Pb(II) Removal from Aqueous Solutions by Adsorption on Stabilized Zero-Valent Iron Nanoparticles—A Green Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Analytical Techniques
2.2. Preparation of RSAC and nZVFe–RSAC
2.3. Procedures for Adsorption Experiments
2.4. Adsorption Kinetics, Isotherms, and Thermodynamics
3. Results and Discussion
3.1. Characterization of RS, RSAC, and nZVFe–RSAC Composite
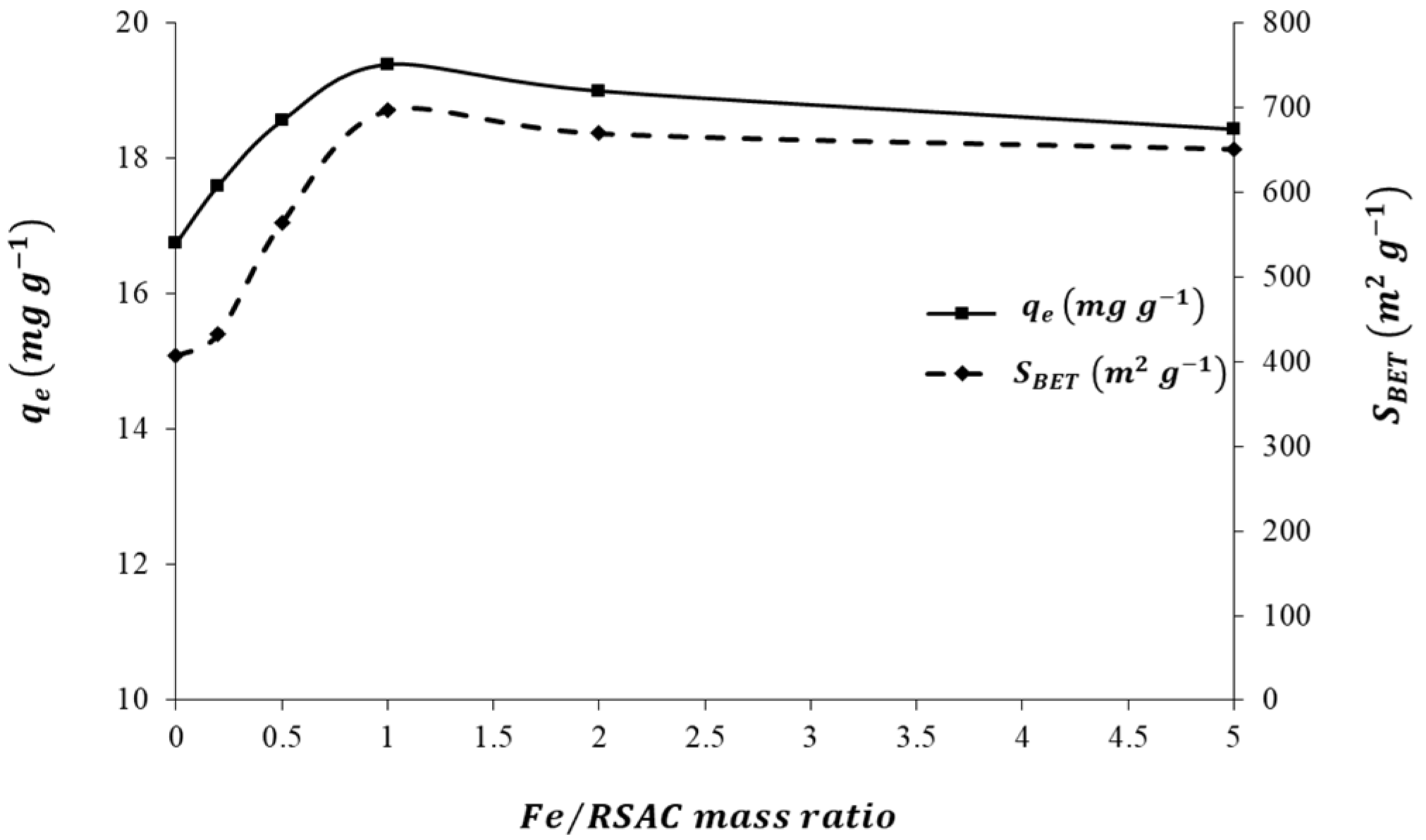
3.2. Effect of Fe Loading Quantity on Nanocomposite Adsorption Capacity
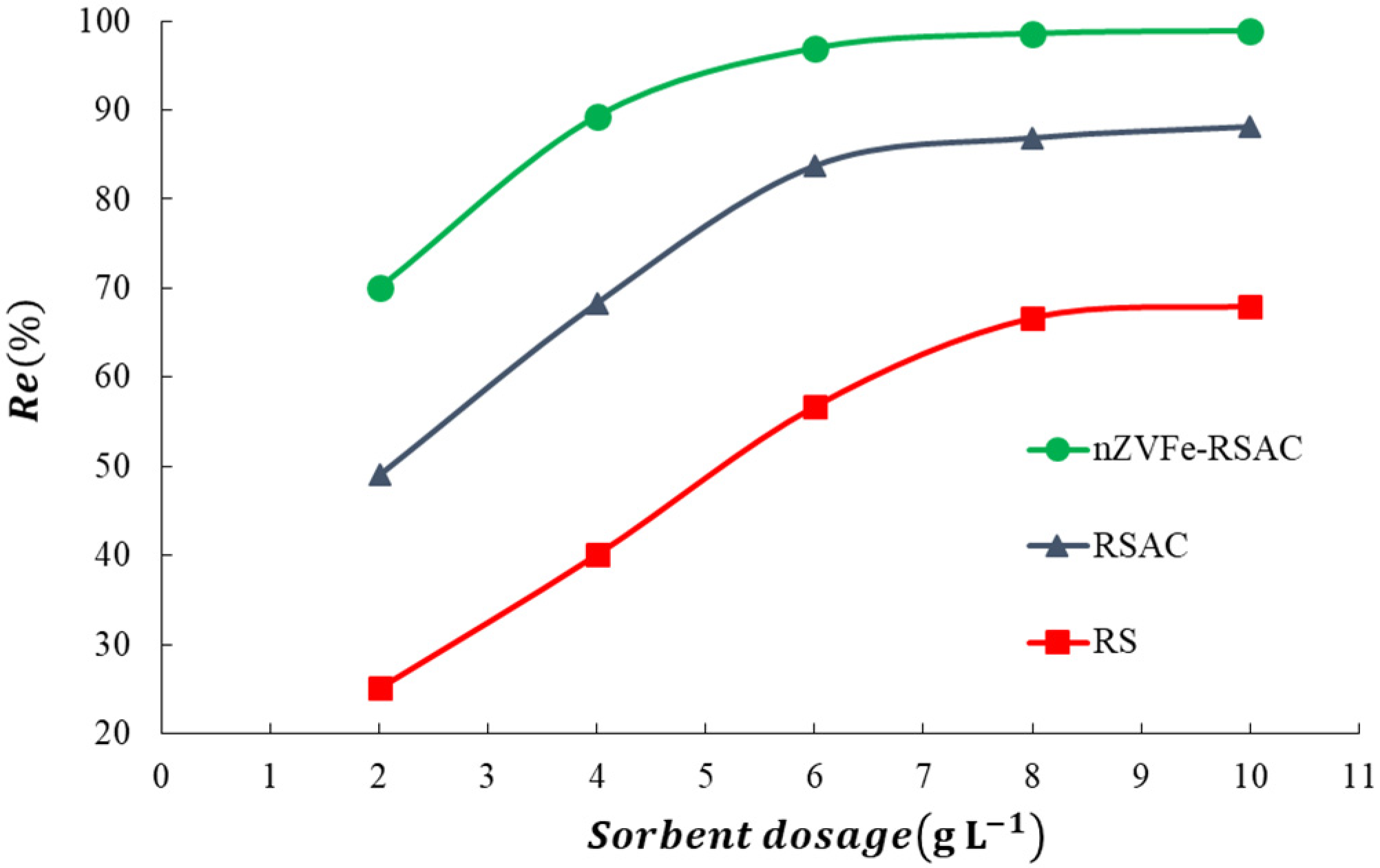
3.3. Effects of pH Value, Zeta Potential, and Absorbent Dosage on Experimental Results
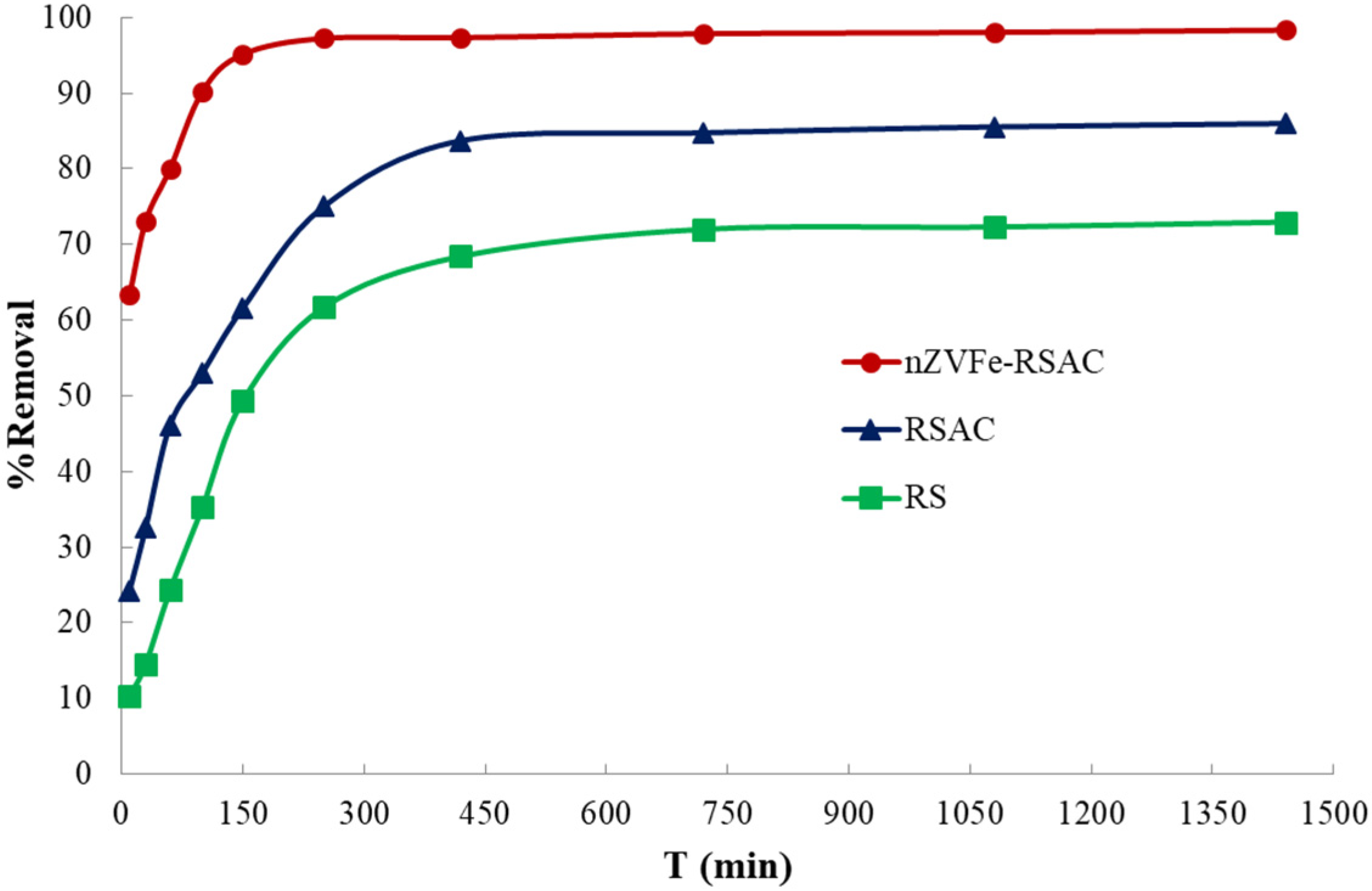
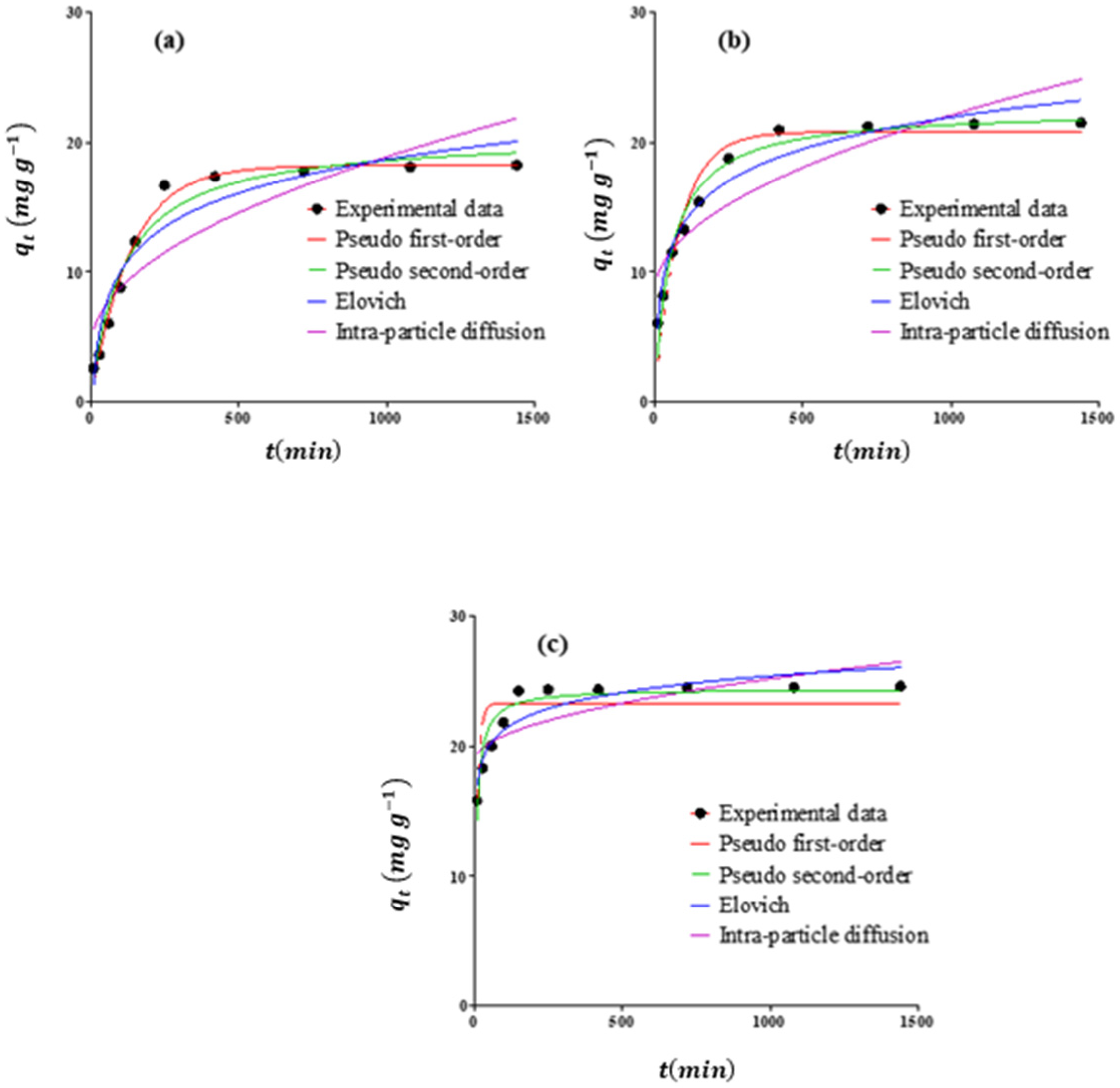
3.4. Kinetic Study and Contact-Time Effects
3.5. Initial Concentration of Lead and Isotherm Study
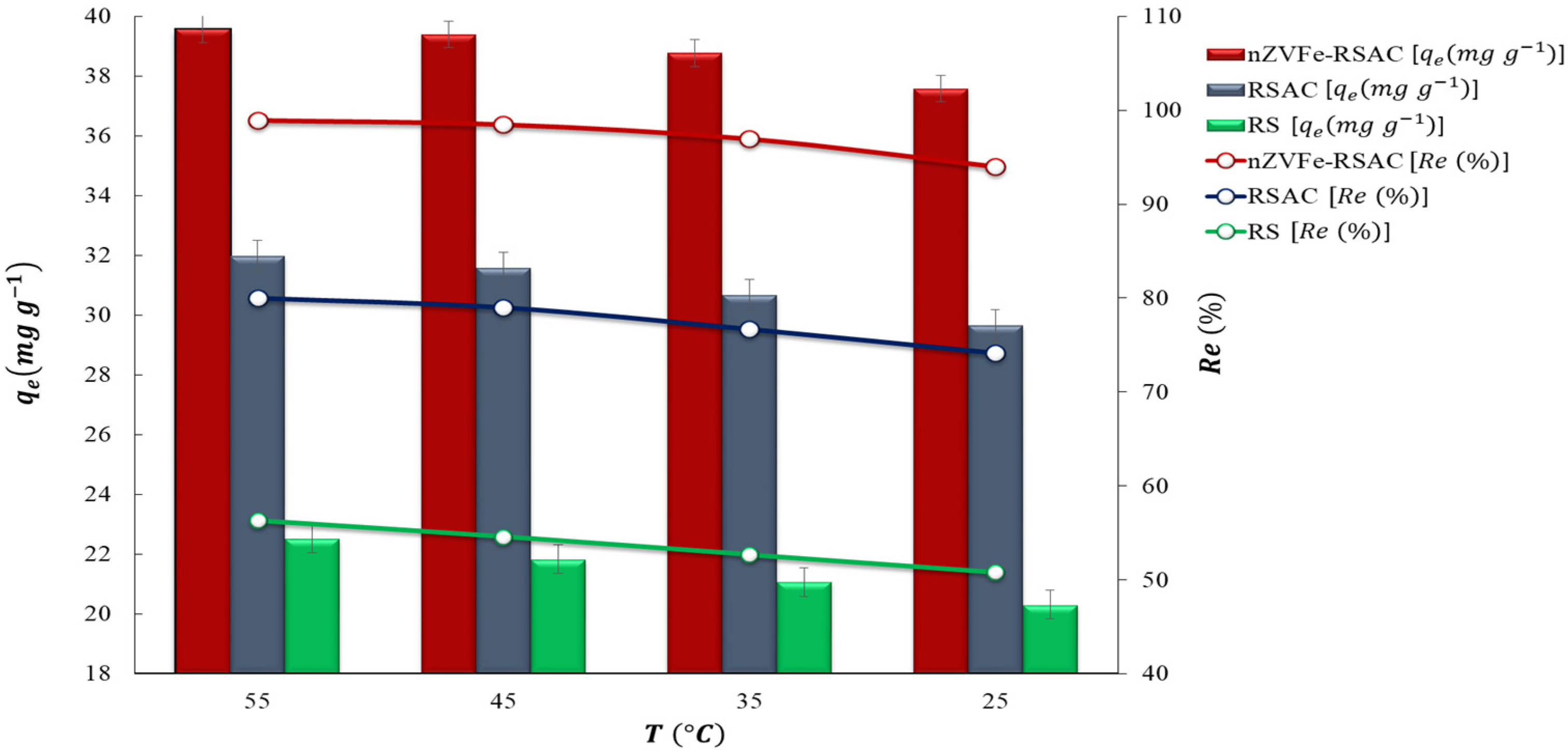
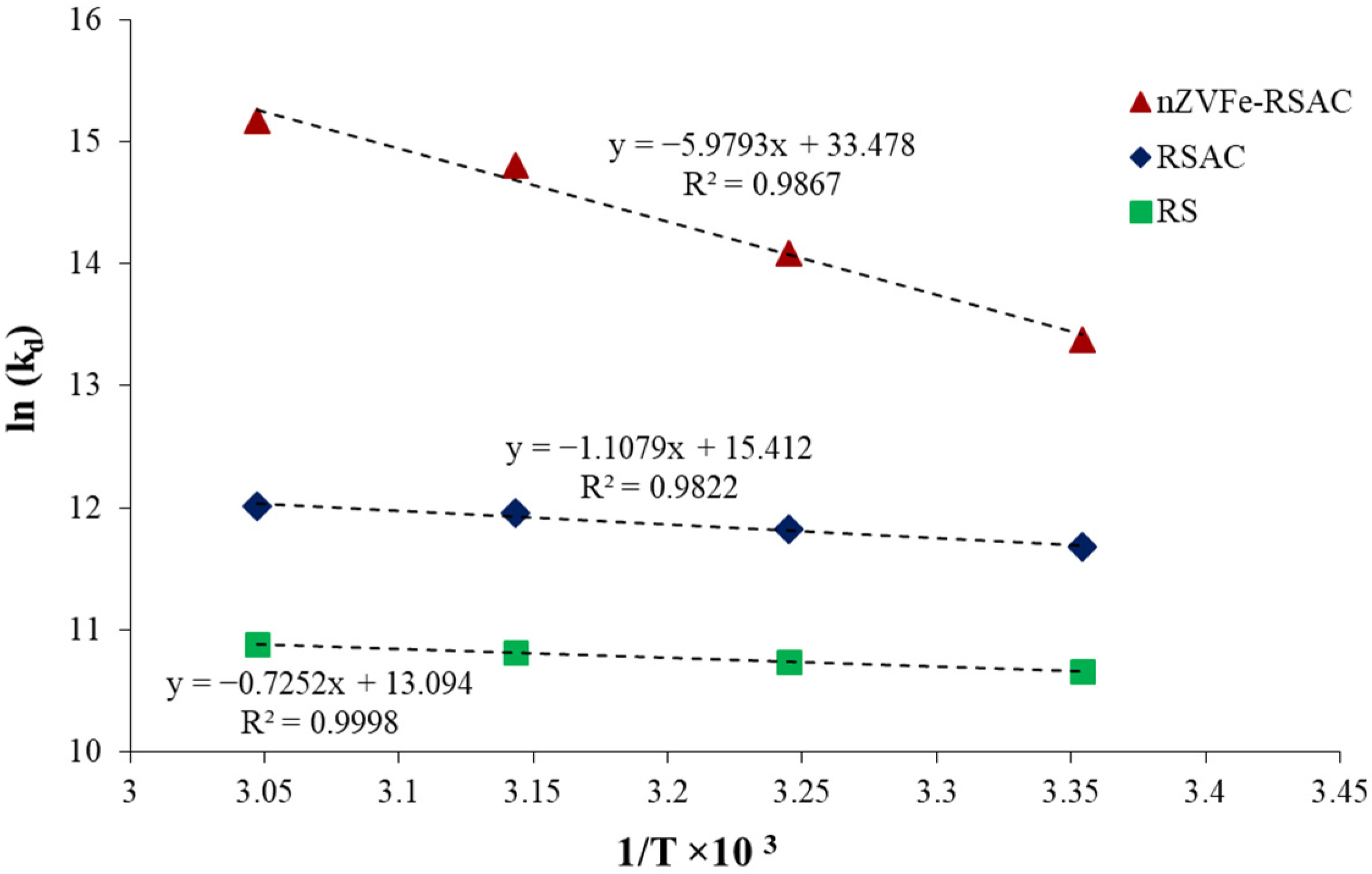
3.6. Effect of Temperature and Thermodynamic Study
3.7. Effect of Competing Ions
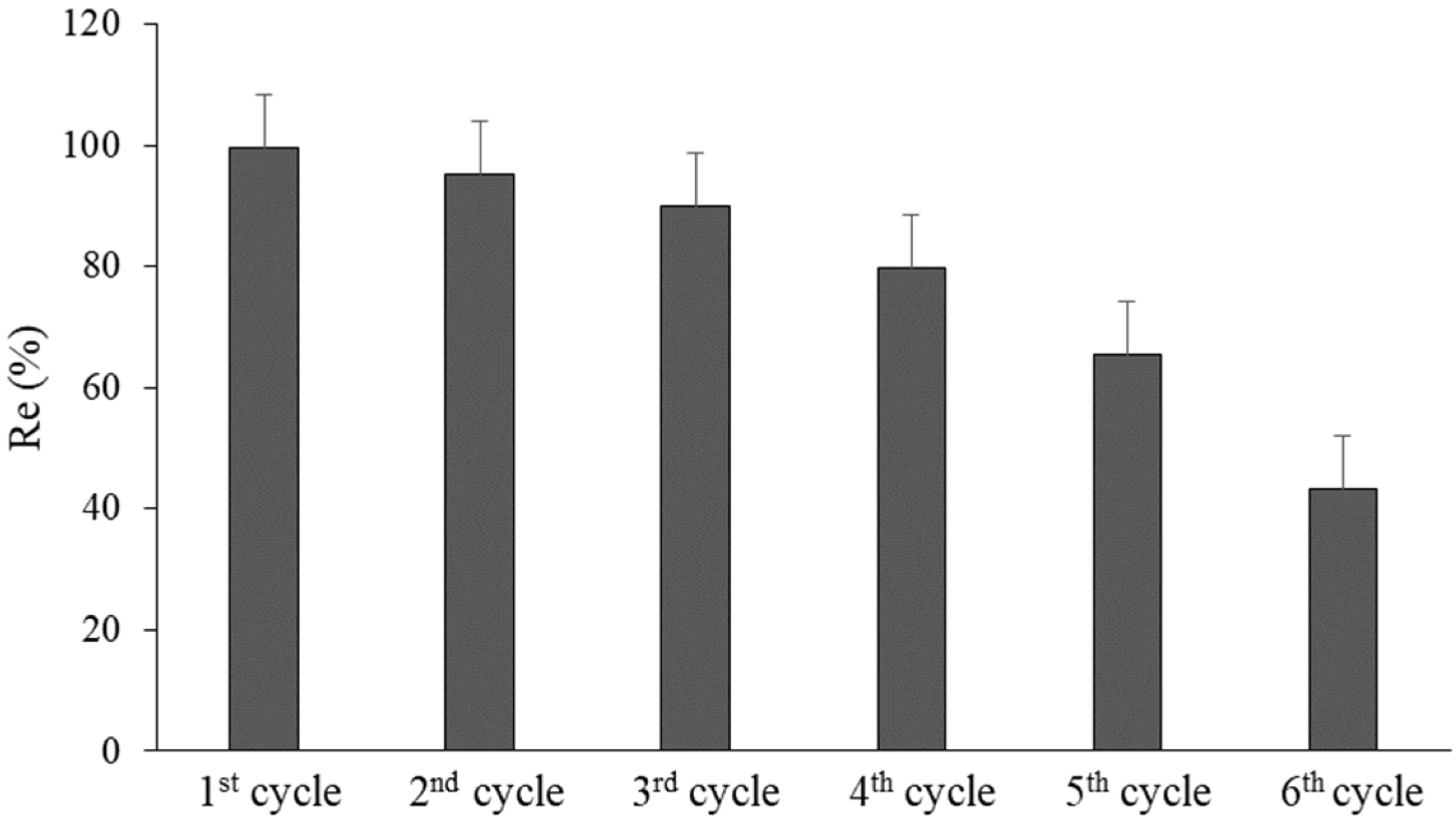
3.8. Recyclability and Reuse of nZVFe–RSAC
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, W.; Zheng, J.; Ou, X.; Liu, X.; Song, Y.; Tian, C.; Rong, W.; Shi, Z.; Dang, Z.; Lin, Z. Effective extraction of Cr (VI) from hazardous gypsum sludge via controlling the phase transformation and chromium species. Environ. Sci. Technol. 2018, 52, 13336–13342. [Google Scholar] [CrossRef] [PubMed]
- Lajayer, B.A.; Najafi, N.; Moghiseh, E.; Mosaferi, M.; Hadian, J. Micronutrient and heavy metal concentrations in basil plant cultivated on irradiated and non-irradiated sewage sludge-treated soil and evaluation of human health risk. Regul. Toxicol. Pharmacol. 2019, 104, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Shahi Khalaf Ansar, B.; Kavusi, E.; Dehghanian, Z.; Pandey, J.; Asgari Lajayer, B.; Price, G.W.; Astatkie, T. Removal of organic and inorganic contaminants from the air, soil, and water by algae. Environ. Sci. Pollut. Res. 2022, 1–29. [Google Scholar] [CrossRef]
- Nobaharan, K.; Abtahi, A.; Asgari Lajayer, B.; van Hullebusch, E.D. Effects of biochar dose on cadmium accumulation in spinach and its fractionation in a calcareous soil. Arab. J. Geosci. 2022, 15, 336. [Google Scholar] [CrossRef]
- Chanthapon, N.; Sarkar, S.; Kidkhunthod, P.; Padungthon, S. Lead removal by a reusable gel cation exchange resin containing nano-scale zero valent iron. Chem. Eng. J. 2018, 331, 545–555. [Google Scholar] [CrossRef]
- Lajayer, B.A.; Ghorbanpour, M.; Nikabadi, S. Heavy metals in contaminated environment: Destiny of secondary metabolite biosynthesis, oxidative status and phytoextraction in medicinal plants. Ecotoxicol. Environ. Saf. 2017, 145, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Asgari Lajayer, B.; Khadem Moghadam, N.; Maghsoodi, M.R.; Ghorbanpour, M.; Kariman, K. Phytoextraction of heavy metals from contaminated soil, water and atmosphere using ornamental plants: Mechanisms and efficiency improvement strategies. Environ. Sci. Pollut. Res. 2019, 26, 8468–8484. [Google Scholar] [CrossRef] [PubMed]
- Pei, K.; Liu, T. Enhanced Cr (VI) removal with Pb (II) presence by Fe2+-activated persulfate and zero-valent iron system. Environ. Technol. 2022, 1–15. [Google Scholar] [CrossRef]
- Mandal, S.; Calderon, J.; Marpu, S.B.; Omary, M.A.; Shi, S.Q. Mesoporous activated carbon as a green adsorbent for the removal of heavy metals and Congo red: Characterization, adsorption kinetics, and isotherm studies. J. Contam. Hydrol. 2021, 243, 103869. [Google Scholar] [CrossRef]
- Ren, J.; Zheng, L.; Su, Y.; Meng, P.; Zhou, Q.; Zeng, H.; Zhang, T.; Yu, H. Competitive adsorption of Cd (II), Pb (II) and Cu (II) ions from acid mine drainage with zero-valent iron/phosphoric titanium dioxide: XPS qualitative analyses and DFT quantitative calculations. Chem. Eng. J. 2022, 445, 136778. [Google Scholar] [CrossRef]
