Does Fipronil Affect on Aquatic Organisms? Physiological, Biochemical, and Histopathological Alterations of Non-Target Freshwater Mussel Species
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Organisms
2.2. Chemicals
2.3. Acute Experiments
2.4. Sublethal Experiments
2.5. Total Haemocyte Counts
2.6. Biochemical Analysis
2.7. Histopathological Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Acute Toxic Effects of Fipronil on the Freshwater Mussels
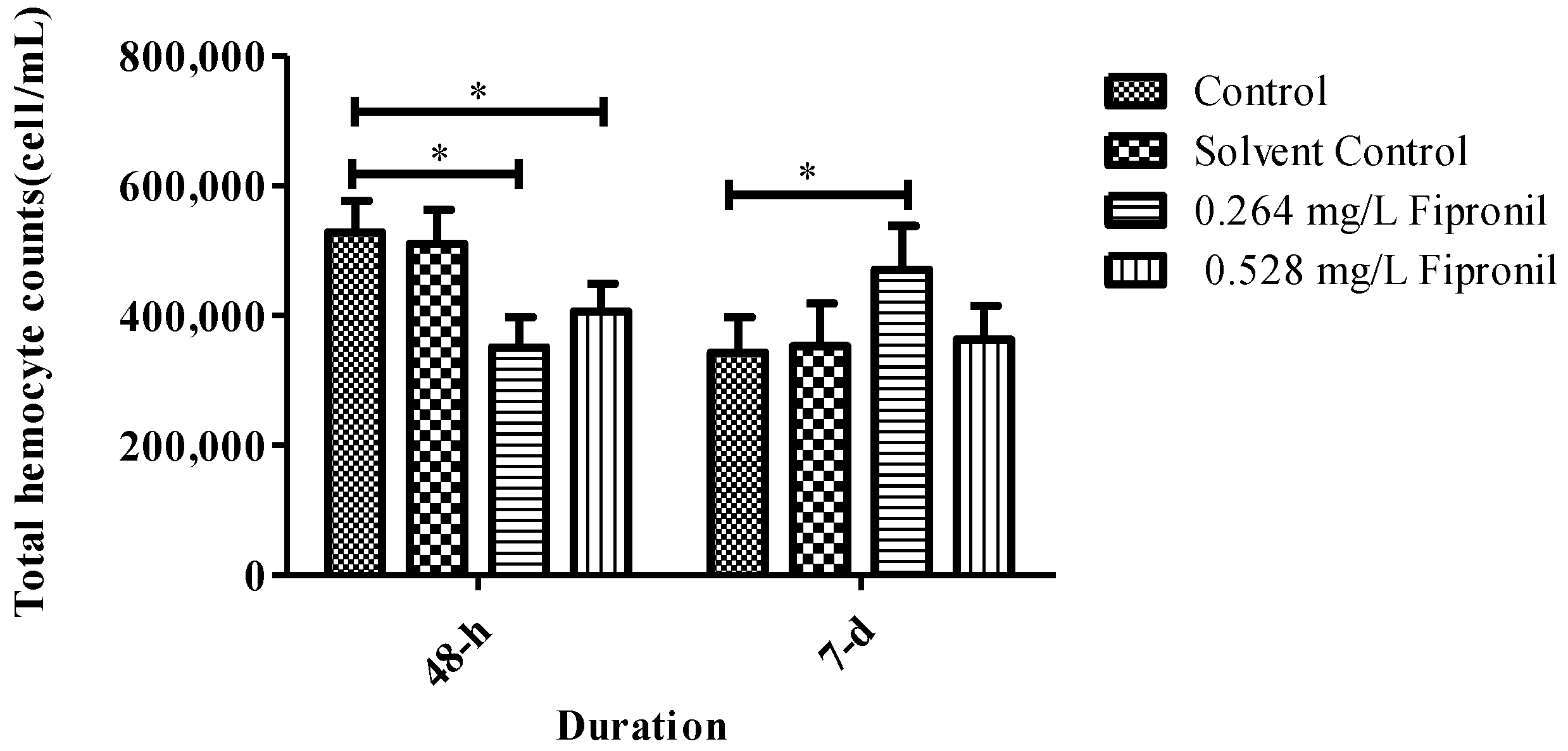
3.2. Total Haemocyte Counts
3.3. Biochemical Parameters
3.4. PCA Results
3.5. Histology Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tingle, C.C.D.; Rother, J.A.; Dewhurst, C.F.; Lauer, S.; King, W.J. Fipronil: Environmental fate, ecotoxicology, and human health concerns. In Reviews of Environmental Contamination and Toxicology; Ware, G.W., Ed.; Springer: New York, NY, USA, 2003; Volume 176. [Google Scholar] [CrossRef]
- Pisa, L.W.; Amaral-Rogers, V.; Belzunces, L.P.; Bonmantin, J.M.; Downs, C.A.; Goulson, D.; Kreutzweiser, D.P.; Krupke, C.; Liess, M.; McField, M.; et al. Effects of neonicotinoids and fipronil on non-target invertebrates. Environ. Sci. Pollut. Res. 2015, 22, 68–102. [Google Scholar] [CrossRef] [Green Version]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 3352, Fipronil. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Fipronil (accessed on 18 November 2022).
- USEPA. Section 18 Ecological Risk Assessment for Fipronil Use to Control Cabbage—Maggot in Turnip and Rutabaga. 2005. Available online: https://www3.epa.gov/pesticides/chem_search/cleared_reviews/csr_PC-129121_31-Aug-05_a.pdf (accessed on 29 August 2022).
- Simon-Delso, N.; Amaral-Rogers, V.; Belzunces, L.P.; Bonmantin, J.M.; Chagnon, M.; Downs, C.; Furlan, L.; Gibbons, D.W.; Giorio, C.; Girolami, V.; et al. Systemic insecticides (neonicotinoids and fipronil): Trends, uses, mode of action and metabolites. Environ. Sci. Pollut. Res. 2015, 22, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Bonmatin, J.M.; Giorio, C.; Girolami, V.; Goulson, D.P.; Kreutzweiser, D.; Krupke, C.; Liess, M.; Long, E.; Marzaro, M.; Mitchell, E.A.D.; et al. Environmental fate and exposure; neonicotinoids and fipronil. Environ. Sci. Pollut. Res. 2015, 22, 35–67. [Google Scholar] [CrossRef] [PubMed]
- Jerome, F.C.; Hassan, A.; Omoniyi-Esan, G.O.; Odujoko, O.O.; Chukwuka, A.V. Metal uptake, oxidative stress and histopathological alterations in gills and hepatopancreas of Callinectes amnicola exposed to industrial effluent. Ecotoxicol. Environ. Saf. 2017, 139, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Bal, A.; Panda, F.; Pati, S.G.; Das, K.; Agrawal, P.K.; Paital, B. Modulation of physiological oxidative stress and antioxidant status by abiotic factors especially salinity in aquatic organisms. Comp. Biochem. Physiol. Part C 2021, 241, 108971. [Google Scholar] [CrossRef] [PubMed]
