Herbicide Fate and Transport in the Great Barrier Reef: A Review of Critical Parameters
Abstract
:1. Introduction
2. Pollutant Load to the GBR
2.1. Natural Resource Management (NRM) Regions of the GBR Catchments
2.2. Nutrient Pesticide and Sediment Loads
2.3. Types of Herbicides Found within the GBR Catchments
3. Critical Parameter in Herbicide Fate and Transport
3.1. Spray Drift
3.2. Type of Crops Harvested
3.3. Degradation through Volatilization and Photodegradation
3.4. Rainfall Occurrence and Intensity
3.5. Washoff Coefficients of Herbicides
3.6. Herbicide Sorption
3.7. Herbicide Half-Life Affecting Microbial Degradation
3.8. Transport Pathways of Persistent Herbicide and Metabolites
3.9. Transport Pathway through Evapotranspiration
3.10. Transport Pathway through Preferential Flow
4. Modelling of Fate and Transport of Herbicides in the GBR Catchment
4.1. Input Parameters on Herbicide Fate and Transport
4.2. Current Status of Modelling
- (1)
- The scenarios represent several management practices relevant to GBR catchments and describe the products applied and their timing and rates.
- (2)
- The application of herbicides is calculated based on product concentration, product rate, and application efficiency, as well as application onto the crop, crop residue or directly to the soil.
- (3)
- Herbicide concentration in surface soil is calculated after leaching losses and extraction coefficients were used to estimate concentrations of herbicides in runoff as a function of soil concentration.
- (4)
- The proportion of herbicides in dissolved and particulate phases was considered using partitioning coefficients between soil and water which were calculated from both soil and herbicide chemistry.
- (5)
- Temperature dependent half-lives of herbicides were obtained from available studies in the literature or from Paddock to Reef monitoring results, where possible, and first order degradation rates are assumed.
4.3. Model Comparison
5. Improvement of Model Results Using Critical Parameters
5.1. Herbicide Usage Data
5.2. Herbicide Degradation Half-Life
5.3. Herbicide Transport Pathway through Aerial Drift
5.4. Herbicide Transport Pathway within Crop Residues
5.5. Herbicide Transport Pathway within Root Zone
5.6. Toxicological Effects of Herbicides
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Valck, J.; Rolfe, J. Linking water quality impacts and benefits of ecosystem services in the Great Barrier Reef. Mar. Pollut. Bull. 2018, 130, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, J.; Brodie, J.; Lewis, S.; Mitchell, A. Quantifying the sources of pollutants in the Great Barrier Reef catchments and the relative risk to reef ecosystems. Mar. Pollut. Bull. 2012, 65, 394–406. [Google Scholar] [CrossRef] [PubMed]
- Emslie, M.J.; Bray, P.; Cheal, A.J.; Johns, K.A.; Osborne, K.; Sinclair-Taylor, T.; Thompson, C.A. Decades of monitoring have informed the stewardship and ecological understanding of Australia’s Great Barrier Reef. Biol. Conserv. 2020, 252, 108854. [Google Scholar] [CrossRef]
- Commonwealth-of-Australia. State Party Report on the State of Conservation of the Great Barrier Reef World Heritage Area (Australia). 2019. Available online: https://www.dcceew.gov.au/ (accessed on 12 October 2022).
- Commonwealth-of-Australia. Reef 2050 Long-Term Sustainability Plan. 2015. Available online: https://www.dcceew.gov.au/parks-heritage/great-barrier-reef/protecting/reef-2050-plan (accessed on 14 October 2022).
- Tarte, D.; Hughes, T. Review of State Party Report on the state of conservation of the Great Barrier Reef World Heritage Area (Australia). 2020. Available online: https://www.marineconservation.org.au/ (accessed on 12 October 2022).
- Australian-and-Queensland-Governments. Pesticide Risk Baseline Results, Reef Water Quality Report Card 2017 and 2018. 2019. Available online: https://www.reefplan.qld.gov.au/__data/assets/pdf_file/0026/82907/report-card-2017-2018-results-pesticide-risk-baseline.pdf (accessed on 10 October 2022).
- Waters, D. Modelling Reductions of Pollutant Loads Due to Improved Management Practices in the Great Barrier Reef Catchments. 2014; pp. 1–120. Available online: https://www.reefplan.qld.gov.au/__data/assets/pdf_file/0027/46098/great-barrier-reef-catchment-modelling-report.pdf (accessed on 8 October 2022).