- Asgari Lajayer, B.; Najafi, N.; Moghiseh, E.; Mosaferi, M.; Hadian, J. Removal of heavy metals (Cu2+ and Cd2+) from effluent using gamma irradiation, titanium dioxide nanoparticles and methanol. J. Nanostructure Chem. 2018, 8, 483–496. [Google Scholar] [CrossRef] [Green Version]
- Heidarpour, A.; Aliasgharzad, N.; Khoshmanzar, E.; Lajayer, B.A. Bio-removal of Zn from contaminated water by using green algae isolates. Environ. Technol. Innov. 2019, 16, 100464. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, X.; Ni, S.-Q.; Zhuang, X.; Lee, T. Nano zero-valent iron improves anammox activity by promoting the activity of quorum sensing system. Water Res. 2021, 202, 117491. [Google Scholar] [CrossRef] [PubMed]
- Danila, V.; Kumpiene, J.; Kasiuliene, A.; Vasarevičius, S. Immobilisation of metal (loid) s in two contaminated soils using micro and nano zerovalent iron particles: Evaluating the long-term stability. Chemosphere 2020, 248, 126054. [Google Scholar] [CrossRef]
- Fajardo, C.; Martín, M.; Nande, M.; Botías, P.; García-Cantalejo, J.; Mengs, G.; Costa, G. Ecotoxicogenomic analysis of stress induced on Caenorhabditis elegans in heavy metal contaminated soil after nZVI treatment. Chemosphere 2020, 254, 126909. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Wang, X.; Khan, A.; Wang, P.; Liu, Y.; Alsaedi, A.; Hayat, T.; Wang, X. Environmental remediation and application of nanoscale zero-valent iron and its composites for the removal of heavy metal ions: A review. Environ. Sci. Technol. 2016, 50, 7290–7304. [Google Scholar] [CrossRef]
- Mortazavian, S.; An, H.; Chun, D.; Moon, J. Activated carbon impregnated by zero-valent iron nanoparticles (AC/nZVI) optimized for simultaneous adsorption and reduction of aqueous hexavalent chromium: Material characterizations and kinetic studies. Chem. Eng. J. 2018, 353, 781–795. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, W.; Sheng, G.; Li, J.; Dong, H.; Chen, Y.; Zhu, L. Synergetic effect of a pillared bentonite support on SE (VI) removal by nanoscale zero valent iron. Appl. Catal. B Environ. 2015, 174, 329–335. [Google Scholar] [CrossRef]
- Liu, M.; Wang, Y.; Chen, L.; Zhang, Y.; Lin, Z. Mg (OH) 2 supported nanoscale zero valent iron enhancing the removal of Pb (II) from aqueous solution. ACS Appl. Mater. Interfaces 2015, 7, 7961–7969. [Google Scholar] [CrossRef]
- Liu, T.; Wang, Z.-L.; Yan, X.; Zhang, B. Removal of mercury (II) and chromium (VI) from wastewater using a new and effective composite: Pumice-supported nanoscale zero-valent iron. Chem. Eng. J. 2014, 245, 34–40. [Google Scholar] [CrossRef]
- Habish, A.J.; Lazarević, S.; Janković-Častvan, I.; Jokić, B.; Kovač, J.; Rogan, J.; Janaćković, Đ.; Petrović, R. Nanoscale zerovalent iron (nZVI) supported by natural and acid-activated sepiolites: The effect of the nZVI/support ratio on the composite properties and Cd2+ adsorption. Environ. Sci. Pollut. Res. 2017, 24, 628–643. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Yang, Y.; Xu, Z.; Zhang, X.; Guo, X.; Bi, D. The removal of chromium (VI) and lead (II) from groundwater using sepiolite-supported nanoscale zero-valent iron (S-NZVI). Chemosphere 2015, 138, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xu, Z.; Ding, G.; Wang, X.; Zhao, M.; Ho, S.S.H.; Li, Y. Comprehensive study on the removal of chromate from aqueous solution by synthesized kaolin supported nanoscale zero-valent iron. Desalin. Water Treat. 2016, 57, 5065–5078. [Google Scholar] [CrossRef]
