Abstract
A comparative analysis of environmental conditions between Alpine and Apennine lakes/ponds which represent different faces of European mountain regions was conducted. The data set was created on the basis of previous works carried out by national and international institutions including biological, physical–chemical, geographic, and precipitation data from 27 lakes/ponds placed at altitudes ranging from 2334 ± 294 m a.s.l. (in the Alps) and 1541 ± 154 m a.s.l. (in the Apennines), with mean maximum depths of about 5.5 ± 4.6 m. A specific focus was dedicated to chironomids as outstanding sentinels for local and global changes in habitat conditions. Species richness and Taxonomic Distinctness Indices were applied to lakes/ponds macroinvertebrates to highlight differences in the biodiversity of the two areas. Subsequently, associations between descriptors of the mountain region climate, lithology, water chemistry, lake morphology, geography, macroinvertebrate assemblage richness, and distinctness were examined through Principal Component Analysis, Analysis of Variance, and Non-metric Multi-dimensional Scaling. Results showed strong positive correlations between mean annual precipitation and temperature with lake macroinvertebrate biodiversity as a whole and with chironomid in particular. Thus, these shore habitats face a threat under climate change conditions (impacting thermal and precipitation regimes). These results are also central in showing that even small ecosystems are important sources of biodiversity for the lower altitudes, stressing the urgency of including them within targeted monitoring and action plans to preserve their peculiar habitat, flora, and fauna.
1. Introduction
Alpine lakes have high societal, ecological, and environmental value, as they support unique plants and animal communities and are excellent sensors of environmental changes [1]. These lakes situated above or beyond the tree line and the peaks, because of their small size, shallow depth, and limited catchment area, are highly influenced by climate harshness, air and organic pollution [2,3,4,5], and hydropower generation [6,7]. Indeed, Alpine lakes at intermediate altitudes (1500–2000 m a.s.l.) have the potential to be more sensitive than high-altitude lakes to climate change because of the impact of air temperature on ice cover duration, precipitation regime, and water permanence [8,9,10]. Therefore, their peculiar flora and fauna will be strongly affected by the ongoing climate warming causing great changes in species distribution and abundance [11,12] and the timing of seasonal events [13]. Our focus is thus on small high-altitude lakes (areas below 0.5 km2) increasingly affected by the local and global change but not under consideration within the European Water Framework Directive (WFD 2000/60/EC). The WFD was fixed on less remote and larger lakes (surface areas above 0.5 km2) mistakenly thinking of mountain waters as clean and in pristine conditions. Mountain lakes represent water towers for downstream ecosystems, lowland lakes, and people [14]. Thus, they should be included in national or international conservation policies and restoration plans, considering their societal value and the impact that climate change is predicted to have on the European Alps in the near future [14]. In order to emphasize the significance of these understudied environments, we included their study into European monitoring programs such as the International Cooperative Programme on Assessment and Monitoring of the Effects of Air Pollution on Rivers and Lakes (ICP Waters; https://www.icp-waters.no/ accessed on 10 July 2023) and the Monitoring System to Detect the Effects of Reduced Pollutants Emissions resulting from NEC Directive adoption (LIFE MODERn (NEC) LIFE20 GIE/IT/000091; https://lifemodernec.eu/ accessed on 10 July 2023).
In this context, macroinvertebrates were selected as bioindicators as they dominate in mountain freshwaters [15], and they are sensitive to changes in the freshwater habitats they inhabit, and of water ecological status [16]. Human activities and impacts, hampering the natural processes of watersheds or lakes, negatively affect macroinvertebrate assemblage structure [17,18]. Nevertheless, they can respond to or counteract a wide range of pressures or climatic and morphological factors [17,18]. At the same time, among macroinvertebrates, chironomids are massively represented, but scarcely studied in these environments, even if they show high diversity [18]. Their larvae play an important role in aquatic food webs acting as a link between primary producers and secondary consumers [19]. Chironomids are characterised by short life cycles so they respond more quickly to climate change than other organisms [19].
The analysis of macroinvertebrates, with insights on chironomids, allows us to highlight environmental differences at a regional scale driven by climate or other factors (e.g., substrate composition, precipitation regime, light and nutrient condition, etc.). These regional differences can be used to extrapolate information to a larger geographical scale [11,12,18,20,21,22]. Therefore, analyses at a fine spatial scale are needed to investigate specific processes and provide useful insights for the implementation of regional-specific policies [23].
Here, we focus on lakes and ponds from two main Italian mountain ranges: the Alps and the Apennines. The latter are subject to the same risks as the Alps, but because of their gentler slope and lower mean altitudes, they are even more vulnerable to direct human impacts (presence of roads, mountain lodges, etc.) [5,24]. Both Italian mountain ranges host several alpine lakes and ponds [15,18,25,26,27,28,29], but there is still little data about their macroinvertebrate and chironomid assemblages, mainly focusing on the northern Apennines [15]. In the Alps, this is partly due to problematic sampling procedures as the lakes are located in hard-to-reach remote areas, which are snow and ice covered for almost all year [15,18,22,30]. On the northern Apennines, limited regional interest in habitat conservation policies led to less collection and publication of data [31].
Despite their importance and the environmental threats they face [5,24], high mountain lakes/ponds and their inhabitants are still poorly known and protected [32]. To assess the ecological status of high-altitude lakes/ponds on Italian main mountain ranges, we collated information from biodiversity surveys with geographical, morphometric, and climatic data. The goal was to understand the ecology of mountain lakes/ponds, and to enhance the knowledge about relationships between macroinvertebrate and chironomid assemblage structure and environmental conditions, starting from local differences. We statistically analysed them to underline which environmental feature drives macroinvertebrate richness and diversity.
The hypotheses to be tested were (H1) that there are differences in the littoral macroinvertebrate and chironomid assemblages between lakes/ponds of each mountain range; (H2) to identify taxa associated with lakes/ponds of the two mountain ranges; (H3) to explore this association at the species level; (H4) and to investigate environmental conditions (e.g., climatic and chemical conditions and lake morphology) that correlate with the littoral macroinvertebrate and chironomid assemblages.
2. Materials and Methods
2.1. Study Area
Italy is bordered to the north by the Alps, which is the largest mountain range in Europe and separates Italy from neighbouring countries such as France, Switzerland, Austria, and Slovenia. The Apennines run from north to south through the length of the Italian peninsula, with their northern end forming the boundary with the Alps. Italy is also represented by two ecoregions, Italy, Corsica and Malta and Alps (WFD, Annex XI, map “A”), split from one another by the 1000 m a.s.l. level line, and by the 44° parallel [33].
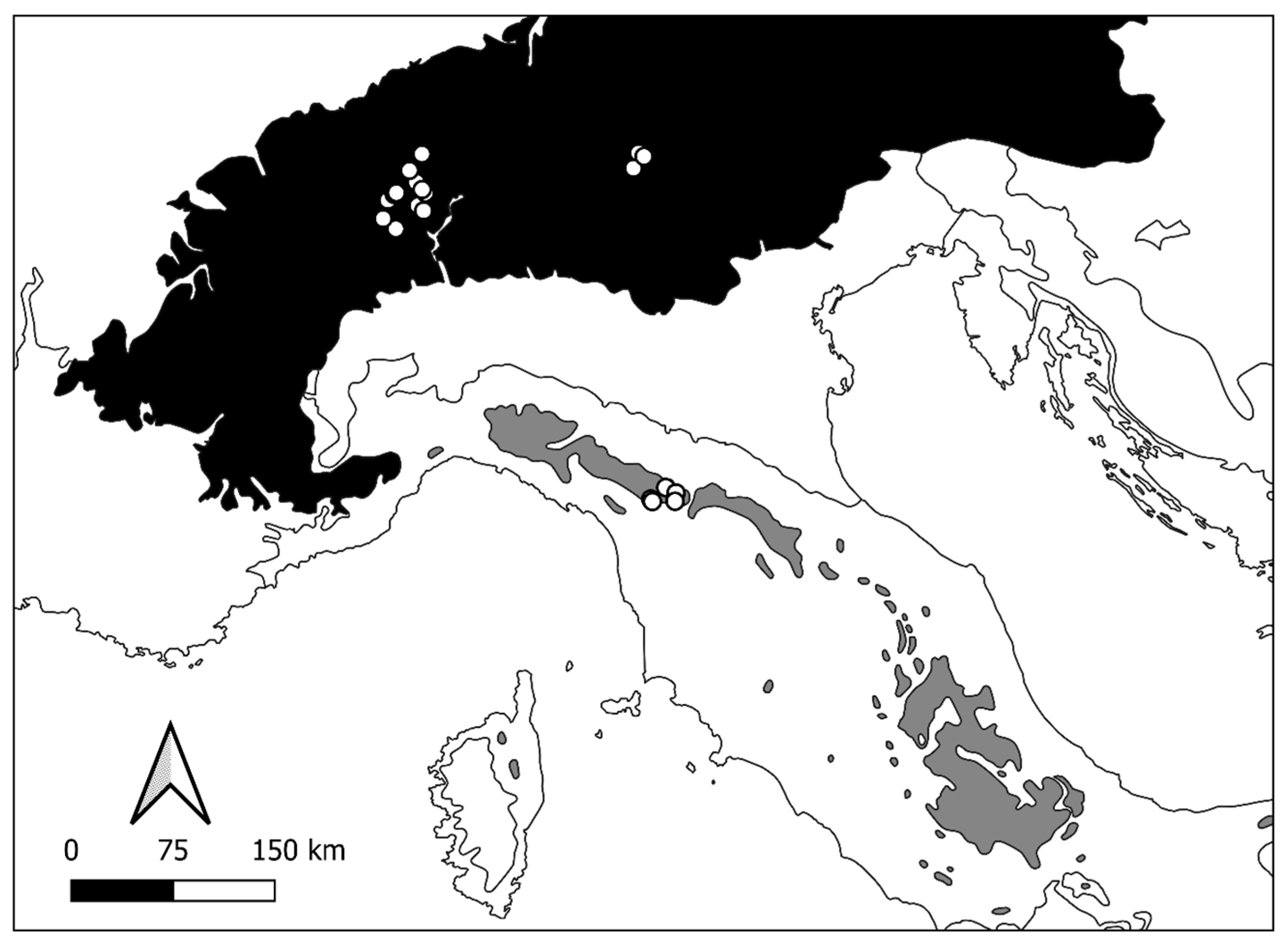
Sampling was performed by the National Research Council-Water Research Institute in the Ecoregion Alps on the Central–Western Alps (CW Alps—21 lakes), by the University of Modena and Reggio-Emilia, and by ARPA Environmental Agency on the Modenese Apennines (MA Apennines—6 lakes) (Figure 1).