- Smith, R.; Middlebrook, R.; Turner, R.; Huggins, R.; Vardy, S.; Warne, M. Large-scale pesticide monitoring across Great Barrier Reef catchments–paddock to reef integrated monitoring, modelling and reporting program. Mar. Pollut. Bull. 2012, 65, 117–127. [Google Scholar] [CrossRef]
- Carroll, C.; Waters, D.; Vardy, S.; Silburn, D.M.; Attard, S.; Thorburn, P.J.; Davis, A.; Halpin, N.; Schmidt, M.; Wilson, B.; et al. A Paddock to reef monitoring and modelling framework for the Great Barrier Reef: Paddock and catchment component. Mar. Pollut. Bull. 2012, 65, 136–149. [Google Scholar] [CrossRef]
- Queensland Government. Reef 2050 Water Quality Improvement Plan; 2019. Available online: https://www.reefplan.qld.gov.au/water-quality-and-the-reef/the-plan (accessed on 4 January 2023).
- Economics, D.A. Economic contribution of the Great Barrier Reef. 2013. Available online: https://www.dcceew.gov.au/parks-heritage/great-barrier-reef/publications/economic-contribution-great-barrier-reef-march-2013 (accessed on 12 October 2022).
- DSITI. Land Use Summary 1999–2013: Cape York NRM Region. 2015. Available online: https://www.publications.qld.gov.au/dataset/9f2aafcd-1994-4958-ac5b-b6a034b2b0ef/resource/dd0dc40a-2930-4e6f-9fcd-41257c35fde2/download/cape-york-land-use-summary.pdf (accessed on 12 October 2022).
- DSITI. Land Use Summary 1999–2015: Wet Tropics NRM Region. 2016. Available online: https://www.publications.qld.gov.au/dataset/1e257ae1-8bdb-4fc1-a742-6f4a22d55ecf/resource/0bf01de4-0e89-4680-90c9-05154f8034e3/download/wet-tropics-land-use-summary-1999-2015.pdf (accessed on 12 October 2022).
- DSITI. Land Use Summary 1999–2015: Burdekin NRM Region. 2016. Available online: https://www.publications.qld.gov.au/dataset/land-use-summary-1999-2015 (accessed on 12 October 2022).
- DSITI. Land Use Summary 1999–2016: Mackay Whitsunday NRM Region. 2017. Available online: https://www.publications.qld.gov.au/dataset/4b4ea2de-0327-4c48-85f3-fe9b5fcd73c3/resource/4956d016-eee4-40b7-85fa-93495a5259fe/download/mackay-whitsunday-land-use-summary-1999-2016.pdf (accessed on 12 October 2022).
- DSITI. Land Use Summary 1999–2017: Fitzroy NRM Region. 2017. Available online: http://qldspatial.information.qld.gov.au/catalogue/custom/search.page?q=%22Land%20use%20mapping%20-%201999%20to%202017%20-%20Fitzroy%20NRM%22 (accessed on 12 October 2022).
- DSITI. Land Use Summary 1999–2017: Burnett Mary NRM Region. 2017. Available online: https://www.publications.qld.gov.au/dataset/51761481-26cc-492b-92f1-1b5ed6667ec7/resource/dff51ef9-fe65-42ee-958a-75853e2fd8a2/download/burnettmarylanduse19992009.pdf (accessed on 12 October 2022).
- Linnenluecke, M.K.; Zhou, C.; Smith, T.; Thompson, N.; Nucifora, N. The impact of climate change on the Australian sugarcane industry. J. Clean. Prod. 2020, 246, 118974. [Google Scholar] [CrossRef]
- DES. Reef 2050 Water Quality Improvement Plan; Department of Environment and Science, Queensland Government: Queensland, Australia, 2020.
- Bartley, R.; Corfield, J.P.; Hawdon, A.A.; Kinsey-Henderson, A.E.; Abbott, B.N.; Wilkinson, S.N.; Keen, R.J. Can changes to pasture management reduce runoff and sediment loss to the Great Barrier Reef? The results of a 10-year study in the Burdekin catchment, Australia. Rangel. J. 2014, 36, 67–84. [Google Scholar] [CrossRef] [Green Version]
- Al-Khatib, K. Herbicide Symptoms. 2020. Available online: http://herbicidesymptoms.ipm.ucanr.edu/ (accessed on 2 November 2022).
- Queensland Government. Reef Water Quality Protection Plan. 2009; pp. 37–56. Available online: https://www.reefplan.qld.gov.au/__data/assets/pdf_file/0025/46177/technical-report.pdf (accessed on 2 November 2022).
- Smith, R.A.; Turner, R.; Vardy, S.; Huggins, R.; Rohan, W. An Evaluation of the Prevalence of Alternate Pesticides of Environmental Concern in Great Barrier Reef Catchments. 2015. Available online: https://www.publications.qld.gov.au/dataset/1bf3951b-e3cc-4727-9d96-262f374f553f/resource/efaa76da-5714-45a5-ba40-30476d9e214e/download/alternatepesticidesreportrp57cfinalmetadata.pdf (accessed on 14 October 2022).
- Davis, A.; Smith, R.; Negri, A.; Thompson, M.; Poggio, M. Advancing our understanding of the source, management, transport and impacts of pesticides on the Great Barrier Reef 2011-2015. 2016. Available online: https://www.researchgate.net/publication/303301289_Advancing_our_understanding_of_the_source_management_transport_and_impacts_of_pesticides_on_the_Great_Barrier_Reef_2011-2015 (accessed on 12 November 2022).
