilolite | 10% NaCl and 0.6% NaOH | Increased desorption: 10–15% NaCl and 0–0.6% NaOH | NH3 | 100% | [229] |
| PO43− | La-doped zeolite from FA | 3 M NaOH (L/S ratio 80:1) at 250 °C for 5 h | High conc. NaOH (<4 M NaOH), high L/S ratio, high temp | Na3PO4 | 95% (five cycles) | [154] |
| NO3− | Polydopamin-coated clinoptilolite | 0.01 M and 0.05 M NaOH | - | NaNO3 | 59–71% (three cycles) | [204] |
| NO3− | HDTMA-modified clinoptilolite | 1 M NaBr (L/S: 5 mL/g) for 6 h | - | NaNO3 | Ca. 100% first cycle | [208] |
6.2.2. Downstream Concentration and Refinement
Concentrating Ammonia by Stripping and Condensation
Concentrating by Precipitation
6.2.3. Regeneration of the Zeolite’s Adsorption Capacity
7. Discussion and Need for Further Studies
7.1. MSW-FA as a Source for Synthetic Zeolites
7.2. Capturing Efficiency
7.3. Acceptance and Need of Recovered End-Products
8. Concluding Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hoornweg, D.; Bhada-Tata, P. What a Waste: A Global Review of Solid Waste Management; The World Bank: Washington, DC, USA, 2012. [Google Scholar]
- Kaza, S.; Yao, L.; Bhada-Tata, P.; Van Woerden, F. What a Waste 2.0: A Global Snapshot of Solid Waste Management to 2050; Urban Development: Washington, DC, USA, 2018. [Google Scholar]
- Chen, D.; Zhang, Y.; Xu, Y.; Nie, Q.; Yang, Z.; Shenga, W.; Qian, G. Municipal solid waste incineration residues recycled for typical construction materials—A review. RSC Adv. 2022, 12, 6279. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, D.; Molin, C.; Hupa, M. Thermal treatment of solid residues from WtE units: A review. Waste Manag. 2015, 37, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Quina, M.J.; Bontempi, E.; Bogush, A.; Schlumberger, S.; Weibel, G.; Braga, R.; Funari, V.; Hyks, J.; Rasumssen, E.; Lederer, J. Technologies for the management of MSW incineration ashes from gas cleaning: New perspectives on recovery of secondary raw materials and circular economy. Sci. Total Environ. 2018, 635, 526–542. [Google Scholar] [CrossRef]
- Weibel, G.; Zappatini, A.; Wolffers, M.; Ringmann, S. Optimization of metal recovery from MSWI fly ash by acid leaching: Findings from laboratory- and industrial-scale experiments. Processes 2021, 9, 352. [Google Scholar] [CrossRef]
- Kanhar, A.H.; Chen, S.; Wang, F. Incineration fly ash and its treatment to possible utilization: A Review. Energies 2020, 13, 6681. [Google Scholar] [CrossRef]
- Millini, R.; Bellussi, G. Zeolite science and perspectives. In Zeolites in Catalysis: Properties and Applications; Cejka, J., Morris, R.E., Nachtigall, P., Eds.; RSC Catalysis Series No. 28; The Royal Society of Chemistry: London, UK, 2017. [Google Scholar]
- Ray, R.L.; Sheppard, R.A. Occurrence of zeolites in sedimentary rocks: An overview, in Natural Zeolites: Occurrence, Properties, Applications. Rev. Miner. Geochem. 2001, 45, 17–34. [Google Scholar]
- MgBemere, H.E.; Ekpe, I.C.; Lawal, G.I. Zeolite synthesis, characterizations, and application areas—A review. Int. Res. J. Environ. Sci. 2017, 6, 45–59. [Google Scholar]
- Khaleque, A.; Alam, M.M.; Hoque, M.; Mondal, S.; Haider, J.B.; Xuc, B.; Johir, M.A.H.; Karmakar, A.K.; Zhoud, J.L.; Ahmedb, M.B.; et al. Zeolite synthesis from low-cost materials and environmental applications: A review. Environ. Adv. 2020, 2, 100019. [Google Scholar] [CrossRef]
- Cho, B.H.; Nam, B.H.; An, J.; Youn, H. Municipal Solid Waste Incineration (MSWI) Ashes as Construction Materials—A Review. Materials 2020, 13, 3143. [Google Scholar] [CrossRef]
- Perego, C.; Bagatin, R.; Tagliabue, M.; Vignola, R. Zeolites and related mesoporous materials for multi-talented environmental solutions. Microp. Mesop. Mater. 2013, 166, 37–49. [Google Scholar] [CrossRef]
- Karlfeldt Fedje, K.; Rauch, S.; Cho, P.; Steenari, B.M. Element associations in ash from waste combustion in fluidized bed. Waste Manag. 2010, 30, 1273–1279. [Google Scholar] [CrossRef] [PubMed]
- Witek-Krowiak, A.; Gorazda, K.; Szopa, D.; Trzaska, K.; Moustakas, K.; Chojnacka, K. Phosphorus recovery from wastewater and bio-based waste: An overview. Bioeng 2022, 13, 13474–13506. [Google Scholar] [CrossRef]
- Cordell, D. Peak phosphorous and the role of P recovery in achieving food security. In Source Separation and Decentralization for Wastewater Management; Larsen, T.A., Udert, K.M., Lienert, J., Eds.; IWA Publishing: London, UK, 2013; pp. 29–44. [Google Scholar]
- Jenssen, T.K.; Kongshaug, G. Energy Consumption and Greenhouse Gas Emissions in Fertiliser Production; Proceeding, No. 509; The International Fertilizer Society: Paris, France, 2003. [Google Scholar]
- European Commission Proposal for a Directive of the European Parliament and of the Council Concerning urban Wastewater Treatment (Recast) 541. 2022. Available online: https://environment.ec.europa.eu/publications/proposal-revised-urban-wastewater-treatment-directive_en (accessed on 25 September 2023).
- van Eekert, M.; Weijma, J.; Verdoes, N.; de Buisonjé, F.; Reitsma, B.; van den Bulk, J.; van Gastel, J. Explorative Research on Innovative Nitrogen Recovery; STOWA Report 51; Stichting Toegepast Onderzoek Waterbeheer: Amersfoort, The Netherlands, 2012. [Google Scholar]
- Ganrot, Z. Use of zeolites for improved nutrient recovery from decentralized domestic wastewater. In Handbook of Natural Zeolites; Vassilis, J., Antonis, A.Z., Eds.; Bentham Science Publishers: Singapore, 2012; Chapter 17; pp. 410–435. [Google Scholar]
- Bandala, E.R.; Liu, A.; Wijesiri, B.; Zeidman, A.B.; Goonetilleke, A. Emerging materials and technologies for landfill leachate treatment: A critical review. Environ. Pollut. 2021, 291, 118133. [Google Scholar] [CrossRef] [PubMed]
- Ferrel-Luna, R.; García-Arreola, M.E.; González-Rodríguez, L.M.; Oredo-Cancino, M.; Escárcega-González, C.E.; de Haro-Del Río, D.A. Reducing toxic element leaching in mine tailings with natural zeolite clinoptilolite. Environ. Sci. Pollut. Res. 2023. [Google Scholar] [CrossRef] [PubMed]
- Helfferich, F. Ion Exchange; Dover Publishing: New York, NY, USA, 1995. [Google Scholar]
- Inglezakis, V.J. The concept of “capacity” in zeolite ion-exchange systems. J. Colloid. Inter. Sci. 2005, 281, 68–79. [Google Scholar] [CrossRef]
- Coombs, D.S.; Alberti, A.; Armbruster, T.; Artioli, G.; Colella, C.; Galli, E.; Grice, J.D.; Liebau, F.; Mandarino, J.A.; Minato, H.; et al. Recommended nomenclature for zeolite minerals: Report of the subcommittee on zeolites of the international mineralogical association, commission on new minerals and mineral names. Can. Mineral. 1997, 35, 1571–1606. [Google Scholar]
- Derbe, T.; Temesgen, S.; Bitew, M. A Short Review on Synthesis, Characterization, and Applications of Zeolites. Hindawi Adv. Mater. Sci. Eng. 2021, 2021, 6637898. [Google Scholar] [CrossRef]
- Wan, W.; Su, J.; Zou, X.D.; Wilhammar, T. Transmission electron microscopy as an important tool for characterization of zeolite structures. Inorg. Chem. Front. 2018, 5, 2836. [Google Scholar] [CrossRef]
- Tsai, Y.T.; Huang, E.; Li, Y.; Hung, H.; Jiang, J.; Liu, T.; Fang, J.; Chen, H. Raman Spectroscopic Characteristics of Zeolite Group Minerals. Minerals 2021, 11, 167. [Google Scholar] [CrossRef]
- Wang, T.; Luo, S.; Tompsett, G.A.; Timko, M.T.; Fan, W.; Auerbach, S.M. Critical Role of Tricyclic Bridges Including Neighboring Rings for Understanding Raman Spectra of Zeolites. J. Am. Chem. Soc. 2019, 141, 20318–20324. [Google Scholar] [CrossRef]
- van Vreeswijk, S.H.; Weckhuysen, B.M. Emerging analytical methods to characterize zeolite-based materials. Natl. Sci. Rev. 2022, 9, nwac047. [Google Scholar] [CrossRef]
- Barelocher, C.; McCuster, L.B.; Olson, H.D. Atlas of Zeolite Framework Types, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Yu, J. Synthesis of zeolites. In Introduction to Zeolite Science and Practice, 3rd ed.; Čejka, J., Hv, B., Corma, A., Schüth, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; Volume 168, Chapter 3. [Google Scholar]
- Brännvall, E.; Kumpiene, J. Fly ash in landfill top covers—A review. Environ. Sci. Proc. Impacts 2016, 18, 11–21. [Google Scholar] [CrossRef]
- Cundy, C.S.; Cox, P.A. The hydrothermal synthesis of zeolites: Precursors, intermediates and reaction mechanism. Microp. Mesop. Mater. 2005, 82, 1–78. [Google Scholar] [CrossRef]
