Insights into the Responses of the Partial Denitrification Process to Elevated Perfluorooctanoic Acid Stress: Performance, EPS Characteristic and Microbial Community
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reactor Setup and Operation
2.2. Chemical and Physical Analysis
2.3. Extraction and Analysis of Bacterial Extracellular Polymeric Substances
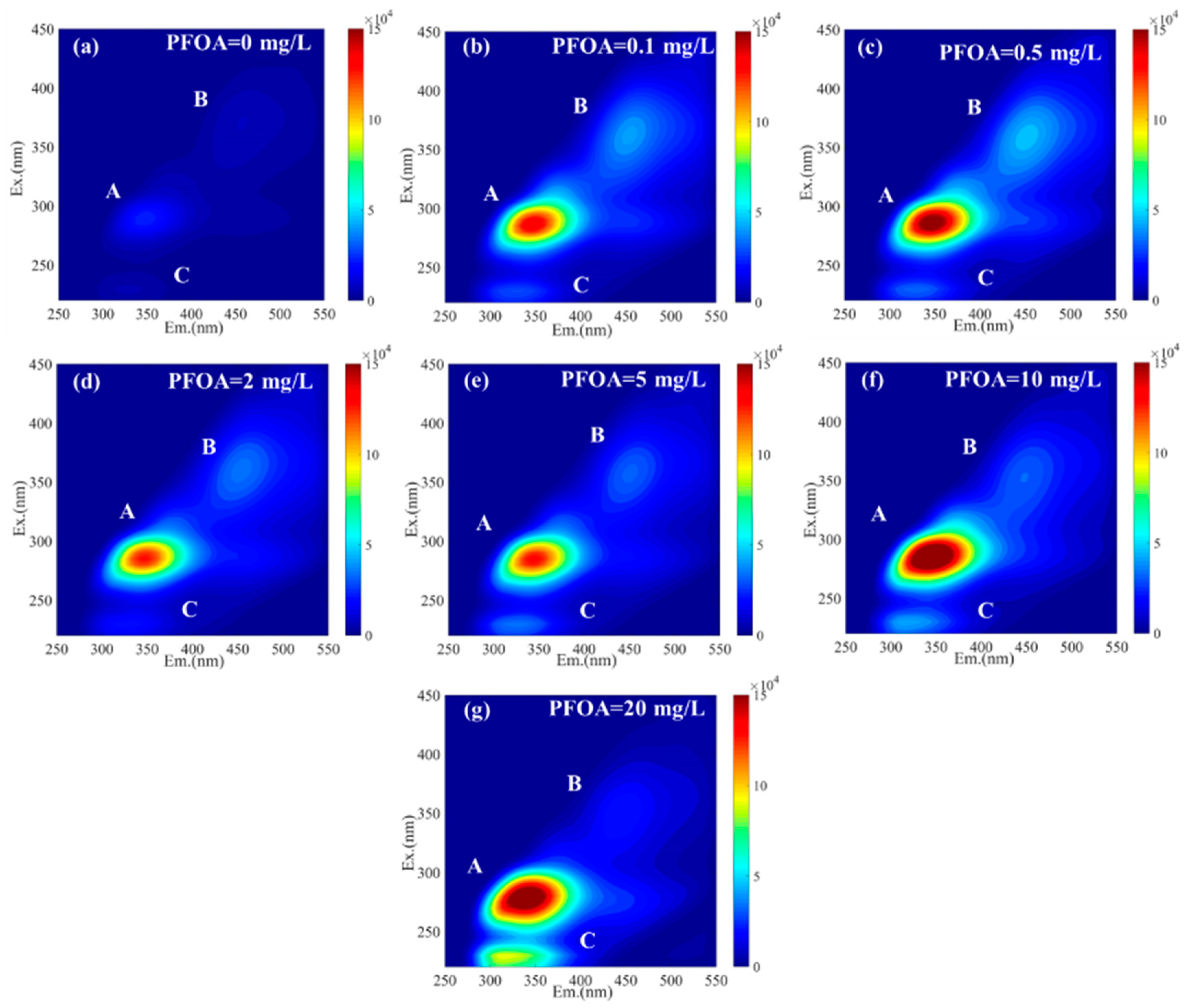
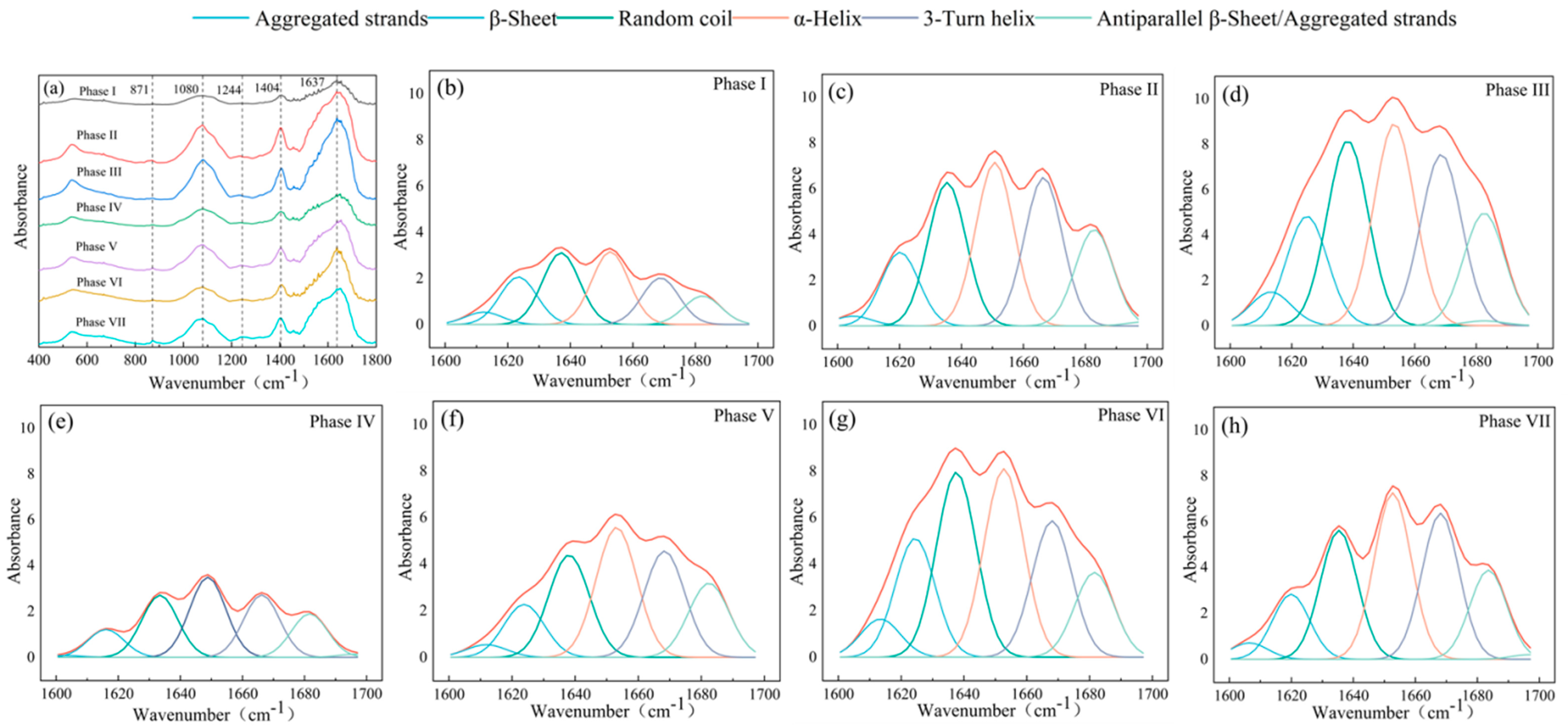
2.4. Excitation–Emission Matrix (EEM) and Fourier Transform Infrared (FTIR) Spectroscopy Analysis
2.5. Sludge Morphology Analysis
2.6. Microbial Community Analysis
3. Results and Discussion
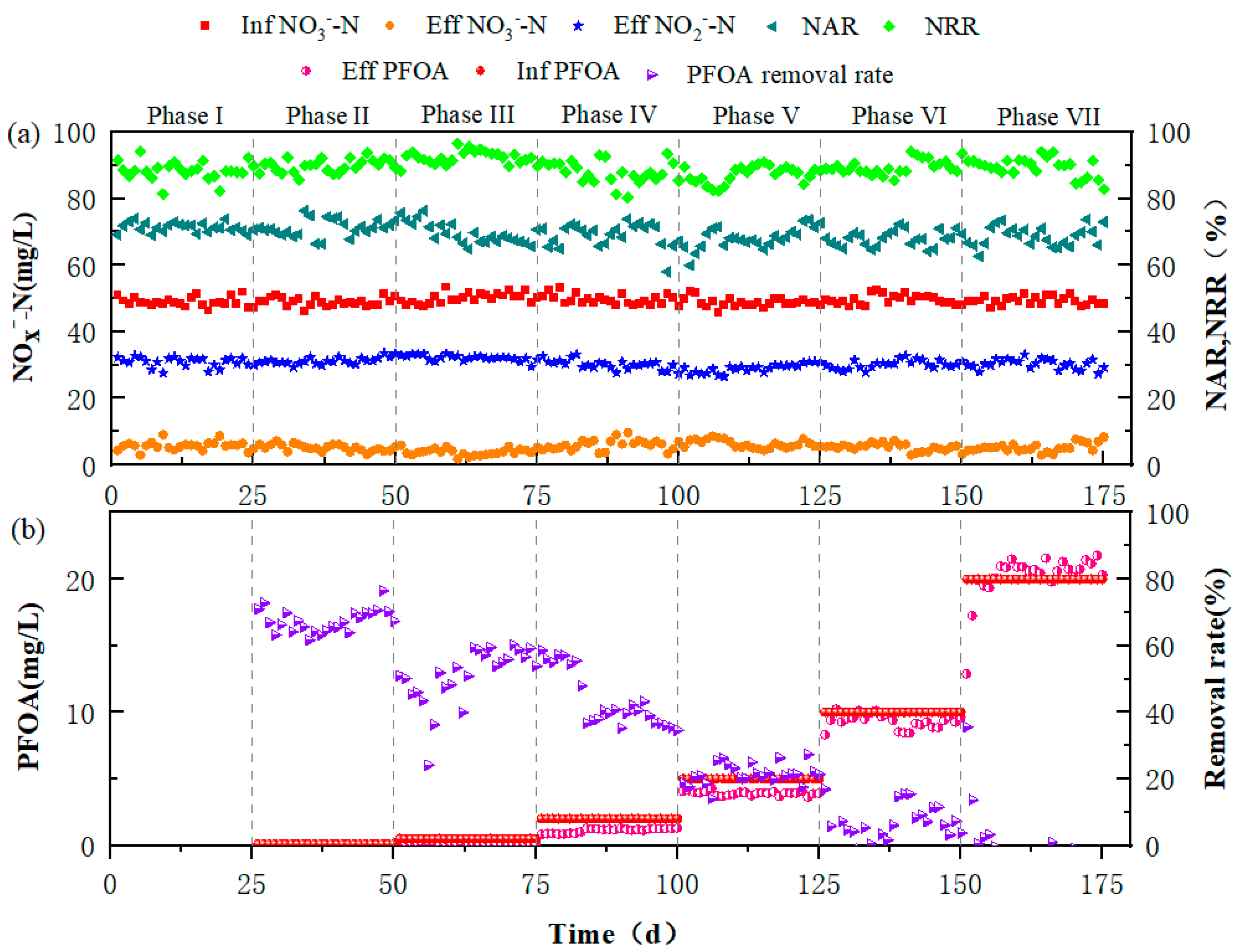
3.1. Performance of the Partial Denitrification Process under PFOA Stress
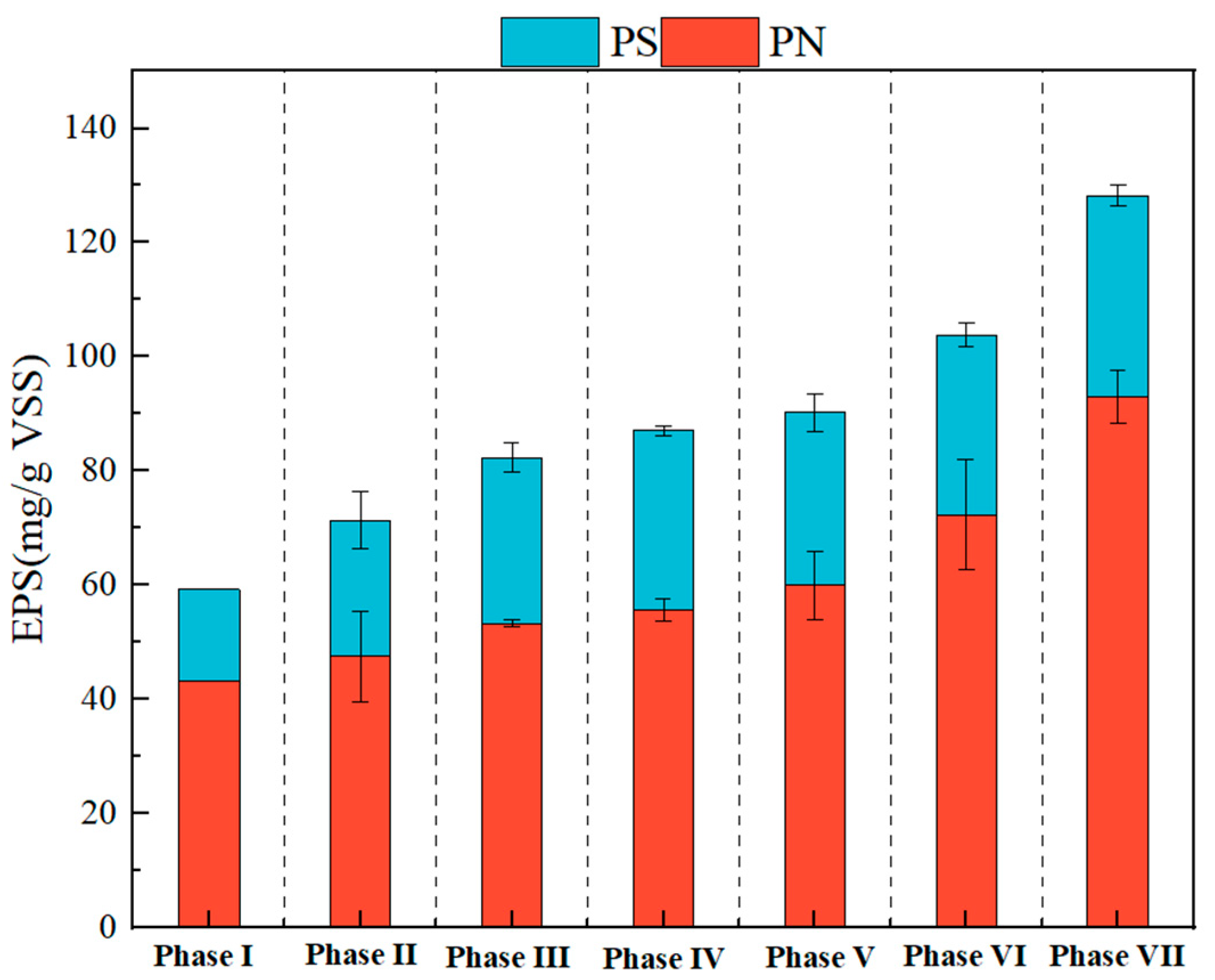
3.2. Contents and Characteristics of EPS in Partial Denitrification Sludge
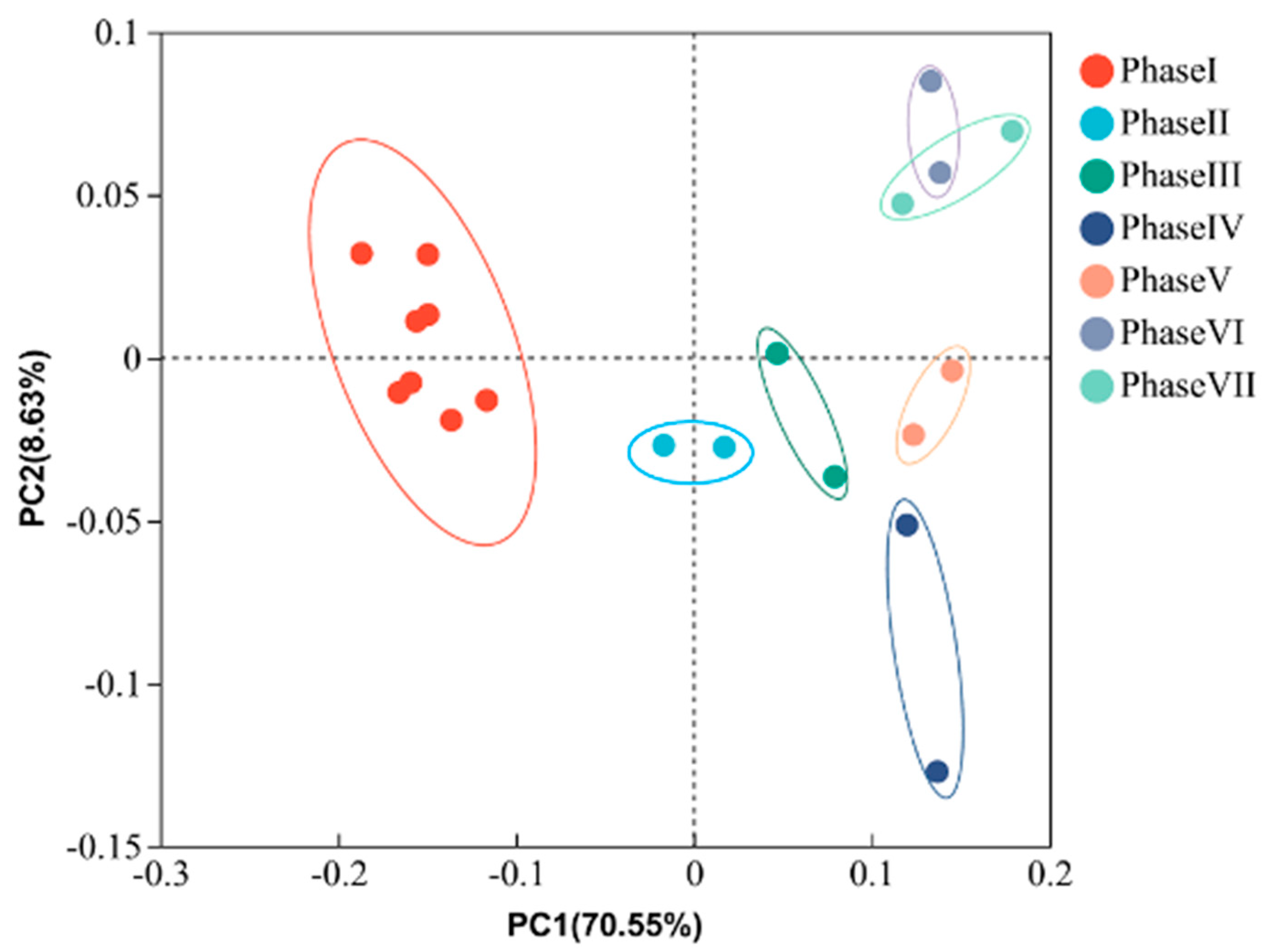
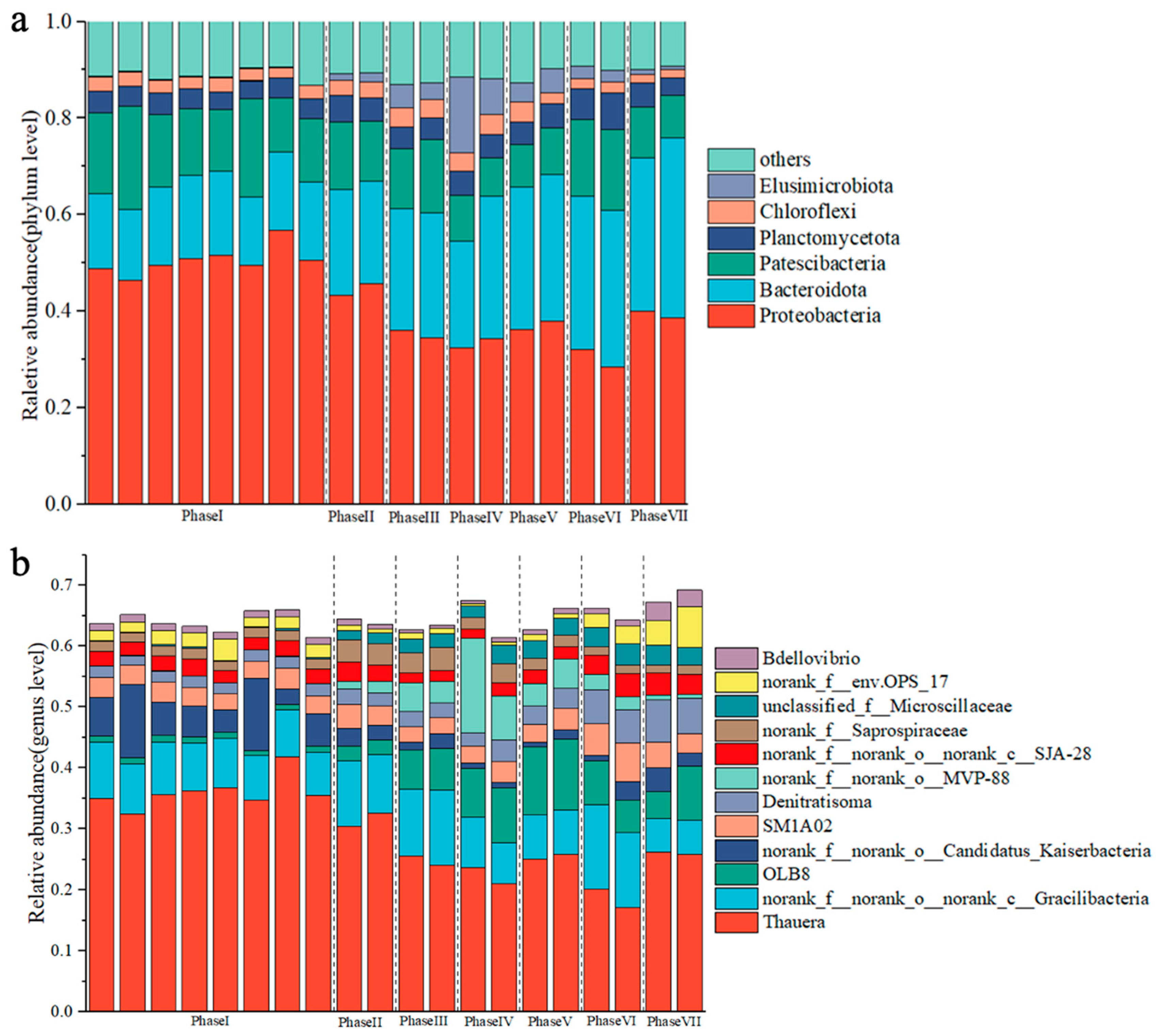
3.3. Effects of PFOA Stress on the Microbial Community Structures
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Du, R.; Cao, S.; Zhang, H.; Li, X.; Peng, Y. Flexible Nitrite Supply Alternative for Mainstream Anammox: Advances in Enhancing Process Stability. Environ. Sci. Technol. 2020, 54, 6353–6364. [Google Scholar] [CrossRef]
- Du, R.; Peng, Y.; Ji, J.; Shi, L.; Gao, R.; Li, X. Partial denitrification providing nitrite: Opportunities of extending application for anammox. Environ. Int. 2019, 131, 105001. [Google Scholar] [CrossRef]
- Zhang, J.; Peng, Y.; Li, X.; Du, R. Feasibility of partial-denitrification/anammox for pharmaceutical wastewater treatment in a hybrid biofilm reactor. Water Res. 2022, 208, 117856. [Google Scholar] [CrossRef]
- Izadi, P.; Sinha, P.; Andalib, M.; Samberger, C.; Lehman, G.; Messologitis, K.; Jacangelo, J. Coupling fundamental mechanisms and operational controls in mainstream partial denitrification for partial denitrification anammox applications: A review. J. Clean. Prod. 2023, 400, 136741. [Google Scholar] [CrossRef]