- Bhowmick, S.; Chakraborty, S.; Mondal, P.; Van Renterghem, W.; Van den Berghe, S.; Roman-Ross, G.; Chatterjee, D.; Iglesias, M. Montmorillonite-supported nanoscale zero-valent iron for removal of arsenic from aqueous solution: Kinetics and mechanism. Chem. Eng. J. 2014, 243, 14–23. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, G.; Wang, X.; Li, S.; Liu, Y.; Yang, G. Removal of hexavalent chromium by bentonite supported organosolv lignin-stabilized zero-valent iron nanoparticles from wastewater. J. Clean. Prod. 2020, 267, 122009. [Google Scholar] [CrossRef]
- Diao, Z.-H.; Xu, X.-R.; Chen, H.; Jiang, D.; Yang, Y.-X.; Kong, L.-J.; Sun, Y.-X.; Hu, Y.-X.; Hao, Q.-W.; Liu, L. Simultaneous removal of Cr (VI) and phenol by persulfate activated with bentonite-supported nanoscale zero-valent iron: Reactivity and mechanism. J. Hazard. Mater. 2016, 316, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Diao, Z.-H.; Xu, X.-R.; Jiang, D.; Kong, L.-J.; Sun, Y.-X.; Hu, Y.-X.; Hao, Q.-W.; Chen, H. Bentonite-supported nanoscale zero-valent iron/persulfate system for the simultaneous removal of Cr (VI) and phenol from aqueous solutions. Chem. Eng. J. 2016, 302, 213–222. [Google Scholar] [CrossRef]
- Kong, X.; Han, Z.; Zhang, W.; Song, L.; Li, H. Synthesis of zeolite-supported microscale zero-valent iron for the removal of Cr6+ and Cd2+ from aqueous solution. J. Environ. Manag. 2016, 169, 84–90. [Google Scholar] [CrossRef]
- Kim, S.A.; Kamala-Kannan, S.; Lee, K.-J.; Park, Y.-J.; Shea, P.J.; Lee, W.-H.; Kim, H.-M.; Oh, B.-T. Removal of Pb (II) from aqueous solution by a zeolite–nanoscale zero-valent iron composite. Chem. Eng. J. 2013, 217, 54–60. [Google Scholar] [CrossRef] [Green Version]
- Sepehri, S.; Heidarpour, M.; Abedi-Koupai, J. Nitrate removal from aqueous solution using natural zeolite-supported zero-valent iron nanoparticles. Soil Water Res. 2014, 9, 224–232. [Google Scholar] [CrossRef]
- Su, H.; Fang, Z.; Tsang, P.E.; Zheng, L.; Cheng, W.; Fang, J.; Zhao, D. Remediation of hexavalent chromium contaminated soil by biochar-supported zero-valent iron nanoparticles. J. Hazard. Mater. 2016, 318, 533–540. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, F.; Hoekman, S.K.; Liu, T.; Gai, C.; Peng, N. Homogeneously dispersed zerovalent iron nanoparticles supported on hydrochar-derived porous carbon: Simple, in situ synthesis and use for dechlorination of PCBs. ACS Sustain. Chem. Eng. 2016, 4, 3261–3267. [Google Scholar] [CrossRef]
- Liu, D.; Liu, Z.; Wang, C.; Lai, Y. Removal of uranium (VI) from aqueous solution using nanoscale zero-valent iron supported on activated charcoal. J. Radioanal. Nucl. Chem. 2016, 310, 1131–1137. [Google Scholar] [CrossRef]
- Sepehri, S.; Nakhjavanimoghaddam, M. Batch removal of aqueous nitrate ions using an effective nano-biocomposite. Glob. Nest J. 2019, 21, 265–275. [Google Scholar] [CrossRef] [Green Version]
- Arshadi, M.; Abdolmaleki, M.; Mousavinia, F.; Foroughifard, S.; Karimzadeh, A. Nano modification of NZVI with an aquatic plant Azolla filiculoides to remove Pb (II) and Hg (II) from water: Aging time and mechanism study. J. Colloid Interface Sci. 2017, 486, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Guerrero, A.; Cortés-Martínez, R.; Alfaro-Cuevas-Villanueva, R.; Rivera-Muñoz, E.M.; Huirache-Acuña, R. Cd (II) and Pb (II) adsorption using a composite obtained from moringa oleifera lam. Cellulose nanofibrils impregnated with iron nanoparticles. Water 2021, 13, 89. [Google Scholar] [CrossRef]
- Yang, F.; He, Y.; Sun, S.; Chang, Y.; Zha, F.; Lei, Z. Walnut shell supported nanoscale Fe0 for the removal of Cu (II) and Ni (II) ions from water. J. Appl. Polym. Sci. 2016, 133, 43304–43309. [Google Scholar] [CrossRef]
- Lee, C.-S.; Gong, J.; Huong, C.V.; Oh, D.-S.; Chang, Y.-S. Macroporous alginate substrate-bound growth of Fe0 nanoparticles with high redox activities for nitrate removal from aqueous solutions. Chem. Eng. J. 2016, 298, 206–213. [Google Scholar] [CrossRef]
