The Ichthyofauna of the Bednja River, Ichthyological ‘Hot Spot’ in the Danube Basin—Exceptional Diversity under Strong Threats
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
4.1. The Importance of the Bednja River for Ichthyodiversity
4.2. Changes in the Composition of the Ichthyofauna of the Bednja River
4.3. Invasive Species in the Bednja River
4.4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Buj, I.; Šanda, R.; Marčić, Z.; Ćaleta, M.; Mrakovčić, M. Combining Morphology and Genetics in Resolving Taxonomy–A Systematic Revision of Spined Loaches (Genus Cobitis; Cypriniformes, Actinopterygii) in the Adriatic Watershed. PLoS ONE 2014, 9, e99833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buj, I.; Ćaleta, M.; Marčić, Z.; Šanda, R.; Vukić, J.; Mrakovčić, M. Different Histories, Different Destinies—Impact of Evolutionary History and Population Genetic Structure on Extinction Risk of the Adriatic Spined Loaches (Genus Cobitis; Cypriniformes, Actinopterygii). PLoS ONE 2015, 10, e0131580. [Google Scholar] [CrossRef] [PubMed]
- Buj, I.; Marčić, Z.; Ćaleta, M.; Šanda, R.; Giger, M.F.; Freyhof, J.; Machordom, A.; Vukić, J. Ancient connections among the European rivers and watersheds revealed from the evolutionary history of the genus Telestes (Actinopterygii; Cypriniformes). PLoS ONE 2017, 12, e0187366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buj, I.; Marčić, Z.; Čavlović, K.; Ćaleta, M.; Tutman, P.; Zanella, D.; Duplić, A.; Raguž, L.; Ivić, L.; Horvatić, S.; et al. Multilocus phylogenetic analysis helps to untangle the taxonomic puzzle of chubs (genus Squalius: Cypriniformes: Actinopteri) in the Adriatic basin of Croatia and Bosnia and Herzegovina. Zool. J. Linn. Soc. 2020, 189, 953–974. [Google Scholar] [CrossRef]
- Buj, I.; Knjaz, M.; Ćaleta, M.; Marčić, Z.; Ivić, L.; Onorato, L.; Šanda, R.; Vukić, J.; Horvatić, S.; Zanella, D.; et al. Phylogeographic pattern, genetic diversity, and evolutionary history of the enigmatic freshwater fish species Aulopyge huegelii (Actinopterygii: Cyprinidae). J. Zool. Syst. Evol. Res. 2021, 59, 2086–2102. [Google Scholar] [CrossRef]
- Marčić, Z.; Buj, I.; Duplić, A.; Ćaleta, M.; Mustafić, P.; Zanella, D.; Zupančič, P.; Mrakovčić, M. A new endemic cyprinid species from the Danube drainage. J. Fish Biol. 2011, 79, 418–430. [Google Scholar] [CrossRef]
- Mustafić, P.; Marčić, Z.; Duplić, A.; Mrakovčić, M.; Ćaleta, M.; Zanella, D.; Buj, I.; Podnar, M.; Dolenec, Z. A new loach species of the genus Cobitis in Croatia. Folia Zool. 2008, 57, 4–9. [Google Scholar]