- Al-Hazmi, H.E.; Maktabifard, M.; Grubba, D.; Majtacz, J.; Hassan, G.K.; Lu, X.; Piechota, G.; Mannina, G.; Bott, C.B.; Mąkinia, J. An advanced synergy of partial denitrification-anammox for optimizing nitrogen removal from wastewater: A review. Bioresour. Technol. 2023, 381, 129168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, L.; Maddela, N.R.; Li, H.; An, Y.; Li, S. Exposure to Cr(VI) affects partial denitrification process-nitrite accumulation, EPS characteristic and microbial community assembly. J. Environ. Chem. Eng. 2023, 11, 109001. [Google Scholar] [CrossRef]
- Liu, Q.; Peng, Y.; Zhao, Y.; Zhao, Q.; Li, X.; Zhang, Q.; Sui, J.; Wang, C.; Li, J. Excellent anammox performance driven by stable partial denitrification when encountering seasonal decreasing temperature. Bioresour. Technol. 2022, 364, 128041. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.F.; Liu, X.H.; Fan, X.Y.; Dai, H.H. Effects of triclosan on performance, microbial community and antibiotic resistance genes during partial denitrification in a sequencing moving bed biofilm reactor. Bioresour. Technol. 2019, 281, 326–334. [Google Scholar] [CrossRef]
- Zeng, Z.; Wang, Y.; Zhu, W.; Xie, T.; Li, L. Effect of COD/NO(3) (-)-N ratio on nitrite accumulation and microbial behavior in glucose-driven partial denitrification system. Heliyon 2023, 9, e14920. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, J.; Liu, Q.; Fan, Y.; Zhu, C.; Liu, Y.; He, C.; Wu, J. Nitrite accumulation and microbial behavior by seeding denitrifying phosphorus removal sludge for partial denitrification (PD): The effect of COD/NO(3)(-) ratio. Bioresour. Technol. 2021, 323, 124524. [Google Scholar] [CrossRef]
- Qian, W.; Ma, B.; Li, X.; Zhang, Q.; Peng, Y. Long-term effect of pH on denitrification: High pH benefits achieving partial-denitrification. Bioresour. Technol. 2019, 278, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, H.; Zhang, S.; Chen, S.; Zhang, L. Stress responses of partial denitrification system under long-term ciprofloxacin exposure in an anaerobic sequencing batch reactor. J. Environ. Chem. Eng. 2023, 11, 110141. [Google Scholar] [CrossRef]
- Alharthi, F.A.; Alshammari, R.H.; Hasan, I. In Situ Polyaniline Immobilized ZnO Nanorods for Efficient Adsorptive Detoxification of Cr (VI) from Aquatic System. Water 2023, 15, 1949. [Google Scholar] [CrossRef]
