Understanding Nutrient Loads from Catchment and Eutrophication in a Salt Lagoon: The Mar Menor Case
Abstract
:Highlights
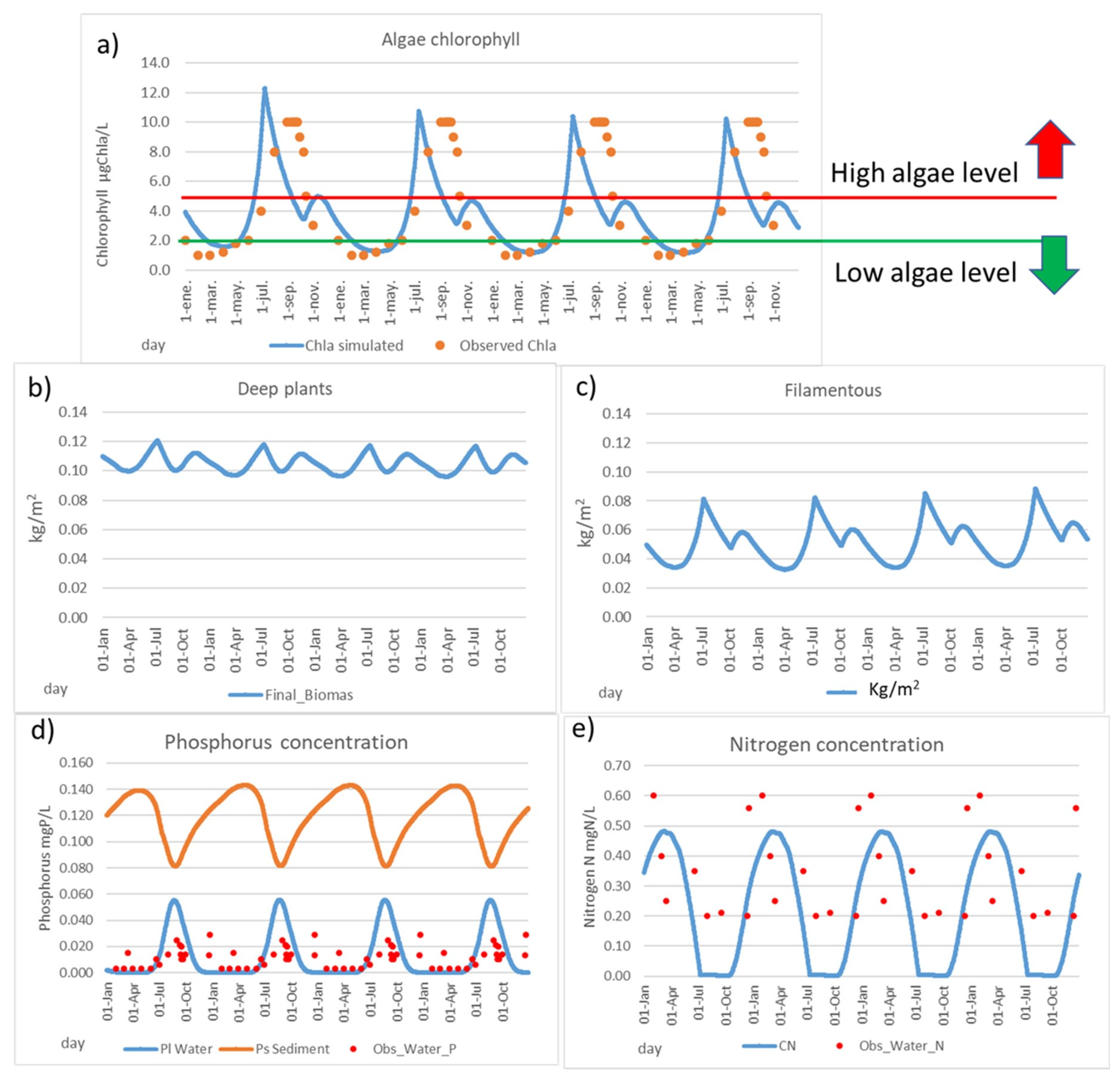
- Mean chlorophyll under 0.5–1 μgChla/L becomes robust and resilient in a salt lagoon.
- Both nitrogen and phosphorus loads contribute to eutrophication in a Mediterranean salt lagoon.
- Key factors in the Mar Menor (MM): phosphorus water–sediment relationship and deep water plants.
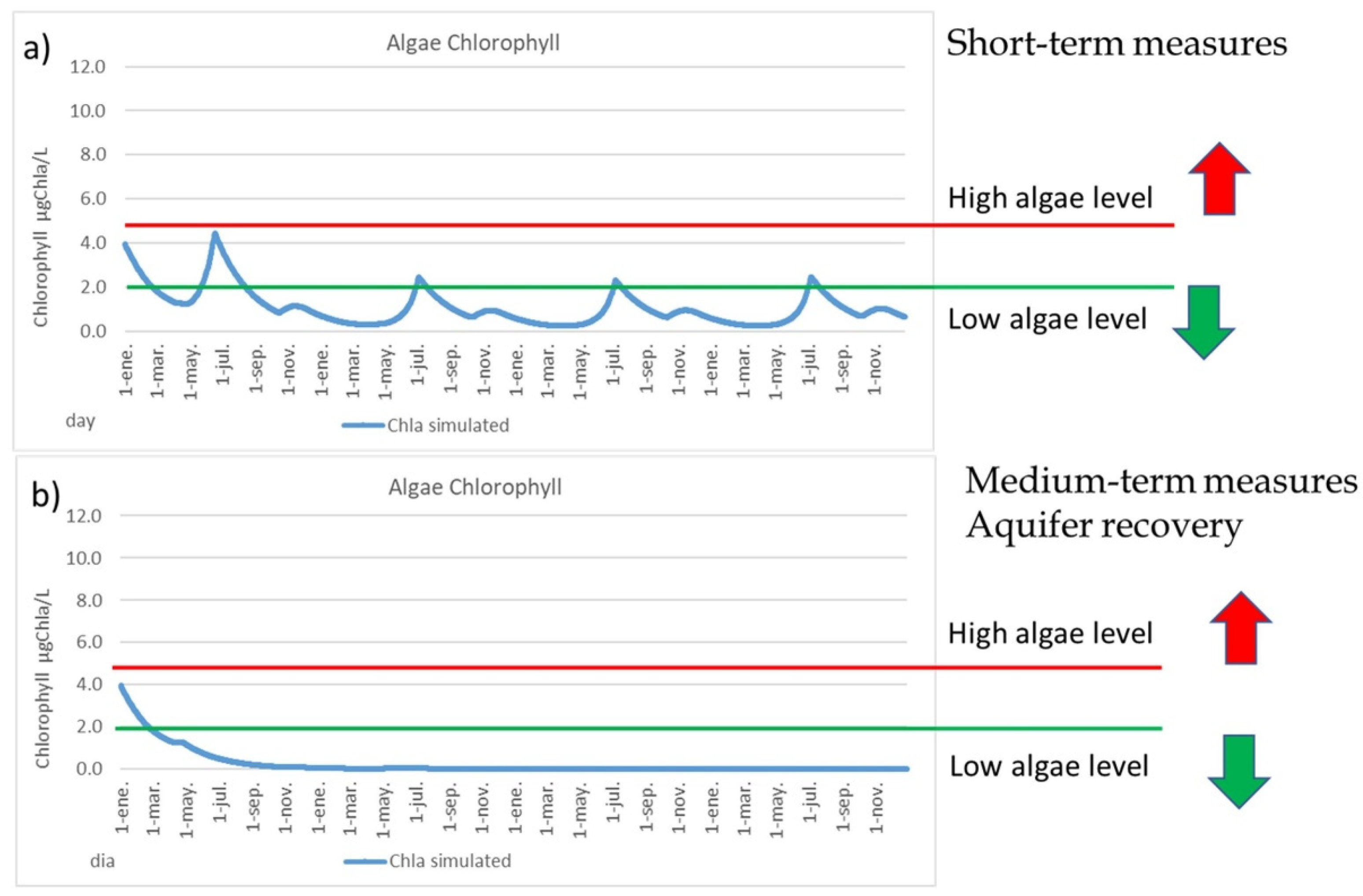
- Recovery nitrate-polluted aquifer makes the MM more robust and resilient.
- High risk of massive mortalities when mean chlorophyll is higher than 5 μgChla/L.
Abstract
1. Introduction
2. Study Case and Data
Study Area
3. Methods and Models
4. Results and Discussion
4.1. Model Calibration and System Response
4.2. Measures to Reduce Algal Bloom Risk
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kakade, A.; Salama, E.-S.; Han, H.; Zheng, Y.; Kulshrestha, S.; Jalalah, M.; Harraz, F.A.; Alsareii, S.A.; Li, X. World eutrophic pollution of lake and river: Biotreatment potential and future perspectives. Environ. Technol. Innov. 2021, 23, 101604. [Google Scholar] [CrossRef]
- Bonilla, S.; Aguilera, A.; Aubriot, L.; Huszar, V.; Almanza, V.; Haakonsson, S.; Izaguirre, I.; O’Farrell, I.; Salazar, A.; Becker, V.; et al. Nutrients and not temperature are the key drivers for cyanobacterial biomass in the Americas. Harmful Algae 2023, 121, 102367. [Google Scholar] [CrossRef] [PubMed]
- García, J. Ecological engineering practice as a global strategy to prevent eutrophication and microalgae blooms. Ecol. Eng. 2021, 161, 106152. [Google Scholar] [CrossRef]
- Le Moal, M.; Gascuel-Odoux, C.; Ménesguen, A.; Souchon, Y.; Étrillard, C.; Levain, A.; Moatar, F.; Pannard, A.; Souchu, P.; Lefebvre, A.; et al. Eutrophication: A new wine in an old bottle? Sci. Total Environ. 2019, 651, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fink, G.; Alcamo, J.; Flörke, M.; Reder, K. Phosphorus loadings to the world’s largest lakes: Sources and trends. Glob. Biogeochem. Cycles 2018, 32, 617–634. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, Y.; Zhang, Y.Y. Plant Nutrients and Trace Elements from the Changjiang Watersheds to the East China Sea. In Ecological Continuum from the Changjiang (Yangtze River) Watersheds to the East China Sea Continental Margin. Estuaries of the World; Zhang, J., Ed.; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- Saturday, A.; Kangume, S.; Bamwerinde, W. Content and dynamics of nutrients in the surface water of shallow Lake Mulehe in Kisoro District, South–western Uganda. Appl. Water Sci. 2023, 13, 150. [Google Scholar] [CrossRef]
- Carpenter, S.R. Eutrophication of aquatic ecosystems: Bistability and soil phosphorus. Proc. Natl. Acad. Sci. USA 2005, 102, 10002–10005. [Google Scholar] [CrossRef]
- Häder, D.-P.; Banaszak, A.T.; Villafañe, V.E.; Narvarte, M.A.; González, R.A.; Helbling, E.W. Anthropogenic pollution of aquatic ecosystems: Emerging problems with global implications. Sci. Total Environ. 2020, 713, 136586. [Google Scholar] [CrossRef]