- Huang, J.-F.; Li, Y.-T.; Wu, J.-H.; Cao, P.-Y.; Liu, Y.-L.; Jiang, G.-B. Floatable, macroporous structured alginate sphere supporting iron nanoparticles used for emergent Cr (VI) spill treatment. Carbohydr. Polym. 2016, 146, 115–122. [Google Scholar] [CrossRef]
- Toli, A.; Chalastara, K.; Mystrioti, C.; Xenidis, A.; Papassiopi, N. Incorporation of zero valent iron nanoparticles in the matrix of cationic resin beads for the remediation of Cr (VI) contaminated waters. Environ. Pollut. 2016, 214, 419–429. [Google Scholar] [CrossRef]
- Liu, F.; Shan, C.; Zhang, X.; Zhang, Y.; Zhang, W.; Pan, B. Enhanced removal of EDTA-chelated Cu (II) by polymeric anion-exchanger supported nanoscale zero-valent iron. J. Hazard. Mater. 2017, 321, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, S.; Li, J.; Xu, J.; Wang, X. Fabrication of Fe/Fe 3 C@ porous carbon sheets from biomass and their application for simultaneous reduction and adsorption of uranium (VI) from solution. Inorg. Chem. Front. 2014, 1, 641–648. [Google Scholar] [CrossRef]
- Shi, J.; Yi, S.; Long, C.; Li, A. Effect of Fe loading quantity on reduction reactivity of nano zero-valent iron supported on chelating resin. Front. Environ. Sci. Eng. 2015, 9, 840–849. [Google Scholar] [CrossRef]
- Hu, B.; Chen, G.; Jin, C.; Hu, J.; Huang, C.; Sheng, J.; Sheng, G.; Ma, J.; Huang, Y. Macroscopic and spectroscopic studies of the enhanced scavenging of Cr (VI) and Se (VI) from water by titanate nanotube anchored nanoscale zero-valent iron. J. Hazard. Mater. 2017, 336, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Chen, C.; Chen, Y.; Li, J.; Wang, D.; Wang, X.; Hu, W. Competitive adsorption of PbII, NiII, and SrII ions on graphene oxides: A combined experimental and theoretical study. ChemPlusChem 2015, 80, 480–484. [Google Scholar] [CrossRef]
- Sheng, G.; Hu, J.; Li, H.; Li, J.; Huang, Y. Enhanced sequestration of Cr (VI) by nanoscale zero-valent iron supported on layered double hydroxide by batch and XAFS study. Chemosphere 2016, 148, 227–232. [Google Scholar] [CrossRef]
- Sheng, G.; Tang, Y.; Linghu, W.; Wang, L.; Li, J.; Li, H.; Wang, X.; Huang, Y. Enhanced immobilization of ReO4− by nanoscale zerovalent iron supported on layered double hydroxide via an advanced XAFS approach: Implications for TcO4− sequestration. Appl. Catal. B Environ. 2016, 192, 268–276. [Google Scholar] [CrossRef]
- Gosu, V.; Gurjar, B.R.; Surampalli, R.Y.; Zhang, T.C. Treatment of pyridine-bearing wastewater by nano zero-valent iron supported on activated carbon derived from agricultural waste. Desalin. Water Treat. 2016, 57, 6250–6260. [Google Scholar] [CrossRef]
- Busch, J.; Meißner, T.; Potthoff, A.; Bleyl, S.; Georgi, A.; Mackenzie, K.; Trabitzsch, R.; Werban, U.; Oswald, S.E. A field investigation on transport of carbon-supported nanoscale zero-valent iron (nZVI) in groundwater. J. Contam. Hydrol. 2015, 181, 59–68. [Google Scholar] [CrossRef]
- Fan, M.; Li, T.; Hu, J.; Cao, R.; Wu, Q.; Wei, X.; Li, L.; Shi, X.; Ruan, W. Synthesis and characterization of reduced graphene oxide-supported nanoscale zero-valent iron (nZVI/rGO) composites used for Pb (II) removal. Materials 2016, 9, 687. [Google Scholar] [CrossRef]
- Fan, H.; Ren, H.; Ma, X.; Zhou, S.; Huang, J.; Jiao, W.; Qi, G.; Liu, Y. High-gravity continuous preparation of chitosan-stabilized nanoscale zero-valent iron towards Cr (VI) removal. Chem. Eng. J. 2020, 390, 124639. [Google Scholar] [CrossRef]
- Sheng, G.; Alsaedi, A.; Shammakh, W.; Monaquel, S.; Sheng, J.; Wang, X.; Li, H.; Huang, Y. Enhanced sequestration of selenite in water by nanoscale zero valent iron immobilization on carbon nanotubes by a combined batch, XPS and XAFS investigation. Carbon 2016, 99, 123–130. [Google Scholar] [CrossRef]
