Study on the Mechanism and Control Strategy of Advanced Treatment of Yeast Wastewater by Ozone Catalytic Oxidation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Main Reaction Device and Online Monitoring System
2.3. Analysis and Test Instruments
2.4. Data Acquisition Conversion
3. Results and Discussions
3.1. Orthogonal Test Results and Optimization
3.2. Catalytic Ozonation Reaction Parameter Study
3.2.1. pH
3.2.2. ORP
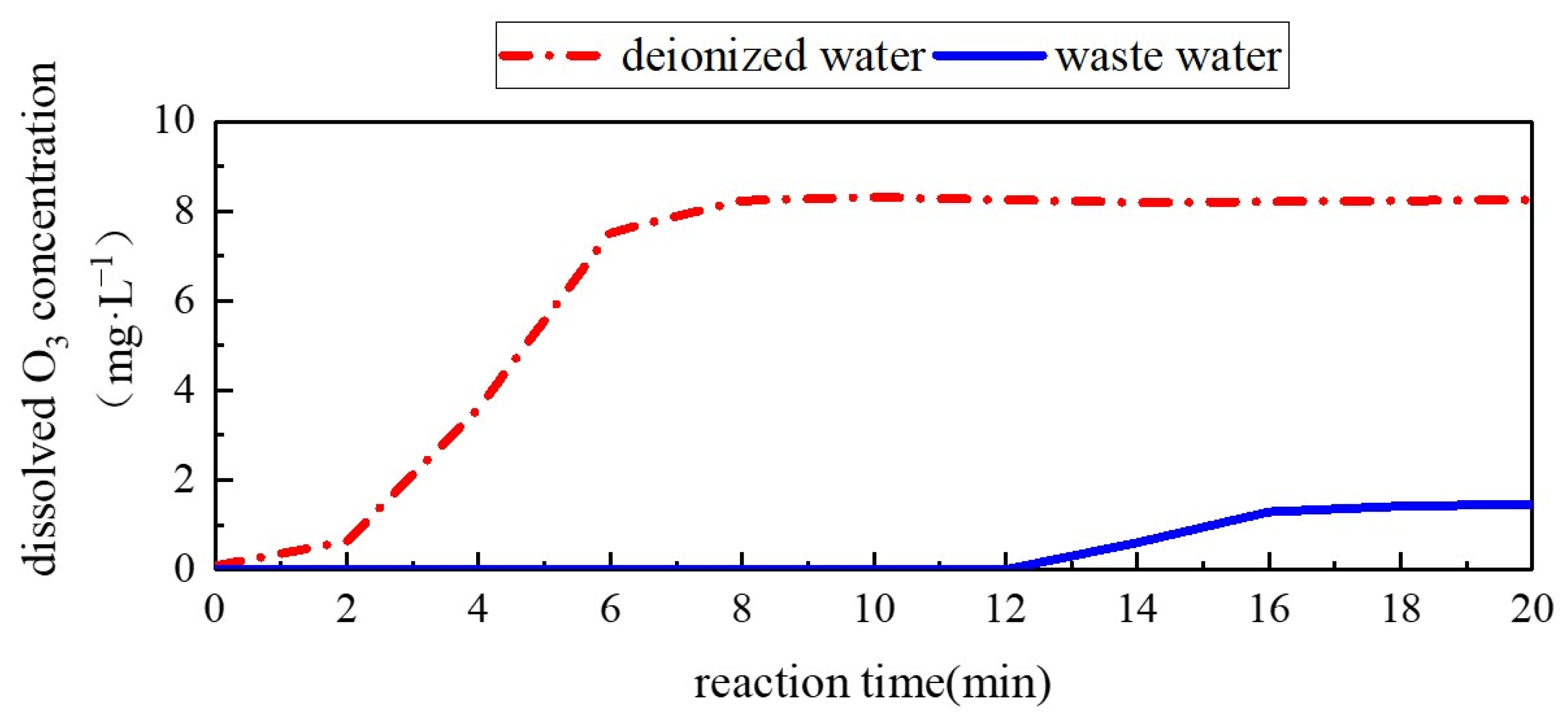
3.2.3. Liquid Phase Ozone Concentration
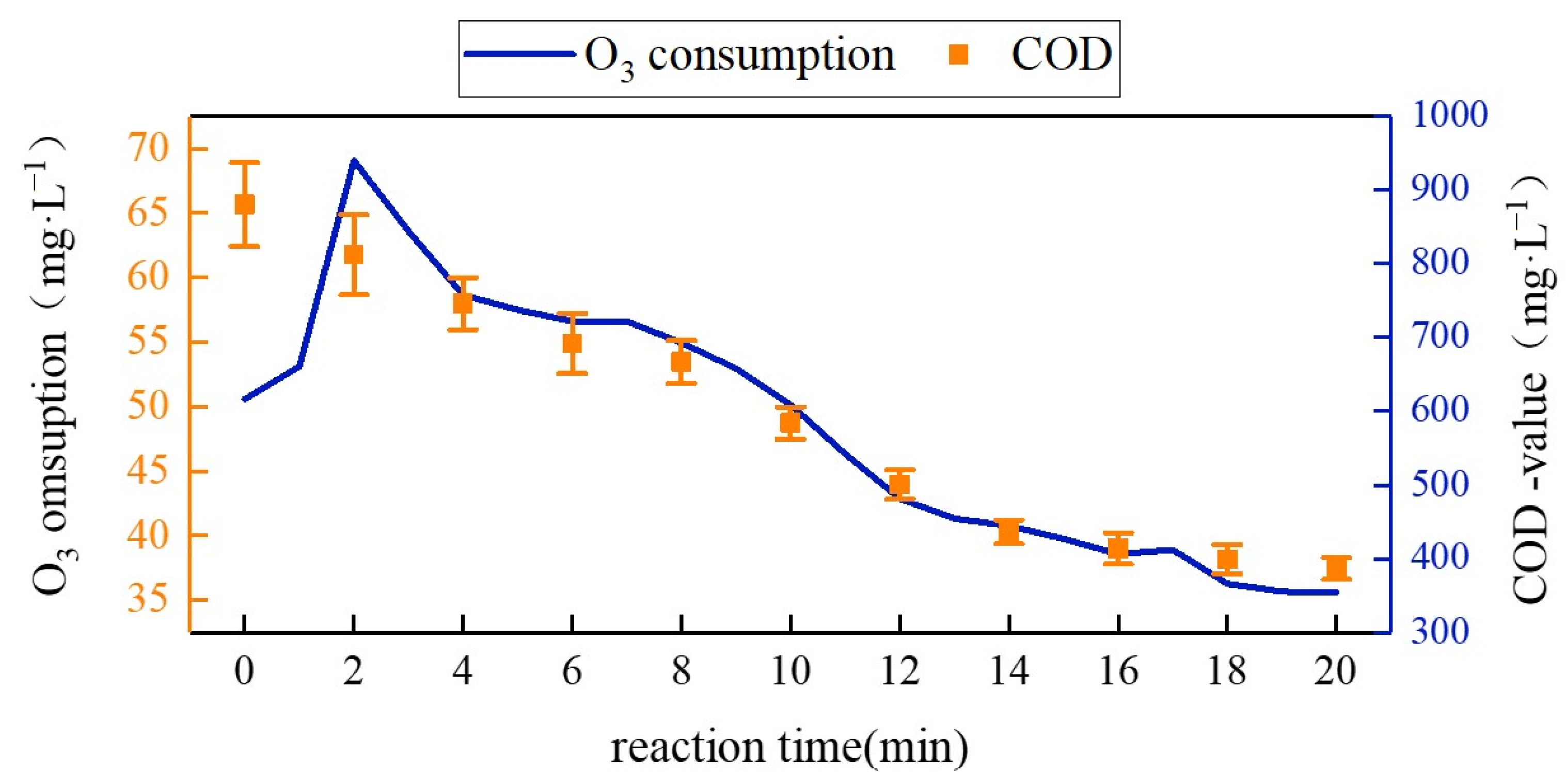
3.3. Study on Ozone Consumption and COD Removal Effect
3.4. Study on Ozone Consumption and Color Removal Effect
3.5. Comprehensive Analysis of Control Strategy for Process Monitoring Data
3.6. Analysis of Control Strategy for Catalytic Ozonation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moradi, M.; Zinatizadeh, A.A.; Zinadini, S.; Azizi, S.; Maaza, M. Decolorization of baker’s yeast wastewater by nanofiltration membrane and performance evaluation using response surface methodology (RSM). J. Iran. Chem. Soc. 2022, 19, 635–644. [Google Scholar] [CrossRef]
- Roshanak, R.K.; Ahmad, J.J.; Ali, E.; Babak, K.; Abdolmajid, G.; Ali, A. Optimization and evaluation of reactive dye adsorption on magnetic composite of activated carbon and iron oxide. Desalin. Water Treat. 2016, 57, 6411–6422. [Google Scholar]
- Ahmadi, E.; Yousefzadeh, S.; Ansari, M.; Ghaffari, H.R.; Azari, A.; Miri, M.; Mesdaghinia, A.; Nabizadeh, R.; Kakavandi, B.; Ahmadi, P.; et al. Performance, kinetic, and biodegradation pathway evaluation of anaerobic fixed film fixed bed reactor in removing phthalic acid esters from wastewater. Sci. Rep. 2017, 7, 41020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hashemi, S.S.; Karimi, K.; Taherzadeh, M.J. Integrated process for protein, pigments, and biogas production from baker’s yeast wastewater using filamentous fungi. Bioresour. Technol. 2021, 337, 125356. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.F.; Xie, X.; Lv, L.L. Study on the Process of Treating Brewery Wastewater by Anaerobic-Aerobic Combined Process. Guangdong Chem. Ind. 2019, 046, 75–77. [Google Scholar]
- Zhao, G.N.; Li, X.L.; Wu, D.D. Study On Yeast Wastewater Treatment Methods. For. Sci. Technol. Inf. 2016, 048, 59–64. [Google Scholar]