- Abbas, E.A.; Mowafy, R.E.; Khalil, A.A.; Sdeek, F.A. The potential role of the dietary addition of bentonite clay powder in mitigating diazinon-induced hepatorenal damage, oxidative stress, and pathological alterations in Nile tilapia. Aquaculture 2021, 533, 736182. [Google Scholar] [CrossRef]
- Vona, R.; Pallotta, L.; Cappelletti, M.; Severi, C.; Matarrese, P. The impact of oxidative stress in human pathology: Focus on gastrointestinal disorders. Antioxidants 2021, 10, 201. [Google Scholar] [CrossRef]
- Xu, Z.; Cao, J.; Qin, X.; Qiu, W.; Mei, J.; Xie, J. Toxic effects on bioaccumulation, hematological parameters, oxidative stress, immune responses and tissue structure in fish exposed to ammonia nitrogen: A Review. Animals 2021, 11, 3304. [Google Scholar] [CrossRef]
- Büyükuslu, N.; Yiğitbaşı, T. Reactive oxygen species and oxidative stress in obesity. Clin. Exp. Health Sci. 2015, 5, 197–203. [Google Scholar] [CrossRef]
- Selmeci, L. Advanced oxidation protein products (AOPP): Novel uremic toxins, or components of the non-enzymatic antioxidant system of the plasma proteome? Free Radic. Res. 2011, 45, 1115–1123. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlahogianni, T.; Dassenakis, M.; Scoullos, M. Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol. Environ. Saf. 2006, 64, 178–189. [Google Scholar] [CrossRef]
- Ficociello, G.; Inverni, A.; Massimi, L.; Buccini, G.; Canepari, S.; Uccelletti, D. Assessment of the effects of atmospheric pollutants using the animal model Caenorhabditis elegans. Environ. Res. 2020, 191, 110209. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Ko, E.; Lee, H.; Kim, K.T.; Choi, M.; Shin, S. Mixed exposure of persistent organic pollutants alters oxidative stress markers and mitochondrial function in the tail of zebrafish depending on sex. Int. J. Environ. Res. Public Health 2021, 18, 9539. [Google Scholar] [CrossRef] [PubMed]
- Duan, S.; Fu, Y.; Dong, S.; Ma, Y.; Meng, H.; Guo, R.; Chen, J.; Liu, Y.; Li, Y. Psychoactive drugs citalopram and mirtazapine caused oxidative stress and damage of feeding behavior in Daphnia magna. Ecotoxicol. Environ. Saf. 2022, 230, 113147. [Google Scholar] [CrossRef]
