Species Richness of Benthic Macrofauna on Rocky Outcrops in the Adriatic Sea by Using Species-Area Relationship (SAR) Tools
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Fieldwork and Photo-Analysis
2.3. Data Analysis
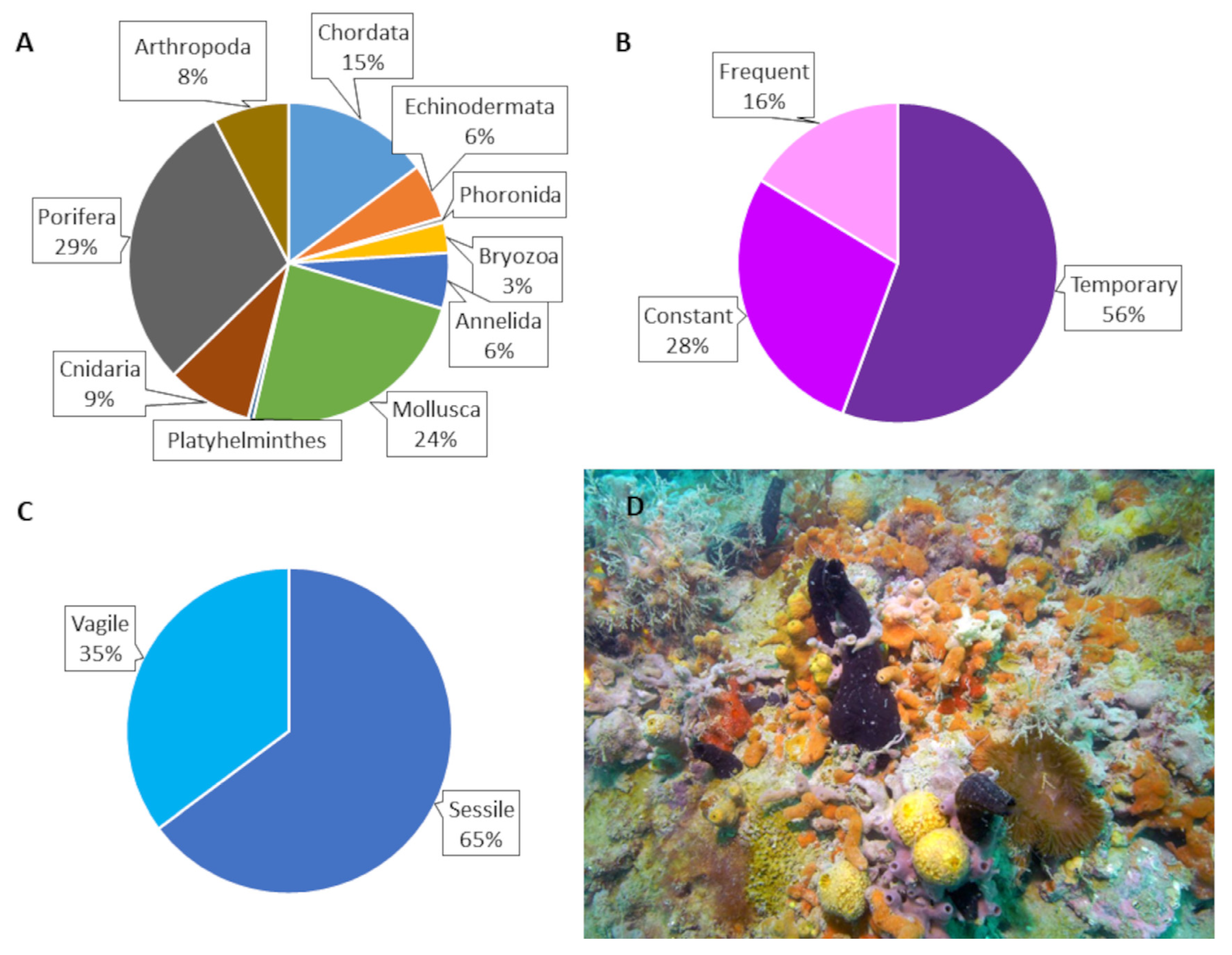
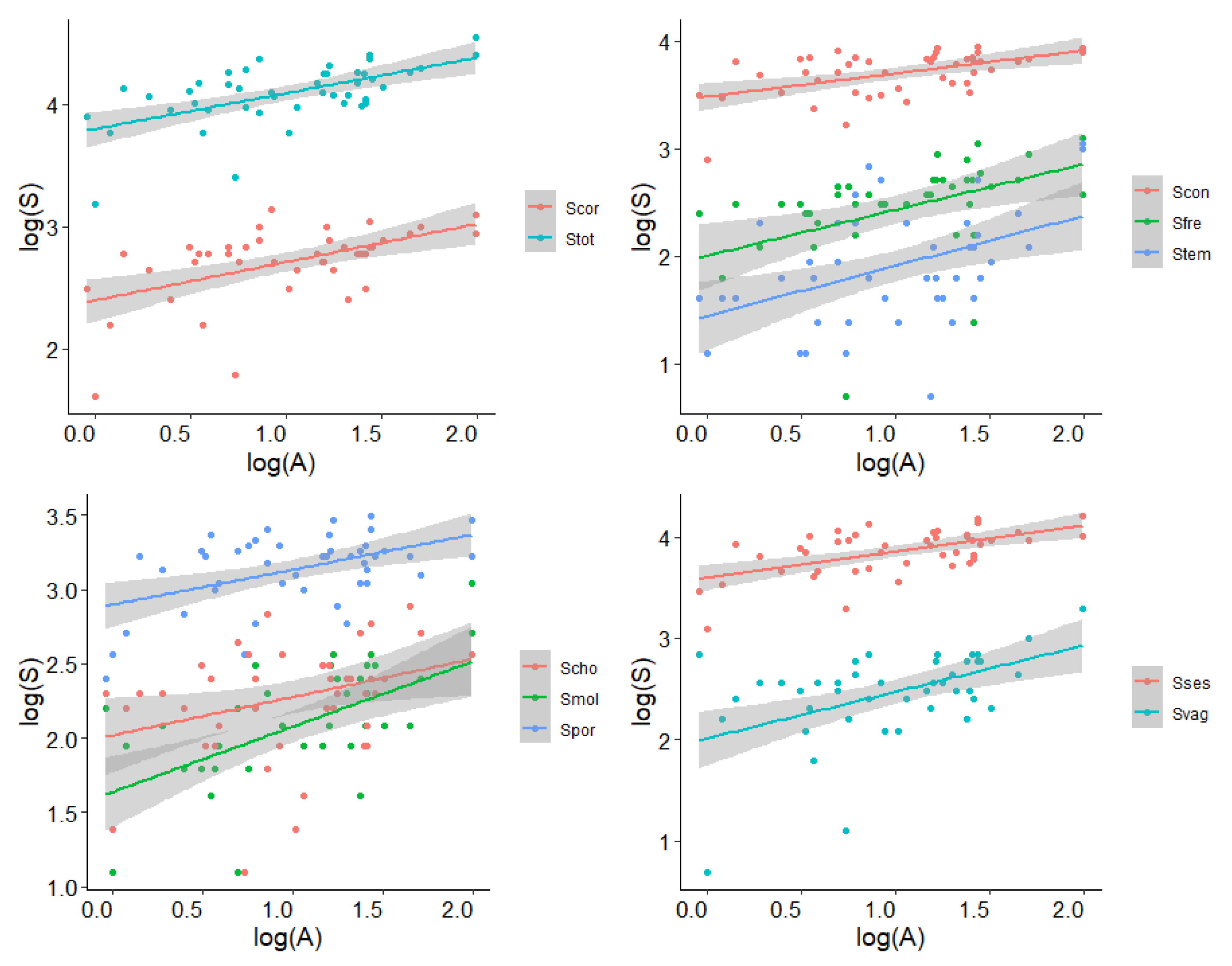
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arrhenius, O. Species and area. J. Ecol. 1921, 9, 95–99. [Google Scholar] [CrossRef] [Green Version]
- Rosenzweig, M. Species Diversity in Space and Time; Cambridge University Press: New York, NY, USA, 1995; 460p. [Google Scholar]
- Fattorini, S.; Lin, G.; Mantoni, C. Avian species–area relationships indicate that towns are not different from natural areas. Environ. Conserv. 2018, 45, 419–424. [Google Scholar] [CrossRef] [Green Version]
- Dengler, J.; Matthews, T.J.; Steinbauer, M.J.; Wolfrum, S.; Boch, S.; Chiarucci, A.; Conradi, T.; Dembicz, I.; Marcenò, C.; García-Mijangos, I.; et al. Species-area relationships in continuous vegetation: Evidence from Palaearctic Grasslands. J. Biogeogr. 2020, 47, 72–86. [Google Scholar] [CrossRef] [Green Version]
- Tittensor, D.P.; Micheli, F.; Nyström, M.; Worm, B. Human impacts on the specie-area relationship in reef fish assemblages. Ecol. Lett. 2007, 10, 760–772. [Google Scholar] [CrossRef]
- Pitacco, V.; Mistri, M.; Lipej, L. Species-Area Relationship (SAR) models as tools for estimating faunal biodiversity associated with habitat builder species in sensitive areas: The case of the Mediterranean stony coral (Cladocora caespitosa). Mar. Environ. Res. 2019, 149, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Connor, E.F.; McCoy, E.D. Species-area relationships. In Encyclopedia of Biodiversity; Levin, S., Ed.; Academic Press: Cambridge, MA, USA, 2001; Volume 5, pp. 397–411. [Google Scholar]
- Ulrich, W. Predicting species numbers using species-area and endemics-area relations. Biodivers. Conserv. 2005, 14, 3351–3362. [Google Scholar] [CrossRef]
- Veech, J. Choice of species-area function affects identification of hotspots. Conserv. Biol. 2001, 14, 140–147. [Google Scholar] [CrossRef]
- Gotelli, N.J.; Colwell, R.K. Quantifying biodiversity: Procedures and pitfalls in the measurement and comparison of species richness. Ecol. Lett. 2001, 4, 379–391. [Google Scholar] [CrossRef] [Green Version]
- MacArthur, R.H.; Wilson, E.O. An equilibrium theory of insular zoogeography. Evolution 1963, 17, 373–387. [Google Scholar] [CrossRef]
- Holt, R.D.; Lawton, J.H.; Polis, G.A.; Martinez, N.D. Trophic rank and species-area relationship. Ecology 1999, 80, 1495–1504. [Google Scholar]
- MacArthur, R.H. Geographical Ecology: Patterns in the Distribution of Species; Princeton University Press: Oxford, UK, 1972; p. 288. [Google Scholar]
- Dawson, M.N. Island and island-like marine environments. Global Ecol. Biogeogr. 2016, 25, 831–846. [Google Scholar] [CrossRef]
- Itescu, Y. Are island-like systems biologically similar to islands? A review of the evidence. Ecography 2019, 42, 1298–1314. [Google Scholar] [CrossRef] [Green Version]
- Gleason, H. On the relation between species and area. Ecology 1922, 3, 158–162. [Google Scholar] [CrossRef]
- Preston, F.W. Time and space and the variation of species. Ecology 1960, 41, 612–627. [Google Scholar] [CrossRef]
- Neigel, J.E. Species-area relationships and marine conservation. Ecol. Appl. 2003, 13, S138–S145. [Google Scholar] [CrossRef] [Green Version]
- McGuinness, K.A. Species-area relations of communities on intertidal boulders: Testing the null hypothesis. J. Biogeogr. 1984, 11, 439–456. [Google Scholar] [CrossRef]