- Chinnadurai, K.; Prema, P.; Veeramanikandan, V.; Kumar, K.R.; Nguyen, V.-H.; Marraiki, N.; Zaghloul, N.S.S.; Balaji, P. Toxicity evaluation and oxidative stress response of fumaronitrile, a persistent organic pollutant (POP) of industrial waste water on tilapia fish (Oreochromis mossambicus). Environ. Res. 2022, 204, 112030. [Google Scholar] [CrossRef] [PubMed]
- Guelfo, J.L.; Korzeniowski, S.; Mills, M.A.; Anderson, J.; Anderson, R.H.; Arblaster, J.A.; Conder, J.M.; Cousins, I.T.; Dasu, K.; Henry, B.J.; et al. Environmental Sources, Chemistry, Fate, and Transport of Per- and Polyfluoroalkyl Substances: State of the Science, Key Knowledge Gaps, and Recommendations Presented at the August 2019 SETAC Focus Topic Meeting. Environ. Toxicol. Chem. 2021, 40, 3234–3260. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, K.; Majeed, I.; Bilal, M.; Rasheed, T.; Shakeel, A.; Iqbal, S. Phytochemistry and Diverse Pharmacology of Genus Mimosa: A Review. Biomolecules 2022, 12, 83. [Google Scholar] [CrossRef]
- Mao, W.; Li, M.; Xue, X.; Cao, W.; Wang, X.; Xu, F.; Jiang, W. Bioaccumulation and toxicity of perfluorooctanoic acid and perfluorooctane sulfonate in marine algae Chlorella sp. Sci. Total Environ. 2023, 870, 161882. [Google Scholar] [CrossRef]
- Riaz, T.; Munnwar, A.; Shahzadi, T.; Zaib, M.; Shahid, S.; Javed, M.; Iqbal, S.; Rizwan, K.; Waqas, M.; Khalid, B.; et al. Phyto-mediated synthesis of nickel oxide (NiO) nanoparticles using leaves’ extract of Syzygium cumini for antioxidant and dyes removal studies from wastewater. Inorg. Chem. Commun. 2022, 142, 109656. [Google Scholar] [CrossRef]
- Sims, J.L.; Stroski, K.M.; Kim, S.; Killeen, G.; Ehalt, R.; Simcik, M.F.; Brooks, B.W. Global occurrence and probabilistic environmental health hazard assessment of per- and polyfluoroalkyl substances (PFASs) in groundwater and surface waters. Sci. Total Environ. 2022, 816, 151535. [Google Scholar] [CrossRef]
- Chiavola, A.; Di Marcantonio, C.; Boni, M.R.; Biagioli, S.; Frugis, A.; Cecchini, G. Experimental investigation on the perfluorooctanoic and perfluorooctane sulfonic acids fate and behaviour in the activated sludge reactor. Process Saf. Environ. Prot. 2020, 134, 406–415. [Google Scholar] [CrossRef]
