Ozone Catalysis Degradation of Sodium Acetate via Vacancy-Driven Radical Oxidation over Fe-Modified Fly Ash
Abstract
:1. Introduction
2. Experimental
2.1. Pretreatment of Fly Ash
2.2. Synthesis of Fe-Modified Fly Ash
2.3. Characterization
2.4. Catalytic Ozonation Reaction
2.5. Reactive Species Detection
3. Results and Discussion
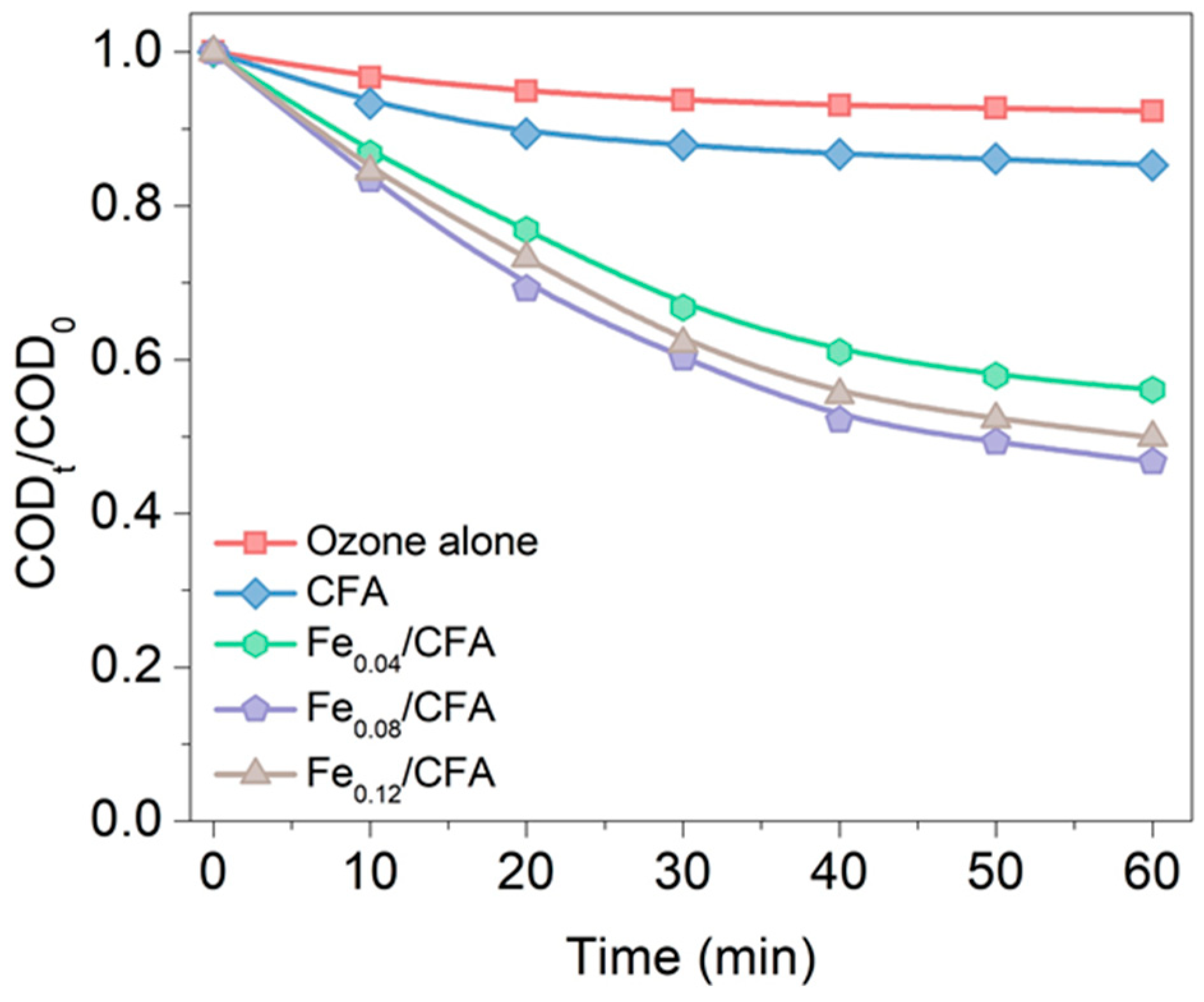
3.1. Enhancing the Degradation of CH3COONa through Catalytic Ozonation
3.2. Structure Characterization of Fe-Modified Fly Ash
3.3. Effect of Reaction Parameters on the Degradation Efficiency of CH3COONa
3.4. Reaction Mechanism Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- You, S.; Ho, S.W.; Li, T.; Maneerung, T.; Wang, C.-H. Techno-economic analysis of geopolymer production from the coal fly ash with high iron oxide and calcium oxide contents. J. Hazard. Mater. 2019, 361, 237–244. [Google Scholar] [CrossRef]
- Forminte, L.; Ciobanu, G.; Buema, G.; Lupu, N.; Chiriac, H.; de Castro, C.G.; Harja, M. New materials synthesized by sulfuric acid attack over power plant fly ash. Rev. Chim. 2020, 71, 48–58. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, B.; Yan, P.; Shen, J.; Kang, J.; Zhao, S.; Zhu, X.; Shen, L.; Wang, S.; Shen, Y. In-situ formation of surface reactive oxygen species on defective sites over N-doped biochar in catalytic ozonation. Chem. Eng. J. 2023, 454, 140232. [Google Scholar] [CrossRef]
- Yao, Z.T.; Ji, X.; Sarker, P.; Tang, J.; Ge, L.; Xia, M.; Xi, Y. A comprehensive review on the applications of coal fly ash. Earth-Sci. Rev. 2015, 141, 105–121. [Google Scholar] [CrossRef]
- Feng, W.; Wan, Z.; Daniels, J.; Li, Z.; Xiao, G.; Yu, J.; Xu, D.; Guo, H.; Zhang, D.; May, E.F. Synthesis of high quality zeolites from coal fly ash: Mobility of hazardous elements and environmental applications. J. Clean. Prod. 2018, 202, 390–400. [Google Scholar] [CrossRef]
- Wang, L.; Huang, X.; Zhang, J.; Wu, F.; Liu, F.; Zhao, H.; Hu, X.; Zhao, X.; Li, J.; Ju, X. Stabilization of lead in waste water and farmland soil using modified coal fly ash. J. Clean. Prod. 2021, 314, 127957. [Google Scholar] [CrossRef]
- Buema, G.; Lupu, N.; Chiriac, H.; Ciobanu, G.; Bucur, R.-D.; Bucur, D.; Favier, L.; Harja, M. Performance assessment of five adsorbents based on fly ash for removal of cadmium ions. J. Mol. Liq. 2021, 333, 115932. [Google Scholar] [CrossRef]