- Chittaro, P.M. Species-area relationships for coral reef fish assemblages of St. Croix, US Virgin Islands. Mar. Ecol. Prog. Ser. 2002, 233, 253–261. [Google Scholar] [CrossRef]
- Balasubramanian, H.; Foster, R.S. Species and space: Role of volume in organizing coral reef fish assemblages in SE Cuba. Mar. Ecol. Prog. Ser. 2007, 345, 229–236. [Google Scholar] [CrossRef] [Green Version]
- Reichert, K.; Ugland, K.I.; Bartsch, I.; Hortal, J.; Bremner, J.; Kraberg, A. Species richness estimation: Estimator performance and the influence of rare species. Limnol. Oceanogr. Methods 2010, 8, 294–303. [Google Scholar] [CrossRef] [Green Version]
- Sabetta, L.; Barbone, E.; Giardino, A.; Galuppo, N.; Basset, A. Species-area patterns of benthic macro-invertebrates in Italian lagoons. Hydrobiologia 2007, 577, 127–139. [Google Scholar] [CrossRef]
- Bevilacqua, S.; Ugland, K.I.; Plicanti, A.; Scuderi, D.; Terlizzi, A. An approach based on the total-species accumulation curve and higher taxon richness to estimate realistic upper limits in regional species richness. Ecol. Evol. 2018, 8, 405–415. [Google Scholar] [CrossRef]
- Casellato, S.; Masiero, L.; Sichirollo, E.; Soresi, S. Hidden secrets of the northern Adriatic: “Tegnúe”, peculiar reefs. Cent. Eur. J. Biol. 2007, 2, 122–136. [Google Scholar] [CrossRef]
- Casellato, S.; Stefanon, A. Coralligenous habitat in the northern Adriatic Sea: An overview. Mar. Ecol. 2008, 29, 321–341. [Google Scholar] [CrossRef]
- Ingrosso, G.; Abbiati, M.; Badalamenti, F.; Bavestrello, G.; Belmonte, G.; Cannas, R.; Benedetti-Cecchi, L.; Bertolino, M.; Bevilacqua, S.; Bianchi, C.N.; et al. Chapter Three—Mediterranean bioconstructions along the Italian coast. In Advances in Marine Biology; Sheppard, C., Ed.; Academic Press: Cambridge, MA, USA, 2018; Volume 79, pp. 61–136. [Google Scholar] [CrossRef]
- Lipej, L.; Orlando-Bonaca, M.; Mavrič, B. Biogenic Formations in the Slovenian Sea; National Institute of Biology, Marine Biology Station Piran, UNEP, RAC/SPA: Tunis, Tunisia, 2016; 206p. [Google Scholar]
- Stefanon, A. Formazioni rocciose del bacino dell’Alto Adriatico. Atti IVSLA 1967, 85, 79–85. [Google Scholar]
- Braga, G.; Stefanon, A. Beachrock e Alto Adriatico: Aspetti paleogeografici, climatici, morfologici ed ecologici del problema. Atti IVSLA 1969, 77, 351–361. [Google Scholar]
- Stefanon, A. The role of beachrock in the study of the evolution of the North Adriatic Sea. Mem. Biogeogr. Adriat. 1970, 8, 79–99. [Google Scholar]
- Newton, R.; Stefanon, A. The ‘Tegnúe de Ciosa’ area: Patch reefs in the northern Adriatic Sea. Mar. Geol. 1975, 8, 27–33. [Google Scholar] [CrossRef]
- Colantoni, P.; Gabbianelli, G.; Ceffa, L.; Ceccolini, C. Bottom features and gas seepages in the Adriatic Sea. In Proceedings of the Vth International Conference on Gas in Marine Sediments, Bologna, Italy, September 1998; pp. 28–31. [Google Scholar]
- Stefanon, A.; Zuppi, G.M. Recent carbonate rock formation in the northern Adriatic Sea. Hydrogeologie 2000, 4, 3–10. [Google Scholar]
- Conti, A.; Stefanon, A.; Zuppi, G.M. Gas seeps and rock formation in the northern Adriatic Sea. Continent. Shelf Res. 2002, 22, 2333–2344. [Google Scholar] [CrossRef]
- Gordini, E.; Marocco, R.; Tunis, G.; Ramella, R. The cemented deposits of the Trieste Gulf (northern Adriatic Sea): Areal distribution, geomorphologic characteristics and high resolution seismic survey. J. Quaternary Sci. 2004, 17, 555–563. [Google Scholar]
- Gordini, E.; Falace, A.; Kaleb, S.; Donda, F.; Marocco, R.; Tunis, G. Methane-related carbonate cementation of marine sediments and related macroalgal coralligenous assemblages in the northern Adriatic Sea. In Seafloor Geomorphology as Benthic Habitat; Harris, P.T., Baker, E.K., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2012; pp. 185–200. [Google Scholar]