- He, J.Q.; Xiao, J.B.; Wang, H.D. Fenton Study on advanced treatment of yeast wastewater by Fenton reagent and magnesium sulfate. Ecol. Environ. 2007, 016, 399–401. [Google Scholar]
- Shi, Y. Coagulation-sedimentation Process for Pretreating Yeast Wastewater. Environ. Sci. Technol. 2010, 033, 141–145. [Google Scholar]
- Li, Z.H.; Xiao, D.G.; Liang, Y. Treatment of yeast wastewater with molasses as raw material. Liquor Making Sci. Technol. 2010, 7, 86–92. [Google Scholar]
- Zhou, X.; Liu, H.; Wang, Y.X. Advance in the treatment techniques of yeast wastewater. Ind. Water Treat. 2007, 27, 8–11. [Google Scholar]
- Wu, Z.Q.; Liang, S.Z.; Yao, R.H. Study on pigment characteristics and removal of sugarcane molasses. Environ. Pollut. Control 1997, 019, 05–43. [Google Scholar]
- Wu, Z.Q.; Liang, S.Z. Study on pigments from molasses alcohol stillage. Environ. Pollut. Control 2002, 2, 13–16. [Google Scholar]
- Wang, W.X.; Wei, C.H.; Wu, C.F.; Zhang, Y.P. Experiment of treating wastewater of a yeast plant by A/O biological fluidized beds and flocculation process. Ind. Water Treat. 2006, 4, 15–18. [Google Scholar]
- Dai, L.L.; Song, Y.H.; Jing, X.L.; Jia, L.M.; Lin, X.J.; Zhu, M. Application of ozone enhanced coagulation to the advanced treatment of yeast wastewater. Ind. Water Treat. 2014, 34, 62–64. [Google Scholar]
- Duran-Morenoa, A.; Garcia-Gonzaleza, S.A.; Gutierrez-Laraa, M.R.; Rigasb, F.; Ramírez-Zamora, R.M. Assessment of Fenton’s reagent and ozonation as pre-treatments for increasing the biodegradability of aqueous diethanolamine solutions from an oil refinery gas sweetening process. J. Hazard. Mater. 2011, 186, 1652–1659. [Google Scholar] [CrossRef]
- Akter, J.; Lee, J.; Kim, W.; Kim, I. Changes in Organics and Nitrogen during Ozonation of Anaerobic Digester Effluent. Water 2022, 14, 1425. [Google Scholar] [CrossRef]
- Pan, X. Degradation of organics in the mature and young landfill leachate by ozonation process. Desalin. Water Treat. 2020, 207, 160–171. [Google Scholar] [CrossRef]
- Al-Amshawee, S.K.A.; Yunus, M.Y.B.M.; Lynam, J.G. Non-catalytic ozonation of palm oil mill effluent (POME). Chem. Eng. Res. Des. 2021, 167, 169–182. [Google Scholar] [CrossRef]
