eDNA Biomonitoring of Macroinvertebrate Communities for the Bioassessment of a River’s Ecological Status
Abstract
1. Introduction
2. Material and Methods
2.1. Study Area and eDNA Sampling
2.2. Determination of the Water Quality Parameters
2.3. eDNA Extraction and the High-Throughput Sequencing
2.4. Bioinformatics Analysis and Biodiversity
2.5. Bioassessment of the River’s Ecological Status
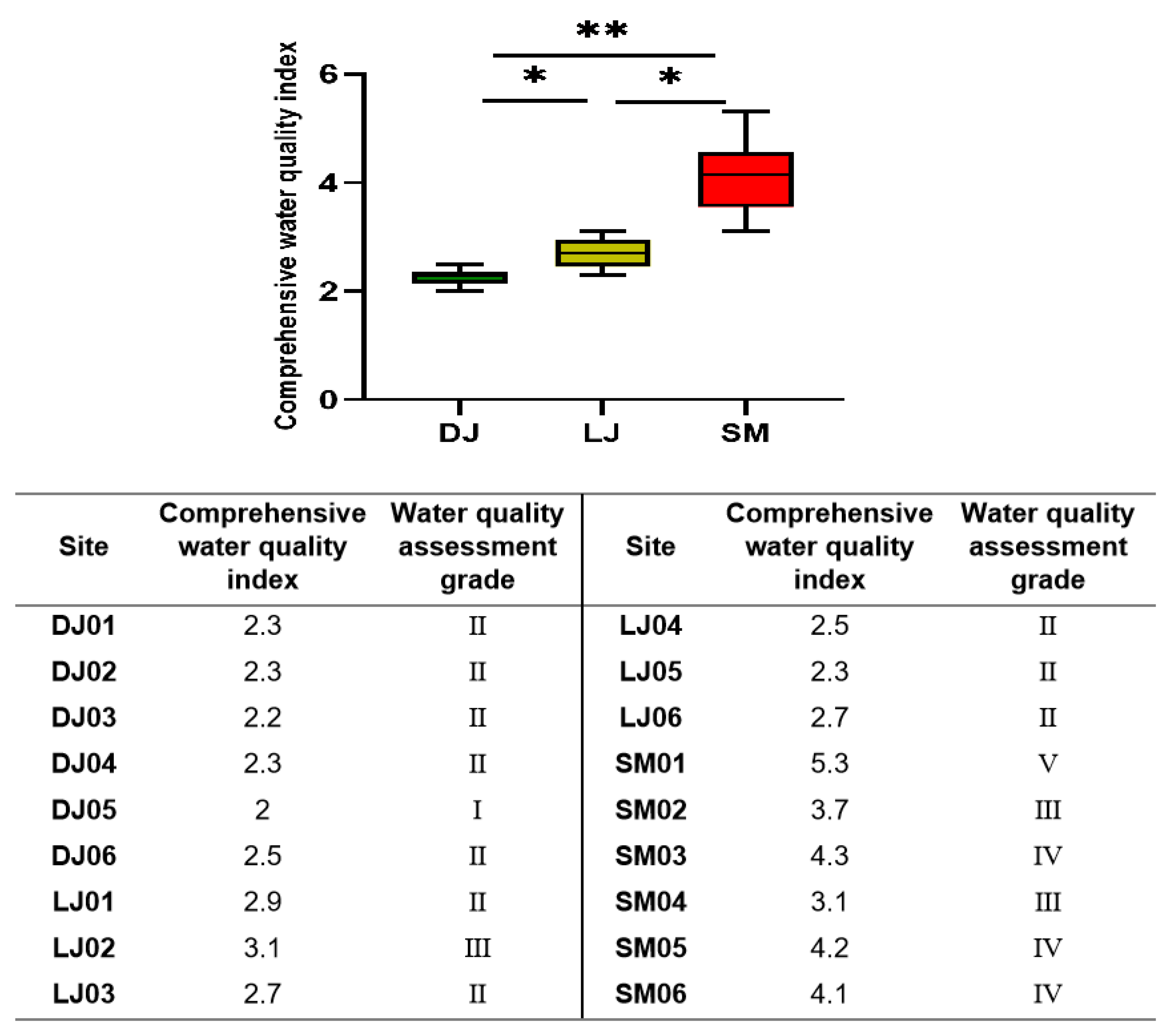
- Calculation of the comprehensive water quality index (WQI)
- 2.
- Biotic Pollution Index
- 3.
