Hydrogeochemistry and Strontium Isotopic Signatures of Mineral Waters from Furnas and Fogo Volcanoes (São Miguel, Azores)
Abstract
:1. Introduction
2. Geological Setting
2.1. Overview of the Geology of São Miguel Island
2.2. Fogo Volcano
2.3. Furnas Volcano
3. Hydrogeological Setting
4. Sampling and Analytical Techniques
5. Results and Discussion
5.1. Hydrogeochemistry
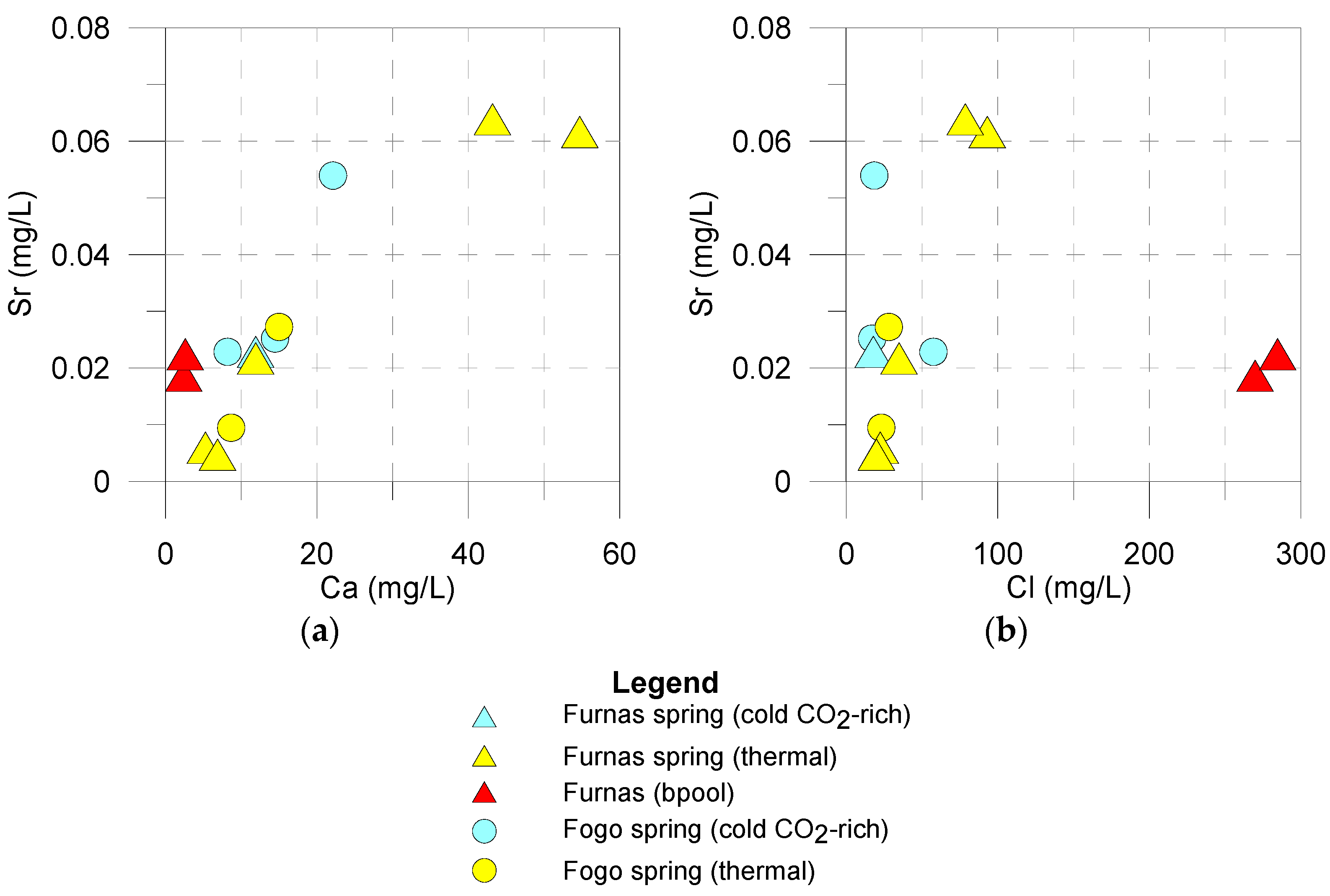
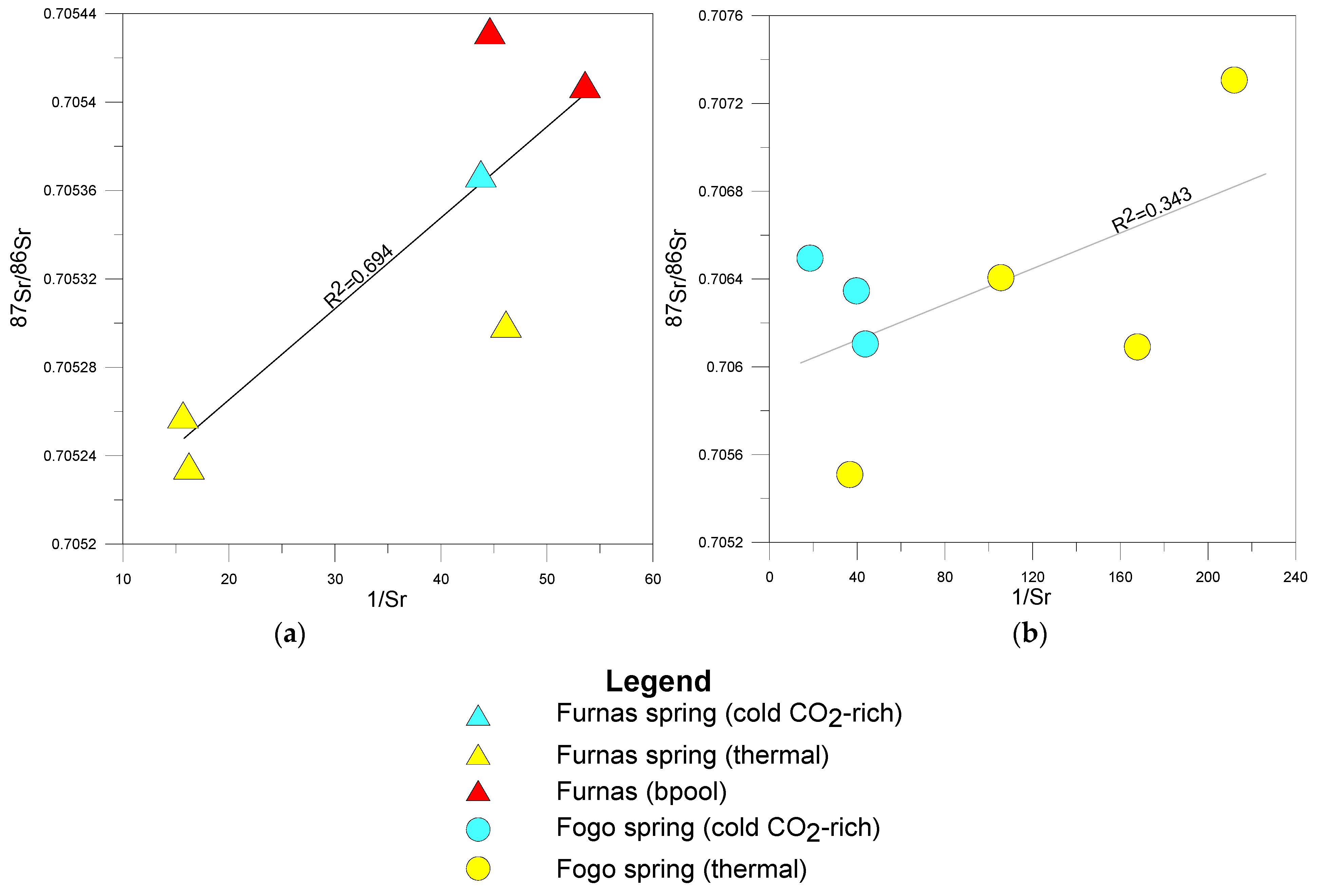
5.2. Strontium Content and 87Sr/86Sr
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Faure, G.; Mensing, T.M. Isotopes, Principles and Applications, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005; p. 897. [Google Scholar]
- Faure, G. Principles of Isotope Geology, 2nd ed.; John Wiley & Sons: New York, NY, USA, 1986. [Google Scholar]
- Ribeiro, S.; Azevedo, M.R.; Santos, J.F.; Medina, J.; Costa, A. Sr isotopic signatures of Portuguese bottled mineral waters and their relationships with the geological setting. Comun. Geol. 2014, 101, 29–37. [Google Scholar]
- Shand, P.; Darbyshire, D.P.F.; Love, A.J.; Edmunds, W.M. Sr isotopes in natural waters: Applications to source characterisation and water-rock interaction in contrasting landscapes. Appl. Geochem. 2009, 24, 574–586. [Google Scholar] [CrossRef]
- Marques, J.M.; Carreira, P.M.; Goff, F.; Eggenkamp HG, M.; da Silva, M.A. Input of 87Sr/86Sr ratios and Sr geochemical signatures to update knowledge on thermal and mineral waters flow paths in fractured rocks (N-Portugal). Appl. Geochem. 2012, 27, 1471–1481. [Google Scholar] [CrossRef]
- Capo, R.C.; Stewart, B.W.; Chadwick, O.A. Strontium isotopes as tracers of ecosystem processes: Theory and methods. Geoderma 1998, 82, 197–225. [Google Scholar] [CrossRef]
- Frei, K.M.; Frei, R. The geographic distribution of strontium isotopes in Danish surface waters—A base for provenance studies in archaeology, hydrology and agriculture. Appl. Geochem. 2011, 26, 326–340. [Google Scholar] [CrossRef]
- Åberg, G.; Jacks, G.; Hamilton, J. Weathering rates and 87Sr/86Sr ratios: An isotopic approach. J. Hydrol. 1989, 109, 65–78. [Google Scholar] [CrossRef]
- Négrel, P.; Fouillac, C.; Brach, M. A strontium isotopic study of mineral and surface waters from the Cézallier (Massif Central, France): Implications for mixing processes in areas of disseminated emergences of mineral waters. Chem. Geol. 1997, 135, 89–101. [Google Scholar] [CrossRef]
- Négrel, P.; Lemiere, B.; De Grammont, H.M.; Billaud, P.; Sengupta, B. Hydrogeochemical processes, mixing and isotope tracing in hard rock aquifers and surface waters from the Subarnarekha River Basin, (east Singhbhum District, Jharkhand State, India). Hydrogeol. J. 2007, 15, 1535–1552. [Google Scholar] [CrossRef]
- Négrel, P.; Petelet-Giraud, E. Strontium isotopes as tracers of groundwater-induced floods: The Somme case study (France). J. Hydrol. 2005, 305, 99–119. [Google Scholar] [CrossRef]
- Wu, C.; Wu, X.; Mu, W.; Zhu, G. Using isotopes (H, O, and Sr) and major ions to identify hydrogeochemical characteristics of groundwater in the Hongjiannao Lake Basin, Northwest China. Water 2020, 12, 1467. [Google Scholar] [CrossRef]
- Bénard, B.; Famin, V.; Agrinier, P.; Aunay, B.; Lebeau, G.; Sanjuan, B.; Vimeux, F.; Bardoux, G.; Dezayes, C. Origin and fate of hydrothermal fluids at Piton des Neiges volcano (Réunion Island): A geochemical and isotopic (O, H, C, Sr, Li, Cl) study of thermal springs. J. Volcanol. Geotherm. Res. 2020, 392, 106682. [Google Scholar] [CrossRef]
- Cruz, J.; França, Z. Hydrogeochemistry of thermal and mineral water springs of the Azores archipelago (Portugal). J. Volcanol. Geotherm. Res. 2006, 151, 382–398. [Google Scholar] [CrossRef]
- Cruz, J.V.; Freire, P.; Costa, A. Mineral waters characterization in the Azores archipelago (Portugal). J. Volcanol. Geotherm. Res. 2010, 190, 353–364. [Google Scholar] [CrossRef]
