Lipid Prospection Based on the Cellular Size of Phytoplankton Communities from Tropical Freshwater Ecosystems: A Systematic Literature Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
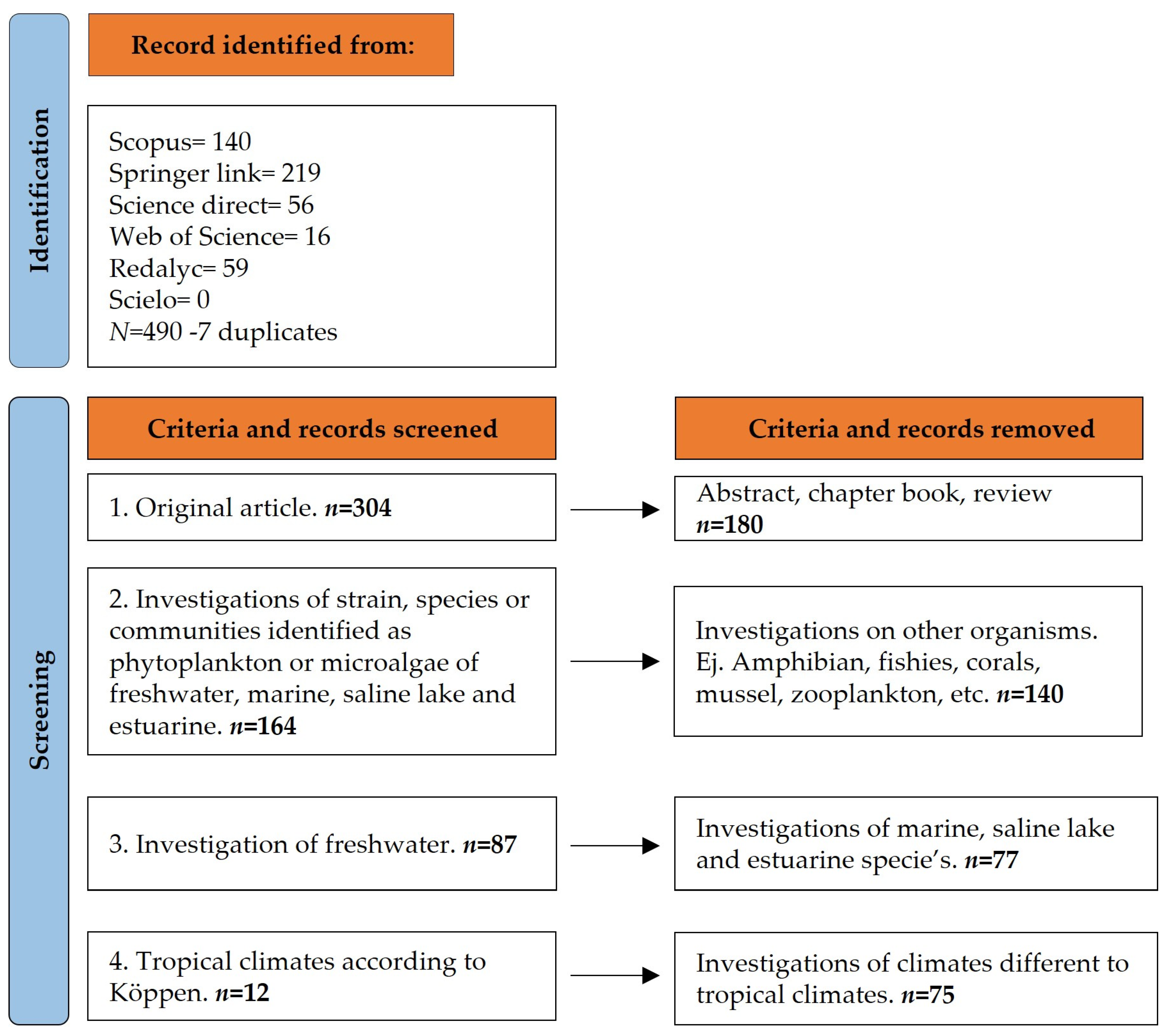
2.2. Stages of the Systematic Review Process
2.3. Data Analysis
3. Results
3.1. Global Data Collection and Phytoplankton Studies over Time
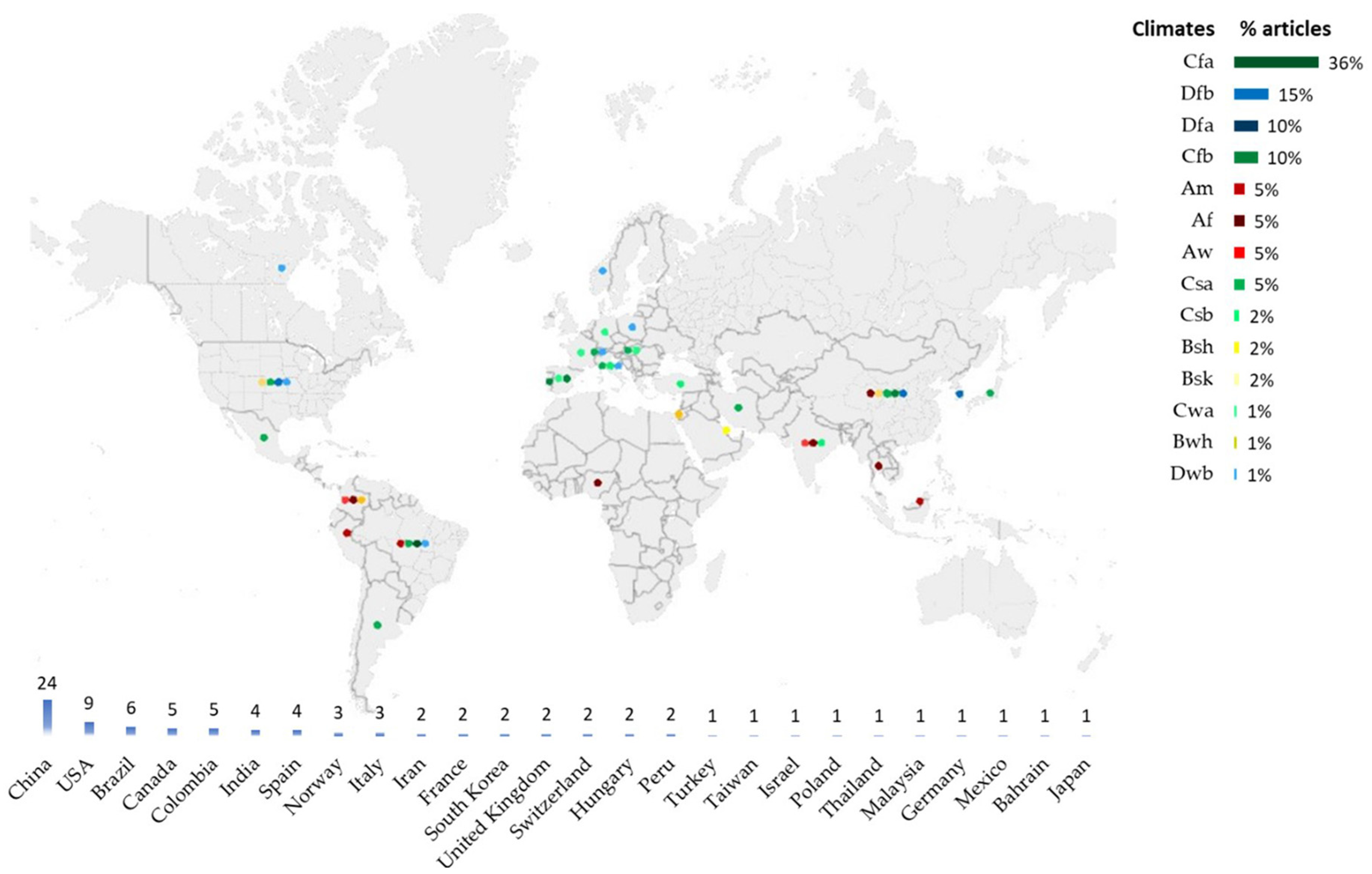
3.2. Distribution of Freshwater Phytoplankton Articles by Climate and Country
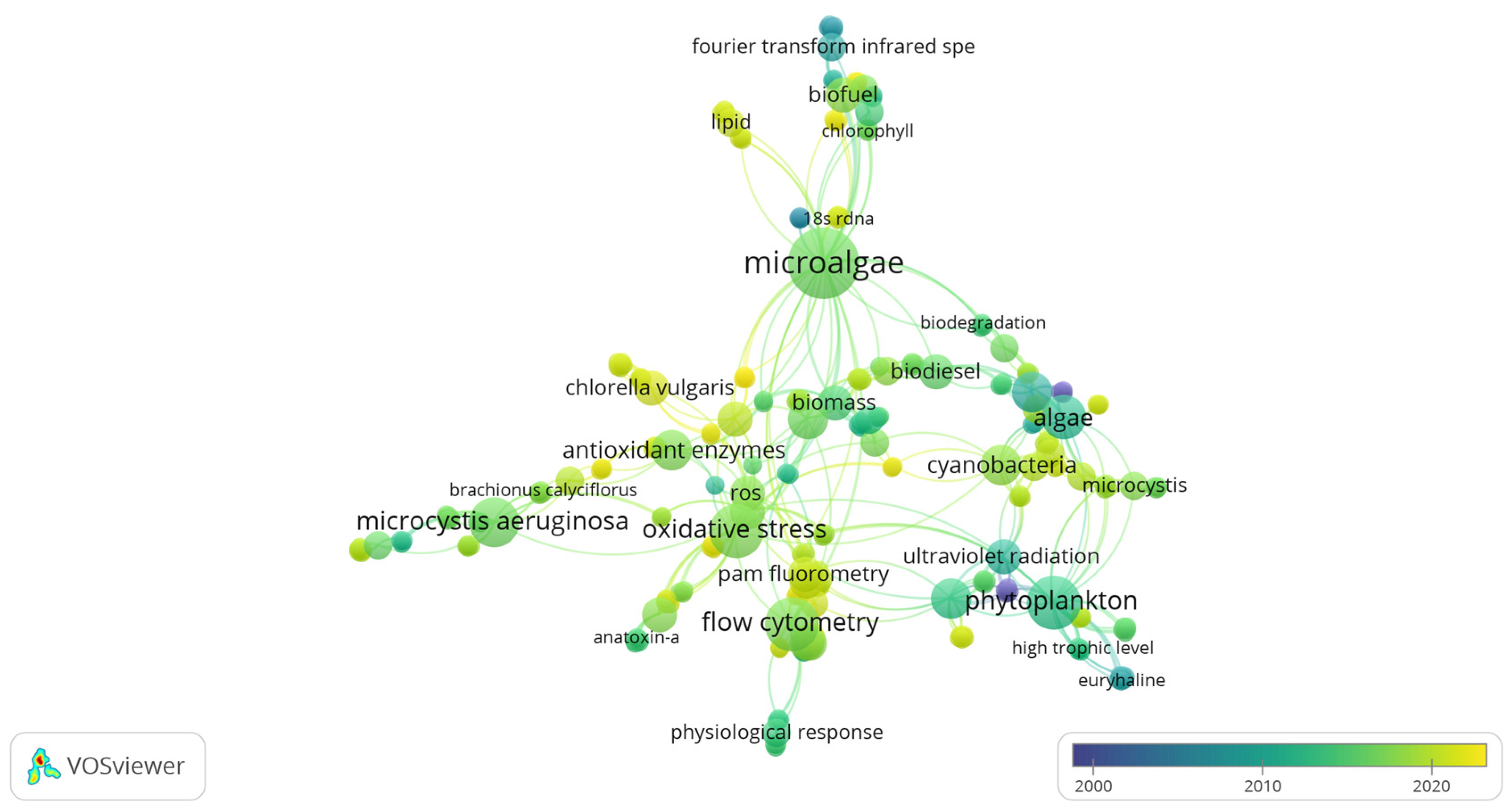
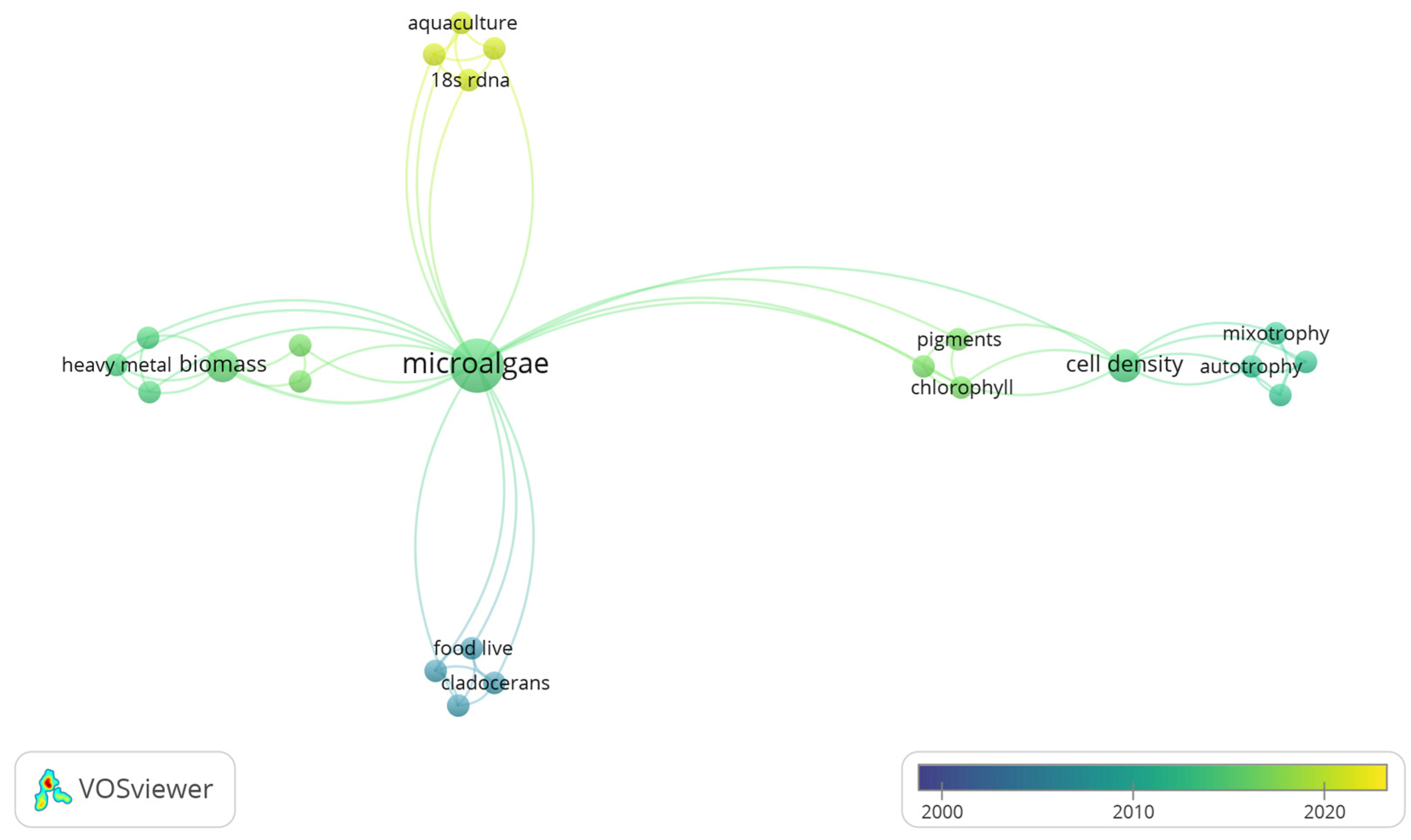
3.3. Co-Occurrence Analysis of Global Freshwater Phytoplankton
Analysis of Freshwater Phytoplankton with Mention of the Eutrophication Process
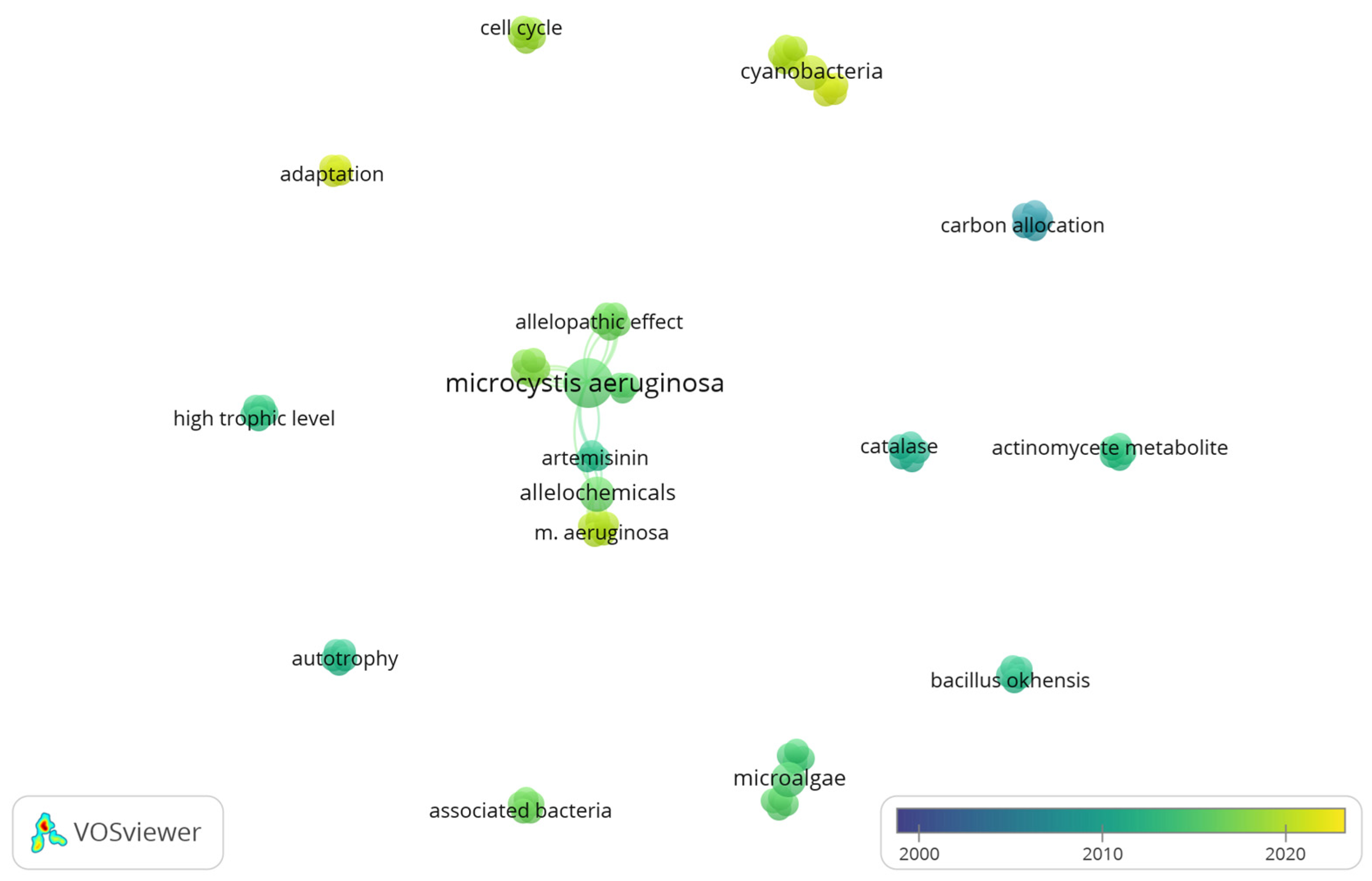
3.4. Co-Occurrence Analysis of Freshwater Phytoplankton Studies in Tropical Environments
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lobus, N.V.; Kulikovskiy, M.S. The co-evolution aspects of the biogeochemical role of phytoplankton in aquatic ecosystems: A review. Biology 2023, 12, 92. [Google Scholar] [CrossRef]
- Maltby, E.; Acreman, M.C. Ecosystem services of wetlands: Pathfinder for a new paradigm. Hydrol. Sci. J. 2011, 56, 341–359. [Google Scholar] [CrossRef]
