Assessment of Water Quality Variations and Trophic State of the Joumine Reservoir (Tunisia) by Multivariate Analysis
Abstract
:1. Introduction
2. Materials and Methods
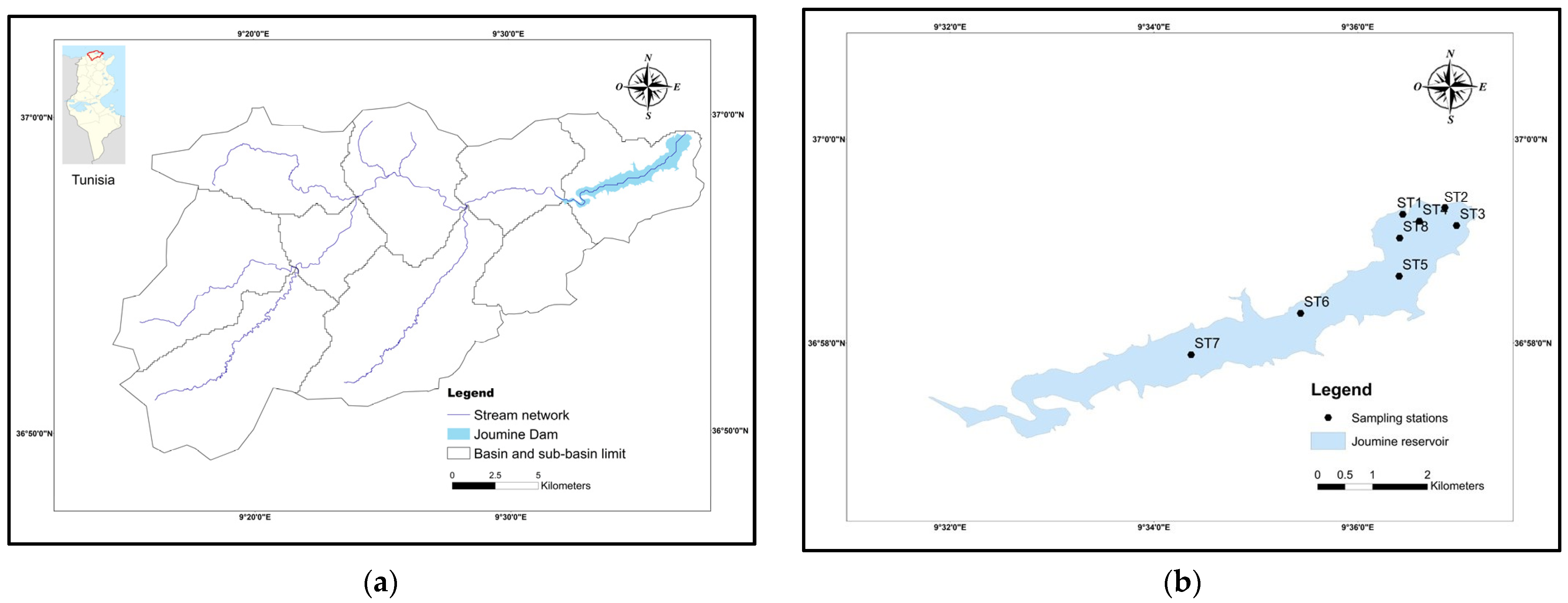
2.1. Study Area, Sampling, and Sample Analysis
2.2. Data Treatment
3. Results
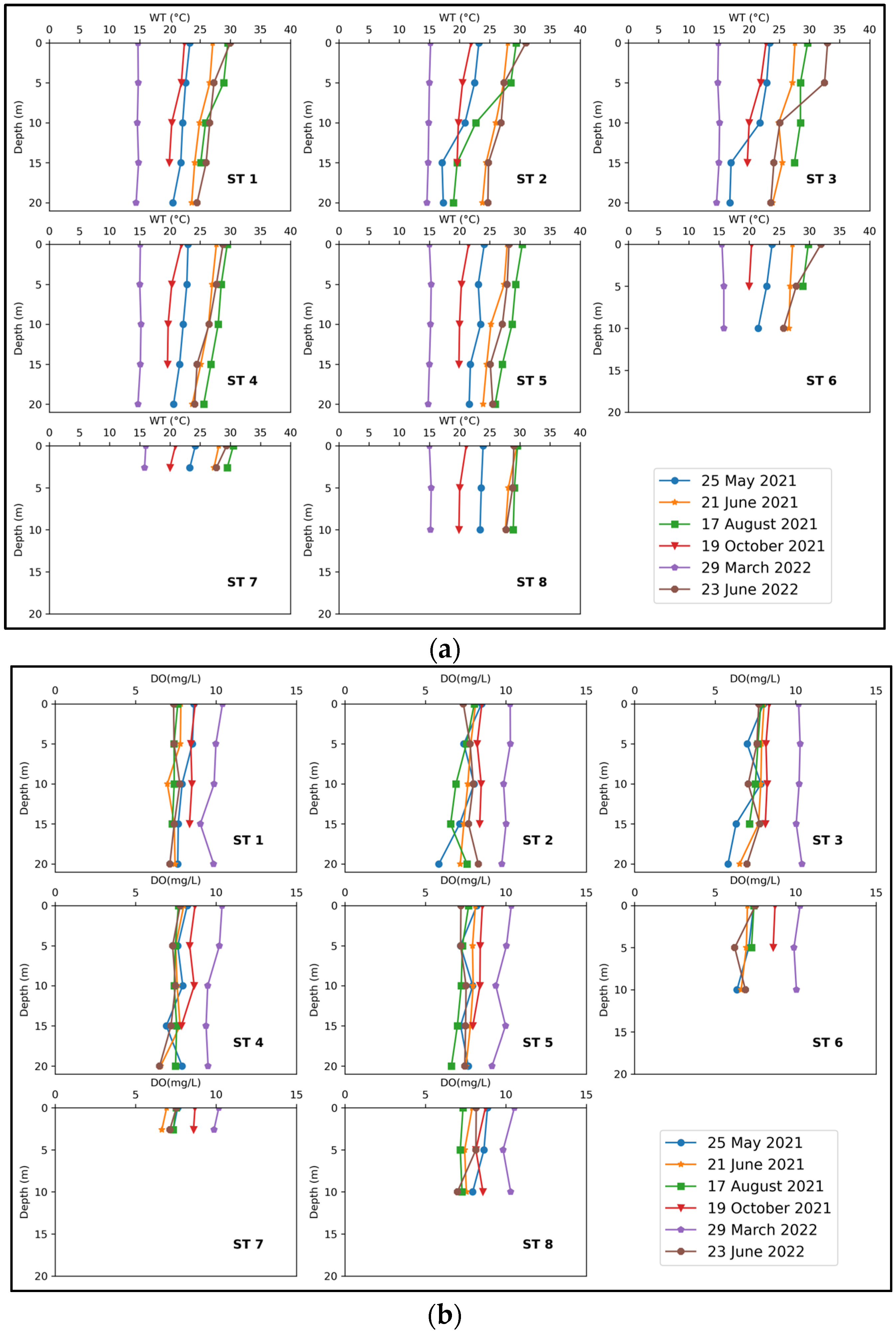
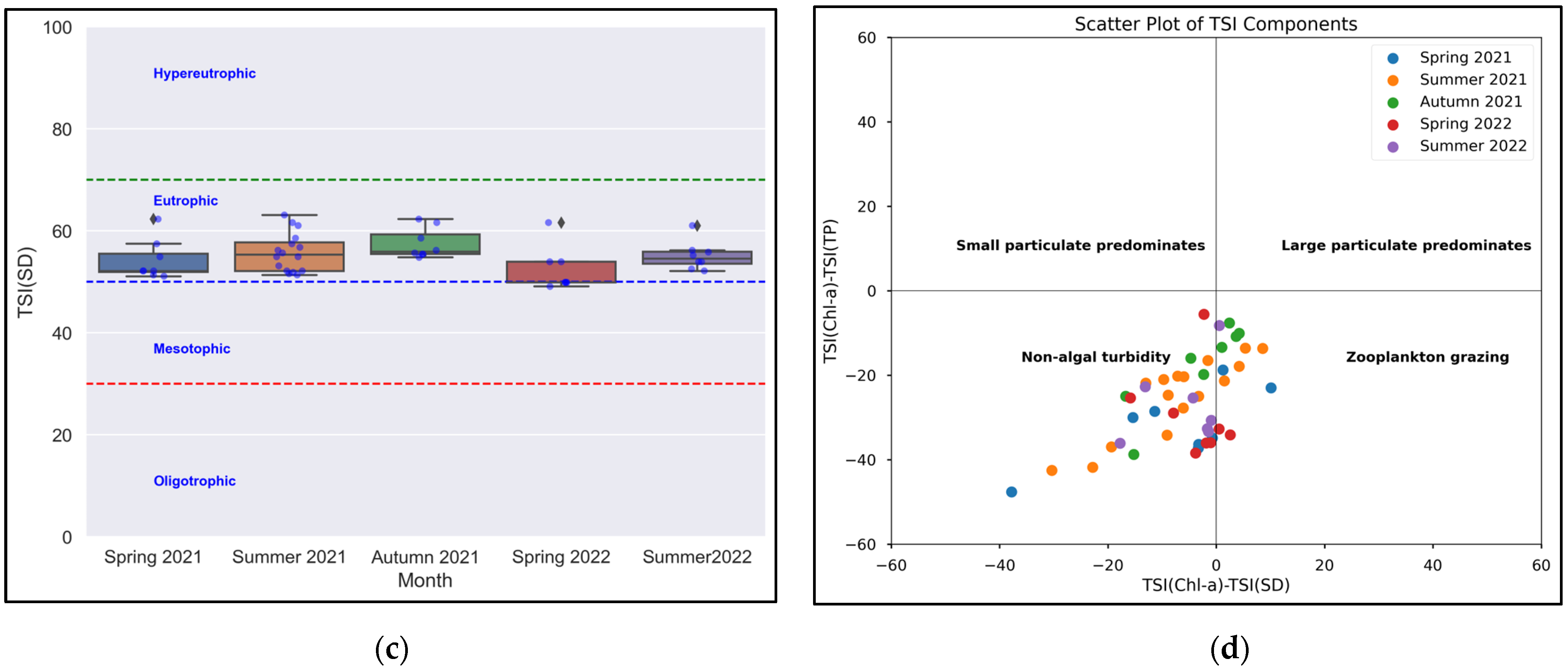
3.1. Trophic Status and Physicochemical Factors of the Joumine Reservoir
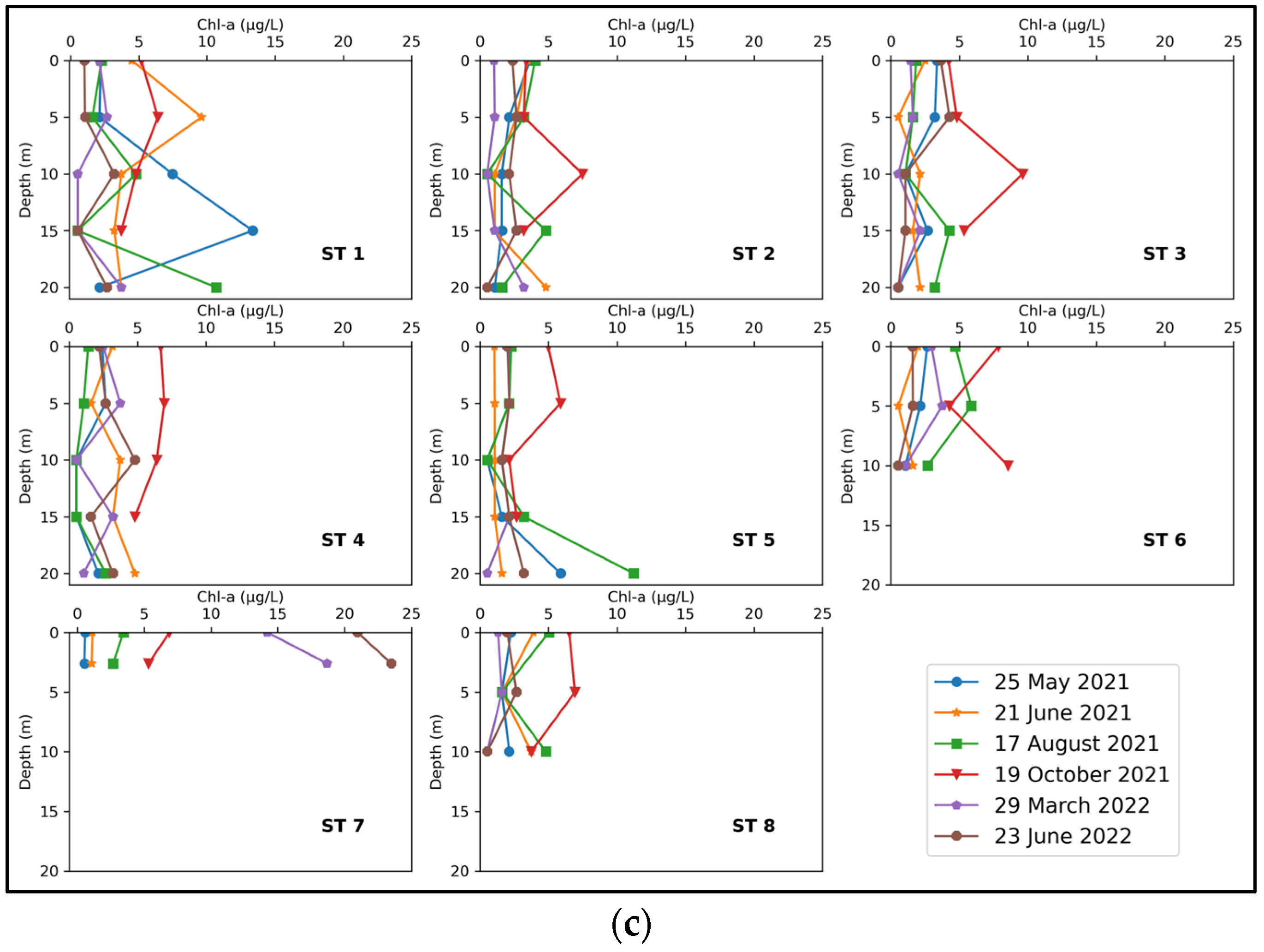
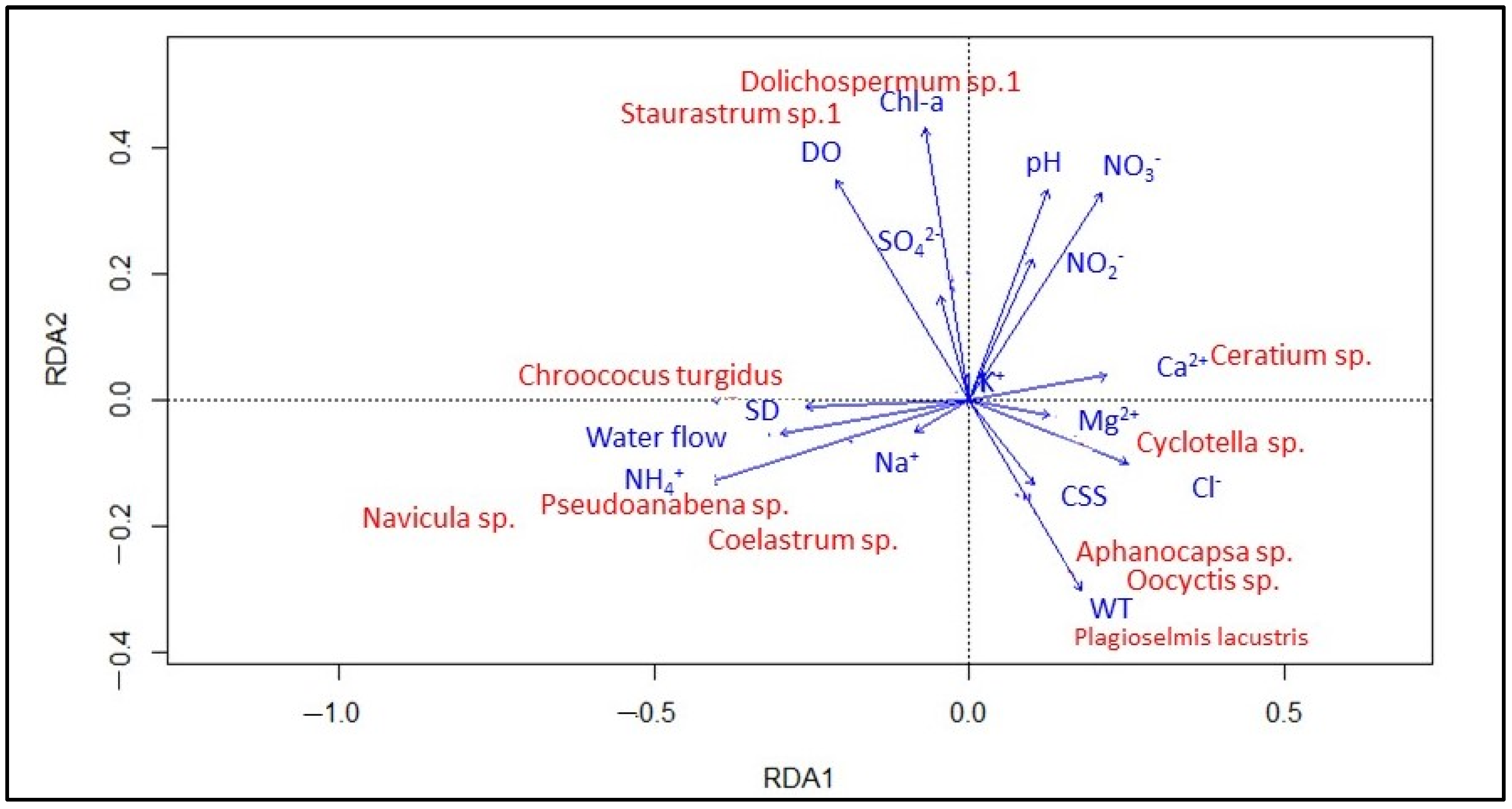
3.2. Joumine Reservoir’s Phytoplankton Community
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Q.; Mei, K.; Dahlgren, R.A.; Wang, T.; Gong, J.; Zhang, M. Impacts of Land Use and Population Density on Seasonal Surface Water Quality Using a Modified Geographically Weighted Regression. Sci. Total Environ. 2016, 572, 450–466. [Google Scholar] [CrossRef]
- Le Moal, M.; Gascuel-Odoux, C.; Ménesguen, A.; Souchon, Y.; Étrillard, C.; Levain, A.; Moatar, F.; Pannard, A.; Souchu, P.; Lefebvre, A.; et al. Eutrophication: A New Wine in an Old Bottle? Sci. Total Environ. 2019, 651, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, S.R.; Caraco, N.F.; Correll, D.L.; Howarth, R.W.; Sharpley, A.N.; Smith, V.H. Non Point Pollution of Surafce Waters with Phosphorus and Nitogen. Ecol. Appl. 1998, 8, 559–568. [Google Scholar] [CrossRef]
- Kazi, T.G.; Arain, M.B.; Jamali, M.K.; Jalbani, N.; Afridi, H.I.; Sarfraz, R.A.; Baig, J.A.; Shah, A.Q. Assessment of Water Quality of Polluted Lake Using Multivariate Statistical Techniques: A Case Study. Ecotoxicol. Environ. Saf. 2009, 72, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Mohseni-Bandpei, A.; Motesaddi, S.; Eslamizadeh, M.; Rafiee, M.; Nasseri, M.; Montazeri Namin, M.; Hashempour, Y.; Mehrabi, Y.; Riahi, S.M. Water Quality Assessment of the Most Important Dam (Latyan Dam) in Tehran, Iran. Environ. Sci. Pollut. Res. 2018, 25, 29227–29239. [Google Scholar] [CrossRef] [PubMed]
- Strobl, R.O.; Robillard, P.D. Network Design for Water Quality Monitoring of Surface Freshwaters: A Review. J. Environ. Manag. 2008, 87, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Varol, M.; Balcı, M. Characteristics of Effluents from Trout Farms and Their Impact on Water Quality and Benthic Algal Assemblages of the Receiving Stream. Environ. Pollut. 2020, 266, 115101. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, F. Multi-Scale Analysis of the Relationship between Landscape Patterns and a Water Quality Index (WQI) Based on a Stepwise Linear Regression (SLR) and Geographically Weighted Regression (GWR) in the Ebinur Lake Oasis. Environ. Sci. Pollut. Res. 2018, 25, 7033–7048. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, X.; Chen, Y.; Cai, Y.; Deng, J. Assessing River Water Quality Using Water Quality Index in Lake Taihu Basin, China. Sci. Total Environ. 2018, 612, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Qadir, A.; Malik, R.N.; Husain, S.Z. Spatio-Temporal Variations in Water Quality of Nullah Aik-Tributary of the River Chenab, Pakistan. Environ. Monit. Assess. 2008, 140, 43–59. [Google Scholar] [CrossRef]
