3.2.2. Validation of the Models
Table 5 shows a detailed summary of the fit and ANOVA for each one of the models describing EY and TPC. The squared-R quantifies the strength between the experimental data and responses predicted by the models. Obtained R
2-values suggest good adjustments, as 90.7% and 88.7% of the variability in EY and TPC are explained by the models. Adjusted determination coefficients (R
2adj) were also calculated, and the obtained values (89.3% and 86.6%, respectively for EY and TPC) are very close to the respective R
2-values, indicating that most of the variation is explained by variables actually affecting the responses [
8]. The accuracy of the models is also visible in the small root mean squared errors (RMS) (
Table 5).
The ANOVA approach separates the overall variance of the results into the variance associated with factor effects, and the variance related to the random error uses F-test to assess whether the former is significant when compared to the latter [
8]. As can be seen from
Table 5, F-ratio values are greater than the tabulated ones for a 95% significance level, and Prob > F are below 0.0001. These results confirm the significance of both models in describing EY and TPC results.
The adequacy of the fitted models was also evaluated by lack-of-fit analysis. In this test, the sum of squares of the residuals is divided into two parts: lack-of-fit error (based on model performance) and pure error (based on replicated measurements). The F-ratio is calculated by dividing the lack-of-fit mean square by the pure error mean square, and this statistic is used to determine whether the lack of fit is significant or not, at a certain significance level (95%, in this study) [
16]. The high F-ratio values obtained (3.6 for EY and 5.1 for TPC) and the corresponding error probabilities (Prob > F of 0.005 and 0.0008, respectively, for EY and TPC, both values lower than 0.05) indicate an undesirable significance of the lack-of-fit.
3.2.3. Effect of Extraction Conditions
This research aimed to optimize the conditions for extracting tannins from eucalyptus bark, and understand how the contact time, solvent volume, and temperature affect the amount and properties of the extract.
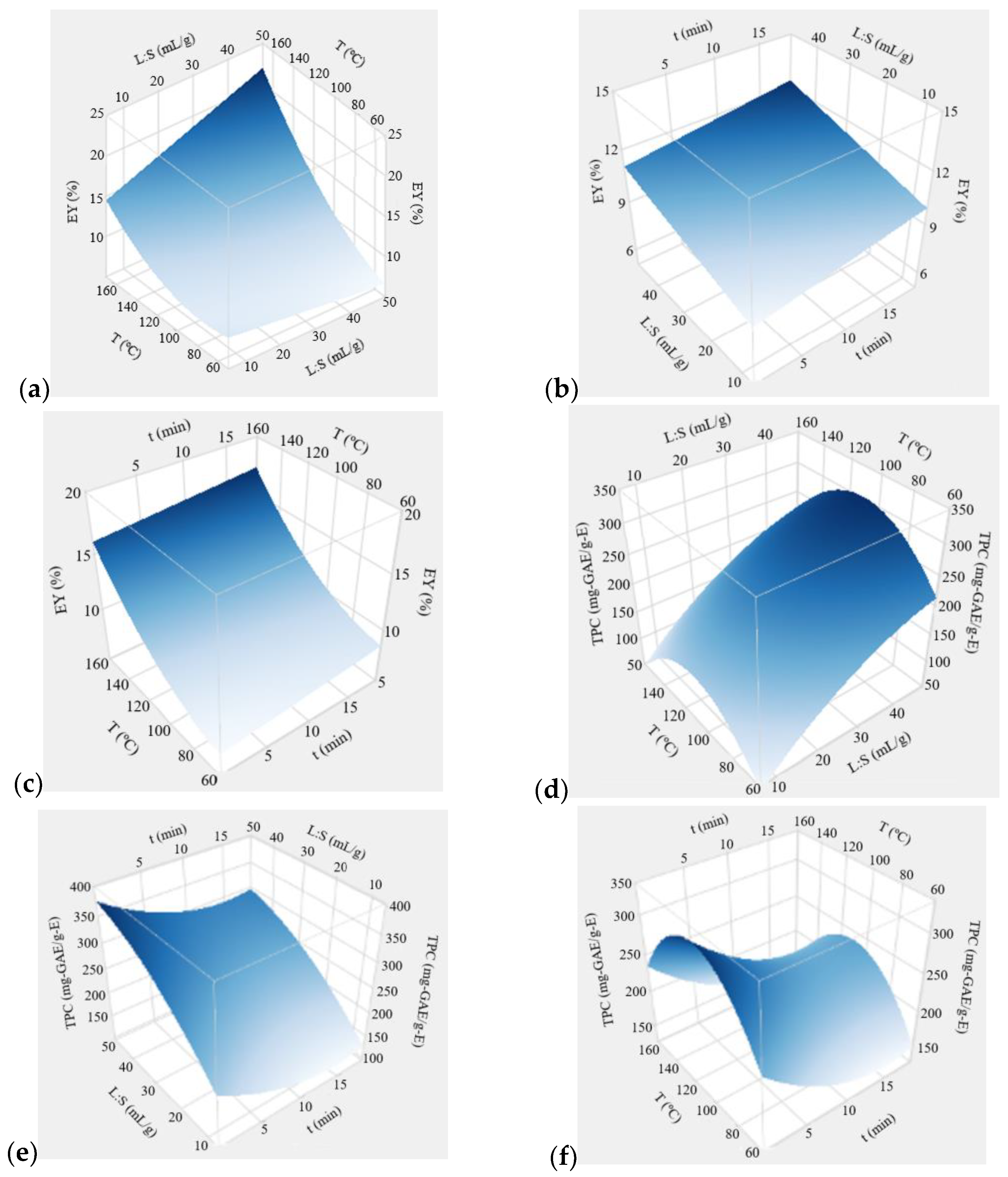
Figure 1 presents the three-dimensional response surface plots for EY and TPC, developed using the fitted models.