- Salcedo, M.Á.; Cruz-Ramírez, A.K.; Sánchez, A.J.; Álvarez-Pliego, N.; Florido, R.; Ruiz-Carrera, V.; Morales-Cuetos, S.S. Water quality indicators in three surface hydraulic connection conditions in tropical floodplain lakes. Water 2022, 14, 3931. [Google Scholar] [CrossRef]
- Edwards, K.F.; Li, Q.; McBeain, K.A.; Schvarcz, C.R.; Steward, G.F. Trophic strategies explain the ocean niches of small eukaryotic phytoplankton. Proc. R. Soc. B Biol. Sci. 2023, 290, 20222021. [Google Scholar] [CrossRef] [PubMed]
- Santana, P.; Caprario, J.; Wu, F.; Azevedo, L.; Uda, P.; Finotti, A. Hydraulic-hydrological modelling for ecological flow analysis in urbanised basins without monitored flow data. Urban Water J. 2022, 1–14. [Google Scholar] [CrossRef]
- Scott, C.; Zilio, M.; Harmon, T.; Zuniga-Teran, A.; Díaz-Caravantes, R.; Hoyos, N.; Perillo, G.; Meza, F.; Varady, R.; Neto, A.; et al. Do ecosystem insecurity and social vulnerability lead to failure of water security? Environ. Dev. 2021, 38, 100606. [Google Scholar] [CrossRef]
- Huggins, X.; Gleeson, T.; Kummu, M.; Zipper, S.; Wada, Y.; Troy, T.; Famiglietti, J. Hotspots for social and ecological impacts from freshwater stress and storage loss. Nat. Commun. 2022, 13, 439. [Google Scholar] [CrossRef]