- Boukari, A.; Benabdallah, S.; Everbecq, E.; Magermans, P.; Grard, A.; Habaieb, H.; Deliège, J.-F. Assessment of Agriculture Pressures Impact on the Joumine River Water Quality Using the PEGASE Model. Environ. Manag. 2019, 64, 520–535. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Warren, R.; He, Y.; Ye, J.; Li, Q.; Wang, G. Impacts of Climate Change on TN Load and Its Control in a River Basin with Complex Pollution Sources. Sci. Total Environ. 2018, 615, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Hamed, Y.; Hadji, R.; Redhaounia, B.; Zighmi, K.; Bâali, F.; El Gayar, A. Climate Impact on Surface and Groundwater in North Africa: A Global Synthesis of Findings and Recommendations. Euro-Mediterr. J. Environ. Integr. 2018, 3, 25. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Stanley, E.H.; Vander Zanden, M.J. State of the World’s Freshwater Ecosystems: Physical, Chemical, and Biological Changes. Annu. Rev. Environ. Resour. 2011, 36, 75–99. [Google Scholar] [CrossRef]
- Boukari, A. Impacts de la Pollution Diffuse en Milieu Agricole sur la Qualité des Eaux de Surface à l’échelle Du Bassin Versant de l’oued Joumine: Application de Différentes Approches de Modélisation; Université de Liège: Liège, Belgium, 2019. [Google Scholar]
- Gayar, A.E.; Hamed, Y. Climate Change and Water Resources Management in Arab Countries. In Recent Advances in Environmental Science from the Euro-Mediterranean and Surrounding Regions; Kallel, A., Ksibi, M., Ben Dhia, H., Khélifi, N., Eds.; Advances in Science, Technology & Innovation; Springer International Publishing: Cham, Switzerland, 2018; pp. 89–91. ISBN 978-3-319-70547-7. [Google Scholar]
- Markad, A.T.; Landge, A.T.; Nayak, B.B.; Inamdar, A.B.; Mishra, A.K. Trophic State Modeling for Shallow Freshwater Reservoir: A New Approach. Environ. Monit. Assess. 2019, 191, 586. [Google Scholar] [CrossRef] [PubMed]
- MARH BPEH. Elaboration de La Vision et de La Stratégie Du Secteur Du l’eau a Li’horizon 250 Pour la Tunisie <<Eau 2050>>. Etape 3: Réalisation des Etudes Prospectives Multithématiques et Etablissement de Modèles Prévisionnels Offre-Demande (Bilans). 2022. Available online: http://www.onagri.tn/uploads/secteur-eau/ETAPE-3-Volume1.pdf (accessed on 25 June 2023).
- Jenhani, A.B.R.; Fathalli, A.; Djemali, I.; Changeux, T.; Romdhane, M.S. Tunisian Reservoirs: Diagnosis and Biological Potentialities. Aquat. Living Resour. 2019, 32, 17. [Google Scholar] [CrossRef]
- Lürling, M.; Van Oosterhout, F.; Faassen, E. Eutrophication and Warming Boost Cyanobacterial Biomass and Microcystins. Toxins 2017, 9, 64. [Google Scholar] [CrossRef]
- Fathalli, A. Caractérisation Moléculaire et Étude Du Potentiel Toxique des Souches de Cyanobactéries Isolées à Partir des Retenues de Barrages de la Tunisie: Cas de la Retenue Du Barrage Bir M’cherga, Tunisie. Ph.D. Thesis, University of Carthage, Tunis, Tunisia, 2012. [Google Scholar]
- Fathalli, A.; Ben Rejeb Jenhani, A.; Moreira, C.; Saker, M.; Romdhane, M.; Vasconcelos, V. First Observation of the Toxic and Invasive Cyanobacterium Species Cylindrospermopsis Raciborskii in Tunisian Freshwaters. Toxic. Assess. Mol. Charact. 2010, 19, 1074–1083. [Google Scholar]