- Liang, Z.; Xu, Y.; Qiu, Q.; Liu, Y.; Lu, W.; Wagner, T. A framework to develop joint nutrient criteria for lake eutrophication management in eutrophic lakes. J. Hydrol. 2021, 594, 125883. [Google Scholar] [CrossRef]
- Yang, X.E.; Wu, X.; Hao, H.L.; He, Z.L. Mechanisms and assessment of water eutrophication. J. Zhejiang Univ. Sci. B 2008, 9, 197–209. [Google Scholar] [CrossRef]
- Zhao, J.; Gao, Q.; Liu, Q.; Fu, G. Lake eutrophication recovery trajectories: Some recent findings and challenges ahead. Ecol. Indic. 2020, 110, 105878. [Google Scholar] [CrossRef]
- Zouiten, H.; Díaz, C.Á.; Gómez, A.G.; Cortezón, J.A.R.; Alba, J.G. An advanced tool for eutrophication modeling in coastal lagoons: Application to the Victoria lagoon in the north of Spain. Ecol. Model. 2013, 265, 99–113. [Google Scholar] [CrossRef]
- Edwards, K.F.; Thomas, M.K.; Klausmeier, C.A.; Litchman, E. Phytoplankton growth and the interaction of light and temperature: A synthesis at the species and community level. Limnol. Oceanogr. 2016, 61, 1232–1244. [Google Scholar] [CrossRef]
- Lee, K.-S.; Park, S.R.; Kim, Y.K. Effects of irradiance, temperature, and nutrients on growth dynamics of seagrasses: A review. J. Exp. Mar. Biol. Ecol. 2007, 350, 144–175. [Google Scholar] [CrossRef]
- Tasnim, B.; Fang, X.; Hayworth, J.S.; Tian, D. Simulating Nutrients and Phytoplankton Dynamics in Lakes: Model Development and Applications. Water 2021, 13, 2088. [Google Scholar] [CrossRef]
- Abomohra, A.E.-F.; El-Naggar, A.H.; Alaswad, S.O.; Elsayed, M.; Li, M.; Li, W. Enhancement of biodiesel yield from a halophilic green microalga isolated under extreme hypersaline conditions through stepwise salinity adaptation strategy. Bioresour. Technol. 2020, 310, 123462. [Google Scholar] [CrossRef]
- Glibert, P.M. Eutrophication, harmful algae and biodiversity—Challenging paradigms in a world of complex nutrient changes. Mar. Pollut. Bull. 2017, 124, 591–606. [Google Scholar] [CrossRef]
- Glibert, P.M.; Wilkerson, F.; Dugdale, R.C.; Parker, A.E.; Alexander, J.A.; Blaser, S.; Murasko, S. Microbial communities from San Francisco Bay Delta respond differently to oxidized and reduced nitrogen substrates—Even under conditions that would otherwise suggest nitrogen sufficiency. Front. Mar. Sci. 2014, 1, 17. [Google Scholar] [CrossRef]
- Baldia, S.F.; Evangelista, A.D.; Aralar, E.V.; Santiago, A.E. Nitrogen and phosphorus utilization in the cyanobacterium Microcystis aeruginosa isolated from Laguna de Bay, Philippines. J. Appl. Phycol. 2007, 19, 607–613. [Google Scholar] [CrossRef]
- Lehman, E.M. Seasonal occurrence and toxicity of Microcystis in impoundments of the Huron River, Michigan, USA. Water Res. 2007, 41, 795–802. [Google Scholar] [CrossRef]
- Ha, J.H.; Hidaka, T.; Tsuno, H. Quantification of toxic Microcystis and evaluation of its dominance ratio in blooms using real-time PCR. Environ. Sci. Technol. 2009, 43, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Gobler, C.J.; Davis, T.W.; Coyne, K.J.; Boyer, G.L. The interactive influences of nutrient loading and zooplankton grazing on the growth and toxicity of cyanobacteria blooms in a eutrophic lake. Harmful Algae 2007, 6, 119–133. [Google Scholar] [CrossRef]