- Xu, B.; Qiu, W.; Du, J.; Wan, Z.; Zhou, J.L.; Chen, H.; Liu, R.; Magnuson, J.T.; Zheng, C. Translocation, bioaccumulation, and distribution of perfluoroalkyl and polyfluoroalkyl substances (PFASs) in plants. iScience 2022, 25, 104061. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Zhang, N.; Yang, J.; Fu, Q.; Wang, Y.; Wang, D.; Tang, L.; Xia, J.; Liu, X.; Li, X.; et al. Interaction between perfluorooctanoic acid and aerobic granular sludge. Water Res. 2020, 169, 115249. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zou, M.; Zhou, Y.; Zeng, L.; Yang, X. Monitoring the nitrous oxide emissions and biological nutrient removal from wastewater treatment: Impact of perfluorooctanoic acid. J. Hazard. Mater. 2021, 402, 123469. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Su, C.; Fan, C.; Li, R.; Wang, Y.; Gao, S.; Chen, M. Long-term effect of perfluorooctanoic acid on the anammox system based on metagenomics: Performance, sludge characteristic and microbial community dynamic. Bioresour. Technol. 2022, 351, 127002. [Google Scholar] [CrossRef]
- Cao, L.; Liao, Y.; Su, C.; Tang, L.; Qi, Z.; Wei, L.; Wu, J.; Gao, S. Effects of PFOA on the physicochemical properties of anaerobic granular sludge: Performance evaluation, microbial community and metagenomic analysis. J. Environ. Manag. 2022, 313, 114936. [Google Scholar] [CrossRef]
- Chen, C.; Fang, Y.; Cui, X.; Zhou, D. Effects of trace PFOA on microbial community and metabolisms: Microbial selectivity, regulations and risks. Water Res. 2022, 226, 119273. [Google Scholar] [CrossRef]
- Cao, L.; Su, C.; Wu, J.; Wei, L.; Zhou, Y.; Tang, L.; Wang, Q.; Xian, Y. Impact of perfluorooctanoic acid on treatment wastewater by a tandem AnSBR-ASBR system: Performance, microbial community and metabolism pathway. Process Saf. Environ. Prot. 2022, 164, 373–383. [Google Scholar] [CrossRef]
- APHA. Standard Methods for Examination Water and Wastewater; American Public Health Association: Washington, DC, USA, 1995. [Google Scholar]