- Fathalli, A.; Romdhane, M.S.; Vasconcelos, V.; Ben Rejeb Jenhani, A. Biodiversity of Cyanobacteria in Tunisian Freshwater Reservoirs: Occurrence and Potent Toxicity—A Review. J. Water Supply: Res. Technol.—AQUA 2015, 64, 755–772. [Google Scholar] [CrossRef]
- Fathalli, A.; Jenhani, A.B.R.; Moreira, C.; Azevedo, J.; Welker, M.; Romdhane, M.; Antunes, A.; Vasconcelos, V. Genetic Variability of the Invasive Cyanobacteria Cylindrospermopsis Raciborskii from Bir M’cherga Reservoir (Tunisia). Arch. Microbiol. 2011, 193, 595–604. [Google Scholar] [CrossRef]
- Aouissi, J.; Chabaane, Z.L.; Benabdallah, S.; Cudennec, C. Assessing the Hydrological Impacts of Agricultural Changes Upstream of the Tunisian World Heritage Sea-Connected Ichkeul Lake. Proc. Int. Assoc. Hydrol. Sci. 2015, 365, 61–65. [Google Scholar] [CrossRef]
- OCDE. Eutrophisation des Eaux, Méthodes de Surveillance, d’évaluation et de Lutte; OCDE: Paris, France, 1982; 165p. [Google Scholar]
- Tian, C.; Lu, X.; Pei, H.; Hu, W.; Xie, J. Seasonal Dynamics of Phytoplankton and Its Relationship with the Environmental Factors in Dongping Lake, China. Environ. Monit. Assess. 2013, 185, 2627–2645. [Google Scholar] [CrossRef]
- Heisler, J.; Glibert, P.M.; Burkholder, J.M.; Anderson, D.M.; Cochlan, W.; Dennison, W.C.; Dortch, Q.; Gobler, C.J.; Heil, C.A.; Humphries, E.; et al. Eutrophication and Harmful Algal Blooms: A Scientific Consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar] [CrossRef] [PubMed]
- WHO. Directives de Qualité Pour L’eau de Boisson: 4e éd. Intégrant le Premier Additif, 4th ed.; Organisation mondiale de la Santé: Geneva, Switzerland, 2017; ISBN 978-92-4-254995-9. [Google Scholar]
- Bouguerne, A.; Boudoukha, A.; Benkhaled, A.; Mebarkia, A.-H. Assessment of Surface Water Quality of Ain Zada Dam (Algeria) Using Multivariate Statistical Techniques. Int. J. River Basin Manag. 2017, 15, 133–143. [Google Scholar] [CrossRef]
- Mamun, M.; Kim, J.Y.; An, K.-G. Multivariate Statistical Analysis of Water Quality and Trophic State in an Artificial Dam Reservoir. Water 2021, 13, 186. [Google Scholar] [CrossRef]
- Singh, K.P.; Malik, A.; Mohan, D.; Sinha, S. Multivariate Statistical Techniques for the Evaluation of Spatial and Temporal Variations in Water Quality of Gomti River (India)—A Case Study. Water Res. 2004, 38, 3980–3992. [Google Scholar] [CrossRef]
- Dutta, S.; Dwivedi, A.; Suresh Kumar, M. Use of Water Quality Index and Multivariate Statistical Techniques for the Assessment of Spatial Variations in Water Quality of a Small River. Environ. Monit. Assess. 2018, 190, 718. [Google Scholar] [CrossRef] [PubMed]
- Koklu, R.; Sengorur, B.; Topal, B. Water Quality Assessment Using Multivariate Statistical Methods—A Case Study: Melen River System (Turkey). Water Resour. Manag. 2010, 24, 959–978. [Google Scholar] [CrossRef]
- Li, T.; Li, S.; Liang, C.; Bush, R.T.; Xiong, L.; Jiang, Y. A Comparative Assessment of Australia’s Lower Lakes Water Quality under Extreme Drought and Post-Drought Conditions Using Multivariate Statistical Techniques. J. Clean. Prod. 2018, 190, 1–11. [Google Scholar] [CrossRef]