- Wang, N.; Sun, X.; Zhao, Q.; Wang, P. Treatment of polymer-flooding wastewater by a modified coal fly ash-catalysed Fenton-like process with microwave pre-enhancement: System parameters, kinetics, and proposed mechanism. Chem. Eng. J. 2021, 406, 126734. [Google Scholar] [CrossRef]
- Niveditha, S.; Gandhimathi, R. Flyash augmented Fe3O4 as a heterogeneous catalyst for degradation of stabilized landfill leachate in Fenton process. Chemosphere 2020, 242, 125189. [Google Scholar] [CrossRef]
- Gupta, P.K.; Mahato, A.; Oraon, P.; Gupta, G.K.; Maity, S. Coal fly ash-derived mesoporous SBA-15 as support material for production of liquid hydrocarbon through Fischer–Tropsch route. Asia-Pac. J. Chem. Eng. 2020, 15, e2471. [Google Scholar] [CrossRef]
- Sikarwar, P.; Kumar, U.A.; Gosu, V.; Subbaramaiah, V. Catalytic oxidative desulfurization of DBT using green catalyst (Mo/MCM-41) derived from coal fly ash. J. Environ. Chem. Eng. 2018, 6, 1736–1744. [Google Scholar] [CrossRef]
- Yuan, S.; Wang, M.; Liu, J.; Guo, B. Recent advances of SBA-15-based composites as the heterogeneous catalysts in water decontamination: A mini-review. J. Environ. Manag. 2020, 254, 109787. [Google Scholar] [CrossRef]
- Sun, X.; Xu, D.; Dai, P.; Liu, X.; Tan, F.; Guo, Q. Efficient degradation of methyl orange in water via both radical and non-radical pathways using Fe-Co bimetal-doped MCM-41 as peroxymonosulfate activator. Chem. Eng. J. 2020, 402, 125881. [Google Scholar] [CrossRef]
- Mohammadi, R.; Feyzi, M.; Joshaghani, M. Synthesis of ZnO-magnetic/ZSM-5 and its application for removal of disperse Blue 56 from contaminated water. Chem. Eng. Process. 2020, 153, 107969. [Google Scholar] [CrossRef]
- Yuan, L.; Shen, J.; Yan, P.; Chen, Z. Interface mechanisms of catalytic ozonation with amorphous iron silicate for removal of 4-chloronitrobenzene in aqueous solution. Environ. Sci. Technol. 2018, 52, 1429–1434. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Li, C.; Ma, J.; Tian, H.; Qiang, Z. Surface hydroxyl groups of synthetic α-FeOOH in promoting OH generation from aqueous ozone: Property and activity relationship. Appl. Catal. B Environ. 2008, 82, 131–137. [Google Scholar] [CrossRef]
- Zhu, H.; Ma, W.; Han, H.; Han, Y.; Ma, W. Catalytic ozonation of quinoline using nano-MgO: Efficacy, pathways, mechanisms and its application to real biologically pretreated coal gasification wastewater. Chem. Eng. J. 2017, 327, 91–99. [Google Scholar] [CrossRef]