- Turicchia, E.; Abbiati, M.; Bettuzzi, M.; Calcinai, B.; Morigi, M.P.; Summers, A.P.; Ponti, M. Bioconstruction and bioerosion in the northern Adriatic coralligenous reefs quantified by X-ray computed tomography. Front. Mar. Sci. 2022, 8, 790869. [Google Scholar] [CrossRef]
- Ponti, M.; Fava, F.; Abbiati, M. Spatial-temporal variability of epibenthic assemblages on subtidal biogenic reefs in the northern Adriatic Sea. Mar. Biol. 2011, 158, 1447–1459. [Google Scholar] [CrossRef]
- Curiel, D.; Falace, A.; Bandelj, V.; Kaleb, S.; Solidoro, C.; Ballesteros, E. Species composition and spatial variability of macroalgal assemblages on biogenic reefs in the northern Adriatic Sea. Bot. Mar. 2012, 55, 625–638. [Google Scholar] [CrossRef] [Green Version]
- Falace, A.; Kaleb, S.; Curiel, D.; Miotti, C.; Galli, G.; Querin, S.; Ballesteros, E.; Solidoro, C.; Bandelj, V. Calcareous bio-concretions in the northern Adriatic Sea: Habitat types, environmental factors that influence habitat distributions, and predictive modeling. PLoS ONE 2015, 10, e0140931. [Google Scholar] [CrossRef]
- Caressa, S.; Gordini, E.; Marocco, R.; Tunis, G. Caratteri geomorfologici degli affioramenti rocciosi del Golfo di Trieste (Adriatico Settentrionale). Gortania Atti Mus. Friul. St. Nat. 2001, 23, 5–29. [Google Scholar]
- Boicourt, W.C.; Kuzmić, M.; Hopkins, T.S. The Inland Sea: Circulation of Chesapeake Bay and the Northern Adriatic. In Ecosystems at the Land-Sea Margin: Drainage Basin to Coastal Sea; Malone, T.C., Malej, A., Harding, L.W., Jr., Smodlaka, N., Turner, R.E., Eds.; Am. Geophysical Union: Washington, DC, USA, 1999; pp. 81–129. [Google Scholar]
- Stravisi, F. The vertical structure annual cycle of the mass field parameters in the Gulf of Trieste. Boll. Oceanol. Teor. Applic. 1983, 1, 239–250. [Google Scholar]
- Stachowitsch, M.; Fuchs, A. Long-term changes in the benthos of the northern Adriatic Sea. Annales Ser. Hist. Nat. 1995, 7, 7–16. [Google Scholar]
- Solis-Weiss, V.; Rossin, P.; Aleffi, F.; Bettoso, N.; Fonda Umani, S. A regional GIS for benthic diversity and environmental impact studies in the Gulf of Trieste, Italy. In Proceedings of the “The Colour of Ocean Data” Symposium, Brussels, Belgium, 25–27 November 2002; Vanden Berghe, E., Brown, M., Costello, M.J., Heip, C., Levitus, S., Pissierssens, P., Eds.; IOC Workshop Report 188. UNESCO: Paris, France, 2004; pp. 245–255. [Google Scholar]
- Mavrič, B.; Orlando-Bonaca, M.; Bettoso, N.; Lipej, L. Soft-bottom macrozoobenthos of the southern part of the Gulf of Trieste: Faunistic, biocoenotic and ecological survey. Acta Adriat. 2010, 51, 203–216. [Google Scholar]
- Pérès, J.M.; Picard, J. Noveau manuel de bionomie bentique de la Mer Méditerranée (New manual on benthic bionomy of the Mediterranean Sea). Réc. Trav. Stat. Mar. Endoume 1964, 31, 5–137. [Google Scholar]
- Solis-Weiss, V.; Rossin, P.; Aleffi, F.; Bettoso, N.; Orel, G.; Vrišer, B. Gulf of Trieste sensitivity areas using Benthos and GIS techniques. In Proceedings of the 5th International Conference on the Mediterranean coastal Environment Medcoast 2001, Hammamet, Tunisia, 23–27 October 2001; Volume 3, pp. 1567–1578. [Google Scholar]
- Gordini, E.; Caressa, S.; Marocco, R. New morpho-sedimentological map of Trieste Gulf (from Punta Tagliamento to Isonzo Mouth). Gortania Atti Mus. Friul. St. Nat. 2003, 25, 5–29. (In Italian) [Google Scholar]
- Orel, G.; Marocco, R.; Vio, E.; Del Piero, D.; Della Seta, G. Sediments and benthic biocoenoses between Po river delta and the Gulf of Trieste (northern Adriatic Sea). Bull. Ecol. 1987, 18, 229–241. (In Italian) [Google Scholar]