- Leonard, R.; Knisel, W.; Davis, F. Modelling pesticide fate with GLEAMS. Eur. J. Agron. 1995, 4, 485–490. [Google Scholar] [CrossRef]
- Wang, R.; Yuan, Y.; Yen, H.; Grieneisen, M.; Arnold, J.; Wang, D.; Wang, C.; Zhang, M. A review of pesticide fate and transport simulation at watershed level using SWAT: Current status and research concerns. Sci. Total Environ. 2019, 669, 512–526. [Google Scholar] [CrossRef]
- de Baan, L. Sensitivity analysis of the aquatic pesticide fate models in SYNOPS and their parametrization for Switzerland. Science of the Total Environment 2020, 715, 136881. [Google Scholar] [CrossRef]
- Marín-Benito, J.; Pot, V.; Alletto, L.; Mamy, L.; Bedos, C.; Barriuso, E.; Benoit, P. Comparison of three pesticide fate models with respect to the leaching of two herbicides under field conditions in an irrigated maize cropping system. Sci. Total. Environ. 2014, 499, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Felix, M.; Holst, N.; Sharp, A. PestTox: An object oriented model for modeling fate and transport of pesticides in the environment and their effects on population dynamics of non-target organisms. Comput. Electron. Agric. 2019, 166. [Google Scholar] [CrossRef]
- GRDC. Understanding Pre-Emergent Herbicides and How They Interact with the Environment. 2007, pp. 1–9. Available online: https://grdc.com.au/ (accessed on 10 October 2022).
- The Pennsylvania State University, Volitilization 2003. Available online: http://www.personal.psu.edu/faculty/a/s/asm4/turfgrass/education/turgeon/lessons/lesson13/corefiles/links/pestdanger/vol1.html (accessed on 10 October 2022).
- Dang, A.; Silburn, M.; Craig, I.; Shaw, M.; Foley, J. Washoff of Residual Photosystem II Herbicides from Sugar Cane Trash under a Rainfall Simulator. J. Agric. Food Chem. 2016, 64, 3967–3974. [Google Scholar] [CrossRef] [PubMed]
- Shaw, M.; Dunlop, J.; McGregor, G. Identifying Pesticides and Locations for Monitoring in the Great Barrier Reef Catchments. In Approach and Assessment Outputs; Queensland Department of Environment and Resource Management: Brisbane, Australia, 2011; p. 86. [Google Scholar]
- Trovato, V.W.; Portilho, I.I.R.; Barizon, R.R.; Scorza, R.P., Jr. Herbicide runoff from a soil with different levels of sugarcane straw coverage in Brazil. Ecotoxicol. Environ. Contam. 2020, 15, 25–35. [Google Scholar] [CrossRef]
- Hussain, S.; Arshad, M.; Springael, D.; Sørensen, S.R.; Bending, G.; Devers-Lamrani, M.; Maqbool, Z.; Martin-Laurent, F. Abiotic and Biotic Processes Governing the Fate of Phenylurea Herbicides in Soils: A Review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 1947–1998. [Google Scholar] [CrossRef]
- Tiktak, A. Application of pesticide leaching models to the Vredepeel dataset: II Pesticide fate. Agric. Water Manag. 2000, 44, 119–134. [Google Scholar] [CrossRef]
- Siimes, K.; Kämäri, J. A review of available pesticide leaching models: Selection of models for simulation of herbicide fate in Finnish sugar beet cultivation. Boreal Environ. Res. 2003, 8, 31–51. [Google Scholar]