- Nawaz, F.; Cao, H.; Xie, Y.; Xiao, J.; Chen, Y.; Ghazi, Z.A. Selection of active phase of MnO2 for catalytic ozonation of 4-nitrophenol. Chemosphere 2017, 168, 1457–1466. [Google Scholar] [CrossRef]
- Kermani, M.; Kakavandi, B.; Farzadkia, M.; Esrafili, A.; Jokandan, S.F.; Shahsavani, A. Catalytic ozonation of high concentrations of catechol over TiO2@Fe3O4 magnetic core-shell nanocatalyst: Optimization, toxicity and degradation pathway studies. J. Clean. Prod. 2018, 192, 597–607. [Google Scholar] [CrossRef]
- Ncanana, Z.; Pullabhotla, V.R. Oxidative degradation of m-cresol using ozone in the presence of pure γ-Al2O3, SiO2 and V2O5 catalysts. J. Environ. Chem. Eng. 2019, 7, 103072. [Google Scholar] [CrossRef]
- Kruanak, K.; Jarusutthirak, C. Degradation of 2, 4, 6-trichlorophenol in synthetic wastewater by catalytic ozonation using alumina supported nickel oxides. J. Environ. Chem. Eng. 2019, 7, 102825. [Google Scholar] [CrossRef]
- Shen, T.; Su, W.; Yang, Q.; Ni, J.; Tong, S. Synergetic mechanism for basic and acid sites of MgMxOy (M = Fe, Mn) double oxides in catalytic ozonation of p-hydroxybenzoic acid and acetic acid. Appl. Catal. B Environ. 2020, 279, 119346. [Google Scholar] [CrossRef]
- Sun, P.; Zhai, S.; Chen, J.; Yuan, J.; Wu, Z.; Weng, X. Development of a multi-active center catalyst in mediating the catalytic destruction of chloroaromatic pollutants: A combined experimental and theoretical study. Appl. Catal. B Environ. 2020, 272, 119015. [Google Scholar] [CrossRef]
- Wang, N.; Jin, L.; Li, C.; Liang, Y.; Wang, P. Preparation of coal fly ash-based Fenton-like catalyst and its application for the treatment of organic wastewater under microwave assistance. J. Clean. Prod. 2022, 342, 130926. [Google Scholar] [CrossRef]
- Li, M.; Yang, K.; Huang, X.; Liu, S.; Jia, Y.; Gu, P.; Miao, H. Efficient degradation of trimethoprim by catalytic ozonation coupled with Mn/FeOx-functionalized ceramic membrane: Synergic catalytic effect and enhanced anti-fouling performance. J. Colloid Interf. Sci. 2022, 616, 440–452. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Feng, D.; Tong, Y. Hierarchical metal sulfides heterostructure as superior bifunctional electrode for overall water splitting. ChemSusChem 2022, 15, e202200590. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Zhang, T.; Zhang, Y.; Du, J.; Han, X.; Chen, J. Enhancing electrocatalytic oxygen reduction on MnO2 with vacancies. Angew. Chem. Int. Ed. 2013, 125, 2534–2537. [Google Scholar] [CrossRef]