- Bandelj, V.; Solidoro, C.; Laurent, C.; Querin, S.; Kaleb, S.; Gianni, F.; Falace, A. Cross-scale connectivity of macrobenthic communities in a patchy network of habitats: The mesophotic biogenic habitats of the northern Adriatic Sea. Estuar. Coast. Shelf Sci. 2020, 245, 1–14. [Google Scholar] [CrossRef]
- Bevilacqua, S.; Katsanevakis, S.; Micheli, F.; Sala, E.; Rilov, G.; Sarà, G.; Malak, D.A.; Abdulla, A.; Gerovasileiou, V.; Gissi, E.; et al. The status of coastal benthic ecosystems in the Mediterranean Sea: Evidence from ecological indicators. Front. Mar. Sci. 2020, 7, 475. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Pronzato, R.; Cattaneo-Vietti, R.; Benedetti Cecchi, L.; Morri, C.; Pansini, M.; Chemello, R.; Milazzo, M.; Fraschetti, S.; Terlizzi, A.; et al. I fondi duri (hard bottoms). In: M. C. Gambi & M. Dappiano (Editors). Manuale di metodologie di campionamento e studio del benthos marino Mediterraneo. Biol. Mar. Medit. 2003, 10, 199–232. [Google Scholar]
- Bachelet, G.; de Montaudouin, X.; Dauvin, J.C. The quantitative distribution of subtidal macrozoobenthic assemblages in Arcachon Bay in relation to environmental factors: Multivariate analysis. Estuar. Coast. Shelf Sci. 1996, 42, 371–391. [Google Scholar] [CrossRef]
- Félix, P.M.; Chainho, P.; Costa, J.L.; Correia, M.J.; Chaves, M.L.; Medeiros, J.P.; Cabral, H.N.; Wouters, N.; Bernardo, J.; Costa, A.M.; et al. Short-term versus long-term changes in the benthic communities of a small coastal lagoon: Implications for ecological status assessment. Vie Milieu 2013, 63, 11–22. [Google Scholar]
- Ballesteros, E. Mediterranean coralligenous assemblages: A synthesis of present knowledge. Oceanogr. Mar. Biol. Ann. Rev. 2006, 44, 123–195. [Google Scholar]
- UNEP/MAP-SPA/RAC SAP/RAC: SPA-BD Protocol—Annex II: List of Endangered or Threatened Species. 2018. Available online: https://www.rac-spa.org/sites/default/files/annex/annex_2_en_20182.pdf (accessed on 9 January 2023).
- Spearman, C. Demonstration of formulae for true measurement of correlation. Am. J. Psychol. 1907, 18, 161–169. [Google Scholar] [CrossRef]
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. The Vegan Package. Community Ecology Package. 2008. Available online: http://r-forge.r-project.org/projects/vegan/ (accessed on 9 January 2023).
- RDevelopmentCoreTeam. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008; Available online: https://www.R-project.org/ (accessed on 9 January 2023).
- Nesto, N.; Simonini, R.; Riccato, F.; Fiorin, R.; Picone, M.; Da Ros, L.; Moschino, V. Macro-zoobenthic biodiversity of northern Adriatic hard substrates: Ecological insights from a bibliographic survey. J. Sea Res. 2020, 160–161, 101903. [Google Scholar] [CrossRef]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Lasram, F.B.R.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.; Corbera, J.; Dailianis, T.; et al. Voultsiadou The biodiversity of the Mediterranean Sea: Estimates, patterns and threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [Green Version]
- Pérès, J.M.; Picard, J. Recherches sur les peuplements benthiques de la Méditerranée nord-orientale. Ann. Inst. Océanogr. Monaco 1958, 34, 213–291. [Google Scholar]
- Garrabou, J.; Kipson, S.; Kaleb, S.; Kružić, P.; Jaklin, A.; Žuljević, A.; Rajković, Z.; Rodić, P.; Jelić, K.; Župan, D. Monitoring Protocol for Reefs—Coralligenous Community. In RAC/SPA—UNEP/MAP; RAC/SPA, Ed.; MedMPAnet Project: Tunis, Tunisia, 2014; p. 35. Available online: https://www.rac-spa.org/sites/default/files/doc_medmpanet/final_docs_croatia/18_testing_monitoring_protocol_coralligenous_2_croatia.pdf (accessed on 9 January 2023).