- Biotic Indices
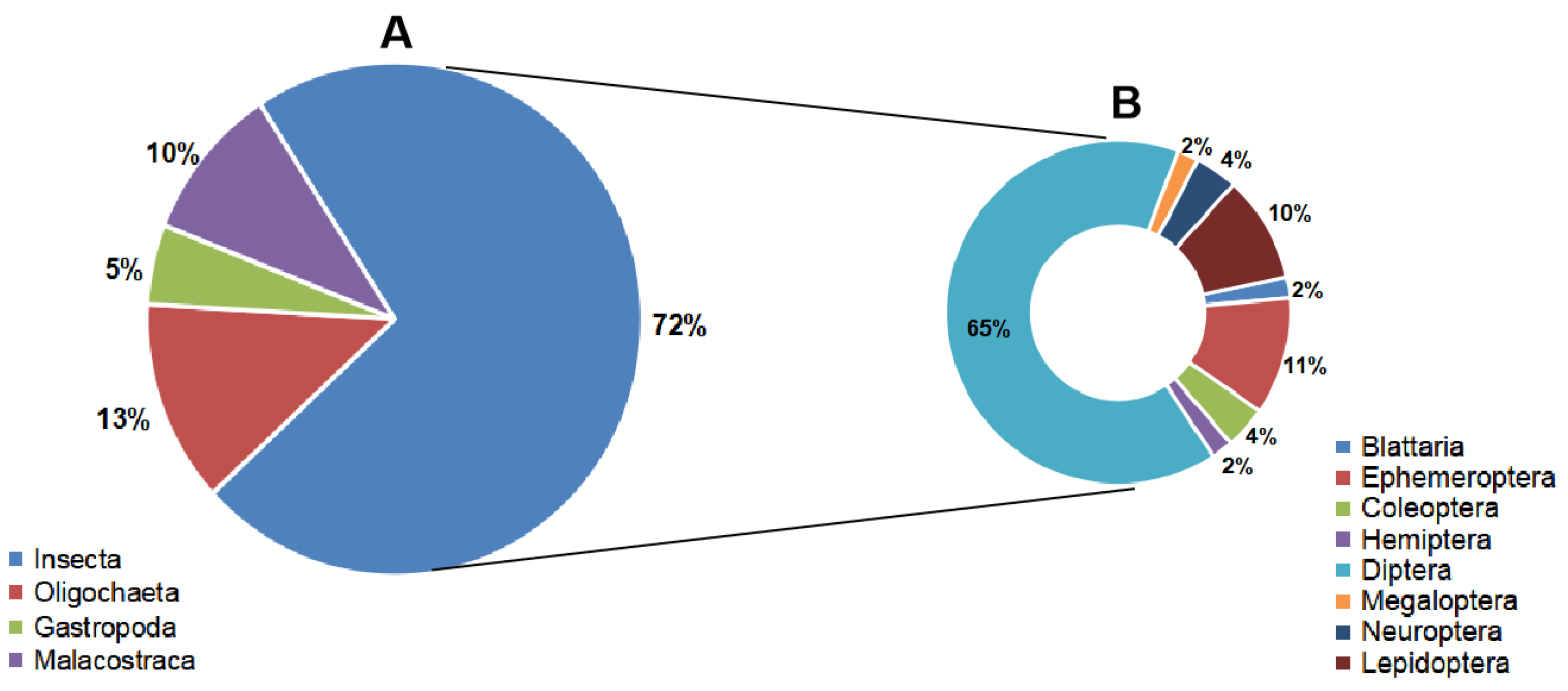
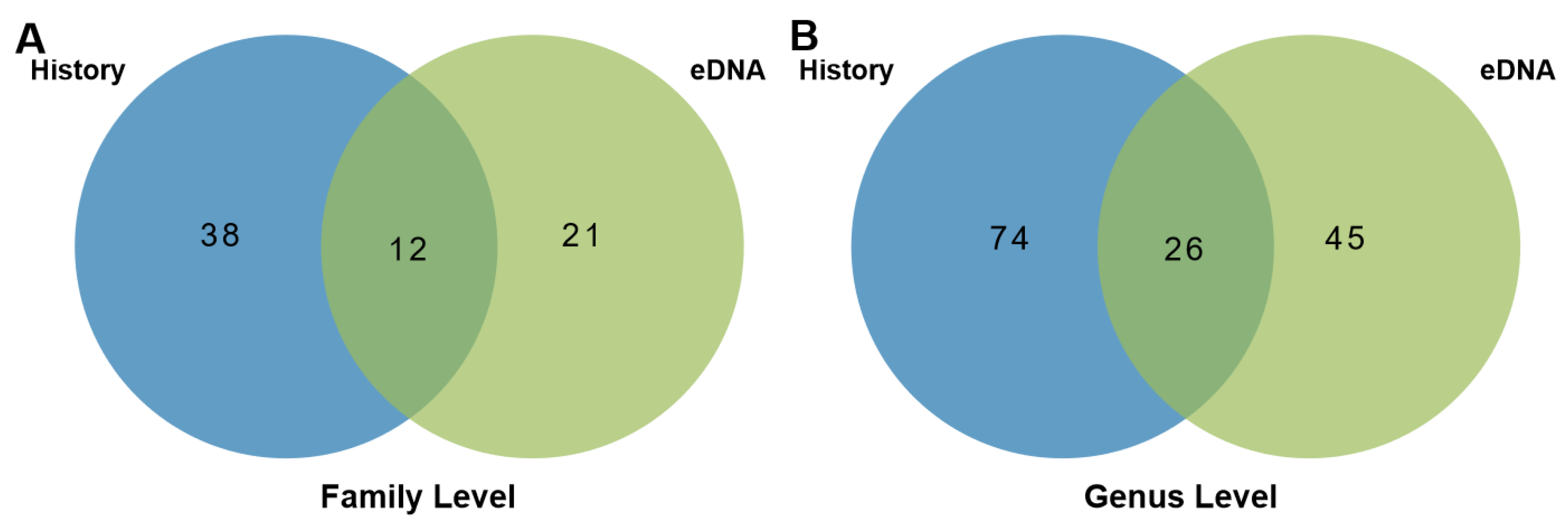
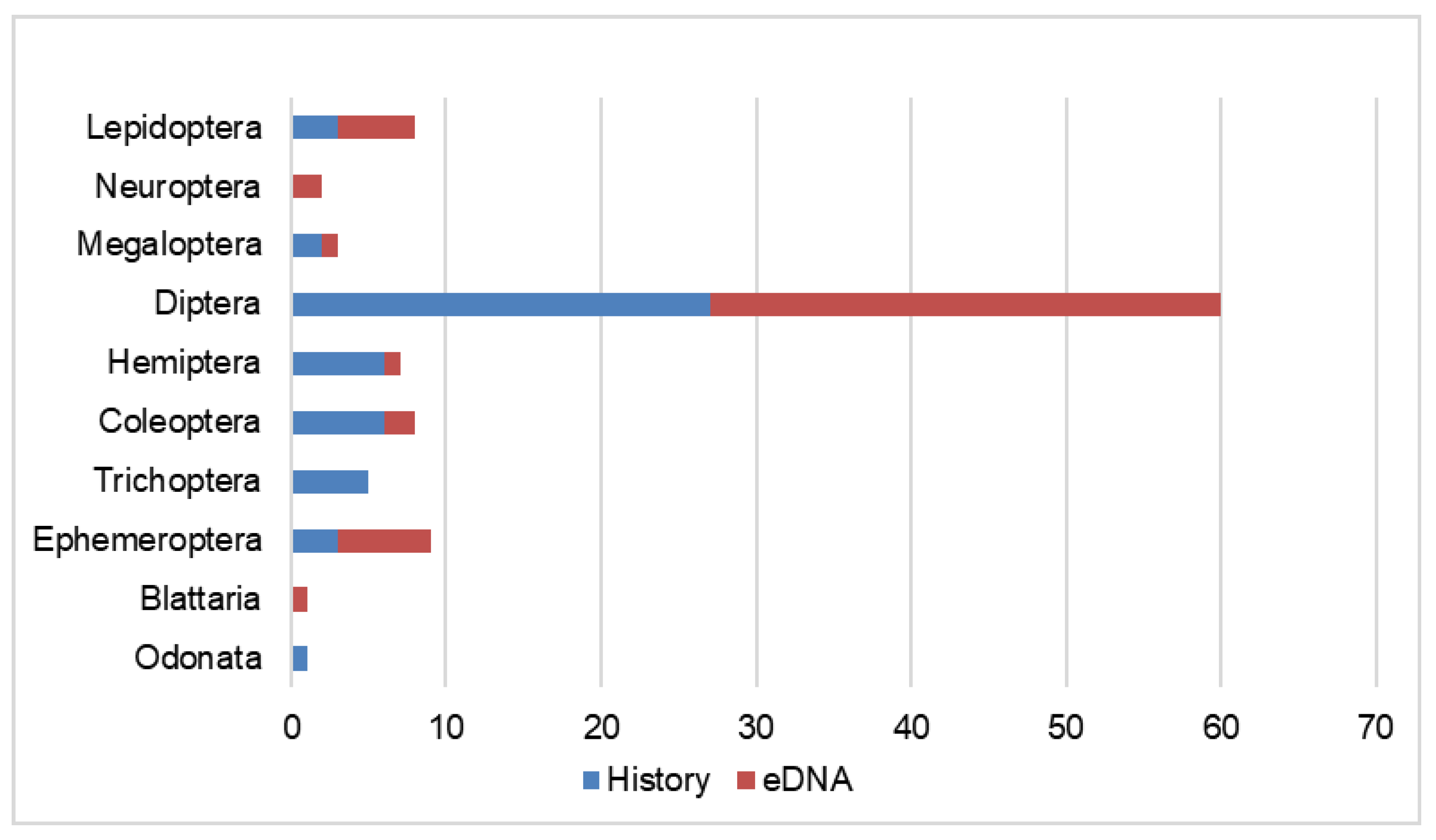
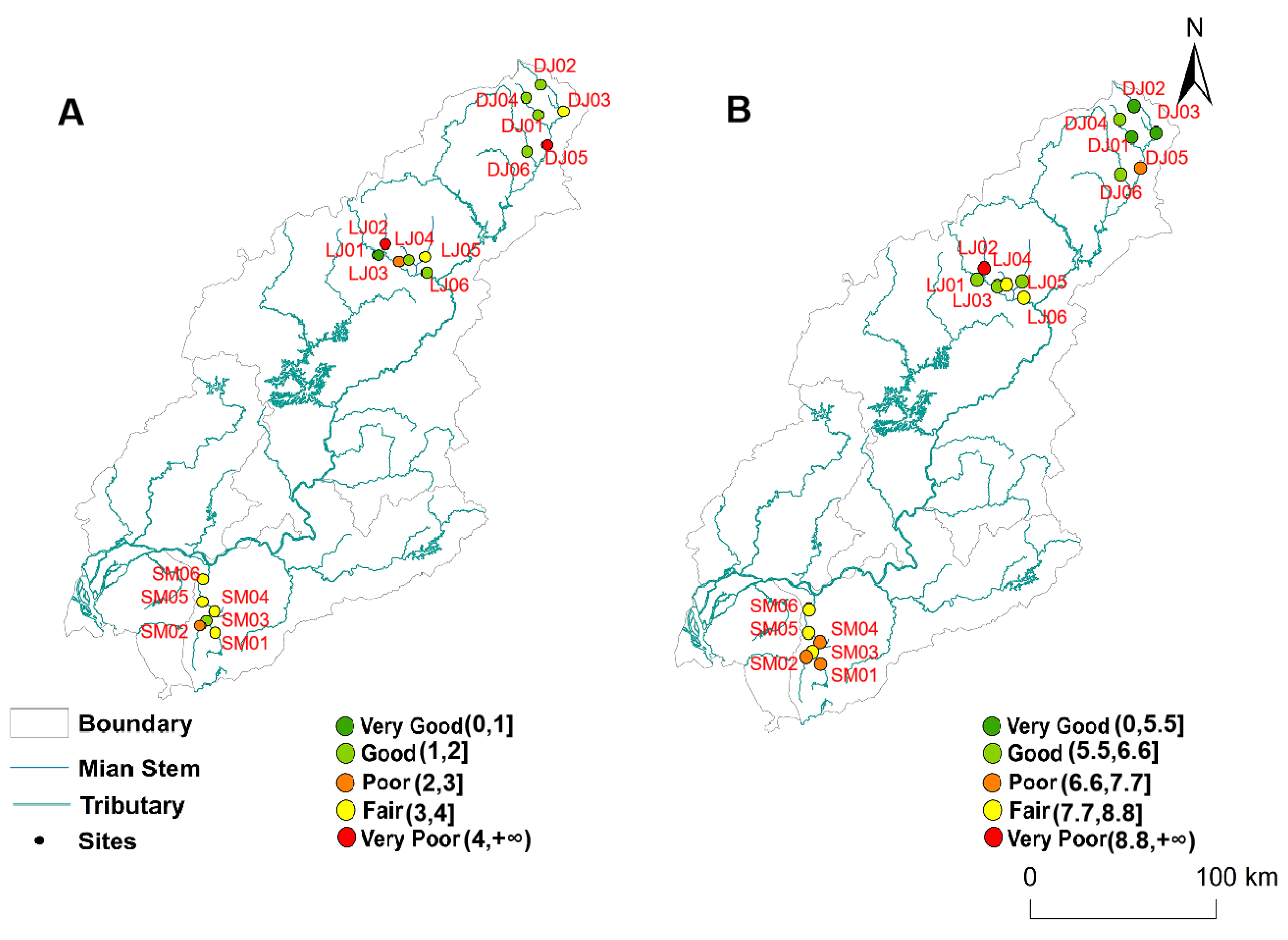
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Clarke, A.; Mac Nally, R.; Bond, N.; Lake, P.S. Macroinvertebrate Diversity in Headwater Streams: A Review. Freshw. Biol. 2008, 53, 1707–1721. [Google Scholar] [CrossRef]
- Sundar, S.; Heino, J.; Roque, F.D.O.; Simaika, J.P.; Melo, A.S.; Tonkin, J.D.; Gomes Nogueira, D.; Silva, D.P. Conservation of Freshwater Macroinvertebrate Biodiversity in Tropical Regions. Aquat. Conserv. Mar. Freshw. Ecosyst. 2020, 30, 1238–1250. [Google Scholar] [CrossRef]
- Masero, J.A.; Pérez-González, M.; Basadre, M.; Otero-Saavedra, M. Food Supply for Waders (Aves: Charadrii) in an Estuarine Area in the Bay of Cádiz (SW Iberian Peninsula). Acta Oecologica 1999, 20, 429–434. [Google Scholar] [CrossRef]
- Lessard, J.L.; Hayes, D.B. Effects of Elevated Water Temperature on Fish and Macroinvertebrate Communities below Small Dams. River Res. Appl. 2003, 19, 721–732. [Google Scholar] [CrossRef]
- Yetter, S.E. Freshwater Macroinvertebrates of the Mid-Atlantic Region. In Mid-Atlantic Freshwater Wetlands: Advances in Wetlands Science, Management, Policy, and Practice; Springer: New York, NY, USA, 2013; pp. 339–379. [Google Scholar] [CrossRef]
- Hill, M.J.; Death, R.G.; Mathers, K.L.; Ryves, D.B.; White, J.C.; Wood, P.J. Macroinvertebrate Community Composition and Diversity in Ephemeral and Perennial Ponds on Unregulated Floodplain Meadows in the UK. Hydrobiologia 2017, 793, 95–108. [Google Scholar] [CrossRef]
- Gore, J.A.; Layzer, J.B.; Mead, J. Macroinvertebrate Instream Flow Studies after 20 Years: A Role in Stream Management and Restoration. Regul. Rivers Res. Manag. 2001, 17, 527–542. [Google Scholar] [CrossRef]
- Nieto, C.; Ovando, X.M.C.; Loyola, R.; Izquierdo, A.; Romero, F.; Molineri, C.; Rodríguez, J.; Rueda Martín, P.; Fernández, H.; Manzo, V.; et al. The Role of Macroinvertebrates for Conservation of Freshwater Systems. Ecol. Evol. 2017, 7, 5502–5513. [Google Scholar] [CrossRef]
