Photocatalytic Degradation of Humic Acid Using Bentonite@Fe3O4@ZnO Magnetic Nanocomposite: An Investigation of the Characterization of the Photocatalyst, Degradation Pathway, and Modeling by Solver Plugin
Abstract
:1. Introduction
2. Materials and Method
2.1. Chemicals and Instruments
2.2. Synthesis of BNTN@Fe3O4@ZnO Magnetic Nanocomposite
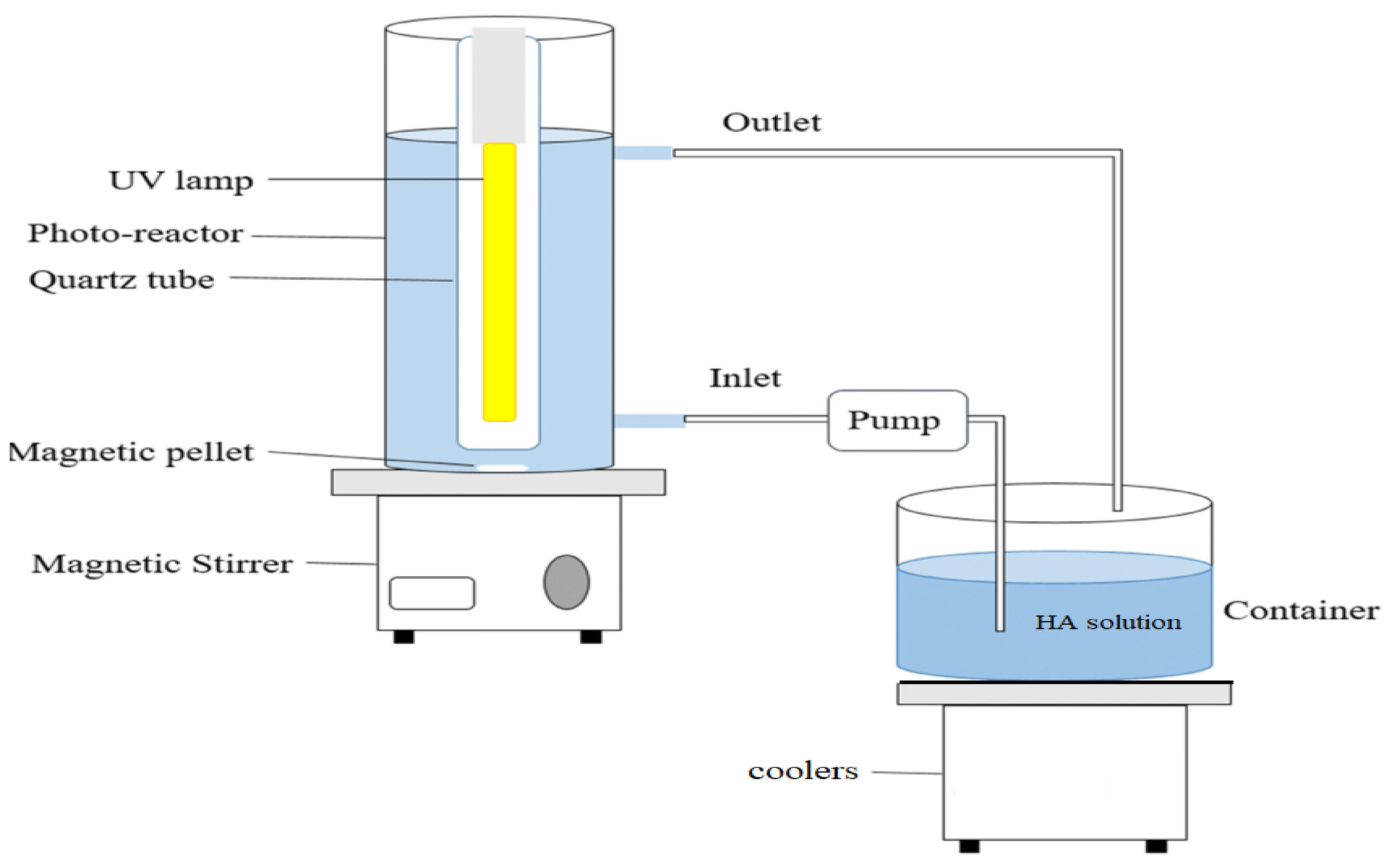
2.3. Photocatalytic Tests
2.4. Kinetic Study of HA Degradation
2.5. Reusability Tests
3. Results and Discussion
3.1. Study of Material Characterization
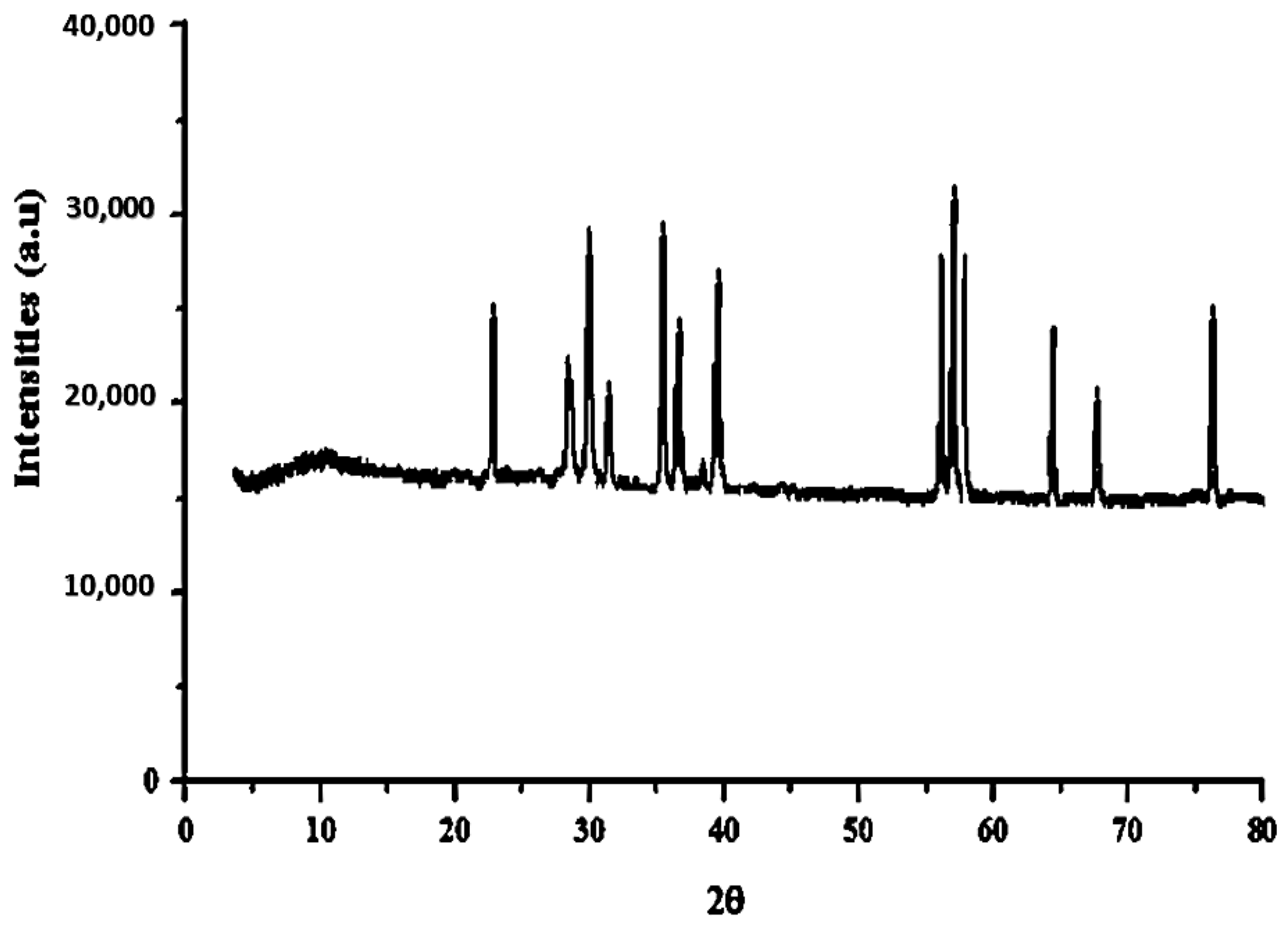
3.1.1. XRD Analysis
3.1.2. FTIR Analysis
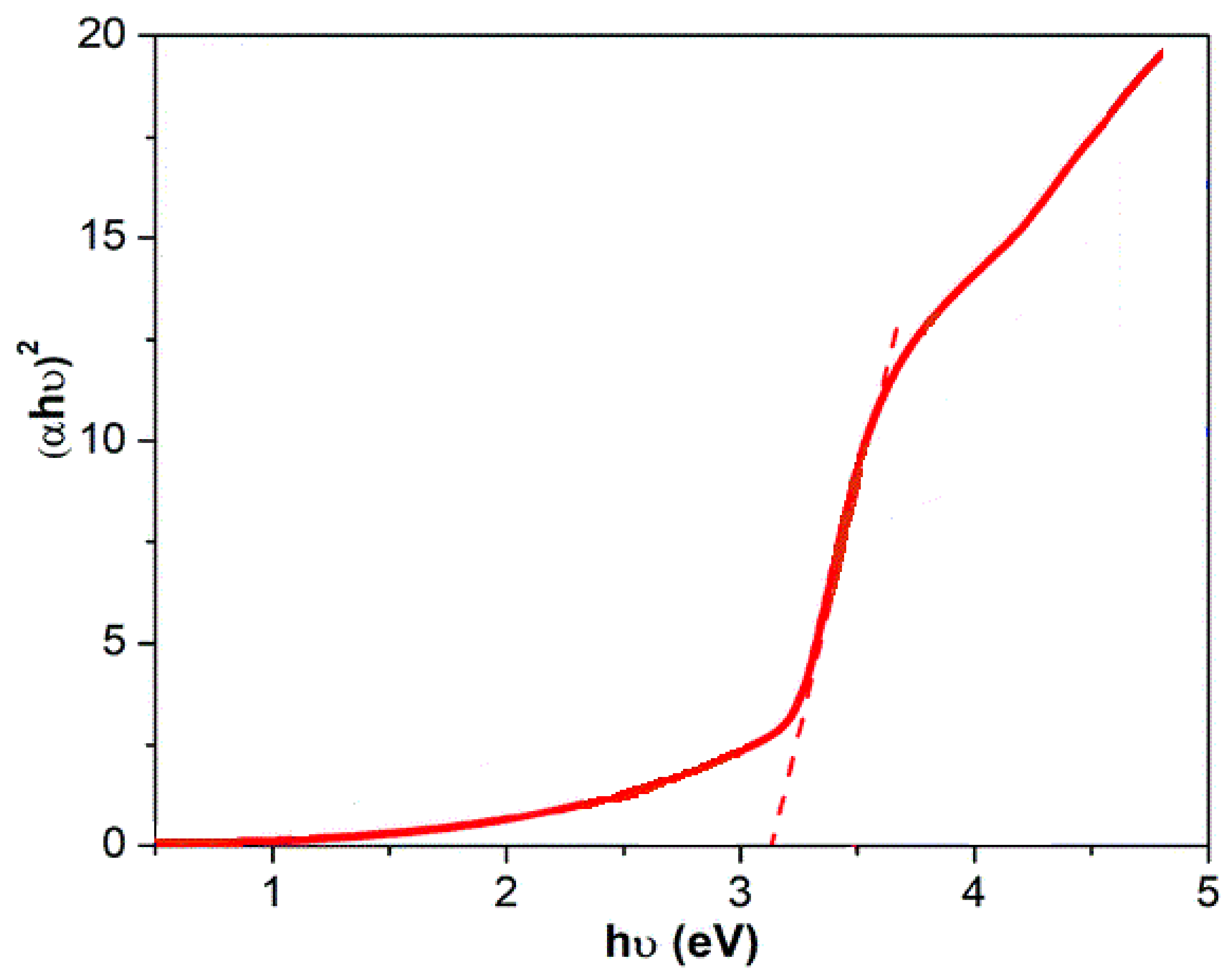
3.1.3. DRS Analysis
3.1.4. FESEM Analysis
3.1.5. EDX Analysis
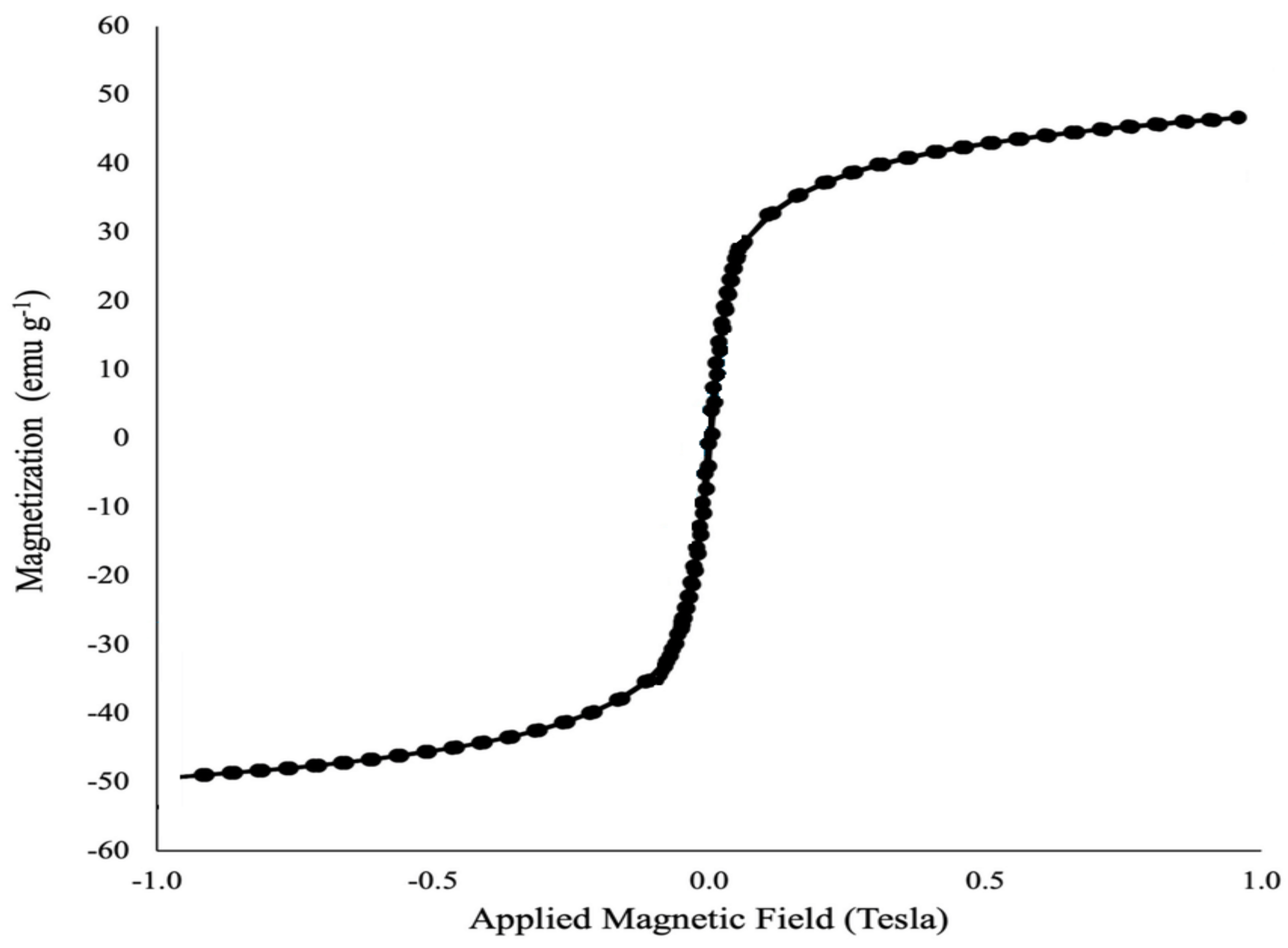
3.1.6. VSM Analysis
3.2. Effect of Environmental Variables
3.2.1. Influence of pH
3.2.2. Influence of Photocatalyst Dose
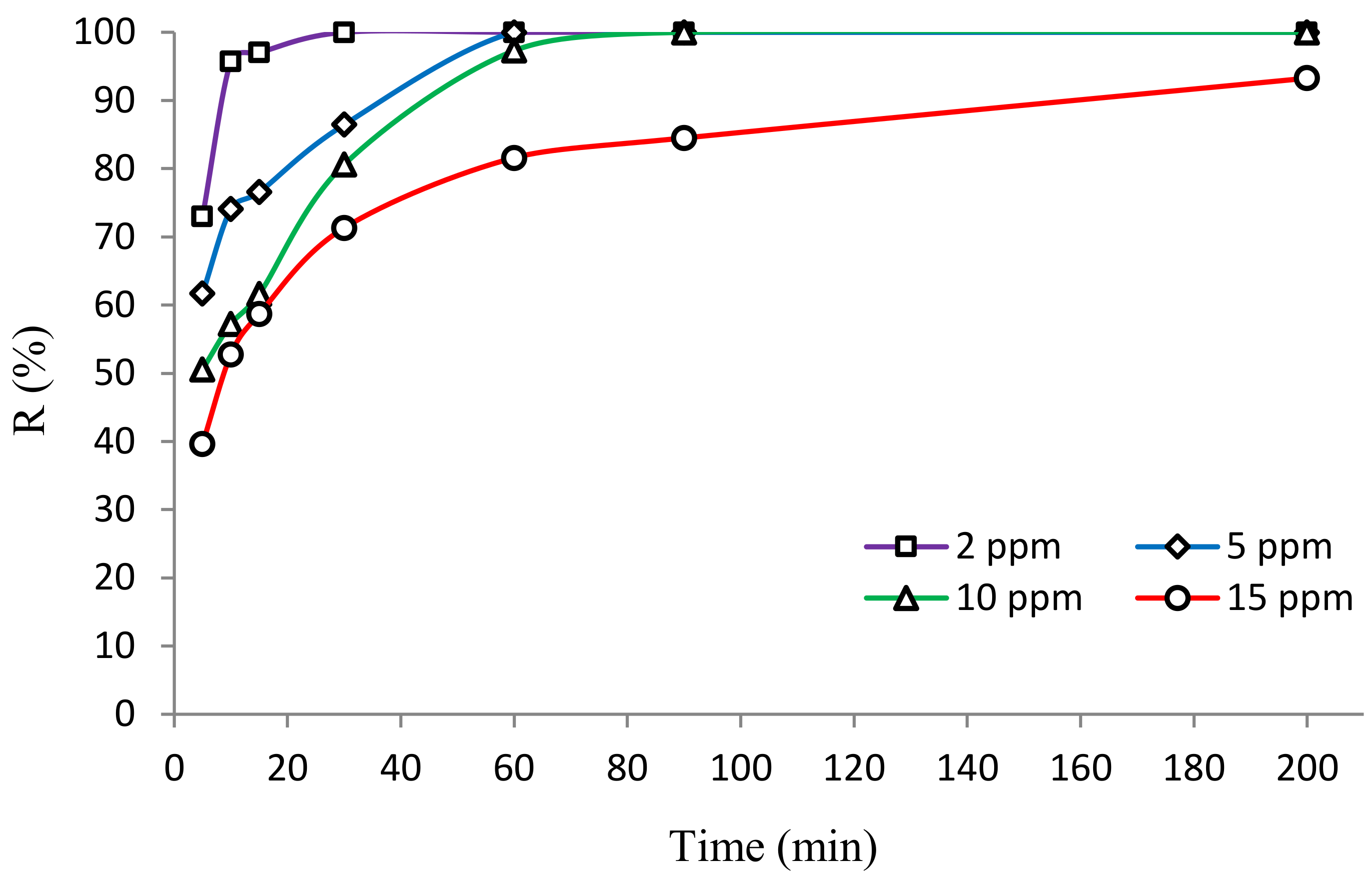
3.2.3. Influence of HA Concentration
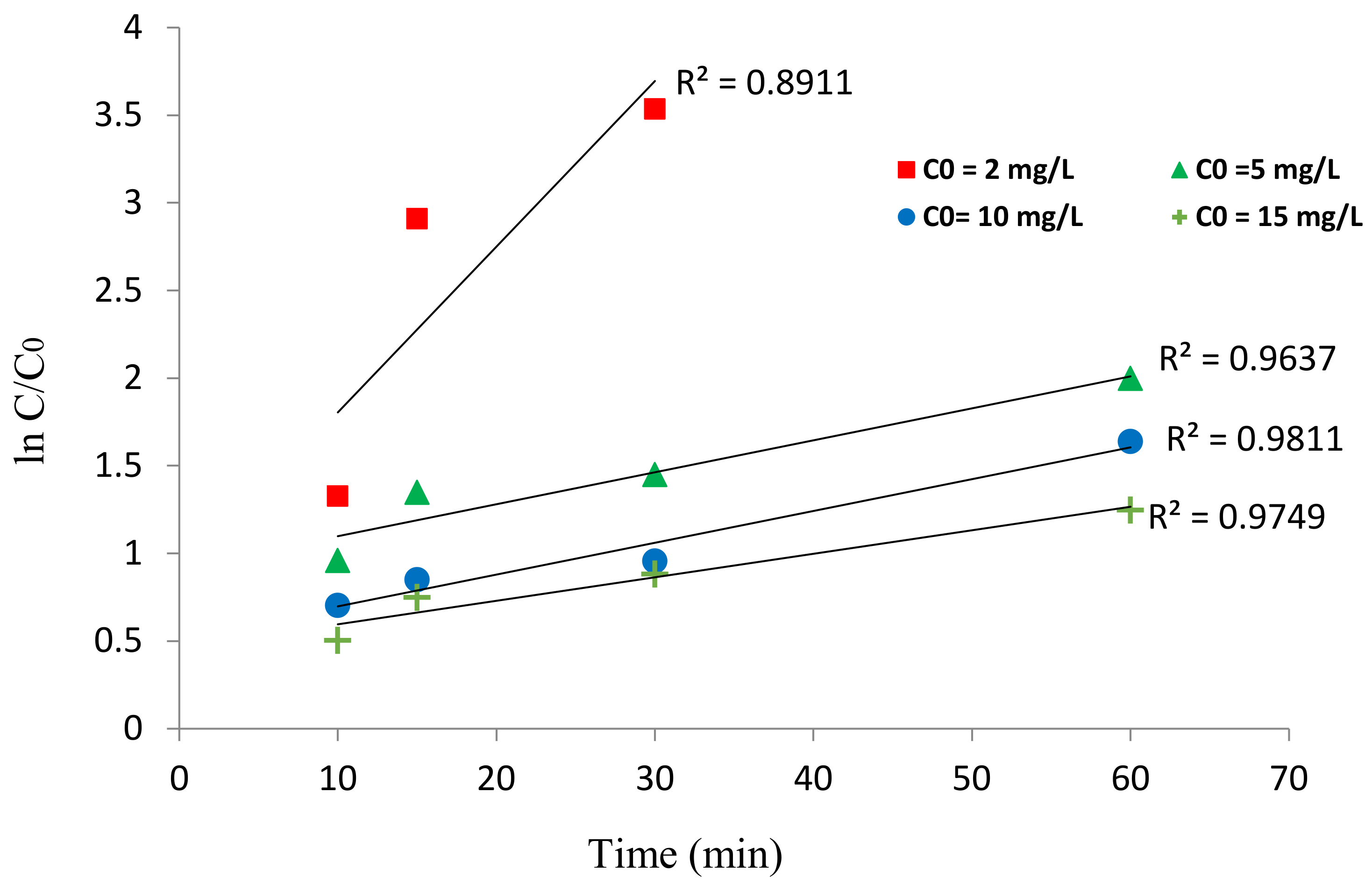
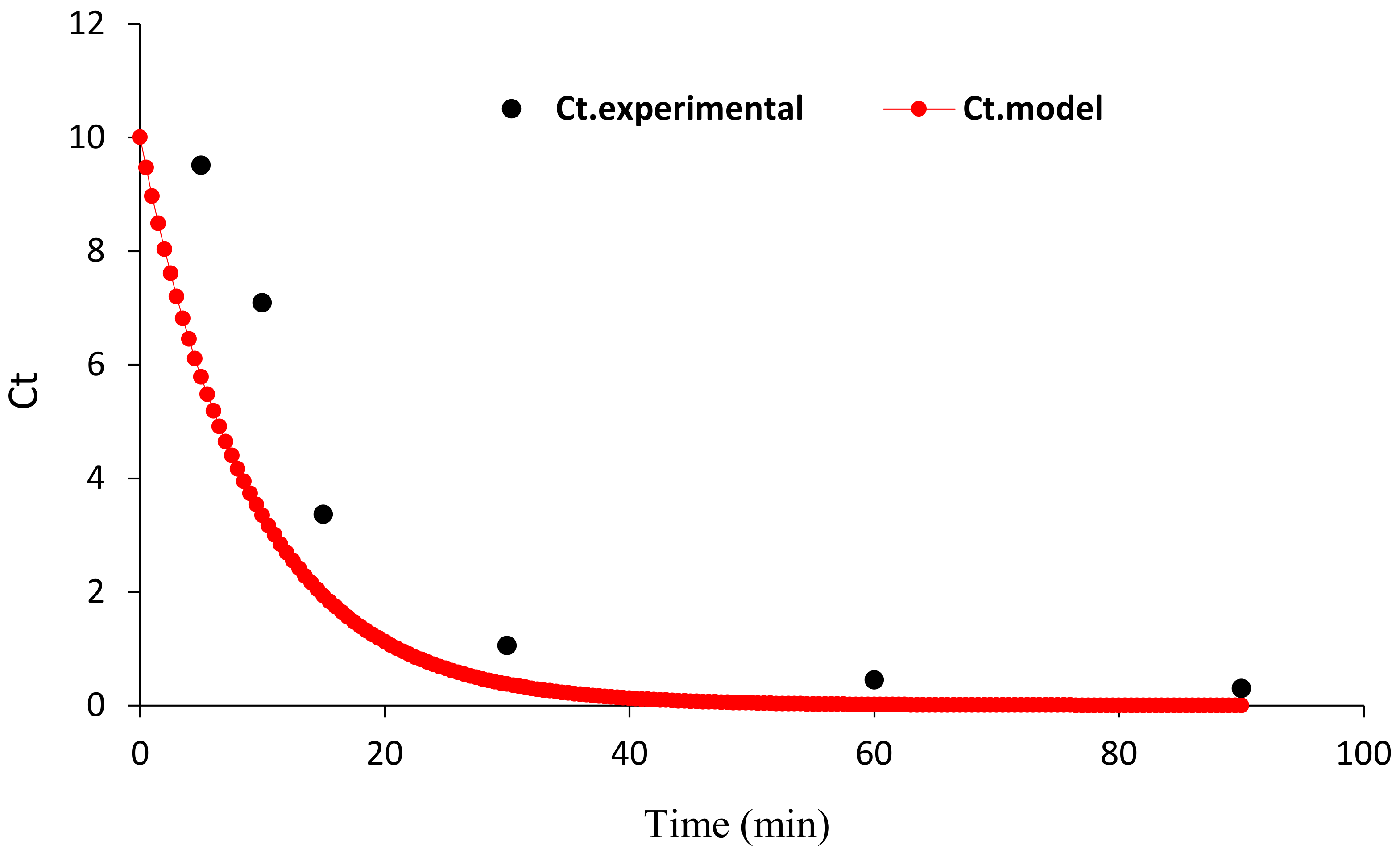
3.2.4. Kinetics of HA Degradation
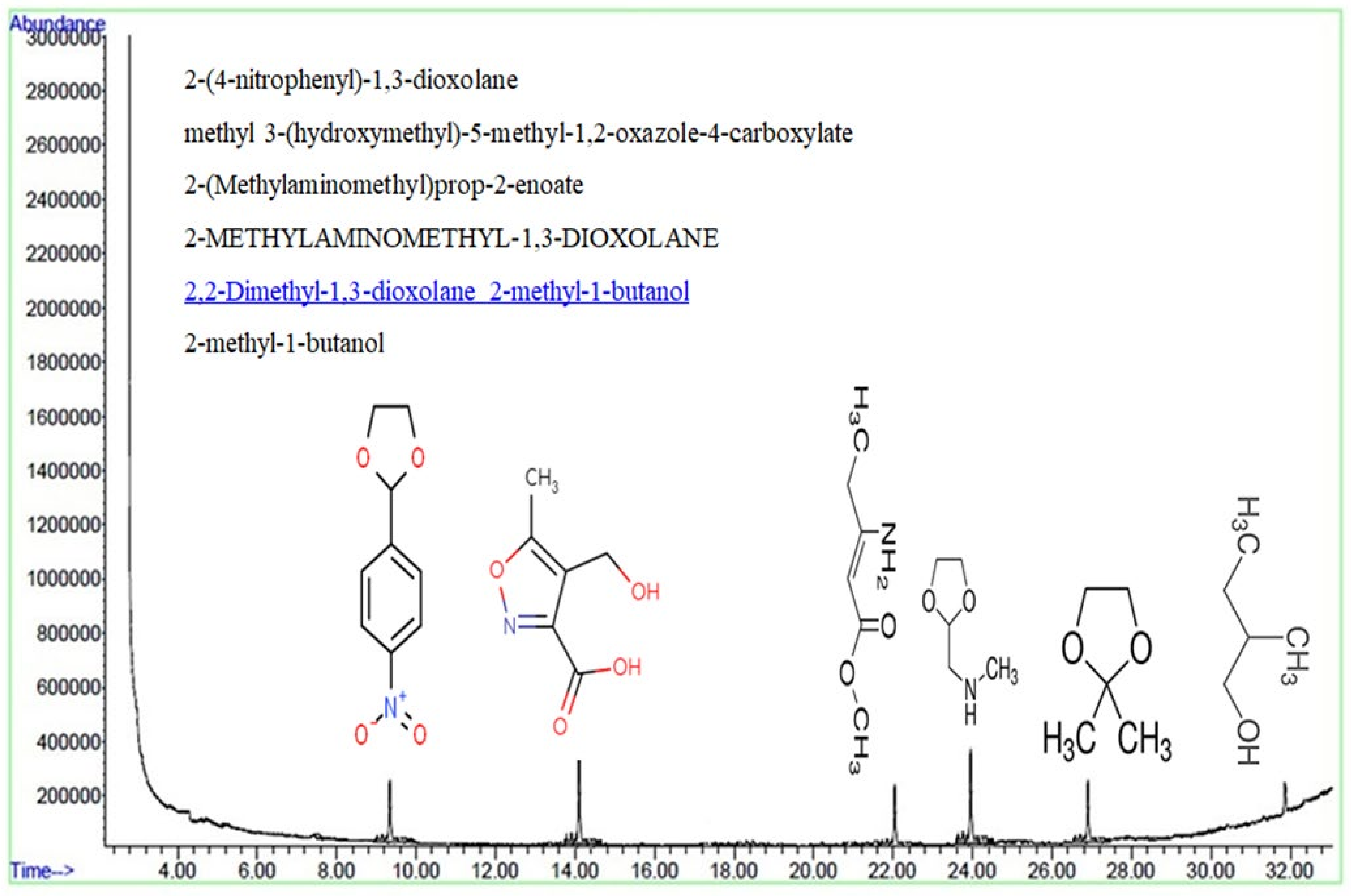
3.2.5. HA Degradation Mechanism
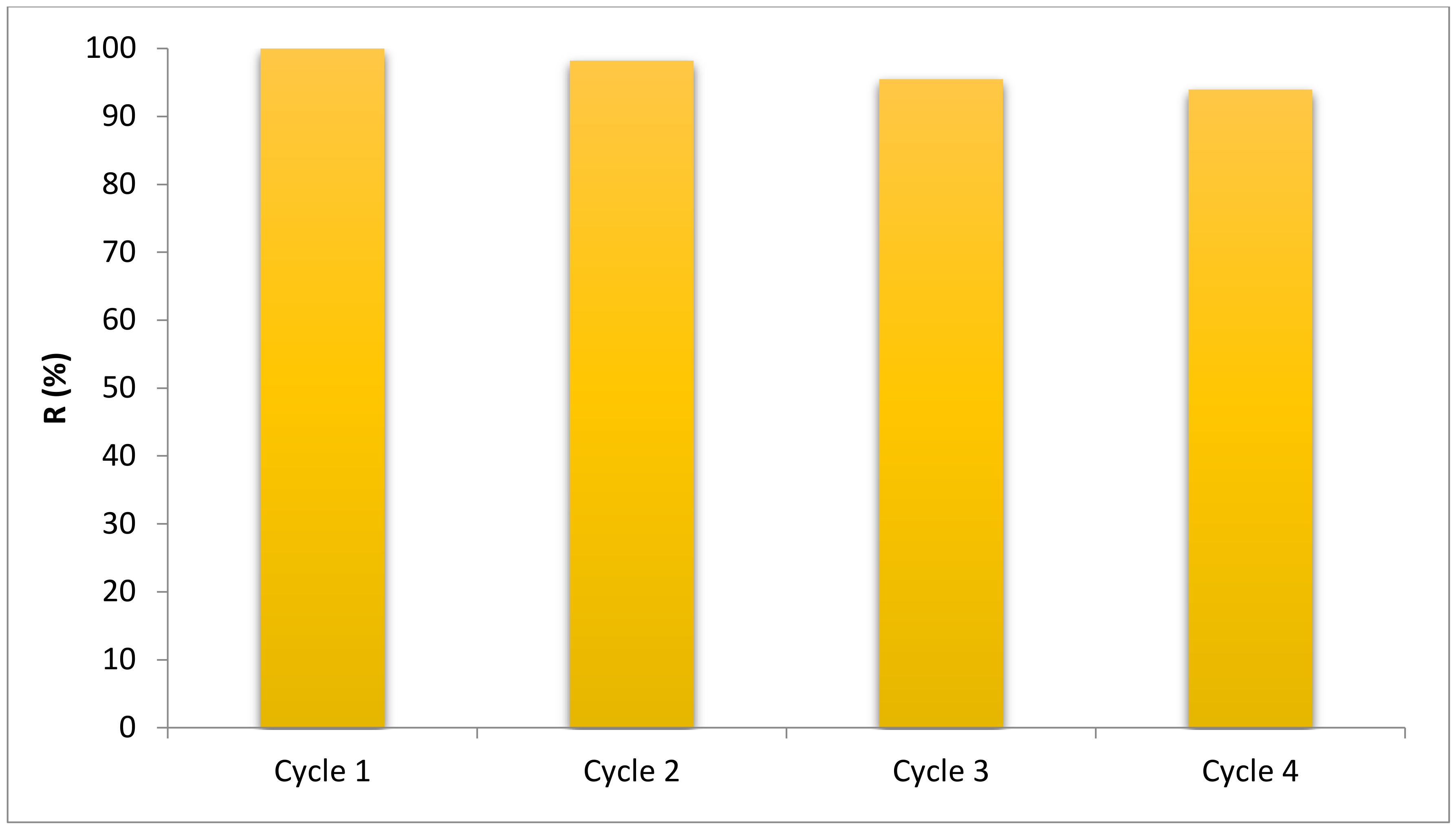
3.2.6. Reusability of BNTN@ Fe3O4@ ZnO
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guo, Z.; Zhan, R.; Shi, Y.; Zhu, D.; Pan, J.; Yang, C.; Wang, Y.; Wang, J. Innovative and green utilization of zinc-bearing dust by hydrogen reduction: Recovery of zinc and lead, and synergetic preparation of Fe/C micro-electrolysis materials. Chem. Eng. J. 2023, 456, 141157. [Google Scholar] [CrossRef]
- Foroutan, R.; Peighambardoust, S.J.; Amarzadeh, M.; Korri, A.K.; Peighambardoust, N.S.; Ahmad, A.; Ramavandi, B. Nickel ions abatement from aqueous solutions and shipbuilding industry wastewater using ZIF-8-chicken beak hydroxyapatite. J. Mol. Liq. 2022, 356, 119003. [Google Scholar] [CrossRef]