- Özkan-Kotiloğlu, S.; Arslan, P.; Akca, G.; Günal, A.Ç. Are BPA-free plastics safe for aquatic life?-Fluorene-9-bisphenol induced thyroid-disrupting effects and histopathological alterations in adult zebrafish (Danio rerio). Comp. Biochem. Physiol. Part C 2022, 260, 109419. [Google Scholar] [CrossRef] [PubMed]
- Wolf, J.C. Alternative Animal Models. In Wallig, Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed.; Wanda, M., Haschek, C., Rousseaux, G., Matthew, A., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 477–518. [Google Scholar]
- Holtze, S.; Gorshkova, E.; Braude, S.; Cellerino, A.; Dammann, P.; Hildebrandt, T.B.; Hoeflich, A.; Hoffmann, S.; Koch, P.; Tozzini, E.T.; et al. Alternative animal models of aging research. Front. Mol. Biosci. 2021, 8, 660959. [Google Scholar] [CrossRef] [PubMed]
- Günal, A.Ç.; Tunca, S.K.; Arslan, P.; Gül, G.; Sepici Dinçel, A. How does sublethal permethrin effect non-target aquatic organisms? Environ. Sci. Pollut. Res. 2021, 28, 52405–52417. [Google Scholar] [CrossRef]
- De Marco, G.; Afsa, S.; Galati, M.; Guerriero, G.; Mauceri, A.; Ben Mansour, H.; Cappello, T. Time-and dose-dependent biological effects of a sub-chronic exposure to realistic doses of salicylic acid in the gills of mussel Mytilus galloprovincialis. Environ. Sci. Pollut. Res. 2022, 29, 88161–88171. [Google Scholar] [CrossRef]
- Afsa, S.; De Marco, G.; Giannetto, A.; Parrino, V.; Cappello, T.; ben Mansour, H.; Maisano, M. Histological endpoints and oxidative stress transcriptional responses in the Mediterranean mussel Mytilus galloprovincialis exposed to realistic doses of salicylic acid. Environ. Toxicol. Pharmacol. 2022, 92, 103855. [Google Scholar] [CrossRef]
- Rahim, N.F.; Yaqin, K. Histological Alteration of Green Mussel Perna viridis Organs Exposed to Microplastics. Squalen Bull. Mar. Fish. Postharvest Biotechnol. 2022, 17, 44–53. [Google Scholar] [CrossRef]
- Günal, A.Ç.; Erkmen, B.; Katalay, S.; Ayhan, M.M.; Gül, G.; Erkoç, F. Determinations of the effects antifouling copper pyrithione on total hemocyte counts of mussel (Mytilus galloprovincialis). Ege JFAS 2018, 35, 15–17. [Google Scholar] [CrossRef]
- Yurdakök-Dikmen, B.; Arslan, P.; Kuzukıran, Ö.; Filazi, A.; Erkoç, F. Unio sp. primary cell culture potential in ecotoxicology research. Toxin Rev. 2017, 37, 75–81. [Google Scholar] [CrossRef]
- Yurdakök Dikmen, B.; Filazi, A.; Arslan, P. Invertebrate Cell Cultures in Ecotoxicology in Alternative Methods Used in Drug Research, Development and Toxicological Studies, 1st ed.; Güvenç, D., Ed.; Turkiye Klinikleri: Ankara, Türkiye, 2018; pp. 58–64. [Google Scholar]