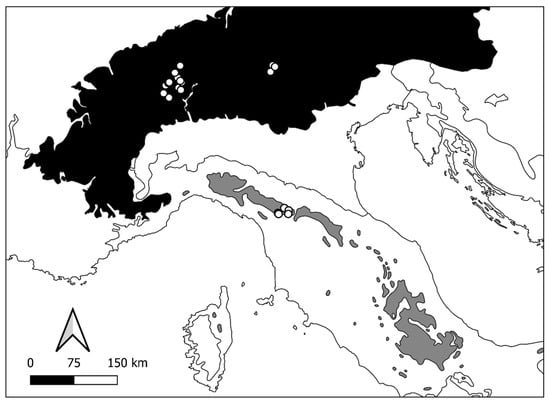
Figure 1.
Study area: location of Central–Western Alps (ecoregion “Alps, conifer and mixed forest” in black) and Modenese Apennines (ecoregion “Apennines, deciduous montane forest” in grey) where sampling was performed (modified by [33]). Solid line: separation among different ecoregions; white circles: sampled lakes.
The focus area was explicitly centred on natural lakes/ponds placed above 1000 m a.s.l. (from 1307 to 2857 m a.s.l.) and characterised by uncovered meadows, cliffs, and landslides on the CW Alps and by uncovered meadows and natural open forests on the MA Apennines. The choice fell on 27 lakes/ponds among those found to have a detailed description of the macroinvertebrate assemblages. The chosen lakes (Table S1: 21 lakes/ponds on CW Alps and 6 lakes/ponds on MA Apennines) have surface areas comprised between 0.001 and 0.078 km2, with maximum depths up to 16 m. The altitude ranges of lakes/ponds of the two mountain ranges are different: 1970–2857 m for the Alps and 1307–1780 m a.s.l. for the Apennines.
For major details on macroinvertebrates of Alpine lakes/ponds, see [15,18,34], and of the Apennines, see [31,35].
2.2. Bibliographic Search
The considered lakes/ponds have been studied since the 90s [11,14,28] and here we collated information on macroinvertebrate and chironomid assemblages derived from the above-mentioned publications covering the period (2000–2011). Only high-altitude natural lakes/ponds were considered, defined as surface standing waters characterised by areas >1 ha (0.01 km2) and maximum depths >1 m [36], placed in proximity of or beyond the tree line determined by tree-growth limiting environmental conditions [37].
The collated data set includes biological information about 170 taxa, considering the lowest possible taxonomic level. The data set was updated with currently accepted names following Fauna Europaea [38] (https://fauna-eu.org accessed on 10 July 2023), to derive a taxonomic accurate and reliable data set.
The sampling carried out during the period 2000–2011 followed a standardised sampling methodology for macroinvertebrates [39,40,41], taking samples from several littoral habitats (gravel, boulders, fine sediments, and within macrophite cover when present) through a D-shaped handle net with 250 µm of mesh aperture in each lake [15,18,31] to reach a representative picture of the lake assemblage. This quick methodology only takes 3 min to carry on each habitat. The external pressures and threats affecting lake littorals make it imperative to prevalently sample this area characterised by a more heterogeneous assemblage than the higher depths [22]. Samples were then sieved, fixed with 85% ethanol, and preserved for further laboratory analysis. Washing, sieving, removing debris, sorting, identifying, and counting by the use of a stereo- (80×) and a micro-scope (1000×) followed in the laboratory [40,41]. Main national [42] and international taxonomic guides [43,44] for chironomids and [45] for oligochaetes were used to identify organisms to the lowest practical taxonomic level (generally, genus or species). The data set underwent taxonomic validation and verification to ensure the accuracy and reliability of the data and to standardize it.
From the previously cited publications, we derived also morphometric, and geographic information on each lake. Physical–chemical information was obtained from CNR-IRSA Verbania and ARPA Emilia Romagna Environmental Agency, responsible for the chemical aspects of the respectively studied mountain lakes.
2.3. Statistical Approaches
To explore the key drivers of macroinvertebrates diversity and richness we derive the mean annual temperatures and the mean annual precipitation amount from the Worldclim 2.1 database [46] (www.worldclim.org accessed on 9 January 2023) with records from a 30-year period (1970–2000) and a spatial resolution of 30 s. We downloaded average monthly temperature and precipitation data and calculated the mean annual value per location aggregating those.
Principal component analysis (PCA) applied to physical–chemical, geographic, and morphometric characteristics of lakes/ponds was performed to explore the occurrence of variability between mountain ranges (CW Alps and MA Apennines) and trends of variability among lakes/ponds. Variables were shifted to be zero-centred and scaled to have unit variance before the analysis took place.
Two different diversity indices were applied: the specific diversity (species richness: α diversity) evaluated by counting the species found in each lake, and the Taxonomic Distinctness index (Δ+) [47], representing the averaged taxonomic distances among species in the community based on data consisting only of presence/absence of taxa from species lists belonging to the 27 lakes described earlier. The combination of these indices is helpful to provide a robust synthesis of taxonomic relationship models within a community assemblage. Ref. [47] found a tendency for these indices to be insensitive to sampling efforts, allowing the comparison of species lists belonging to the same faunal group of different sample sizes collected over different areas or times, and identified by different researchers, and to be related to environmental impact. The indices, applied both to the whole macroinvertebrate assemblage and to chironomids in particular, were evaluated through the “vegan” package (version 2.5-6) [48] in R project software (version 4.3.1) [49].
H1.
To evaluate differences in environmental characteristics and biodiversity indices among mountain range lakes/ponds, we used Kruskal–Wallis one-way ANOVA by ranks (α < 0.05) because normality and homogeneity of variance could not be achieved in most log-transformed data.
Immediately afterwards, a Non-metric Multi-dimensional Scaling (NMDS), an indirect gradient analysis approach that produces an ordination based on a Bray Curtis dissimilarity matrix, was performed using the “vegan” package to highlight differences in the macroinvertebrate assemblages between the two groups of lakes. Taxa present in less than 5% of the samples and consequently present in only one lake (rare taxa) were excluded from all the multivariate analyses to improve robustness.
H2.
Then, differences in assemblage composition between mountain range lakes/ponds were quantitatively explored within a permutational multivariate analysis of variance (PERMANOVA) via the adonis2 function. Significance was tested by 5000 Monte Carlo permutations, again within the “vegan” package.
H3.
Later, indicator species analysis (IndVal.g) [50] was employed to search for significant indicator taxa discriminating Apennine vs. Alpine lakes. IndVal combines the information on the concentration of species abundances in a group (A) and the degree of occurrence in that group (B). Ideal indicator species are those always present at sites in a given group and which never occur in other groups [51]. The indicator values range from 0 to 1, the value presented in this study corresponding to the square root of the product between A and B. The significance of the indicator values evaluated via the multipatt function was tested by 999 Monte Carlo permutations, where the observed indicator value was tested against those derived from randomised data, alpha was set to 0.05 and the Holm method was used to correct for multiple comparisons.
H4.
Lastly, the correlation among indices (richness and Distinctness, for the whole macroinvertebrate assemblage and for chironomids only) and seven main descriptors of each mountain range were examined using Spearman’s rank correlation coefficient. pH and conductivity were used as proxies of lithological and water chemical conditions; mean annual temperature and mean annual precipitation amount as proxies of climate conditions; lake maximum depth, surface area, and altitude as proxies of lake morphology and geography.
3. Results
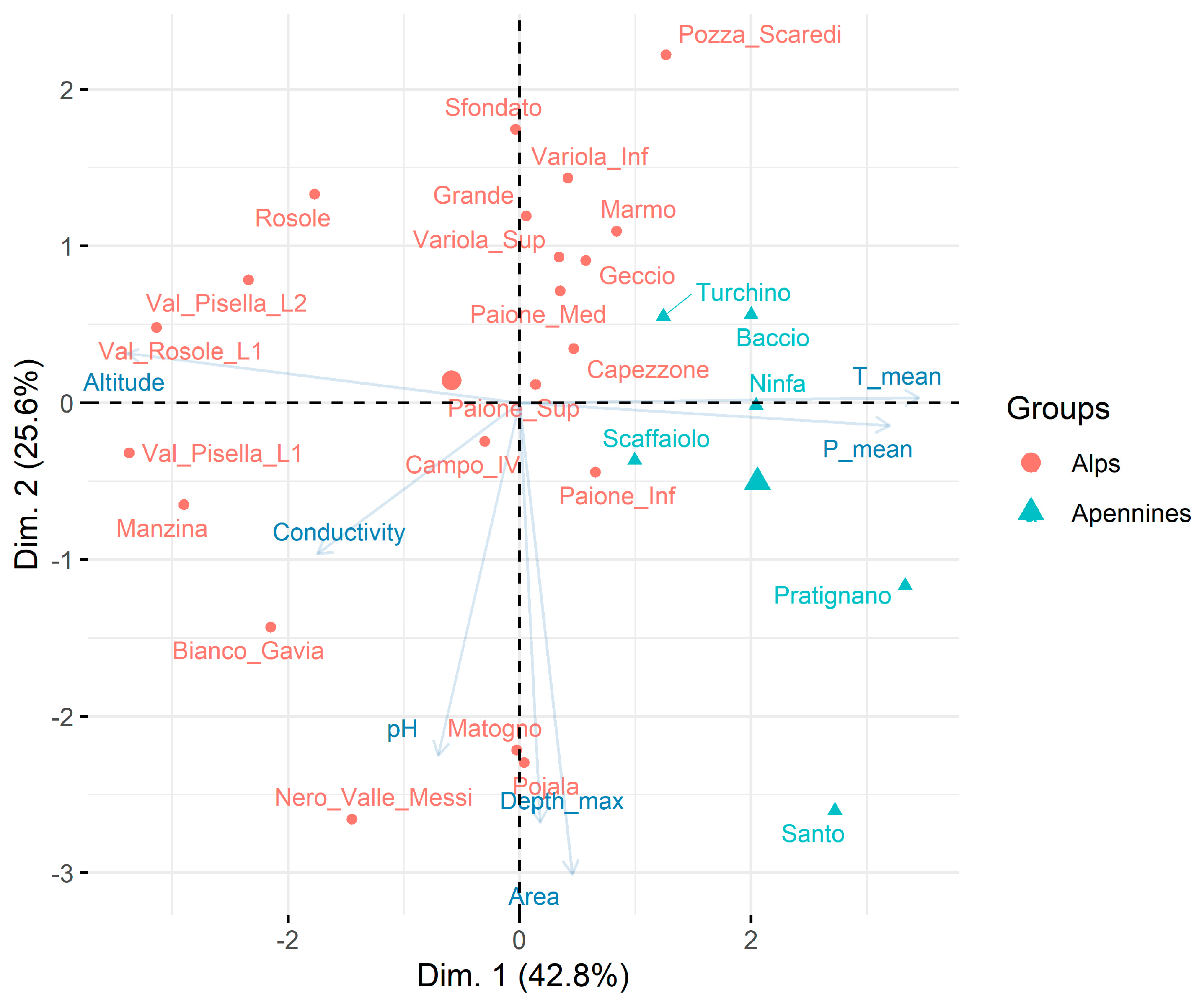
PCA applied to morphometric, geographic, and physical–chemical characteristics of lakes/ponds showed the separation of the two mountain groups (Figure 2). The first two axes explained 68.4% of the total variability. The first axis (42.8%) is negatively related to altitude and positively related to mean annual temperatures and precipitations. The second axis (25.6%) is negatively related to pH, maximum depth, and lake area.