- Na, K.; Choi, M.; Ryoo, R. Recent advances in the synthesis of hierarchically nanoporous zeolites. Microp. Mesop. Mater. 2013, 166, 3–19. [Google Scholar] [CrossRef]
- Gao, M.; Ma, Q.; Lin, Q.; Chang, J.; Bao, W.; Ma, M. Combined modification of fly ash with Ca(OH)2/Na2FeO4 and its adsorption of Methyl orange. App. Surf. Sci. 2015, 359, 323–330. [Google Scholar] [CrossRef]
- Perez-Ramırez, J.; Verboekend, D.; Bonilla, A.; Abello, S. Zeolite catalysts with tunable hierarchy factor by pore-growth moderators. Adv. Funct. Mater 2009, 19, 3972. [Google Scholar] [CrossRef]
- Ivanova, I.I.; Knyazeva, E.E. Micro–mesoporous materials obtained by zeolite recrystallization: Synthesis, characterization and catalytic applications. Chem. Soc. Rev. 2013, 42, 3671–3688. [Google Scholar] [CrossRef]
- Hanache, L.E.; Sundermann, L.; Lebeau, B.; Toufaily, J.; Hamieh, T.; Daou, T.J. Surfactant-modified MFI-type nanozeolites: Super-adsorbents for nitrate removal from contaminated water. Microp. Mesop. Mater. 2019, 283, 1–13. [Google Scholar] [CrossRef]
- Schick, J.; Daou, T.J.; Caullet, P.; Paillaud, J.L.; Patarin, J.; Mangold-Callarec, C. Surfactant-modified MFI nanosheets: A high capacity anion-exchanger. Chem. Comm. 2011, 47, 902–904. [Google Scholar] [CrossRef]
- Hanache, L.E.; Lebeau, B.; Nouali, H.; Toufaily, J.; Hamieh, T.; Daou, T.J. Performance of surfactant-modified *BEA-type zeolite nanosponges for the removal of nitrate in contaminated water: Effect of the external surface. J. Hazard. Mater. 2019, 364, 206–217. [Google Scholar] [CrossRef]
- Mao, Y.; Wu, H.; Wang, W.; Jia, M.; Che, X. Pretreatment of municipal solid waste incineration fly ash and preparation of solid waste source sulphoaluminate cementitious material. J. Hazard. Mater. 2020, 385, 121580. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Cui, R.; Yang, T.; Zhai, Z.; Li, R. Distribution characteristics of heavy metals in different size fly ash from a sewage sludge circulating fluidized bed incinerator. Energy Fuels 2017, 31, 2044–2051. [Google Scholar] [CrossRef]
- Fabricius, A.-L.; Renner, M.; Voss, M.; Funk, M.; Perfoll, A.; Gehring, F.; Graf, R.; Fromm, S.; Duester, L. Municipal waste incineration fly ashes: From a multi-element approach to market potential evaluation. Environ. Sci. Eur. 2020, 32, 88. [Google Scholar] [CrossRef] [PubMed]
- Bodénan, F.; Deniard, P. Characterization of flue gas cleaning residues from European solid waste incinerators: Assessment of various Ca-based sorbent processes. Chemosphere 2003, 51, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Chiang, K.-Y.; Jih, J.-C.; Chien, M.-D. The acid extraction of metals from municipal solid waste incinerator products. Hydrometallurgy 2008, 93, 16–22. [Google Scholar] [CrossRef]
- Zhu, J.; Hao, Q.; Chen, J.; Hu, M.; Tu, T.; Jiang, C. Distribution characteristics and comparison of chemical stabilization ways of heavy metals from MSW incineration fly ashes. Waste Manag. 2020, 113, 488–496. [Google Scholar] [CrossRef]
- Saakshy, K.; Singh, A.B.; Gupta Sharma, A.K. Fly ash as low cost adsorbent for treatment of effluent of handmade paper industry-Kinetic and modelling studies for direct black dye. J. Clean. Prod. 2016, 112, 1227–1240. [Google Scholar] [CrossRef]
- Wesche, K. Fly Ash in Concrete Properties and Performance; RILEM Report 7; International Union of Testing and Research Laboratories: France; London, UK, 1991; p. 356. [Google Scholar]
- Chen, Z.; Lu, S.; Tang, M.; Ding, J.; Buekens, A.; Yang, J.; Qiu, Q.; Yan, J. Mechanical activation of fly ash from MSWI for utilization in cementitious materials. Waste Manag. 2019, 88, 182–190. [Google Scholar] [CrossRef]
- Fan, X.F.; Yuan, R.; Gan, M.; Ji, Z.; Sun, Z. Subcritical hydrothermal treatment of municipal solid waste incineration fly ash: A review. Sci. Total Environ. 2023, 865, 160745. [Google Scholar] [CrossRef]
- Kim, S.Y.; Tanaka, N.; Matsuto, T.; Tojo, Y. Leaching behaviour of elements and evaluation of pre-treatment methods for municipal solid waste incinerator residues in column leaching tests. Waste Manag. Res. 2005, 23, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Kasina, M.; Kowalski, P.R.; Kajdas, B.; Michalik, M. Assessment of valuable and critical elements recovery potential in ashes from processes of solid municipal waste and sewage sludge thermal treatment. Resources 2020, 9, 131. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, F.-S.; Zhu, J.; Liu, Z. Effective utilization of waste ash from MSW and coal co-combustion power plant—Zeolite synthesis. J. Hazard. Mater. 2008, 153, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Ohbuchi, A.; Koike, Y.; Nakamura, T. Crystal morphology analysis for heavy elements in municipal solid waste incineration fly ash and bottom ash by X-ray characterization techniques. Anal. Sci. 2020, 6, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Siddique, R. Utilization of municipal solid waste (MSW) ash in cement and mortar. Resour. Conserv. Recycl. 2010, 54, 1037–1047. [Google Scholar] [CrossRef]
- Kowalski, P.R.; Kasina, M.; Michalik, M. Metallic elements fractionation in municipal solid waste incineration residues. Energy Procedia 2016, 97, 31–36. [Google Scholar] [CrossRef]
- Weibel, G.; Eggenberger, U.; Schlumberger, S.; Mäder, U.K. Chemical associations and mobilization of heavy metals in fly ash from municipal solid waste incineration. Waste Manag. 2017, 62, 147–159. [Google Scholar] [CrossRef]
- Rani, D.A.; Boccaccini, A.R.; Deegan, D.; Cheeseman, C.R. Air pollution control residues from waste incineration: Current UK situation and assessment of alternative technologies. Waste Manag. 2008, 28, 2279–2292. [Google Scholar] [CrossRef] [PubMed]
- Saikia, N.; Kato, S.; Kojima, T. Production of cement clinkers from municipal solid waste incineration (MSWI) fly ash. Waste Manag. 2007, 27, 1178–1189. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Ye, T.; Guo, Y.; Gao, X. A study on the chemical and mineralogical characterization of MSWI fly ash using a sequential extraction procedure. J. Hazard. Mater. 2006, 134, 197–201. [Google Scholar] [CrossRef]
- IAWG (International Ash Working Group); Chandler, A.J.; Eighmy, T.T.; Hartlén, J.; Hjelmar, O.; Kosson, D.S.; Sawell, S.E.; van der Sloot, H.; Vehlow, J. Municipal Solid Waste Incinerator Residues; Studies in Environmental Science; Elsevier: Amsterdam, The Netherlands, 1997; Volume 67. [Google Scholar]
- Zhao, Y.; Li, H. Understanding municipal solid waste production and diversion factors utilizing deep-learning methods. Util. Policy 2023, 83, 101612. [Google Scholar] [CrossRef]
- Joseph, A.M.; Snellings, R.; van den Heede, P.; Matthys, S.; de Belie, N. The use of municipal solid waste incineration ash in various building materials: A Belgian Point of View. Materials 2018, 11, 141. [Google Scholar] [CrossRef]
- Deng, L.; Xu, Q.; Wu, H. Synthesis of zeolite-like material by hydrothermal and fusion methods using municipal solid waste fly ash. Proc. Environ. Sci. 2016, 31, 662–667. [Google Scholar] [CrossRef]
- Eighmy, T.T.; Eusden, J.D.; Krzanowski, J.E.; Domiago, D.S.; Stampfli, D.; Martin, P.M.; Erickson, P.M. Comprehensive approach toward understanding element speciation and leaching behavior in municipal solid waste incineration electrostatic precipitator ash. Environ. Sci. Technol. 1995, 29, 629–646. [Google Scholar] [CrossRef]
- Hjelmar, O. Disposal strategies for municipal solid waste incineration residues. J. Hazard. Mater. 1996, 47, 345–368. [Google Scholar] [CrossRef]
- Forestier, L.L.; Libourel, G. Characterization of flue gas residues from municipal solid waste combustors. Environ. Sci. Technol. 1998, 32, 2250–2256. [Google Scholar] [CrossRef]
- Quina, M.J.; Bordado, J.C.; Quinta-Ferreira, R.M. Treatment and use of air pollution control residues from MSW incineration: An overview. Waste Manag. 2008, 28, 2097–2121. [Google Scholar] [CrossRef] [PubMed]
- Song, G.J.; Kim, K.; Seo, Y.; Kim, S. Characteristics of ashes from different locations at the MSW incinerator equipped with various air pollution control devices. Waste Manag. 2004, 24, 99–106. [Google Scholar] [CrossRef]