- Simunek, J.; Jacques, D.; Langergraber, G.; Bradford, S.A.; Šejna, M.; van Genuchten, M.T. Numerical modeling of contaminant transport using HYDRUS and its specialized modules. J. Indian Inst. Sci. 2013, 93, 265–284. [Google Scholar]
- Shaw, M.; Silburn, D.; Thornton, C.; Robinson, B.; McClymont, D. Modelling pesticide runoff from paddocks in the Great Barrier Reef with HowLeaky. In Proceedings of the International Congress on Modelling and Simulation (MODSIM), Perth, WA, Australia, 12–16 December 2011. [Google Scholar]
- Connolly, R.; Kennedy, I.; Silburn, D.; Simpson, B.; Freebairn, D. Simulating Endosulfan Transport in Runoff from Cotton Fields in Australia with the GLEAMS Model. J. Environ. Qual. 2001, 30, 702–713. [Google Scholar] [CrossRef] [Green Version]
- King, O.; Smith, R.; Mann, R.; Warne, M. Proposed Aquatic Ecosystem Protection Guideline Values for Pesticides Commonly Used in the Great Barrier Reef Catchment Area: Part 1–2, 4-D, Ametryn, Diuron, Glyphosate, Hexazinone, Imazapic, Imidacloprid, Isoxaflutole, Metolachlor, Metribuzin, Metsulfuron-methyl, Simazine and Tebuthiuron; Department of Science, Information Technology and Innovation: Brisbane, Australia, 2017; p. 211. [Google Scholar]
- Aslam, S.; Iqbal, A.; Lafolie, F.; Recous, S.; Benoit, P.; Garnier, P. Mulch of plant residues at the soil surface impact the leaching and persistence of pesticides: A modelling study from soil columns. J. Contam. Hydrol. 2018, 214, 54–64. [Google Scholar] [CrossRef] [Green Version]
- Queyrel, W.; Habets, F.; Blanchoud, H.; Ripoche, D.; Launay, M. Pesticide fate modeling in soils with the crop model STICS: Feasibility for assessment of agricultural practices. Sci. Total Environ. 2016, 542, 787–802. [Google Scholar] [CrossRef] [PubMed]
- Simpson, B.W.; Fraser, G.; Armour, J.; Hargreaves, P.; Ruddle, L. Pesticide Studies—Australia. In Offsite Movement of Agrochemicals in Tropical Sugarcane Production: Extension Workshop, Bundaberg, Australia; Department of Natural Resources and Mines: Bundaberg, Australia, 2001; pp. 87–100. [Google Scholar]
- King, G.K.K.; Larras, F.; Charles, S.; Delignette-Muller, M.L. Hierarchical modelling of species sensitivity distribution: Development and application to the case of diatoms exposed to several herbicides. Ecotoxicol. Environ. Saf. 2015, 114, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Bontje, D.; Kooi, B.; Liebig, M.; Kooijman, S. Modelling long-term ecotoxicological effects on an algal population under dynamic nutrient stress. Water Res. 2009, 43, 3292–3300. [Google Scholar] [CrossRef] [PubMed]
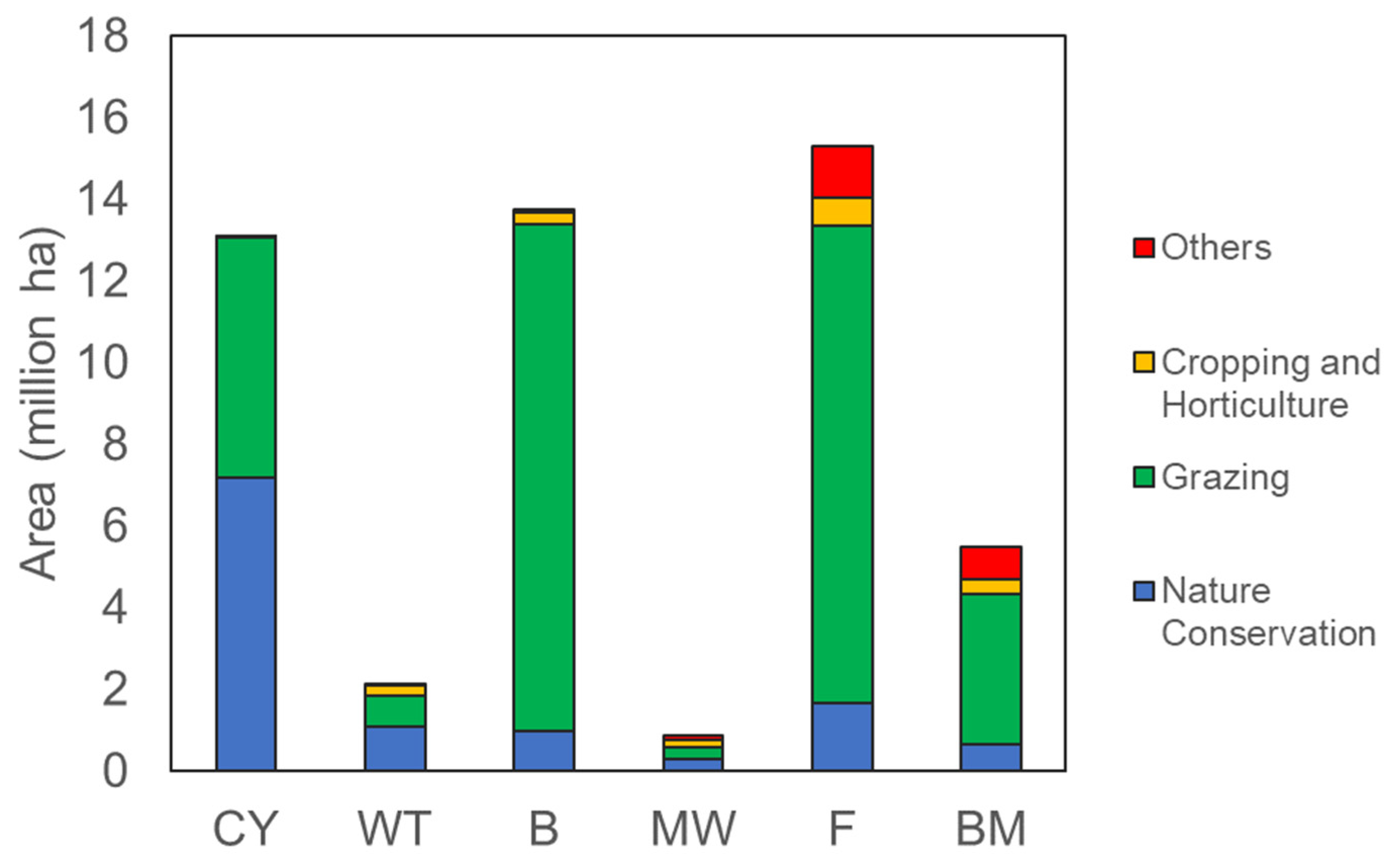

| Catchment No. | Catchment Name | Natural Resource Management (NRM) Regions | Land Use (% of Land Area, Excluding Water Bodies) | Average Pollutant Loads to the GBR (Relative Proportion in %) [1] | ||
|---|---|---|---|---|---|---|