- Moisander, P.H.; Ochiai, M.; Lincoff, A. Nutrient limitation of Microcystis aeruginosa in northern California Klamath River reservoirs. Harmful Algae 2009, 8, 889–897. [Google Scholar] [CrossRef]
- Davis, T.W.; Harke, M.J.; Marcoval, M.A.; Goleski, J.; Orano-Dawson, C.; Berry, D.L.; Gobler, C.J. Effects of nitrogenous compounds and phosphorus on the growth of toxic and non-toxic strains of Microcystis during cyanobacterial blooms. Aquat. Microb. Ecol. 2010, 61, 149–162. [Google Scholar] [CrossRef]
- López-Ballesteros, A.; Trolle, D.; Srinivasan, R.; Senent-Aparicio, J. Assessing the effectiveness of potential best management practices for science-informed decision support at the watershed scale: The case of the Mar Menor coastal lagoon, Spain. Sci. Total Environ. 2023, 859, 160144. [Google Scholar] [CrossRef] [PubMed]
- Maté-Sánchez-Val, M.; Aparicio-Serrano, G. The impact of marine pollution on the probability of business failure: A case study of the Mar Menor lagoon. J. Environ. Manag. 2023, 332, 117381. [Google Scholar] [CrossRef]
- Conesa, H.M.; Jiménez-Cárceles, F.J. The Mar Menor lagoon (SE Spain): A singular natural ecosystem threatened by human activities. Mar. Pollut. Bull. 2007, 54, 839–849. [Google Scholar] [CrossRef]
- Lloret, J.; Marín, A.; Marín-Guirao, L. Is coastal lagoon eutrophication likely to be aggravated by global climate change? Estuar. Coast. Shelf Sci. 2008, 78, 403–412. [Google Scholar] [CrossRef]
- Gilabert, J. Seasonal plankton dynamics in a Mediterranean hypersaline coastal lagoon: The Mar Menor. J. Plankton Res. 2001, 23, 207–218. [Google Scholar] [CrossRef]
- Vázquez-Luis, M.; Álvarez, E.; Barrajón, A.; García-March, J.R.; Grau, A.; Hendriks, I.E.; Jiménez, S.; Kersting, D.; Moreno, D.; Pérez, M.; et al. S.O.S. Pinna nobilis: A Mass Mortality Event in Western Mediterranean Sea. Front. Mar. Sci. 2017, 4, 220. [Google Scholar] [CrossRef]
- Cabanellas-Reboredo, M.; Vázquez-Luis, M.; Mourre, B.; Álvarez, E.; Deudero, S.; Amores, Á.; Addis, P.; Ballesteros, E.; Barrajón, A.; Coppa, S.; et al. Tracking a mass mortality outbreak of pen shell Pinna nobilis populations: A collaborative effort of scientists and citizens. Sci. Rep. 2019, 9, 13355. [Google Scholar] [CrossRef] [PubMed]
- Cortés-Melendreras, E.; Gomariz-Castillo, F.; Alonso-Sarría, F.; Martín, F.J.G.; Murcia, J.; Canales-Cáceres, R.; Esplá, A.A.R.; Barberá, C.; Giménez-Casalduero, F. The relict population of Pinna nobilis in the Mar Menor is facing an uncertain future. Mar. Pollut. Bull. 2022, 185, 114376. [Google Scholar] [CrossRef] [PubMed]
- Castejón-Porcel, G.; Espín-Sánchez, D.; Ruiz-Álvarez, V.; García-Marín, R.; Moreno-Muñoz, D. Runoff water as a resource in the Campo de Cartagena (Region of Murcia): Current possibilities for use and benefits. Water 2018, 10, 456. [Google Scholar] [CrossRef]
- Medidas Para la Recuperación del Acuífero del Campo de Cartagena y Reducción de los Aportes de Nitrógeno al Mar Menor. Instituto de Ingeniería del Agua y Medio Ambiente. Universitat Politècnica de València. IIAMA-UPV. 2020. Available online: https://www.chsegura.es/export/sites/chs/descargas/cuenca/marmenor/docsdescarga/InformeUPVSeguraCampoCartagena_20210914.pdf (accessed on 1 June 2023).