- Manfrin, C.; Ciriaco, S.; Segarich, M.; Fioravanti, M.; Tedesco, P.; Florio, D.; Carella, F.; Giulianini, P.G.; Terlizzi, A.; Pallavicini, A.; et al. First detection of Haplosporidium pinnae inhabiting the Gulf of Trieste. Biol. Mar. Medit. 2022; in press. [Google Scholar]
- Kersting, D.K.; Vázquez-Luis, M.; Mourre, B.; Belkhamssa, F.Z.; Álvarez, E.; Bakran-Petricioli, T.; Barberá, C.; Barrajón, A.; Cortés, E.; Deudero, S.; et al. Recruitment Disruption and the Role of Unaffected Populations for Potential Recovery After the Pinna nobilis Mass Mortality Event. Front. Mar. Sci. 2020, 7, 594378. [Google Scholar] [CrossRef]
- Zotou, M.; Gkrantounis, P.; Karadimou, E.; Tsirintanis, K.; Sini, M.; Poursanidis, D.; Azzolin, M.; Dailianis, T.; Kytinou, E.; Issaris, Y. Pinna nobilis in the Greek seas (NE Mediterranean): On the brink of extinction? Mediterr. Mar. Sci. 2020, 21, 575–591. [Google Scholar] [CrossRef]
- MacArthur, R.; Wilson, E. The Theory of Island Biogeography; Princeton University Press: Princeton, NJ, USA, 1967; 203p. [Google Scholar]
- Whittaker, R.J.; Triantis, K.A.; Ladle, R.J. A general dynamic theory of oceanic island biogeography. J. Biogeogr. 2008, 35, 977–994. [Google Scholar] [CrossRef]
- Hortal, J.; Triantis, K.A.; Meiri, S.; Thébault, E.; Sfenthourakis, S. Island species richness increases with habitat diversity. Am. Nat. 2009, 174, E205–E217. [Google Scholar] [CrossRef] [Green Version]
- Lomolino, M.V. The unifying, fundamental principles of biogeography: Understanding Island Life. Front. Biogeogr. 2016, 8, e29920. [Google Scholar] [CrossRef]
- Schoener, A. Colonization curves for planar marine islands. Ecology 1974, 55, 818–827. [Google Scholar] [CrossRef]
- Smith, G.B. Relationship of eastern Gulf of Mexico reef-fish communities to the species equilibrium theory of insular biogeography. J. Biogeogr. 1979, 6, 49–61. [Google Scholar] [CrossRef]
- Schoener, A.; Long, E.R.; DePalma, J. Geographic variation in artificial island colonization curves. Ecology 1978, 59, 367–382. [Google Scholar] [CrossRef]
- Abele, L.G. Comparative species richness in fluctuating and constant environments: Coral-associated decapod crustaceans. Science 1976, 192, 461–463. [Google Scholar] [CrossRef]
- Abele, L.G.; Patton, W.K. The size of coral heads and the community biology of associated decapod crustaceans. J. Biogeogr. 1976, 3, 35–47. [Google Scholar] [CrossRef]
- Stortini, C.; Petrie, B.; Frank, K.; Leggett, W. Marine macroinvertebrate species-area relationships, assemblage structure and their environmental drivers on submarine banks. Mar. Ecol. Prog. Ser. 2020, 641, 25–47. [Google Scholar] [CrossRef]
- Hachich, N.F.; Bonsall, M.B.; Arraut, E.M.; Barneche, D.R.; Lewinsohn, T.M.; Floeter, S.R. Island biogeography: Patterns of marine shallow-water organisms in the Atlantic Ocean. J. Biogeogr. 2015, 42, 1871–1882. [Google Scholar] [CrossRef]
- Shepherd, U.; Brantley, S. Expanding on Watson’s framework for classifying patches: When is an island not an island? J. Biogeogr. 2005, 32, 951–960. [Google Scholar] [CrossRef]
- Giangrande, A.; Gambi, M.C.; Gravina, M.F. Paradigm shifts in community ecology: Open versus closed units, challenges and limits of connectivity studies. Mar. Ecol. 2017, 38, e12480. [Google Scholar] [CrossRef]
- Cardone, F.; Corriero, G.; Longo, C.; Pierri, C.; Gimenez, G.; Gravina, M.F.; Giangrande, A.; Lisco, S.; Moretti, M.; De Giosa, F.; et al. A system of marine animal bioconstructions in the mesophotic zone along the southern Italian coast. Front. Mar. Sci. 2022, 9, 948836. [Google Scholar] [CrossRef]
- Drakare, S.; Lennon, J.J.; Hillebrand, H. The imprint of the geographical, evolutionary and ecological context on species–area relationships. Ecol. Lett. 2006, 9, 215–227. [Google Scholar] [CrossRef]
- Belmaker, J. Species richness of resident and transient coral-dwelling fish responds differentially to regional diversity. Global Ecol. Biogeogr. 2009, 18, 426–436. [Google Scholar] [CrossRef]
- Triantis, K.A.; Guilhaumon, F.; Whittaker, R.J. The island species–area relationship: Biology and statistics. J. Biogeogr. 2012, 39, 215–231. [Google Scholar] [CrossRef]