- Zhou, J.Q.; Geng, C.Y.; Zhao, A.X. Pilot-scale experimental study on advanced treatment of printing and dyeing wastewater by catalytic ozonation. Ind. Water Treat. 2022, 048, 75–80. [Google Scholar]
- Erme, K.; Jogi, I. Metal Oxides as Catalysts and Adsorbents in Ozone Oxidation of NOx. Environ. Sci. Technol. 2019, 53, 5266–5271. [Google Scholar] [CrossRef]
- Wang, W.X.; Yao, H.; Yue, L.Z. Supported-catalyst CuO/AC with reduced cost and enhanced activity for the degradation of heavy oil refinery wastewater by catalytic ozonation process. Environ. Sci. Pollut. R. 2020, 27, 7199–7210. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.Y.; Zhang, F.Z.; Wu, H.Z.; Wei, C.H. The mineralization of oxalic acid and bio-treated coking wastewater by catalytic ozonation using nickel oxide. Environ. Sci. Pollut. R. 2018, 25, 2389–2400. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.W.; Shao, S.J.; Ding, X.; Li, Z.X.; Jing, J.X.; Jiao, W.Z.; Liu, Y.Z. Removal of phenol from wastewater by high-gravity intensified heterogeneous catalytic ozonation with activated carbon. Environ. Sci. Pollut. R. 2022, 29, 34830–34840. [Google Scholar] [CrossRef] [PubMed]
- Tichonovas, M.; Krugly, E.; Jankunaite, D.; Racys, V.; Martuzevicius, D. Ozone-UV-catalysis based advanced oxidation process for wastewater treatment. Environ. Sci. Pollut. R. 2017, 24, 17584–17597. [Google Scholar] [CrossRef] [PubMed]
- Olmez-Hanci, T.; Imren, C.; Arslan-Alaton, I.; Kabdasli, I.; Tunay, O. H2O2/UV-C oxidation of potential endocrine disrupting compounds: A case study with dimethyl phthalate. Photoch. Photobio. Sci. 2009, 8, 620–627. [Google Scholar] [CrossRef]
- Xu, J.; Tu, Y.; Wu, Q.; Zhang, Y.H.; Tang, M.; Li, J.; Chen, Y. Research on the ozone, ozone/hydrogen peroxide catalytic oxidation to the advanced treatment of chemical industrial wastewater. Ind. Water Treat. 2017, 037, 62–65. [Google Scholar]
- Souza, F.S.; da Silva, V.V.; Rosin, C.K.; Hainzenreder, L.; Arenzon, A.; Feris, L.A. Comparison of different advanced oxidation processes for the removal of amoxicillin in aqueous solution. Environ. Technol. 2018, 39, 549–557. [Google Scholar] [CrossRef]
