Equation Chapter 1 Section 1 Techno-Economic Analysis for the Selection of Cost-Effective Treatment for Algae Removal in Drinking Water Treatment Plants
Abstract
:1. Introduction
2. Data Sources and Methods
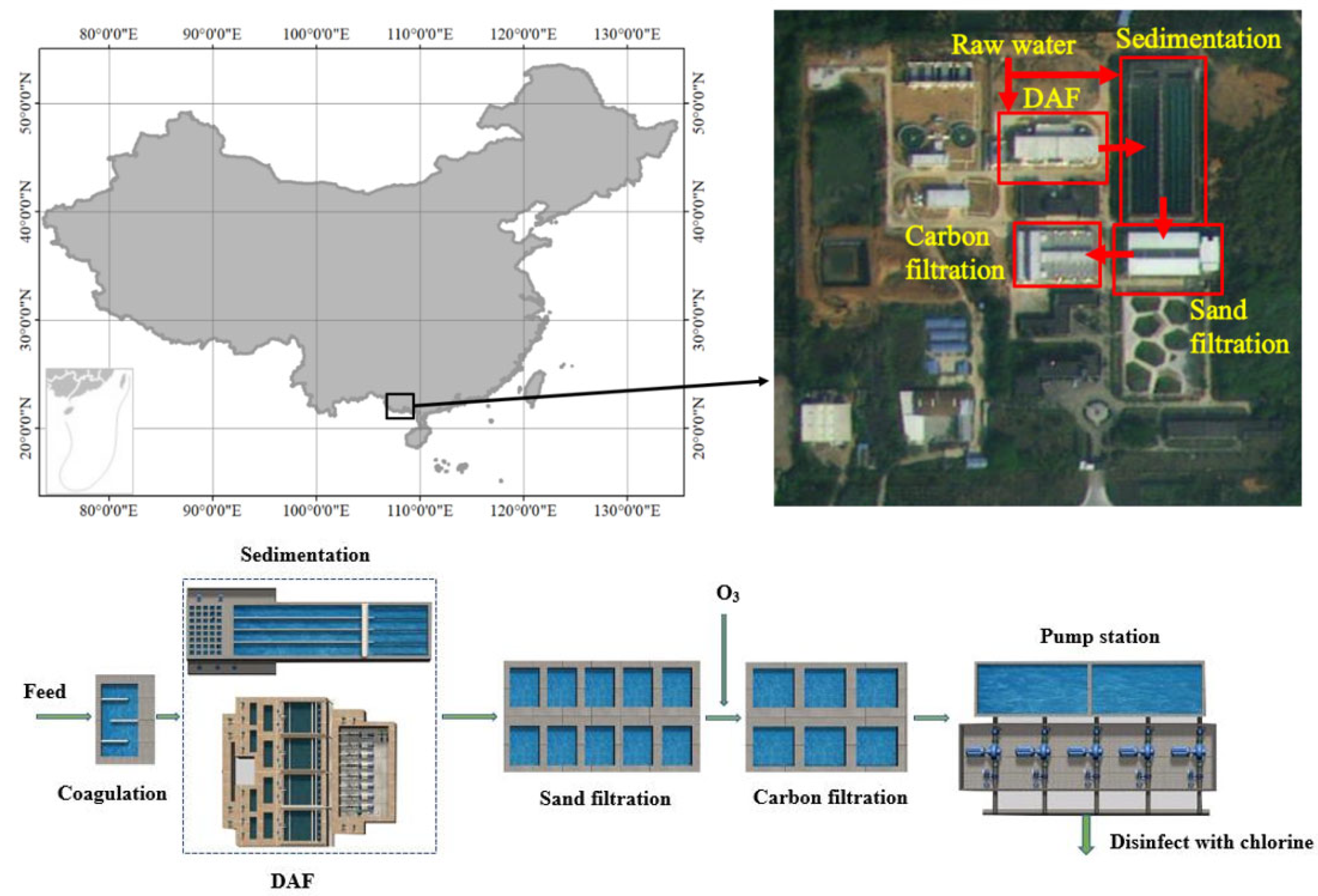
2.1. Treatment Processes in Beijiao DWTP
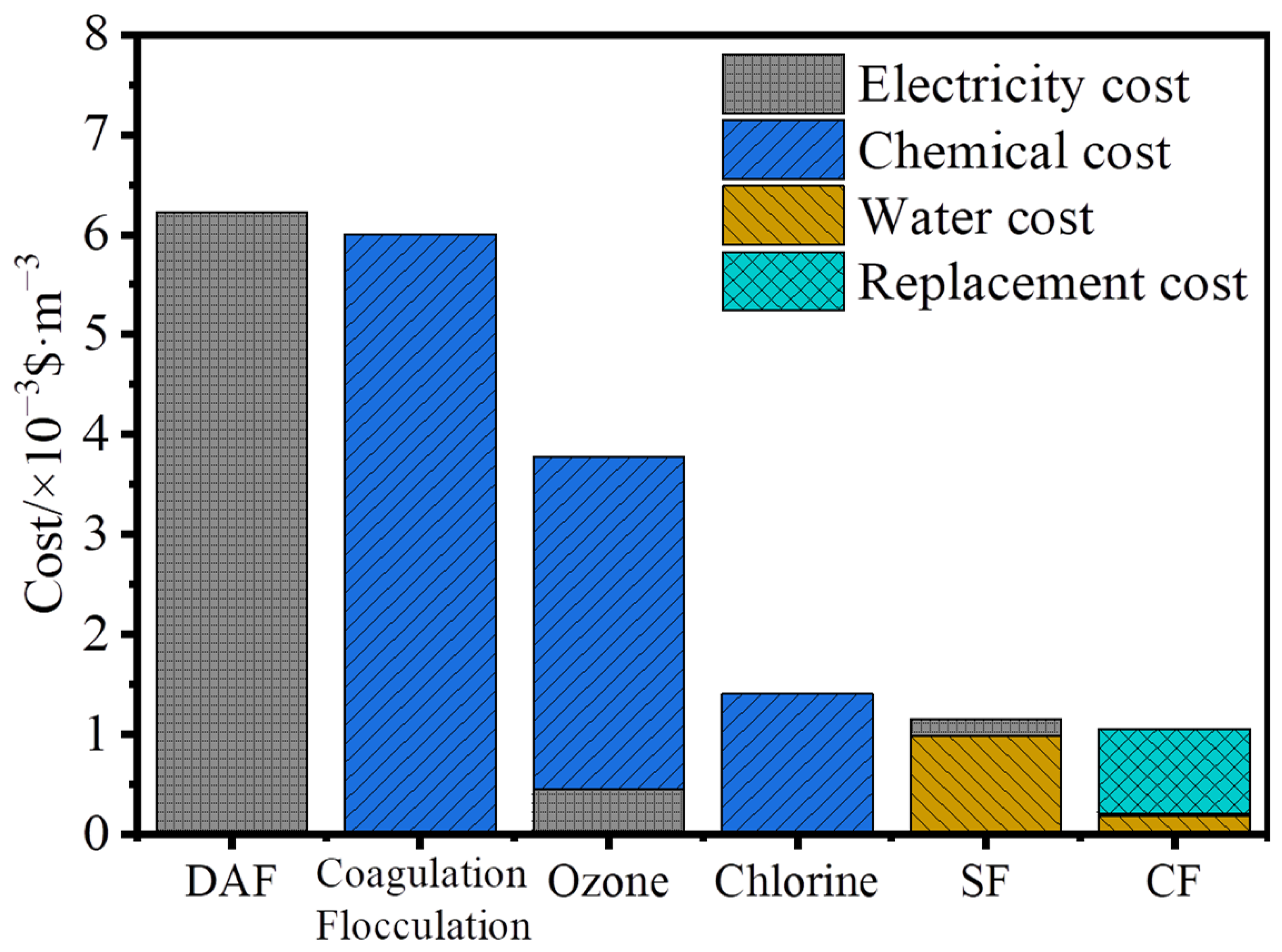
2.2. Operational Costs of the Beijiao DWTP
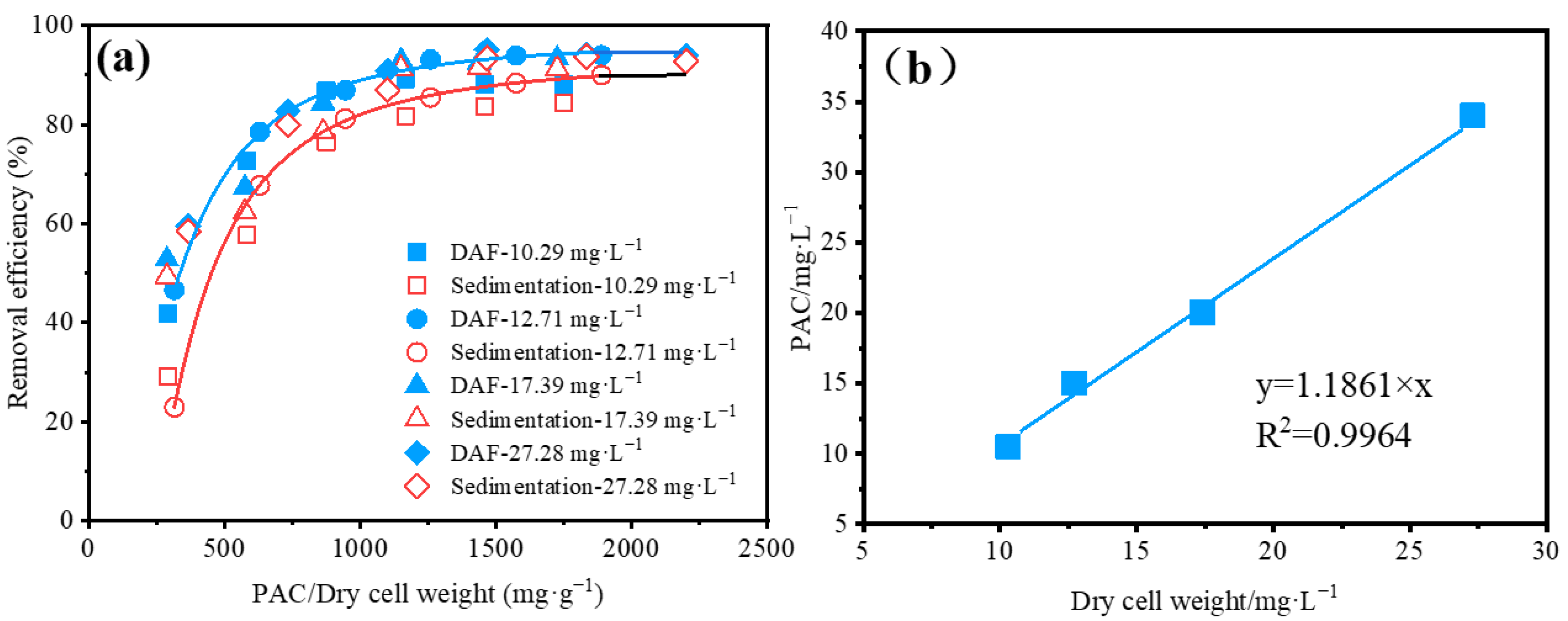
2.3. Algae Removal Using DAF or Sedimentation Batch Experiment in the Beijiao DWTP
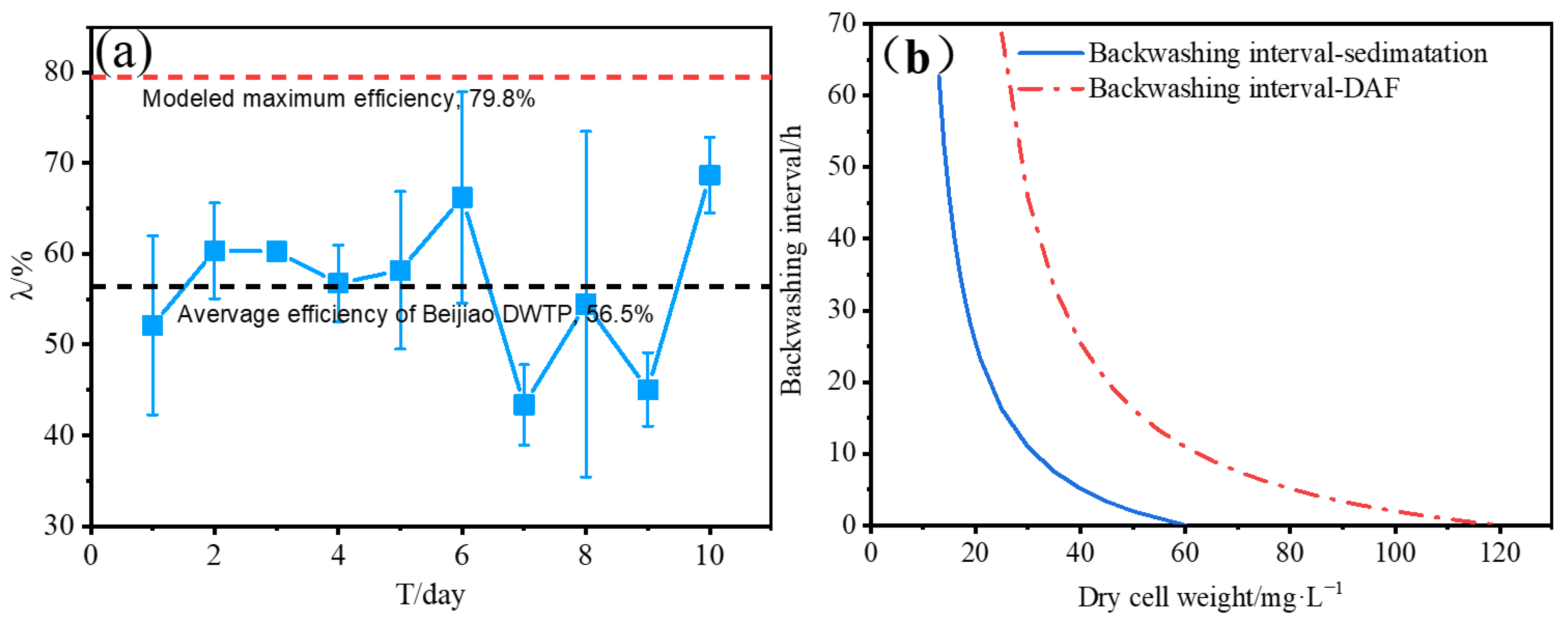
2.4. Simulation Removal Efficiency and Backwash Interval of the Sand Filtration Process
3. Results and Discussion
3.1. Economic Costs of the Beijiao DWTP
3.2. Evaluation of the Performance of Algae Removal Using DAF or Sedimentation
3.3. Influence of Algae Concentration on the Backwash Interval of Sand Filtration
3.4. Protocol for Cost-Effective Treatment Processes for Algae Removal
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of U.S. Freshwaters: Analysis of Potential Economic Damages. Environ. Sci. Technol. 2009, 43, 12–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Howarth, R.W.; Marino, R. Nitrogen as the limiting nutrient for eutrophication in coastal marine ecosystems: Evolving views over three decades. Limnol. Oceanogr. 2006, 51, 364–376. [Google Scholar] [CrossRef] [Green Version]
- Li, K.; Xu, T.; Xi, J.; Jia, H.; Gao, Z.; Sun, Z.; Yin, D.; Leng, L. Multi-factor analysis of algal blooms in gate-controlled urban water bodies by data mining. Sci. Total Environ. 2021, 753, 141821. [Google Scholar] [CrossRef] [PubMed]
- Moss, B.; Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Liu, Z. Nitrogen, macrophytes, shallow lakes and nutrient limitation: Resolution of a current controversy? Hydrobiologia 2013, 710, 3–21. [Google Scholar] [CrossRef]