- Cardone, F.; Corriero, G.; Longo, C.; Mercurio, M.; Tarantini, S.O.; Gravina, M.F.; Lisco, S.; Moretti, M.; De Giosa, F.; Giangrande, A.; et al. Massive bioconstructions built by Neopycnodonte cochlear (Mollusca, Bivalvia) in a mesophotic environment in the central Mediterranean Sea. Sci. Rep. 2020, 10, 6337. [Google Scholar] [CrossRef] [Green Version]
- Orel, G.; Mennea, B. I popolamenti bentonici di alcuni tipi di fondo mobile del Golfo di Trieste. Pubbl. Della Stn. Zool. Napoli 1969, 37, 261–276. [Google Scholar]
- Gamulin-Brida, H. Biocenoses benthiques de la Mer Adriatique. Acta Adriat. 1974, 15, 1–102. [Google Scholar]
- Fedra, K.; Ölscher, E.M.; Scherübel, C.; Stachowitsch, M.; Wurzian, R.S. On the ecology of a North Adriatic benthic community: Distribution, standing crop and composition of the macrobenthos. Mar. Biol. 1976, 38, 129–145. [Google Scholar] [CrossRef]
- Molin, E.; Riccato, F.; Fiorin, R.; Artico, G.; Campaci, P. Hard substrate macrozoobenthos Communities of three rocky outcrops in the Gulf of Venice (North Adriatic). Boll. Mus. St. Nat. Venezia 2011, 62, 5–18. [Google Scholar]
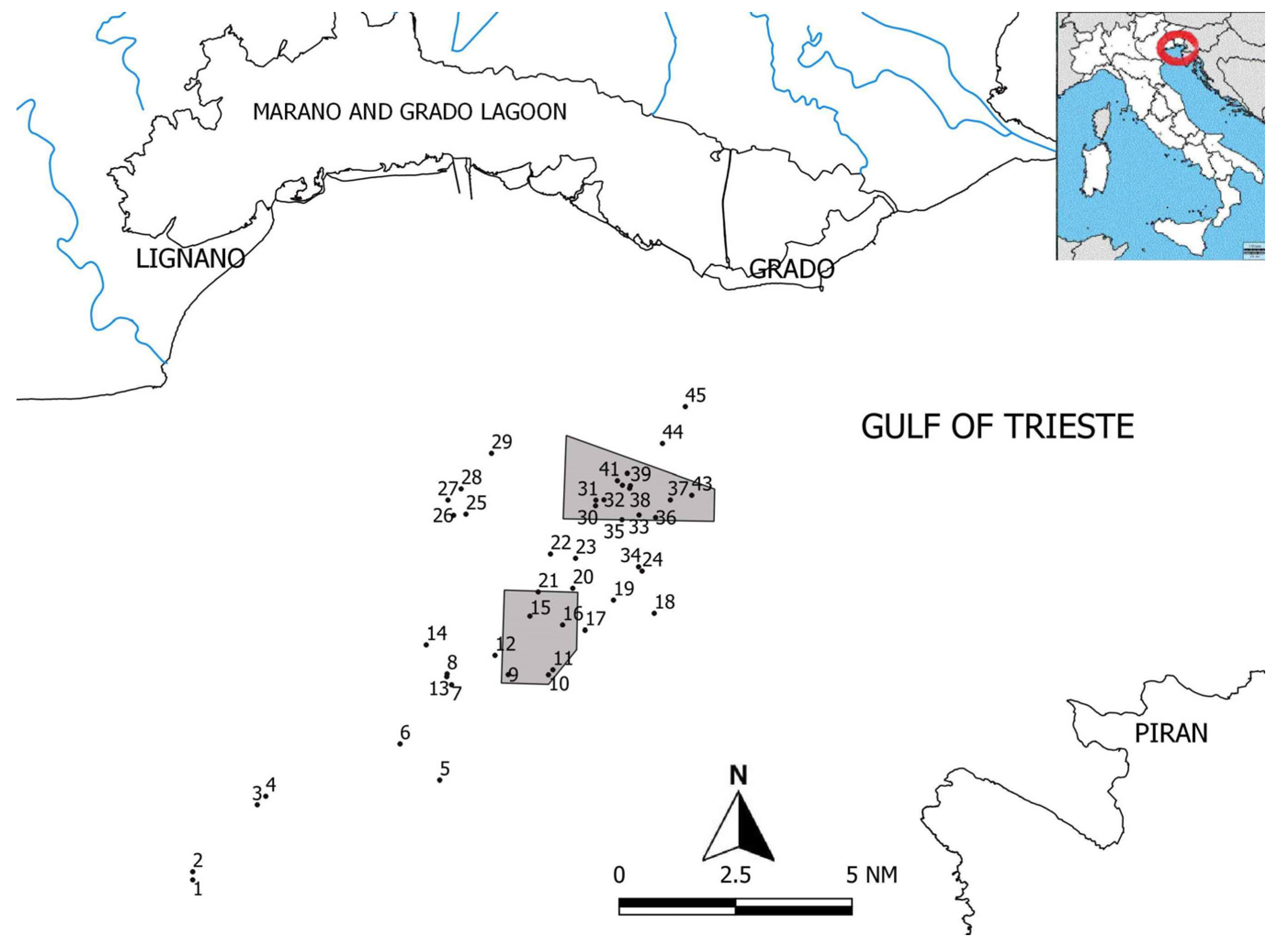
| Outcrop Name | Sampling Date | Coast Distance (nm) | Depth (m) | n. Point Intercepts | n. Taxa | |
|---|---|---|---|---|---|---|
| 1 | COLOMBA * | 17 September 2014 | 10 | 24 | 55 | 53 |
| 2 | COLOMBA 2 * | 24 September 2014 | 10 | 24 | 72 | 53 |
| 3 | STRUCOLO * | 24 September 2014 | 9.5 | 25 | 59 | 51 |
| 4 | GUBANA * | 17 September 2014 | 9.5 | 24 | 63 | 60 |
| 5 | BARDELLI * | 18 September 2014 | 10 | 23 | 24 | 49 |
| 6 | NICOLA * | 9 July 2013 | 9 | 23 | 101 | 54 |
| 7 | DAVE * | 5 July 2013 | 9 | 21 | 130 | 71 |
| 8 | LA LONGA | 4 July 2014 | 8.6 | 20.5 | 33 | 58 |
| 9 | AGENORE * SCI | 29 August 2014 | 8.8 | 21 | 41 | 61 |
| 10 | PESCECANE * SCI | 31 October 2014 | 9.5 | 21 | 59 | 79 |
| 11 | SCARPENE * SCI | 19 January 2015 | 8.6 | 19 | 50 | 64 |
| 12 | ALDEBARAN * | 5 July 2013 | 9.1 | 19 | 37 | 52 |
| 13 | LA BOMBA * | 4 July 2014 | 8.3 | 20 | 80 | 65 |
| 14 | SARATOGA * | 1 August 2013 | 8.3 | 18 | 138 | 73 |
| 15 | DORSALE * SCI | 6 August 2013 | 9.4 | 18 | 107 | 67 |
| 16 | SUDPIASTRA * SCI | 29 August 2014 | 7.7 | 21 | 43 | 65 |
| 17 | SPARI * | 1 August 2013 | 7.9 | 20 | 103 | 57 |
| 18 | LE GATTE | 29 August 2014 | 7.1 | 20 | 99 | 65 |
| 19 | ALBERT | 31 October 2014 | 7 | 19 | 64 | 58 |
| 20 | MISTO * | 5 July 2013 | 7.2 | 18 | 103 | 55 |
| 21 | BIRO * SCI | 29 August 2014 | 7.6 | 18 | 53 | 62 |
| 22 | PESCHERIA * | 29 August 2014 | 6.6 | 17 | 50 | 71 |
| 23 | S. PAOLO * | 19 January 2015 | 6.6 | 17 | 94 | 59 |
| 24 | MORO * | 22 December 2014 | 6.2 | 21 | 69 | 43 |
| 25 | SALIENT * | 4 July 2014 | 6.5 | 13 | 99 | 71 |
| 26 | SARGASSI | 27 May 2014 | 6.5 | 15 | 92 | 55 |
| 27 | PINNACOLI * | 9 July 2013 | 6.5 | 14 | 113 | 63 |
| 28 | NORDALTI * | 6 August 2013 | 6 | 13 | 87 | 59 |
| 29 | MUSSOLI | 27 March 2013 | 4.5 | 13 | 27 | 43 |
| 30 | W FUMAPIL SCI | 10 April 2015 | 9.9 | 17 | 105 | 82 |
| 31 | FUMAPIL SCI | 22 December 2014 | 5.2 | 16.5 | 42 | 55 |
| 32 | TIMOTEO * SCI | 26 January 2015 | 5.3 | 16 | 55 | 72 |
| 33 | SEPA SCI | 21 May 2014 | 5.2 | 18 | 184 | 82 |
| 34 | CP * | 26 January 2015 | 5.3 | 17 | 82 | 60 |
| 35 | AREA 4 SCI | 3 April 2014 | 5 | 17.5 | 45 | 52 |
| 36 | PALO LARGO SCI | 3 July 2014 | 4.8 | 19 | 85 | 75 |
| 37 | PALI * SCI | 11 July 2013 | 4.5 | 18 | 52 | 30 |
| 38 | S. PIETRO SCI | 18 September 2014 | 4.6 | 16 | 29 | 62 |
| 39 | S. PIEROTERRA SCI | 10 August 2014 | 4.6 | 17 | 83 | 70 |
| 40 | CORVINE * SCI | 6 August 2013 | 4.6 | 15 | 84 | 70 |
| 41 | SPERLIG SCI | 10 April 2015 | 8.6 | 16.5 | 105 | 79 |
| 42 | AMERIGO * SCI | 10 May 2014 | 4.4 | 17 | 184 | 94 |
| 43 | MALUSA SCI | 26 August 2014 | 3.5 | 16.5 | 102 | 70 |
| 44 | MENEGHEL * | 11 July 2013 | 3.4 | 14 | 25 | 24 |
| 45 | AREA 3 | 1 April 2014 | 3 | 14 | 44 | 43 |
| Taxonomic list | ||
| Porifera | Phymanthus pulcher (Andres, 1883) | Protula tubularia (Montagu, 1803) |
| Agelas oroides (Schmidt, 1864) | Calliactis parasitica (Couch, 1842) | Bispira volutacornis (Montagu, 1804) |
| Axinella polypoides Schmidt, 1862 | Sagartia elegans (Dalyell, 1848) | Myxicola infundibulum (Montagu, 1808) |
| Axinella cannabina (Esper, 1794) | Anemonia viridis (Forsskål, 1775) | Arthropoda |
| Axinella damicornis (Esper, 1794) | Cornularia cornucopiae (Pallas, 1766) | Paguristes eremita (Linnaeus, 1767) |
| Raspailia (Raspailia) viminalis Schmidt, 1862 | Maasella edwardsii (de Lacaze-Duthiers, 1888) | Dromia personata (Linnaeus, 1758) |