- Lindegaard, C. The Role of Zoobenthos in Energy Flow in Two Shallow Lakes. In Nutrient Dynamics and Biological Structure in Shallow Freshwater and Brackish Lakes; Springer Science & Business Media: Cham, Switzerland, 1994; pp. 313–322. [Google Scholar] [CrossRef]
- Borja, A.; Franco, J.; Pérez, V. A Marine Biotic Index to Establish the Ecological Quality of Soft-Bottom Benthos Within European Estuarine and Coastal Environments. Mar. Pollut. Bull. 2000, 40, 1100–1114. [Google Scholar] [CrossRef]
- Muxika, I.; Borja, Á.; Bald, J. Using Historical Data, Expert Judgement and Multivariate Analysis in Assessing Reference Conditions and Benthic Ecological Status, According to the European Water Framework Directive. Mar. Pollut. Bull. 2007, 55, 16–29. [Google Scholar] [CrossRef]
- Mistri, M.; Munari, C. BITS: A SMART Indicator for Soft-Bottom, Non-Tidal Lagoons. Mar. Pollut. Bull. 2008, 56, 587–599. [Google Scholar] [CrossRef]
- Resh, V.H. Which Group Is Best? Attributes of Different Biological Assemblages Used in Freshwater Biomonitoring Programs. Environ. Monit. Assess. 2008, 138, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Shogren, A.J.; Tank, J.L.; Egan, S.P.; Bolster, D.; Riis, T. Riverine Distribution of Mussel Environmental DNA Reflects a Balance among Density, Transport, and Removal Processes. Freshw. Biol. 2019, 64, 1467–1479. [Google Scholar] [CrossRef]
- Thomsen, P.F.; Willerslev, E. Environmental DNA—An Emerging Tool in Conservation for Monitoring Past and Present Biodiversity. Biol. Conserv. 2015, 183, 4–18. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, H. Combining Multiple Markers in Environmental DNA Metabarcoding to Assess Deep-Sea Benthic Biodiversity. Front. Mar. Sci. 2021, 8, 684955. [Google Scholar] [CrossRef]
- Bohmann, K.; Evans, A.; Gilbert, M.T.P.; Carvalho, G.R.; Creer, S.; Knapp, M.; Yu, D.W.; De Bruyn, M. Environmental DNA for Wildlife Biology and Biodiversity Monitoring. Trends Ecol. Evol. 2014, 29, 358–367. [Google Scholar] [CrossRef]
- Rees, H.C.; Maddison, B.C.; Middleditch, D.J.; Patmore, J.R.M.; Gough, K.C. REVIEW: The Detection of Aquatic Animal Species Using Environmental DNA—A Review of EDNA as a Survey Tool in Ecology. J. Appl. Ecol. 2014, 51, 1450–1459. [Google Scholar] [CrossRef]