Results show that the extraction time exerts a statistically significant (albeit small) influence on the EY through a positive linear effect. The extraction yield presents very low improvements (typically 1.3%) when the extraction time increases from 0.25 to 18.8 min (
Figure 1b,c). The influence of this factor on the TPC is more marked, resulting from the a combination of linear negative and quadratic effects of time (
Table 4). The surface plots (
Figure 1e,f) show a decrease in TPC with increasing extraction times up to 16 min, followed by a very slight improvement in TPC. Maximum TPC values are observed for the shortest extraction time (0.25 min). This TPC versus time pattern has been also observed for flavonoids MAE from
Cannabis sativa L. [
17]. Ašperger et al. (2022) tested irradiation times in the range 5–60 min to extract polyphenols from grape skins and reported optimum conditions using the shortest studied time. This is explained by the ionic conduction and water dipole rotation effects caused by microwave heating, which quickly generates pressure within the plant cells, leading to a molecular interaction with the electromagnetic field and a fast energy transfer between the solvent and the plant material [
18]. Moreover, prolonged extraction and long-term thermal exposure may cause structural defects in sensitive extracted compounds [
20]. These results support one of the main advantages of MAE, the extraction speed, which is also interesting from economic and environmental points of view.
The L:S ratio significantly affected both extraction responses. According to the mass transfer principles, extraction will take place until an equilibrium is established between the concentrations of extracted compounds in the solid and the liquid phases. Higher L:S means a greater driving force for extraction, due to an increased concentration gradient, and an equilibrium shifts to the extraction side [
7]. This explains the results depicted in
Figure 1a, showing an almost linear increase in EY with increasing amounts of solvent, especially for average and high temperatures of the operating range. For low temperatures (below 80 °C), L:S exerted a very small decrease in EY. In these conditions, the extraction efficiency is low, and the effect of low pressure and agitation inside the vessel when higher L:S ratios (lower amount of EGB particles) are used may overlap the positive influence of the higher amount of solvent. Regarding the extract quality, the amount of solvent was the factor that most affected TPC.
Figure 1d,e show a marked increase in the phenolics content with raising L:S, also compatible with the enunciated mass transfer principle, and in line with other MAE studies. For instance, TPC values in carob kibble extracts obtained through MAE using ethanol aqueous mixtures increased with the increase of L:S, and the best-optimized value was found at the highest evaluated level (30 mL/g) [
21]. A similar effect was observed on polyphenols MAE from
Eucalyptus robusta leaves, using water as a solvent, where the optimal L:S was identified as 50 mL/g [
22].
The temperature was the most influential parameter for the EY response. Higher temperatures are associated with higher extraction yields as the result of positive linear, quadratic, and interaction effects of the factor (
Table 4). The variation of EY is especially significant for high liquid-to-solid ratios. Temperature directly affects the mass transfer coefficients and the solubility of compounds, being responsible for enhanced values. Increasing temperature reduces solvent viscosity and surface tension, which improves the wetting of the solid, the matrix penetration, and the solubility of compounds [
20]. The increase in the extraction yield with temperature is also reported for MAE applied to other plant materials, e.g., Russian olive leaves and flowers [
23]. However, the excessive temperature may cause thermal degradation of extracted compounds and a decline in extraction performance [
23]. The combination of two opposed effects (enhanced mass transfer, thermal degradation) explains the TPC variation observed in
Figure 1d,f. The phenolic content of the extract increased with the extraction temperature up to a maximum value observed at 111 °C. Above this level, an increase in temperature caused a decline in TPC. These results seem to corroborate a literature study on phenolics stability under MAE conditions, which indicates that all the evaluated compounds are stable up to 100 °C, but the degradation of a few phenolic compounds occurred at 125 °C [
24]. It is noteworthy that some works on MAE evaluate irradiation power as a factor instead of temperature, possibly because the operating temperature cannot be regulated in some microwave devices [
25]. The behavior of TPC as a function of power reported in those studies seems to be quite similar to that found here for temperature: an increase in TPC values with rising microwave power to a certain point, and a gradual decrease or stagnation as the irradiation power continued to rise [
25,
26]. The extraction temperature is expected to increase with the microwave power, although limited to the boiling point of the solvents at the observed pressure conditions. Whenever the temperature is not measured, different patterns of TPC variation with microwave power may be observed. Moreover, the importance of considering the irradiation power combined with sample size, i.e., power density, as a factor instead of the microwave power alone has been suggested [
25].