| Nutrients (Dissolve Inorganic Nitrogen, DIN) | Sediments (Total Suspended Sediment, TSS) | Pesticides | ||||
| 101 | Jacky-Jacky Creek | I. Cape York Region Total area: ~136,520 km2 | Grazing (~44.7%) Cropping and Horticulture (~0.1%) Nature conservation (~55%) Others (~0.2%) [13] | 0.13 | 5.03 | 0.01 |
| 102 | Olive-Pascoe | |||||
| 103 | Lockhart River | |||||
| 104 | Stewart River | |||||
| 105 | Normanby River | |||||
| 106 | Jeannie River | |||||
| 107 | Endeavour River | |||||
| 108 | Daintree River | II. Wet Tropics Region Total area: ~22,230 km2 | Grazing (~51%) Cropping and Horticulture (~35%) Nature conservation (~11%) Others (~3%) [14] | 36.33 | 11.84 | 35.79 |
| 109 | Mossman River | |||||
| 110 | Barron River | |||||
| 111 | Mulgrave-Russell River | |||||
| 112 | Johnstone River | |||||
| 113 | Tully River | |||||
| 114 | Murray River | |||||
| 115 | Hinchinbrook Island | |||||
| 116 | Herbert River | |||||
| 117 | Black River | III. Burdekin Region Total area: ~140,874 km2 | Grazing (~90.4%) Cropping and Horticulture (~2%) Nature cons. (~7%) Others (~0.6%) [15] | 29.46 | 40.43 | 18.93 |
| 118 | Ross River | |||||
| 119 | Haughton River | |||||
| 120 | Burdekin River | |||||
| 121 | Don River | |||||
| 122 | Proserpine River | IV. Mackay Whitsunday Total area: ~9335 km2 | Grazing (~35%) Cropping and Horticulture (~18%) Nature cons. (~34%) Others (~13%) [16] | 11.89 | 7.43 | 43.2 |
| 123 | Whitsunday Island | |||||
| 124 | O’Connell River | |||||
| 125 | Pioneer River | |||||
| 126 | Plane Creek | |||||
| 127 | Styx River | V. Fitzroy Region Total area: ~156,762 km2 | Grazing (~76.3%) Cropping and Horticulture (~4.3%) Nature conservation (11%) Others (~8.4%) [17] | 10.96 | 20.28 | 0.74 |
| 128 | Shoalwater | |||||
| 129 | Waterpark Creek | |||||
| 130 | Fitzroy River | |||||
| 131 | Curtis Island | |||||
| 132 | Calliope River | |||||
| 133 | Boyne River | |||||
| 134 | Baffle Creek | VI. Burnett Mary Region Total area: ~55,777 km2 | Grazing (~67.2%) Cropping and Horticulture (~6.7%) Nature conservation (12%) Others (~14.1%) [18] | 11.23 | 14.99 | 1.33 |
| 135 | Kolan River | |||||
| 136 | Burnett River | |||||
| 137 | Burrum River | |||||
| 138 | Mary River | |||||
| 139 | Fraser Island | |||||
| No. | Catchments | Largest Pesticide Loads Contributor [9] | Risk Category | Pesticide Risk Baseline (% Species Protected) | Contribution of Pesticide Groups (Relative Proportion in %) | ||
|---|---|---|---|---|---|---|---|
| PSII Herbicides | Other Herbicides | Insecticides | |||||
| Australian and Queensland Governments (2019) | |||||||
| Wet Tropics Region | Moderate | 95 | - | - | - | ||
| 108 | Daintree River | Very Low | 100 | 2 | 0 | 98 | |
| 109 | Mossman River | Moderate | 91 | 53 | 36 | 11 | |
| 110 | Barron River | Very Low | 100 | 14 | 69 | 17 | |
| 111 | Mulgrave-Russell River | Moderate | 91 | 47 | 26 | 27 | |
| 112 | Johnstone River | √ | Moderate | 92 | 50 | 32 | 18 |