- Qin, Z.; Jin, J.; Liu, L.; Zhang, Y.; Du, Y.; Yang, Y. Reuse of soil-like material solidified by a biomass fly ash-based binder as engineering backfill material and its performance evaluation. J. Clean. Prod. 2023, 402, 136824. [Google Scholar] [CrossRef]
- Cao, T.-T.; Tie-Fu, X.; Zhao, M.-N.; Xu, J.; Cui, C.-W. Application of vacuum-ultraviolet (VUV) for phenolic homologues removal in humic acid solution: Efficiency, pathway and DFT calculation. J. Hazard. Mater. 2020, 384, 121464. [Google Scholar] [CrossRef]
- Zhan, Y.; Zhu, Z.; Lin, J.; Qiu, Y.; Zhao, J. Removal of humic acid from aqueous solution by cetylpyridinium bromide modified zeolite. J. Environ. Sci. 2010, 22, 1327–1334. [Google Scholar] [CrossRef]
- Wang, T.; Qu, G.; Ren, J.; Yan, Q.; Sun, Q.; Liang, D.; Hu, S. Evaluation of the potentials of humic acid removal in water by gas phase surface discharge plasma. Water Res. 2016, 89, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Han, C.; Nadagouda, M.N.; Dionysiou, D.D. An innovative zinc oxide-coated zeolite adsorbent for removal of humic acid. J. Hazard. Mater. 2016, 313, 283–290. [Google Scholar] [CrossRef] [Green Version]
- Tahmasebi, F.; Alimohammadi, M.; Nabizadeh, R.; Khoobi, M.; Karimian, K.; Zarei, A. Performance evaluation of graphene oxide coated on cotton fibers in removal of humic acid from aquatic solutions. Korean J. Chem. Eng. 2019, 36, 894–902. [Google Scholar] [CrossRef]
- Zhou, X.; Zhou, S.; Ma, F.; Xu, Y. Synergistic effects and kinetics of rGO-modified TiO2 nanocomposite on adsorption and photocatalytic degradation of humic acid. J. Environ. Manag. 2019, 235, 293–302. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Pang, W.Y.; Shafie, Z.M.H.M.; Zaulkiflee, N.D. PES/PVP/TiO2 mixed matrix hollow fiber membrane with antifouling properties for humic acid removal. J. Water Process Eng. 2019, 31, 100827. [Google Scholar] [CrossRef]