- Mitroi, V.; Maleval, V.; Deroubaix, J.F.; Vinçon-Leite, B.; Humbert, J.-F. What urban lakes and ponds quality is about? conciliating water quality and ecological indicators with users’ perceptions and expectations about urban lakes and ponds quality in urban areas. J. Environ. Policy Plan. 2022, 24, 701–718. [Google Scholar] [CrossRef]
- Häder, N.; Gao, K. Aquatic productivity under multiple stressors. Water 2023, 15, 817. [Google Scholar] [CrossRef]
- Peltomaa, E.; Hällfors, H.; Taipale, S.J. Comparison of diatoms and dinoflagellates from different habitats as sources of PUFAs. Mar. Drugs. 2019, 17, 233. [Google Scholar] [CrossRef]
- Jónasdóttir, S. Fatty acid profiles and production in marine phytoplankton. Mar. Drugs 2019, 17, 151. [Google Scholar] [CrossRef] [PubMed]
- Meyer, N.; Rettner, J.; Werner, M.; Werz, O.; Pohnert, G. Algal oxylipins mediate the resistance of diatoms against algicidal bacteria. Mar. Drugs 2018, 16, 486. [Google Scholar] [CrossRef] [PubMed]
- Gorokhova, E. Individual body size as a predictor of lipid storage in Baltic Sea zooplankton. J. Plankton Res. 2019, 41, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Robinson, A.; Bouman, H.; Tilstone, G.; Sathyendranath, S. Size