Acute Thermal Stress Reduces Skeletal Muscle Growth and Quality in Gibel Carp (Carassius gibelio)
Abstract
:1. Introduction
2. Materials and Methods
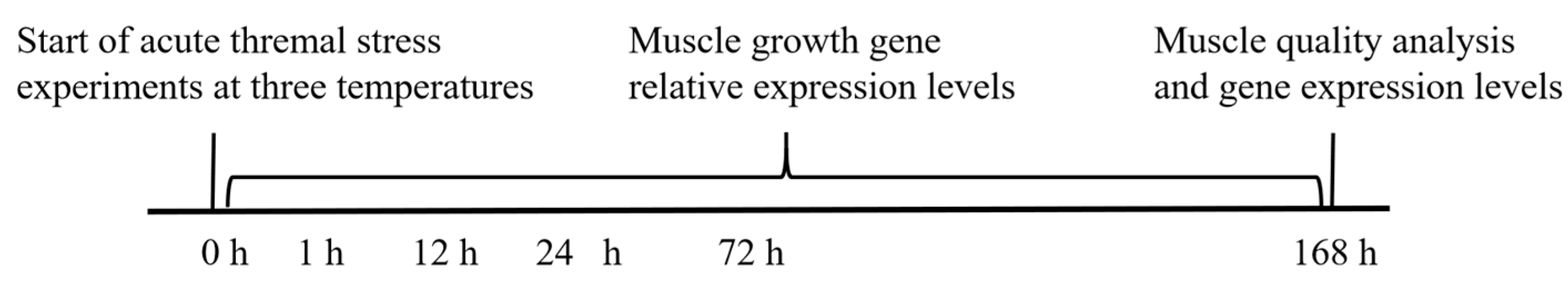
2.1. Fish, Experimental Design and Sample Collection
2.2. Cumulative Mortality Rate and Histological Observation
2.3. Muscle Texture Measurements
2.4. Chemical Analysis