- Varol, M.; Gökot, B.; Bekleyen, A.; Şen, B. Water Quality Assessment and Apportionment of Pollution Sources of Tigris River (Turkey) Using Multivariate Statistical Techniques—A Case Study: Water Quality Assessment. River Res. Appl. 2012, 28, 1428–1438. [Google Scholar] [CrossRef]
- Becker, V.; Huszar, V.L.M.; Crossetti, L.O. Responses of Phytoplankton Functional Groups to the Mixing Regime in a Deep Subtropical Reservoir. Hydrobiologia 2009, 628, 137–151. [Google Scholar] [CrossRef]
- Slimani, M.; Cudennec, C.; Feki, H. Structure du gradient pluviométrique de la transition Méditerranée–Sahara en Tunisie: Déterminants géographiques et saisonnalité/Structure of the rainfall gradient in the Mediterranean–Sahara transition in Tunisia: Geographical determinants and seasonality. Hydrol. Sci. J. 2007, 52, 1088–1102. [Google Scholar] [CrossRef]
- Chakroun, H.; Benabdallah, S.; Lili Chabaane, Z. Spatial Decision Support Systems Integrating Ecohydrology in Limited Water Resources Regions; Advances in Environmental Research; Nova Science Publishers: Hauppauge, NY, USA, 2015; Volume 39. [Google Scholar]
- Limam, A. Contribution à l’étude des Conditions Du Milieu et des Peuplements Phytoplanctoniques des Eaux de la Retenue Du Barrage Joumine en Relation Avec le Réseau de Distribution; Université de Carthage: Carthage, Tunisia, 2003; p. 107. [Google Scholar]
- Rodier, J.; Legube, B.; Merlet, N. Coll L’analyse de l’eau, 9th ed.; Dunod: Paris, France, 2009. [Google Scholar]
- Neuveux, J. Recherche Sur La Chlorophylle a et à La Phétophytinea En Milieu Oligotrophe et En Milieu Eutrophe (Méditérrannée). Ph.D. Thesis, University of Paris 6, Paris, France, 1974; p. 116. [Google Scholar]
- Utermöhl, H. On the Perfection of Quantitative Phytoplankton Method. Int. Ass. Theo. Appl. Limnol. Commun. 1958, 9, 1–38. [Google Scholar]
- Muangthong, S.; Shrestha, S. Assessment of Surface Water Quality Using Multivariate Statistical Techniques: Case Study of the Nampong River and Songkhram River, Thailand. Environ. Monit. Assess. 2015, 187, 548. [Google Scholar] [CrossRef] [PubMed]
- Vega, M.; Pardo, R.; Barrado, E.; Debán, L. Assessment of Seasonal and Polluting Effects on the Quality of River Water by Exploratory Data Analysis. Water Res. 1998, 32, 3581–3592. [Google Scholar] [CrossRef]
- Liu, C.-W.; Lin, K.-H.; Kuo, Y.-M. Application of Factor Analysis in the Assessment of Groundwater Quality in a Blackfoot Disease Area in Taiwan. Sci. Total Environ. 2003, 313, 77–89. [Google Scholar] [CrossRef]
- Xu, Y.; Xiang, Z.; Rizo, E.Z.; Naselli-Flores, L.; Han, B.-P. Combination of Linear and Nonlinear Multivariate Approaches Effectively Uncover Responses of Phytoplankton Communities to Environmental Changes at Regional Scale. J. Environ. Manag. 2022, 305, 114399. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.A.; Ansari, A.A. Eutrophication: An Ecological Vision. Bot. Rev. 2005, 71, 449–482. [Google Scholar] [CrossRef]
- Carlson, R.E. A Trophic State Index for Lakes1: Trophic State Index. Limnol. Oceanogr. 1977, 22, 361–369. [Google Scholar] [CrossRef]
- Champiat, D.; Larpent, J.P. Biologie des Eaux. Méthodes et Etchniques; Masson: Paris, France, 1988; p. 347. [Google Scholar]