- Mächler, E.; Deiner, K.; Steinmann, P.; Altermatt, F. Utility of Environmental DNA for Monitoring Rare and Indicator Macroinvertebrate Species. Freshw. Sci. 2014, 33, 1174–1183. [Google Scholar] [CrossRef]
- Valentini, A.; Taberlet, P.; Miaud, C.; Civade, R.; Herder, J.; Thomsen, P.F.; Bellemain, E.; Besnard, A.; Coissac, E.; Boyer, F.; et al. Next-Generation Monitoring of Aquatic Biodiversity Using Environmental DNA Metabarcoding. Mol. Ecol. 2016, 25, 929–942. [Google Scholar] [CrossRef]
- Jerde, C.L.; Mahon, A.R.; Chadderton, W.L.; Lodge, D.M. “Sight-Unseen” Detection of Rare Aquatic Species Using Environmental DNA. Conserv. Lett. 2011, 4, 150–157. [Google Scholar] [CrossRef]
- Harper, L.R.; Buxton, A.S.; Rees, H.C.; Bruce, K.; Brys, R.; Halfmaerten, D.; Read, D.S.; Watson, H.V.; Sayer, C.D.; Jones, E.P.; et al. Prospects and Challenges of Environmental DNA (EDNA) Monitoring in Freshwater Ponds. Hydrobiologia 2019, 826, 25–41. [Google Scholar] [CrossRef]
- Tzafesta, E.; Zangaro, F.; Specchia, V.; Pinna, M. An Overview of DNA-Based Applications for the Assessment of Benthic Macroinvertebrates Biodiversity in Mediterranean Aquatic Ecosystems. Diversity 2021, 13, 112. [Google Scholar] [CrossRef]
- Duarte, S.; Leite, B.R.; Feio, M.J.; Costa, F.O.; Filipe, A.F. Integration of DNA-Based Approaches in Aquatic Ecological Assessment Using Benthic Macroinvertebrates. Water 2021, 13, 331. [Google Scholar] [CrossRef]
- Blackman, R.C.; Ho, H.-C.; Walser, J.-C.; Altermatt, F. Spatio-Temporal Patterns of Multi-Trophic Biodiversity and Food-Web Characteristics Uncovered across a River Catchment Using Environmental DNA. Commun. Biol. 2022, 5, 259. [Google Scholar] [CrossRef]
- Liu, J.; Du, L.; Sun, Z.; Zhang, H. Comparison of Structure and Diversity of Benthic Communities in the Okinawa Trough and Mariana Trench by Environmental DNA Metabarcoding. Deep. Sea Res. Part I Oceanogr. Res. Pap. 2022, 185, 103806. [Google Scholar] [CrossRef]
- Serrana, J.M.; Yaegashi, S.; Kondoh, S.; Li, B.; Robinson, C.T.; Watanabe, K. Ecological Influence of Sediment Bypass Tunnels on Macroinvertebrates in Dam-Fragmented Rivers by DNA Metabarcoding. Sci. Rep. 2018, 8, 10185. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Zhang, S.; Lu, Q.; Chen, X.; Zhang, S.; Kong, Y.; Zhao, J. Fishing for Fish Environmental DNA: Ecological Applications, Methodological Considerations, Surveying Designs, and Ways Forward. Mol. Ecol. 2022, 31, 5132–5164. [Google Scholar] [CrossRef]