- Trottet, A.; George, C.; Drillet, G.; Lauro, F.M. Aquaculture in coastal urbanized areas: A comparative review of the challenges posed by Harmful Algal Blooms. Crit. Rev. Environ. Sci. Technol. 2022, 52, 2888–2929. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, Y.; Han, R.; Song, K.; Zhou, X.; Wang, G.; Wang, Q. Eutrophication triggers the shift of nutrient absorption pathway of submerged macrophytes: Implications for the phytoremediation of eutrophic waters. J. Environ. Manag. 2019, 239, 376–384. [Google Scholar] [CrossRef]
- Ma, J.; Wang, P. Effects of rising atmospheric CO2 levels on physiological response of cyanobacteria and cyanobacterial bloom development: A review. Sci. Total Environ. 2021, 754, 141889. [Google Scholar] [CrossRef]
- Zamyadi, A.; MacLeod, S.L.; Fan, Y.; McQuaid, N.; Dorner, S.; Sauve, S.; Prevost, M. Toxic cyanobacterial breakthrough and accumulation in a drinking water plant: A monitoring and treatment challenge. Water Res. 2012, 46, 1511–1523. [Google Scholar] [CrossRef]
- Chapra, S.C.; Boehlert, B.; Fant, C.; Bierman, V.J.; Henderson, J.; Mills, D.; Mas, D.M.L.; Rennels, L.; Jantarasami, L.; Martinich, J.; et al. Climate Change Impacts on Harmful Algal Blooms in U.S. Freshwaters: A Screening-Level Assessment. Environ. Sci. Technol. 2017, 51, 8933–8943. [Google Scholar] [CrossRef] [Green Version]
- Smith, R.B.; Bass, B.; Sawyer, D.; Depew, D.; Watson, S.B. Estimating the economic costs of algal blooms in the Canadian Lake Erie Basin. Harmful Algae 2019, 87, 101624. [Google Scholar] [CrossRef]
- Dunlap, C.R.; Sklenar, K.S.; Blake, L.J. A Costly Endeavor: Addressing Algae Problems in a Water Supply. J. AWWA 2015, 107, E255–E262. [Google Scholar] [CrossRef]
- K.D.o. Health, Environment, Water Quality Standards White Paper: Chlorophyll—A Criteria for Public Water Supply Lakes and Reservoirs; Kansas Department of Health and Environment Bureau of Water: Topeka, KS, USA, 2011. [Google Scholar]
- Treuer, G.; Kirchhoff, C.; Lemos, M.C.; McGrath, F. Challenges of managing harmful algal blooms in US drinking water systems. Nat. Sustain. 2021, 4, 958–964. [Google Scholar] [CrossRef]