- Coutinho, R.; Fontiela, J.; Freire, P.; Cruz, J.V. Hydrogeology of São Miguel Island, Azores: A review. In Volcanic Geology of São Miguel Island (Azores Archipelago); Gaspar, J.L., Guest, J.E., Duncan, A.M., Barriga, F.J.A.S., Chester, D.K., Eds.; Geological Society of London, Memoirs: London, UK, 2015; Volume 44, pp. 289–296. [Google Scholar]
- Freire, P.; Andrade, C.; Viveiros, F.; Silva, C.; Coutinho, R.; Cruz, J.V. Mineral water occurrence and geochemistry in the Azores volcanic archipelago (Portugal): Insight from an extended database on water chemistry. Environ. Earth Sci. 2015, 73, 2749–2762. [Google Scholar] [CrossRef]
- Caliro, S.; Viveiros, F.; Chiodini, G.; Ferreira, T. Gas geochemistry of hydrothermal fluids of the S. Miguel and Terceira Islands, Azores. Geochim. Cosmochim. Acta 2015, 168, 43–57. [Google Scholar] [CrossRef]
- Viveiros, F.; Cardellini, C.; Ferreira, T.; Caliro, S.; Chiodini, G.; Silva, C. Soil CO2 emissions at Furnas volcano, São Miguel Island, Azores archipelago: Volcano monitoring perspectives, geomorphologic studies, and land use planning application. J. Geophys. Res. Solid Earth 2010, 115, B12208. [Google Scholar] [CrossRef]
- Viveiros, F.; Chiodini, G.; Cardellini, C.; Caliro, S.; Zanon, V.; Silva, C.; Rizzo, A.L.; Hipólito, A.; Moreno, L. Deep CO2 emitted at Furnas do Enxofre geothermal area (Terceira Island, Azores archipelago). An approach for determining CO2 sources and total emissions using carbon isotopic data. J. Volcanol. Geotherm. Res. 2020, 401, 106968. [Google Scholar] [CrossRef]
- Freire, P.; Andrade, C.; Coutinho, R.; Cruz, J.V. Fluvial geochemistry in São Miguel Island (Azores, Portugal): Source and fluxes of inorganic solutes in an active volcanic environment. Sci. Total Environ. 2013, 454, 154–169. [Google Scholar] [CrossRef]
- Woitischek, J.; Dietzel, M.; Inguaggiato, C.; Böttcher, M.E.; Leis, A.; Cruz, J.V.; Gehre, M. Characterization and origin of hydrothermal waters at São Miguel (Azores) inferred by chemical and isotopic composition. J. Volcanol. Geotherm. Res. 2017, 346, 104–117. [Google Scholar] [CrossRef]
- Cruz, J.; Coutinho, R.M.; Carvalho, M.; Oskarsson, N.; Gislason, S.R. Chemistry of waters from Furnas volcano, São Miguel, Azores: Fluxes of volcanic carbon dioxide and leached material. J. Volcanol. Geotherm. Res. 1999, 92, 151–167. [Google Scholar] [CrossRef]
- Morell, I.; Pulido-Bosch, A.; Daniele, L.; Cruz, J.V. Chemical and isotopic assessment in volcanic thermal waters: Cases of Ischia (Italy) and São Miguel (Azores, Portugal). Hydrol. Process. 2008, 22, 4386–4399. [Google Scholar] [CrossRef]
- Louvat, P.; Allègre, C.J. Riverine erosion rates on São Miguel volcanic island, Azores archipelago. Chem. Geol. 1998, 148, 177–200. [Google Scholar] [CrossRef]
- Cruz, J.V.; Andrade, C. Groundwater salinization in Graciosa and Pico islands (Azores archipelago, Portugal): Processes and impacts. J. Hydrol. Reg. Stud. 2017, 12, 69–87. [Google Scholar] [CrossRef]
- Pacheco, J.M.; Ferreira, T.; Queiroz, G.; Wallenstein, N.; Coutinho, R.; Cruz, J.V.; Pimentel, A.; Silva, R.; Gaspar, J.L.; Goulart, C. Notas sobre a Geologia do Arquipélago dos Açores. In Geologia de Portugal; Dias, R., Araújo, A., Terrinha, P., Kullberg, J.C., Eds.; Escolar Editora: Lisboa, Portugal, 2013; Volume 2, pp. 595–690. [Google Scholar]
- Gaspar, J.L.; Guest, J.E.; Duncan, A.; Chester, D.; Barriga, F. Volcanic Geology of São Miguel Island (Azores Archipelago): Introduction. In Volcanic Geology of São Miguel Island (Azores Archipelago); Gaspar, J.L., Guest, J.E., Duncan, A.M., Barriga, F.J.A.S., Chester, D.K., Eds.; Geological Society of London, Memoirs: London, UK, 2015; Volume 44, pp. 1–3. [Google Scholar]