- Liang, Y.; Hong, H.C.; Dong, L.H.; Lan, C.Y.; Han, B.P.; Wong, M.H. Sources and Properties of Natural Organic Matter (NOM) in Water Along the Dongjiang River (the Source of Hong Kong’s Drinking Water) and Toxicological Assay of Its Chlorination By-Products. Arch. Environ. Contam. Toxicol. 2008, 54, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Jiang, Y.; Liu, Q.; Hou, Z.; Liao, J.; Fu, L.; Peng, Q. Influences of the Land Use Pattern on Water Quality in Low-Order Streams of the Dongjiang River Basin, China: A Multi-Scale Analysis. Sci. Total Environ. 2016, 551–552, 205–216. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, Q.-Q.; Zhang, S.-X.; Xing, C.; Ying, G.-G. Emission Estimation and Fate Modelling of Three Typical Pesticides in Dongjiang River Basin, China. Environ. Pollut. 2020, 258, 113660. [Google Scholar] [CrossRef]
- Zhou, T.; Wu, J.; Peng, S. Assessing the Effects of Landscape Pattern on River Water Quality at Multiple Scales: A Case Study of the Dongjiang River Watershed, China. Ecol. Indic. 2012, 23, 166–175. [Google Scholar] [CrossRef]
- Li, F.; Qin, S.; Wang, Z.; Zhang, Y.; Yang, Z. Environmental DNA Metabarcoding Reveals the Impact of Different Land Use on Multitrophic Biodiversity in Riverine Systems. Sci. Total Environ. 2023, 855, 158958. [Google Scholar] [CrossRef] [PubMed]
- Arimoro, F.O.; Keke, U.N. Stream Biodiversity and Monitoring in North Central, Nigeria: The Use of Macroinvertebrate Indicator Species as Surrogates. Environ. Sci. Pollut. Res. 2021, 28, 31003–31012. [Google Scholar] [CrossRef] [PubMed]
- Edegbene, A.O.; Odume, O.N.; Arimoro, F.O.; Keke, U.N. Identifying and Classifying Macroinvertebrate Indicator Signature Traits and Ecological Preferences along Urban Pollution Gradient in the Niger Delta. Environ. Pollut. 2021, 281, 117076. [Google Scholar] [CrossRef] [PubMed]
- Pinel-Alloul, B.; Méthot, G.; Lapierre, L.; Willsie, A. Macroinvertebrate Community as a Biological Indicator of Ecological and Toxicological Factors in Lake Saint-François (Québec). Environ. Pollut. 1996, 91, 65–87. [Google Scholar] [CrossRef]