- Siciliano, A. Removal of Cr (VI) from water using a new reactive material: Magnesium Oxide supported nanoscale zero-valent iron. Materials 2016, 9, 666. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, Q.; Li, G.; Zhang, S.; Song, J.; Yang, F. High-dispersion zero-valent iron particles stabilized by artificial humic acid for lead ion removal. J. Hazard. Mater. 2020, 383, 121170. [Google Scholar] [CrossRef]
- Wang, X.; Liang, D.; Wang, Y.; Peijnenburg, W.J.; Monikh, F.A.; Zhao, X.; Dong, Z.; Fan, W. A critical review on the biological impact of natural organic matter on nanomaterials in the aquatic environment. Carbon Res. 2022, 1, 13. [Google Scholar] [CrossRef]
- Yan, B.; Feng, L.; Zheng, J.; Zhang, Q.; Jiang, S.; Zhang, C.; Ding, Y.; Han, J.; Chen, W.; He, S. High performance supercapacitors based on wood-derived thick carbon electrodes synthesized via green activation process. Inorg. Chem. Front. 2022, 9, 6108–6123. [Google Scholar] [CrossRef]
- Yan, B.; Zheng, J.; Feng, L.; Du, C.; Jian, S.; Yang, W.; Wu, Y.A.; Jiang, S.; He, S.; Chen, W. Wood-derived biochar as thick electrodes for high-rate performance supercapacitors. Biochar 2022, 4, 50. [Google Scholar] [CrossRef]
- Obey, G.; Adelaide, M.; Ramaraj, R. Biochar derived from non-customized matamba fruit shell as an adsorbent for wastewater treatment. J. Bioresour. Bioprod. 2022, 7, 109–115. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, Z.; Xu, L.; Buyong, F.; Chay, T.C.; Li, Z.; Cai, Y.; Hu, B.; Zhu, Y.; Wang, X. Modified biochar: Synthesis and mechanism for removal of environmental heavy metals. Carbon Res. 2022, 1, 8. [Google Scholar] [CrossRef]
- Awang, N.A.; Wan Salleh, W.N.; Aziz, F.; Yusof, N.; Ismail, A.F. A review on preparation, surface enhancement and adsorption mechanism of biochar-supported nano zero-valent iron adsorbent for hazardous heavy metals. J. Chem. Technol. Biotechnol. 2022, 98, 22–44. [Google Scholar] [CrossRef]
- Ahmad, S.; Liu, X.; Tang, J.; Zhang, S. Biochar-supported nanosized zero-valent iron (nZVI/BC) composites for removal of nitro and chlorinated contaminants. Chem. Eng. J. 2021, 431, 133187. [Google Scholar] [CrossRef]
- FAO. FAOSTAT Statistical Database; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Motlagh, E.K.; Asasian-Kolur, N.; Sharifian, S.; Pirbazari, A.E. Sustainable rice straw conversion into activated carbon and nano-silica using carbonization-extraction process. Biomass Bioenergy 2021, 144, 105917. [Google Scholar] [CrossRef]
- Jiang, S.-F.; Ling, L.-L.; Chen, W.-J.; Liu, W.-J.; Li, D.-C.; Jiang, H. High efficient removal of bisphenol A in a peroxymonosulfate/iron functionalized biochar system: Mechanistic elucidation and quantification of the contributors. Chem. Eng. J. 2019, 359, 572–583. [Google Scholar] [CrossRef]
- Ruiz-Torres, C.A.; Araujo-Martínez, R.F.; Martínez-Castañón, G.A.; Morales-Sánchez, J.E.; Guajardo-Pacheco, J.M.; González-Hernández, J.; Lee, T.-J.; Shin, H.-S.; Hwang, Y.; Ruiz, F. Preparation of air stable nanoscale zero valent iron functionalized by ethylene glycol without inert condition. Chem. Eng. J. 2018, 336, 112–122. [Google Scholar] [CrossRef]
- Charoensook, K.; Huang, C.-L.; Tai, H.-C.; Lanjapalli, V.V.K.; Chiang, L.-M.; Hosseini, S.; Lin, Y.-T.; Li, Y.-Y. Preparation of porous nitrogen-doped activated carbon derived from rice straw for high-performance supercapacitor application. J. Taiwan Inst. Chem. Eng. 2021, 120, 246–256. [Google Scholar] [CrossRef]
- Momčilović, M.; Purenović, M.; Bojić, A.; Zarubica, A.; Ranđelović, M. Removal of lead (II) ions from aqueous solutions by adsorption onto pine cone activated carbon. Desalination 2011, 276, 53–59. [Google Scholar] [CrossRef]