- Zghal, S.; Jedidi, I.; Cretin, M.; Cerneaux, S.; Abdelmouleh, M. One-step synthesis of highly porous carbon graphite/carbon nanotubes composite by in-situ growth of carbon nanotubes for the removal of humic acid and copper (II) from wastewater. Diam. Relat. Mater. 2020, 101, 107557. [Google Scholar] [CrossRef]
- Lin, Q.; Dong, F.; Li, C.; Cui, J. Disinfection byproduct formation from algal organic matters after ozonation or ozone combined with activated carbon treatment with subsequent chlorination. J. Environ. Sci. 2021, 104, 233–241. [Google Scholar] [CrossRef]
- Qin, J.J.; Oo, M.H.; Kekre, K.A.; Knops, F.; Miller, P. Impact of coagulation pH on enhanced removal of natural organic matter in treatment of reservoir water. Sep. Purif. Technol. 2006, 49, 295–298. [Google Scholar] [CrossRef]
- Eskandarinezhad, S.; Khosravi, R.K.R.; Amarzadeh, M.; Mondal, P.; Filho, F.J.C.M. Application of different Nanocatalysts in industrial effluent treatment: A review. J. Compos. Compd. 2021, 2, 43–56. [Google Scholar] [CrossRef]
- Liu, W.; Huang, F.; Liao, Y.; Zhang, J.; Ren, G.; Zhuang, Z.; Zhen, J.; Lin, Z.; Wang, C. Treatment of CrVI-Containing Mg(OH)2 Nanowaste. Angew. Chem. Int. Ed. 2008, 47, 5619–5622. [Google Scholar] [CrossRef]
- Gonçalves, N.P.; Minella, M.; Fabbri, D.; Calza, P.; Malitesta, C.; Mazzotta, E.; Prevot, A.B. Humic acid coated magnetic particles as highly efficient heterogeneous photo-Fenton materials for wastewater treatments. Chem. Eng. J. 2020, 390, 124619. [Google Scholar] [CrossRef]
- Geng, N.; Chen, W.; Xu, H.; Lin, T.; Ding, M.; Wang, Y.; Tao, H.; Hu, K. Preparation of Fe3O4/TiO2-N-GO sonocatalyst and using for humic acid removal with the assist of ultrasound. Mater. Sci. Semicond. Process. 2019, 102, 104593. [Google Scholar] [CrossRef]