- Li, F.; Guo, F.; Gao, W.; Cai, Y.; Zhang, Y.; Yang, Z. Environmental DNA Biomonitoring Reveals the Interactive Effects of Dams and Nutrient Enrichment on Aquatic Multitrophic Communities. Environ. Sci. Technol. 2022, 56, 16952–16963. [Google Scholar] [CrossRef]
- Leese, F.; Sander, M.; Buchner, D.; Elbrecht, V.; Haase, P.; Zizka, V.M.A. Improved Freshwater Macroinvertebrate Detection from Environmental DNA through Minimized Nontarget Amplification. Environ. DNA 2021, 3, 261–276. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Mahé, F. VSEARCH: A Versatile Open Source Tool for Metagenomics. PeerJ 2016, 4, e2584. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME Allows Analysis of High-Throughput Community Sequencing Data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and Clustering Orders of Magnitude Faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Chutter, F.M. An Empirical Biotic Index of the Quality of Water in South African Streams and Rivers. Water Res. 1972, 6, 19–30. [Google Scholar] [CrossRef]
- Whittier, T.R.; Sickle, J.V. Macroinvertebrate Tolerance Values and an Assemblage Tolerance Index (ATI) for Western USA Streams and Rivers. J. North Am. Benthol. Soc. 2010, 29, 852–866. [Google Scholar] [CrossRef]
- Pawlowski, J.; Bruce, K.; Panksep, K.; Aguirre, F.; Amalfitano, S.; Apotheloz-Perret-Gentil, L.; Baussant, T.; Bouchez, A.; Carugati, L.; Cermakova, K.; et al. Environmental DNA Metabarcoding for Benthic Monitoring: A Review of Sediment Sampling and DNA Extraction Methods. Sci. Total Environ. 2022, 818, 151783. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, K.D.; Thompson, L.R.; Duarte, B.; Kahlke, T.; Thompson, A.R.; Marques, J.C.; Cacador, I. DNA Sequencing as a Tool to Monitor Marine Ecological Status. Front. Mar. Sci. 2017, 4, 107. [Google Scholar] [CrossRef]
- Cai, Y.; Gong, Z.; Qin, B. Benthic Macroinvertebrate Community Structure in Lake Taihu, China: Effects of Trophic Status, Wind-Induced Disturbance and Habitat Complexity. J. Great Lakes Res. 2012, 38, 39–48. [Google Scholar] [CrossRef]