- Vavva, C.; Voutsas, E.; Magoulas, K. Process development for chemical stabilization of fly ash from municipal solid waste incineration. Chem. Eng. Res. Des. 2017, 125, 57–71. [Google Scholar] [CrossRef]
- Li, X.; Chen, Q.; Zhou, Y.; Tyrer, M.; Yu, Y. Stabilization of heavy metals in MSWI fly ash using silica fume. Waste Manag. 2014, 34, 2494–2504. [Google Scholar] [CrossRef]
- Karlfeldt, K.; Steenari, B.-M. Assessment of metal mobility in MSW incineration ashes using water as the reagent. Fuel 2007, 86, 1983–1993. [Google Scholar] [CrossRef]
- Alba, N.; Gasso, S.; Lacorte, T.; Baldasano, J.M. Characterization of municipal solid waste incineration residues from facilities with different air pollution control systems. J. Air Waste Manag. Assoc. 1997, 47, 1170–1179. [Google Scholar] [CrossRef]
- Romero, M.; Rincon, J.M.; Rawlings, R.D.; Boccaccini, A.R. Use of vitrified urban incinerator waste as raw material for production of sintered glass–ceramics. Mat. Res. Bull. 2001, 36, 383–395. [Google Scholar] [CrossRef]
- Cheng, T.W.; Chen, Y.S. Characterisation of glass–ceramics made from incinerator fly ash. Ceram. Intern. 2004, 30, 343–349. [Google Scholar] [CrossRef]
- Miyake, M.; Tamura, C.; Matsuda, M. Resource recovery of waste incineration fly ash: Synthesis of zeolites a and p. J. Am. Ceram. Soc. 2002, 85, 1873–1875. [Google Scholar] [CrossRef]
- Yang, G.C.C.; Yang, T. Synthesis of zeolites from municipal incinerator fly ash. J. Hazard. Mater. 1998, 62, 75–89. [Google Scholar] [CrossRef]
- Bac, B.H.; Song, Y.; Moon, Y.; Kim, M.H.; Kang, I.M. Effective utilization of incinerated municipal solid waste incineration ash: Zeolitic material synthesis and silica extraction. Waste Manag. Res. 2010, 28, 714–722. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Chen, J.-C. Resourcization and valorization of waste incineration fly ash for the synthesis of zeolite and applications. J. Environ. Chem. Eng. 2021, 9, 106549. [Google Scholar] [CrossRef]
- Qiu, Q.; Jiang, X.; Lv, G.; Lu, S.; Ni, M. Stabilization of heavy metals in municipal solid waste incineration fly ash in circulating fluidized bed by microwave-assisted hydrothermal treatment with additives. Energy Fuels 2016, 30, 7588–7595. [Google Scholar] [CrossRef]
- Qiu, Q.; Jiang, X.; Chen, Z.; Lu, S.; Ni, M. Microwave-assisted hydrothermal treatment with soluble phosphate added for heavy metals solidification in MSWI fly ash. Energy Fuels 2017, 31, 5222–5232. [Google Scholar] [CrossRef]
- Chen, Q.; Long, L.; Liu, X.; Jiang, X.; Chi, Y.; Yan, J.; Zhao, X.; Kong, L. Low-toxic zeolite fabricated from municipal solid waste incineration fly ash via microwave-assisted hydrothermal process with fusion pretreatment. J. Mater. Cycles Waste Manag. 2020, 22, 1196–1207. [Google Scholar] [CrossRef]
- Wang, K.-S.; Chiang, K.-Y.; Lin, K.-L.; Sun, C.-J. Effects of a water-extraction process on heavy metal behavior in municipal solid waste incinerator fly ash. Hydrometallurgy 2001, 62, 73–81. [Google Scholar] [CrossRef]
- Chuai, X.; Yang, Q.; Zhang, T.; Zhao, Y.; Wang, J.; Zhao, G.; Cui, X.; Zhang, Y.; Zhang, T.; Xiong, Z.; et al. Speciation and leaching characteristics of heavy metals from municipal solid waste incineration fly ash. Fuel 2022, 328, 125338. [Google Scholar] [CrossRef]
- Todorovic, J.; Ecke, H.; Lagerkvist, A. Solidification with water as a treatment method for air pollution control residues. Waste Manag. 2003, 23, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Glasser, F.P. Fundamental aspects of cement solidification and stabilisation. J. Hazard. Mater. 1997, 52, 151–170. [Google Scholar] [CrossRef]
- Kiyaki, T.; Matsuda, M.; Wajima, T.; Matsui, T. Synthesis of zeolite from municipal solid waste incineration fly ash. Waste Manag. 2002, 22, 893–899. [Google Scholar] [CrossRef]
- Shoppert, A.A.; Loginova, I.V.; Chaikin, L.I.; Rogozhnikov, D.A. Alkali fusion leaching method for comprehensive processing of fly ash. KnE Mater. Sci. 2017, 2, 89–96. [Google Scholar] [CrossRef]
- Verboekend, D.; Pérez-Ramiréz, J. Design of hierarchical zeolitecatalysts by desilication. Catal. Sci. Technol. 2011, 1, 879–890. [Google Scholar] [CrossRef]
- Sayehi, M.; Tounsi, H.; Garbarino, G.; Riani, P.; Busca, G. Reutilization of silicon- and aluminum- containing wastes in the perspective of the preparation of SiO2-Al2O3 based porous materials for adsorbents and catalysts. Waste Manag. 2020, 103, 146–158. [Google Scholar] [CrossRef]
- Boukerche, I.; Djerada, S.; Benmansoura, L.; Tifoutia, L.; Saleh, K. Degradability of aluminum in acidic and alkaline solutions. Corros. Sci. 2014, 78, 343–352. [Google Scholar] [CrossRef]
- Lenntech. Iron Removal by Physical-Chemical Ways. Available online: https://www.lenntech.com/processes/iron-manganese/iron/iron-removal-physicalchemical-way.htm (accessed on 25 October 2023).
- Faust, S.D.; Aly, O.M. Chemistry of Water Treatment; CRC Press: Boca Raton, FL, USA, 1988; p. 224. [Google Scholar]
- Okada, K.; Arimitsu, N.; Kameshima, Y.; Nakajima, A.; MacKenzie, K.J.D. Preparation of porous silica from chlorite by selective acid leaching. Appl. Clay Sci. 2005, 30, 116–124. [Google Scholar] [CrossRef]
- Temuujin, J.; Okada, K.; MacKenzie, K.J.D. Preparation of porous silica from vermiculite by selective leaching. Appl. Clay Sci. 2003, 22, 187–195. [Google Scholar] [CrossRef]
- Adans, Y.F.; Martins, A.R.; Coelho, R.E.; das Virgens, C.F.; Ballarini, A.D.; Carvalho, L.S. A simple way to produce c-alumina from aluminum cans by precipitation reactions. Mat. Res. 2016, 19, 977–982. [Google Scholar] [CrossRef]
- Zong, Y.Z.; Chen, W.; Liu, Z. Extraction of alumina from high-alumina coal ash using an alkaline hydrothermal method. SN Appl. Sci. 2019, 1, 783. [Google Scholar] [CrossRef]
- Zhao, X.; Zeng, L.; Guo, J.; Zhu, Q.; Huang, Z.; Lin, L.; Chen, X.; Cao, J.; Zhou, Z. Efficient separation and comprehensive extraction of aluminum, silicon, and iron from coal fly ash by a cascade extraction method. J. Clean. Prod. 2023, 406, 137090. [Google Scholar] [CrossRef]
- Zhu, P.; Dai, H.; Han, L.; Xu, X.; Cheng, L.; Wang, Q.; Shi, Z. Aluminum extraction from coal ash by a two-step acid leaching method. J Zhejiang Univ.-Sci. A (Appl. Phys. Eng.) 2015, 16, 161–169. [Google Scholar] [CrossRef]
- Shoppert, A.; Veleev, D.; Loginova, I.; Chaikin, L. Complete extraction of amorphous aluminosilicate from coal fly ash by alkali leaching under atmospheric pressure. Metals 2020, 10, 1684. [Google Scholar] [CrossRef]
- Kumar, A.; Agrawal, S.; Dhawan, N. Processing of coal fly ash for the extraction of alumina values. J. Sustain. Metall. 2020, 6, 294–306. [Google Scholar] [CrossRef]
- Wu, C.; Yu, H.; Zhang, H. Extraction of aluminum by pressure acid-leaching method from coal fly ash. Trans. Nonferrous Met. Soc. China 2012, 22, 2282–2288. [Google Scholar] [CrossRef]
- Zhang, B.H.; Wu, D.Y.; Wang, C.; He, S.B.; Zhang, Z.J.; Kong, H.N. Simultaneous removal of ammonium and phosphate by zeolite synthesized from coal fly ash as influenced by acid treatment. J. Environ. Sci. 2007, 19, 540–554. [Google Scholar] [CrossRef]
- Wajima, T.; Ikegami, Y. Synthesis of zeolitic materials from waste porcelain at low temperature via a two-step alkali conversion. Ceram. Int. 2007, 33, 1269–1274. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Weng, C.-H.; Chen, C.-H. Synthesis of zeolite P from coal fly ash and its application in the removal of ammonium and phosphate ions. Environ. Sci. Pollut. Res. 2018, 25, 8339–8350. [Google Scholar]
- Jin, Y.; Ma, X.; Jiang, X.; Liu, H.; Li, X.; Yan, J.; Cen, K. Effects of hydrothermal treatment on the major heavy metals in fly ash from municipal solid waste incineration. Energy Fuels 2013, 27, 394–400. [Google Scholar] [CrossRef]
- Mossop, K.F.; Davidson, C.M. Comparison of original and modified BCR sequential extraction procedures for the fractionation of copper, iron, lead, manganese and zinc in soils and sediments. Anal. Chim. Acta 2003, 478, 111–118. [Google Scholar] [CrossRef]
- Bayuseno, A.P.; Schmahl, W.W.; Muellejans, T. Hydrothermal processing of MSWI fly ash-towards new stable minerals and fixation of heavy metals. J. Hazard. Mater. 2009, 167, 250–259. [Google Scholar] [CrossRef]