class dependent relationships between temperature and phytoplankton photosynthesis-irradiance parameters in the Atlantic Ocean. Front. Mar. Sci. 2018, 4, 435. [Google Scholar] [CrossRef]
- Hillebrand, H.; Acevedo-trejos, E.; Moorthi, S.; Ryabov, A.; Striebel, M.; Thomas, P.; Schneider, M. Cell size as driver and sentinel of phytoplankton community structure and functioning. Funct. Ecol. 2022, 36, 276–293. [Google Scholar] [CrossRef]
- Finkel, Z.; Follows, M.; Irwin, A. Size-scaling of macromolecules and chemical energy content in the eukaryotic microalgae. J. Plankton Res. 2016, 38, 1151–1162. [Google Scholar] [CrossRef]
- Olenina, I.; Wasmund, N.; Hajdu, S.; Jurgensone, I.; Gromisz, S.; Kownacka, J.; Toming, K.; Vaiciūtė, D.; Olenin, S. Assessing impacts of invasive phytoplankton: The baltic sea case. Mar. Pollut. Bull. 2006, 60, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Berglund, O.; Larsson, P.; Ewald, G.; Okla, L. The effect of lake trophy on lipid content and PCB concentrations in planktonic food webs. Ecology 2001, 82, 1078–1088. [Google Scholar] [CrossRef]
- Arts, M.; Robarts, R.; Evans, M. Seasonal changes in particulate and dissolved lipids in a eutrophic prairie lake. Freshw. Biol. 1997, 38, 525–537. [Google Scholar] [CrossRef]