- Třešňáková, N.; Günal, A.Ç.; Başaran Kankılıç, G.; Paçal, E.; Uyar, R.; Erkoç, F. Sub-lethal toxicities of zinc pyrithione, copper pyrithione alone and in combination to the indicator mussel species Unio crassus Philipsson, 1788 (Bivalvia, Unionidae). Chem. Ecol. 2020, 36, 292–308. [Google Scholar] [CrossRef]
- Arslan, P.; Yurdakok-Dikmen, B.; Kuzukiran, O.; Ozeren, S.C.; Filazi, A. Effects of acetamiprid and flumethrin on Unio sp. primary cells. Biologia 2021, 76, 1359–1365. [Google Scholar] [CrossRef]
- Arslan, P.; Yurdakok-Dikmen, B.; Ozeren, S.C.; Kuzukiran, O.; Filazi, A. In vitro effects of erythromycin and florfenicol on primary cell lines of Unio crassus and Cyprinus carpio. Environ. Sci. Pollut. Res. 2021, 28, 48408–48416. [Google Scholar] [CrossRef]
- Bogan, A.E. Global diversity of freshwater mussels (Mollusca, Bivalvia) in freshwater. Hydrobiologia 2008, 595, 139–147. [Google Scholar] [CrossRef]
- Lopes-Lima, M.; Gürlek, M.E.; Kebapçı, Ü.; Şereflişan, H.; Yanık, T.; Mirzajani, A.; Neubet, E.; Prie, V.; Teixeria, A.; Gomes-dos-Santos, A.; et al. Diversity, biogeography, evolutionary relationships, and conservation of Eastern Mediterranean freshwater mussels (Bivalvia: Unionidae). Mol. Phylogenet. Evol. 2021, 163, 107261. [Google Scholar] [CrossRef]
- OECD (Organization for Economic Co-Operation and Development). OECD Guidelines for Testing of Chemicals; OECD: Paris, France, 1993. [Google Scholar]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Sprague, J.B. Measurement of pollutant toxicity to fish—III: Sublethal effects and “safe” concentrations. Water Res. 1971, 5, 245–266. [Google Scholar] [CrossRef]
- Yavuzcan, H.Y.; Benli, A.Ç.K. Nitrite toxicity to crayfish, Astacus leptodactylus, the effects of sublethal nitrite exposure on hemolymph nitrite, total hemocyte counts, and hemolymph glucose. Ecotoxicol. Environ. Saf. 2004, 59, 370–375. [Google Scholar]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Wıtko-Sarsat, V.; Frıedlander, M.; Capeillere-Blandin, C.; Nguyen-Khoa, T.; Nguyen, A.T.; Zingraff, J.; Jungers, P.; Descamps-Latscha, B. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996, 49, 1304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luna, L.G. Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology; Blackiston Division, McGraw-Hill: New York, NY, USA, 1968. [Google Scholar]
- Benli, A.C.K.; Köksal, B.; Özkul, A. Sublethal ammonia exposure of Nile tilapia (Oreochromis niloticus L.): Effects on gill, liver and kidney histology. Chemosphere 2008, 72, 1355–1358. [Google Scholar] [CrossRef]
- LC50 Software Program, Version 1.00. Center for Exposure Assessment Modeling (CEAM). US EPA: Washington, DC, USA, 1999.