- Weibel, G.; Eggenberger, U.; Dmitrii, A.K.; Hummel, W.; Schlumberger, S.; Klink, W.; Fisch, M.; Mäder, U.K. Extraction of heavy metals from MSWI fly ash using hydrochloric acid and sodium chloride solution. Waste Manag. 2018, 76, 457–471. [Google Scholar] [CrossRef] [PubMed]
- AWEL. Stand der Technik für die Aufbereitung von Rauchgasreinigungsrückständen (RGRR) aus Kehrichtverbrennungsanlagen. Amt für Abfall, Wasser, Energie und Luft Abteilung Abfallwirtschaft und Betriebe Walchetor; Baudirektion Kanton Zürich: Zurich, Switzerland, 2013; p. 24. [Google Scholar]
- Vassilev, S.V.; Vassileva, C.G. A new approach for the classification of coal fly ashes based on their origin, composition, properties, and behaviour. Fuel 2007, 86, 1490–1512. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Chen, J.-C. The optimum operating conditions for the synthesis of zeolite from waste incineration fly ash by alkali fusion and hydrothermal methods. Int. Sch. Sci. Res. Innov. 2018, 12, 669–675. [Google Scholar]
- Xie, J.; Hu, Y.; Chen, D.; Zhou, B. Hydrothermal treatment of MSWI fly ash for simultaneous dioxins decomposition and heavy metal stabilization. Front. Environ. Sci. Eng. China 2010, 4, 108–115. [Google Scholar] [CrossRef]
- Shi, D.; Hu, C.; Zhang, J.; Li, P.; Zhang, C.; Wang, X.; Ma, H. Silicon-aluminum additives assisted hydrothermal process for stabilization of heavy metals in fly ash from MSW incineration. Fuel Process. Technol. 2017, 165, 44–53. [Google Scholar] [CrossRef]
- Qiu, Q.; Jiang, X.; Lv, G.; Lu, S.; Ni, M. Effects of microwave-assisted hydrothermal treatment on the major heavy metals of municipal solid waste incineration fly ash in a circulating fluidized bed. Energy Fuels 2016, 30, 5945–5952. [Google Scholar] [CrossRef]
- Tong, H.; Shi, D.; Cai, H.; Liu, J.; Lv, M.; Gu, L.; Luo, L.; Wang, B. Novel hydroxyapatite (HAP)-assisted hydrothermal solidification of heavy metals in fly ash from MSW incineration: Effect of HAP liquid-precursor and HAP seed crystal derived from eggshell waste. Fuel Process. Technol. 2022, 236, 107400. [Google Scholar] [CrossRef]
- Xu, Z.; Liang, Z.; Shao, H.; Zhao, Q. Heavy metal stabilization in MSWI fly ash using an additive-assisted microwave hydrothermal method. J. Ind. Eng. Chem. 2023, 117, 352–360. [Google Scholar] [CrossRef]
- Kulkarni, P.S.; Crespo, J.G.; Afonso, C.A.M. Dioxins sources and current remediation technologies—A review. Environ. Int. 2008, 34, 139–153. [Google Scholar] [CrossRef]
- Xue, Y.; Liu, X. Detoxification, solidification and recycling of municipal solid waste incineration fly ash: A review. Chem. Eng. J. 2021, 420, 130349. [Google Scholar] [CrossRef]
- Hwang, I.-H.; Aoyama, H.; Matsuto, T.; Nakagishi, T.; Matsuo, T. Recovery of solid fuel from municipal solid waste by hydrothermal treatment using subcritical water. Waste Manag. 2012, 32, 410–416. [Google Scholar] [CrossRef]
- Toor, S.S.; Rosendahl, L.; Rudolf, A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy 2011, 36, 2328–2342. [Google Scholar] [CrossRef]
- Jin, Y.-Q.; Ma, X.-J.; Jiang, X.-G.; Liu, H.-M.; Li, X.-D.; Yan, J.-H. Hydrothermal degradation of polychlorinated dibenzop-dioxins and polychlorinated dibenzofurans in fly ash from municipal solid waste incineration under non-oxidative and oxidative conditions. Energy Fuel 2013, 27, 414–420. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Shibuya, E.; Kanamaru, Y.; Uyama, K.; Nishioka, M.; Yamasaki, N. Hydrothermal decomposition of PCDDs/PCDFs in MSWI fly ash. Chemosphere 1996, 32, 203–208. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, P.; Chen, D.; Zhou, B.; Li, J.; Li, X.-W. Hydrothermal treatment of municipal solid waste incineration fly ash for dioxin decomposition. J. Hazard. Mater. 2012, 207, 79–85. [Google Scholar] [CrossRef]
- Chen, X.; Khunjar, W.; Jun, Z.; Li, J.; Yu, X.; Zhang, Z. Synthesis of nano-zeolite from coal fly ash and its potential for nutrient sequestration from anaerobically digested swine wastewater. Bioresour. Technol. 2012, 110, 79–85. [Google Scholar] [CrossRef]
- Qiu, Q.; Chen, Q.; Jiang, X.; Lv, G.; Chen, Z.; Lu, S.; Ni, M.; Yan, J.; Lin, X.; Song, H.; et al. Improving microwave-assisted hydrothermal degradation of PCDD/Fs in fly ash with added Na2HPO4 and water-washing pretreatment. Chemosphere 2019, 220, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.; Wu, Z.; Yip, A.C.K. Advances in the Green Synthesis of Microporous and Hierarchical Zeolites: A Short Review. Catalysts 2019, 9, 274. [Google Scholar] [CrossRef]
- Liu, Z.; Okabe, K.; Anand, C.; Yonezawa, Y.; Zhu, J.; Yamada, H.; Endo, A.; Yanaba, Y.; Yoshikawa, T.; Ohara, K.; et al. Continuous flow synthesis of ZSM-5 zeolite on the order of seconds. Proc. Natl. Acad. Sci. USA 2016, 113, 14091–14096. [Google Scholar] [CrossRef] [PubMed]
- López-Delgado, A.; Robla, J.I.; Padilla, I.; López-Andrés, S. Zero-waste process for the transformation of a hazardous aluminum waste into a raw material to obtain zeolites. J. Clean. Prod. 2020, 255, 120178. [Google Scholar] [CrossRef]
- Colella, C.; Wise, W.S. The IZA Handbook of Natural Zeolites: A tool of knowledge on the most important family of porous minerals. Microp. Mesop. Mater. 2014, 189, 4–10. [Google Scholar] [CrossRef]
- de Magalhães, L.F.; da Silva, G.R.; Peres, A.C.E. Zeolite application in wastewater treatment. Adsorp. Sci. Technol. 2022. [Google Scholar] [CrossRef]
- Moshoeshoe, M.; Nadiye-Tabbiruka, M.S.; Obuseng, V. A review of the chemistry, structure, properties and applications of zeolites. Am. J. Mater. Sci. 2017, 7, 196–221. [Google Scholar] [CrossRef]
- Mumpton, F.A. La roca magica: Uses of natural zeolites inagriculture and industry. Proc. Natl. Acad. Sci. USA 1999, 96, 3463–3470. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.; Liu, S.; Chai, Y.; Wu, G.; Guan, N.; Li, L. Zeolites for separation: Fundamental and application. J. Energy Chem. 2022, 71, 288–303. [Google Scholar] [CrossRef]
- Pérez-Botella, E.; Palomino, M.; Valencia, S.; Rey, F. Nanoporous Materials for Gas Storage; Springer: Singapore, 2019; pp. 173–208. [Google Scholar]
- Lin, H.; Wu, X.; Zhu, J. Kinetics, equilibrium, and thermodynamics of ammonium sorption from swine manure by natural chabazite. Sep. Sci. Technol. 2015, 51, 202–213. [Google Scholar] [CrossRef]
- Dong, L.D.; Zhu, Z.; Qiu, Y.; Zhao, J. Removal of lead from aqueous solution by hydroxyapatite/magnetite composite adsorbent. Chem. Eng. J. 2010, 165, 827–834. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solid sand liquids. Part-I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Freundlich, H. Colloid and Capillary Chemistry; Metheum: London, UK, 1926; p. 993. [Google Scholar]
- Millar, G.J.; Winnett, A.; Thompson, T.; Couperthwaite, S.J. Equilibrium studies of ammonium exchange with Australian natural zeolites. J. Water Process Eng. 2016, 9, 47–57. [Google Scholar] [CrossRef]
- Sprynskyy, M.; Buszewski, B.; Terzyk, A.P.; Namieśnik, J. Study of the selection mechanism of heavy metal (Pb2+, Cu2+, Ni2+, and Cd2+) adsorption on clinoptilolite. J. Colloid Interface Sci. 2006, 304, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Ugurlu, M.; Karaoglu, M.H. Adsorption of ammonium from an aqueous solution by fly ash and sepiolite: Isotherm, kinetic and thermodynamic analysis. Microp. Mesop. Mater. 2011, 139, 173–178. [Google Scholar] [CrossRef]
- Ji, X.D.; Zhang, M.L.; Ke, Y.Y.; Song, Y.C. Simultaneous immobilization of ammonium and phosphate from aqueous solution using zeolites synthesized from fly ashes. Water Sci. Technol. 2013, 67, 6. [Google Scholar] [CrossRef] [PubMed]
- Makgabutlane, B.; Nthunya, L.N.; Musyoka, N.; Dladla, B.S.; Nxumalo, E.N.; Mhlanga, S.D. Microwave assisted synthesis of coal fly ash-based zeolites for removal of ammonium from urine. RSC Adv. 2020, 10, 2416–2427. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Xu, D.; Han, L.; Niu, D.; Zhang, L.; Wu, W.; Tianm, B. Ammonium removal from aqueous solution by zeolites synthesized from low-calcium and high-calcium fly ashes. Desalination 2011, 277, 46–53. [Google Scholar] [CrossRef]