- de Carvalho, C.; Caramujo, M. The various roles of fatty acids. Molecules 2018, 23, 2583. [Google Scholar] [CrossRef]
- Parrish, C. Dissolved and particulate marine lipid classes: A review. Mar. Chem. 1988, 23, 17–40. [Google Scholar] [CrossRef]
- Lobus, N.V. Biogeochemical role of algae in aquatic ecosystems: Basic research and applied biotechnology. J. Mar. Sci. Eng. 2022, 10, 1846. [Google Scholar] [CrossRef]
- Basu, S.; Mackey, K.R. Phytoplankton as key mediators of the biological carbon pump: Their responses to a changing climate. Sustainability 2018, 10, 869. [Google Scholar] [CrossRef]
- Birner, R. Bioeconomy Concepts. In Bioeconomy, Shaping the Transition to a Sustainable, Biobased Economy; Lewandowski, I., Ed.; Springer Nature: Stuttgart, Germany, 2018; pp. 17–38. [Google Scholar] [CrossRef]
- Page, M.; Mckenzie, J.; Bossuyt, P.; Boutron, I.; Hoffmann, T.; Mulrow, C.; Shamseer, L.; Tetzlaff, J.; Akl, E.; Brennan, S.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. J. Clin. Epidemiol. 2021, 134, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Climate Data. Available online: https://es.climate-data.org/ (accessed on 1 April 2021).