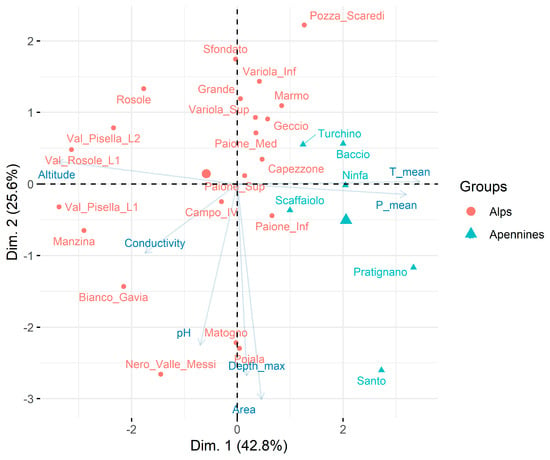
Figure 2.
PCA ordination of physical–chemical, geographic, and morphometric characteristics of the studied lakes/ponds.
3.1. Environmental Characterisation
In the second step, we applied different analyses to characterise the two regions. In particular, the Worldclim database allowed us to detail the climatic conditions to which lakes/ponds are subject in both regions. Mean monthly temperatures and mean monthly precipitations (Figure S1) showed that the study areas undergo differently distributed rainfall and temperature regimes.
From a geographic point of view (Figure S2a–e), box and whisker plots [52] showed the lake altitude, mean annual precipitation, and mean annual temperature distribution in the two mountain ranges. Altitudes and mean annual precipitations on the MA are non-symmetrically distributed with a central tendency to lower values than those found in the Alps for altitudes, and higher values for precipitations. The opposite is valid for mean annual temperatures on the CW Alps, which are lower than the Modenese Apennines, distributed more downward and near the zero-degree line (Figure S1). Mean annual precipitation and mean annual temperatures are significantly different between the two mountain ranges as highlighted in Figure S2b,c. In contrast to previous results, mean maximum depths and surface areas are not significantly different in the two study areas (Figure S2d,e), even if on the CW Alps a higher measure of dispersion is present for maximum depths and on the MA for surface lake areas.
Both mountain ranges showed a wide interval of pH values varying from 4.97 to 8.03 in the CW Alps and from 6.48 to 8.49 in MA lakes (Figure S2f). No significant differences were found for both pH and conductivity values between the two mountain ranges. It is worth noting that, in general, the highest pH and conductivity values were observed in the MA (Modenese Apennines) compared to the CW Alps (Central–Western Alps) (Table S1, Figure S2f,g). The MA region exhibited relatively high levels of mineralization in the waters, with conductivity ranging from 42 to 87 µS cm−1, primarily attributed to calcium and bicarbonate content. This mineralization accounts for the higher pH values observed in this particular mountain range. However, exceptions were found in lakes L1 Pisella, L1 Rosole, and Manzina in the CW Alps, where elevated conductivity and pH values were observed due to the presence of carbonatic rocks within a catchment area primarily characterised by acidic lithology.
3.2. Macroinvertebrate Assemblage Structure
The total amount of tagged taxa is 160, considering mainly species and genus levels, and families when organisms were damaged or not fully preserved (Table S2). We found 88 taxa in the Alps and 97 in the Apennines. The main taxonomic rank categories (7 classes, 18 orders, and 59 families) are differently represented on the two mountain ranges. Among them, 25 taxa (16%) are shared and widespread on both mountain ranges, 63 taxa (39%) are exclusive and characteristic of the CW Alps, and 72 taxa (45%) of the MA Apennines.
In general, lake littorals were insect dominated (78% of all taxa—Table S2): Diptera were the most abundant (52% of insects), followed by Trichoptera (13%) and Coleoptera (12%). Another well-represented taxonomic rank was oligochaetes with 13% of taxa.
A detailed picture (Table S2) of the macrofauna of the lakes on both mountain ranges showed similar percentages of the dominant groups (insects—75%, oligochaetes—17% in the CW Alps, and insects—75%, oligochaetes—12% in the MA). Among insects, the differences between the two mountain ranges are evident with a clear dominance of Diptera on the CW Alps (64%), and a less marked dominance on the MA (45%), flanked by the appearance of mayflies (10%) and a higher presence of dragonflies (8% vs. 3%). At a lower taxonomical rank, the distribution of families among Diptera showed a similar distribution, with 6 families registered on the CW Alps and 8 families on the MA Apennines (almost exclusively represented by chironomids—86% on the CW Alps, and 73% on the MA Apennines). Finally, focusing only on chironomids, the CW Alps were mainly represented by Orthocladiinae and the MA by Chironominae with similar relative abundances of taxa (around 60%). On the CW Alps, Chironominae held the second position with 19% of the abundances, while on MA, Tanypodinae and Orthocladiinae kept the same position almost equally (21% and 17% of the taxa).
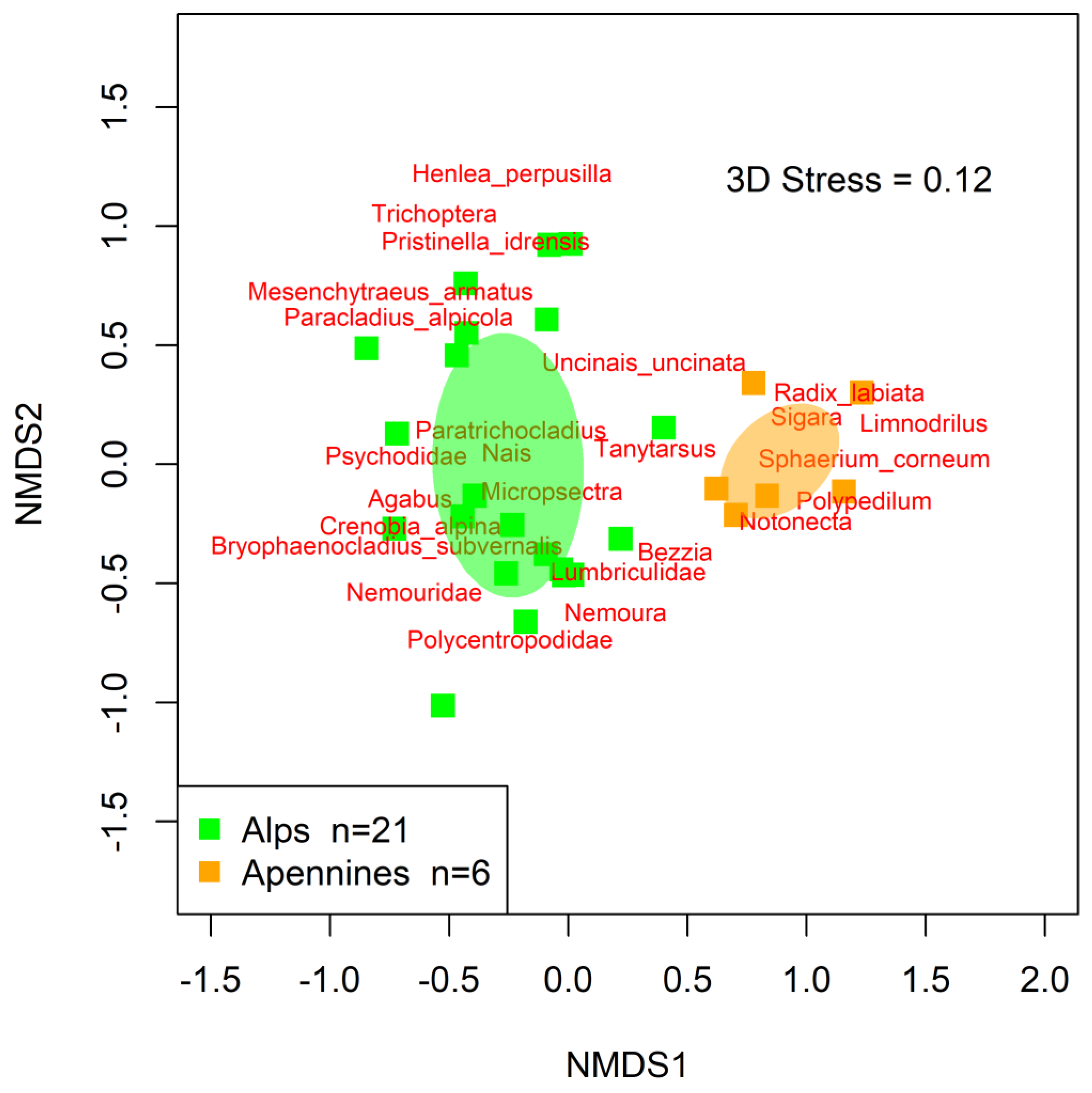
Differences in macroinvertebrate assemblages between the two mountain ranges (H2) were also emphasized by the results of NMDS, showing that macroinvertebrates inhabiting the two regions clustered separately (Figure 3). Such results were confirmed by PERMANOVA, which highlighted significant differences among the assemblage structures of the two regions (F = 4.82, p-value < 0.001, R2 = 0.16).