- Menya, E.; Olupot, P.W.; Storz, H.; Lubwama, M.; Kiros, Y. Synthesis and evaluation of activated carbon from rice husks for removal of humic acid from water. Biomass Convers. Biorefinery 2022, 12, 3229–3248. [Google Scholar] [CrossRef]
- Li, S.; Li, Z.; Ke, B.; He, Z.; Cui, Y.; Pan, Z.; Li, D.; Huang, S.; Lai, C.; Su, J. Magnetic multi-walled carbon nanotubes modified with polyaluminium chloride for removal of humic acid from aqueous solution. J. Mol. Liq. 2019, 279, 241–250. [Google Scholar] [CrossRef]
- Liu, J.; Chen, C.; Zhang, K.; Zhang, L. Applications of metal–organic framework composites in CO2 capture and conversion. Chin. Chem. Lett. 2021, 32, 649–659. [Google Scholar] [CrossRef]
- Ghanbari, F.; Yaghoot-Nezhad, A.; Wacławek, S.; Lin, K.Y.A.; Rodríguez-Chueca, J.; Mehdipour, F. Comparative investigation of acetaminophen degradation in aqueous solution by UV/Chlorine and UV/H2O2 processes: Kinetics and toxicity assessment, process feasibility and products identification. Chemosphere 2021, 285, 131455. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Luo, H.; Jiang, Z.; Fang, D.; Chi, J.; Shangguan, W.; Wang, Z.; Wang, L.; Lee, A.F. Ge-Doped Cobalt Oxide for Electrocatalytic and Photocatalytic Water Splitting. ACS Catal. 2022, 12, 12000–12013. [Google Scholar] [CrossRef]
- Huang, Z.; Ding, J.; Yang, X.; Liu, H.; Song, P.; Guo, Y.; Guo, Y.; Wang, L.; Zhan, W. Highly Efficient Oxidation of Propane at Low Temperature over a Pt- Based Catalyst by Optimization Support. Environ. Sci. Technol. 2022, 56, 17278–17287. [Google Scholar] [CrossRef]
- Shi, Y.; Huang, J.; Zeng, G.; Cheng, W.; Yu, H.; Gu, Y.; Shi, L.; Yi, K. Stable, metal-free, visible-light-driven photocatalyst for efficient removal of pollutants: Mechanism of action. J. Colloid Interface Sci. 2018, 531, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Xin, Z.; Zhao, X.; Ji, H.; Ma, T.; Li, H.; Zhong, S.; Shen, Z. Amorphous carbon-linked TiO2/carbon nanotube film composite with enhanced photocatalytic performance: The effect of interface contact and hydrophilicity. Chin. Chem. Lett. 2021, 32, 2151–2154. [Google Scholar] [CrossRef]