| Dictyonella incisa (Schmidt, 1880) | Cladocora caespitosa (Linnaeus, 1767) | Galathea strigosa (Linnaeus, 1761) |
| Bubaris vermiculata (Bowerbank, 1866) | Caryophyllia (Caryophyllia) inornata (Duncan, 1878) | Galathea intermedia Lilljeborg, 1851 |
| Acanthella acuta Schmidt, 1862 | Leptopsammia pruvoti Lacaze-Duthiers, 1897 | Lysmata seticaudata (Risso, 1816) |
| Chondrilla nucula Schmidt, 1862 | Cerianthus membranaceus (Gmelin, 1791) | Maja crispata Risso, 1827 in [Risso, 1826–1827] |
| Chondrosia reniformis Nardo, 1847 | Epizoanthus arenaceus (Delle Chiaje, 1836) | Homarus gammarus (Linnaeus, 1758) |
| Cliona viridis (Schmidt, 1862) | Parazoanthus axinellae (Schmidt, 1862) | Pagurus anachoretus Risso, 1827 in [Risso, 1826–1827] |
| Cliona celata Grant, 1826 | Mollusca | Pagurus prideaux Leach, 1815 [in Leach, 1815–1875] |
| Cliona rhodensis Rützler & Bromley, 1981 | Philinopsis depicta (Renier, 1807) | Periclimenes amethysteus (Risso, 1827 in [Risso, 1826–1827]) |
| Aplysilla sulfurea Schulze, 1878 | Haliotis tuberculata Linnaeus, 1758 | Pilumnus hirtellus (Linnaeus, 1761) |
| Dysidea avara (Schmidt, 1862) | Thylacodes arenarius (Linnaeus, 1758) | Scyllarus arctus (Linnaeus, 1758) |
| Ircinia variabilis (Schmidt, 1862) | Rapana venosa (Valenciennes, 1846) | Bryozoa |
| Sarcotragus spinosulus Schmidt, 1862 | Muricopsis cristata (Brocchi, 1814) | Schizobrachiella sanguinea (Norman, 1868) |
| Dysidea fragilis (Montagu, 1814) | Hexaplex trunculus (Linnaeus, 1758) | Chartella papyracea (Ellis and Solander, 1786) |
| Sarcotragus foetidus Schmidt, 1862 | Bolinus brandaris (Linnaeus, 1758) | Reteporella beaniana (King, 1846) |
| Pleraplysilla spinifera (Schulze, 1879) | Felimare villafranca (Risso, 1818) | Phoronida |
| Scalarispongia scalaris (Schmidt, 1862) | Edmundsella pedata (Montagu, 1816) | Phoronis muelleri Selys-Lonchamps, 1903 |
| Spongia (Spongia) officinalis Linnaeus, 1759 | Felimida krohni (Vérany, 1846) | Echinodermata |
| Cacospongia mollior Schmidt, 1862 | Dendrodoris grandiflora (Rapp, 1827) | Marthasterias glacialis (Linnaeus, 1758) |
| Haliclona (Reniera) mediterranea Griessinger, 1971 | Paradoris indecora (Bergh, 1881) | Echinaster (Echinaster) sepositus (Retzius, 1783) |
| Petrosia (Petrosia) ficiformis (Poiret, 1789) | Felimare tricolor (Cantraine, 1835) | Sphaerechinus granularis (Lamarck, 1816) |
| Haliclona (Haliclona) simulans (Johnston, 1842) | Trapania maculata Haefelfinger, 1960 | Paracentrotus lividus (Lamarck, 1816) |
| Haliclona (Soestella) mamillata (Griessinger, 1971) | Dendrodoris limbata (Cuvier, 1804) | Echinus melo Lamarck, 1816 |
| Haliclona (Soestella) mucosa (Griessinger, 1971) | Flabellina affinis (Gmelin, 1791) | Holothuria (Holothuria) tubulosa Gmelin, 1791 |
| Haliclona (Halichoclona) fulva (Topsent, 1893) | Paraflabellina ischitana (Hirano and T. E. Thompson, 1990) | Ocnus planci (Brandt, 1835) |
| Oscarella lobularis (Schmidt, 1862) | Tethys fimbria Linnaeus, 1767 | Holothuria (Panningothuria) forskali Delle Chiaje, 1823 |
| Clathria (Clathria) compressa Schmidt, 1862 | Crimora papillata Alder and Hancock, 1862 | Ophioderma longicaudum (Bruzelius, 1805) |
| Tedania (Tedania) anhelans (Vio in Olivi, 1792) | Discodoris stellifera (Vayssière, 1903) | Ophiura ophiura (Linnaeus, 1758) |
| Mycale (Aegogropila) tunicata (Schmidt, 1862) | Doriopsilla areolata Bergh, 1880 | Chordata |
| Phorbas tenacior (Topsent, 1925) | Doris pseudoargus Rapp, 1827 | Aplidium tabarquensis Ramos-Espla, 1991 |
| Crella (Yvesia) rosea (Topsent, 1892) | Felimare fontandraui (Pruvot-Fol, 1951) | Aplidium conicum (Olivi, 1792) |
| Phorbas fictitius (Bowerbank, 1866) | Paraflabellina gabinierei (Vicente, 1975) | Polycitor adriaticus (Drasche, 1883) |
| Crambe crambe (Schmidt, 1862) | Polycera quadrilineata (O. F. Müller, 1776) | Didemnum commune (Della Valle, 1877) |
| Antho (Antho) inconstans (Topsent, 1925) | Berthella ocellata (Delle Chiaje, 1830) | Didemnum coriaceum (Drasche, 1883) |
| Myxilla (Myxilla) rosacea (Lieberkühn, 1859) | Calliostoma zizyphinum (Linnaeus, 1758) | Diplosoma listerianum (Milne Edwards, 1841) |
| Ulosa digitata (Schmidt, 1866) | Bolma rugosa (Linnaeus, 1767) | Aplidium elegans (Giard, 1872) |