- Westrick, J.A.; Szlag, D.C.; Southwell, B.J.; Sinclair, J. A review of cyanobacteria and cyanotoxins removal/inactivation in drinking water treatment. Anal. Bioanal. Chem. 2010, 397, 1705–1714. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Gu, X.; Chen, H.; Mao, Z.; Shen, R.; Zeng, Q.; Ge, Y. Co-occurrence of multiple cyanotoxins and taste-and-odor compounds in the large eutrophic Lake Taihu, China: Dynamics, driving factors, and challenges for risk assessment. Environ. Pollut. 2022, 294, 118594. [Google Scholar] [CrossRef] [PubMed]
- Pichel, N.; Vivar, M.; Fuentes, M. The problem of drinking water access: A review of disinfection technologies with an emphasis on solar treatment methods. Chemosphere 2019, 218, 1014–1030. [Google Scholar] [CrossRef]
- Liu, X.; Chen, L.; Yang, M.; Tan, C.; Chu, W. The occurrence, characteristics, transformation and control of aromatic disinfection by-products: A review. Water Res. 2020, 184, 116076. [Google Scholar] [CrossRef]
- Cho, K.J.; Choi, Y.; Shin, J.-K.; Lee, J. Problematic algae in the sedimentation and filtration process of water treatment plants. Aqua 2011, 60, 219. [Google Scholar] [CrossRef]
- Mahmoud, E.; Gad, A.; Farrag, A.; Aboeldahb, S. Evaluation of the most promising techniques overcoming the algal problems takes place during the purification of drinking water. Environ. Sci. Pollut. Res. 2021, 28, 44239–44248. [Google Scholar] [CrossRef]
- Sze, Y.S.; Aris, A.B.; Zaidi, N.S.; Bahrodin, M.B. Performance of Sand Filtration System with Different Sand Bed Depth for Polishing Wastewater Treatment. J. Environ. Treat. Tech. 2021, 9, 452–457. [Google Scholar]
- Saravanan, A.; Kumar, P.S.; Varjani, S.; Jeevanantham, S.; Yaashikaa, P.R.; Thamarai, P.; Abirami, B.; George, C.S. A review on algal-bacterial symbiotic system for effective treatment of wastewater. Chemosphere 2021, 271, 129540. [Google Scholar] [CrossRef]