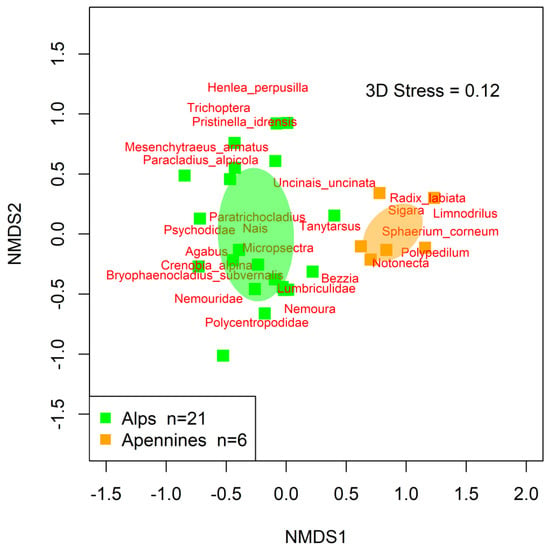
Figure 3.
NMDS ordination plot for high-altitude macroinvertebrate assemblages where the a priori identified lakes groups are colored. Shaded ellipses: 95% confidence interval surrounding the centroid of each lake group in the ordination space. Each square represents the overall macroinvertebrate assemblage at each lake. Macroinvertebrate taxa are positioned in the ordination space with red labels, as weighted averages.
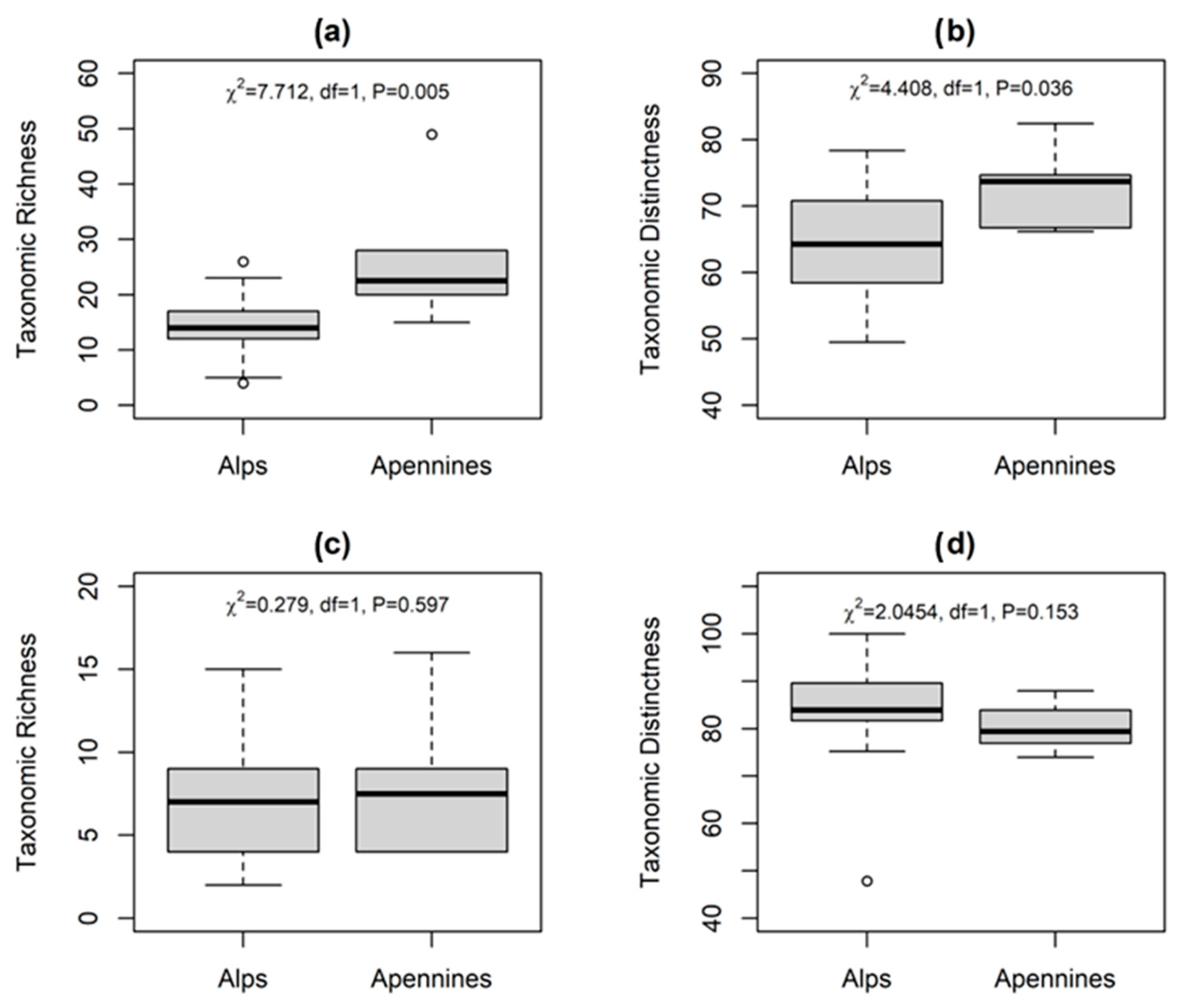
The application of different diversity indices (α and Δ+) on the macroinvertebrate assemblages highlighted that α-diversity (Figure 4a) was higher on the MA lakes, with a mean value of 26 ± 12 taxa per lake and a maximum of 49 taxa in Lake Pratignano, thus supporting our H1 hypothesis. On the contrary, CW lakes showed a mean value of 14 ± 6 taxa per lake with a minimum of 4 taxa in Lake Sfondato. In support of α-diversity values, the Taxonomic Distinctness (Δ+, Figure 4b) resulted higher on the MA lakes with a mean value of 72.9 ± 6.0 (distributed between 66.2 and 82.4), close to the mean CW lakes value of 64.9 ± 8.5, but where values were scattered between 49.5 and 78.4.
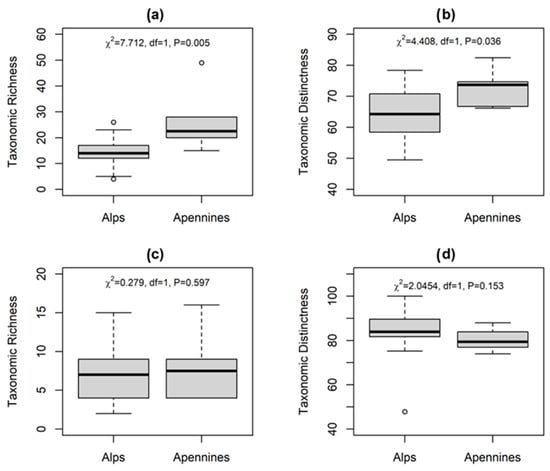
Figure 4.
Box plots of Taxonomic Richness and Distinctness for the whole macroinvertebrate assemblage (panels a,b) and for chironomids only (panels c,d) on the CW Alps and the MA Apennines. Statistical significance through Kruskal–Wallis test (p < 0.05) between categories is reported.
The application of the same diversity indices on chironomids led to different results (Figure 4c,d). The α-diversity (Figure 4c) was not significantly different in the two mountain ranges: on the MA lakes the mean value is 8.0 ± 4.4 taxa per lake and a maximum of 16 taxa was found in Lake Pratignano. Similarly, CW lakes showed a mean value of 7.0 ± 3.7 taxa per lake with a minimum of 2 taxa in Lake Val Rosole. Moreover, the Taxonomic Distinctness (Δ+) results were similar in the two mountain ranges with a mean value of 80.3 ± 5.0 on the MA lakes and 83.6 ± 10.2 on the CW lakes (Figure 4d).
Indicator species analysis did not highlight any macroinvertebrate taxa with high frequency to show significant differences between the two regions (H3). Only the chironomids Zavrelimyia sp. (A = 1.00, B = 0.62, IndVal = 0.79) and Heterotrissocladius marcidus (A = 1.00, B = 0.52, IndVal = 0.72) displayed greater affinities for CW Alpine lakes, while several taxa, spanning across several taxonomic rank levels, showed greater affinity for the MA lakes (Table S3). For species associated with MA lakes the “A” probability values were generally higher than “B”, suggesting these taxa are good indicators of this mountain group, even if not always present.