- Peleyeju, M.G.; Viljoen, E.L. WO3-based catalysts for photocatalytic and photoelectrocatalytic removal of organic pollutants from water—A review. J. Water Process Eng. 2021, 40, 101930. [Google Scholar] [CrossRef]
- Shi, L.; Ren, Z.; Zhou, C.; Shen, L.; Bai, H. Numerical simulation of an entangled wire-silicone rubber continuous interpenetration structure based on domain meshing superposition method. Compos. Part B 2023, 256, 110648. [Google Scholar] [CrossRef]
- Zhang, X.; Ma, F.; Dai, Z.; Wang, J.; Chen, L.; Ling, H.; Soltanian, M.R. Radionuclide transport in multi-scale fractured rocks: A review. J. Hazard. Mater. 2022, 424, 127550. [Google Scholar] [CrossRef]
- Tang, X.; Tang, R.; Xiong, S.; Zheng, J.; Li, L.; Zhou, Z.; Gong, D.; Deng, Y.; Su, L.; Liao, C. Application of natural minerals in photocatalytic degradation of organic pollutants: A review. Sci. Total Environ. 2022, 812, 152434. [Google Scholar] [CrossRef]
- Zheng, R.; Gao, H.; Ren, Z.; Cen, D.; Chen, Z. Preparation of activated bentonite and its adsorption behavior on oil-soluble green pigment. Physicochem. Probl. Miner. Process. 2017, 53, 829–845. [Google Scholar] [CrossRef]
- Patehkhor, H.A.; Fattahi, M.; Khosravi-Nikou, M. Synthesis and characterization of ternary chitosan–TiO2–ZnO over graphene for photocatalytic degradation of tetracycline from pharmaceutical wastewater. Sci. Rep. 2021, 11, 1–17. [Google Scholar] [CrossRef]
- Tobajas, M.; Belver, C.; Rodriguez, J.J. Degradation of emerging pollutants in water under solar irradiation using novel TiO2-ZnO/clay nanoarchitectures. Chem. Eng. J. 2017, 309, 596–606. [Google Scholar] [CrossRef]