- Alcolea, A.; Contreras, S.; Huninkb, J.E.; García-Aróstegui, J.L.; Jiménez-Martínez, J. Hydrogeological modelling for the watershed management of the Mar Menor coastal lagoon (Spain). Sci. Total Environ. 2019, 663, 901–914. [Google Scholar] [CrossRef]
- Servicios de Consultoría y Asistencia Técnica para la Simulación Bajo Distintos Escenarios y Mediante Modelo Hidrológico, de la Evolución del Contenido en Nitratos de la Masa de Agua Subterránea 070.052 Campo de Cartagena. (TTMM: Varios. Provincias: Murcia y Alicante). Instituto de Ingeniería del Agua y Medio Ambiente. Universitat Politècnica de València. IIAMA-UPV. 2021. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwiS9IrJw--BAxWomlYBHdGHBRAQFnoECAgQAQ&url=https%3A%2F%2Fwww.chsegura.es%2Fexport%2Fsites%2Fchs%2Fdescargas%2Fcuenca%2Fmarmenor%2Fdocsdescarga%2FInformeUPVSeguraCampoCartagena_20210914.pdf&usg=AOvVaw3Odm17Z-nHQ-7gnh_J1s-p&opi=89978449 (accessed on 1 June 2023).
- Modelo de Flujo. Acuífero Cuaternario del Campo de Cartagena: “Cuantificación, Control de la Calidad y Seguimiento Piezométrico de la Descarga de Agua Subterránea del Acuífero Cuaternario del Campo de Cartagena al Mar Menor. 2021 Tragsatec. Available online: https://www.chsegura.es/static/marmenor/DescargasMarMenorCuaternarioCampoCartagena.zip (accessed on 1 June 2023).
- Pérez-Martín, M.; Benedito-Castillo, S. Fertigation to Recover Nitrate-Polluted Aquifer and Improve a Long Time Eutrophicated Lake, Spain. Sci. Total Environ. 2023, 894, 165020. [Google Scholar] [CrossRef]
- López-Andreu, F.J.; López-Morales, J.A.; Juárez, J.F.A.; Alcaraz, R.; Hernández, M.D.; Erena, M.; Domínguez-Gómez, J.A.; Galiano, S.G. Monitoring System of the Mar Menor Coastal Lagoon (Spain) and Its Watershed Basin Using the Integration of Massive Heterogeneous Data. Sensors 2022, 22, 6507. [Google Scholar] [CrossRef]
- Erena, M.; Domínguez, J.A.; Aguado, F.; Soria, J.; Galiano, G. Monitoring Coastal Lagoon Water Quality through Remote Sensing: The Mar Menor as a Case Study. Water 2019, 11, 1468. [Google Scholar] [CrossRef]
- Angel, P.-R.; Marcos, C.; Gilabert, J. The Ecology of the Mar Menor Coastal Lagoon: A Fast Changing Ecosystem under Human Pressure. Coastal Lagoons. In Ecosystem Processes and Modeling for Sustainable Use and Development; Gönenç, I.E., Wolflin, J.P., Eds.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- García-Oliva, M.; Marcos, C.; Umgiesser, G.; McKiver, W.; Ghezzo, M.; De Pascalis, F.; Pérez-Ruzafa, A. Modelling the impact of dredging inlets on the salinity and temperature regimes in coastal lagoons. Ocean Coast. Manag. 2019, 180, 104913. [Google Scholar] [CrossRef]
- Ruiz, J.M.; Albentsosa, M.; Aldeguer, B.; Alvarez-Rogel, J.; Yebra, L. Informe de Evolución y Estado Actual del Mar Menor en Relación al Proceso de Eutrofización y Sus Causas 2020; Technical Report Instituto Español de Oceanografía (IEO); Spanish Institute of Oceanography: Madrid, Spain, 2020; p. 165. Available online: https://www.miteco.gob.es/es/prensa/informe-ieo-mar-menor.aspx (accessed on 11 December 2022).
- Bernardeau-Esteller, J.; Sandoval-Gil, J.M.; Belando, M.D.; Ramos-Segura, A.; García-Muñoz, R.; Marín-Guirao, L.; Ruiz, J.M. The Role of Cymodocea nodosa and Caulerpa prolifera Meadows as Nitrogen Sinks in Temperate Coastal Lagoons. Diversity 2023, 15, 172. [Google Scholar] [CrossRef]
- Arévalo, L. El Mar Menor como sistema forzado por el Mediterráneo. Boletín Instituo Español Oceanogr. 1988, 5, 61–95. [Google Scholar]
- IEO. Estudio Oceanográfico Integral de alta Resolución de la Laguna Costera del Mar Menor, Murcia. Primer Informe Técnico: Campaña MM1116 (Otoño). 2018. Available online: https://www.ulpgc.es/sites/default/files/ArchivosULPGC/noticia/2017/Feb/estudio-ieo-mar-menor.pdf (accessed on 1 June 2023).
- Martínez-Alvarez, V.; Gallego-Elvira, B.; Maestre-Valero, J.; Tanguy, M. Simultaneous solution for water, heat and salt balances in a Mediterranean coastal lagoon (Mar Menor, Spain). Estuar. Coast. Shelf Sci. 2011, 91, 250–261. [Google Scholar] [CrossRef]
- López-Castejón, F. Caracterización de la Hidrodinámica del Mar Menor y los Flujos de Intercambio con el Mediterráneo Mediante Datos in situ y Modelado Numérico. Ph.D. Thesis, Technical University of Cartagena, Cartagena, Spain, 2017. [Google Scholar] [CrossRef]
- Senent-Aparicio, J.; López-Ballesteros, A.; Nielsen, A.; Trolle, D. A holistic approach for determining the hydrology of the mar menor coastal lagoon by combining hydrological & hydrodynamic models. J. Hydrol. 2021, 603, 127150. [Google Scholar] [CrossRef]
- Torrente, M.D.B.; Ruiz, J.M.; Muñoz, R.G.; Segura, A.R.; Esteller, J.B.; Casero, J.J.; Guirao, L.M.; Moreno, P.G.; Navarro, I.F.; Nuez, E.F.; et al. Collapse of macrophytic communities in a eutrophicated coastal lagoon. Front. Mar. Sci. 2019. [Google Scholar] [CrossRef]
- IEO. Informe de Síntesis Sobre el Estado Actual del Mar Menor y sus Causas en Relación a los Contenidos de Nutrients. 2019. Available online: https://www.ieo.es/documents/10640/7708614/1_Informe+sobre+la+evolucion+del+Mar+Menor+y+sus+causas_noviembre+2019+final+B.pdf/564902d9-d41e-4e45-b730-9e561411c977 (accessed on 1 June 2023).