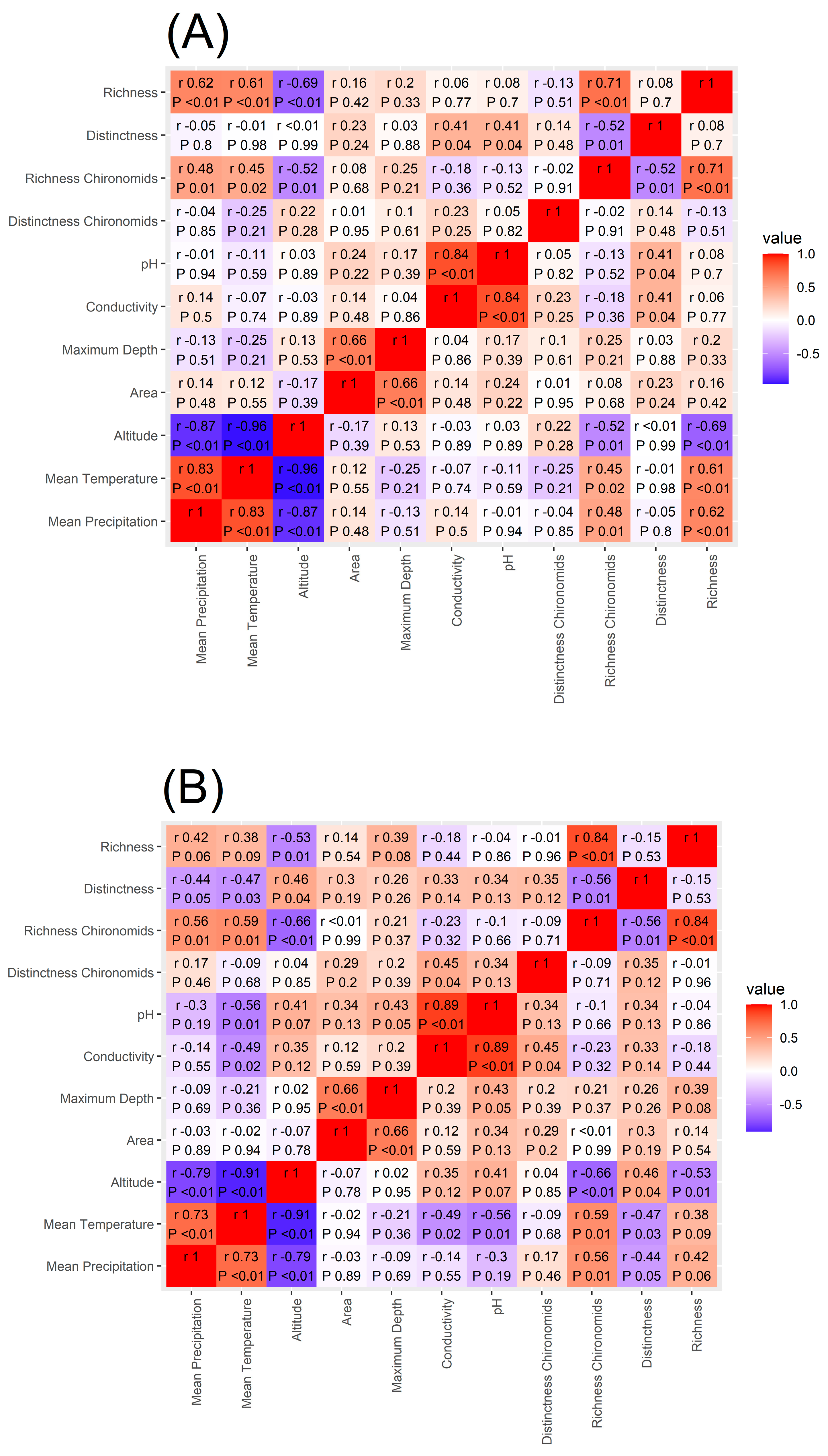
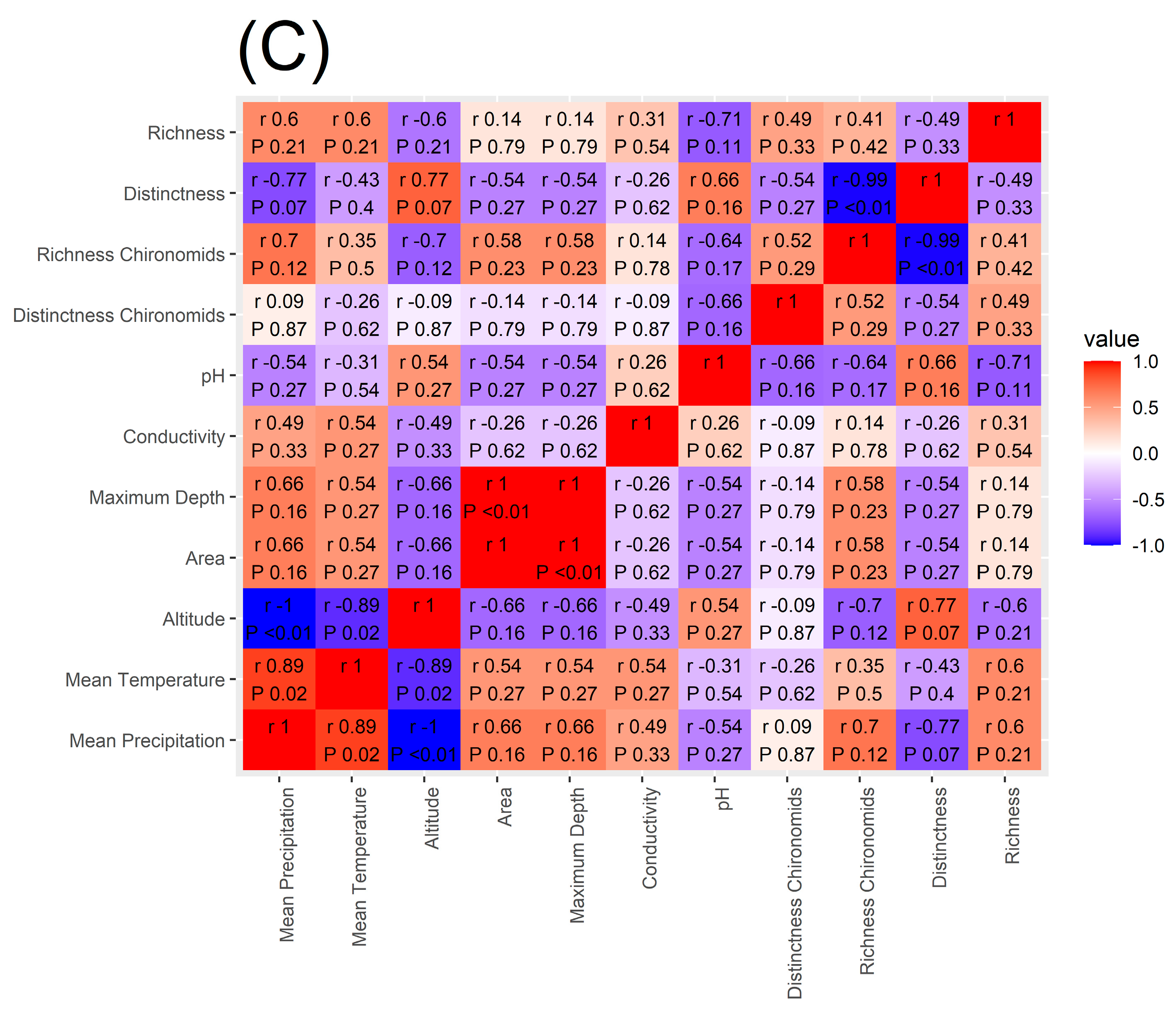
Spearman’s rank correlation coefficient applied to macroinvertebrate richness showed that the number of taxa present in each lake is significantly and positively correlated to mean annual precipitation and mean annual temperature while being negatively correlated to lake altitude (Figure 5) supporting our H4 hypothesis. Similar results were found for chironomid richness (Figure 5). On the other hand, macroinvertebrate Distinctness was not significantly correlated to any of the environmental variables investigated and chironomid Distinctness was significantly and positively correlated with conductivity.
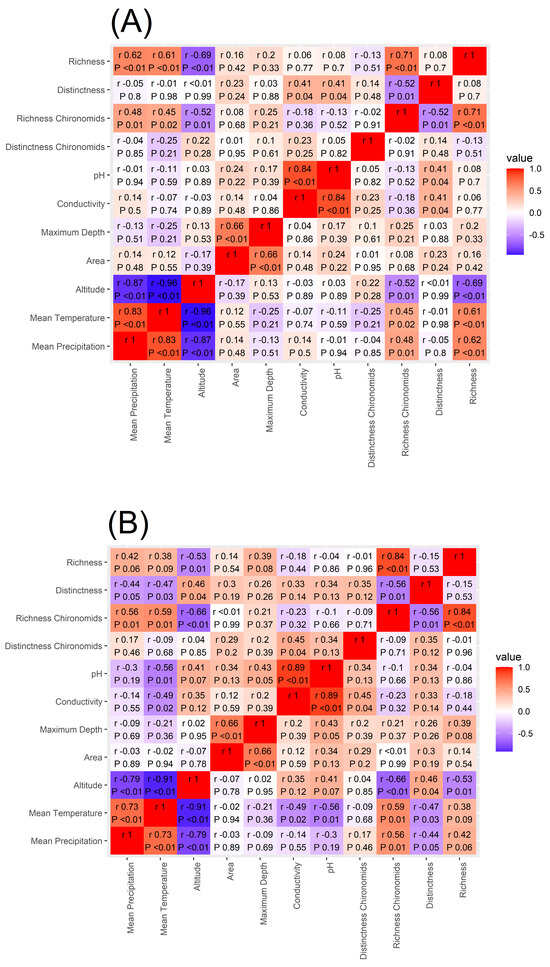
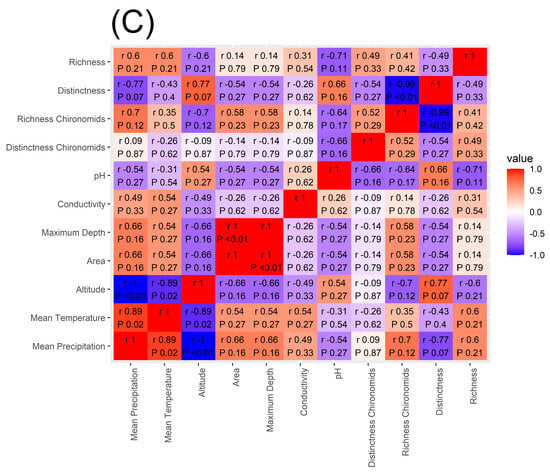
Figure 5.
Heatmap of the correlation between macroinvertebrate and chironomid richness and Distinctness, mean annual temperature and mean annual precipitation amount, altitude, lake area, maximum depth, conductivity, and pH. In panel (A), the Spearman’s correlation values are reported for the whole data set, in panel (B) for the CW Alpine lakes, and in panel (C) for the MA lakes.
Results allowed us to envisage possible future scenarios of these ecosystems considering the ongoing climate change and highlighted the importance of specific monitoring plans and active actions to preserve the maintenance of natural lake processes and the peculiar biodiversity high altitude lakes/ponds host, which reflect moderately clean and poorly disturbed water habitats.
4. Discussion
Due to climate change and human activities, high-altitude ecosystems are experiencing rapid changes including erosion, sediment flows, and surface area modification [9]. It is therefore pivotal a detailed study to assess their impacts on high altitude threatened freshwater ecosystems and their biota, since these lakes favour the local microclimate regulation and act as touristic attractions.
4.1. Environmental Characterisation
Although lake basins on the CW Alps present only small human disturbances, they are susceptible to long-range transport of atmospheric pollutants through air masses from the industrialized area of the Po Plain [53,54]. On the contrary, lake basins on the MA, placed at lower altitudes with gentler slopes, and less degraded grounds, show direct human impacts (e.g., unpaved roads, mountain huts, and a larger number of tourists) [55].
The PCA analysis reflects the differences between the two study areas, which are placed at different latitudes. Therefore, they undergo differently distributed rainfall and temperature regimes, with shorter freezing periods in the Apennines, while the Alpine area is influenced by the continental climate, defined by heavy rainfalls in early summer, and by dry and cold winters [5]. Their respective grounds are in very different stages of development depending on altitude, slope, and exposure.
In both mountain ranges, lakes/ponds are distributed away from the mountain ridges where gentler slopes are present, and water can stagnate for longer times [56]. Morphometry shows that, notwithstanding the Alps showed higher lake maximum depths, the surface lake areas are quite similar in between because of the very limited space characterising the high altitudes [20,57].
The water chemical aspects corroborate the different situations highlighted by the two study areas (ANOVA results): a wide interval of pH and conductivity values with higher values found in MA lakes/ponds due to highly mineralized waters, mainly represented by calcium and bicarbonates, and explaining the higher pH values and the lower acidification risk undergoing by this mountain range. As previously reported [58], differences in lake chemistry are explained by differences in the catchment mineral composition, mainly in the presence of weatherable minerals.