- Mauri, M.; Elli, T.; Caviglia, G.; Uboldi, G.; Azzi, M. RAWGraphs: A visualization platform to create open outputs. In Proceedings of the 12th Biannual Conference on Italian SIGCHI Chapter, Cagliari, Italy, 18–20 September 2017; ACM: Cagliari, Italy, 2017; pp. 1–5. [Google Scholar] [CrossRef]
- Van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Li, M.; Reynolds, C.S. Colony formation in the cyanobacterium Microcystis. Biol. Rev. Camb. Philos. Soc. 2018, 93, 1399–1420. [Google Scholar] [CrossRef]
- Liu, C.; Shi, X.; Wu, F.; Ren, M.; Gao, G.; Wu, Q. Genome analyses provide insights into the evolution and adaptation of the eukaryotic picophytoplankton Mychonastes homosphaera. BMC Genom. 2020, 21, 477. [Google Scholar] [CrossRef] [PubMed]
- Maraşlıoğlu, F.; Öterler, B.; Sevindik, T.O.; Soylu, E.N.; Demir, N.; Çelekli, A.; Sömek, H.; Coşkun, T.; Solak, C.N.; Çetin, T.; et al. New records for the Turkish freshwater algal flora in twenty-five river basins of Turkey, Part II: Chlorophyta, cyanobacteria, euglenozoa. Turk. J. Fish. Aquat. Sci. 2022, 22. [Google Scholar] [CrossRef]
- Komárek, J.; Anagnostidis, K. Nomenclatural novelties in chroococcalean cyanoprokaryotes. Preslia 1995, 67, 15–23. [Google Scholar]
- Kim, Y.J. Taxonomic and ecological study of the families hydrodictyaceae and coelastraceae, order chlorococcales, and class chlorophyceae in Korea. J. Ecol. Environ. 2013, 36, 421–437. [Google Scholar] [CrossRef]
- Škaloud, P.; Škaloudová, M.; Procházková, A.; Němcová, Y. Morphological delineation and distribution patterns of four newly described species within the Synura petersenii species complex (chrysophyceae, stramenopiles). Eur. J. Phycol. 2014, 49, 213–229. [Google Scholar] [CrossRef]
- Lin, S.; Gobler, C.J.; Carpenter, E.J. Cytological and biochemical responses of Dunaliella tertiolecta (volvocales, chlorophyta) to iron stress. Phycologia 2001, 40, 403–410. [Google Scholar] [CrossRef]
- Shiraishi, H. Cryopreservation of the edible alkalophilic cyanobacterium Arthrospira platensis. Biosci. Biotechnol. Biochem. 2016, 80, 2051–2057. [Google Scholar] [CrossRef] [PubMed]
- Lizzul, A.M.; Lekuona-Amundarain, A.; Purton, S.; Campos, L.C. Characterization of Chlorella sorokiniana, UTEX 1230. Biology 2018, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Engel, B.D.; Schaffer, M.; Kuhn Cuellar, L.; Villa, E.; Plitzko, J.M.; Baumeister, W. Native architecture of the Chlamydomonas chloroplast revealed by in situ cryo-electron tomography. eLife 2015, 13, e04889. [Google Scholar] [CrossRef]
- Vander Wiel, J.B.; Mikulicz, J.D.; Boysen, M.R.; Hashemi, N.; Kalgren, P.; Nauman, L.M.; Baetzold, S.J.; Powell, G.G.; He, Q.; Hashemi, N.N. Characterization of Chlorella vulgaris and Chlorella protothecoides using multi-pixel photon counters in a 3D focusing optofluidic system. RSC Adv. 2017, 7, 4402–4408. [Google Scholar] [CrossRef]
- Moestrup, Ø.; Hansen, G.; Daugbjerg, N. Studies on woloszynskioid dinoflagellates III: On the ultrastructure and phylogeny of Borghiella dodgei gen. et sp. nov., a cold-water species from Lake Tovel, N. Italy, and on B. tenuissima comb. nov. (syn. Woloszynskia tenuissima). Phycologia 2008, 47, 54–78. [Google Scholar] [CrossRef]
- Zielinski, R. Motility in Scenedesmus Quadricauda. Student Work. 1975. Available online: https://digitalcommons.unomaha.edu/studentwork/3377 (accessed on 23 October 2023).
- Obertegger, U.; Camin, F.; Guella, G.; Flaim, G. Adaptation of a psychrophilic freshwater dinoflagellate to ultraviolet radiation. J. Phycol. 2011, 47, 811–820. [Google Scholar] [CrossRef]
- Ji, M.K.; Kabra, A.N.; Choi, J.; Hwang, J.H.; Kim, J.R.; Abou-Shanab, R.A.; Oh, Y.K.; Jeon, B.H. Biodegradation of bisphenol A by the freshwater microalgae Chlamydomonas mexicana and Chlorella vulgaris. Ecol. Eng. 2014, 73, 260–269. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.-J.; Li, Y.-W.; Du, J.-G.; Wang, Z.-H.; Deng, S.-H. Effects of single-walled carbon nanotubes on growth and physiological characteristics of Microcystis aeruginosa. J. Cent. South Univ. 2018, 25, 1628–1641. [Google Scholar] [CrossRef]
- Sigee, D.C.; Bahrami, F.; Estrada, B.; Webster, R.E.; Dean, A.P. The influence of phosphorus availability on carbon allocation and P quota in Scenedesmus subspicatus: A synchrotron-based FTIR analysis. Phycologia 2007, 46, 583–592. [Google Scholar] [CrossRef]
- Jusoh, M.; Kasan, N.A.; Hashim, F.S.; Haris, N.; Zakaria, M.F.; Mohamed, N.N.; Rasdi, N.W.; Abd Wahid, M.E.; Katayama, T.; Takahashi, K. Isolation of freshwater and marine indigenous microalgae species from Terengganu water bodies for potential uses as live feeds in aquaculture industry. Int. Aquat. Res. 2020, 12, 74–83. [Google Scholar] [CrossRef]