- Esen, M.; Yuksel, T. Experimental evaluation of using various renewable energy sources for heating a greenhouse. Energy Build. 2013, 65, 340–351. [Google Scholar] [CrossRef]
- Taner, T.; Sivrioglu, M. A techno-economic & cost analysis of a turbine power plant: A case study for sugar plant. Renew. Sustain. Energy Rev. 2017, 78, 722–730. [Google Scholar]
- Taner, T. Optimisation processes of energy efficiency for a drying plant: A case of study for Turkey. Appl. Therm. Eng. 2015, 80, 247–260. [Google Scholar] [CrossRef]
- Roy, S.; Ghosh, S.; Bhowmick, N. Application of Colloidal Filtration Theory on Textile Fibrous Media: Effect of Fiber Orientation on Bacterial Removal Efficiency and Attachment. J. Inst. Eng. India Ser. E 2018, 99, 111–117. [Google Scholar] [CrossRef]
- Tufenkji, N.; Elimelech, M. Correlation Equation for Predicting Single-Co llector Efficiency in Physicochemical Filtration in Saturated Porous Media. Environ. Sci. Technol. 2004, 38, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Wu, W.; Liu, H.; Huang, Y.; Bi, X.; Liao, R.; Yin, S. Dimensional Analysis Model of Head Loss for Sand Media Filters in a Drip Irrigation System Using Reclaimed Water. Water 2022, 14, 961. [Google Scholar] [CrossRef]
- Najdawi, F.; Neptune, K. Optimizing Reverse Osmosis Membrane Parameters through the Use of the Solution-Diffusion Model: A Review. Engineering 2022, 14, 9–32. [Google Scholar] [CrossRef]
- Li, C.; Luo, F.; Duan, H.; Dong, F.; Chen, X.; Feng, M.; Zhang, Z.; Cizmas, L.; Sharma, V.K. Degradation of chloramphenicol by chlorine and chlorine dioxide in a pilot-scale water distribution system. Sep. Purif. Technol. 2019, 211, 564–570. [Google Scholar] [CrossRef]
- Zhang, J.; Liao, Y.; Wang, Q.; Wang, C.; Yu, J. Degradation of odorous sulfide compounds by different oxidation processes in drinking water: Performance, reaction kinetics and mechanism. Water Res. 2020, 189, 116643. [Google Scholar] [CrossRef]