4.2. Macroinvertebrate Assemblage Structure
The geological and geographic position of Apennine lakes/ponds enables them to be inhabited by a greater number of macroinvertebrate taxa (higher diversity) than the lakes/ponds in the Alps, confirming their importance for conservation purposes and supporting our first hypothesis (H1). Moreover, NMDS and PERMANOVA analyses corroborate the findings of highly and significantly different macroinvertebrate assemblage structures in the two regions (H2).
Comparing the results of MA Apennine lakes/ponds with other European lake ecosystems [12,21,59,60,61,62,63,64,65] or with high-altitude systems in the Alps (CW Alps), the MA Apennine lakes/ponds are extremely rich in taxa, comparable only with outcomes on the Pyrenees [11], and in contrast with the Alpine ecosystems.
The percentage distribution of different taxa, with a preponderance of insects, reflects the usual distribution at these altitudes [18,22,31,34,35] but the lower altitude lakes (MA) also showed mayflies (10%) and dragonflies (8%) more typical of the area [31,35].
The main differences between the two areas are attributed to Diptera chironomids which represented the dominant group, but with differently distributed subfamilies: Orthocladiinae are the main taxa group on the CW Alps, and Chironominae on the MA as was deeply discussed by [34] suggesting the former are cold adapted and mostly scrapers feeding on periphyton, while Chironominae dominate in lowland still waters where higher temperatures and the abundance of detritus and algae increased. Hence, food supply changes from alpine (uncovered meadows, cliffs, and landslides) to lowland (forested) areas, and it was essential in determining, together with temperature and oxygen content, the altitudinal pattern of chironomid assemblage structure diversity [22,65,66].
Indicator species analysis does not allow us to identify taxa significantly associated with CW Alpine vs. MA Apennine lakes (H3). Those results are probably driven by the relatively small sample size for Apennines lakes, therefore, in our opinion, more lakes should be sampled to identify the specific taxa responsible for the discrimination observed between the two mountain ranges. Although not statistically significant, some patterns can be observed: CW Alpine lakes are associated only with the presence of Zavrelimyia sp. (Chironomidae Tanypodinae), one of the most frequent genera all over the Alps and present frequently >2000 m a.s.l. and at pH < 7 [34]. On MA lakes, the analysis highlights several taxonomic rank levels of macroinvertebrate taxa linked to the area. Among them, Helobdella stagnalis (Hirudinea), Homochaeta spp. (Oligochaeta), Cladotanytarsus spp., and Cladopelma sp. (Chironomidae) are more frequently found in warmer, vegetated lakes/ponds inhabited by a prey-rich fauna [22,67,68]. MA lakes are also represented by still waters adapted species, like Caenis horaria (Ephemeroptera), acknowledged to be very resistant also to eutrophic conditions [69] characterising most likely lower altitude lakes/ponds, and Anax imperator (Odonata) whose occurrence is proven to be influenced by larger lakes/ponds size [70]. Oligochaeta Homochaeta spp. and Pristina bilobata are frequently found in well-oxygenated oligo-saprobic mountain waters [71] or on the bottom of ponds, lake littorals and slowly running waters [72]. Gastropoda and Bivalvia occur with Radix peregra and Sphaerium corneum, very common molluscs usually found both in the mountain and the lower altitude lakes/ponds [73], characterised by higher values of pH and alkalinity, being the latter also tolerant to hypoxia [74].
Altitude, often viewed as a proxy of temperature and length of ice-free season, is considered one of the main drivers of Alpine lakes/ponds’ macroinvertebrate richness [12,18,70,75]. We found a correlation (altitude, temperature, and several biodiversity metrics) in our analyses, supporting our last hypothesis (H4). Macroinvertebrate assemblages are shown to become less diverse with altitude (and decreasing temperature), leading to a species richness decrease with higher elevation (and lower temperature) in line with previous studies [14,58,75]. The Distinctness of macroinvertebrates as a whole is greater in the MA lakes where richness is higher, and the taxa present are less specialized to high-altitude habitats. We could not discriminate between altitude and mean annual temperature influence on biodiversity, since these parameters are highly correlated in our study sites.
Focusing on chironomids, on the contrary, our analyses reveal that richness, as well as Distinctness, are generally higher at higher altitudes or in CW Alpine lakes, confirming that chironomids taxa present in mountain areas are often well adapted to the harsh environmental conditions prevailing in Alpine lakes/ponds where they frequently dominate. Similar effects were found by [75] on cold stenotherm species richness of Dytiscidae along an altitudinal gradient in the Swiss Alps.
Mean annual precipitation showed strong correlations with all the biodiversity metrics considered. Higher water availability is thus associated with higher richness both considering macroinvertebrates as a whole and chironomids only. Chironomid Distinctness is also linked to an increase in mean annual precipitation, whereas the Distinctness of macroinvertebrates as a whole exhibits an opposite trend. The increase in macroinvertebrate Distinctness in relatively dry habitats suggests that the average taxonomic distance among those taxa increases, while the total number of taxa decreases. This pattern could be attributed to the adaptation of a few species to extremely dry conditions or the representativeness of high-level groups (e.g., Heteroptera and Coleoptera) constituted by only a small number of taxa. Chironomids confirmed to represent a pivotal component of high-altitude lake macroinvertebrate assemblages with patterns that can differ from that of the macroinvertebrates as a whole. This aspect needs to be further investigated and a detailed taxonomic study is essential to properly assess the conservation status of these environments. A confused taxonomy, based on doubtful and problematic species, makes the conservation and management of threatened species and their environment weaker and more problematic.
Even if more data are needed to properly define the relative importance of climatic variables, and, consequently, predict the effect of future climate change scenarios, our results highlighted the close relationship between high-altitude lakes/ponds macroinvertebrates and the climatic conditions of the area they inhabit.
At high altitudes, species are frequently obliged univoltine, cold steno-thermic, showing adaptation to the long winter period and ice cover, though [76] justifies the exclusive occurrence of some chironomid species at these altitudes due to the cold stenothermy of larval stages. Another important factor impacting macroinvertebrates as a whole is the very low colonization rate in remote areas, accounting for low species diversity. In general, dispersal and colonization rates condition the arrival of a species in a mountain lake, while its abiotic and biotic features (i.e., availability of diversified habitats, water chemistry, temperature fluctuations, and biological interactions) determine their survival, reproduction, and dispersion ability. Therefore, the combined effect of diversity decline and abundance increase in single species with altitude demonstrates the well-known phenomenon of an impact shift from biotic to abiotic conditions, e.g., [77,78,79]. Even if the colonization of an alpine ecosystem is in some cases stochastic, most of the colonizing species are recruited from taxa pools in nearby water bodies. Thus, differences in species composition are due to biogeographic factors, such as the presence of diversified environmental gradients among high-altitude lake valleys or catchments, and to the historical evolution of single areas influenced differently by the presence of glaciers and their retreat.
5. Conclusions
In this study, we gathered all the available information about the macroinvertebrate fauna of two mountain ranges in Italy. Strong differences were highlighted in the environmental conditions and the associated fauna both within and between the two groups of lakes. These results showed the importance of biodiversity research on those ecosystems that host peculiar assemblages. Long-term studies are essential to properly understand the link between climate habitat conditions and biodiversity. This is crucial, but observational studies or surveys are hardly financed on priority water bodies such as small lakes/ponds. These environments host peculiar fauna and flora and are thus considered more vulnerable to climate changes and human impacts than lowland lakes.
In conclusion, the impact of climate change on macroinvertebrates may be even more detrimental for smaller-sized lakes/ponds flora and fauna, as they are highly susceptible to climatic changes. Therefore, there is an urgent need to promote the protection, preservation, and naturalistic enhancement of the many fragile and hidden lakes/ponds currently not covered by any legislation. Given their importance as essential biodiversity sources for lower altitude areas, their role in supporting the local biodiversity, and their function as regional meteorological regulators, these ecosystems hold an inestimable socio-economic–environmental value.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/w15213814/s1, Figure S1: mean monthly temperatures (blue lines) and mean monthly precipitations (boxplots) for the CW Alps (left) and for the MA (right); Figure S2: box plots of selected geographic and morphometric characteristic for the studied Italian mountain ranges (CW Alps and MA Apennines). Statistical significance through Kruskal–Wallis test between categories is reported; Table S1: list of lakes studied distributed on the two Italian mountain chains. Columns represent region where the lakes are, river basin, lake name, main geology, year of sampling, latitude and longitude (WGS 84), altitude (m a.s.l.), maximum depth (m), lake area (km2), water temperature (°C), conductivity (μS cm−1), and pH; Table S2: presence/absence matrix of lakes macroinvertebrates in the two Italian mountain chains. Columns represent lake name, region where the lakes are and presence (1) or absence (0) for all the targeted taxa. Table S3: Supplementary Table. IndVal analysis for Alpine and Apennines sites. A: probability that the surveyed site belongs to the target site group given the fact that the taxon has been found; B: probability of finding the taxon in sites belonging to the site group. All the reported associations were not significant after 999 permutations at α = 0.05 and the Holm correction for multiple comparisons.
Author Contributions
Conceptualization, A.B., G.C. and R.F.; methodology, A.B., G.C., S.Z., I.A., D.P., M.R. and R.F.; software, R.F.; validation, A.B., G.C., S.Z., I.A., D.P., M.R. and R.F.; formal analysis, G.C. and R.F.; investigation, S.Z. and M.R.; resources, A.B. and D.P.; data curation, G.C., S.Z. and R.F.; writing—original draft preparation, A.B., G.C. and R.F.; writing—review and editing, A.B., G.C., S.Z., I.A., D.P., M.R. and R.F.; visualization, R.F.; supervision, A.B. and D.P.; project administration, A.B. and D.P.; funding acquisition, A.B. and D.P. All authors have read and agreed to the published version of the manuscript.
Funding
The authors have no relevant financial or non-financial interests to disclose.
Data Availability Statement
All data generated or analysed during this study are included in this published article, and its Supplementary Information is available in Excel and Word files.
Acknowledgments
Thanks are due to researchers, technicians, and students helping from time to time in field campaigns and chemical laboratory analysis. Thanks are due to Hamerlík L. (Comenius University, Bratislava, Slovakia) for comments and input on the draft manuscript.
Conflicts of Interest
The authors declare that they have no conflict of interest relevant to the content of this article.