- UPCT (Universidad Politécnica de Cartagena). Servidor de Datos Científicos del Mar Menor (SDC). 2023. Available online: https://marmenor.upct.es/ (accessed on 10 February 2023).
- Ruiz, J.M.; Clemente-Navarro, P.; Mercado, J.M.; Fraile-Nuez, E.; Albentosa, M.; Marín-Guirao, L.; Santos, J. Nuevo Evento de Mortalidad Masiva de Organismos Marinos en el Mar Menor: Contexto y Factores. Instituto Español de Oceanografía IEO. September 2021. Available online: https://www.ieo.es/documents/10640/7708614/3_informe+IEO_FINAL2_MGB_060921.pdf/cb4859a4-4da4-4855-82ec-8fb4167790c3 (accessed on 1 June 2023).
- Royal Decrete 817/2015, de 11 de Septiembre, por el que se Establecen los Criterios de Seguimiento y Evaluación del Estado de las Aguas Superficiales y las Normas de Calidad Ambiental. Available online: https://www.boe.es/buscar/pdf/2015/BOE-A-2015-9806-consolidado.pdf (accessed on 1 June 2023).
- Pérez-Martín, M.A.; Estrela, T.; Del-Amo, P. Measures required to reach the nitrate objectives in groundwater based on a long-term nitrate model for large river basins (Júcar, Spain). Sci. Total Environ. 2016, 566–567, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Martín, M.A.; Estrela, T.; Andreu, J.; Ferrer, J. Modeling Water Resources and River-Aquifer Interaction in the Júcar River Basin, Spain. Water Resour. Manag. 2014, 28, 4337–4358. [Google Scholar] [CrossRef]
- Mapama. “Balance del Nitrógeno en la Agricultura Española, 2019”. Ministerio de Agricultura y Pesca, Alimentación y Medio Ambiente. 2021. Available online: https://www.mapa.gob.es/es/agricultura/temas/medios-de-produccion/01_modelnationallevelncpsa_ae_110n_spain_junio2021_tcm30-620003.xlsx (accessed on 1 June 2023).
- Jensen, J.P.; Pedersen, A.R.; Jeppesen, E.; Søndergaard, M. An empirical model describing the seasonal dynamics of phosphorus in 16 shallow eutrophic lakes after external loading reduction. Limnol. Oceanogr. 2006, 51, 791–800. [Google Scholar] [CrossRef]
- Savva, I.; Bennett, S.; Roca, G.; Jordà, G.; Marbà, N. Thermal tolerance of Mediterranean marine macrophytes: Vulnerability to global warming. Ecol. Evol. 2018, 8, 12032–12043. [Google Scholar] [CrossRef]
- Caraco, N.; Cole, J.; Likens, G.E. A comparison of phosphorus immobilization in sediments of freshwater and coastal marine systems. Biogeochemistry 1990, 9, 277–290. [Google Scholar] [CrossRef]
- Bennett, S.; Vaquer-Sunyer, R.; Jorda, G.; Forteza, M.; Roca, G.; Marbà, N. Thermal Performance of Seaweeds and Seagrasses Across a Regional Climate Gradient. Front. Mar. Sci. 2022, 9, 733315. [Google Scholar] [CrossRef]
- Marbà, N.; Jordà, G.; Bennett, S.; Duarte, C.M. Seagrass Thermal Limits and Vulnerability to Future Warming. Front. Mar. Sci. 2022, 9, 860826. [Google Scholar] [CrossRef]
- Tsioli, S.; Orfanidis, S.; Papathanasiou, V.; Katsaros, C.; Exadactylos, A. Effects of salinity and temperature on the performance of Cymodocea nodosa and Ruppia cirrhosa: A medium-term laboratory study. Bot. Mar. 2018, 62, 97–108. [Google Scholar] [CrossRef]
- Servicio y desarrollo de la red de control y vigilancia de la calidad de las aguas litorales para el seguimiento del estado de las masas de agua costeras en la Región de Murcia. 2021 Ref: DT2021/014 2021 Servicio de planificación y evaluación ambiental (CARM). Informe Final Taxon.