- Beler-Baykal, B.; Allar, A.D. Upgrading fertilizer production wastewater effluent quality for ammonium discharges through ion exchange with clinoptilolite. Environ. Technol. 2008, 29, 665–672. [Google Scholar] [CrossRef]
- Liu, Y.; Yan, C.; Zhao, J.; Zhang, Z.; Wang, H.; Zhou, S. Synthesis of zeolite P1 from fly ash under solvent-free conditions for ammonium removal from water. J. Clean. Prod. 2018, 202, 11–22. [Google Scholar] [CrossRef]
- Fu, H.; Li, Y.; Yu, Z.; Shen, J.; Li, J.; Zhang, M.; Ding, T.; Xu, L.; Lee, S.S. Ammonium removal using a calcined natural zeolite modified with sodium nitrate. J. Hazard. Mater. 2020, 393, 122481. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wang, N.; Kang, Z.; Yang, X.; Pan, M. An Investigation into the Adsorption of Ammonium by Zeolite-Magnetite Composites. Minerals 2022, 12, 256. [Google Scholar] [CrossRef]
- Shaban, M.; AbuKhadra, M.R.; Nasief, F.M.; El-Salam, H.M.A. Removal of Ammonia from Aqueous Solutions, Ground Water, and Wastewater Using Mechanically Activated Clinoptilolite and Synthetic Zeolite-A: Kinetic and Equilibrium Studies. Water Air Soil Pollut. 2017, 228, 450. [Google Scholar] [CrossRef]
- Otal, E.; Vilches, L.F.; Luna, Y.; Poblete, R.; García-Maya, J.M.; Fernández-Pereira, C. Ammonium ion adsorption and settleability improvement achieved in a synthetic zeolite-amended activated sludge. Chin. J. Chem. Eng. 2013, 21, 1062–1068. [Google Scholar] [CrossRef]
- Franus, M.; Wdowin, M.; Bandura, L.; Franus, W. Removal of environmental pollutions using zeolites from fly ash: A review. Fresenius Environ. Bull. 2015, 24, 854–866. [Google Scholar]
- Xie, J.; Wang, Z.; Fang, D.; Li, C.; Wu, D. Green synthesis of a novel hybrid sorbent of zeolite/lanthanum hydroxide and its application in the removal and recovery of phosphate from water. J. Colloid Inter. Sci. 2014, 423, 13–19. [Google Scholar] [CrossRef]
- Hui, K.S.; Chao, C.Y.; Kot, S. Removal of mixed heavy metal ions in wastewater by zeolite 4A and residual products from recycled coal fly ash. J. Hazard. Mater. 2005, 127, 89–101. [Google Scholar] [CrossRef]
- Alvarez-Ayuso, E.; Garcia-Sanchez, A.; Querol, X. Purification of metal electroplating waste waters using zeolites. Water Res. 2003, 37, 4855–4862. [Google Scholar] [CrossRef]
- Nightingale, E.R. Phenomenological theory of ion solvation. Effective radii of hydrated ions. J. Phys. Chem. 1959, 63, 1381–1387. [Google Scholar] [CrossRef]
- Marcus, Y. Thermodynamics of solvation of ions. Part 5.—Gibbs free energy of hydration at 298.15 K. J. Chem. Soc. Faraday Trans. 1991, 87, 2995–2999. [Google Scholar] [CrossRef]
- Joseph, I.V.; Tosheva, L.; Doyle, A.M. Simultaneous removal of Cd(II), Co(II), Cu(II), Pb(II), and Zn(II) ions from aqueous solutions via adsorption on FAU-type zeolites prepared from coal fly ash. J. Environ. Chem. Eng. 2020, 8, 103895. [Google Scholar] [CrossRef]
- Ibrahim, H.S.; Jamil, T.S.; Hegazy, E.Z. Application of zeolite prepared from Egyptian kaolin for the removal of heavy metals: II. Isotherm models. J. Hazard. Mater. 2010, 182, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Kesraoui-Ouki, S.; Cheeseman, C.R.; Perry, R. Natural zeolite utilization in pollution-control—A review of applications to metals effluents. J. Chem. Technol. Biotechnol. 1994, 59, 121–126. [Google Scholar] [CrossRef]
- Oter, O.; Akcay, H. Use of natural clinoptilolite to improve, water quality: Sorption and selectivity studies of lead(II), copper(II), zinc(II), and nickel(II). Water Environ. Res 2007, 79, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Caputo, D.; Pepe, F. Experiments and data processing of ion exchange equilibria involving Italian natural zeolites: A review. Microp. Mesop. Mater. 2007, 105, 222–231. [Google Scholar] [CrossRef]
- Bosso, S.T.; Enzweiler, J. Evaluation of heavy metal removal from aqueous solution onto scolecite. Water Res. 2002, 36, 4795–4800. [Google Scholar] [CrossRef]
- Deng, Q.; Dhar, B.R.; Elbeshbishy, E.; Lee, H.S. Ammonium nitrogen removal from the permeates of anaerobic membrane bioreactors: Economic regeneration of exhausted zeolite. Environ. Technol. 2014, 35, 2008–2017. [Google Scholar] [CrossRef]
- Lind, B.-B.; Ban, Z.; Byden, S. Nutrient recovery from human urine by struvite crystallization with ammonia adsorption on zeolite and wollastonite. Biores. Technol. 2000, 73, 169–174. [Google Scholar] [CrossRef]
- Zhao, Y.; Niu, Y.; Hu, X.; Xi, B.; Peng, X.; Liu, W.; Guan, W.; Wang, L. Removal of ammonium ions from aqueous solutions using zeolite synthesized from red mud. Desalination Water Treat. 2016, 57, 4720–4731. [Google Scholar] [CrossRef]
- Zanin, E.; Scapinello, J.; de Oliveira, M.; Rambo, C.L.; Franscescon, F.; Freitas, L.; Maria, J.; de Mello, M.; Fiori, M.A.; Oliveira, J.V.; et al. Adsorption of heavy metals from wastewater graphic industry using clinoptilolite zeolite as adsorbent. Process. Saf. Environ. Prot. 2017, 105, 194–200. [Google Scholar] [CrossRef]
- Dal Bosco, S.M.; Jimenez, R.S.; Carvalho, W. Removal of toxic metals from wastewater by Brazilian natural scolecite. J. Colloid Interface. Sci. 2005, 281, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Ćurković, L.; Cerjan-Stefanović, Š.; Filipan, T. Metal ion exchange by natural and modified zeolites. Water Res. 1997, 31, 1379–1382. [Google Scholar] [CrossRef]
- Pérez-Botella, E.; Valncia, S.; Rey, F. Zeolites in Adsorption Processes: State of the Art and Future Prospects. Chem. Rev. 2022, 122, 17647–17695. [Google Scholar] [CrossRef] [PubMed]
- Margeta, K.; Zabukovec, N.; Siljeg, M.; Farkas, A. Natural zeolites in water treatment—How effective is their use. In Water Treatment; IntechOpen: London, UK, 2013; Chapter 5. [Google Scholar]
- Haji, S.; Al-Buqaishi, B.A.; Bucheeri, A.A.; Bu-Ali, Q.; Al-Aseeri, M.; Ahmed, S. The dynamics and equilibrium of ammonium removal from aqueous solution by Na-Y zeolite. Desalination Water Treat. 2016, 57, 18992–19001. [Google Scholar] [CrossRef]
- Walton, K.S.; Abney, M.B.; LeVan, M.D. CO2 adsorption in Y and X zeolites modified by alkali metal cation exchange. Microporous Mesoporous Mater. 2006, 91, 78. [Google Scholar] [CrossRef]
- Rabo, J.A.; Schoonover, M.W. Early discoveries in zeolite chemistry and catalysis at Union Carbide, and follow-up in industrial catalysis. Appl. Catal. A 2001, 222, 261–275. [Google Scholar] [CrossRef]
- Bujnova, A.; Lesny, J. Sorption characteristics of zinc and cadmium by some natural, modified, and synthetic zeolites. Electron. J. Chem. Technol. Biotechnol. 2004, 59, 121–126. [Google Scholar]
- Inglezakis, V.J.; Loizidou, M.M.; Grigoropoulou, H.P. Ion exchange studies on natural and modified zeolites and the concept of exchange site accessibility. J. Colloid Inter. Sci. 2004, 275, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Oren, A.H.; Kaya, A. Factors affecting adsorption characteristics of Zn2+ on two natural zeolites. J. Hazard. Mater. 2006, 131, 59–65. [Google Scholar] [CrossRef]
- Kosmulski, M. The pH dependent surface charging and points of zero charge. VII. Update. Adv. Colloid Inter. Sci. 2018, 251, 115–138. [Google Scholar] [CrossRef]
- Kosmulski, M. The pH dependent surface charging and points of zero charge. VIII. Update. Adv. Colloid Inter. Sci. 2020, 275, 102064. [Google Scholar] [CrossRef] [PubMed]
- Monhemius, A.J. Precipitation diagrams for metal hydroxides, sulphides, arsenates and phosphates. Trans. Inst. Min. Metall. 1977, 6, 203–206. [Google Scholar]
- Goscianska, J.; Ptaszkowska-Koniarz, M.; Frankowski, M.; Franus, M.; Panek, R.; Franus, W. Removal of phosphate from water by lanthanum-modified zeolites obtained from fly ash. J. Colloid Inter. Sci. 2018, 513, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.Y.; Zhang, B.H.; Li, C.J.; Zhang, Z.J.; Kong, H.N. Simultaneous removal of ammonium and phosphate by zeolite synthesized from fly ash as influenced by salt treatment. J. Colloid Inter. Sci. 2006, 304, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.G.; Kong, H.N.; Wu, D.Y.; Hu, Z.B.; Wang, Z.S.; Wang, Y.H. Removal of phosphate from aqueous solution by zeolite synthesized from fly ash. J. Colloid Interface Sci. 2006, 300, 491–497. [Google Scholar] [CrossRef]