References
- Kernan, M.; Catalan, J.F.; Ventura, M.; Curtis, C.J. A biological survey of high mountain and high latitude lakes across Europe: Aims, sampling strategy, methods and main achievements. Adv. Limnol. 2009, 62, 3–16. [Google Scholar] [CrossRef]
- Battarbee, R.V. Mountain lakes, pristine o polluted? Limnetica 2005, 24, 1–8. [Google Scholar] [CrossRef]
- Battarbee, R.V.; Patrick, S.; Kernan, M.; Psenner, R.; Thies, H.; Grimalt, J.; Rosseland, B.O.; Wathne, B.; Catalan, J.; Mosello, R.; et al. High Mountain Lakes and Atmospherically Transported Pollutants. In Global Change and Mountain Regions: An Overview of Current Knowledge; Huber, U.M., Bugmann, H.K.M., Reasoner, M.A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 113–121. [Google Scholar]
- Machate, O.; Schmeller, D.S.; Schulze, T.; Brack, W. Review: Mountain lakes as freshwater resources at risk from chemical pollution. Environ. Sci. Eur. 2023, 35, 3. [Google Scholar] [CrossRef]
- Rogora, M.; Frate, L.; Carranza, M.L.; Freppaz, M.; Stanisci, A.; Bertani, I.; Bottarin, R.; Brambilla, A.; Canullo, R.; Carbognani, M.; et al. Assessment of climate change effects on mountain ecosystems through a cross-site analysis in the Alps and Apennines. Sci. Total Environ. 2018, 624, 429–1442. [Google Scholar] [CrossRef]
- Sutela, T.; Aroviita, J.; Keto, A. Assessing ecological status of regulated lakes with littoral macrophyte, macroinvertebrate and fish assemblages. Ecol. Indic. 2013, 24, 185–192. [Google Scholar] [CrossRef]
- Caruso, B.S.; King, R.; Newton, S.; Zammit, C. Simulation of Climate Change Effects on Hydropower Operations in Mountain Headwater Lakes, New Zealand. River Res. Appl. 2017, 33, 147–161. [Google Scholar] [CrossRef]
- Thompson, R.; Kamenik, C.; Schmidt, R. Ultra-sensitive Alpine lakes and climate change. J. Limnol. 2005, 64, 139–152. [Google Scholar] [CrossRef]
- Salerno, F.; Gambelli, S.; Viviano, G.; Thakuri, S.; Guyennon, N.; D’Agata, C.; Diolaiuti, G.; Smiraglia, C.; Stefani, F.; Bocchiola, D.; et al. High alpine ponds shift upwards as average temperatures increase: A case study of the Ortles-Cevedale mountain group (Southern Alps, Italy) over the last 50 years. Glob. Planet Chang. 2014, 120, 81–91. [Google Scholar] [CrossRef]
- Sadro, S.; Melack, J.M.; Sickman, J.O.; Skeen, K. Climate warming response of mountain lakes affected by variations in snow. Limnol. Oceanogr. Lett. 2019, 4, 9–17. [Google Scholar] [CrossRef]
- Fjellheim, A.; Boggero, A.; Nocentini, A.M.; Rieradevall, M.; Raddum, G.G.; Schnell, Ø.A. Distribution of benthic invertebrates in relation to environmental factors—A study of European remote alpine lake ecosystems. Verh. Int. Ver. Limnol. 2000, 27, 484–488. [Google Scholar] [CrossRef]
- Fjellheim, A.; Raddum, G.G.; Vandvik, V.; Cogalniceanu, D.; Bitusik, P.; Boggero, A.; Brancelj, A.; Dumnicka, E.; Galas, J.; Gâldean, N.; et al. Diversity and distribution patterns of benthic invertebrates along alpine gradients. A study of remote European freshwater lakes. Adv. Limnol. 2009, 62, 167–190. [Google Scholar] [CrossRef]
- Carlson, B.Z.; Corona, M.C.; Dentant, C.; Bonet, R.; Thuiller, W.; Choler, P. Observed long-term greening of alpine vegetation—A case study in the French Alps. Environ. Res. Lett. 2017, 12, 114017. [Google Scholar] [CrossRef]
- Gobiet, A.; Kotlarski, S.; Beniston, M.; Heinrich, G.; Rajczak, J.; Stoffel, M. 21st century climate change in the European Alps—A review. Sci. Total Environ. 2014, 493, 1138–1151. [Google Scholar] [CrossRef]
- Boggero, A. Macroinvertebrates of Italian mountain lakes: A review. Redia J. Zool. 2018, 101, 35–45. [Google Scholar] [CrossRef]
- Allan, J.D.; Castillo, M.M. Stream Ecology: Structure and Function of Running Waters; Springer: Dordrecht, The Netherlands, 2007; pp. 135–161. [Google Scholar]
- Reice, S.R.; Wohlenberg, M. Monitoring freshwater benthic macroinvertebrates and benthic processes: Measures for assessment of ecosystem health. In Freshwater Biomonitoring and Benthic Macroinvertebrates; Rosenberg, D.M., Resh, V.H., Eds.; Chapman and Hall: New York, NY, USA, 1992; pp. 287–305. [Google Scholar]
- Boggero, A.; Zaupa, S.; Musazzi, S.; Rogora, M.; Dumnicka, E.; Lami, A. Environmental factors as drivers for macroinvertebrate and diatom diversity in Alpine lakes: New insights from the Stelvio National Park (Italy). J. Limnol. 2019, 78, 147–162. [Google Scholar] [CrossRef]
- Armitage, P.D.; Pardo, I. Impact assessment of regulation at the reach level using macroinvertebrate information from mesohabitats. Regul. Rivers Res. Manag. 1995, 10, 147–158. [Google Scholar] [CrossRef]
- Catalán, J.; Camarero, L.; Felip, M.; Pla, S.; Ventura, M.; Buchaca, T.; Bartumeus, F.; de Mendoza, G.; Miró, A.; Casamayor, E.O.; et al. High mountain lakes: Extreme habitats and witnesses of environmental changes. Limnetica 2006, 25, 551–584. [Google Scholar] [CrossRef]
- Catalán, J.; Curtis, C.; Kernan, M. Remote European mountain lake ecosystems: Regionalisation and ecological status. Freshw. Biol. 2009, 54, 2419–2432. [Google Scholar] [CrossRef]
- Füreder, L.; Ettinger, R.; Boggero, A.; Thaler, B.; Thies, H. Macroinvertebrate Diversity in Alpine Lakes: Effects of Altitude and Catchment Properties. Hydrobiologia 2006, 562, 123–144. [Google Scholar] [CrossRef]
- Marcheggiani, S.; Cesarini, G.; Puccinelli, C.; Chiudioni, F.; Mancini, L.; Angelici, C.; Martinoli, M.; Tancioni, L. An Italian local study on assessment of the ecological and human impact of water abstraction. Microchem. J. 2019, 149, 104016. [Google Scholar] [CrossRef]
- Morales-Molino, C.; Steffen, M.; Samartin, S.; van Leeuwen, J.F.N.; Hürlimann, D.; Vescovi, E.; Tinner, W. Long-Term Responses of Mediterranean Mountain Forests to Climate Change, Fire and Human Activities in the Northern Apennines (Italy). Ecosystems 2021, 24, 1361–1377. [Google Scholar] [CrossRef]
- Di Giorgio, M.; Zuppa, A.M. Macrobenthos dei laghetti di quota del Gran Sasso e del tratto sorgentizio del torrente Nora Appennino abruzzese, nota preliminare. In Monitoraggio Biologico Del Gran Sasso; Cicolani, B., Ed.; Consorzio di ricerca Gran Sasso: Teramo, Italy, 1996; pp. 284–289. [Google Scholar]
- Ruggiero, A.; Solimini, A.G.; Mutschlechner, A.; Anello, M.; Carchini, G. Aspetti limnologici del Lago Racollo (1568 m s.l.m.), Campo Imperatore (AQ). Studi Trentini Di Sci. Nat. Acta Biol. 2001, 78, 173–180. [Google Scholar]
- Ruggiero, A.; Solimini, A.G.; Carchini, G. Limnological aspects of an Apennine shallow lake. Ann. Limnol. Int. J. Lim. 2004, 40, 89–99. [Google Scholar] [CrossRef]
- Osella, G.; Pannunzio, G. Macrobenthos dei laghetti del Gran Sasso d’Italia. Quad. Mus. Civ. Di Stor. Nat. Ferrara 2013, 1, 57–67. [Google Scholar]
- Khamis, K.; Hannah, D.M.; Clarvis, M.H.; Brown, L.E.; Castella, E.; Milner, A.M. Alpine aquatic ecosystem conservation policy in a changing climate. Environ. Sci. Policy 2014, 43, 39–55. [Google Scholar] [CrossRef]
- Velle, G.; Bodin, C.L.; Arle, J.; Austnes, K.; Boggero, A.; Bojkova, J.; Fornaroli, R.; Fölster, J.; Goedkoop, W.; Jones, I.; et al. Responses of Benthic Invertebrates to Chemical Recovery from Acidification; NIVA report SNO 7881-2023. ICP Waters report 153/2023; NIVA: Bergen, Norway, 2023. [Google Scholar]
- Ansaloni, I.; Prevedelli, D.; Ruocco, M.; Simonini, R. Checklist of benthic macroinvertebrates of the Lago Pratignano (northern Apennines, Italy): An extremely rich ecosystem. Check List 2016, 12, 1821. [Google Scholar] [CrossRef]
- Nelson, S.J.; Hovel, R.A.; Daly, J.; Gavin, A.; Dykema, S.; McDowell, W.H. Northeastern mountain ponds as sentinels of change: Current and emerging research and monitoring in the context of shifting chemistry and climate interactions. Atmos. Environ. 2021, 264, 118694. [Google Scholar] [CrossRef]
- European Environment Agency. Europe’s Environment: The Third Assessment; Environmental assessment report 10; EEA: Luxembourg, 2003. [Google Scholar]
- Boggero, A.; Füreder, L.; Lencioni, V.; Simcic, T.; Thaler, B.; Ferrarese, U.; Lotter, A.F.; Ettinger, R. Littoral Chironomid communities of Alpine lakes in relation to environmental factors. Hydrobiologia 2006, 562, 145–165. [Google Scholar] [CrossRef]
- Ansaloni, I.; Benassi, A.; Manzieri, A.M.; Ruocco, M.; Sala, L.; Tintorri, A. Il Lago della Ninfa (Appennino modenese): Comunità macrozoobentonica, fauna vertebrata e considerazioni ecologiche. Atti Soc. Nat. Mat. Modena 2015, 146, 249–262. [Google Scholar]
- British Standards Institution. Guidance Standard on Assessing the Hydromorphological Features of Lakes; BS EN 16039:2011 Water quality; BSI: London, UK, 2011. [Google Scholar]
- Körner, C. A re-assessment of high elevation treeline positions and their explanation. Oecologia 1998, 115, 445–459. [Google Scholar] [CrossRef]
- de Jong, Y. Fauna Europaea. Fauna Europaea Consortium. 2016. Available online: https://www.gbif.org/dataset/90d9e8a6-0ce1-472d-b682-3451095dbc5a (accessed on 14 September 2023).