- dos Santos, P.R.; Daniel, L.A. A review: Organic matter and ammonia removal by biological activated carbon filtration for water and wastewater treatment. Int. J. Environ. Sci. Technol. 2020, 17, 591–606. [Google Scholar] [CrossRef]
- Teixeira, M.R.; Rosa, M.J. Comparing dissolved air flotation and conventional sedimentation to remove cyanobacterial cells of Microcystis aeruginosa: Part II. The effect of water background organics. Sep. Purif. Technol. 2007, 53, 126–134. [Google Scholar] [CrossRef]
- Khiadani, M.; Kolivand, R.; Ahooghalandari, M.; Mohajer, M. Removal of turbidity from water by dissolved air flotation and conventional sedimentation systems using poly aluminum chloride as coagulant. Desalination Water Treat. 2014, 52, 985–989. [Google Scholar] [CrossRef]
- Edzwald, J.K. Principles and applications of dissolved air flotation. Water Sci. Technol. 1995, 31, 1–23. [Google Scholar] [CrossRef]
- Song, Z.; Zhang, X.; Ngo, H.H.; Guo, W.; Wen, H.; Li, C. Occurrence, fate and health risk assessment of 10 common antibiotics in two drinking water plants with different treatment processes. Sci. Total Environ. 2019, 674, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Kato, R.; Asami, T.; Utagawa, E.; Furumai, H.; Katayama, H. Pepper mild mottle virus as a process indicator at drinking water treatment plants employing coagulation-sedimentation, rapid sand filtration, ozonation, and biological activated carbon treatments in Japan. Water Res. 2018, 132, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Yang, J.; Ou, M.; Wang, Y.; Jia, J. Study of Microcystis aeruginosa inhibition by electrochemical method. Biochem. Eng. J. 2007, 36, 215–220. [Google Scholar] [CrossRef]
- Liang, H.; Gong, W.; Chen, J.; Li, G. Cleaning of fouled ultrafiltration (UF) membrane by algae during reservoir water treatment. Desalination 2008, 220, 267–272. [Google Scholar] [CrossRef]