- Aylward, G.H.; Findlay, T.J.V. SI Chemical Data, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 1974. [Google Scholar]
- Hamdi, N.; Srasra, E. Removal of phosphate ions from aqueous solution using Tunisian clays minerals and synthetic zeolite. J. Environ. Sci. 2012, 24, 617–623. [Google Scholar] [CrossRef]
- Zhang, Y.; Kou, X.; Lu, H.; Lv, X. The feasibility of adopting zeolite in phosphorus removal from aqueous solutions. Desalination Water Treat. 2014, 52, 4298–4304. [Google Scholar] [CrossRef]
- Alshameri, A.; Yan, C.; Lei, X. Enhancement of phosphate removal from water by TiO2/Yemeni natural zeolite: Preparation, characterization and thermodynamic. Microporous Mesoporous Mater. 2014, 196, 145–157. [Google Scholar] [CrossRef]
- Naghash, A.; Nezamzadeh-Ejhieh, A. Comparison of the efficiency of modified clinoptilolite with HDTMA and HDP surfactants for the removal of phosphate in aqueous solutions. J. Ind. Eng. Chem. 2015, 31, 185–191. [Google Scholar] [CrossRef]
- Hermassi, M.; Valderrama, C.; Font, O.; Moreno, N.; Querol, X.; Batis, N.; Cortina, J. Phosphate recovery fromaqueous solution by K-zeolite synthesized from fly ash for subsequent valorisation as slow release fertilizer. Sci. Total Environ. 2020, 731, 139002. [Google Scholar] [CrossRef]
- Ciesla, J.; Franus, W.; Franus, M.; Kedziora, K.; Gluszczyk, J.; Szerement, J.; Jozefaciuk, G. Environmental-friendly modifications of zeolite to increase its sorption and anion exchange Properties, Physicochemical Studies of the Modified Materials. Material 2019, 12, 3213. [Google Scholar] [CrossRef] [PubMed]
- Mokrzycki, J.; Fedyna, M.; Marzec, M.; Szerement, J.; Panek, R.; Klimek, A.; Bajda, T.; Mierzwa-Hersztek, M. Copper ion-exchanged zeolite X from fly ash as an efficient adsorbent of phosphate ions from aqueous solutions. J. Environ. Chem. Eng. 2022, 10, 108567. [Google Scholar] [CrossRef]
- Mahmoodi, N.; Saffar-Dastgerdi, M.H. Zeolite nanoparticle as a superior adsorbent with high capacity: Synthesis, surface modification and pollutant adsorption ability from wastewater. Microchem. J. 2019, 145, 74–83. [Google Scholar] [CrossRef]
- Swiderska-Dabrowska, R.; Schmidt, R.; Sikora, A. Effect of calcination temperature on chemical stability of zeolite modified by iron ions. In Polish Environmental Engineering 2012; Environmental Engineering Committee PAS, Ed.; Environmental Engineering Committee PAS: Lublin, Poland, 2012; pp. 307–317. [Google Scholar]
- Glassman, H.N. Surface active agents and their application in bacteriology. Bacteriol. Rev. 1948, 12, 105. [Google Scholar] [CrossRef]
- Reeve, P.J.; Fallowfield, H.J. Natural and surfactant modified zeolites: A review of their applications for water remediation with a focus on surfactant desorption and toxicity towards microorganisms. J. Environ. Manag. 2018, 205, 253–261. [Google Scholar] [CrossRef]
- Ivankovic, T.; Hrenovic, J. Surfactants in the environment. Arch. Ind. Hyg. Toxicol. 2010, 61, 95–110. [Google Scholar] [CrossRef]
- Li, Z. Sorption kinetics of hexadecyltrimethylammonium on natural clinoptilolite. Langmuir 1999, 15, 6438–6445. [Google Scholar] [CrossRef]
- Bowman, R.S. Applications of surfactant-modified zeolites to environmental remediation. Microporous Mesoporous Mater. 2003, 61, 43–56. [Google Scholar] [CrossRef]
- Baez-Alvarado, M.D.; Olguín, M.T. Surfactant-modified clinoptilolite-rich tuff to remove barium (Ba2+) and fulvic acid from mono- and bi-component aqueous media. Microporous Mesoporous Mater. 2011, 139, 81–86. [Google Scholar] [CrossRef]
- Abdellaoui, Y.; Oualid, H.A.; Hsini, A.; Ibrahimi, B.E.; Laabd, M.; Ouardi, M.E.; Giácoman-Vallejos, G.; Gamero-Melo, P. Synthesis of zirconium-modified Merlinoite from fly ash for enhanced removal of phosphate in aqueous medium: Experimental studies supported by Monte Carlo/SA simulations. Chem. Eng. J. 2021, 404, 126600. [Google Scholar] [CrossRef]
- Li, Z.; Willims, C.; Roy, S.; Bowman, R.S. Desorption of hexadecyltrimethylammonium from charged mineral surfaces. Environ. Geosci. 2017, 10, 37–45. [Google Scholar] [CrossRef]
- de Gennaro, B.; Catalanotti, L.; Bowman, R.S.; Mercurio, M. Anion exchange selectivity of surfactant modified clinoptilolite-rich tuff for environmental remediation. J. Colloid Inter. Sci. 2014, 430, 178–183. [Google Scholar] [CrossRef]
- Gouran-Orimi, R.; Mirzayi, B.; Nematollahzadeh, A.; Tardast, A. Competitive adsorption of nitrate in fixed-bed column packed with bio-inspired polydopamine coated zeolite. J. Environ. Chem. Eng. 2018, 6, 2232–2240. [Google Scholar] [CrossRef]
- Lin, H.; Huang, X.; Chang, J.; Li, B.; Bai, Y.; Su, B.; Shi, L.; Dong, Y. Improving sludge settling performance of secondary settling tank and simultaneously adsorbing nitrate and phosphate with surfactant modified zeolite (SMZ) ballasted flocculation. J. Environ. Chem. Eng. 2023, 11, 109650. [Google Scholar] [CrossRef]
- Zhan, Y.; Lin, J.; Zhu, Z. Removal of nitrate from aqueous solution using cetylpyridinium bromide (CPB) modified zeolite as adsorbent. J. Hazard. Mater. 2011, 186, 1972–1978. [Google Scholar] [CrossRef]
- Schick, J.; Caullet, P.; Paillaud, J.-L.; Patarin, J.; Mangold-Callarec, C. Batch-wise nitrate removal from water on a surfactant-modified zeolite. Microporous Mesoporous Mater. 2010, 132, 395–400. [Google Scholar] [CrossRef]
- Schick, J.; Caullet, P.; Paillaud, J.-L.; Patarin, J.; Mangold-Callarec, C. Nitrate sorption from water on a Surfactant-Modified Zeolite. Fixed-bed column experiments. Microporous Mesoporous Mater. 2011, 142, 549–556. [Google Scholar] [CrossRef]
- Li, Z.; Roy, S.J.; Zou, Y.; Bowman, R.S. Long-Term chemical and biological stability of surfactant-modified zeolite. Environ. Sci. Technol. 1998, 32, 2628–2632. [Google Scholar] [CrossRef]
- Hrenovic, J.; Rozic, M.; Sekovanic, L.; Anic-Vucinic, A. Interaction of surfactant-modified zeolites and phosphate accumulating bacteria. J. Hazard. Mater. 2008, 156, 576–582. [Google Scholar] [CrossRef]
- Li, Z. Chromate transport through surfactant-modified zeolite columns. Groundw. Monit. Remediat. 2006, 26, 117–124. [Google Scholar] [CrossRef]
- Li, Z.; Hong, H. Retardation of chromate through packed columns of surfactant-modified zeolite. J. Hazard. Mater. 2009, 162, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Bowman, R.S. Retention of inorganic oxyanions by organo-kaolinite. Water Res. 2001, 35, 3771–3776. [Google Scholar] [CrossRef]
- Nye, J.V.; Guerin, W.F.; Boyd, S.A. Heterotrophic activity of microorganisms in soils treated with quaternary ammonium compounds. Environ. Sci. Technol. 1994, 28, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Hrenovic, J.; Ivankovic, T. Toxicity of anionic and cationic surfactant to Acinetobacter junii in pure culture Cent. Eur. J. Biol. 2007, 2, 405–414. [Google Scholar]
- Bingre, R.; Louis, B.; Nguyen, P. An overview on zeolite shaping technology and solutions to overcome diffusion limitations. Catalysts 2018, 8, 163. [Google Scholar] [CrossRef]
- Gleichmann, K.; Unger, B.; Brandt, A. Manufacturing of industrial zeolite molecular sieves. Chem. Ing. Technol. 2017, 89, 851–862. [Google Scholar] [CrossRef]
- Amir, C.; Mohammad, K.; Javad, A.S.; Sareh, A.A. Effect of bentonite binder on adsorption and cation exchange properties of granulated nano nay zeolite. Adv. Mater. Res. 2011, 335–336, 423–428. [Google Scholar] [CrossRef]
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering: Treatment and Reuse, 4th ed.; Metcalf & Eddy, Inc.; McGraw-Hill: New York, NY, USA, 2003; pp. 1138–1162. [Google Scholar]
- Loiola, A.R.; Bessa, R.A.; Oliveira, C.P.; Freitas, A.D.L.; Soares, S.A.; Bohn, F.; Pergher, S.B.C. Magnetic zeolite composites: Classification, synthesis routes, and technological applications. J. Magn. Magn. Mater. 2022, 560, 169651. [Google Scholar] [CrossRef]
- Lu, A.-H.; Salabas, E.L.; Schüth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef]
- Kharisov, B.I.; Dias, H.V.R.; Kharissova, O.V.; Jiménez-Pérez, V.M.; Pérez, B.O.; Flores, B.M. Iron-containing nanomaterials: Synthesis, properties, and environmental applications. RSC Adv. 2012, 2, 9325–9358. [Google Scholar] [CrossRef]