- Foroutan, R.; Peighambardoust, S.J.; Latifi, P.; Ahmadi, A.; Alizadeh, M.; Ramavandi, B. Carbon nanotubes/β-cyclodextrin/MnFe2O4 as a magnetic nanocomposite powder for tetracycline antibiotic decontamination from different aqueous environments. J. Environ. Chem. Eng. 2021, 9, 106344. [Google Scholar] [CrossRef]
- Xu, R.; Wang, Y.-N.; Sun, Y.; Wang, H.; Gao, Y.; Li, S.; Guo, L.; Gao, L. Ecotoxicology and Environmental Safety External sodium acetate improved Cr (VI) stabilization in a Cr-spiked soil during chemical-microbial reduction processes: Insights into Cr (VI) reduction performance, microbial community and metabolic functions. Ecotoxicol. Environ. Saf. 2023, 251, 114566. [Google Scholar] [CrossRef] [PubMed]
- Aragaw, T.A.; Bogale, F.M.; Aragaw, B.A. Iron-based nanoparticles in wastewater treatment: A review on synthesis methods, applications, and removal mechanisms. J. Saudi Chem. Soc. 2021, 25, 101280. [Google Scholar] [CrossRef]
- Norabadi, E.; Panahi, A.H.; Ghanbari, R.; Meshkinian, A.; Kamani, H.; Ashrafi, S.D. Optimizing the parameters of amoxicillin removal in a photocatalysis/ozonation process using Box–Behnken response surface methodology. Desalin. Water Treat. 2020, 192, 234–240. [Google Scholar] [CrossRef]
- Dai, Z.; Ma, Z.; Zhang, X.; Chen, J.; Ershadnia, R.; Luan, X.; Soltanian, M.R. An integrated experimental design framework for optimizing solute transport monitoring locations in heterogeneous sedimentary media. J. Hydrol. 2022, 614, 128541. [Google Scholar] [CrossRef]
- Birben, N.C.; Paganini, M.C.; Calza, P.; Bekbolet, M. Photocatalytic degradation of humic acid using a novel photocatalyst: Ce-doped ZnO. Photochem. Photobiol. Sci. 2017, 16, 24–30. [Google Scholar] [CrossRef]
- Khodadadi, M.; Al-Musawi, T.J.; Kamani, H.; Silva, M.F.; Panahi, A.H. The practical utility of the synthesis FeNi3@SiO2@TiO2 magnetic nanoparticles as an efficient photocatalyst for the humic acid degradation. Chemosphere 2020, 239, 124723. [Google Scholar] [CrossRef]