- Hawkesworth, C.J.; Norry, M.J.; Roddick, J.C.; Vollmer, R. 143Nd/144Nd and 87Sr/86Sr ratios from the Azores and their significance in LIL-element enriched mantle. Nature 1979, 280, 28–31. [Google Scholar] [CrossRef]
- Beier, C.; Stracke, A.; Haase, K.M. The peculiar geochemical signatures of São Miguel (Azores) lavas: Metasomatised or recycled mantle sources? Earth Planet. Sci. Lett. 2007, 259, 186–199. [Google Scholar] [CrossRef]
- Zanon, V. Conditions for mafic magma storage beneath fissure zones at oceanic islands. The case of São Miguel Island (Azores archipelago). In Chemical, Physical and Temporal Evolution of Magmatic Systems; Geological Society of London: London, UK, 2015; Volume 422, pp. 85–104. [Google Scholar]
- Widom, E.; Carlson, R.W.; Gill, J.B.; Schmincke, H.U. Th-Sr-Nb-Pb isotope and trace element evidence for the origin of the São Miguel, Azores, enriched mantle source. Chem. Geol. 1997, 140, 49–68. [Google Scholar] [CrossRef]
- Jeffery, A.J.; Gertisser, R.; O’Driscoll, B.; Pacheco, J.M.; Whitley, S.; Pimentel, A.; Self, S. Temporal evolution of a post-caldera, mildly peralkaline magmatic system: Furnas volcano, São Miguel, Azores. Contrib. Mineral. Petrol. 2016, 171, 42. [Google Scholar] [CrossRef] [Green Version]
- Elliott, T.; Blichert-Toft, J.; Heumann, A.; Koestsier, G.; Forjaz, V. The origin of enriched mantle beneath São Miguel, Azores. Geochim. Cosmochim. Acta 2007, 71, 219–240. [Google Scholar] [CrossRef]
- Turner, S.; Hawkesworth, C.; Rogers, N.; King, P. U-Th isotope disequilibria and ocean island basalt generation in the Azores. Chem. Geol. 1997, 139, 145–164. [Google Scholar] [CrossRef]
- Beier, C.; Haase, K.M.; Hansteen, T.H. Magma evolution of the Sete Cidades Volcano, São Miguel, Azores. J. Petrol. 2006, 47, 1375–1411. [Google Scholar] [CrossRef] [Green Version]
- Muecke, G.K.; Ade-Hall, J.M.; Aumento, F.; MacDonald, A.; Reynolds, P.H.; Hyndman, R.D.; Quintino, J.; Opdyke, N.; Lowrie, W. Deep drilling in an activite geothermal area in the Azores. Nature 1974, 252, 281–285. [Google Scholar] [CrossRef]
- Wallenstein, N.; Duncan, A.; Guest, J.E.; Almeida, M.H. Eruptive History of Fogo Volcano, São Miguel, Azores. In Volcanic Geology of São Miguel Island (Azores Archipelago); Gaspar, J.L., Guest, J.E., Duncan, A.M., Barriga, F.J.A.S., Chester, D.K., Eds.; Geological Society of London, Memoirs: London, UK, 2015; Volume 44, pp. 105–123. [Google Scholar]
- Wallenstein, N. Estudo da História Recente e do Comportamento Eruptivo do Vulcão do Fogo (S.; Miguel, Açores). Ph.D. Thesis, Universidade dos Açores, Ponta Delgada, Portugal, 1999; p. 266. (In Portuguese). [Google Scholar]
- Carmo, R.; Madeira, J.; Ferreira, T.; Queiroz, G.; Hipólito, A. Volcano-Tectonic Structures of São Miguel Island, Azores. In Volcanic Geology of São Miguel Island (Azores Archipelago); Gaspar, J.L., Guest, J.E., Duncan, A.M., Barriga, F.J.A.S., Chester, D.K., Eds.; Geological Society of London, Memoirs: London, UK, 2015; Volume 44, pp. 65–86. [Google Scholar]
- Santos, J.F.; Acciaioli, M.H.; França, Z.; Nunes, J.C.; Pinto, M.S.; Forjaz, V.H. Contribuição para o estudo das Variações Litogeoquímicas do Vulcão do Fogo (ilha de S.; Miguel, Açores). In Proceedings of theActas da XIV Semana de Geoquımica e do VIII Congresso de Geoquımica de Paıses de Lıngua Portuguesa, Aveiro, Universidade de Aveiro, Aveiro, Portugal, 11–16 July 2005; Volume 1, pp. 141–144. [Google Scholar]
- Snyder, D.C.; Widom, E.; Pietruszka, A.J.; Carlson, R.W. The role of open-system processes in the development of silicic magma chambers: A chemical and isotopic investigation of the Fogo A trachyte deposit, São Miguel, Azores. J. Petrol. 2004, 45, 723–738. [Google Scholar] [CrossRef] [Green Version]