- Ortiz-Moreno, M.L.; Cortés-Castillo, C.E.; Sánchez-Villarraga, J.; Padilla, J.; Otero-Paternina, A.M. Evaluación del crecimiento de la microalga Chlorella sorokiniana en diferentes medios de cultivo en condiciones autotróficas y mixotróficas. Orinoquia 2012, 16, 11–20. [Google Scholar] [CrossRef]
- Cobos, M.; Castro, J.; Cerdeira, L. Potencial biotecnológico para la producción sustentable de biodiesel de microalgas oleaginosas aisladas del Río Itaya, Loreto, Perú. Ecol. Apl. 2014, 13, 169–175. [Google Scholar] [CrossRef]
- Cobos, M.; Paredes, J.; Castro, J. Inducción de la producción de lípidos totales en microalgas sometidas a estrés nutritivo. Acta Biol. Colomb. 2016, 21, 17–26. [Google Scholar]
- Sarker, N.K.; Salam, P.A. Design of batch algal cultivation systems and ranking of the design parameters. Energy Ecol. Environ. 2020, 5, 196–210. [Google Scholar] [CrossRef]
- Bemal, S.; Anil, A.C. Effects of salinity on cellular growth and exopolysaccharide production of freshwater Synechococcus strain CCAP1405. J. Plankton Res. 2018, 40, 46–58. [Google Scholar] [CrossRef]
- Burgos, R.C.A.; Ramírez-Merlano, J.A.; Jiménez-Forero, J.A. Uso de fertilizante comercial en la cinética celular de Desmodesmus opoliensis (Chlorophyceae), reporte preliminar. Orinoquia 2016, 20, 18–25. [Google Scholar] [CrossRef]
- Prieto, M.; De la Cruz, L.; Morales, M. Cultivo experimental del cladocero Moina sp. alimentado con Ankistrodesmus sp. y Saccharomyces cereviseae. Rev. MVZ Cordoba 2006, 11, 705–714. [Google Scholar] [CrossRef]
- Adams, J.; Pendlebury, D.; Potter, R.; Szomszor, M. Global Research Report Latin America: South and Central America, Mexico, and the Caribbean. ISI. 2021. Available online: https://clarivate.com/lp/latin-america-south-and-central-america-mexico-and-the-caribbean/ (accessed on 23 October 2023).
- United Nations. Water the United Nations World Water Development Report 2023: Partnerships and Cooperation for Water; UNESCO: Paris, France, 2023; ISBN 978-92-3-100576-3. [Google Scholar]
- Rotondo, F.; Perchinunno, P.; L’Abbate, S.; Mongelli, L. Ecological transition and sustainable development: Integrated statistical indicators to support public policies. Sci. Rep. 2022, 12, 18513. [Google Scholar] [CrossRef]
- Oliver, S.; Corburn, J.; Ribeiro, H. Challenges regarding water quality of eutrophic reservoirs in urban landscapes: A mapping literature review. Int. J. Environ. Res. Public Health 2019, 16, 40. [Google Scholar] [CrossRef] [PubMed]
- Growns, I.; Ryder, D.; Mcinerney, P.; Bond, N.; Holt, G.; Lester, R.; Thompson, R. The use of fatty acids to identify food sources of secondary consumers in wetland mesocosms. J. Freshw. Ecol. 2020, 35, 173–189. [Google Scholar] [CrossRef]
- Marie, D.; Simon, N.; Guillou, L.; Partensky, F.; Vaulot, D. Flow cytometry analysis of marine picoplankton. In Living Color; Springer Lab Manuals; Diamond, R.A., Demaggio, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2000; pp. 421–454. [Google Scholar] [CrossRef]
- Ni, L.; Acharya, K.; Hao, X.; Li, S. Isolation and identification of an anti-algal compound from Artemisia annua and mechanisms of inhibitory effect on algae. Chemosphere 2012, 88, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhou, Q.-H.; Liu, B.-Y.; Cheng, L.; Tian, Y.; Zhang, Y.-Y.; Wu, Z.-B. Programmed cell death in the cyanobacterium Microcystis aeruginosa induced by allelopathic effect of submerged macrophyte Myriophyllum spicatum in co-culture system. J. Appl. Phycol. 2016, 28, 2805–2814. [Google Scholar] [CrossRef]
- Chen, L.; Wang, Y.; Shi, L.; Zhao, J.; Wang, W. Identification of allelochemicals from pomegranate peel and their effects on Microcystis aeruginosa growth. Environ. Sci. Pollut. Res. 2019, 26, 22389–22399. [Google Scholar] [CrossRef] [PubMed]
- Chia, M.A.; Chimdirim, P.K.; Japhet, W.S. Lead induced antioxidant response and phenotypic plasticity of Scenedesmus quadricauda (Turp.) de Brébisson under different nitrogen concentrations. J. Appl. Phycol. 2015, 27, 293–302. [Google Scholar] [CrossRef]
- Tarafdar, L.; Mohapatra, M.; Muduli, P.R.; Kumar, A.; Mishra, D.R.; Rastogi, G. Co-occurrence patterns and environmental factors associated with rapid onset of Microcystis aeruginosa bloom in a tropical coastal lagoon. J. Environ. Manag. 2023, 325, 116580. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, Y.; Huang, S.; Peng, C.; Hao, Z.; Li, D. Nitrogen limitation significantly reduces the competitive advantage of toxic Microcystis at high light conditions. Chemosphere 2019, 237, 124508. [Google Scholar] [CrossRef]
- Costa, J.A.D.; Souza, J.P.D.; Teixeira, A.P.; Nabout, J.C.; Carneiro, F.M. Eutrophication in aquatic ecosystems: A scientometric study. Acta Limnol. Bras. 2018, 30, 1–11. [Google Scholar] [CrossRef]