- Üzüm, Ç.; Shahwan, T.; Eroğlu, A.E.; Hallam, K.R.; Scott, T.B.; Lieberwirth, I. Synthesis and characterization of kaolinite-supported zero-valent iron nanoparticles and their application for the removal of aqueous Cu2+ and Co2+ ions. Appl. Clay Sci. 2009, 43, 172–181. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.-Y.; Chen, S.S.; Liao, W.; Wang, W.; Jang, M.-F.; Chen, W.-H.; Ahamad, T.; Alshehri, S.M.; Hou, C.-H.; Lin, K.-S. Assessment of agricultural waste-derived activated carbon in multiple applications. Environ. Res. 2020, 191, 110176. [Google Scholar] [CrossRef]
- Rajahmundry, G.K.; Garlapati, C.; Kumar, P.S.; Alwi, R.S.; Vo, D.-V.N. Statistical analysis of adsorption isotherm models and its appropriate selection. Chemosphere 2021, 276, 130176. [Google Scholar] [CrossRef]
- Menezes, J.M.C.; da Silva Bento, A.M.; da Silva, J.H.; de Paula Filho, F.J.; da Costa, J.G.M.; Coutinho, H.D.M.; Teixeira, R.N.P. Equilibrium, kinetics and thermodynamics of lead (II) adsorption in bioadsorvent composed by Caryocar coriaceum Wittm barks. Chemosphere 2020, 261, 128144. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Fosso-Kankeu, E.; Spiro, M.; Waanders, F.; Kumar, N.; Ray, S.S.; Kim, J.; Kang, M. Equilibrium, kinetic, and thermodynamic studies of lead ion adsorption from mine wastewater onto MoS2-clinoptilolite composite. Mater. Today Chem. 2020, 18, 100376. [Google Scholar] [CrossRef]
- Qin, C.; Chen, Y.; Gao, J.-M. Manufacture and characterization of activated carbon from marigold straw (Tagetes erecta L) by H3PO4 chemical activation. Mater. Lett. 2014, 135, 123–126. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Bhaduri, D.; Adak, T.; Munda, S.; Satapathy, B.; Dash, P.; Padhy, S.; Pattanayak, A.; Routray, S.; Chakraborti, M. Characterization of rice straw from major cultivars for best alternative industrial uses to cutoff the menace of straw burning. Ind. Crops Prod. 2020, 143, 111919. [Google Scholar] [CrossRef]
- Birks, L.; Friedman, H. Particle size determination from X-ray line broadening. J. Appl. Phys. 1946, 17, 687–692. [Google Scholar] [CrossRef]
- Li, S.; Yang, F.; Zhang, Y.; Lan, Y.; Cheng, K. Performance of lead ion removal by the three-dimensional carbon foam supported nanoscale zero-valent iron composite. J. Clean. Prod. 2021, 294, 125350. [Google Scholar] [CrossRef]
- Bae, S.; Collins, R.N.; Waite, T.D.; Hanna, K. Advances in surface passivation of nanoscale zerovalent iron: A critical review. Environ. Sci. Technol. 2018, 52, 12010–12025. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, S.; Lu, X.-Q.; Chen, Z.-l. Removal of Pb (II) from water using synthesized kaolin supported nanoscale zero-valent iron. Chem. Eng. J. 2010, 163, 243–248. [Google Scholar] [CrossRef]
- Li, W.; Brunner, P.; Franssen, H.-J.H.; Li, Z.; Wang, Z.; Zhang, Z.; Wang, W. Potential evaporation dynamics over saturated bare soil and an open water surface. J. Hydrol. 2020, 590, 125140. [Google Scholar] [CrossRef]
- Gao, C.; Wang, X.-L.; An, Q.-D.; Xiao, Z.-Y.; Zhai, S.-R. Synergistic preparation of modified alginate aerogel with melamine/chitosan for efficiently selective adsorption of lead ions. Carbohydr. Polym. 2021, 256, 117564. [Google Scholar] [CrossRef]
- Zhang, W.-X. Nanoscale iron particles for environmental remediation: An overview. J. Nanopart. Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Arshadi, M.; Soleymanzadeh, M.; Salvacion, J.; SalimiVahid, F. Nanoscale zero-valent iron (NZVI) supported on sineguelas waste for Pb (II) removal from aqueous solution: Kinetics, thermodynamic and mechanism. J. Colloid Interface Sci. 2014, 426, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.C.; Rubner, M.F.; Cohen, R.E. Polyelectrolyte multilayer nanoreactors for preparing silver nanoparticle composites: Controlling metal concentration and nanoparticle size. Langmuir 2002, 18, 3370–3375. [Google Scholar] [CrossRef]