- Liang, L.; Cao, P.; Qin, X.; Wu, S.; Bai, H.; Chen, S.; Yu, H.; Su, Y.; Quan, X. Oxygen vacancies-driven nonradical oxidation pathway of catalytic ozonation for efficient water decontamination. Appl. Catal. B Environ. 2023, 325, 122321. [Google Scholar] [CrossRef]
- Yu, J.; Li, X.; Wu, M.; Lin, K.; Xu, L.; Zeng, T.; Shi, H.; Zhang, M. Synergistic role of inherent calcium and iron minerals in paper mill sludge biochar for phosphate adsorption. Sci. Total. Environ. 2022, 834, 155193. [Google Scholar] [CrossRef]
- Cai, C.; Duan, X.; Xie, X.; Kang, S.; Liao, C.; Dong, J.; Liu, Y.; Xiang, S.; Dionysiou, D.D. Efficient degradation of clofibric acid by heterogeneous catalytic ozonation using CoFe2O4 catalyst in water. J. Haz. Mat. 2021, 410, 124604. [Google Scholar] [CrossRef]
- Li, Z.; Lyu, J.; Ge, M. Synthesis of magnetic Cu/CuFe2O4 nanocomposite as a highly efficient Fenton-like catalyst for methylene blue degradation. J. Mater. Sci. 2018, 53, 15081–15095. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Y.; Guo, J.; Kan, Z.; Jia, Y. Catalytic ozonation mechanisms of Norfloxacin using Cu–CuFe2O4. Environ. Res. 2023, 216, 114521. [Google Scholar] [CrossRef]
- Gao, P.; Zhang, Z.; Feng, L.; Liu, Y.; Du, Z.; Zhang, L. Novel Ti3C2/Bi@BiOI nanosheets with gradient oxygen vacancies for the enhancement of spatial charge separation and photocatalytic performance: The roles of reactive oxygen and iodine species. Chem. Eng. J. 2021, 426, 130764. [Google Scholar] [CrossRef]
- Liu, J.; Ke, L.; Liu, J.; Sun, L.; Yuan, X.; Li, Y.; Xia, D. Enhanced catalytic ozonation towards oxalic acid degradation over novel copper doped manganese oxide octahedral molecular sieves nanorods. J. Haz. Mater. 2019, 371, 42–52. [Google Scholar] [CrossRef]
- Saputra, E.; Muhammad, S.; Sun, H.; Ang, H.-M.; Tadé, M.O.; Wang, S. Manganese oxides at different oxidation states for heterogeneous activation of peroxymonosulfate for phenol degradation in aqueous solutions. Appl. Catal. B Environ. 2013, 142, 729–735. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, W.; Yin, X.; Liu, Y. The role of Mn-doping for catalytic ozonation of phenol using Mn/γ-Al2O3 nanocatalyst: Performance and mechanism. J. Environ. Chem. Eng. 2016, 4, 3415–3425. [Google Scholar] [CrossRef]