- Wild, A. (Ed.) Russell’s Soil Conditions and Plant Growth; Longman Scientific and Technical: Harlow, UK, 1988. [Google Scholar]
- McGlathery, K.; Sundbäck, K.; Anderson, I. Eutrophication in shallow coastal bays and lagoons: The role of plants in the coastal filter. Mar. Ecol. Prog. Ser. 2007, 348, 1–18. [Google Scholar] [CrossRef]
- Jilbert, T.; Couture, R.-M.; Huser, B.J.; Salonen, K. Preface: Restoration of eutrophic lakes: Current practices and future challenges. Hydrobiologia 2020, 847, 4343–4357. [Google Scholar] [CrossRef]
- Pérez-Ruzafa, A.; Campillo, S.; Fernández-Palacios, J.M.; García-Lacunza, A.; García-Oliva, M.; Ibañez, H.; Navarro-Martínez, P.C.; Pérez-Marcos, M.; Pérez-Ruzafa, I.M.; Quispe-Becerra, J.I.; et al. Long-Term Dynamic in Nutrients, Chlorophyll a, and Water Quality Parameters in a Coastal Lagoon During a Process of Eutrophication for Decades, a Sudden Break and a Relatively Rapid Recovery. Front. Mar. Sci. 2019, 6, 26. [Google Scholar] [CrossRef]

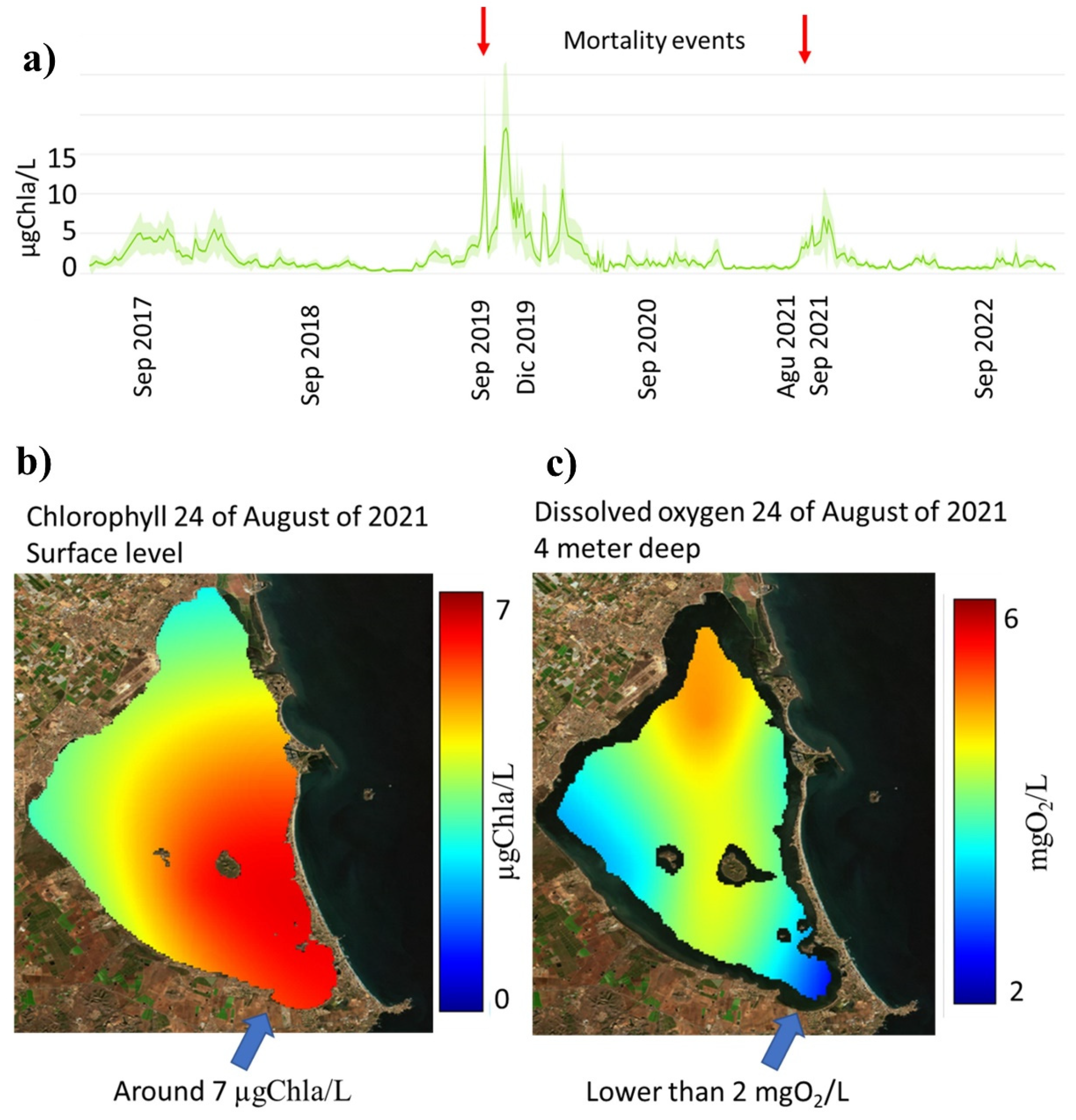
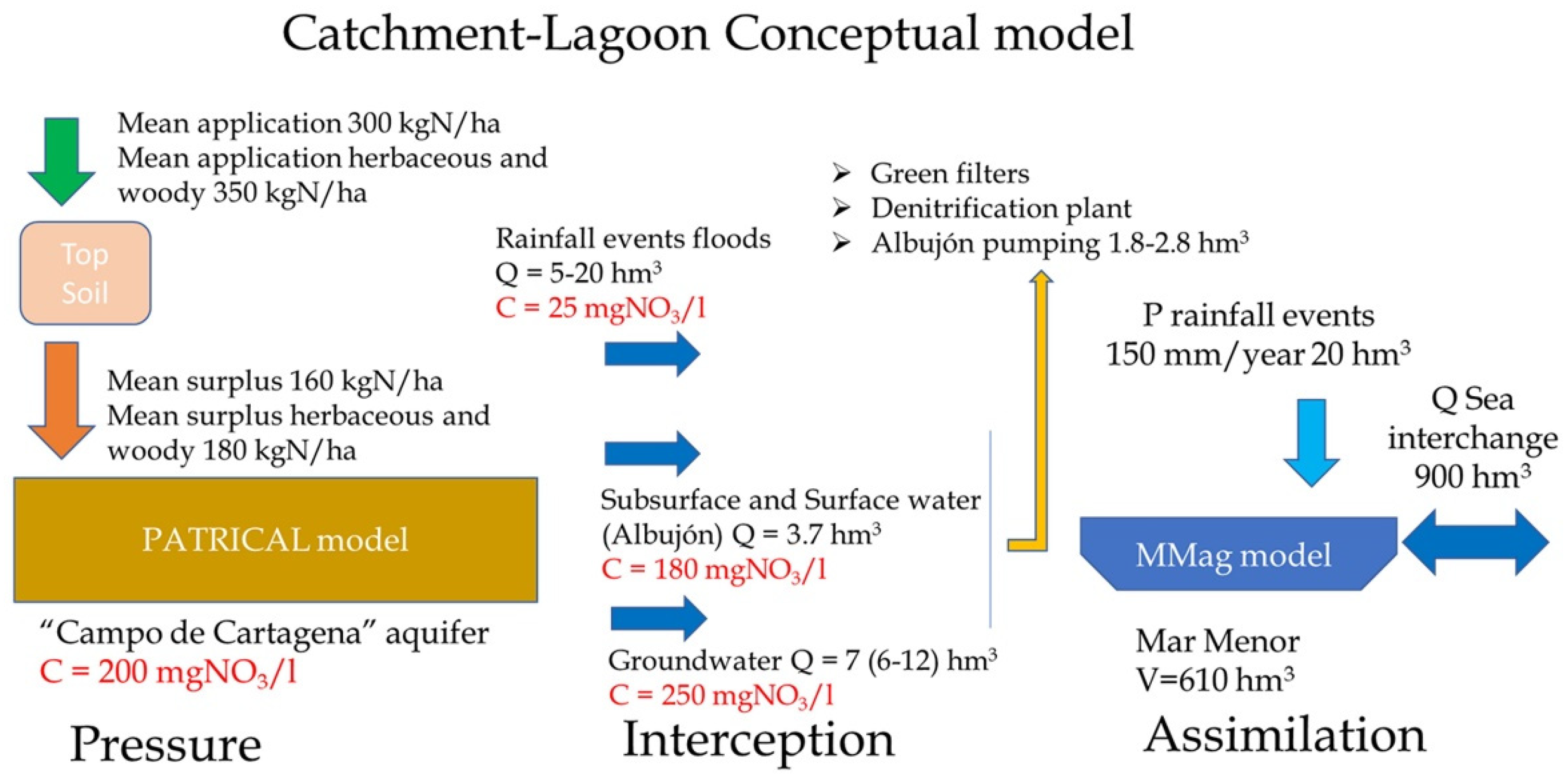
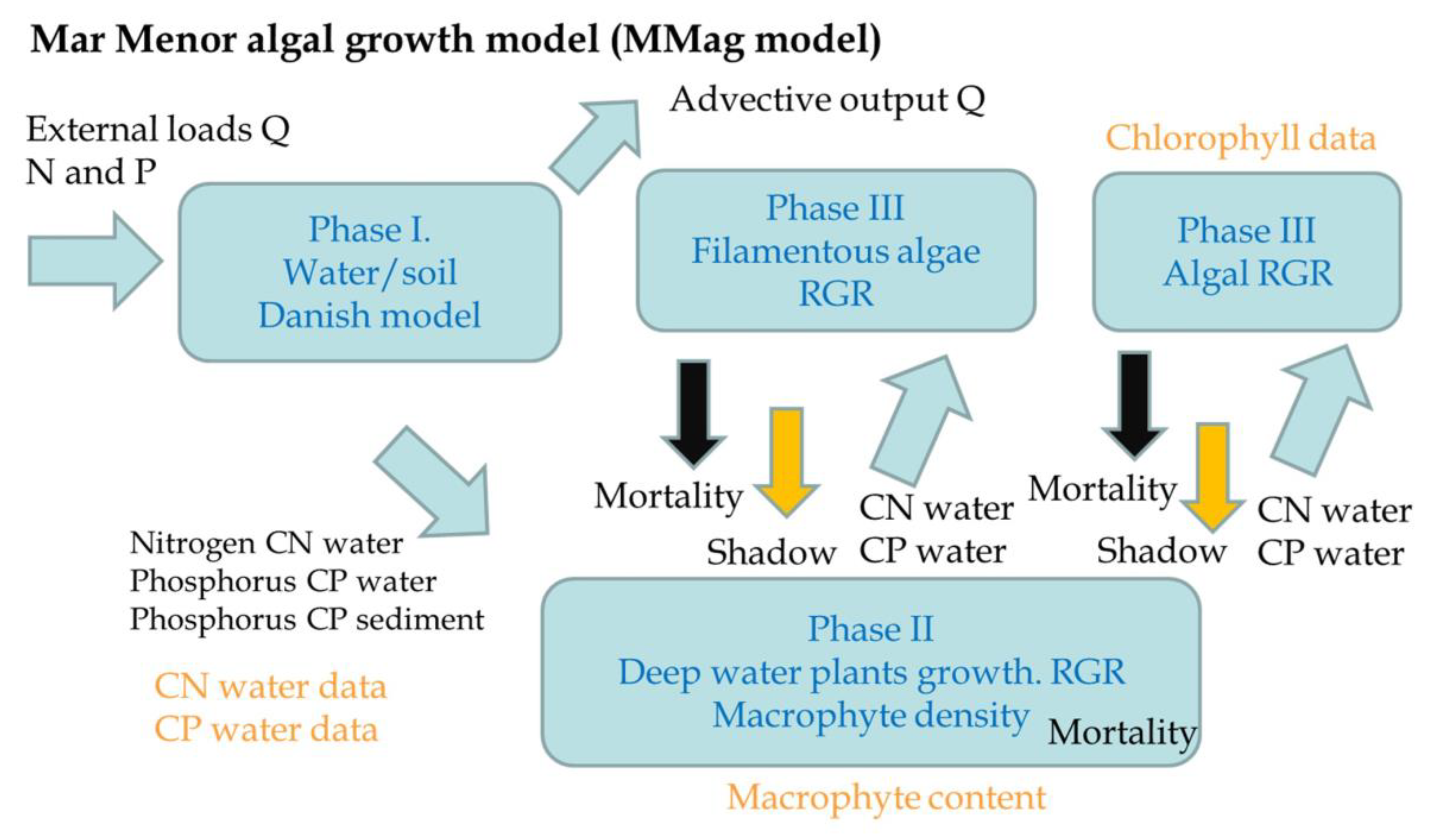
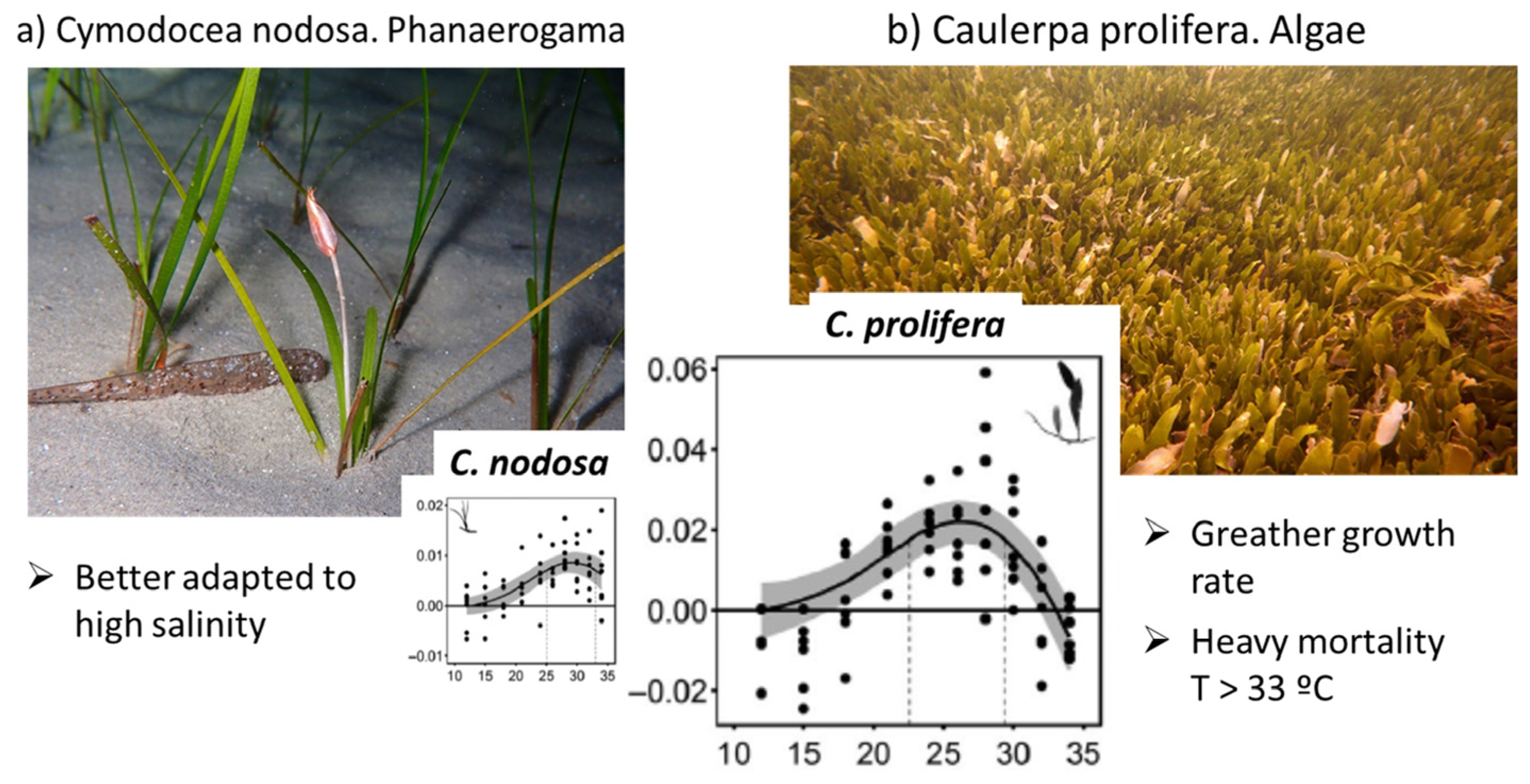

| Flow Temporally | Flow Source | Volume hm3/Year hm3/Event (a) | Nitrate mgNO3/L (b) | Nitrate tnNO3/Year tnNO3/Event a × b | Total Phosphorus mgP/L (c) | Total Phosphorus tnP/Year tnP/Event a × c |