- Pereira, M.L.; Matias, D.; Viveiros, F.; Moreno, L.; Silva, C.; Zanon, V.; Uchôa, J. The contribution of hydrothermal mineral alteration analysis and gas geothermometry for understanding high-temperatura geothermal fields. The case of Ribeira Grande geothermal field, Azores. Geothermics 2022, 105, 102519. [Google Scholar] [CrossRef]
- Guest, J.E.; Gaspar, J.L.; Cole, P.D.; Queiroz, G.; Duncan, A.M.; Wallenstein, N.; Ferreira, T.; Pacheco, J.M. Volcanic geology of Furnas volcano, São Miguel, Azores. J. Volcanol. Geotherm. Res. 1999, 92, 1–29. [Google Scholar] [CrossRef]
- Guest, J.E.; Pacheco, J.M.; Cole, P.D.; Duncan, A.M.; Wallenstein, N.; Queiroz, G.; Gaspar, J.L.; Ferreira, T. The volcanic History of Furnas Volcano, São Miguel, Azores. In Volcanic Geology of São Miguel Island (Azores Archipelago); Gaspar, J.L., Guest, J.E., Duncan, A.M., Barriga, F.J.A.S., Chester, D.K., Eds.; Geological Society of London, Memoirs: London, UK, 2015; Volume 44, pp. 125–134. [Google Scholar]
- Moore, R.B. Volcanic geology and eruption frequency, São Miguel, Azores. Bull. Volcanol. 1990, 52, 602–614. [Google Scholar] [CrossRef]
- Cole, P.D.; Queiroz, G.; Wallenstein, N.; Gaspar, J.L.; Guest, J.E. An historic subplinianrphreatomagmatic eruption: The 1630 AD eruption of Furnas volcano, Azores. J. Volcanol. Geotherm. Res. 1995, 69, 117–135. [Google Scholar] [CrossRef]
- Andrade, C.; Viveiros, F.; Cruz, J.V.; Coutinho, R.; Silva, C. Estimation of the CO2 flux from Furnas volcanic Lake (São Miguel, Azores). J. Geotherm. Res. 2016, 315, 51–64. [Google Scholar] [CrossRef]
- Cruz, J.V.; Amaral, C.S. Major ion chemistry of groundwater from perched-water bodies of the Azores (Portugal) volcanic archipelago. Appl. Geochem. 2004, 19, 445–459. [Google Scholar] [CrossRef]
- Cruz, J.V.; Andrade, C. Natural background groundwater composition in the Azores archipelago (Portugal): A hydrogeochemical study and threshold value determination. Sci. Total Environ. 2015, 520, 127–135. [Google Scholar] [CrossRef]
- Cruz, J.V.; Silva, M.O.; Dias, M.I.; Prudêncio, M.I. Groundwater composition and pollution due to agricultural pratices at Sete Cidades volcano (Azores, Portugal). Appl. Geochem. 2013, 29, 162–173. [Google Scholar] [CrossRef]
- Cruz, J.V. Groundwater and volcanoes: Examples from the Azores archipelago. Environ. Geol. 2003, 44, 343–355. [Google Scholar] [CrossRef]
- Cruz, J.V.; Silva, M.O. Hydrogeologic framework of the Pico Island (Azores, Portugal). Hydrogeol. J. 2001, 9, 177–189. [Google Scholar] [CrossRef]
- Cruz, J.V.; Freire, P.; Costa, A.; Fontiela, J.; Cabral, L.; Coutinho, R.M. Hydrogeochemical Characterization of Mineral Waters in São Miguel Island, Azores. In Volcanic Geology of São Miguel Island (Azores Archipelago); Gaspar, J.L., Guest, J.E., Duncan, A.M., Barriga, F.J.A.S., Chester, D.K., Eds.; Geological Society of London, Memoirs: London, UK, 2015; Volume 44, pp. 257–269. [Google Scholar]
- Thornthwaite, C.W. An approach toward a national classification of climate. Geogr. Rev. 1948, 38, 55–94. [Google Scholar] [CrossRef]
- Ricardo, R.P.; Madeira, M.A.V.; Medina, J.M.B.; Marques, M.M.; Sanches Furtado, A.F.A. Esboço pedológico da ilha de S. Miguel (Açores). An. Inst. Super. Agron. 1977, 37, 275–385. [Google Scholar]
- Cruz, J.V.; Pacheco, D.; Cymbron, R.; Mendes, S. Monitoring of the groundwater chemical status in the Azores archipelago (Portugal) in the context of the EU water framework directive. Sci. Total Environ. 2010, 61, 173–186. [Google Scholar] [CrossRef]
- DROTRH-INAG. Plano Regional da Água-Relatório Técnico; DROTRH-INAG: Ponta Delgada, Portugal, 2001. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwju9ZDKuK_8AhVGtVYBHflEDVkQFnoECBkQAQ&url=https%3A%2F%2Fservicos-sraa.azores.gov.pt%2Fgrastore%2FDRA%2FQSIGA%2FPP_DocumentoApoio.pdf&usg=AOvVaw22BHEM-kkz1K7wwgvAS4ci (accessed on 14 November 2022).