- Stara, A.; Pagano, M.; Capillo, G.; Fabrello, J.; Sandova, M.; Vazzana, I.; Zuskova, E.; Velisek, J.; Matozzo, V.; Faggio, C. Assessing the effects of neonicotinoid insecticide on the bivalve mollusc Mytilus galloprovincialis. Sci. Total Environ. 2020, 700, 134914. [Google Scholar] [CrossRef] [PubMed]
- Khudhur, S.M.; Shekha, Y.A.; Ahmed Shekha, Y. Histopathological and Biochemical Biomarker Response of Mussel, Unio pictorum, to Carbamate Pesticide Carbaryl: A Laboratory Study. Indian J. Ani. Res. 2018, 1157, 1–5. [Google Scholar] [CrossRef]
- Stara, A.; Pagano, M.; Albano, M.; Savoca, S.; Di Bella, G.; Albergamo, A.; Koutkova, Z.; Sandova, M.; Velisek, J.; Fabrello, J.; et al. Effects of long-term exposure of Mytilus galloprovincialis to thiacloprid: A multibiomarker approach. Environ. Pollut. 2021, 289, 117892. [Google Scholar] [CrossRef]
- Arslan, P. How Does Cyphenothrin Affect the Freshwater Mussel as In Vitro and In Vivo Models? Water Air Soil Pollut. 2022, 233, 386. [Google Scholar] [CrossRef]
- Dos Santos, G.P.C.; de Assis, C.R.D.; Oliveira, V.M.; Cahu, T.B.; Silva, V.L.; Santos, J.F.; Yogui, G.T.; Bezerra, R.S. Acetylcholinesterase from the charru mussel Mytella charruana: Kinetic characterization, physicochemical properties and potential as in vitro biomarker in environmental monitoring of mollusk extraction areas. Comp. Biochem. Physiol. Part C 2022, 252, 109225. [Google Scholar] [CrossRef]
- Connelly, P. Environmental Fate of Fipronil; Environmental Monitoring Branch, Department of Pesticide Regulation, California Environmental Protection Agency: Sacramento, CA, USA, 2001; pp. 1–17. [Google Scholar]
- Gunasekara, A.S.; Truong, T.; Goh, K.S.; Spurlock, F.; Tjeerdema, R.S. Environmental fate and toxicology of fipronil. J. Pest Sci. 2007, 32, 189–199. [Google Scholar] [CrossRef] [Green Version]
- Qu, H.; Ma, R.; Liu, D.; Jing, X.; Wang, F.; Zhou, Z.; Wang, P. The toxicity, bioaccumulation, elimination, conversion of the enantiomers of fipronil in Anodonta woodiana. J. Hazard. Mater. 2016, 312, 169–174. [Google Scholar] [CrossRef]
- Bringolf, R.B.; Cope, W.G.; Eads, C.B.; Lazaro, P.R.; Barnhart, M.C.; Shea, D. Acute and chronic toxicity of technical-grade pesticides to glochidia and juveniles of freshwater mussels (Unionidae). Environ. Toxicol. Chem. 2007, 26, 2086–2093. [Google Scholar] [CrossRef] [PubMed]
- Qyli, M.; Aliko, V.; Faggio, C. Physiological and biochemical responses of Mediterranean green crab, Carcinus aestuarii, to different environmental stressors: Evaluation of hemocyte toxicity and its possible effects on immune response. Comp. Biochem. Physiol. Part C 2020, 231, 108739. [Google Scholar] [CrossRef]
- Suresh, K.; Mohandas, A. Effect of sublethal concentrations of copper on hemocyte number in bivalves. J. Invertebr. Pathol. 1990, 55, 325–331. [Google Scholar] [CrossRef]