- Yaghoot-Nezhad, A.; Saebnoori, E.; Danaee, I.; Elahi, S.; Panah, N.B.; Khosravi-Nikou, M.R. Evaluation of the oxidative degradation of aromatic dyes by synthesized nano ferrate(VI) as a simple and effective treatment method. J. Water Process Eng. 2022, 49, 103017. [Google Scholar] [CrossRef]
- Yaghoot-Nezhad, A.; Moradi, M.; Rostami, M.; Danaee, I.; Khosravi-Nikou, M.R. Dual Z-Scheme CuO-ZnO@Graphitic Carbon Nitride Ternary Nanocomposite with Improved Visible Light-Induced Catalytic Activity for Ultrasound-Assisted Photocatalytic Desulfurization. Energy Fuels 2020, 34, 13588–13605. [Google Scholar] [CrossRef]
- Kavurmaci, S.S.; Bekbolet, M. Photocatalytic degradation of humic acid in the presence of montmorillonite. Appl. Clay Sci. 2013, 75–76, 60–66. [Google Scholar] [CrossRef]
- Li, X.; Yu, P.; Niu, X.; Yamaguchi, H.; Li, D. Non-contact manipulation of nonmagnetic materials by using a uniform magnetic field: Experiment and simulation. J. Magn. Magn. Mater. 2019, 497, 165957. [Google Scholar] [CrossRef]
- Wang, T.; Zhou, J.; Wang, W.; Zhu, Y.; Niu, J. Ag-single atoms modified S1.66-N1.91/TiO2-x for photocatalytic activation of peroxymonosulfate for bisphenol A degradation. Chin. Chem. Lett. 2022, 33, 2121–2124. [Google Scholar] [CrossRef]
- Li, F.; Wang, L.; Qu, G.; Hou, P.; Kong, L.; Huang, J.; Xu, X. An integrated approach to configure rGO/VS4/S composites with improved catalysis of polysulfides for advanced lithium–sulfur batteries. Chin. Chem. Lett. 2022, 33, 3909–3915. [Google Scholar] [CrossRef]