- Qu, F.S.; Cao, A.K.; Yang, Y.; Mahmud, S.; Su, P.Y.; Yang, J.X.; He, Z.J.; Lai, Q.Y.; Zhu, L.J.; Tu, Z.P.; et al. Hierarchically superhydrophilic poly(vinylidene fluoride) membrane with self-cleaning fabricated by surface mineralization for stable separation of oily wastewater. J. Membrane Sci. 2021, 640, 119864. [Google Scholar] [CrossRef]
- Wang, H.; Lin, X.Z.; Huang, Y.X.; Bian, W.L.; Ma, L.M. Two advanced oxidation pathways of modified iron-shavings participation in ozonation. Sep. Purif. Technol. 2020, 244, 116838. [Google Scholar] [CrossRef]
- Faridul Hasan, K.M.; Wang, H.N.; Sakil, M.; Muhammad, A.T.; Cao, C.Y. Wool functionalization through AgNPs: Coloration, antibacterial and wastewater treatment. Surf. Innov. 2021, 9, 25–36. [Google Scholar] [CrossRef]
- Yun, Z.; Mahmud, S.; He, Z.K.; Yang, Y.; Zhang, Z.; Guo, F.; Chen, Z.H.; Zhu, X.; Zhao, Y.B. Graphene oxide modified membrane for highly efficient wastewater treatment by dynamic combination of nanofiltration and catalysis. J. Hazard. Mater. 2020, 397, 122774. [Google Scholar]
- Yang, S.S.; Guo, W.Q.; Meng, Z.H.; Zhou, X.J.; Feng, X.C.; Zheng, H.S.; Liu, B.; Ren, N.Q.; Cui, Y.S. Characterizing the fluorescent products of waste activated sludge in dissolved organic matter following ultrasound assisted ozone pretreatments. Bioresoure Technol. 2013, 131, 560–563. [Google Scholar] [CrossRef] [PubMed]
- Balcıoğlu, G.; Beril Gönder, Z. Baker’s yeast wastewater advanced treatment using ozonation and membrane process for irrigation reuse. Process Saf. Environ. 2018, 117, 43–50. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, D.X.; Feng, L.; Cheng, X.; Zhang, L.Q. Analyses on removal characteristics of organic compounds during activated carbon catalytic ozonation process treating yeast fermentation wastewater. J. Environ. Eng. 2017, 11, 5820–5827. [Google Scholar]
- Manh Khai, N.; Van Son, T.; Thi Thuy, P.; Hoang Giang, P.; Bich Loan, H.; Thao Huong, N.; Thi Ha, N.; Thi Huong, T.; Huu Hao, N. Fenton/ozone-based oxidation and coagulation processes for removing metals (Cu, Ni)-EDTA from plating wastewater. J. Water Process Eng. 2021, 39, 101836. [Google Scholar]
- Wang, T.; Le, T.; Hu, J.; Ravindra, A.V.; Xv, H.; Zhang, L.; Wang, S.; Yin, S. Ultrasonic-assisted ozone degradation of organic pollutants in industrial sulfuric acid. Ultrason. Sonochem. 2022, 86, 106043. [Google Scholar] [CrossRef]