- Zhu, K.; Wu, Y.; Li, C.; Xu, J.; Zhang, M. Ecosystem-Based Restoration to Mitigate Eutrophication: A Case Study in a Shallow Lake. Water 2020, 12, 2141. [Google Scholar] [CrossRef]
- Dai, T.; Liu, R.; Zhou, X.; Zhang, J.; Song, M.; Zou, P.; Bi, X.; Li, S. Role of Lake Aquatic–Terrestrial Ecotones in the Ecological Restoration of Eutrophic Water Bodies. Toxics 2023, 11, 560. [Google Scholar] [CrossRef]
- Rohr, J.R.; Bernhardt, E.S.; Cadotte, M.W.; Clements, W.H. The ecology and economics of restoration. Ecol. Soc. 2018, 23, 1–15. [Google Scholar] [CrossRef]
- Lindig-Cisneros, R.; Zambrano, L. Aplicaciones prácticas para la conservación y restauración de humedales y otros ecosistemas acuáticos. In Perspectivas Sobre la Conservación de Ecosistemas Acuáticos en México; Sánchez, O., Herzig, M., Peters, E., Márquez, R., Zambrano, L., Eds.; INE, SEMARNAT: Distrito Federal, Mexico, 2007; pp. 167–188. ISBN 978-968-817-856-0. [Google Scholar]
- Rast, W.; Thornton, J.A. Trends in eutrophication research and control. Hydrol. Process. 1996, 10, 295–313. [Google Scholar] [CrossRef]
- Tanaka, Y.; Minggat, E.; Roseli, W. The impact of tropical land-use change on downstream riverine and estuarine water properties and biogeochemical cycles: A review. Ecol. Process. 2021, 10, 1–21. [Google Scholar] [CrossRef]
- Olabi, A.G.; Shehata, N.; Sayed, E.T.; Rodriguez, C.; Anyanwu, R.C.; Russell, C.; Abdelkareem, M.A. Role of microalgae in achieving sustainable development goals and circular economy. Sci. Total Environ. 2023, 854, 158689. [Google Scholar] [CrossRef]
 freshwater,
freshwater,  marine,
marine,  marine and freshwater,
marine and freshwater,  saline lakes,
saline lakes,  estuarine.
estuarine.
 freshwater,
freshwater,  marine,
marine,  marine and freshwater,
marine and freshwater,  saline lakes,
saline lakes,  estuarine.
estuarine.

| AND | |||||
|---|---|---|---|---|---|
| OR | Phytoplankton 1 | Lipids 1,2 | Cell size 2 | Freshwater 1,2 | Environmental stress 3 |
| Lipidomic | Microplankton | Limnetic | Metabolic stress | ||
| Fatty acids | Nanoplankton | Wetlands | |||
| Lipidic | Picoplankton | Tropical | |||
| Platform | Search Equation |
|---|---|
| Scopus SpringerLink Web of science | (phytoplankton) AND (lipids OR lipidomic OR “fatty acids” OR lipidic) AND (“cell size” OR microplankton OR nanoplankton OR picoplankton) AND (freshwater OR limnetic OR wetlands OR tropical) AND (“environmental stress” OR “metabolic stress”). |
| ScienceDirect | (phytoplankton) AND (lipids OR lipidic) AND (“cell size”) AND (freshwater OR tropical) AND (“environmental stress” OR “metabolic stress”). |
| Redalyc * | (fitoplancton) AND (lípidos OR lipidómica) AND (“tamaño celular” OR microplancton OR nanoplancton OR picoplancton) AND (agua dulce OR limnético OR humedales OR tropical) *. |
| Topics | Lipidic Analysis Reported | Year | Reference |
|---|---|---|---|
| Isolation of microalgae with a high lipid content for potential use in aquaculture industry | Polar lipid content | 2020 | [46] |
| High production of Chlorella sorokiniana for its ability to synthesize fatty acids of industrial interest | Total lipid content | 2012 | [47] |
| Biodiesel production | Total lipid content Triglycerides | 2014 | [48] |
| Production of total lipids | Total lipid content Intracellular lipid presence | 2016 | [49] |
| Commercial use of microalgae | N/A | 2020 | [50] |
| Production of exopolysaccharides | N/A | 2018 | [51] |
| Biotechnological potential as fish feed, supplementary nutrients, pharmaceuticals, and the bioremediation of contaminated water | N/A | 2016 | [52] |
| Production of potentially useful biomass for aquaculture | N/A | 2006 | [53] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bautista-Regil, J.; Sánchez, A.J.; Salcedo, M.Á.; Arredondo-Vega, B.O.; Ruiz-Carrera, V. Lipid Prospection Based on the Cellular Size of Phytoplankton Communities from Tropical Freshwater Ecosystems: A Systematic Literature Review. Water 2023, 15, 3774. https://doi.org/10.3390/w15213774
Bautista-Regil J, Sánchez AJ, Salcedo MÁ, Arredondo-Vega BO, Ruiz-Carrera V. Lipid Prospection Based on the Cellular Size of Phytoplankton Communities from Tropical Freshwater Ecosystems: A Systematic Literature Review. Water. 2023; 15(21):3774. https://doi.org/10.3390/w15213774
Chicago/Turabian StyleBautista-Regil, Jesús, Alberto J. Sánchez, Miguel Ángel Salcedo, Bertha Olivia Arredondo-Vega, and Violeta Ruiz-Carrera. 2023. "Lipid Prospection Based on the Cellular Size of Phytoplankton Communities from Tropical Freshwater Ecosystems: A Systematic Literature Review" Water 15, no. 21: 3774. https://doi.org/10.3390/w15213774