- Tsai, M.-H.; Hua, L.-C.; Huang, K.; Huang, C. NOM removal and residual Al minimization by enhanced coagulation: Roles of sequence dosing with PACl–FeCl3. J. Water Supply: Res. Technol. Aqua 2020, 69, 616–628. [Google Scholar] [CrossRef]
- Di Marcantonio, C.; Bertelkamp, C.; van Bel, N.; Pronk, T.E.; Timmers, P.H.A.; van der Wielen, P.; Brunner, A.M. Organic micropollutant removal in full-scale rapid sand filters used for drinking water treatment in The Netherlands and Belgium. Chemosphere 2020, 260, 127630. [Google Scholar] [CrossRef] [PubMed]
- Matilainen, A.; Vieno, N.; Tuhkanen, T. Efficiency of the activated carbon filtration in the natural organic matter removal. Environ. Int. 2006, 32, 324–331. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, L.; Sommerfeld, M.; Hu, Q. Harvesting microalgal biomass using magnesium coagulation-dissolved air flotation. Biomass Bioenergy 2016, 93, 43–49. [Google Scholar] [CrossRef]
- Ashour, M.; Alprol, A.E.; Heneash, A.M.M.; Saleh, H.; Abualnaja, K.M.; Alhashmialameer, D.; Mansour, A.T. Ammonia Bioremediation from Aquaculture Wastewater Effluents Using Arthrospira platensis NIOF17/003: Impact of Biodiesel Residue and Potential of Ammonia-Loaded Biomass as Rotifer Feed. Materials 2021, 14, 5460. [Google Scholar] [CrossRef]
- Mansour, A.T.; Ashour, M.; Alprol, A.E.; Alsaqufi, A.S. Aquatic Plants and Aquatic Animals in the Context of Sustainability: Cultivation Techniques, Integration, and Blue Revolution. Sustainability 2022, 14, 3257. [Google Scholar] [CrossRef]
- Yin, Z.; Chu, R.; Zhu, L.; Li, S.; Mo, F.; Hu, D.; Liu, C. Application of chitosan-based flocculants to harvest microalgal biomass for biofuel production: A review. Renew. Sustain. Energy Rev. 2021, 145, 111159. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Leng, L.; Wei, L.; Xiong, Q.; Xu, S.; Li, W.; Lv, S.; Lu, Q.; Wan, L.; Wen, Z.; Zhou, W. Use of microalgae based technology for the removal of antibiotics from wastewater: A review. Chemosphere 2020, 238, 124680. [Google Scholar] [CrossRef] [PubMed]