|---|---|---|---|---|---|---|
| Continuous year | Surface and subsurface water | 3.7 | 180 | 672 | 0.5 | 1.9 |
| Continuous year | Groundwater | 7.0 | 250 | 1750 | 0.0 | 0.0 |
| Annual | Total | 10.7 | 2421 | 0.5 | 1.9 | |
| Small and frequent events | Surface | 5 | 25 | 125 | 0.5–1.0 | 2.5–5.0 |
| Extreme Events | Surface | 20 | 25 | 500 | 0.5–1.0 | 10–20 |
| Gates, Sea Connection | Main Flow Direction | hm3/Day | hm3/Year |
|---|---|---|---|
| Las Encañizadas | outflow | −0.30 | −109.5 |
| El Estacio | inflow | 2.06 | 751.9 |
| outflow | −1.76 | −642.4 | |
| netflow | 0.30 | 109.5 | |
| Marchamalo | inflow | 0.47 | 171.6 |
| Total input flow | inflow | 2.53 | 923.5 |
| Total Net Flow | inflow | 0.47 | 171.6 |
| Phase | Component | Parameter | Calibrated | Dimension | Description | Sensitivity |
|---|---|---|---|---|---|---|
| I | Danish model Phosphorus | bF | 0.002975 | - | Sedimentation rate | High |
| tF | 0.4 | - | T dependence | High | ||
| bS | 0.047 | - | Sedimentation release | Low | ||
| Tl | 20 | °C | Temperature threshold | High | ||
| Nitrogen | K’ | 2.6 | 1/year | Nitrogen sedimentation | Medium | |
| II | Deep species | RGR 30 °C | 10 | mg/g/day | Relative Growth Rate | Very High |