| Mycale (Aegogropila) contarenii (Lieberkühn, 1859) | Tylodina perversa (Gmelin, 1791) | Polysyncraton lacazei (Giard, 1872) |
| Hemimycale columella (Bowerbank, 1874) | Elysia viridis (Montagu, 1804) | Cystodytes dellechiajei (Della Valle, 1877) |
| Mycale (Carmia) macilenta (Bowerbank, 1866) | Arca noae Linnaeus, 1758 | Didemnum lahillei Hartmeyer, 1909 |
| Polymastia mamillaris (Müller, 1806) | Glycymeris glycymeris (Linnaeus, 1758) | Polyclinella azemai Harant, 1930 |
| Suberites domuncula (Olivi, 1792) | Rocellaria dubia (Pennant, 1777) | Aplidium nordmanni (Milne Edwards, 1841) |
| Terpios fugax Duchassaing and Michelotti, 1864 | Limaria hians (Gmelin, 1791) | Aplidium turbinatum (Savigny, 1816) |
| Halichondria (Halichondria) semitubulosa (Lamarck, 1814) | Mytilus galloprovincialis Lamarck, 1819 | Clavelina sabbadini Brunetti, 1987 |
| Suberites massa Nardo, 1847 | Ostrea edulis Linnaeus, 1758 | Diazona violacea Savigny, 1816 |
| Tethya aurantium (Pallas, 1766) | Pinna nobilis Linnaeus, 1758 | Phallusia fumigata (Grube, 1864) |
| Tethya citrina Sarà and Melone, 1965 | Mimachlamys varia (Linnaeus, 1758) | Phallusia mammillata (Cuvier, 1815) |
| Geodia cydonium (Linnaeus, 1767) | Talochlamys multistriata (Poli, 1795) | Ascidiella aspersa (Müller, 1776) |
| Aplysina aerophoba (Nardo, 1833) | Pecten jacobaeus (Linnaeus, 1758) | Ascidia mentula Müller, 1776 |
| Aplysina cavernicola (Vacelet, 1959) | Venus verrucosa Linnaeus, 1758 | Distomus variolosus Gaertner, 1774 |
| Hexadella racovitzai Topsent, 1896 | Octopus vulgaris Cuvier, 1797 | Halocynthia papillosa (Linnaeus, 1767) |
| Cnidaria | Sepia officinalis Linnaeus, 1758 | Styela plicata (Lesueur, 1823) |
| Nausithoe punctata Kölliker, 1853 | Annelida | Botrylloides leachii (Savigny, 1816) |
| Cereus pedunculatus (Pennant, 1777) | Bonellia viridis Rolando, 1822 | Botryllus schlosseri (Pallas, 1766) |
| Aiptasia mutabilis (Gravenhorst, 1831) | Serpula vermicularis Linnaeus, 1767 | Botrylloides violaceus Oka, 1927 |
| Condylactis aurantiaca (Delle Chiaje, 1825) | Sabella spallanzanii (Gmelin, 1791) | |
| Phymanthus pulcher (Andrès, 1883) | Protula tubularia (Montagu, 1803) | |
| rs | p | |
|---|---|---|
| Total | 0.57 | <0.05 |
| Porifera | 0.32 | <0.05 |
| Mollusca | 0.56 | <0.05 |
| Chordata | 0.35 | <0.05 |
| Cnidaria | 0.28 | n.s. |
| Constant | 0.46 | <0.05 |
| Frequent | 0.60 | <0.05 |
| Temporary | 0.44 | <0.05 |
| Sessile | 0.51 | <0.05 |
| Vagile | 0.44 | <0.05 |
| Coralligenous | 0.51 | <0.05 |
| Groups | Adj R-Squared | p-Value | Slope | Intercept | Expected | Observed |
|---|---|---|---|---|---|---|
| Total | 0.338 | <0.001 | 0.291 | 3.796 | 22 | 21 ± 7 |
| Porifera | 0.210 | <0.001 | 0.236 | 2.896 | 10 | 9 ± 3 |
| Chordata | 0.091 | <0.001 | 0.257 | 2.017 | 4 | 5 ± 2 |
| Mollusca | 0.297 | <0.001 | 0.563 | 1.45 | 1 | 2 ± 2 |
| Constant | 0.246 | <0.001 | 0.214 | 3.481 | 19 | 17 ± 6 |
| Frequent | 0.176 | <0.01 | 0.426 | 2.003 | 3 | 3 ± 2 |
| Temporary | 0.179 | <0.01 | 0.466 | 1.443 | 1 | 1 ± 1 |
| Sessile | 0.312 | <0.001 | 0.258 | 3.597 | 20 | 18 ± 6 |
| Vagile | 0.234 | <0.001 | 0.459 | 2.012 | 4 | 3 ± 2 |
| Coralligenous | 0.259 | <0.001 | 0.312 | 2.395 | 5 | 6 ± 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bettoso, N.; Faresi, L.; Pitacco, V.; Orlando-Bonaca, M.; Aleffi, I.F.; Lipej, L. Species Richness of Benthic Macrofauna on Rocky Outcrops in the Adriatic Sea by Using Species-Area Relationship (SAR) Tools. Water 2023, 15, 318. https://doi.org/10.3390/w15020318
Bettoso N, Faresi L, Pitacco V, Orlando-Bonaca M, Aleffi IF, Lipej L. Species Richness of Benthic Macrofauna on Rocky Outcrops in the Adriatic Sea by Using Species-Area Relationship (SAR) Tools. Water. 2023; 15(2):318. https://doi.org/10.3390/w15020318
Chicago/Turabian StyleBettoso, Nicola, Lisa Faresi, Valentina Pitacco, Martina Orlando-Bonaca, Ida Floriana Aleffi, and Lovrenc Lipej. 2023. "Species Richness of Benthic Macrofauna on Rocky Outcrops in the Adriatic Sea by Using Species-Area Relationship (SAR) Tools" Water 15, no. 2: 318. https://doi.org/10.3390/w15020318
APA StyleBettoso, N., Faresi, L., Pitacco, V., Orlando-Bonaca, M., Aleffi, I. F., & Lipej, L. (2023). Species Richness of Benthic Macrofauna on Rocky Outcrops in the Adriatic Sea by Using Species-Area Relationship (SAR) Tools. Water, 15(2), 318. https://doi.org/10.3390/w15020318