- Bel Haj Zekri, S. Conditions Du Milieu et Recherche de la Toxicité des Cyanoprocaryotes et de Leurs Potentiels Toxiques Dans Les Retenues des Barrage Joumine et Séjenène; Université du 7 novembre à Carthage: Tunis, Tunisia, 2003; p. 126. [Google Scholar]
- El Herry, S. Biodiversité En Cyanobactéries Toxiques Dans La Retenue de Barrage Lebna (Tunisie). Caractérisations Morphologique et Moléculaire et Évaluation Du Potentiel Toxique des Différentes Morphoespèces Du Genre Microcystis Spp. Isolées des Divers Plans d’eau En Algérie et En Tunisie. Ph.D. Thesis, Université de Cartage-Université Paris-Sud 11, Tunis, Tunisia, 2008; p. 202. [Google Scholar]
- Jenhani, A.B.R.; Fathalli, A.; Romdhane, M.S. Phytoplankton Assemblages in Bir Mcherga Freshwater Reservoir (Tunisia). In Proceedings of the Conference Proceedings, Tulcea, Romania, 14–16 September 2012; p. 6. [Google Scholar]
- Abrantes, N.; Antunes, S.C.; Pereira, M.J.; Gonçalves, F. Seasonal Succession of Cladocerans and Phytoplankton and Their Interactions in a Shallow Eutrophic Lake (Lake Vela, Portugal). Acta Oecologica 2006, 29, 54–64. [Google Scholar] [CrossRef]
- Varol, M.; Şen, B. Assessment of Surface Water Quality Using Multivariate Statistical Techniques: A Case Study of Behrimaz Stream, Turkey. Environ. Monit. Assess. 2009, 159, 543–553. [Google Scholar] [CrossRef]
- Carlson, R.; Simpson, J. A Coordinator’s Guide to Volunteer Lake Monitoring Methods. N. Am. Lake Manag. Soc. 1996, 96, 305. [Google Scholar]
- Carlson, R.E.; Havens, K.E. Simple Graphical Methods for the Interpretation of Relationships Between Trophic State Variables. Lake Reserv. Manag. 2005, 21, 107–118. [Google Scholar] [CrossRef]
- Aouissi, J.; Benabdallah, S.; Chabaâne, Z.L.; Cudennec, C. Modeling Water Quality to Improve Agricultural Practices and Land Management in a Tunisian Catchment Using the Soil and Water Assessment Tool. J. Environ. Qual. 2014, 43, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Barakat, A.; El Baghdadi, M.; Rais, J.; Aghezzaf, B.; Slassi, M. Assessment of Spatial and Seasonal Water Quality Variation of Oum Er Rbia River (Morocco) Using Multivariate Statistical Techniques. Int. Soil Water Conserv. Res. 2016, 4, 284–292. [Google Scholar] [CrossRef]
- Conceição, L.P.; De Jesus Affe, H.M.; Da Silva, D.M.L.; De Castro Nunes, J.M. Spatio-Temporal Variation of the Phytoplankton Community in a Tropical Estuarine Gradient, under the Influence of River Damming. Reg. Stud. Mar. Sci. 2021, 43, 101642. [Google Scholar] [CrossRef]
- Barbosa, A.B.; Domingues, R.B.; Galvão, H.M. Environmental Forcing of Phytoplankton in a Mediterranean Estuary (Guadiana Estuary, South-Western Iberia): A Decadal Study of Anthropogenic and Climatic Influences. Estuaries Coasts 2010, 33, 324–341. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Huszar, V.; Kruk, C.; Naselli-Flores, L.; Melo, S. Towards a Functional Classification of the Freshwater Phytoplankton. J. Plankton Res. 2002, 24, 417–428. [Google Scholar] [CrossRef]