| 113 | Tully River | √ | Moderate | 93 | 59 | 20 | 21 |
| 114 | Murray River | Moderate | 91 | 63 | 23 | 14 | |
| 116 | Herbert River | Moderate | 94 | 61 | 22 | 17 | |
| Burdekin Region | Low | 98 | - | - | - | ||
| 117 | Black River | Very Low | 99 | 0 | 97 | 3 | |
| 118 | Ross River | Low | 97 | 1 | 79 | 20 | |
| 119 | Haughton River | √ | High | 86 | 61 | 34 | 5 |
| 120 | Burdekin River | √ | Very Low | 99 | 42 | 54 | 4 |
| Sub | Belyando River | √ | - | - | - | - | - |
| Sub | Suttor River | √ | - | - | - | - | - |
| 121 | Don River | Very Low | 100 | 9 | 73 | 18 | |
| Mackay Whitsunday Region | High | 81 | - | - | - | ||
| 122 | Proserpine River | Moderate | 91 | 48 | 28 | 24 | |
| 124 | O’Connell River | High | 84 | 60 | 27 | 13 | |
| 125 | Pioneer River | √ | Very High | 76 | 59 | 24 | 17 |
| 126 | Plane Creek | √ | Very High | 71 | 62 | 26 | 12 |
| Fitzroy Region | Low | 96 | - | - | - | ||
| 127 | Styx River | Very Low | 99 | 48 | 50 | 2 | |
| 128 | Shoalwater | Very Low | 99 | 0 | 5 | 95 | |
| 129 | Waterpark Creek | Very Low | 100 | 48 | 35 | 17 | |
| 130 | Fitzroy River | √ | Low | 96 | 35 | 65 | 0 |
| Sub | Comet River | √ | - | - | - | - | - |
| 132 | Calliope River | Low | 98 | 24 | 70 | 6 | |
| 133 | Boyne River | Very Low | 99 | 17 | 21 | 62 | |
| Burnett Mary Region | Low | 97 | - | - | - | ||
| 134 | Baffle Creek | Very Low | 99 | 16 | 47 | 37 | |
| 135 | Kolan River | Low | 96 | 19 | 56 | 25 | |
| 136 | Burnett River | √ | Low | 97 | 51 | 43 | 6 |
| 137 | Burrum River | Moderate | 92 | 34 | 60 | 6 | |
| 138 | Mary River | Moderate | 95 | 23 | 69 | 8 | |
| Types of Herbicides Detected/Commonly Used in the GBR Catchments | Land Use | GBR Catchments | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sugarcane | Horticulture | Cotton | Urban/STP | Banana | Grazing | Forestry | Broadacre Cropping | Wet Topics | Burdekin | Mackay Whitsunday | Fitzroy | Burnett Mary | |
| ‘Priority’ PSII herbicides | |||||||||||||
| Ametryn | / | / | / | / | / | ||||||||
| Atrazine | / | / | / | / | / | / | / | / | / | ||||
| Diuron | / | / | / | / | / | / | / | / | / | / | |||
| Hexazinone | / | / | / | / | / | / | / | ||||||
| Tebuthiuron | / | / | / | / | / | / | |||||||
| ‘Alternative’ PSII herbicides | |||||||||||||
| Metribuzin | / | / | / | / | |||||||||
| Simazine | / | / | / | / | / | / | / | / | / | ||||
| Terbutryn | / | / | |||||||||||
| Bromacil | / | / | / | / | |||||||||
| Prometryn | / | / | / | ||||||||||
| Fluometuron | / | / | / | ||||||||||
| ‘Alternative’ herbicides | |||||||||||||
| Glyphosate | / | / | |||||||||||
| Imazapic | / | ||||||||||||
| Isoxaflutole | / | / | / | / | |||||||||
| Metolachlor | / | / | / | / | / | / | / | / | / | ||||
| S-metolachlor | |||||||||||||
| Pendimethalin | / | / | |||||||||||
| Picloram | / | ||||||||||||
| 2,4-D | / | / | / | / | / | / | |||||||
| Triclopyr | / | / | / | / | |||||||||
| Trifluralin | / | ||||||||||||
| Imazethapyr | / | ||||||||||||