- NIVA. International Cooperative Programme for Assessment and Monitoring of Acidification of Rivers and Lakes: Programme Manual; Programme Centre, NIVA: Oslo, Norway, 1987. [Google Scholar]
- ICP Waters Programme Centre. Programme Manual; NIVA report SNO 3547-96; Programme Centre, NIVA: Oslo, Norway, 1996. [Google Scholar]
- ICP Waters Programme Centre. ICP Waters Programme Manual 2010; NIVA SNO 6074-2010. ICP Waters report 105/2010; Programme Centre, NIVA: Oslo, Norway, 2010. [Google Scholar]
- AA.VV. Guide Per il Riconoscimento Delle Specie Animali delle Acque Interne Italiane; CNR Progetto Finalizzato Promozione della qualità dell’ambiente: Verona, Italy; pp. 1977–1985.
- Andersen, T.; Cranston, P.S.; Epler, J.H. (Eds.) Chironomidae of the Holarctic Region: Keys and Diagnoses; Part 1—Larvae; Series Insects Systematics and Evolution Supplement 66; Scandinavian Entomology LTD: Lund, Sweden, 2013. [Google Scholar]
- Wiederholm, T. (Ed.) Chironomidae of the Holartic region Keys and Diagnoses Part II: Pupae; Entomologica Scandinavica; Btj Datfailm: Lund, Sweden, 1986. [Google Scholar]
- Timm, T. A Guide to the Freshwater Oligochaeta and Polychaeta of Northern and Central Europe. Lauterbornia 2009, 66, 1–235. [Google Scholar]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, M.R. A taxonomic distinctness index and its statistical properties. J. Appl. Ecol. 1998, 35, 523–531. [Google Scholar] [CrossRef]
- Oksanen, J.; Friendly, F.G.B.M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; et al. Vegan: Community Ecology Package, R Package Version 2.5-6. 2019. Available online: https://CRAN.R-Project.Org/Package=vegan (accessed on 7 July 2023).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- De Cáceres, M.; Legendre, P. Associations between Species and Groups of Sites: Indices and Statistical Inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef]
- Dufrěne, M.; Legendre, P. Species Assemblages and Indicator Species: The Need for a Flexible Asymmetrical Approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- Tukey, J.W. Exploratory Data Analysis. In The Concise Encyclopedia of Statistics; Springer: New York, NY, USA, 1977. [Google Scholar] [CrossRef]
- Rogora, M.; Colombo, L.; Lepori, F.; Marchetto, A.; Steingruber, S.; Tornimbeni, O. Thirty years of chemical changes in alpine acid-sensitive lakes in the Alps. Wat. Air Soil Poll. 2013, 224, 1746. [Google Scholar] [CrossRef]
- Poma, G.; Salerno, F.; Roscioli, C.; Novati, S.; Guzzella, L. Persistent organic pollutants in sediments of high-altitude Alpine ponds within Stelvio National Park, Italian Alps. Inland Waters 2017, 7, 34–44. [Google Scholar] [CrossRef]
- Fott, J. (Ed.) Limnology of mountain lakes. In Developments in Hydrobiology; Springer: Dordrecht, The Netherlands, 1994; Volume 93. [Google Scholar]
- Mosquera, P.V.; Hampel, H.; Vazquez, R.F.; Alonso, M.; Catalan, J. Abundance and morphometry changes across the high-mountain lake-size gradient in the tropical Andes of Southern Ecuador. Water Resour. Res. 2017, 53, 7269–7280. [Google Scholar] [CrossRef]
- Seekell, D.A.; Pace, M.L.; Tranvik, L.J.; Verpoorter, C. A fractal-based approach to lake size-distributions. Geophys. Res. Lett. 2013, 40, 517–521. [Google Scholar] [CrossRef]
- Kamenik, C.; Schmidt, R.; Kum, G.; Psenner, R. The Influence of Catchment Characteristics on the Water Chemistry of Mountain Lakes. Arct Antarct Alp. Res. 2001, 33, 404–409. [Google Scholar] [CrossRef]
- Boix, D.; Sala, J.; Moreno-Amich, R. The faunal composition of Espolla Pond (NE Iberian Peninsula): The neglected biodiversity of temporary waters. Wetlands 2001, 21, 577–592. [Google Scholar] [CrossRef]
- Sahuquillo, M.; Poquet, J.M.; Rueda, J.; Miracle, M.R. Macroinvertebrate communities in sediment and plants in coastal Mediterranean water bodies (central Iberian Peninsula). Ann. Limnol. Int. J. Lim. 2007, 43, 11–30. [Google Scholar] [CrossRef]
- Oertli, B.; Indermuehle, N.; Angélibert, S.; Hinden, H.; Stoll, A. Macroinvertebrate assemblages in 25 high Alpine ponds of the Swiss National Park (Cirque of Macun) and relation to environmental variables. Hydrobiologia 2008, 597, 29–41. [Google Scholar] [CrossRef]
- Céréghino, R.; Oertli, B.; Bazzanti, M.; Coccia, C.; Compin, A.; Biggs, J.; Bressi, N.; Grillas, P.; Hull, A.; Kalettka, T.; et al. Biological traits of European pond macroinvertebrates. Hydrobiologia 2012, 689, 51–61. [Google Scholar] [CrossRef]
- Guareschi, S.; Gutiérrez-Cánovas, C.; Picazo, F.; Sánchez-Fernández, D.; Abellán, P.; Velasco, J.; Millán, A. Aquatic Macroinvertebrate Biodiversity: Patterns and Surrogates in Mountainous Spanish National Parks. Aquat. Conserv. 2012, 22, 598–615. [Google Scholar] [CrossRef]
- Novikmec, M.; Veselská, M.; Bitušík, P.; Hamerlík, L.; Matúšová, Z.; Klementová, B.R.; Svitok, M. Checklist of benthic macroinvertebrates of high altitude ponds of the Tatra Mountains (Central Europe) with new records of two species for Slovakia. Check List 2015, 11, 1522. [Google Scholar] [CrossRef]
- Hamerlík, L.; Svitok, M.; Novikmec, M.; Veselská, M.; Bitušík, P. Weak altitudinal pattern of overall chironomid richness is a result of contrasting trends of subfamilies in high-altitude ponds. Hydrobiologia 2017, 793, 67–81. [Google Scholar] [CrossRef]
- Hamerlík, L.; Bitušík, P. The Distribution of Littoral Chironomids along an Altitudinal Gradient in High Tatra Mountain Lakes: Could They Be Used as Indicators of Climate Change? Ann. Limnol. Int. J. Lim. 2009, 45, 145–156. [Google Scholar] [CrossRef]
- Sağlam, N.; Dörücü, M. Observations on the Ecology of the Freshwater Leech Helobdella stagnalis (Hirudinoidea: Glossiphoniidae), New for Turkey. Zool. Middle East 2002, 25, 115–120. [Google Scholar] [CrossRef]
- Boggero, A.; Lencioni, V. Macroinvertebrates assemblages of high altitude lakes, inlets and outlets in the southern. Alps. Arch. Hydrobiol. 2006, 165, 37–61. [Google Scholar] [CrossRef]
- Kelly-Quinn, M.; Bracken, J.J. Ephemeropteran assemblages in Ireland. Ver. Int. Ver. Theoret. Angew. Limnol. 2000, 27, 963–969. [Google Scholar] [CrossRef]
- Oertli, B.; Joye, D.A.; Castella, E.; Juge, R.; Cambin, D.; Lachavanne, J.B. Does pond size matter? The relationship between pond area and biodiversity. Biol. Conserv. 2002, 104, 59–70. [Google Scholar] [CrossRef]
- Schenková, J.; Kroča, J. Seasonal changes of an oligochaetous Clitellata (Annelida) community in a mountain stream. Acta Univ. Carol. Environ. 2007, 21, 143–150. [Google Scholar]
- Di Chiara Paoletti, A.; Sambugar, B. Aquatic Oligochaeta in Italy, with special reference to Naididae. Hydrobiologia 1996, 334, 37–49. [Google Scholar] [CrossRef]
- Schniebs, K.; Glöer, P.; Vinarski, M.V.; Hundsdoerfer, A.K. Intraspecific morphological and genetic variability in the European freshwater snail Radix labiata (Rossmaessler, 1835) (Gastropoda: Basommatophora: Lymnaeidae). Contrib. Zool. 2013, 82, 55–68. [Google Scholar] [CrossRef]
- Mackie, G.L. Biology of Freshwater Corbiculid and Sphaeriid Clams of North America; Bulletin Ohio Biological Survey: Columbus, OH, USA, 2007; Volume 15. [Google Scholar]
- Ilg, C.; Oertli, B. How Can We Conserve Cold Stenotherm Communities In Warming Alpine Ponds? Hydrobiologia 2014, 723, 53–62. [Google Scholar] [CrossRef]
- Reiss, F. Verbreitung lakustriner Chironomiden (Diptera) des Alpengebiets. Ann. Zool. Fenn. 1968, 5, 119–125. [Google Scholar]
- Tockner, K.; Malard, F.; Burgherr, P.; Robinson, C.T.; Uehlinger, U.; Zah, R.; Ward, J.V. Physico-chemical characterization of channel types in a glacial floodplain ecosystem (Val Roseg, Switzerland). Arch. Hydrobiol. 1997, 140, 433–463. [Google Scholar] [CrossRef]
- Füreder, L.; Schütz, C.; Burger, R.; Wallinger, M. Seasonal Abundance and Community Structure of Chironomidae in Two Contrasting High Alpine Streams. Verh. Int. Ver. Limnol. 2000, 27, 1597–1601. [Google Scholar] [CrossRef]
- Brittain, J.E.; Milner, A.M. (Eds.) Glacier-fed rivers—Unique lotic ecosystems. Freshw. Biol. 2001, 46, 1571–1874. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).