3.2.4. Extraction Optimization
The extraction yield and the total phenolic content were examined to determine the optimum conditions to extract polyphenols from the
Eucalyptus globulus bark. In the first approach, these two responses were optimized independently, and the results are presented in
Table 6. The extraction time, liquid-to-solid ratio, and temperature that maximize the extraction yield (optimum value: 20%) were obtained at the highest values evaluated for the three factors: 18.8 min, solvent amount of 48.5 mL/g, and temperature of 160 °C. Regarding the polyphenolic content of the extract, the maximum predicted value is 374 mg-GAE/g-E, expected to be obtained when the extraction is carried out for 15 s, using a water volume of 48.5 mL per gram of bark and at a temperature of 111 °C. In order to validate these predicted responses, experiments were performed under optimal conditions (
Table 6). Experimental results, named as EY
exp and TPC
exp (average values ± uncertainty), agree quite well with the values calculated from the developed models (EY
pred and TPC
pred; values ± 95% confidence interval). Condensed tannin contents (CT) were measured in the generated extracts. This class of tannins is the most interesting for coagulant production, due to the reactivity of nucleophilic sites. High contents of condensed tannins were obtained (no statistically significant difference among CT values in both extracts).
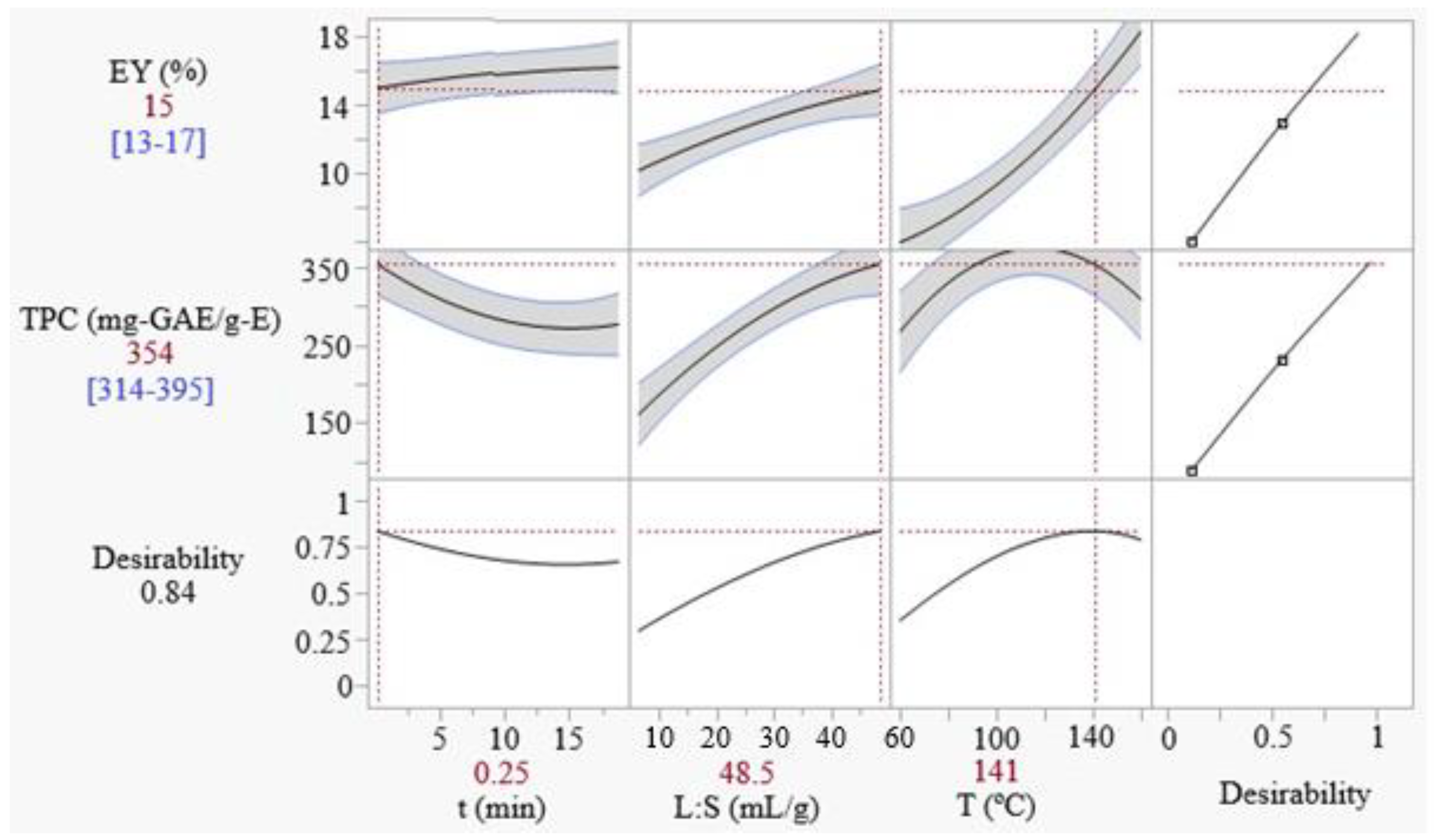
As seen in the previous section, antagonistic responses of EY and TPC are observed with the variation of the extraction time and temperature. Therefore, the desirability approach was used to maximize a function D that combines both responses, as explained in
Section 2.4.3. The optimal conditions that maximize the overall desirability D, representing an optimum compromise between EY and TPC, were found as: 15 s, 48.5 mL/g, and 141 °C. The predicted values for EY and TPC are 15 ± 2% and 354 ± 40 mg-GAE/g-E, respectively, corresponding to an overall desirability of 0.84. These predicted responses values are confirmed by experimental values of EY and TPC, obtained under the same conditions (15.3 ± 0.3% and 330 ± 64 mg-GAE/g).
Figure 2 shows the prediction profile of the responses near the overall optimum point. As it can be seen, the liquid-to-solid ratio is the factor that most dramatically affects the desirability value, followed by temperature. The optimization study here conducted has considered three operating variables (time, liquid-to-solid ratio, and temperature) and aimed to optimize the extraction parameters. From the point of view of reducing production costs, the lowest possible values of irradiation time, solvent amount, and temperature are desirable. In the case of extraction time, the optimum was obtained as the minimum tested value (15 s), which is interesting considering the