- Hou, Z.; Dong, W.; Wang, H.; Zhao, Z.; Li, Z.; Liu, H.; Li, Y.; Zeng, Z.; Xie, J.; Zhang, L.; et al. Response of nitrite accumulation to elevated C/NO- 3-N ratio during partial denitrification process: Insights of extracellular polymeric substance, microbial community and metabolic function. Bioresour. Technol. 2023, 384, 129269. [Google Scholar] [CrossRef]
- Tan, C.H.; Koh, K.S.; Xie, C.; Tay, M.; Zhou, Y.; Williams, R.; Ng, W.J.; Rice, S.A.; Kjelleberg, S. The role of quorum sensing signalling in EPS production and the assembly of a sludge community into aerobic granules. ISME J. 2014, 8, 1186–1197. [Google Scholar] [CrossRef]
- Yu, G.-H.; He, P.-J.; Shao, L.-M.; Zhu, Y.-S. Extracellular proteins, polysaccharides and enzymes impact on sludge aerobic digestion after ultrasonic pretreatment. Water Res. 2008, 42, 1925–1934. [Google Scholar] [CrossRef]
- Yu, J.; Xiao, K.; Xu, H.; Li, Y.; Xue, Q.; Xue, W.; Zhang, A.; Wen, X.; Xu, G.; Huang, X. Spectroscopic fingerprints profiling the polysaccharide/protein/humic architecture of stratified extracellular polymeric substances (EPS) in activated sludge. Water Res. 2023, 235, 119866. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, B.; Li, X.; Wang, S.; Wang, W.; Peng, Y. Enrichment of anammox biomass during mainstream wastewater treatment driven by achievement of partial denitrification through the addition of bio-carriers. J. Environ. Sci. 2024, 137, 181–194. [Google Scholar] [CrossRef]
- Yu, X.; Nishimura, F.; Hidaka, T. Impact of Long-Term Perfluorooctanoic Acid (PFOA) Exposure on Activated Sludge Process. Water Air Soil Pollut. 2018, 229, 134. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, D.; Zhu, X.; Zheng, X.; Feng, L. Long-term effects of copper nanoparticles on wastewater biological nutrient removal and N2O generation in the activated sludge process. Environ. Sci. Technol. 2012, 46, 12452–12458. [Google Scholar] [CrossRef]
- Jiao, Y.; Zou, M.; Yang, X.; Tsang, Y.F.; Chen, H. Perfluorooctanoic acid triggers oxidative stress in anaerobic digestion of sewage sludge. J. Hazard. Mater. 2022, 424, 127418. [Google Scholar] [CrossRef]