- Mustafić, P.; Marčić, Z.; Zanella, D.; Ćaleta, M.; Buj, I.; Horvatić, S.; Mrakovčić, M. Ichthyofauna of the Vrana Lake Nature park. In Managing Lakes and Reservoirs in Croatia-Process, Protection and Evaluation. Scientific and Technical Conference with International Participation; Rubnić, J., Ed.; Hrvatsko društvo za zaštitu voda: Zagreb, Croatia, 4–6 May 2017; pp. 227–235. [Google Scholar]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes, 1st ed.; Kottelat: Cornol, Switzerland; Freyhof: Berlin, Germany, 2007; p. 646. [Google Scholar]
- Stagl, J.C.; Hattermann, F.F. Impacts of Climate Change on Riverine Ecosystems: Alterations of Ecologically Relevant Flow Dynamics in the Danube River and Its Major Tributaries. Water 2016, 8, 566. [Google Scholar] [CrossRef] [Green Version]
- Lenhardt, M.; Smederevac-Lalić, M.; Hegediš, A.; Skorić, S.; Cvijanović, C.; Višnjić-Jeftić, Ž.; Djikanović, V.; Jovičić, K.; Jaćimović, M.; Jarić, I. Human Impacts on Fish Fauna in the Danube River in Serbia: Current Status and Ecological Implications, 1st ed.; Bănăduc, D., Curtean-Bănăduc, A., Pedrotti, F., Cianfaglione, K., Akeroyd, J., Eds.; Springer: Cham, Switzerland, 2020; pp. 257–279. [Google Scholar]
- Petrić, H. On some settlements in the Bednja River basin during Middle and Early New Age. Kaj Časopis Za Književnost Umjet. I Kult. 2010, 43, 91–116. (In Croatian) [Google Scholar]
- Počkal, M. Hidrographic sizes of the Bednja River basin. Geogr. Glas. 1982, 44, 85–88. (In Croatian) [Google Scholar]
- Šloser, J.K.K. Kalnička gora and its natural features. Rad Jugosl. Akad. Znan. I Umjet. 1870, 11, 145–227. (In Croatian) [Google Scholar]
- Sebišanović, G. Ichthyological Reflections, 1st ed.; Tiskarski zavod Platzera sina: Varaždin, Croatia, 1890; p. 82. [Google Scholar]
- Brusina, S. Scraps for Croatian ichthyology and for fisheries. Glas. Hrvat. Naravosl. Društva 1892, 1–6, 221–288. [Google Scholar]
- Medić, M. Second round of ichthyological notes. Rad Jugosl. Akad. Znan. I Umjet. 1901, 30, 139–191. [Google Scholar]
- Medić, M. Ziege (Pelecus cultratus Linné) and Cactus roach (Leuciscus virgo Heckel). Glas. Zemalj. Muz. U Bosni I Hercegovini 1902, 1, 113–122. [Google Scholar]
- Hirc, D. Contributions to the fauna and flora of Kalnička Gora. Glas. Hrvat. Naravosl. Društva 1911, 23, 46–53. [Google Scholar]
- Langhoffer, A. Notizen aus dem kroatische zoologischen Landesmuseum. Glas. Hrvat. Naravosl. Društva 1915, 27, 58–59. [Google Scholar]
- Samobor, S. Fishing in the Bednja Stream. Lovačko Ribarski Vjesn. 1935, 44, 371–374. [Google Scholar]
- Taler, Z. Huchen (Salmo hucho L.) and Neretva Marble Trout (Salmo marmoratus Cuv.) Zoogeographic Dana and Commercial Value, 1st ed.; Pučka knjižara Priroda: Zagreb, Croatia, 1945; pp. 71–77. [Google Scholar]
- Zanandrea, G. Le lamprede dei musei di Zagabria e Lubiana. Biološki Glas. 1958, 11, 45–54. [Google Scholar]