- Jalu, R.G.; Chamada, T.A.; Kasirajan, R. Calcium oxide nanoparticles synthesis from hen eggshells for removal of lead (Pb (II)) from aqueous solution. Environ. Chall. 2021, 4, 100193. [Google Scholar] [CrossRef]
- Kavand, M.; Eslami, P.; Razeh, L. The adsorption of cadmium and lead ions from the synthesis wastewater with the activated carbon: Optimization of the single and binary systems. J. Water Process Eng. 2020, 34, 101151. [Google Scholar] [CrossRef]
- Tabesh, S.; Davar, F.; Loghman-Estarki, M.R. Preparation of γ-Al2O3 nanoparticles using modified sol-gel method and its use for the adsorption of lead and cadmium ions. J. Alloys Compd. 2018, 730, 441–449. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Azizian, S.; Douven, S. Implications of apparent pseudo-second-order adsorption kinetics onto cellulosic materials: A review. BioResources 2019, 14, 7582–7626. [Google Scholar] [CrossRef]
- Abo El-Reesh, G.Y.; Farghali, A.A.; Taha, M.; Mahmoud, R.K. Novel synthesis of Ni/Fe layered double hydroxides using urea and glycerol and their enhanced adsorption behavior for Cr (VI) removal. Sci. Rep. 2020, 10, 587. [Google Scholar] [CrossRef] [Green Version]
- Alqadami, A.A.; Naushad, M.; ALOthman, Z.A.; Alsuhybani, M.; Algamdi, M. Excellent adsorptive performance of a new nanocomposite for removal of toxic Pb (II) from aqueous environment: Adsorption mechanism and modeling analysis. J. Hazard. Mater. 2020, 389, 121896. [Google Scholar] [CrossRef]
- Patil, S.A.; Suryawanshi, U.P.; Harale, N.S.; Patil, S.K.; Vadiyar, M.M.; Luwang, M.N.; Anuse, M.A.; Kim, J.H.; Kolekar, S.S. Adsorption of toxic Pb (II) on activated carbon derived from agriculture waste (Mahogany fruit shell): Isotherm, kinetic and thermodynamic study. Int. J. Environ. Anal. Chem. 2020, 1–17. [Google Scholar] [CrossRef]
- Senol-Arslan, D. Isotherms, kinetics and thermodynamics of pb (ii) adsorption by crosslinked chitosan/sepiolite composite. Polym. Bull. 2022, 79, 3911–3928. [Google Scholar] [CrossRef]
- Liang, W.; Wang, G.; Peng, C.; Tan, J.; Wan, J.; Sun, P.; Li, Q.; Ji, X.; Zhang, Q.; Wu, Y. Recent advances of carbon-based nano zero valent iron for heavy metals remediation in soil and water: A critical review. J. Hazard. Mater. 2022, 426, 127993. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Chen, N.; Qin, J.; Yang, Y.; Feng, C.; Li, M.; Gao, Y. Biochar stabilized nano zero-valent iron and its removal performance and mechanism of pentavalent vanadium (V (V)). Colloids Surf. A Physicochem. Eng. Asp. 2020, 599, 124882. [Google Scholar] [CrossRef]
- Wang, W.; Hu, B.; Wang, C.; Liang, Z.; Cui, F.; Zhao, Z.; Yang, C. Cr (VI) removal by micron-scale iron-carbon composite induced by ball milling: The role of activated carbon. Chem. Eng. J. 2020, 389, 122633. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, S.; Sun, Y.; Cheng, K.; Li, J.; Tsang, D.C. Fabrication and characterization of hydrophilic corn stalk biochar-supported nanoscale zero-valent iron composites for efficient metal removal. Bioresour. Technol. 2018, 265, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Li, J.; Liu, J.; Qu, X.; Ma, H. Advances in continuous flow aerobic granular sludge: A review. Process Saf. Environ. Prot. 2022, 163, 27–35. [Google Scholar] [CrossRef]
- Nobaharan, K.; Bagheri Novair, S.; Asgari Lajayer, B.; van Hullebusch, E.D. Phosphorus removal from wastewater: The potential use of biochar and the key controlling factors. Water 2021, 13, 517. [Google Scholar] [CrossRef]
- Guan, Q.; Zeng, G.; Song, J.; Liu, C.; Wang, Z.; Wu, S. Ultrasonic power combined with seed materials for recovery of phosphorus from swine wastewater via struvite crystallization process. J. Environ. Manag. 2021, 293, 112961. [Google Scholar] [CrossRef]