- Feng, C.; Lu, Z.; Zhang, Y.; Liang, Q.; Zhou, M.; Li, X.; Yao, C.; Li, Z.; Xu, S. A magnetically recyclable dual Z-scheme GCNQDs-CoTiO3/CoFe2O4 composite photocatalyst for efficient photocatalytic degradation of oxytetracycline. Chem. Eng. J. 2022, 435, 134833. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Q.; Cui, Q.; Ni, S.Q. Nitrogen recovery through fermentative dissimilatory nitrate reduction to ammonium (DNRA): Carbon source comparison and metabolic pathway. Chem. Eng. J. 2022, 441, 135938. [Google Scholar] [CrossRef]





| Molecular Structure | pKa | Molar Mass | Chemical Structure |
|---|---|---|---|
| C187H186O89N9S1 | <2 | 227.17 g/mol |  |
| Concentration (mg/L) | Equation | Kobs (min−1) | R2 | t1/2 (min) |
|---|---|---|---|---|
| 2 | y = 0.2207x + 0.3849 | 0.2207 | 0.9411 | 3.140009 |
| 5 | y = 0.0389x + 0.8563 | 0.0389 | 0.9637 | 17.81491 |
| 10 | y = 0.0380x + 0.4676 | 0.0538 | 0.9811 | 12.88104 |
| 15 | y = 0.0284x + 0.4211 | 0.0155 | 0.9749 | 44.70968 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hossein Panahi, A.; Al-Musawi, T.J.; Masihpour, M.; Fard, S.F.T.; Nasseh, N. Photocatalytic Degradation of Humic Acid Using Bentonite@Fe3O4@ZnO Magnetic Nanocomposite: An Investigation of the Characterization of the Photocatalyst, Degradation Pathway, and Modeling by Solver Plugin. Water 2023, 15, 2931. https://doi.org/10.3390/w15162931
Hossein Panahi A, Al-Musawi TJ, Masihpour M, Fard SFT, Nasseh N. Photocatalytic Degradation of Humic Acid Using Bentonite@Fe3O4@ZnO Magnetic Nanocomposite: An Investigation of the Characterization of the Photocatalyst, Degradation Pathway, and Modeling by Solver Plugin. Water. 2023; 15(16):2931. https://doi.org/10.3390/w15162931
Chicago/Turabian StyleHossein Panahi, Ayat, Tariq J. Al-Musawi, Mahdieh Masihpour, Seyedeh Fatemeh Tabatabaei Fard, and Negin Nasseh. 2023. "Photocatalytic Degradation of Humic Acid Using Bentonite@Fe3O4@ZnO Magnetic Nanocomposite: An Investigation of the Characterization of the Photocatalyst, Degradation Pathway, and Modeling by Solver Plugin" Water 15, no. 16: 2931. https://doi.org/10.3390/w15162931