- Ayhan, M.M.; Katalay, S.; Günal, A.Ç. How pollution effects the immune systems of invertebrate organisms (Mytilus galloprovincialis Lamark, 1819). Mar. Pollut. Bull. 2021, 172, 112750. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.A.; Sarker, T.R.; Sutradhar, L.; Hussain, M.; Iqbal, M.M. Toxic effects of chlorpyrifos on the growth, hemocytes counts, and vital organ’s histopathology of freshwater mussel, Lamellidens marginalis. J. King Saud Uni. Sci. 2022, 35, 102482. [Google Scholar] [CrossRef]
- Ewere, E.E.; Reichelt-Brushett, A.; Benkendorff, K. The neonicotinoid insecticide imidacloprid, but not salinity, impacts the immune system of Sydney rock oyster, Saccostrea glomerata. Sci. Total Environ. 2020, 742, 140538. [Google Scholar] [CrossRef] [PubMed]
- Arslan, P. Determinations of the effects of cyfluthrin on the hemocytes parameters of freshwater mussel (Unio delicatus). Ege JFAS 2022, 39, 39–45. [Google Scholar] [CrossRef]
- Ali, M.M.; Sahar, T.; Firyal, S.; Ijaz, M.; Majeed, K.A.; Awan, F.; Adil, M.; Akbar, H.; Rashid, M.I.; Ciğerci, İ.H. Assessment of cytotoxic, genotoxic, and oxidative stress of dibutyl phthalate on cultured bovine peripheral lymphocytes. Oxid. Med. Cell. Longev. 2022, 9961513. [Google Scholar] [CrossRef]
- Li, X.; Naseem, S.; Hussain, R.; Ghaffar, A.; Li, K.; Khan, A. Evaluation of DNA Damage, Biomarkers of Oxidative Stress, and Status of Antioxidant Enzymes in Freshwater Fish (Labeo rohita) Exposed to Pyriproxyfen. Oxid. Med. Cell. Longev. 2022, 5859266. [Google Scholar] [CrossRef]
- de Almeida, E.A.; Bainy, A.C.D.; de Melo Loureiro, A.P.; Martinez, G.R.; Miyamoto, S.; Onuki, J.; Barbaso, L.F.; Garcia, C.C.M.; Prado, F.M.; Ronsein, G.E.; et al. Oxidative stress in Perna perna and other bivalves as indicators of environmental stress in the Brazilian marine environment: Antioxidants, lipid peroxidation and DNA damage. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007, 146, 588–600. [Google Scholar] [CrossRef]
- Mishchuk, O.V.; Stoliar, O.B. The effect of pesticide acetamiprid on biochemical markers in tissues of freshwater bivalve mussels Anodonta cygnea L. (Unionidae). Ukr. Kyi Biokhimichnyi Zhurnal 2008, 80, 117–124. [Google Scholar]
- Chen, S.; Yu, Y.; Gao, Y.; Yin, P.; Tian, L.; Niu, J.; Liu, Y. Exposure to acute ammonia stress influences survival, immune response and antioxidant status of pacific white shrimp (Litopenaeus vannamei) pretreated with diverse levels of inositol. Fish Shellfish Immunol. 2019, 89, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Belek, N.; Erkmen, B.; Dinçel, A.S.; Gunal, A.C. Does persistent organic pollutant PFOS (perfluorooctane sulfonate) negative impacts on the aquatic invertebrate organism, Astacus leptodactylus [Eschscholtz, 1823]. Ecotoxicology 2022, 31, 1217–1230. [Google Scholar] [CrossRef]