- Tang, L.; Su, C.; Wang, Q.; Cao, L.; Xian, Y.; Wen, S.; Zhou, Y.; Gao, S. Use of iron-loaded biochar to alleviate anammox performance inhibition under PFOA stress conditions: Integrated analysis of sludge characteristics and metagenomics. Sci. Total Environ. 2023, 865, 161178. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Song, M.; Zhou, Y. Redistribution of perfluorooctanoic acid in sludge after thermal hydrolysis: Location of protein plays a major role. Water Res. 2023, 241, 120135. [Google Scholar] [CrossRef]
- Du, M.; Xu, D.; Trinh, X.; Liu, S.; Wang, M.; Zhang, Y.; Wu, J.; Zhou, Q.; Wu, Z. EPS solubilization treatment by applying the biosurfactant rhamnolipid to reduce clogging in constructed wetlands. Bioresour. Technol. 2016, 218, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Chen, Y.; Wang, Y.; Kong, Z.; Cao, W.; Zhang, Y. Insight into the Impacts and Removal Pathways of Perfluorooctanoic Acid (PFOA) in Anaerobic Digestion. Water 2022, 14, 2255. [Google Scholar] [CrossRef]
- Yan, W.; Qian, T.; Zhang, L.; Wang, L.; Zhou, Y. Interaction of perfluorooctanoic acid with extracellular polymeric substances—Role of protein. J. Hazard. Mater. 2021, 401, 123381. [Google Scholar] [CrossRef]
- Huang, D.-Q.; Wang, Y.; Li, Z.-Y.; Huang, B.-C.; Yang, M.; Fan, N.-S.; Jin, R.-C. Metabolomics and molecular simulation reveal the responding mechanism of anammox consortia to perfluorooctanoic acid by regulating metabolic network. Chem. Eng. J. 2023, 460, 141712. [Google Scholar] [CrossRef]
- Li, W.; Li, L.; Li, B.; Peng, L.; Xu, Y.; Li, R.; Song, K. Effect and mechanism of perfluorooctanoic acid (PFOA) on anaerobic digestion sludge dewaterability. Chemosphere 2023, 335, 139142. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhou, J.; Lv, M.; Yu, H.; Zhao, H.; Xu, X. Specific component comparison of extracellular polymeric substances (EPS) in flocs and granular sludge using EEM and SDS-PAGE. Chemosphere 2015, 121, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, J.; Deng, D.; Li, R.; Guo, C.; Ma, J.; Chen, M. Investigation of extracellular polymeric substances (EPS) in four types of sludge: Factors influencing EPS properties and sludge granulation. J. Water Process Eng. 2021, 40, 101924. [Google Scholar] [CrossRef]