| Structural Components (%) | Elemental Analysis (wt.%) | |||||||
|---|---|---|---|---|---|---|---|---|
| Cellulose | Hemicelluloses | Lignin | Extractives | Ash | Carbon | Oxygen | Hydrogen | Nitrogen |
| 39.25 | 29.18 | 25.42 | 5.89 | 2.83 | 39.65 | 45.63 | 8.21 | 3.14 |
| Bulk Density (kg m−3) | Iodine Number (mg g−1) | Elemental Analysis (wt.%) | ||||
|---|---|---|---|---|---|---|
| Ash | Carbon | Oxygen | Hydrogen | Nitrogen | ||
| 470 | 830 | 3.53 | 80.77 | 9.10 | 3.02 | 3.54 |
| Sample | Pore Volume (cm3 g−1) | Mean Pore Width (nm) | ||
|---|---|---|---|---|
| SBET (m2 g−1) | VT (cm−1 g−1) | VM (cm−1 g−1) | ||
| RS | 5.14 | 0.024 | - | - |
| RSAC | 406.87 | 0.384 | 0.062 | 2.7 |
| nZVFe–RSAC | 697.19 | 0.641 | 0.094 | 1.8 |
| Model | Parameters | Adsorbent | ||
|---|---|---|---|---|
| RS | RSAC | nZVFe–RSAC | ||
| (Exp) | 16.67 | 20.93 | 24.24 | |
| Pseudo-first order | (Cal) | 18.21 | 20.78 | 23.28 |
| 0.008 | 0.012 | 0.091 | ||
| 0.982 | 0.916 | 0.604 | ||
| SEE | 0.070 | 0.149 | 0.457 | |
| Pseudo-second order | (Cal) | 20.62 | 22.51 | 24.39 |
| 0.0004 | 0.001 | 0.006 | ||
| 0.17 | 0.51 | 3.57 | ||
| 0.968 | 0.964 | 0.896 | ||
| SEE | 0.044 | 0.058 | 0.153 | |
| Intraparticle diffusion | 0.467 | 0.435 | 0.203 | |
| −4.089 | −8.31 | −18.73 | ||
| 0.770 | 0.793 | 0.586 | ||
| SEE | 0.287 | 0.250 | 0.673 | |
| Elovich | 0.530 | 0.283 | 2.182 | |
| 0.265 | 1.75 | 0.552 | ||
| 0.919 | 0.955 | 0.852 | ||
| SEE | 0.139 | 0.121 | 0.159 | |
| Model | Parameters | Adsorbent | ||
|---|---|---|---|---|
| RS | RSAC | nZVFe–RSAC | ||
| Langmuir | 23.87 | 65.04 | 124.4 | |
| 0.039 | 0.017 | 0.040 | ||
| 0.027–0.959 | 0.033–0.966 | 0.38–0.989 | ||
| 0.989 | 0.984 | 0.989 | ||
| SEE | 0.17 | 0.29 | 0.20 | |
| Freundlich | 5.70 | 7.91 | 19.79 | |
| n | 4.432 | 3.071 | 3.178 | |
| 0.832 | 0.928 | 0.964 | ||
| SEE | 0.60 | 0.49 | 0.36 | |
| Langmuir–Freundlich | 0.026 | 0.024 | 0.065 | |
| 23.27 | 67.83 | 140.8 | ||
| n | 0.874 | 1.118 | 1.322 | |
| 0.995 | 0.987 | 0.996 | ||
| SEE | 0.03 | 0.24 | 0.05 | |
| Redlich–Peterson | a | 0.759 | 0.990 | 6.14 |
| b | 0.021 | 0.010 | 0.078 | |
| g | 1.068 | 1.059 | 0.921 | |
| 0.991 | 0.981 | 0.985 | ||
| SEE | 0.05 | 0.31 | 0.27 | |
| Thermodynamic Parameters | T (°K) | ΔH0 | ΔS0 | ΔG0 | |
|---|---|---|---|---|---|
| Adsorbent | |||||
| RS | 328 | 6.03 | 108.86 | −29.70 | |
| 318 | −28.61 | ||||
| 308 | −27.51 | ||||
| 298 | −26.43 | ||||
| RSAC | 328 | 9.21 | 128.135 | −32.78 | |
| 318 | −31.62 | ||||
| 308 | −30.29 | ||||
| 298 | −28.96 | ||||
| nZVFe–RSAC | 328 | 49.72 | 278.34 | −41.38 | |
| 318 | −39.16 | ||||
| 308 | −36.09 | ||||
| 298 | −33.16 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sepehri, S.; Kanani, E.; Abdoli, S.; Rajput, V.D.; Minkina, T.; Asgari Lajayer, B. Pb(II) Removal from Aqueous Solutions by Adsorption on Stabilized Zero-Valent Iron Nanoparticles—A Green Approach. Water 2023, 15, 222. https://doi.org/10.3390/w15020222
Sepehri S, Kanani E, Abdoli S, Rajput VD, Minkina T, Asgari Lajayer B. Pb(II) Removal from Aqueous Solutions by Adsorption on Stabilized Zero-Valent Iron Nanoparticles—A Green Approach. Water. 2023; 15(2):222. https://doi.org/10.3390/w15020222
Chicago/Turabian StyleSepehri, Saloome, Elahe Kanani, Sima Abdoli, Vishnu D. Rajput, Tatiana Minkina, and Behnam Asgari Lajayer. 2023. "Pb(II) Removal from Aqueous Solutions by Adsorption on Stabilized Zero-Valent Iron Nanoparticles—A Green Approach" Water 15, no. 2: 222. https://doi.org/10.3390/w15020222
APA StyleSepehri, S., Kanani, E., Abdoli, S., Rajput, V. D., Minkina, T., & Asgari Lajayer, B. (2023). Pb(II) Removal from Aqueous Solutions by Adsorption on Stabilized Zero-Valent Iron Nanoparticles—A Green Approach. Water, 15(2), 222. https://doi.org/10.3390/w15020222