- Popescu, R.C.; Andronescu, E.; Vasile, B.S. Recent advances in magnetite nanoparticle functionalization for nanomedicine. Nanomaterials 2019, 9, 1791. [Google Scholar] [CrossRef] [PubMed]
- Phouthavong, V.; Yan, R.; Nijpanich, S.; Hagio, T.; Ichino, R.; Kong, L.; Li, L. Magnetic adsorbents for wastewater treatment: Advancements in their synthesis methods. Materials 2022, 15, 1053. [Google Scholar] [CrossRef]
- Cataldo, E.; Salvi, L.; Paoli, F.; Fucile, M.; Masciandaro, G.; Manzi, D.; Masini, C.M.; Mattii, G.B. Application of zeolites in agriculture and other potential uses: A review. Agronomy 2021, 11, 1547. [Google Scholar] [CrossRef]
- Pond, W.G.; Mumpton, F.A. Zeo-Agriculture—Use of Natural Zeolites in Agriculture and Aquaculture; Westview Press: Brockport, NY, USA, 1984. [Google Scholar]
- Li, Z. Use of surfactant-modified zeolite as fertilizer carriers to control nitrate release. Microporous Mesoporous Mater. 2003, 61, 181–188. [Google Scholar] [CrossRef]
- Filatova, E.G.; Pozhidaev, Y.N. Development of natural zeolites regeneration scheme. IOP Conf. Ser. Earth Environ. Sci. 2020, 459, 032035. [Google Scholar] [CrossRef]
- Zhang, Y.; Prigent, B.; Geißen, S.-U. Adsorption and regenerative oxidation of trichlorophenol with synthetic zeolite: Ozone dosage and its influence on adsorption performance. Chemosphere 2016, 154, 132–137. [Google Scholar] [CrossRef]
- Lubensky, J.; Ellersdorfer, M.; Stocker, K. Ammonium recovery from model solutions and sludge liquor with a combined ion exchange and air stripping process. J. Water Process Eng. 2019, 32, 100909. [Google Scholar] [CrossRef]
- Malovanyy, A.; Sakalova, H.; Yatchyshyn, Y.; Plaza, E.; Malovanyy, M. Concentration of ammonium from municipal wastewater using ion exchange process. Desalination 2013, 329, 93–102. [Google Scholar] [CrossRef]
- Salvador, F.; Martin-Sanchez, N.; Sanchez-Hernandez, R.; Sanchez-Montero, M.J.; Izquierdo, C. Regeneration of carbonaceous adsorbents. Part I: Thermal Regeneration. Microporous Mesoporous Mater. 2015, 202, 259–276. [Google Scholar] [CrossRef]
- Sireesha, S.; Agarwal, A.; Sopanrao, K.S.; Sreedhar, L.; Anitha, K.L. Modified coal fly ash as a low-cost, efficient, green, and stable adsorbent for heavy metal removal from aqueous solution. Biomass Convers. Biorefinery 2022. [Google Scholar] [CrossRef]
- Batjargal, T.; Yang, J.-S.; Kim, D.-H.; Baek, K. Removal characteristics of Cd(II), Cu(II), Pb(II), and Zn(II) by natural Mongolian zeolite through batch and column experiments. Sep. Sci. Technol. 2011, 46, 1313–1320. [Google Scholar] [CrossRef]
- Bolan, N.S.; Mowatt, C.; Adriano, D.C.; Blennerhassett, J.D. Removal of ammonium ions from fellmongery effluent by zeolite. Commun. Soil Sci. Plant Anal. 2003, 34, 1861–1872. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Xu, D.; Han, L.; Niu, D.; Tian, B.; Zhang, J.; Zhang, L.; Wu, W. Removal of ammonium from aqueous solutions using zeolite synthesized from fly ash by a fusion method. Desalination 2011, 271, 111–121. [Google Scholar] [CrossRef]
- Karadag, D.; Tok, S.; Akgul, E.; Turan, M.; Ozturk, M.; Demir, A. Ammonium removal from sanitary landfill leachate using natural G¨ordes clinoptilolite. J. Hazard. Mater. 2008, 153, 60–66. [Google Scholar] [CrossRef]
- Lorick, D.; Ahlström, M.; Grimvall, A.; Harder, R. Effectiveness of struvite precipitation and ammonia stripping for recovery of phosphorus and nitrogen from anaerobic digestate: A systematic review. Environ. Evid. 2020, 9, 27. [Google Scholar] [CrossRef]
- Huang, J.-C.; Shang, C. Air stripping. In Handbook of Environmental Engineering. Vol. 4: Advanced Physicochemical Treatment Processes; Wang, L.L., Hung, Y.-T., Shammas, N.K., Eds.; The Humana Press Inc.: Totowa, NJ, USA, 2006. [Google Scholar]
- Katehis, D.; Diyamandoglu, V.; Fillos, J. Stripping and recovery of ammonia from centrate of anaerobically digested biosolids at elevated temperatures. Water Environ. Res. 1998, 70, 231–240. [Google Scholar] [CrossRef]
- Değermenci, N.; Yildiz, E. Ammonia stripping using a continuous flow jet loop reactor: Mass transfer of ammonia and effect on stripping performance of influent ammonia concentration, hydraulic retention time, temperature, and air flow rate. Environ. Sci. Pollut. Res. 2021, 28, 31462–31469. [Google Scholar] [CrossRef]
- Guisnet, M.; Ribeiro, F.R. Chapter 1 Deactivation and regenaration of solid catalysts. In Deactivation and Regeneration of Zeolite Catalysts; Catalytic Science Series; Guisnet, M., Robeiro, F.R., Eds.; Imperial College Press: London, UK, 2011; Volume 9, p. 360. [Google Scholar] [CrossRef]
- Chen, S.; Popovich, J.; Zhang, W.; Ganser, C.; Hadel, S.E.; Seo, D.-K. Superior ion release properties and antibacterial efficacy of nanostructured zeolites ion-exchanged with zinc, copper, and iron. RSC Adv. 2018, 8, 37949–37957. [Google Scholar] [CrossRef]
- Daligaux, V.; Richard, R.; Manero, M.-H. Deactivation and Regeneration of Zeolite Catalysts Used in Pyrolysis of PlasticWastes—A Process and Analytical Review. Catalysts 2021, 11, 770. [Google Scholar] [CrossRef]
- Guisnet, M.; Ribeiro, F.R. Fundamental description of deactivation and regeneration of acid zeolites. Stud. Surf. Sci. Catal. 1994, 88, 53–68. [Google Scholar]
- Salvador, F.; Martin-Sanchez, N.; Sanchez-Hernandez, R.; Sanchez-Montero, M.J.; Izquierdo, C. Regeneration of carbonaceous adsorbents. Part II: Chemical, Microbiological and Vacuum Regeneration. Microporous Mesoporous Mater. 2015, 202, 277–296. [Google Scholar] [CrossRef]
- Wang, S.; Li, H.; Xie, S.; Liu, S.; Xu, L. Physical and chemical regeneration of zeolitic adsorbents for dye removal in wastewater treatment. Chemosphere 2006, 65, 82–87. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Shi, T.B.; Jia, C.Z.; Ji, W.J.; Chen, Y.; He, M.Y. Adsorptive removal of aromatic organosulfur compounds over the modified Na-Y zeolites. Appl. Catal. B Environ. 2008, 82, 1–10. [Google Scholar] [CrossRef]
- Fujita, H.; Izumi, J.; Sagehashi, M.; Fujii, T.; Sakoda, A. Decomposition of trichloroethene on ozone-adsorbed high silica zeolites. Water Res. 2004, 38, 166–172. [Google Scholar] [CrossRef]
- Lee, D.-G.; Kim, J.-H.; Lee, C.-H. Adsorption and thermal regeneration of acetone and toluene vapors in dealuminated Y-zeolite bed. Sep. Purif. Technol. 2011, 77, 312–324. [Google Scholar] [CrossRef]
- Simancas, R.; Chokkalingam, A.; Elangovan, S.P.; Liu, Z.; Sano, T.; Iyoki, K.; Wakihara, T.; Okubo, T. Recent progress in the improvement of hydrothermal stability of zeolites. Chem. Sci. 2021, 12, 7677. [Google Scholar] [CrossRef]
- Bathen, D. Physical waves in adsorption technology—An overview. Sep. Purif. Technol. 2003, 33, 163–177. [Google Scholar] [CrossRef]
- Abdelsayed, V.; Shekhawat, D.; Tempke, R.S. Zeolites interactions with microwaves during methane non-oxidative coupling. Catal. Today 2021, 365, 88–102. [Google Scholar] [CrossRef]
- Delgado, L.; Catarino, A.S.; Eder, P.; Litten, D.; Luo, Z.; Villanueva, A. End-of-Waste Criteria; EUR—Scientific and Technical Research series; European Commission Joint Research Centre: Seville, Spain, 2009. [Google Scholar] [CrossRef]
- Grand View Research, Market Analysis Report: Zeolite Market Size, Share & Trends Analysis Report By Application (Catalyst, Adsorbent, Detergent Builder), By Product (Natural, Synthetic), By Region (North America, Europe, APAC, CSA, MEA), And Segment Forecasts, 2022–2030. Grand View Research, India & US, Report 978-1-68038-601-1, p. 114. Available online: https://www.grandviewresearch.com/industry-analysis/zeolites-market (accessed on 25 October 2023).
- Markets and Markets, Market Research Report: Zeolites Market by Type (Natural, Synthetic), Function (Ion-Exchange, Catalyst, Molecular Sieve), Synthetic Zeolites Application (Detergent, Catalyst), Natural Zeolites Application, and Regional-Global Forecast to 2026. Markets and Markets, India, Report CH 8006. 2021. Available online: https://www.marketsandmarkets.com/Market-Reports/zeolites-market-76442083.html (accessed on 25 October 2023).
- Coherent Market Insights (2023) Nutrient Recycling Market. Coherent Market Insights, India, Report CM15972, p. 154. Available online: https://www.coherentmarketinsights.com/market-insight/nutrient-recycling-market-5972 (accessed on 25 October 2023).