- Treer, T.; Habeković, D.; Aničić, I.; Safner, R.; Kolak, A. Standard growth curve for chub (Leuciscus cephalus L. 1758) in Croatia. Ribarstvo 1997, 55, 47–52. [Google Scholar]
- Treer, T.; Habeković, D.; Safner, R.; Kolak, A.; Aničić, I. Length-mass relationship in chub (Leuciscus cephalus) from five Croatian rivers. Poljopr. Znan. Smotra 1999, 64, 137–142. [Google Scholar]
- Treer, T.; Safner, R.; Aničić, I.; Kolak, A.; Dražić, M. Morphological variation among four strains of common carp Cyprinus carpio in Croatia. Folia Zool. 2000, 49, 69–74. [Google Scholar]
- Miranda, L.E.; Spencer, A.B. Understanding the output of a Smith-Root GPP electrofisher. North Am. J. Fish. Manag. 2005, 25, 848–852. [Google Scholar] [CrossRef]
- Dodds, W.K.; Whiles, M.R. Fish Ecology and Fisheries. In Aquatic Ecology, Freshwater Ecology, 2nd ed.; Dodds, W.K., Whiles, M.R., Eds.; Academic Press: Cambridge, MA, USA, 2010; pp. 611–633. [Google Scholar]
- McAleece, N.; Gage, J.D.G.; Lambshead, P.J.D.; Paterson, G.L.J. BioDiversity Professional Statistics Analysis Software, 1st ed.; Jointly developed by the Scottish Association for Marine Science and the Natural History Museum: London, United Kingdom, 1997. [Google Scholar]
- Mrakovčić, M.; Brigić, A.; Buj, I.; Ćaleta, M.; Mutafić, P.; Zanella, D. Red Book of Freshwater Fish of Croatia, 1st ed.; Ministry of Culture, State Institute for Nature Protection: zagreb, Republic of Croatia, 2006. [Google Scholar]
- Tougard, C.; Vukić, J.; Ahnelt, H.; Buj, I.; Kovačić, M.; Moro, G.A.; Tutman, P.; Šanda, R. Quaternary climatic cycles promoted (re)colonization and diversification events in Adriatic sand gobies. J. Zool. Syst. Evol. Res. 2021, 59, 1037–1052. [Google Scholar] [CrossRef]
- Primack, R.B. Essentials of Conservation Biology, 4th ed.; Sinauer Associates: Sunderland, UK, 2006. [Google Scholar]
- Witkowski, A.; Bajić, A.; Treer, T.; Hegediš, A.; Marić, S.; Šprem, N.; Piria, M.; Kapusta, A. Past and present of and perspectives for the Danube huchen, Hucho hucho (L.), in the Danube basin. Arch. Pol. Fish. 2013, 21, 129–142. [Google Scholar] [CrossRef]
- Buj, I.; Raguž, L.; Marčić, Z.; Ćaleta, M.; Duplić, A.; Zanella, D.; Mustafić, P.; Ivić, L.; Horvatić, S.; Karlović, R. Plitvice Lakes National park harbors ancient, yet endangered diversity of trout (genus Salmo). J. Appl. Ichthyol. 2020, 37, 20–37. [Google Scholar] [CrossRef]
- Corbacho, C.; Sánchez, J. Patterns of species richness and introduced species in native freshwater fish faunas of a Mediterranean-type basin: The Guadiana River (southwest Iberian Peninsula). Regul. Rivers Res. Manag. 2001, 17, 699–707. [Google Scholar] [CrossRef]