- Domingues, R.B.; Barbosa, A.B.; Galvão, H.M. River Damming Leads to Decreased Phytoplankton Biomass and Disappearance of Cyanobacteria Blooms. Estuar. Coast. Shelf Sci. 2014, 136, 129–138. [Google Scholar] [CrossRef]
- Bonilla, S.; Aguilera, A.; Aubriot, L.; Huszar, V.; Almanza, V.; Haakonsson, S.; Izaguirre, I.; O’Farrell, I.; Salazar, A.; Becker, V.; et al. Nutrients and Not Temperature Are the Key Drivers for Cyanobacterial Biomass in the Americas. Harmful Algae 2023, 121, 102367. [Google Scholar] [CrossRef]
- Butrón, A.; Iriarte, A.; Madariaga, I. Size-Fractionated Phytoplankton Biomass, Primary Production and Respiration in the Nervión–Ibaizabal Estuary: A Comparison with Other Nearshore Coastal and Estuarine Ecosystems from the Bay of Biscay. Cont. Shelf Res. 2009, 29, 1088–1102. [Google Scholar] [CrossRef]
- Maier, G.; Glegg, G.A.; Tappin, A.D.; Worsfold, P.J. A High Resolution Temporal Study of Phytoplankton Bloom Dynamics in the Eutrophic Taw Estuary (SW England). Sci. Total Environ. 2012, 434, 228–239. [Google Scholar] [CrossRef]
- Béjaoui, B.; Basti, L.; Canu, D.M.; Feki-Sahnoun, W.; Salem, H.; Dahmani, S.; Sahbani, S.; Benabdallah, S.; Blake, R.; Norouzi, H.; et al. Hydrology, Biogeochemistry and Metabolism in a Semi-arid Mediterranean Coastal Wetland Ecosystem. Sci. Rep. 2022, 12, 9367. [Google Scholar] [CrossRef]
- Chislock, M.F. Eutrophicati on: Causes, Consequences, and Controls in Aquati c Ecosystems. Nat. Educ. Knowl. 2013, 4, 10. [Google Scholar]
- Boyd, C.E.; Tucker, C.S. Pond Aquaculture Water Quality Management; Kluwer: Norwell, MA, USA, 1998. [Google Scholar]
- Vollenweider, R.A. A Manual on Methods for Measuring Primary Production in Aquatic Environments; I.B.P. HandbooK N°12; Blackwell: Oxford, UK; Edinburgh, Scotland, 1969. [Google Scholar]
- Brettum, P. Changes in the Volume and Composition of Phytoplankton after Experimental Acidification of a Humic Lake. Environ. Int. 1996, 22, 619–628. [Google Scholar] [CrossRef]
- Rao, D.S.M.; Rao, G.M.N. Seasonal Abundance of Phytoplankton in Relation to PhysicoChemical Features of Venkammacheruvu Veeraghattam, Sriakulam (Dist.) A.P, India. IOSR J. Pharm. Biol. Sci. 2016, 11, 91–98. [Google Scholar] [CrossRef]
- Parakkandi, J.; Saha, A.; Sarkar, U.K.; Das, B.K.; Puthiyottil, M.; Muhammadali, S.A.; Ramteke, M.; Johnson, C.; Kumari, S. Spatial and Temporal Dynamics of Phytoplankton in Association with Habitat Parameters in a Tropical Reservoir, India. Arab. J. Geosci. 2021, 14, 827. [Google Scholar] [CrossRef]
- Devercelli, M.; O’Farrell, I. Factors Affecting the Structure and Maintenance of Phytoplankton Functional Groups in a Nutrient Rich Lowland River. Limnologica 2013, 43, 67–78. [Google Scholar] [CrossRef]
- Klemer, A.R.; Cullen, J.J.; Mageau, M.T.; Hanson, K.M.; Sundell, R.A. Cyanobacterial Buoyancy Regualtion: The Paradoxical Roles of Carbon1. J. Phycol. 1996, 32, 47–53. [Google Scholar] [CrossRef]
- Steinberg, C.E.W.; Hartmann, H.M. Planktonic Bloom-Forming Cyanobacteria and the Eutrophication of Lakes and Rivers. Freshw. Biol. 1988, 20, 279–287. [Google Scholar] [CrossRef]
- Onagri Observatoire National de l’Agriculture. Available online: http://www.onagri.nat.tn/barrages (accessed on 9 February 2023).