- Cruz, J.V. Ensaio Sobre a Água Subterrânea nos Açores. História, Ocorrência e Eualidade. SRA, Ponta Delgada; 2004. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwjmn5Spt6_8AhU8s1YBHctVAlgQFnoECAgQAQ&url=http%3A%2F%2Fwww.ivar.azores.gov.pt%2Fpublicacoes%2FPaginas%2Fcms_908_CRUZ-J-V-2004-Ensaio-sobre-a-agua-subterranea-nos-Acores-Historia-ocorrencia-e-qualidade-Ed-Secretaria-Regional-do.aspx&usg=AOvVaw1zfHmlRlK5w206nxIKy1q4 (accessed on 14 November 2022).
- Krásný, J. Classification of transmissivity magnitude and variation. Ground Water 1993, 31, 230–236. [Google Scholar] [CrossRef]
- APHA-AWWA-WPCF. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1985. [Google Scholar]
- Clark, I.D.; Fritz, P. Environmental Isotopes in Hydrology; Lewis Publishers: New York, NY, USA, 1997; p. 328. [Google Scholar]
- IAEA. Sampling Procedures for Isotope Hydrology; Water Resources Programe: Vienna, Austria, 2017. [Google Scholar]
- Evans, W.; Sorey, M.; Cook, A.; Kennedy, B.; Shuster, D.; Colvard, E.; White, L.; Huebner, M. Tracing and quantifying magmatic carbon discharge in cold groundwaters: Lessons learned from Mammoth Mountain, USA. J. Volcanol. Geotherm. Res. 2002, 114, 291–312. [Google Scholar] [CrossRef]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water, 3rd ed.; Water-Supply Paper 2254; U.S. Geological Survey: Reston, VA, USA; p. 263.
- Bestland, E.; George, A.; Green, G.; Olifent, V.; Mackay, D.; Whallen, M. Groundwater dependent pools in seasonal and permanent streams in the Clare Valley of South Australia. J. Hydrol. Reg. Stud. 2017, 9, 216–235. [Google Scholar] [CrossRef] [Green Version]
- Kelly, W.R.; Panno, S.V.; Hackley, K.C. The Sources, Distribution and Trends of Chloride in the Waters of Illinois; Bulletin Illinois State Water Survey B-74; Illinois State Water Survey: Champaign, IL, USA, 2012; pp. 1–59. [Google Scholar]
- Berner, E.K.; Berner, R.A. Global Environment: Water, Air, and Geochemical Cycles, 2nd ed.; Princeton University Press: Oxfordshire, UK; Princeton, NJ, USA, 2012; p. xvi + 444. [Google Scholar]