- Liess, M.; Henz, S.; Knillmann, S. Predicting low-concentration effects of pesticides. Sci. Rep. 2019, 9, 15248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qu, C.; Liu, S.; Tang, Z.; Li, J.; Liao, Z.; Qi, P. Response of a novel selenium-dependent glutathione peroxidase from thick shell mussel Mytilus coruscus exposed to lipopolysaccharide, copper and benzo[α]pyrene. Fish Shellfish Immunol. 2019, 89, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, J.M.; Sousa, V.S.; Teixeira, M.R.; Bebianno, M.J. Chronic toxicity of polystyrene nanoparticles in the marine mussel Mytilus galloprovincialis. Chemosphere 2022, 287, 132356. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, J.M.; Beckmann, C.; Bebianno, M.J. Assessing the effects of the cytostatic drug 5-Fluorouracil alone and in a mixture of emerging contaminants on the mussel Mytilus galloprovincialis. Chemosphere 2022, 305, 135462. [Google Scholar] [CrossRef]
- Canli, E.G.; Canli, M. Antioxidant system biomarkers of freshwater mussel (Unio tigridis) respond to nanoparticle (Al2O3, CuO, TiO2) exposures. Biomarkers 2021, 26, 434–442. [Google Scholar] [CrossRef]
- Rodrigo, A.P.; Costa, P.M. The Role of the Cephalopod Digestive Gland in the Storage and Detoxification of Marine Pollutants. Front. Physiol. 2017, 8, 232. [Google Scholar] [CrossRef] [Green Version]
- Arslan, P. Determination of The Oxidative Stress Effect of Chlorpyrifos Ethyl on the in vitro Models of the Freshwater Organisms. Future Biochemistry and Bioscience 2001, 3, 50–55. [Google Scholar]
- Bacha, O.; Khazri, A.; Mezni, A.; Mezni, A.; Touaylia, S. Protective effect of the Spirulina platensis against toxicity induced by Diuron exposure in Mytilus galloprovincialis. Int. J. Phytoremediation 2022, 24, 778–786. [Google Scholar] [CrossRef]
- Cappello, T.; De Marco, G.; Conti, G.O.; Giannetto, A.; Ferrante, M.; Mauceri, A.; Maisano, M. Time-dependent metabolic disorders induced by short-term exposure to polystyrene microplastics in the Mediterranean mussel Mytilus galloprovincialis. Ecotoxicol. Environ. Saf. 2021, 209, 111780. [Google Scholar] [CrossRef] [PubMed]
- Izral, N.M.; Brua, R.B.; Culp, J.M.; Yates, A.G. Crayfish tissue metabolomes effectively distinguish impacts of wastewater and agriculture in aquatic ecosystems. Sci. Total Environ. 2021, 760, 143322. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.; Costa, M.; Leite, C.; Borges, C.; Coppola, F.; Henriques, B.; Freitas, R. Ecotoxicological effects of lanthanum in Mytilus galloprovincialis: Biochemical and histopathological impacts. Aquat. Toxicol. 2019, 211, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Ardeshir, R.A.; Zolgharnein, H.; Movahedinia, A.; Salamat, N.; Zabihi, E. Comparison of waterborne and intraperitoneal exposure to fipronil in the Caspian white fish (Rutilus frisii) on acute toxicity and histopathology. Toxicol. Rep. 2017, 4, 348–357. [Google Scholar] [CrossRef] [PubMed]
- El-Murr, A.E.; Imam, T.S.; Hakim, Y.; Ghonimi, W.A.M. Histopathological, Immunological, Hematological and Biochemical Effects of Fipronil on Nile Tilapia (Oreochromis niloticus). J. Veterinar. Sci. Technol. 2015, 6, 252. [Google Scholar] [CrossRef]