- Lampert, W. Laboratory Studies on Zooplankton-cyanobacteria Interactions. N. Z. J. Mar. Freshw. Res. 1987, 21, 483–490. [Google Scholar] [CrossRef]



| 2020/2021–2021/2022 | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WT 1 | pH | DO 1 | NO3− 1 | NO2− 1 | NH4+ 1 | Chl-a 1 | CSS 1 | Na+ 1 | K+ 1 | Ca2+ 1 | Mg2+ 1 | Cl− 1 | SO42− 1 | SD 1 | Zeu 1 | ||
| ST1 | Mean | 21.82 | 8.87 | 8.28 | 6.12 | 0.22 | 1.61 | 4.33 | 11.37 | 46.14 | 3.31 | 40.64 | 13.43 | 88.09 | 67.00 | 1.50 | 3.84 |
| Min | 14.40 | 8.03 | 6.97 | 0.07 | 0.01 | 0.30 | 0.53 | 1.00 | 17.42 | 0.28 | 0.97 | 4.66 | 21.84 | 30.56 | 0.70 | 1.79 | |
| Max | 29.60 | 10.73 | 10.40 | 18.47 | 0.65 | 7.40 | 13.35 | 39.00 | 202.04 | 14.82 | 123.34 | 40.37 | 133.00 | 120.40 | 2.50 | 6.40 | |
| +/−SD 1 | 4.66 | 0.93 | 0.98 | 6.74 | 0.20 | 1.93 | 3.30 | 12.08 | 54.78 | 4.09 | 30.65 | 9.94 | 31.73 | 24.76 | 0.62 | 1.58 | |
| ST2 | Mean | 21.31 | 8.95 | 8.14 | 6.80 | 0.23 | 1.48 | 2.63 | 10.00 | 37.46 | 4.02 | 39.92 | 13.22 | 105.34 | 69.87 | 1.51 | 3.87 |
| Min | 14.60 | 8.19 | 5.84 | 0.09 | 0.02 | 0.18 | 0.53 | 1.00 | 21.81 | 1.22 | 23.81 | 0.13 | 20.45 | 31.54 | 0.80 | 2.05 | |
| Max | 29.40 | 10.45 | 10.28 | 17.97 | 0.65 | 6.25 | 7.48 | 92.00 | 144.79 | 25.70 | 91.11 | 29.80 | 319.40 | 105.70 | 2.50 | 6.40 | |
| +/−SD | 4.67 | 0.71 | 1.18 | 6.92 | 0.23 | 1.59 | 1.69 | 18.72 | 29.97 | 5.56 | 23.18 | 7.08 | 55.97 | 21.74 | 0.63 | 1.60 | |
| ST3 | Mean | 21.78 | 8.80 | 8.13 | 5.94 | 0.23 | 1.50 | 2.63 | 12.22 | 30.58 | 2.62 | 41.47 | 12.38 | 87.02 | 63.90 | 1.54 | 3.94 |
| Min | 14.60 | 7.69 | 5.80 | 0.03 | 0.00 | 0.36 | 0.53 | 1.00 | 18.30 | 0.81 | 17.96 | 8.76 | 18.13 | 28.70 | 0.90 | 2.31 | |
| Max | 29.70 | 10.39 | 10.39 | 16.62 | 0.69 | 7.10 | 9.61 | 83.00 | 58.09 | 6.94 | 91.14 | 22.20 | 124.40 | 93.90 | 2.50 | 6.40 | |
| +/−SD | 4.97 | 0.85 | 1.29 | 6.41 | 0.25 | 1.72 | 2.07 | 18.48 | 13.89 | 1.99 | 24.26 | 4.97 | 35.80 | 22.24 | 0.62 | 1.60 | |
| ST4 | Mean | 22.30 | 8.91 | 8.17 | 6.19 | 0.23 | 1.47 | 2.80 | 5.83 | 30.93 | 2.52 | 39.90 | 11.41 | 84.04 | 64.04 | 1.63 | 4.17 |
| Min | 14.70 | 8.14 | 6.55 | 0.04 | 0.02 | 0.18 | 0.53 | 1.00 | 22.83 | 0.95 | 21.11 | 2.36 | 16.22 | 25.38 | 0.80 | 2.05 | |
| Max | 29.50 | 10.80 | 10.37 | 16.51 | 0.71 | 5.14 | 6.94 | 16.00 | 57.46 | 6.72 | 92.68 | 22.11 | 120.13 | 93.96 | 3.00 | 7.68 | |
| +/−SD | 4.75 | 0.77 | 0.99 | 6.43 | 0.23 | 1.50 | 2.04 | 5.65 | 12.00 | 1.87 | 21.82 | 4.62 | 36.38 | 22.86 | 0.80 | 2.04 | |
| ST5 | Mean | 22.56 | 8.77 | 8.15 | 5.92 | 0.25 | 1.76 | 2.59 | 7.07 | 30.75 | 2.50 | 41.48 | 12.49 | 88.07 | 62.48 | 1.50 | 3.84 |
| Min | 14.80 | 8.08 | 6.62 | 0.10 | 0.01 | 0.44 | 0.53 | 1.00 | 21.26 | 1.11 | 24.38 | 8.02 | 14.43 | 23.27 | 0.80 | 2.05 | |
| Max | 30.40 | 10.20 | 10.34 | 16.76 | 1.46 | 8.24 | 11.21 | 22.00 | 60.48 | 7.02 | 94.40 | 22.63 | 140.80 | 91.97 | 2.50 | 6.40 | |
| +/−SD | 4.88 | 0.70 | 0.99 | 6.17 | 0.34 | 2.12 | 2.41 | 6.03 | 12.77 | 1.68 | 23.59 | 4.78 | 36.95 | 22.02 | 0.57 | 1.47 | |