- Weerakoon, S.N.; Chandrasekara, W.U.; Amarasinghe, U.S. Seasonal Water--level Fluctuations and Changes in Macro--benthic Community Structure in Tropical Reservoirs: A Sri Lankan Case Study. Lakes Reserv. Res. Manag. 2021, 26, e12358. [Google Scholar] [CrossRef]
- Dai, S.; Bai, M.; Jia, H.; Xian, W.; Zhang, H. An Assessment of Seasonal Differences in Fish Populations in Laizhou Bay Using Environmental DNA and Conventional Resource Survey Techniques. Fishes 2022, 7, 250. [Google Scholar] [CrossRef]
- Piñol, J.; Mir, G.; Gomez-Polo, P.; Agustí, N. Universal and Blocking Primer Mismatches Limit the Use of High-Throughput DNA Sequencing for the Quantitative Metabarcoding of Arthropods. Mol. Ecol. Resour. 2015, 15, 819–830. [Google Scholar] [CrossRef]
- Hu, W.; Su, C.; Liu, Q.; Kong, Y.; Hua, S.; Hu, Z. Comparison of Fish Communities Using Environmental DNA Metabarcoding and Capture Methods in a Freshwater Lake: A New Set of Universal PCR Primers. Fish. Res. 2022, 253, 106365. [Google Scholar] [CrossRef]
- Gibson, J.; Shokralla, S.; Porter, T.M.; King, I.; Van Konynenburg, S.; Janzen, D.H.; Hallwachs, W.; Hajibabaei, M. Simultaneous Assessment of the Macrobiome and Microbiome in a Bulk Sample of Tropical Arthropods through DNA Metasystematics. Proc. Natl. Acad. Sci. USA 2014, 111, 8007–8012. [Google Scholar] [CrossRef]
- Wineland, S.M.; Welch, S.M.; Pauley, T.K.; Apodaca, J.J.; Olszack, M.; Mosher, J.J.; Holmes, J.N.; Waldron, J.L. Using Environmental DNA and Occupancy Modelling to Identify Drivers of Eastern Hellbender (Cryptobranchus Alleganiensis Alleganiensis) Extirpation. Freshw. Biol. 2019, 64, 208–221. [Google Scholar] [CrossRef]
- Shang, G.; Wang, X.; Zhu, L.; Liu, S.; Li, H.; Wang, Z.; Wang, B.; Zhang, Z. Heavy Metal Pollution in Xinfengjiang River Sediment and the Response of Fish Species Abundance to Heavy Metal Concentrations. Int. J. Environ. Res. Public Health 2022, 19, 11087. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Wu, N.; Tang, T.; Cai, Q.; Park, Y.-S. Effects of Heavy Metals on Benthic Macroinvertebrate Communities in High Mountain Streams. Ann. De Limnol. Int. J. Limnol. 2010, 46, 291–302. [Google Scholar] [CrossRef]