| Ref. | Al, Si Sources | Pre-Treatment | Hydrothermal Treatment | Products | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type | In MSW-FA | BET | CEC | Initial Si/Al Ratio | Type | Type | Additives | L/S | Temp | Time | Zeolite | BET | CEC | ||
| Si | Al | m2/g | meq/g | - | mL/g | °C | h | m2/g | meq/g | ||||||
| [77] | MSW-FA | 9.5% SiO2 | 5.5% Al2O3 | N.S. | N.S. | 1.5 | DW, Cal 1, Acid 1, DI | Autoclave w/ST | 1–2 M NaOH | 25 | 60 | 20–30 | Zeolite A | N.S. | N.S. |
| 0.5–2 M NaOH | 25 | 100–120 | 20–48 | Zeolite P | N.S. | N.S. | |||||||||
| [78] | MSW-FA (<75 µm, WMS) | 22.0% SiO2 | 10.2% Al2O3 | 2.54 | 0.078 | 1.9 | No | N.S. | 3.5 M NaOH | 5–15 | 130–190 | 24 | Gismondine | 45 | 0.8 |
| 6 M NaOH | Gmelinite | N.S. | 0.65 | ||||||||||||
| [79] | MSW-FA (water-cooled) | 47.1% SiO2 | 12.4% Al2O3 | 0.5 | N.S. | 4 | Gr | Autoclave | 2 M NaOH | 30 | 200 | >24 | Zeolite-like | 54 | N.S. |
| [65] | MSW-FA, WGP, Al2O3 powder | 0.3% Si | 2.2% Al | N.S. | 0.20 | 1 | No | Heater w/ST | 2.5 M NaOH | 10 | 60 | 24 | Zeolites Y, A and L (perlialite) | N.S. | 1.00 |
| AF (1.2 g NaOH/g FA, 550 °C, 1 h) | Aging w/ST | WGP, Al2O3 powder | 10 | Room | 24 | - | - | ||||||||
| CRY | No | - | 90 | 24 | Zeolite-like | N.S. | 1.00 | ||||||||
| [80] | MSW-FA | 12.2% Si | 0.8% Al | 8.59 | N.S. | 40 | AF (1.5 g NaOH/g FA, 400 °C, 40 min) | HYD | DI | 100 | 105 | 24 | - | - | - |
| Sealed reactor | 1.2% CTAB 2 | Filtrate | 105 | 24 | - | - | - | ||||||||
| Cal | - | - | 550 °C | 3 | ZSM-23 | 651 | N.S. | ||||||||
| [54] | MSW-FA and coal-FA | 45.5% SiO2 | 22.6% Al2O3 | N.S. | 0.64 | 1.8 | Gr, AF (1.2 g NaOH/g FA, 550 °C, 1 h) | Aging w/ST | DI | 9 | Room | 24 | - | - | - |
| CRY | No | - | 90 | 6–10 | Zeolite X | 200 | 2.5 | ||||||||
| ≥130 | 14–18 | Zeolite HS | N.S. | N.S. | |||||||||||
| [81] | MSW-FA | N.S. | N.S. | N.S. | N.S. | N.S. | No | MWA | DI, 1.5 mol NaH2PO4/kg FA | 2 | 200 | 1/6 | Zeolite-like | N.S. | N.S. |
| [82] | MSW-FA | N.S. | N.S. | N.S. | N.S. | N.S. | No | MWA | 1 M Na2HPO4 | 3 | 150 | 1/3 | Zeolite-like | N.S. | N.S. |
| [83] | MSW-FA | 4.3% Si | 2.4% Al | 0.023 | AF (2 g NaOH/g FA, 550 °C, 1 h) | MWA | DI | 3.3 | 100 | 0.5 | Zeolite-like | N.S. | 1.17 | ||
| No | MWA | DI | 3.3 | 100 | 0.5 | Zeolite-like | N.S. | 0.43 | |||||||
| Waste Material | Al2O3 | SiO2 | CaO |
|---|---|---|---|
| Aluminium scrap | Almet > 90–99% | ||
| Aluminium dust | Altotal 25–40 Almet 15–25 | 6–11 | 1–4 |
| Black aluminium dross | 42–88 | 1.3–14 | 0.6–1 |
| White aluminium dross | 40–50 | ||
| Spent Fluid Catalytic Cracking catalysts | 40–50 | 40–50 | 0–1 |
| Coal combustion ashes | 15–40 | 40–60 | 3–15 |
| Aluminium salt slag | 20–30 Almet 5–10 | 2–10 | |
| Coal gasification ashes | 5–30 | 25–60 | 2–30 |
| Liquid Crystal Displays glass panel | 15–25 | 50–75 | 0–7 |
| MSW-FA | 5–24 | 12–41 | 15–50 |
| Electric furnace steel reduction slag | 15–20 | 15–20 | 50–60 |
| Lithium slag | 15–20 | 50–55 | 10–12 |
| Red mud from the Bayer process (dried) | 10–20 | 3–50 | 2–40 |
| Drilling and cutting muds (dried) | 5–20 | 30–70 | 2–30 |
| MSW-BA | 1–20 | 5–50 | 10–50 |
| Waste porcelain | 19 | 70 | 3 |
| Blast furnace iron slag | 10–15 | 30–40 | 40–50 |
| Wood ash | 0.5–15 | 10–70 | 10–70 |
| Waste foundry sand | 0–15 | 75–90 | 0–5 |
| Palm oil fuel ash (POFA) | 0.5–12 | 45–75 | 3–15 |
| Zinc slag | 7–10 | 15–20 | 15–20 |
| Electric furnace steel oxidation slag | 5–10 | 10–15 | 20–25 |
| Structure | Chemistry | ||||
|---|---|---|---|---|---|
| Zeolite | FTC | Window | Si/Al | Cation | CEC |
| Å | mol/mol | - | meq/g | ||
| Natural zeolites | |||||
| Clinoptilolite | HEU | 3.1 × 7.5 | 4.0–5.7 | Na, K, Ca | 2.0–2.6 |
| Chabazite | CHA | 3.8 | 1.4–4.0 | Na, K, Ca | 2.5–4.7 |
| Phillipsite | PHI | 3.8 | 1.1–3.3 | Na, K, Ca | 2.9–5.6 |
| Analcime | ANA | 1.6 × 4.2 | 1.5–2.8 | Na | 3.6–5.3 |
| Erionite | ERI | 3.6 × 5.1 | 2.6–3.8 | Na, K, Ca | 2.7–3.4 |
| Faujasite | FAU | 7.4 | 2.1–2.8 | Na, K, Mg | 3.0–3.4 |
| Ferrierite | FER | 4.2 × 5.4 | 4.9–5.7 | Ca | 2.1–2.3 |
| Heulandite | HEU | 3.1 × 7.5 | 4.0–6.2 | Na, K, Ca, Sr | 2.2–2.5 |
| Laumontite | LAU | 6.5 × 7.0 | 1.9–2.4 | Na, K, Mg | 3.8–4.3 |
| Synthetic zeolites | |||||
| X | FAU | 7.4 | 1.0–1.5 | - | 2.7–6.0 |
| Y | FAU | 7.4 | <3 | - | 3.9 |
| Mordenite 1 | MOR | 6.5 × 7.0 | 4.0–5.7 | Na, K, Ca | 2.0–2.4 |
| A | LTA | 4.1 × 4.5 | 1.0–3.2 | - | 3.9–5.3 |
| NaP1 | GIS | 2.9 | 1.7–3.9 | - | 2.0 |
| Zeolite | Source | Si/Al | BET | NH4+ Adsorption | CEC | References |
|---|---|---|---|---|---|---|
| mol/mol | m2/g | mg NH4/g | meq/g | |||
| Magnetic clinoptilolite | Natural | N.S. | 43.1 | 172 | N.S. | [150] |
| Na-A | Natural | N.S. | 430 | 116 | N.S. | [151] |
| Mechanically activated clinoptilolite | Natural | N.S. | 258 | 109 | N.S. | [151] |
| NaP1 | Coal FA | 2.7 | 56.9 | 34.5 | 2.56 | [126] |
| X, some A, P, and hydroxysodalite | Low-Ca coal FA | 6.0 | 27.0 | 23.8 | 2.79 | [146] |
| P1 | Coal FA | 3.5 | 18.5 | 22.9 | N.S. | [148] |
| NaOH-treated zeolite Australia | Natural | N.S. | N.S. | 19.5–20.0 | N.S. | [141] |
| Clinoptilolite-Ca/-Na, Stilbite-Ca | Natural | 2.9 | 25.8 | 17.0 | N.S. | [149] |
| Sodalite | Coal FA | N.S. | 15.5 | 16.0 | 2.92 | [145] |
| NaP1, some analcime, chabazite | Coal FA | N.S. | N.S. | 13.7 | N.S. | [152] |
| Zeolite Australia as received | Natural | 9.8 | N.S. | 8.6 | N.S. | [141] |
| Na-X | Coal FA | 1.12 | 165 | 5.0 | 18 | [153] |
| NaP1/Fe2O3 | Coal FA | 2.8 | 162 | 4.5 | 1.54 | [154] |
| Gismondine | High Ca coal FA | 4.7 | 45.5 | 3.2 | 0.69 | [146] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vogelsang, C.; Umar, M. Municipal Solid Waste Fly Ash-Derived Zeolites as Adsorbents for the Recovery of Nutrients and Heavy Metals—A Review. Water 2023, 15, 3817. https://doi.org/10.3390/w15213817
Vogelsang C, Umar M. Municipal Solid Waste Fly Ash-Derived Zeolites as Adsorbents for the Recovery of Nutrients and Heavy Metals—A Review. Water. 2023; 15(21):3817. https://doi.org/10.3390/w15213817
Chicago/Turabian StyleVogelsang, Christian, and Muhammad Umar. 2023. "Municipal Solid Waste Fly Ash-Derived Zeolites as Adsorbents for the Recovery of Nutrients and Heavy Metals—A Review" Water 15, no. 21: 3817. https://doi.org/10.3390/w15213817
APA StyleVogelsang, C., & Umar, M. (2023). Municipal Solid Waste Fly Ash-Derived Zeolites as Adsorbents for the Recovery of Nutrients and Heavy Metals—A Review. Water, 15(21), 3817. https://doi.org/10.3390/w15213817