| ST6 | Mean | 22.69 | 8.86 | 7.97 | 3.31 | 0.20 | 1.42 | 3.48 | 6.54 | 30.41 | 2.18 | 41.63 | 12.27 | 94.39 | 61.99 | 1.06 | 2.71 |
| Min | 15.50 | 8.30 | 6.36 | 0.08 | 0.01 | 0.35 | 0.53 | 1.00 | 23.66 | 0.94 | 25.16 | 9.24 | 19.53 | 31.99 | 0.70 | 1.79 | |
| Max | 29.80 | 10.01 | 10.28 | 10.15 | 0.49 | 5.70 | 8.54 | 15.00 | 55.21 | 5.80 | 88.50 | 20.74 | 161.80 | 92.32 | 2.00 | 5.12 | |
| +/−SD | 5.03 | 0.66 | 1.37 | 4.01 | 0.17 | 1.86 | 2.51 | 5.51 | 11.26 | 1.48 | 22.06 | 4.06 | 46.04 | 24.56 | 0.49 | 1.26 | |
| ST7 | Mean | 23.56 | 8.79 | 8.07 | 3.07 | 0.23 | 1.40 | 4.49 | 23.17 | 30.47 | 2.10 | 39.14 | 12.55 | 86.22 | 66.43 | 0.62 | 1.59 |
| Min | 15.80 | 8.15 | 6.63 | 0.08 | 0.01 | 0.53 | 0.53 | 5.00 | 24.61 | 1.32 | 24.27 | 9.47 | 21.81 | 36.22 | 0.50 | 1.28 | |
| Max | 30.50 | 9.93 | 10.17 | 10.40 | 0.69 | 6.07 | 18.69 | 73.00 | 56.20 | 4.30 | 90.43 | 21.31 | 119.25 | 91.89 | 0.70 | 1.79 | |
| +/−SD | 5.33 | 0.57 | 1.21 | 4.56 | 0.26 | 1.76 | 5.77 | 27.26 | 11.45 | 1.01 | 23.51 | 4.06 | 39.71 | 26.26 | 0.08 | 0.20 | |
| ST8 | Mean | 22.52 | 8.97 | 8.61 | 7.76 | 0.22 | 1.62 | 3.69 | 12.00 | 29.90 | 2.29 | 39.36 | 11.21 | 95.99 | 67.98 | 1.18 | 3.03 |
| Min | 15.00 | 8.24 | 7.17 | 0.12 | 0.02 | 0.20 | 0.53 | 1.00 | 17.90 | 1.50 | 22.86 | 7.14 | 17.39 | 28.33 | 0.70 | 1.79 | |
| Max | 29.60 | 9.99 | 10.53 | 12.32 | 0.40 | 5.62 | 6.94 | 60.00 | 56.88 | 4.96 | 90.31 | 20.56 | 152.20 | 88.21 | 2.00 | 5.12 | |
| +/−SD | 6.01 | 0.66 | 1.19 | 4.72 | 0.16 | 1.96 | 2.17 | 17.43 | 13.88 | 1.38 | 27.18 | 4.36 | 46.72 | 24.38 | 0.60 | 1.53 | |
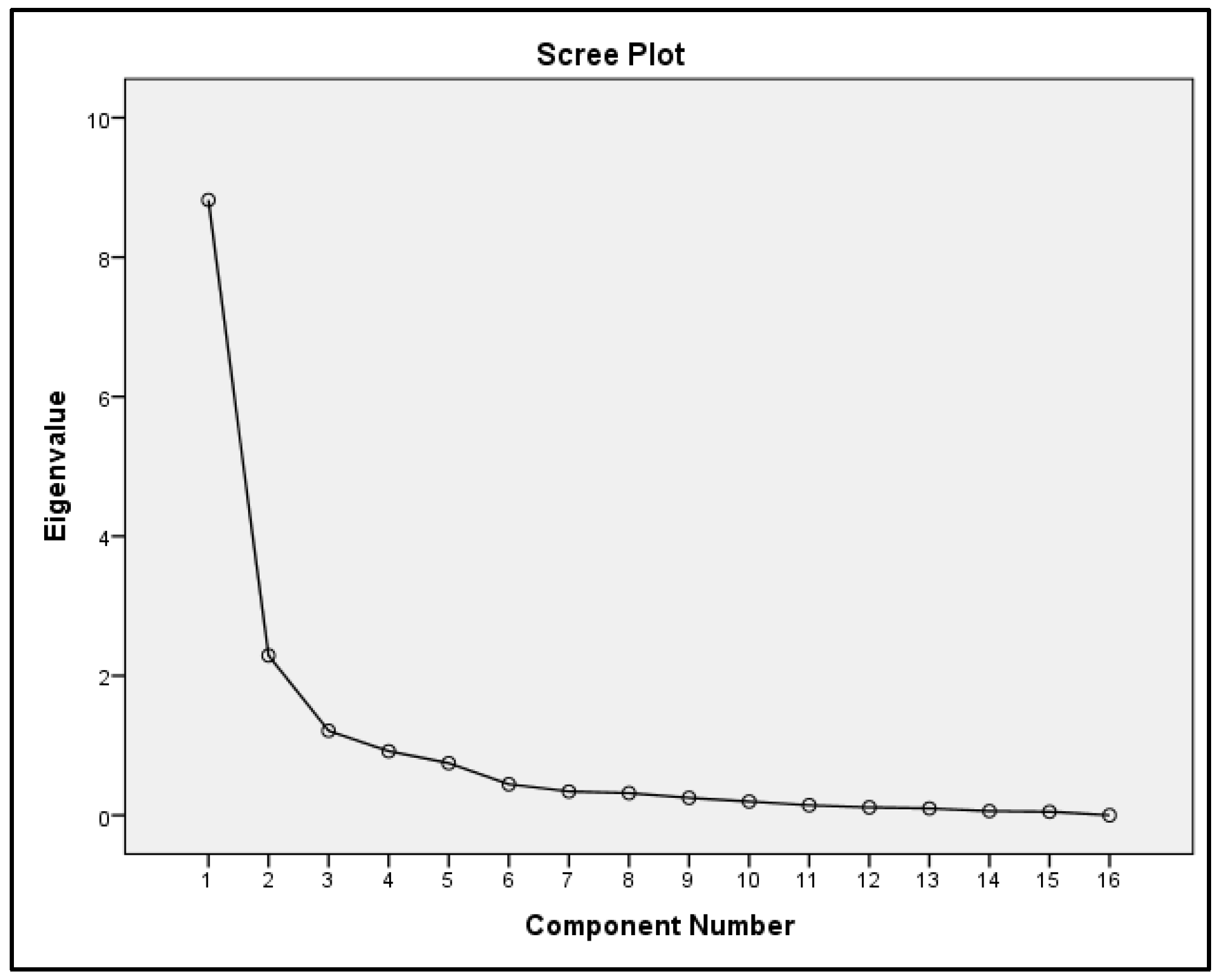
| Components | |||
|---|---|---|---|
| PC1 | PC2 | PC3 | |
| WT | −0.79 | −0.09 | 0.25 |
| pH | 0.83 | 0.44 | −0.81 |
| DO | 0.79 | 0.30 | −0.21 |
| NO2− | −0.25 | 0.89 | 0.42 |
| NO3− | −0.44 | 0.79 | 0.04 |
| NH4+ | 0.89 | 0.19 | −0.12 |
| Chl-a | −0.17 | 0.21 | −0.72 |
| CSS | 0.37 | −0.02 | 0.49 |
| Na+ | 0.78 | 0.28 | 0.32 |
| K+ | 0.68 | 0.23 | 0.27 |
| Ca2+ | 0.96 | 0.08 | −0.02 |
| Mg2+ | 0.89 | 0.20 | 0.15 |
| Cl− | 0.77 | 0.10 | 0.30 |
| SO42− | 0.84 | 0.37 | 0.13 |
| SD | 0.87 | −0.27 | 0.03 |
| Zeu | 0.87 | −0.27 | 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madyouni, H.; Almanza, V.; Benabdallah, S.; Joaquim-Justo, C.; Romdhane, M.S.; Habaieb, H.; Deliege, J.-F. Assessment of Water Quality Variations and Trophic State of the Joumine Reservoir (Tunisia) by Multivariate Analysis. Water 2023, 15, 3019. https://doi.org/10.3390/w15173019
Madyouni H, Almanza V, Benabdallah S, Joaquim-Justo C, Romdhane MS, Habaieb H, Deliege J-F. Assessment of Water Quality Variations and Trophic State of the Joumine Reservoir (Tunisia) by Multivariate Analysis. Water. 2023; 15(17):3019. https://doi.org/10.3390/w15173019
Chicago/Turabian StyleMadyouni, Haifa, Viviana Almanza, Sihem Benabdallah, Celia Joaquim-Justo, Mohamed Salah Romdhane, Hamadi Habaieb, and Jean-François Deliege. 2023. "Assessment of Water Quality Variations and Trophic State of the Joumine Reservoir (Tunisia) by Multivariate Analysis" Water 15, no. 17: 3019. https://doi.org/10.3390/w15173019