- Hermoso, V.; Clavero, M.; Blanco-Garrido, F.; Prenda, J. Invasive species and habitat degradation in Iberian streams: An analysis of their role in freshwater fish diversity loss. Ecol. Appl. 2011, 21, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Maceda-Veiga, A.; Mac Nally, R.; de Sostoa, A. The presence of non-native species is not associated with native fish sensitivity to water pollution in greatly hydrologically altered rivers. Sci. Total Environ. 2017, 607–608, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Vitule, J.; Freire, C.; Simberloff, D. Introduction of non-native freshwater fish can certainly be bad. Fish Fish. 2009, 10, 98–108. [Google Scholar] [CrossRef]
- Gozlan, R.; Britton, J.; Cowx, I.; Copp, G. Current knowledge on non-native freshwater fish introductions. J. Fish Biol. 2010, 76, 751–786. [Google Scholar] [CrossRef]
- Wacker, S.; Aronsen, T.; Karlsson, S.; Ugedal, O.; Diserud, O.H.; Ulvan, E.M.; Hindar, K.; Næsje, T.F. Selection against individuals from genetic introgression of escaped farmed salmon in a natural population of Atlantic salmon. Evol. Appl. 2021, 14, 1450–1460. [Google Scholar] [CrossRef] [PubMed]
- Mooney, H.A.; Cleland, E.E. The evolutionary impact of invasive species. Proc. Natl. Acad. Sci. 2001, 98, 5446–5451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Britton, J.; Davies, G.; Brazier, M. Eradication of the invasive Pseudorasbora parva results in increased growth and production of native fishes. Ecol. Freshw. Fish 2009, 18, 8–14. [Google Scholar] [CrossRef]
- Carpentier, A.; Gozlan, R.; Cucherousset, J.; Paillisson, J.; Marion, L. Is topmouth gudgeon Pseudorasbora parva responsible for the decline in sunbleak Leucaspius delineatus populations? J. Fish Biol. 2007, 71, 274–278. [Google Scholar] [CrossRef]
- Copp, G.; Fox, M. Growth and life history traits of introduced pumpkinseed (Lepomis gibbosus) in Europe, and the relevance to its potential invasiveness. Biol. Invaders Inland Waters: Profiles Distrib. Threat. 2007, 2, 289–306. [Google Scholar]
- Cucherousset, J.; Copp, G.; Fox, M.; Sterud, E.; van Kleef, H.; Verreycken, H.; Záhorská, E. Life-history traits and potential invasiveness of introduced pumpkinseed Lepomis gibbosus populations in northwestern Europe. Biol. Invasions 2009, 11, 2171–2180. [Google Scholar] [CrossRef] [Green Version]
- Jakovlić, I.; Piria, M.; Šprem, N.; Tomljanović, T.; Matulić, D.; Treer, T. Distribution, abundance and condition of invasive Ponto-Caspian gobies Ponticola kessleri (Günther, 1861), Neogobius fluviatilis (Pallas, 1814), and Neogobius melanostomus (Pallas, 1814) in the Sava River basin, Croatia. J. Appl. Ichthyol. 2015, 31, 888–894. [Google Scholar] [CrossRef]
- Tutman, P.; Zanella, D.; Horvatić, S.; Hamzić, A.; Adrović, A.; Dulčić, J.; Glamuzina, B. Freshwater gobies (Gobiidae) of Bosnia and Herzegovina: A review of the current status and distribution. J. Vertebr. Biol. 2020, 69, 20046–20061. [Google Scholar] [CrossRef]