- Ghaffar, A.; Hussain, R.; Abbas, G.; Kalim, M.; Khan, A.; Ferrando, S.; Gallus, L.; Ahmed, Z. Fipronil (Phenylpyrazole) induces hemato-biochemical, histological and genetic damage at low doses in common carp, Cyprinus carpio (Linnaeus, 1758). Ecotoxicology 2018, 27, 1261–1271. [Google Scholar] [CrossRef]
- Faggio, C.; Tsarpali, V.; Dailianis, S. Mussel digestive gland as a model tissue for assessing xenobiotics: An overview. Sci. Total Environ. 2018, 636, 220–229. [Google Scholar] [CrossRef]
- Bignell, J.P.; Dodge, M.J.; Feist, S.W.; Lyons, B.; Martin, P.D.; Taylor, N.G.H.; Stone, D.; Travalent, L.; Stentiford, G.D. Mussel histopathology: Effects of season, disease and species. Aquat. Biol. 2008, 2, 1–15. [Google Scholar] [CrossRef] [Green Version]
- da Cunha, E.L.R.; da Silva Matos, R.; Pereira, N.R.C.; de Oliveira, P.R.; Daemon, E.; Camargo-Mathias, M.I. Histopathological changes in the liver and thyroid of mice (Mus musculus) caused by the acaricides: Fipronil and thymol. J. Histol. Histopathol. 2017, 4, 9. [Google Scholar] [CrossRef] [Green Version]
- Kartheek, R.M.; David, M. Assessment of fipronil toxicity on Wistar rats: A hepatotoxic perspective. Toxicol. Rep. 2018, 5, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Balamurugan, S.; Subramanian, P. Histopathology of the Foot, Gill and Digestive Gland Tissues of Freshwater Mussel, Lamellidens marginalis Exposed to Oil Effluent. Austin J. Environ. Toxicol. 2021, 7, 1033. [Google Scholar]






| Exposure Times | LC50 Values | 95% Confidence Interval |
|---|---|---|
| 24-h | 18.71 mg/L | 13.20–25.75 |
| 48-h | 8.66 mg/L | 5.02–13.81 |
| 72-h | 5.87 mg/L | 3.28–9.88 |
| 96-h | 2.64 mg/L | 1.45–4.56 |
| Experiment Groups | Control | Control (DMSO) | 0.264 mg/L | 0.528 mg/L | ||||
|---|---|---|---|---|---|---|---|---|
| Fipronil | Fipronil | |||||||
| Tissues/Duration | 48-h | 7-d | 48-h | 7-d | 48-h | 7-d | 48-h | 7-d |
| Digestive Gland | ||||||||
| Degenerations of the tubules | - | - | - | - | + | ++ | +++ | +++ |
| Tubule necrosis | - | - | - | - | + | + | ++ | +++ |
| Gill | ||||||||
| Deformations of the lamella | - | - | - | - | + | + | + | ++ |
| Haemocytic infiltration | - | - | - | - | - | + | + | + |
| Epithelial hyperplasia of lamella | - | - | - | - | - | + | ++ | ++ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arslan, P.; Günal, A.Ç. Does Fipronil Affect on Aquatic Organisms? Physiological, Biochemical, and Histopathological Alterations of Non-Target Freshwater Mussel Species. Water 2023, 15, 334. https://doi.org/10.3390/w15020334
Arslan P, Günal AÇ. Does Fipronil Affect on Aquatic Organisms? Physiological, Biochemical, and Histopathological Alterations of Non-Target Freshwater Mussel Species. Water. 2023; 15(2):334. https://doi.org/10.3390/w15020334
Chicago/Turabian StyleArslan, Pınar, and Aysel Çağlan Günal. 2023. "Does Fipronil Affect on Aquatic Organisms? Physiological, Biochemical, and Histopathological Alterations of Non-Target Freshwater Mussel Species" Water 15, no. 2: 334. https://doi.org/10.3390/w15020334