| Fluroxypyr | / | / | |||||||||||
| MCPA | / | / | / | / | |||||||||
| Propazine | / | ||||||||||||
| Mecoprop | / | ||||||||||||
| Acifluorfen | / | ||||||||||||
| Metsulfuron-methy | / | / | / | ||||||||||
| Glufosinate-ammonium | / | ||||||||||||
| Pyrithiobac sodium | / | / | |||||||||||
| Haloxyfop | / | / | |||||||||||
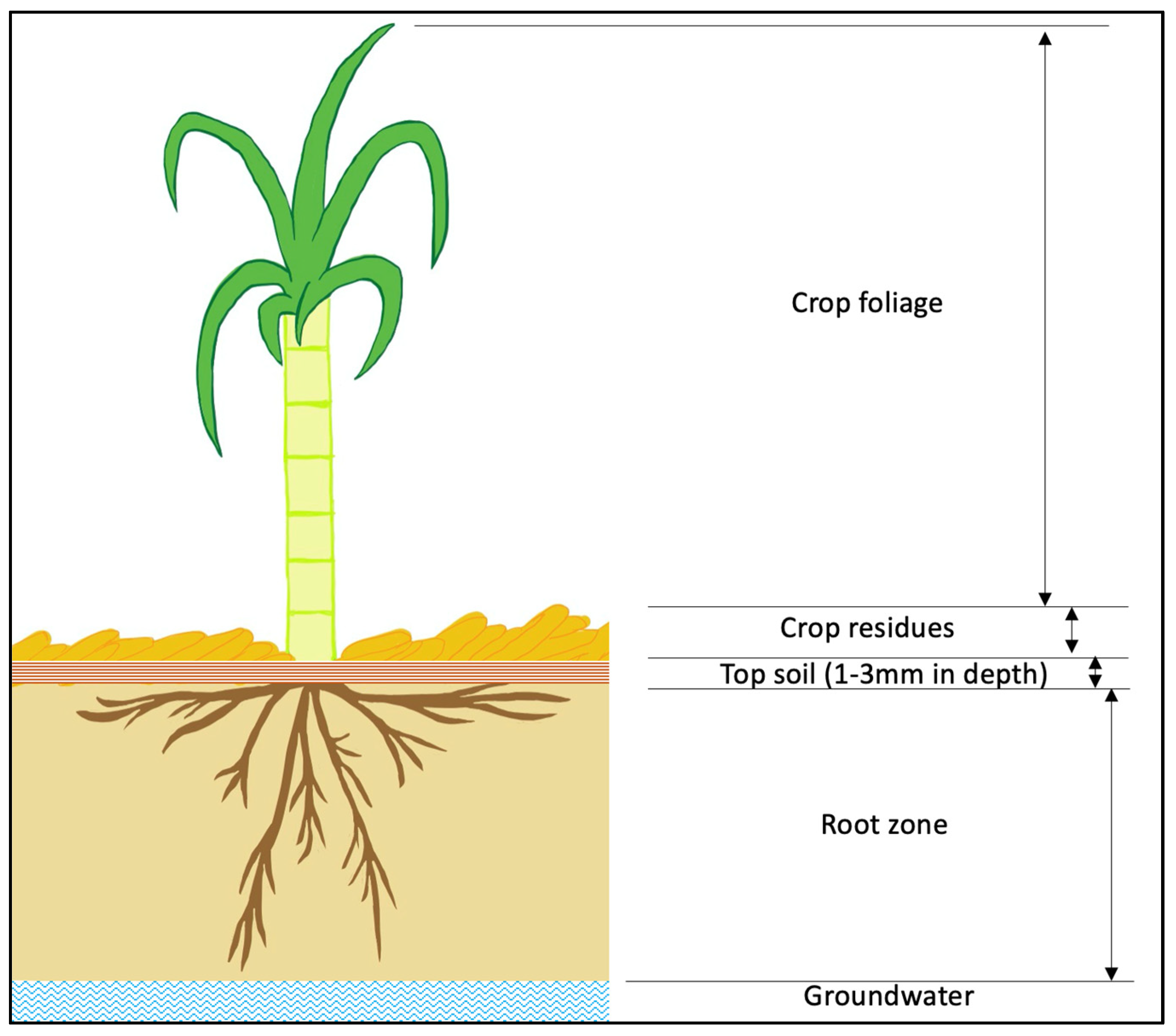
| Fate and Transport | Field Scale Modelling | Basin Level Modelling | End of Catchment | ||
|---|---|---|---|---|---|
| Input Parameters | Fate Behaviour | Mechanism of Transport | Channel Entry Route | Instream Processes | |
| Crop type Agricultural land use Crop coverage Growth stages Photo-degradation Availability of crop residues Transpiration Rooting depth and density Crop residues Amount, thickness, density Residue water content Chemical composition Characteristics of origin Degree of decomposition Biological parameters Herbicide application Mode of application Application rate No. of application Product concentration Application efficiency Time to rain Irrigation management Herbicide properties Half life Degradation Active ingredients Chemical properties Sorption coefficient Wash off coefficient Partition coefficient Environment Slope Precipitation Temperature Distance to water body Water type Evaporation Rainfall data (intensity, amount) Soil properties Soil density and porosity Soil organic carbon Soil water content Soil drainage Soil temperature Soil pH and Eh Hydraulic conductivity Soil layers | Air Wind loss—spray drifts Foliage Uptake by plant Volatilization Photolysis Washoff Crop residues Sorption Volatilization Photolysis Microbial degradation Generation of metabolites Washoff Topsoil Volatilization Photolysis Biolysis Distribution Run-off Leaching Root Zone Uptake by plant Abiotic transformation Hydrolysis (Degradation) Microbial degradation Generation of metabolites Sorption/desorption on organic matter and clays Resurface due to evaporation Leaching into groundwater Subsurface lateral flow Preferential flow through micropores into groundwater | Drift Soil erosions Transported during soil erosion process Liquid Phase Transported by soil water fluxes Transformed and Attenuated Herbicide Dissolved herbicide, break-down products and organic carbon transported by groundwater | Surface waters: Run-off Drift Erosion Subsurface water: Lateral flow Groundwater: Percolation | Losses Outflow Degradation Volatilization Setting Burial Herbicide Distribution: Herbicide transferred between liquid and solid phases via diffusion | Herbicide loads to GBR |
| Authors | Wang et al., (2019) | Shaw et al., (2011) | Anzooman et al., (2013) | Baan (2020) | Mwema et al., (2019) | Jaikaew et al., (2017) | Siimes and Kamari (2003) | Suarez (2013) | de Oliveira et al., (2019) | Boivin et al., (2006) | Leonard et al., (1995) | Aslam et al., (2018) | Marin Benito et al., (2014) | Tiktak (2000) | Bannwarth et al., (2014) | Connoly et al., (2001) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model | ||||||||||||||||