- Perdikaris, C.; Ergolavou, A.; Gouva, E.; Nathanailides, C.; Chantzaropoulos, A.; Paschos, I. Carassius gibelio in Greece: The dominant naturalised invader of freshwaters. Rev. Fish Biol. Fish. 2011, 22, 17–27. [Google Scholar] [CrossRef]
- Copp, G.; Bianco, P.; Bogutskaya, N.; Eros, T.; Falka, I.; Ferreira, M.; Fox, M.; Freyhof, J.; Gozlan, R.; Grabowska, J.; et al. To be, or not to be, a non-native freshwater fish? J. Appl. Ichthyol. 2005, 21, 242–262. [Google Scholar] [CrossRef]
- Jaćimović, M.; Lenhardt, M.; Krpo-Ćetković, J.; Jarić, I.; Gačić, Z.; Hegediš, A. Boom-bust like dynamics of invasive black bullhead (Ameiurus melas) in Lake Sava (Serbia). Fish. Manag. Ecol. 2019, 26, 153–164. [Google Scholar] [CrossRef]
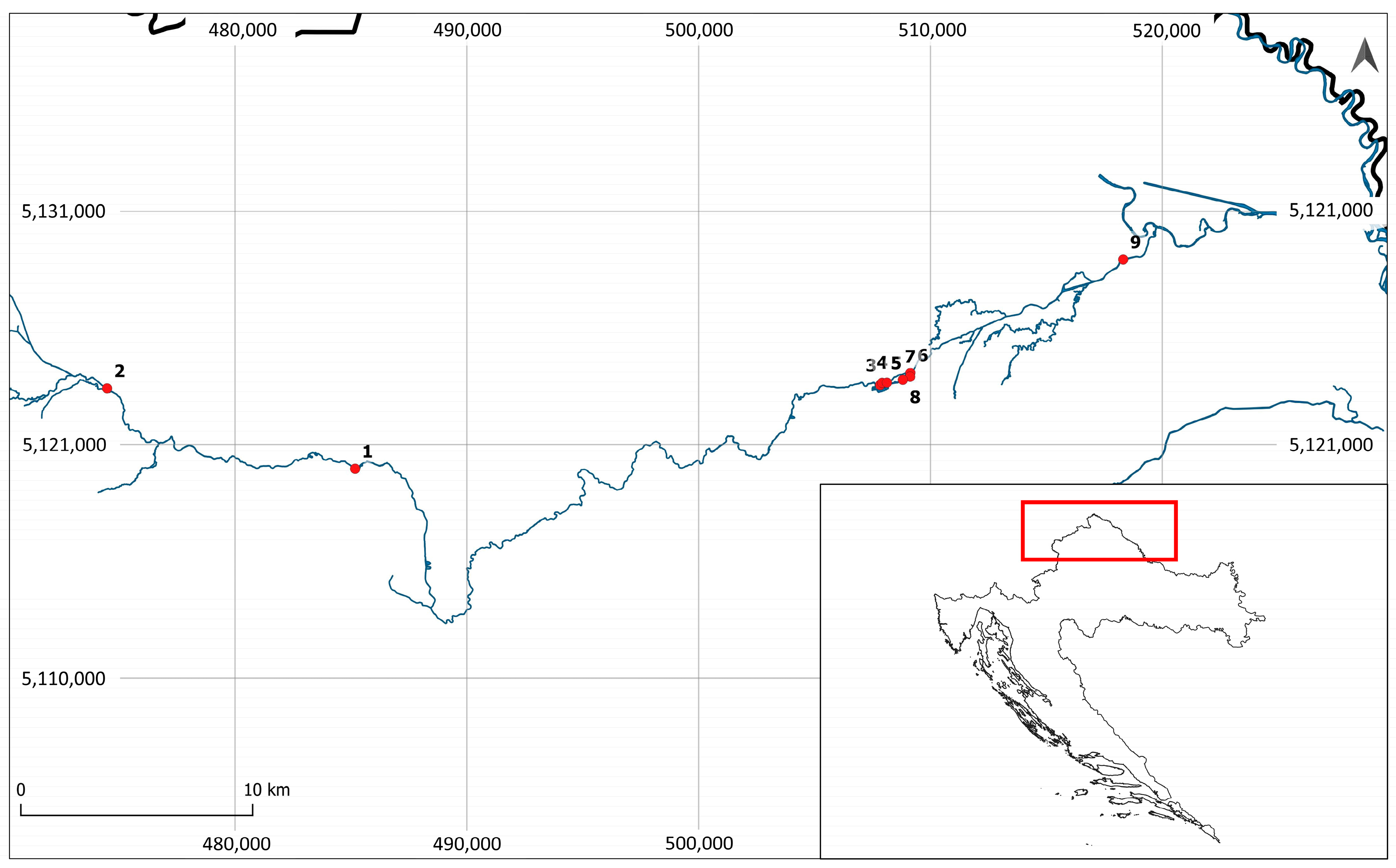
| Locality Number | Longitude | Latitude | Locality Name |
|---|---|---|---|
| 1 | 485181 | 5118966.75 | Beletinec |
| 2 | 474480 | 5122412 | Stažnjevec |
| 3 | 507830.4 | 5122559 | Bednja—accumulation above the dam |
| 4 | 507916.2 | 5122654 | Channel along the accumulation |
| 5 | 508122 | 5122660 | Bednja under the dam |
| 6 | 509132.8 | 5123079 | Accumulation above the dam in Ludbreg |
| 7 | 509122.8 | 5122911 | Bednja in Ludbreg |
| 8 | 508808 | 5122780 | Ludbreg Lake |
| 9 | 518315.9 | 5127936.12 | Mali Bukovec |
| Species Name | Locality 1 | Locality 2 | Locality 4 | Locality 6 | Locality 8 | Locality 7 | Locality 3 | Locality 5 | Locality 9 |
|---|---|---|---|---|---|---|---|---|---|
| Abramis brama | 12 | 18 | 4 | 7 | 2 | ||||
| Alburnoides bipunctatus | 13 | 140 | 25 | 95 | 31 | ||||
| Alburnus alburnus | 11 | 336 | 14 | 32 | 4 | 40 | 18 | ||
| Ameiurus melas1 | 4 | ||||||||
| Barbatula barbatula | 6 | ||||||||
| Barbus barbus | 1 | 135 | 6 | 26 | 17 | ||||
| Blicca bjoerkna | 2 | 2 | 11 | 5 | 1 | ||||
| Carassius carassius | 1 | ||||||||
| Carassius gibelio2 | 20 | 13 | 9 | ||||||
| Chondrostoma nasus | 22 | 20 | 3 | 17 | 18 | ||||
| Cobitis elongatoides | 2 | 38 | 7 | 20 | 33 | 6 | |||
| Cyprinus carpio | 2 | ||||||||
| Eudontomyzon vladykovi | 1 | ||||||||
| Esox lucius | 3 | 12 | 2 | 7 | |||||
| Gobio obtusirostris | 27 | 4 | 1 | ||||||
| Lepomis gibbosus3 | 1 | 2 | 2 | 18 | 3 | 5 | |||
| Leuciscus aspius | 2 | 1 | |||||||
| Leuciscus leuciscus | 3 | ||||||||
| Neogobius fluviatilis4 | 1 | ||||||||
| Perca fluviatilis | 2 | 7 | 5 | ||||||
| Pseudorasbora parva5 | 1 | 1 | |||||||
| Rhodeus amarus | 4 | 130 | 127 | 12 | 9 | ||||
| Romanogobio kesslerii | 1 | ||||||||
| Romanogobio vladykovi | 11 | 4 | 17 | 15 | 48 | ||||
| Rutilus rutilus | 1 | 60 | 18 | 147 | 16 | 115 | 41 | 15 | |
| Rutilus virgo | 1 | 35 | 4 | 53 | 4 | 43 | 4 | ||