- Custódio, E.; Llamas, M.R. Hidrología Subterrânea, 2nd ed.; Tomo, I., Ed.; Ediciónes Ómega, S.A.: Barcelona, Spain, 1983; p. 1157. [Google Scholar]
- IAEA, Water Resource Programme. Measurements of the Isotopic Composition of Meteoric Water of São Miguel, Azores from 1962 till 2009; Water Resources Programe: Vienna, Austria, 2014. [Google Scholar]
- Cartigny, P.; Harris, J.W.; Javoy, M. Diamond genesis, mantle fractionations and mantle nitrogen content: A study of δ13C–N concentrations in diamonds. Earth Planet. Sci. Lett. 2001, 185, 85–98. [Google Scholar] [CrossRef]
- Clark, T. Groundwater Geochemistry and Isotopes; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: London, UK, 2015. [Google Scholar]
- O’Sullivan, P.E. Palaeolimnology. In The Lakes Handbook, Limnology and Limnetic Ecology; O’Sullivan, P.E., Reynolds, C.S., Eds.; Blackwell: Oxford, UK, 2004; Volume 1, pp. 609–666. [Google Scholar]
- Ferreira, T.; Oskarsson, N. Chemistry and isotopic composition of fumarole discharges of Furnas caldera. J. Volcanol. Geotherm. Res. 1999, 92, 169–179. [Google Scholar] [CrossRef]
- Andrade, C.; Viveiros, F.; Cruz, J.V.; Coutinho, R. Global carbon dioxide output of volcanic lakes in the Azores archipelago, Portugal. J. Geochem. Explor. 2021, 229, 106835. [Google Scholar] [CrossRef]
- Kendall, C.; Doctor, D. Stable Isotopic Applications in Hydrological Studies. In Surface and Groundwater, Weathering, and Soils; Drever, J.I., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 9, pp. 319–364. [Google Scholar]
- Negrel, P.; Guerrot, C.; Millot, R. Chemical and strontium isotope characterization of rainwater in France: Influence of sources and hydrogeochemical implications. Isot. Environ. Health Stud. 2007, 3, 179–196. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Ohba, T.; Fujimaki, H. Sr isotope diversity of hot spring and volcanic lake waters from Zao volcano, Japan. J. Volcanol. Geotherm. Res. 2007, 166, 7–16. [Google Scholar] [CrossRef]
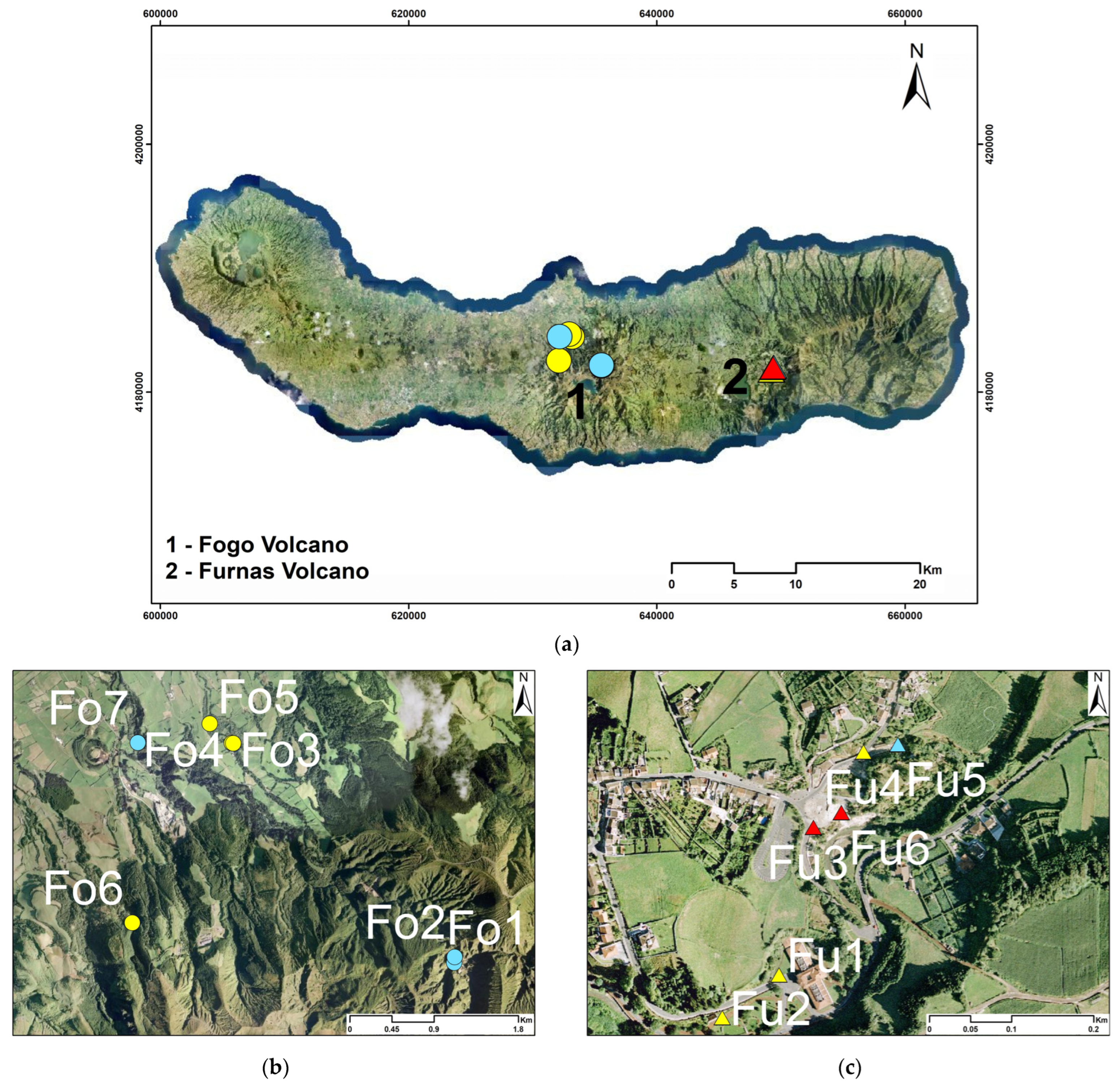
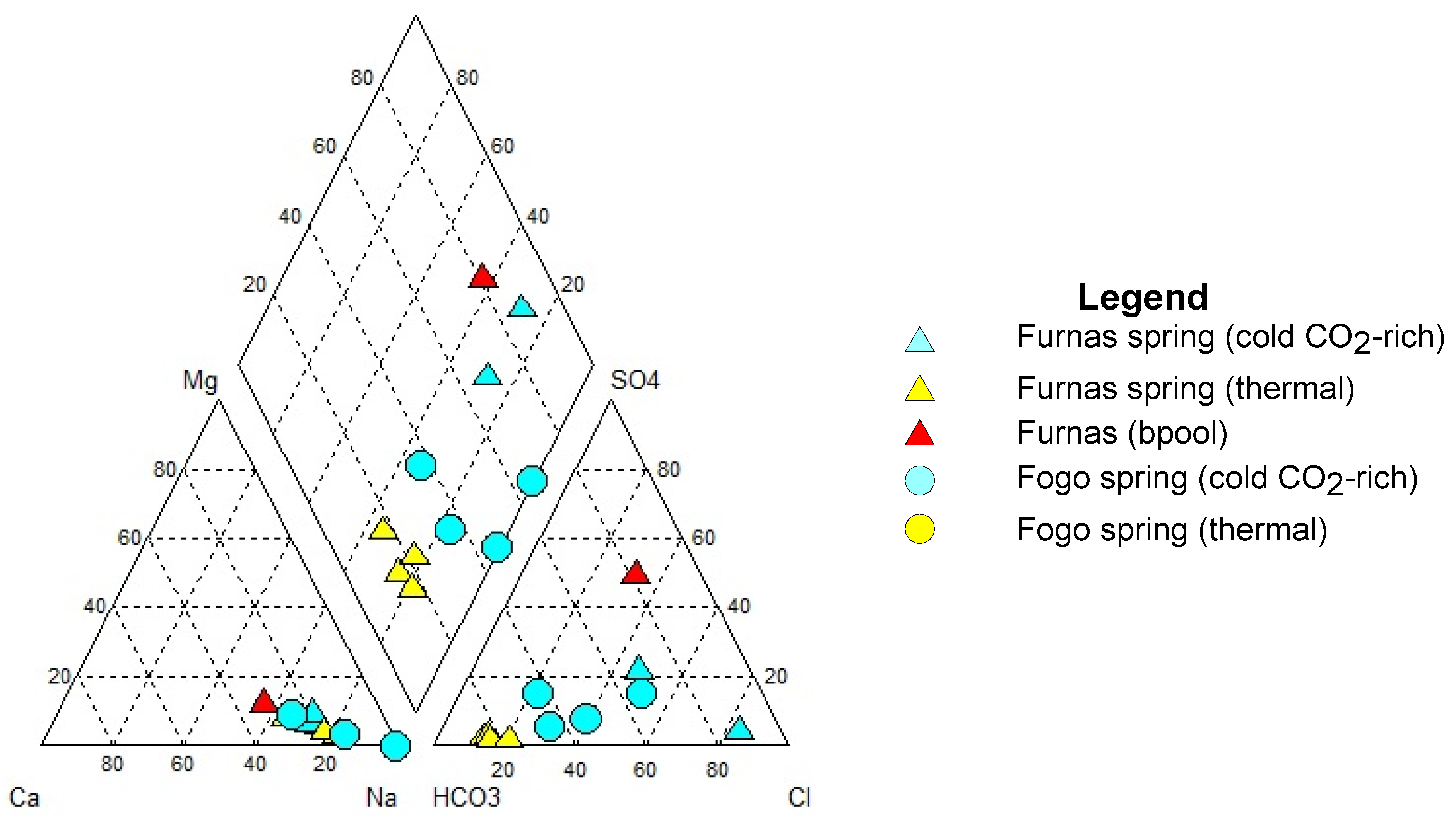