| FATE | ||||||||||||||||
| Air | ||||||||||||||||
| Spray drifts | Y | Y | N | |||||||||||||
| Crop foliage | ||||||||||||||||
| Crop interception | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | ||||||
| Degradation | Y | Y | Y | Y | Y | Y | ||||||||||
| Uptake by plant | ||||||||||||||||
| Washoff | Y | Y | Y | Y | Y | Y | Y | |||||||||
| Crop residues | ||||||||||||||||
| Crop residue interception | Y | |||||||||||||||
| Sorption on crop residues | Y | |||||||||||||||
| Degradation | Y | |||||||||||||||
| Microbial degradation | Y | |||||||||||||||
| Generation of metabolites | Y | |||||||||||||||
| Washoff | Y | |||||||||||||||
| Topsoil (0–3 mm) | ||||||||||||||||
| Degradation | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Distribution phase | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Run-off | Y | Y | Y | Y | Y | Y | Y | N | Y | |||||||
| Leaching | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | |||
| Within Root Zone | ||||||||||||||||
| Uptake by plant | Y | Y | Y | Y | ||||||||||||
| Abiotic transformation | Y | Y | ||||||||||||||
| Degradation | Y | Y | Y | Y | Y | Y | ||||||||||
| Microbial degradation | Y | Y | ||||||||||||||
| Generation of metabolites | Y | Y | Y | |||||||||||||
| Sorption/ | Y | Y | Y | Y | Y | Y | Y | Y | ||||||||
| Resurface | Y | Y | Y | |||||||||||||
| Preferential flow | Y | Y | Y | Y | Y | |||||||||||
| Leaching | Y | Y | ||||||||||||||
| TRANSPORT | ||||||||||||||||
| Surface waters: | ||||||||||||||||
| Runoff | Y | Y | Y | Y | Y | Y | Y | Y | ||||||||
| Drift | Y | Y | ||||||||||||||
| Erosion | Y | Y | Y | Y | Y | Y | ||||||||||
| Subsurface water: | ||||||||||||||||
| Lateral flow | Y | Y | Y | Y | ||||||||||||
| Groundwater: | ||||||||||||||||
| Percolation | Y | Y | Y | Y | Y | Y | Y | |||||||||
| Adjustment for lag | Y | |||||||||||||||
| INSTREAM PROCESSES | ||||||||||||||||
| Pesticides removed via: | ||||||||||||||||
| Outflow | Y | |||||||||||||||
| Degradation | Y | |||||||||||||||
| Volatilization | Y | |||||||||||||||
| Setting | Y | |||||||||||||||
| Burial | Y | |||||||||||||||
| Pesticides Distribution: | ||||||||||||||||
| Pesticides transferred between liquid and solid phases via diffusion | Y |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nahar, K.; Baillie, J.; Zulkarnain, N.A. Herbicide Fate and Transport in the Great Barrier Reef: A Review of Critical Parameters. Water 2023, 15, 237. https://doi.org/10.3390/w15020237
Nahar K, Baillie J, Zulkarnain NA. Herbicide Fate and Transport in the Great Barrier Reef: A Review of Critical Parameters. Water. 2023; 15(2):237. https://doi.org/10.3390/w15020237
Chicago/Turabian StyleNahar, Kamrun, Justine Baillie, and Noor Azwa Zulkarnain. 2023. "Herbicide Fate and Transport in the Great Barrier Reef: A Review of Critical Parameters" Water 15, no. 2: 237. https://doi.org/10.3390/w15020237