- Badireddy, A.R.; Korpol, B.R.; Chellam, S.; Gassman, P.L.; Engelhard, M.H.; Lea, A.S.; Rosso, K.M. Spectroscopic Characterization of Extracellular Polymeric Substances from Escherichia coli and Serratia marcescens: Suppression Using Sub-Inhibitory Concentrations of Bismuth Thiols. Biomacromolecules 2008, 9, 3079–3089. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Liu, S.; Zhang, Z. Role of extracellular polymeric substance in determining the high aggregation ability of anammox sludge. Water Res. 2015, 75, 51–62. [Google Scholar] [CrossRef]
- Huang, D.; Xu, R.; Sun, X.; Li, Y.; Xiao, E.; Xu, Z.; Wang, Q.; Gao, P.; Yang, Z.; Lin, H.; et al. Effects of perfluorooctanoic acid (PFOA) on activated sludge microbial community under aerobic and anaerobic conditions. Environ. Sci. Pollut. Res. 2022, 29, 63379–63392. [Google Scholar] [CrossRef]
- Xu, R.; Tao, W.; Lin, H.; Huang, D.; Su, P.; Gao, P.; Sun, X.; Yang, Z.; Sun, W. Effects of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonic Acid (PFOS) on Soil Microbial Community. Microb. Ecol. 2022, 83, 929–941. [Google Scholar] [CrossRef]
- Xiao, J.; Huang, J.; Wang, Y.; Qian, X. The fate and behavior of perfluorooctanoic acid (PFOA) in constructed wetlands: Insights into potential removal and transformation pathway. Sci. Total Environ. 2023, 861, 160309. [Google Scholar] [CrossRef]
- Uzun, M.; Koziaeva, V.; Dziuba, M.; Alekseeva, L.; Krutkina, M.; Sukhacheva, M.; Baslerov, R.; Grouzdev, D. Recovery and genome reconstruction of novel magnetotactic Elusimicrobiota from bog soil. ISME J. 2023, 17, 204–214. [Google Scholar] [CrossRef]
- Zhao, F.; Yang, L.; Zhang, T.; Zhuang, D.; Wu, Q.; Yu, J.; Tian, C.; Zhang, Z. Gut microbiome signatures of extreme environment adaption in Tibetan pig. NPJ Biofilms Microbiomes 2023, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Qian, W.; Yuan, C.; Yuan, Z.; Peng, Y. Achieving Mainstream Nitrogen Removal through Coupling Anammox with Denitratation. Environ. Sci. Technol. 2017, 51, 8405–8413. [Google Scholar] [CrossRef] [PubMed]