- Ma, N.; Ru, Y.; Weng, M.; Chen, L.; Chen, W.; Dai, Q. Synergistic mechanism of supported Mn–Ce oxide in catalytic ozonation of nitrofurazone wastewater. Chemosphere 2022, 308, 136192. [Google Scholar] [CrossRef]
- Liu, Y.; Bian, C.; Li, Y.; Sun, P.; Xiao, Y.; Xiao, X.; Wang, W.; Dong, X. Aminobenzaldehyde convelently modified graphitic carbon nitride photocatalyst through Schiff base reaction: Regulating electronic structure and improving visible-light-driven photocatalytic activity for moxifloxacin degradation. J. Colloid. Interface Sci. 2023, 630, 867–878. [Google Scholar] [CrossRef]
- Li, Y.; Sun, P.; Liu, T.; Cheng, L.; Chen, R.; Bi, X.; Dong, X. Efficient photothermal conversion for oxidation removal of formaldehyde using an rGO-CeO2 modified nickel foam monolithic catalyst. Sep. Purif. Technol. 2023, 311, 123236. [Google Scholar] [CrossRef]
- Bian, C.; Zhou, B.; Mo, F.; Liu, X.; Sun, P.; Dong, X. Post-synthetically covalent modification of g-C3N4 to regulate electronic structure and investigation of photocatalytic activity for eliminating antibiotics. Sep. Purif. Technol. 2023, 325, 124556. [Google Scholar] [CrossRef]







| Samples | SBET (m2/g) | Vtotal (cm3/g) | Pore Size (nm) | XPS Analysis | ICP Test of Fe Element (g/g) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Fe2+/Fe3+ | Oads | Osur | Olat | Theoretical | Actual | ||||
| CFA | 59.64 | 0.13 | 12.76 | / | 0.56 | 0.39 | 0.05 | / | 0.0098 |
| Fe0.04/CFA | 39.14 | 0.11 | 11.46 | 0.85 | 0.45 | 0.42 | 0.13 | 0.04 | 0.034 |
| Fe0.08/CFA | 20.26 | 0.08 | 13.44 | 0.88 | 0.33 | 0.52 | 0.15 | 0.08 | 0.061 |
| Fe0.12/CFA | 16.37 | 0.06 | 8.47 | 0.83 | 0.40 | 0.39 | 0.21 | 0.12 | 0.087 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Chen, R.; Chang, X.; Yan, J.; Gu, Y.; Xi, S.; Sun, P.; Dong, X. Ozone Catalysis Degradation of Sodium Acetate via Vacancy-Driven Radical Oxidation over Fe-Modified Fly Ash. Water 2023, 15, 3801. https://doi.org/10.3390/w15213801
Chen Y, Chen R, Chang X, Yan J, Gu Y, Xi S, Sun P, Dong X. Ozone Catalysis Degradation of Sodium Acetate via Vacancy-Driven Radical Oxidation over Fe-Modified Fly Ash. Water. 2023; 15(21):3801. https://doi.org/10.3390/w15213801
Chicago/Turabian StyleChen, Yaoji, Ruifu Chen, Xinglan Chang, Jingying Yan, Yajie Gu, Shuang Xi, Pengfei Sun, and Xiaoping Dong. 2023. "Ozone Catalysis Degradation of Sodium Acetate via Vacancy-Driven Radical Oxidation over Fe-Modified Fly Ash" Water 15, no. 21: 3801. https://doi.org/10.3390/w15213801