| RGR 20 °C | 4 | mg/g/day | Relative Growth Rate | Very High | ||
| Return P | 35% | % | High | |||
| Return N | 10% | % | High | |||
| Mortality | 0.018 | days−1 | High | |||
| III | Filamentous | RGR 30 °C | 30 | mg/g/day | Relative Growth Rate | High |
| RGR 20 °C | 12 | mg/g/day | Relative Growth Rate | High | ||
| Return P | 40% | % | Low | |||
| Return N | 10% | % | Low | |||
| Mortality | 0.006 | days−1 | High | |||
| Algae | RGR 30° | 75 | mg/g/day | Relative Growth Rate | High | |
| RGR 20° | 22 | mg/g/day | Relative Growth Rate | High | ||
| Return P | 40% | % | Low | |||
| Return N | 10% | % | Low | |||
| Mortality | 0.014 | days−1 | High |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Martín, M.Á. Understanding Nutrient Loads from Catchment and Eutrophication in a Salt Lagoon: The Mar Menor Case. Water 2023, 15, 3569. https://doi.org/10.3390/w15203569
Pérez-Martín MÁ. Understanding Nutrient Loads from Catchment and Eutrophication in a Salt Lagoon: The Mar Menor Case. Water. 2023; 15(20):3569. https://doi.org/10.3390/w15203569
Chicago/Turabian StylePérez-Martín, Miguel Ángel. 2023. "Understanding Nutrient Loads from Catchment and Eutrophication in a Salt Lagoon: The Mar Menor Case" Water 15, no. 20: 3569. https://doi.org/10.3390/w15203569