- de Almeida Fernandes, L.; Pereira, A.D.; Leal, C.D.; Davenport, R.; Werner, D.; Filho, C.R.M.; Bressani-Ribeiro, T.; de Lemos Chernicharo, C.A.; de Araujo, J.C. Effect of temperature on microbial diversity and nitrogen removal performance of an anammox reactor treating anaerobically pretreated municipal wastewater. Bioresour. Technol. 2018, 258, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, Y.; Chen, P.; Sathishkumar, K.; Lu, Y.; Naraginti, S.; Wu, Y.; Wu, H. Biological mediated synthesis of reduced graphene oxide (rGO) as a potential electron shuttle for facilitated biological denitrification: Insight into the electron transfer process. J. Environ. Chem. Eng. 2022, 10, 108225. [Google Scholar] [CrossRef]
- Huang, X.; Yao, K.; Yu, J.; Dong, W.; Zhao, Z. Nitrogen removal performance and microbial characteristics during simultaneous chemical phosphorus removal process using Fe(3). Bioresour. Technol. 2022, 363, 127972. [Google Scholar] [CrossRef]
- Li, J.; Ma, J.; Liao, H.; Li, X.; Shen, L.; Lin, H.; Sun, L.; Ou, R.; He, D. Hot-pressed membrane assemblies enhancing the biofilm formation and nitrogen removal in a membrane-aerated biofilm reactor. Sci. Total Environ. 2022, 833, 155003. [Google Scholar] [CrossRef]
- Kondrotaite, Z.; Valk, L.C.; Petriglieri, F.; Singleton, C.; Nierychlo, M.; Dueholm, M.K.D.; Nielsen, P.H. Diversity and Ecophysiology of the Genus OLB8 and Other Abundant Uncultured Saprospiraceae Genera in Global Wastewater Treatment Systems. Front. Microbiol. 2022, 13, 917553. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Zhang, H.; Feng, Y.; Maddela, N.R.; Li, S.; Zhang, L. Insights into the Responses of the Partial Denitrification Process to Elevated Perfluorooctanoic Acid Stress: Performance, EPS Characteristic and Microbial Community. Water 2023, 15, 2977. https://doi.org/10.3390/w15162977
Zhang S, Zhang H, Feng Y, Maddela NR, Li S, Zhang L. Insights into the Responses of the Partial Denitrification Process to Elevated Perfluorooctanoic Acid Stress: Performance, EPS Characteristic and Microbial Community. Water. 2023; 15(16):2977. https://doi.org/10.3390/w15162977
Chicago/Turabian StyleZhang, Shaoqing, Hang Zhang, Yucheng Feng, Naga Raju Maddela, Shugeng Li, and Liqiu Zhang. 2023. "Insights into the Responses of the Partial Denitrification Process to Elevated Perfluorooctanoic Acid Stress: Performance, EPS Characteristic and Microbial Community" Water 15, no. 16: 2977. https://doi.org/10.3390/w15162977