- Chen, Z.H.; Mahumb, S.; Cai, L.R.; He, Z.J.; Yang, Y.; Zhang, L.; Zhao, S.F.; Zhu, X. Hierarchical poly(vinylidene fluoride)/active carbon composite membrane with self-confining functional carbon nanotube layer for intractable wastewater remediation. J. Membrane Sci. 2020, 603, 118041. [Google Scholar] [CrossRef]
- Han, R.; Fang, X.; Song, Y.; Wang, L.; Lu, Y.; Ma, H.; Xiao, H.; Shao, L. Study on the oxidation of ammonium sulfite by ozone in a rotating packed bed. Chem. Eng. Process. 2022, 173, 108820. [Google Scholar] [CrossRef]
- Jiang, G.; Zhao, Y.; Ma, C.; Wang, X. Research Progress of Ozone Oxidation Reaction Kinetics. Contemp. Chem. Ind. 2018, 47, 1071–1074. [Google Scholar]
- Xu, Z.L.; Qi, W.X.; Gu, P.P.; Guo, W.Z.; Zhao, L. Kinetics of Degradation of Unsymmetrical Dimethylhydrazine in Water by Hydrogen Peroxide Enhanced Ultraviolet Photocatalytic Ozone. Chem. Eng. Des. Commun. 2019, 45, 146. [Google Scholar]
- Li, J.; Zhang, L.; Liu, J.; Lin, J.; Peng, Y. Hydroxylamine addition and real-time aeration control in sewage nitritation system for reduced start-up period and improved process stability. Bioresour. Technol. 2019, 294, 122183. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Xu, L.P.; Li, H.P.; Li, J.; Tang, J.S. Research on and Application of Oxidation-reduction Potential. Earth Environ. 2014, 42, 430–436. [Google Scholar]
- Wang, G.; Jia, S.; Li, R.; Ma, S.; Chen, X.; Wu, Z.; Shi, G.; Niu, X. Seasonal variation characteristics of hydroxyl radical pollution and its potential formation mechanism during the daytime in Lanzhou. J. Environ. Sci. 2020, 95, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Cashman, M.A.; Kirschenbaum, L.; Holowachuk, J.; Boving, T.B. Identification of hydroxyl and sulfate free radicals involved in the reaction of 1,4-dioxane with peroxone activated persulfate oxidant. J. Hazard. Mater. 2019, 380, 120875. [Google Scholar] [CrossRef]
- Jing, X.L.; Li, Z.F. Process Research on Advanced Treatment of Yeast Wastewater through Enhanced Catalytic Ozonation. J. Residuals Sci. Tech. 2016, 13, S101–S109. [Google Scholar] [CrossRef]





| Num. | Parameter | Numerical Value | Unit |