minimization of energy. The optimum L:S ratio, on the other side, was obtained at the highest level tested (48.5 mL/g). A higher L:S means increased energy consumption to remove the solvent and concentrate the dry extract product (drying, freeze-drying, or spray-drying drying). The overall desirability shows to be relatively sensitive to the L:S ratio, which means that the use of reduced L:S ratios, compared to the optimum values, may cause significant losses to D. For instance, the use of 27.5 mL/g (average value of the range) would result in EY and TPC drops from 15% and 354 mg-GAE/g-E to 13% and 285 mg-GAE/g-E, respectively. A 20% reduction in L:S in respect to the optimum value, i.e. 40 mL/g, causes little variation in D (EY reduces from 15 to 14% and TPC from 354 to 333 mg-GAE/g-E). The extract drying is not a matter of the present study, but the models here developed for EY and TPC are useful, allowing to set the L:S to desired levels and calculate the predicted impact on the extraction parameters. Regarding the temperature, 141 °C was defined as the optimum. However, the extraction temperature can be significantly reduced to values of 100 °C, with a small impact on D. For instance, the desirability level that would be obtained if 111 °C was used is 0.76, representing a significant decrease in the EY (from 15 to 11%) and an increase in TPC (from 354 to 374 mg-GAE/g), compared to the use of the optimum temperature (141 °C).
3.2.5. Comparison to Literature
The specific characteristics of each extraction method make direct comparisons challenging. Eucalyptus bark origin, species, conservation, and particle size have an impact on the extraction results and are an additional source of variation. A literature survey on the extraction of eucalyptus bark and other plant parts led to the results shown in
Table 7.
Considering eucalyptus bark extractions through different methods and solvents, the reported values for EY and TPC are respectively in the ranges of 0.5–22% (typically above 7%) and 61–407 mg-GAE/g-E. The highest TPC in bark and other plant parts extracts has been obtained using organic solvents, which are undesirable from an environmental point of view. EY and TPC results obtained in this work by MAE, using water as the only extractant, are higher than the ones reported in the literature for SLE with the same solvent [
10,
12]. Compared to the values found using organic solvents, EY and TPC values obtained here, although lower, are quite significant. At optimized conditions, the number of polyphenols that can be extracted from EGB is 50.5 mg-GAE per gram of bark, which compares favorably with 37.8 mg-GAE/g obtained through SLE using methanol 50%
v/
v solution as extractant [
29] or 25.8 mg-GAE/g obtained with sodium hydroxide/sodium sulfite solution [
13]. However, the values are lower than the amounts that can be extracted from eucalyptus leaves.