- He, X.X.; Liu, Y.L.; Conklin, A.; Westrick, J.; Weavers, L.K.; Dionysiou, D.D.; Lenhart, J.J.; Mouser, P.J.; Szlag, D.; Walker, H.W. Toxic cyanobacteria and drinking water: Impacts, detection, and treatment. Harmful Algae 2016, 54, 174–193. [Google Scholar] [CrossRef]


| Parameters | Value | Unit |
|---|---|---|
| Ozonator | 35 | kw |
| Ozonator-supporting equipment | 8.9 | kw |
| Ozone dosing | 16.08 | kw |
| Air floating return pump | 37 | kw |
| Air floating supporting equipment | 32 | kw |
| Chemical dosing | 2.7 | kw |
| Backwash pump of sand filtration | 45 | kw |
| Backwash pump of activated carbon filtration | 55 | kw |
| Air blower | 75 | kw |
| Parameters | Value | Unit |
|---|---|---|
| Electricity | 0.1126 | $·kwh−1 |
| Water | 0.0563 | $·m−3 |
| PACl | 0.4927 | $·kg−1 |
| Liquid oxygen | 0.2393 | $·kg−1 |
| Liquid chlorine | 0.5631 | $·kg−1 |
| Activated carbon | 0.357 | $·kg−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, M.; Li, L.; Mubashar, M.; Su, X.; Liang, Y.; Zhang, H.; Zhang, X. Equation Chapter 1 Section 1 Techno-Economic Analysis for the Selection of Cost-Effective Treatment for Algae Removal in Drinking Water Treatment Plants. Water 2023, 15, 243. https://doi.org/10.3390/w15020243
Liu M, Li L, Mubashar M, Su X, Liang Y, Zhang H, Zhang X. Equation Chapter 1 Section 1 Techno-Economic Analysis for the Selection of Cost-Effective Treatment for Algae Removal in Drinking Water Treatment Plants. Water. 2023; 15(2):243. https://doi.org/10.3390/w15020243
Chicago/Turabian StyleLiu, Mingmeng, Lili Li, Muhammad Mubashar, Xuhui Su, Yangchun Liang, Haiyang Zhang, and Xuezhi Zhang. 2023. "Equation Chapter 1 Section 1 Techno-Economic Analysis for the Selection of Cost-Effective Treatment for Algae Removal in Drinking Water Treatment Plants" Water 15, no. 2: 243. https://doi.org/10.3390/w15020243