|---|---|---|---|
| 1 | COD | 880 | mg·L−1 |
| 2 | Biochemical oxygen demand (BOD5) | 41 | mg·L−1 |
| 3 | Bicarbonate | 182 | mg·L−1 |
| 4 | Carbonate | <0.10 | mg·L−1 |
| 5 | Total dissolved solids (TDS) (TDS) | 13,300 | mg·L−1 |
| 6 | pH | 8.10 | - |
| 7 | Chroma | 700 | times |
| Catalyst Type | Molecular Weight | Purity Quotient |
|---|---|---|
| MnO2 | 86.94 | ≥85.0 |
| Ni2O3 | 165.39 | 70.0–75.0 |
| Al2O3 | 101.96 | ≥90.0 |
| TiO2 | 79.87 | ≥98.0 |
| Catalyst Type | Reaction Time (mg·L−1) | pH Value | Catalyst Dosage (g·L−1) | COD Value (mg·L−1) | |
|---|---|---|---|---|---|
| 1 | MnO2 | 200 | 5 | 3 | 807 |
| 2 | MnO2 | 300 | 8 | 6 | 682 |
| 3 | MnO2 | 400 | 10 | 9 | 465 |
| 4 | MnO2 | 500 | 12 | 12 | 387 |
| 5 | Al2O3 | 200 | 8 | 9 | 703 |
| 6 | Al2O3 | 300 | 5 | 12 | 763 |
| 7 | Al2O3 | 400 | 12 | 3 | 508 |
| 8 | Al2O3 | 500 | 10 | 6 | 467 |
| 9 | Ni2O3 | 200 | 10 | 12 | 680 |
| 10 | Ni2O3 | 300 | 12 | 9 | 647 |
| 11 | Ni2O3 | 400 | 8 | 6 | 708 |
| 12 | Ni2O3 | 500 | 5 | 3 | 743 |
| 13 | TiO2 | 200 | 12 | 6 | 735 |
| 14 | TiO2 | 300 | 10 | 3 | 689 |
| 15 | TiO2 | 400 | 8 | 12 | 723 |
| 16 | TiO2 | 500 | 5 | 9 | 730 |
| K1 | 2341 | 2925 | 3043 | 2747 | 10,437 |
| K2 | 2441 | 2781 | 2816 | 2592 | - |
| K3 | 2778 | 2404 | 2301 | 2545 | - |
| K4 | 2877 | 2327 | 2277 | 2553 | - |
| k1 | 585 | 731 | 761 | 687 | - |
| k2 | 610 | 695 | 704 | 648 | - |
| k3 | 695 | 601 | 575 | 636 | - |
| k4 | 719 | 582 | 569 | 638 | - |
| R | 134 | 130 | 192 | 51 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jing, X.; Cheng, S.; Men, C.; Zhu, H.; Luo, M.; Li, Z. Study on the Mechanism and Control Strategy of Advanced Treatment of Yeast Wastewater by Ozone Catalytic Oxidation. Water 2023, 15, 274. https://doi.org/10.3390/w15020274
Jing X, Cheng S, Men C, Zhu H, Luo M, Li Z. Study on the Mechanism and Control Strategy of Advanced Treatment of Yeast Wastewater by Ozone Catalytic Oxidation. Water. 2023; 15(2):274. https://doi.org/10.3390/w15020274
Chicago/Turabian StyleJing, Xianglong, Shikun Cheng, Cong Men, Huimin Zhu, Mei Luo, and Zifu Li. 2023. "Study on the Mechanism and Control Strategy of Advanced Treatment of Yeast Wastewater by Ozone Catalytic Oxidation" Water 15, no. 2: 274. https://doi.org/10.3390/w15020274