| Sabanejewia balcanica | 4 | ||||||||
| Sander lucioperca | 2 | 2 | 4 | ||||||
| Scardinius erythrophthalmus | 69 | 26 | |||||||
| Silurus glanis | 1 | 1 | |||||||
| Squalius cephalus | 71 | 23 | 136 | 48 | 61 | 41 | |||
| Tinca tinca | 1 | ||||||||
| Vimba vimba | 53 | 32 | 1 | 7 |
| Species Name | Native/Foreign | Annexes of the Habitat Directive | EU Threatened Category (IUCN) | National Threatened Category [30] | Strictly Protected Species in Croatia |
|---|---|---|---|---|---|
| Abramis brama (Linnaeus, 1758) | Native | LC | / | ||
| Alburnoides bipunctatus (Bloch, 1782) | Native | LC | LC | ||
| Alburnus alburnus (Linnaeus, 1758) | Native | LC | / | ||
| Ameiurus melas Rafinesque, 1820 | Non-native | ||||
| Barbatula barbatula (Linnaeus, 1758) | Native | LC | / | ||
| Barbus balcanicus Kotlík, Tsigenopoulos, Ráb & Berrebi, 2002 | Native | II | LC | VU | |
| Barbus barbus (Linnaeus, 1758) | Native | V | LC | / | |
| Blicca argyroleuca Heckel, 1843 | Native | LC | / | ||
| Carassius gibelio (Bloch, 1782) | Non-native | ||||
| Carassius carassius (Linnaeus, 1758) | Native | LC | VU | yes | |
| Chondrostoma nasus (Linnaeus, 1758) | Native | LC | / | ||
| Cobitis elongatoides Băcescu & Mayer, 1969 | Native | II | LC | / | |
| Ctenopharyngodon idella Valenciennes, 1844 * | Non-native | ||||
| Cyprinus carpio Linnaeus, 1758 | Native | VU | EN | ||
| Eudontomyzon vladykovi Oliva & Zanandrea, 1959 | Native | II | LC | NT | yes |
| Esox lucius Linnaeus, 1758 | Native | LC | / | ||
| Gobio obtusirostris Valenciennes, 1842 | Native | LC | LC | ||
| Gymnocephalus cernua (Linnaeus, 1758) * | Native | LC | / | ||
| Hucho hucho (Linnaeus, 1758) * | Native | II, V | EN | EN | |
| Lepomis gibbosus (Linnaeus, 1758) | Non-native | ||||
| Leuciscus aspius (Linnaeus, 1758) | Native | II, V | LC | VU | |
| Leuciscus idus (Linnaeus, 1758) * | Native | LC | VU | ||
| Leuciscus leuciscus (Linnaeus, 1758) | Native | LC | / | ||
| Lota lota (Linnaeus, 1758) * | Native | LC | VU | ||
| Misgurnus fossilis (Linnaeus, 1758) * | Native | II | LC | VU | yes |
| Neogobius fluviatilis (Pallas, 1814) | Non-native | ||||
| Perca fluviatilis Linnaeus, 1758 | Native | LC | / | ||
| Phoxinus lumaireul Schinz, 1840 ** | Native | LC | / | ||
| Pseudorasbora parva (Temminck & Schlegel, 1846) | Non-native | ||||
| Rhodeus amarus (Bloch, 1782) | Native | II | LC | / | |
| Romanogobio kesslerii (Dybowski, 1862) | Native | II | LC | NT | yes |
| Romanogobio vladykovi (Fang, 1943) | Native | II | LC | DD | yes |
| Rutilus rutilus (Linnaeus, 1758) | Native | LC | / | ||
| Rutilus virgo (Heckel, 1852) | Native | II, V | LC | NT | |
| Sabanejewia balcanica (Karaman, 1922) | Native | II | LC | VU | yes |