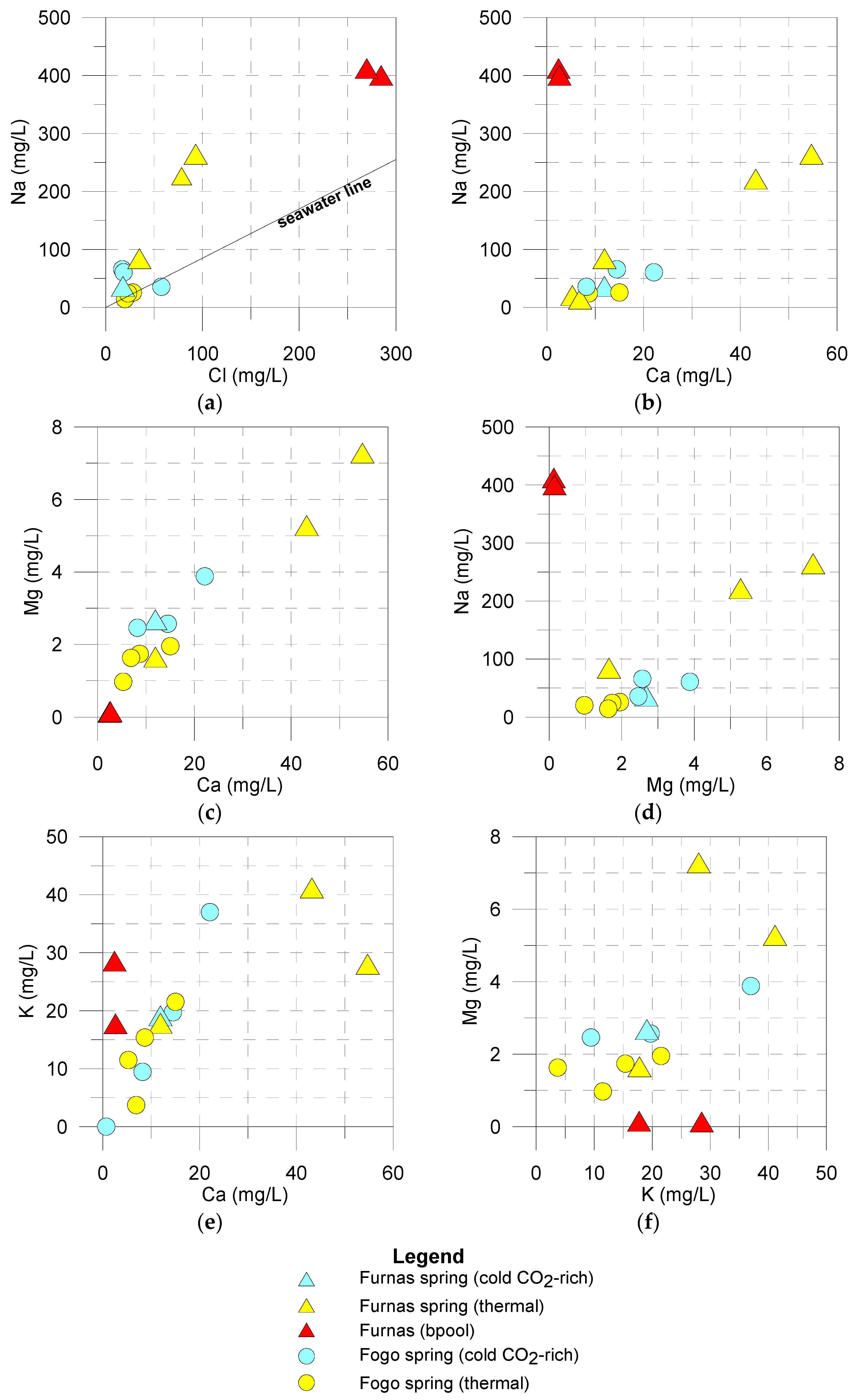
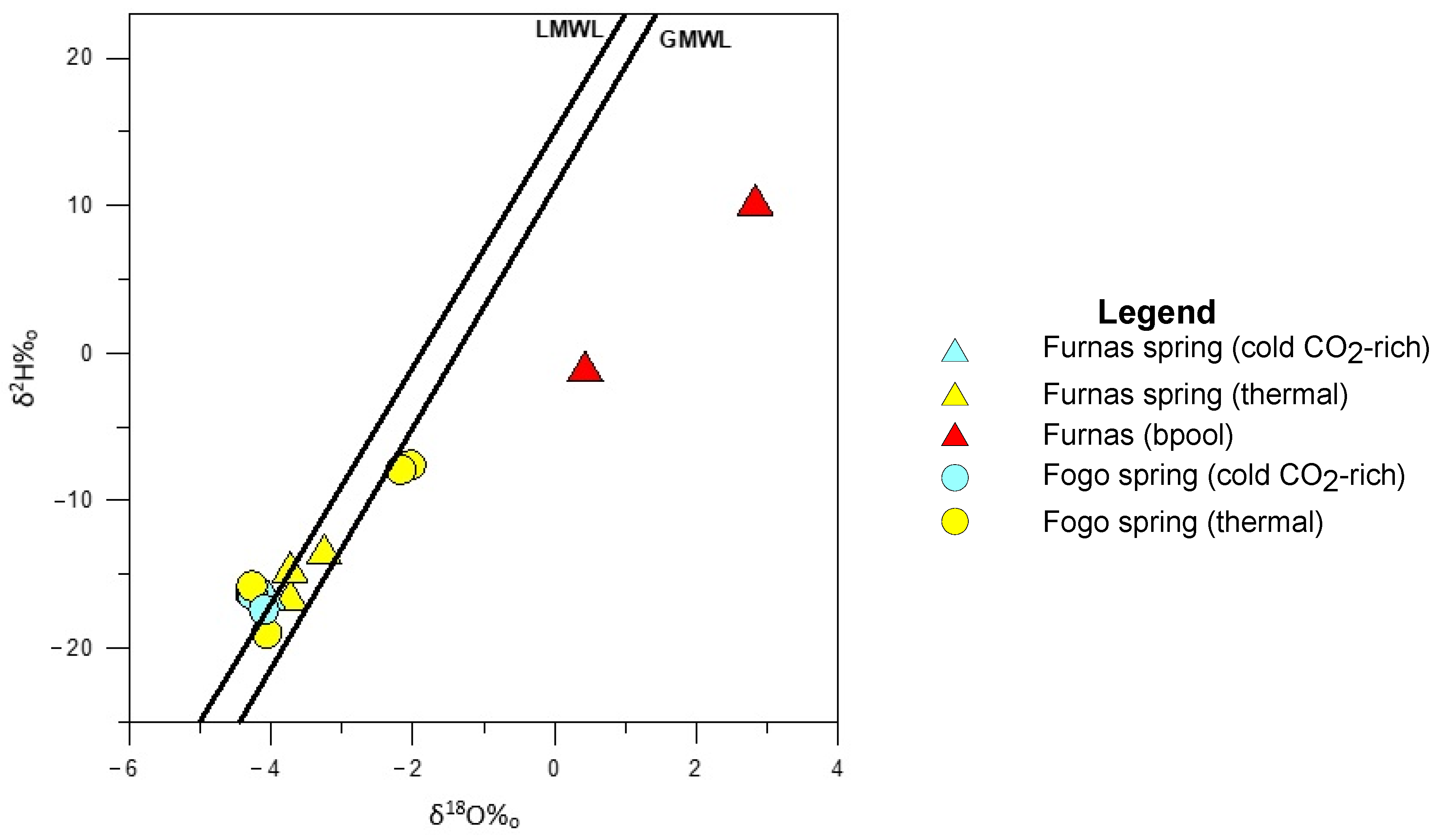

| Location | Ref. | Name | Type | Altitude | T | pH | EC | CO2 | Na | K | Ca | Mg | Cl | SO4 | HCO3 | SiO2 | Sr | 87Sr/86Sr | δ18O | δ2H | δ13C | δ34S |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| m | °C | µS cm−1 | mg L−1 | |||||||||||||||||||
| Furnas | Fu1 | Quenturas | Thermal spr | 199 | 57.6 | 6.67 | 1328 | 212.60 | 221.37 | 41.16 | 43.19 | 5.28 | 78.46 | 12.63 | 736.88 | 213.30 | 0.0638 | 0.705258 | −3.73 | −16.4 | −7.04 | 11.1 |
| Fu2 | Torno | Thermal spr | 199 | 39.6 | 6.59 | 1412 | 234.00 | 264.19 | 28.01 | 54.73 | 7.28 | 93.01 | 10.28 | 608.78 | 148.81 | 0.0616 | 0.705235 | −3.25 | −13.3 | −5.96 | 12.76 | |
| Fu3 | Caldeira Grande | bpool | 203 | 96.8 | 7.96 | 2110 | 14.90 | 400.03 | 17.75 | 2.62 | 0.15 | 284.71 | 74.93 | 668.56 | 344.93 | 0.0224 | 0.705432 | 0.43 | −0.9 | −4.36 | 6.03 | |
| Fu4 | Caldeirão | Thermal spr | 202 | 75.6 | 6.69 | 476 | 127.60 | 83.94 | 17.81 | 11.91 | 1.65 | 34.79 | 8.74 | 128.10 | 160.47 | 0.0217 | 0.705325 | −3.73 | −14.5 | −4.54 | 1.77 | |
| Fu5 | Azeda | Cold spr | 201 | 16.6 | 4.98 | 308 | 936.30 | 36.19 | 19.10 | 11.91 | 2.68 | 17.75 | 16.62 | 88.45 | 94.45 | 0.0228 | 0.705368 | −4.04 | −16.4 | −5.76 | 3.28 | |
| Fu6 | Caldeira Asmodeu | bpool | 199 | 92.3 | 7.91 | 2370 | 46.40 | 411.98 | 28.53 | 2.43 | 0.13 | 269.80 | 111.43 | 311.10 | 338.59 | 0.0187 | 0.705408 | 2.83 | 10.4 | −3.00 | −4.02 | |
| Fogo | Fo1 | Lombadas | Cold spr | 578 | 17.8 | 5.36 | 326 | 803.70 | 65.73 | 19.73 | 14.48 | 2.57 | 17.04 | 4.23 | 192.76 | 100.99 | 0.0252 | 0.706346 | −4.29 | −16.3 | −5.4 | 4.85 |
| Fo2 | Lombadas Furo II | Cold spr | 578 | 18.6 | 5.69 | 564 | 881.00 | 60.57 | 37.00 | 22.13 | 3.88 | 18.46 | 4.71 | 184.22 | 103.39 | 0.0539 | 0.706495 | −4.26 | −16.4 | −6.64 | 10.48 | |
| Fo3 | Cald. Pequena—CRG | Thermal spr | 263 | 53.3 | 2.26 | 683 | 259.40 | 25.69 | 21.52 | 15.01 | 1.95 | 28.05 | 92.22 | - | 196.34 | 0.0272 | 0.705509 | −2.03 | −7.6 | −6.82 | 0.73 | |