- Bian, B.; Zhou, Y.; Fang, B.B. Distribution of Heavy Metals and Benthic Macroinvertebrates: Impacts from Typical Inflow River Sediments in the Taihu Basin, China. Ecol. Indic. 2016, 69, 348–359. [Google Scholar] [CrossRef]
- Kang, S.-R.; King, S.L. Influence of Salinity and Prey Presence on the Survival of Aquatic Macroinvertebrates of a Freshwater Marsh. Aquat. Ecol. 2012, 46, 411–420. [Google Scholar] [CrossRef]
- Spellerberg, I.F.; Fedor, P.J. A Tribute to Claude Shannon (1916–2001) and a Plea for More Rigorous Use of Species Richness, Species Diversity and the ‘Shannon–Wiener’ Index. Glob. Ecol. Biogeogr. 2003, 12, 177–179. [Google Scholar] [CrossRef]
- Ding, Y.; Pan, B.; Zhao, G.; Sun, C.; Han, X.; Li, M. Geo-Climatic Factors Weaken the Effectiveness of Phytoplankton Diversity as a Water Quality Indicator in a Large Sediment-Laden River. Sci. Total Environ. 2021, 792, 148346. [Google Scholar] [CrossRef]
- Resh, V.H.; Norris, R.H.; Barbour, M.T. Design and Implementation of Rapid Assessment Approaches for Water Resource Monitoring Using Benthic Macroinvertebrates. Austral. Ecol. 1995, 20, 108–121. [Google Scholar] [CrossRef]
- Liu, L.; Xu, Z.; Yin, X.; Li, F.; Wang, M. Development of an Index Based on Fish, Macroinvertebrates, Phytoplankton, and Physicochemical Properties to Assess Urban Aquatic Ecosystems in Jinan, China. River Res. Appl. 2022, 39, 73–83. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, F.; Zou, Y.; Qin, S.; Li, F.; Zhang, Y. eDNA Biomonitoring of Macroinvertebrate Communities for the Bioassessment of a River’s Ecological Status. Water 2023, 15, 308. https://doi.org/10.3390/w15020308
Wu F, Zou Y, Qin S, Li F, Zhang Y. eDNA Biomonitoring of Macroinvertebrate Communities for the Bioassessment of a River’s Ecological Status. Water. 2023; 15(2):308. https://doi.org/10.3390/w15020308
Chicago/Turabian StyleWu, Feifei, Yanting Zou, Shan Qin, Feilong Li, and Yuan Zhang. 2023. "eDNA Biomonitoring of Macroinvertebrate Communities for the Bioassessment of a River’s Ecological Status" Water 15, no. 2: 308. https://doi.org/10.3390/w15020308
APA StyleWu, F., Zou, Y., Qin, S., Li, F., & Zhang, Y. (2023). eDNA Biomonitoring of Macroinvertebrate Communities for the Bioassessment of a River’s Ecological Status. Water, 15(2), 308. https://doi.org/10.3390/w15020308