| Salmo labrax Pallas, 1814 ** | Native | / | / | ||
| Sander lucioperca (Linnaeus, 1758) | Native | LC | / | ||
| Scardinius erythrophthalmus (Linnaeus, 1758) | Native | LC | / | ||
| Silurus glanis Linnaeus, 1758 | Native | LC | / | ||
| Squalius cephalus (Linnaeus, 1758) | Native | LC | / | ||
| Tinca tinca (Linnaeus, 1758) | Native | LC | / | ||
| Vimba vimba (Linnaeus, 1758) | Native | LC | VU |
| Species Richness, S | Shannon Index, H | Reciprocal Simpson Index, 1/D | Alpha Index, A | ||
|---|---|---|---|---|---|
| Indices based on the native species | |||||
| Beletinec | 6 | 0.411 | 1.489 | 1.463 | |
| Stažnjevec | 9 | 0.621 | 2.601 | 1.892 | |
| Channel along the accumulation in Ludbreg area | 15 | 0.767 | 6.192 | 2.461 | |
| Accumulation above the dam in Ludbreg | 7 | 0.870 | 5.167 | 1.948 | |
| Ludbreg Lake | 7 | 0.488 | 2.092 | 1.358 | |
| Bednja in Lubreg | 14 | 0.892 | 9.204 | 3.083 | |
| Channel Lubreg | 14 | 0.642 | 3.129 | 3.364 | |
| Bednja under the dam | 14 | 0.857 | 8.282 | 2.779 | |
| Mali Bukovec | 14 | 0.818 | 7.204 | 3.655 | |
| TOTAL SAMPLE | 28 | 0.755 | 9.834 | 4.336 | |
| Indices based on the entire samples, also including non-native species | |||||
| Beletinec | 6 | 0.411 | 1.489 | 1.463 | |
| Stažnjevec | 10 | 0.603 | 2.625 | 2.159 | |
| Channel along the accumulation in Ludbreg area | 18 | 0.726 | 6.236 | 3.060 | |
| Accumulation above the dam in Ludbreg | 9 | 0.830 | 5.618 | 2.716 | |
| Ludbreg Lake | 9 | 0.552 | 2.504 | 1.812 | |
| Bednja in Lubreg | 14 | 0.892 | 9.204 | 3.083 | |
| Channel Lubreg | 18 | 0.702 | 3.981 | 3.841 | |
| Bednja under the dam | 16 | 0.844 | 8.723 | 3.261 | |
| Mali Bukovec | 15 | 0.8123 | 7.609 | 3.970 | |
| TOTAL SAMPLE | 33 | 0.743 | 10.371 | 5.234 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buj, I.; Pleše, S.; Onorato, L.; Marčić, Z.; Mustafić, P.; Zanella, D.; Ćaleta, M.; Ivić, L.; Novoselec, L.; Renić, N.; et al. The Ichthyofauna of the Bednja River, Ichthyological ‘Hot Spot’ in the Danube Basin—Exceptional Diversity under Strong Threats. Water 2023, 15, 311. https://doi.org/10.3390/w15020311
Buj I, Pleše S, Onorato L, Marčić Z, Mustafić P, Zanella D, Ćaleta M, Ivić L, Novoselec L, Renić N, et al. The Ichthyofauna of the Bednja River, Ichthyological ‘Hot Spot’ in the Danube Basin—Exceptional Diversity under Strong Threats. Water. 2023; 15(2):311. https://doi.org/10.3390/w15020311
Chicago/Turabian StyleBuj, Ivana, Sara Pleše, Lucija Onorato, Zoran Marčić, Perica Mustafić, Davor Zanella, Marko Ćaleta, Lucija Ivić, Lucija Novoselec, Nikola Renić, and et al. 2023. "The Ichthyofauna of the Bednja River, Ichthyological ‘Hot Spot’ in the Danube Basin—Exceptional Diversity under Strong Threats" Water 15, no. 2: 311. https://doi.org/10.3390/w15020311