| Fo4 | Cald. Grande—CRG | Thermal spr | 263 | 48.0 | 3.15 | 624 | 177.30 | 24.17 | 15.37 | 8.68 | 1.74 | 23.08 | 67.24 | - | 112.71 | 0.0095 | 0.706406 | −2.17 | −7.9 | −5.7 | 0.13 | |
| Fo5 | Pocinha | Thermal spr | 252 | 29.3 | 5.05 | 220 | 362.80 | 20.31 | 11.49 | 5.28 | 0.97 | 22.37 | 13.93 | 25.62 | 84.92 | 0.0060 | 0.706091 | −4.06 | −19 | −6.46 | 0.87 | |
| Fo6 | Caldeira Velha Spr. | Themal spr | 417 | 31.6 | 5.46 | 166 | 222.10 | 14.25 | 3.72 | 6.89 | 1.63 | 19.88 | 41.31 | 19.52 | 41.17 | 0.0047 | 0.707307 | −4.27 | −15.8 | −7.18 | 6.63 | |
| Fo7 | Magarça | Cold spr | 194 | 18.8 | 5.01 | 292 | 398.60 | 35.55 | 9.45 | 8.21 | 2.46 | 57.51 | 3.94 | 14.03 | 68.05 | 0.0229 | 0.706104 | −4.1 | −17.4 | −5.9 | 11.36 | |
| ---- | SW | Seawater | spr | - | - | - | - | - | - | - | - | - | - | - | - | 7.8390 | 0.709199 | 0.49 | 5.9 | −9.71 | 21.37 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, L.; Cruz, J.V.; Viveiros, F.; Durães, N.; Coutinho, R.; Andrade, C.; Santos, J.F.; Acciaioli, M.H. Hydrogeochemistry and Strontium Isotopic Signatures of Mineral Waters from Furnas and Fogo Volcanoes (São Miguel, Azores). Water 2023, 15, 245. https://doi.org/10.3390/w15020245
Ferreira L, Cruz JV, Viveiros F, Durães N, Coutinho R, Andrade C, Santos JF, Acciaioli MH. Hydrogeochemistry and Strontium Isotopic Signatures of Mineral Waters from Furnas and Fogo Volcanoes (São Miguel, Azores). Water. 2023; 15(2):245. https://doi.org/10.3390/w15020245
Chicago/Turabian StyleFerreira, Letícia, José Virgílio Cruz, Fatima Viveiros, Nuno Durães, Rui Coutinho, César Andrade, José Francisco Santos, and Maria Helena Acciaioli. 2023. "Hydrogeochemistry and Strontium Isotopic Signatures of Mineral Waters from Furnas and Fogo Volcanoes (São Miguel, Azores)" Water 15, no. 2: 245. https://doi.org/10.3390/w15020245
APA StyleFerreira, L., Cruz, J. V., Viveiros, F., Durães, N., Coutinho, R., Andrade, C., Santos, J. F., & Acciaioli, M. H. (2023). Hydrogeochemistry and Strontium Isotopic Signatures of Mineral Waters from Furnas and Fogo Volcanoes (São Miguel, Azores). Water, 15(2), 245. https://doi.org/10.3390/w15020245