2.5. RNA Extraction and RT-qPCR
2.6. Statistical Analysis
3. Results
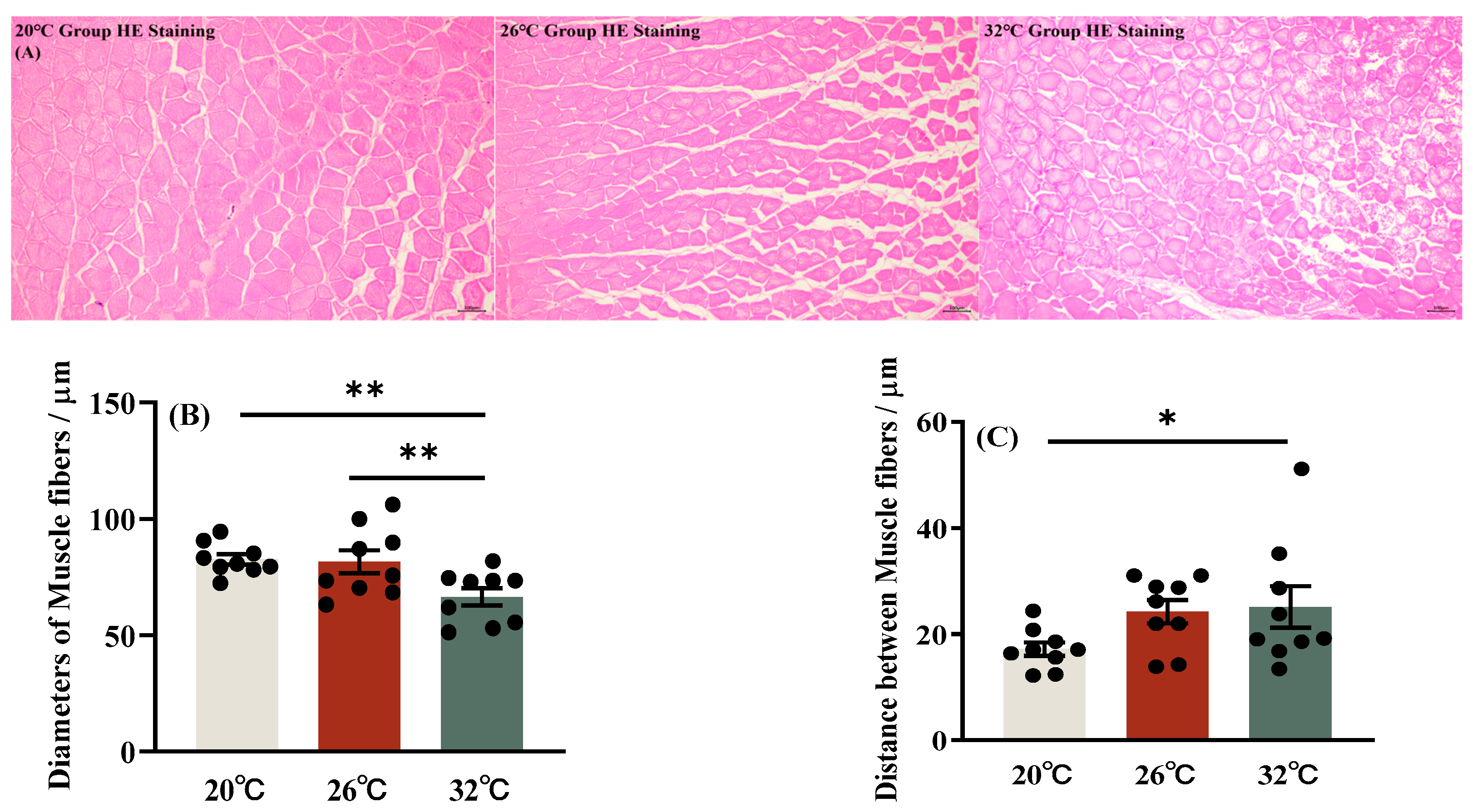
3.1. Cumulative Mortality Rate and Structure of Muscle Fiber of C. gibelio
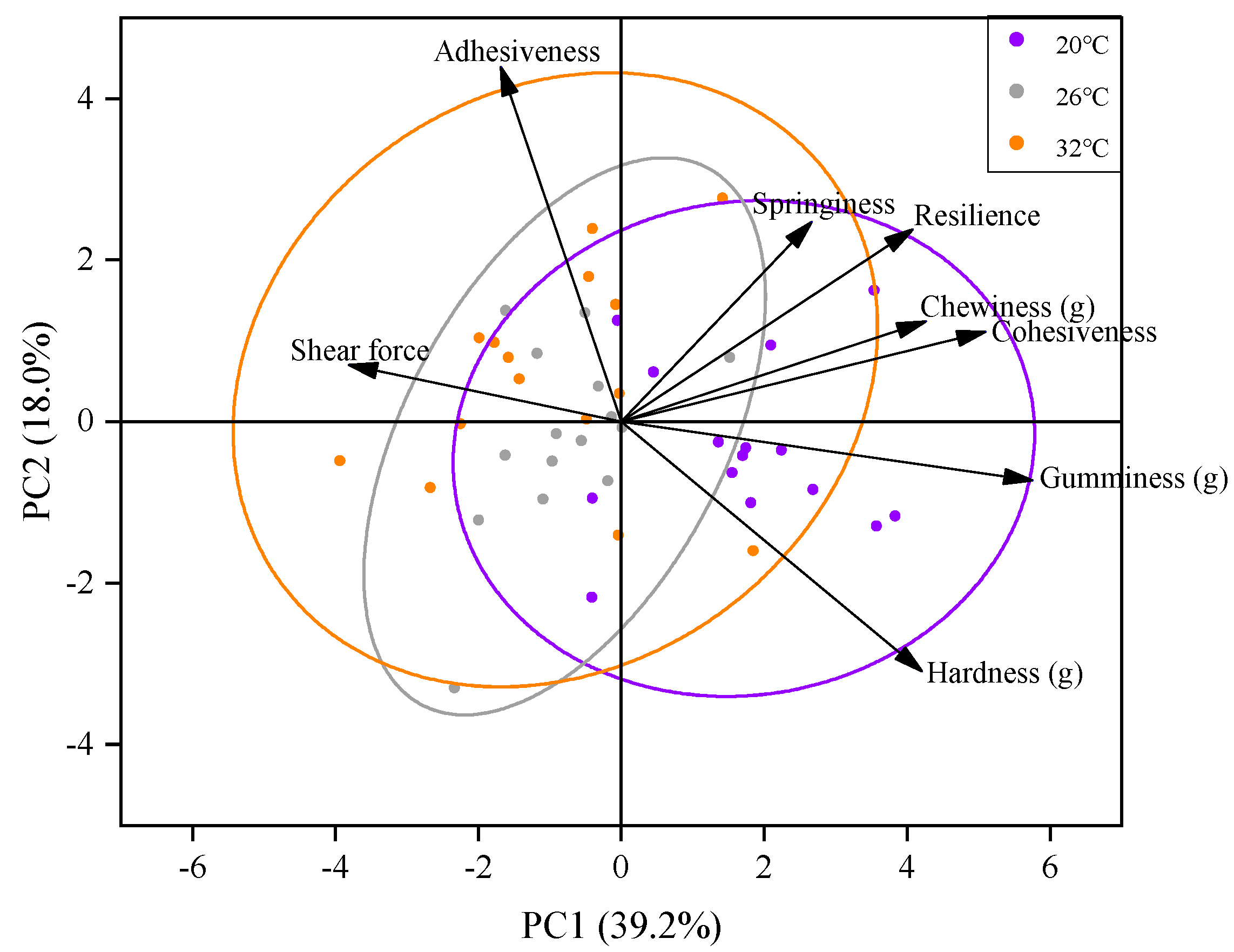
3.2. Effect of Acute Thermal Stress on Muscle Texture of C. gibelio
3.3. Muscle Nutrient Contents of C. gibelio
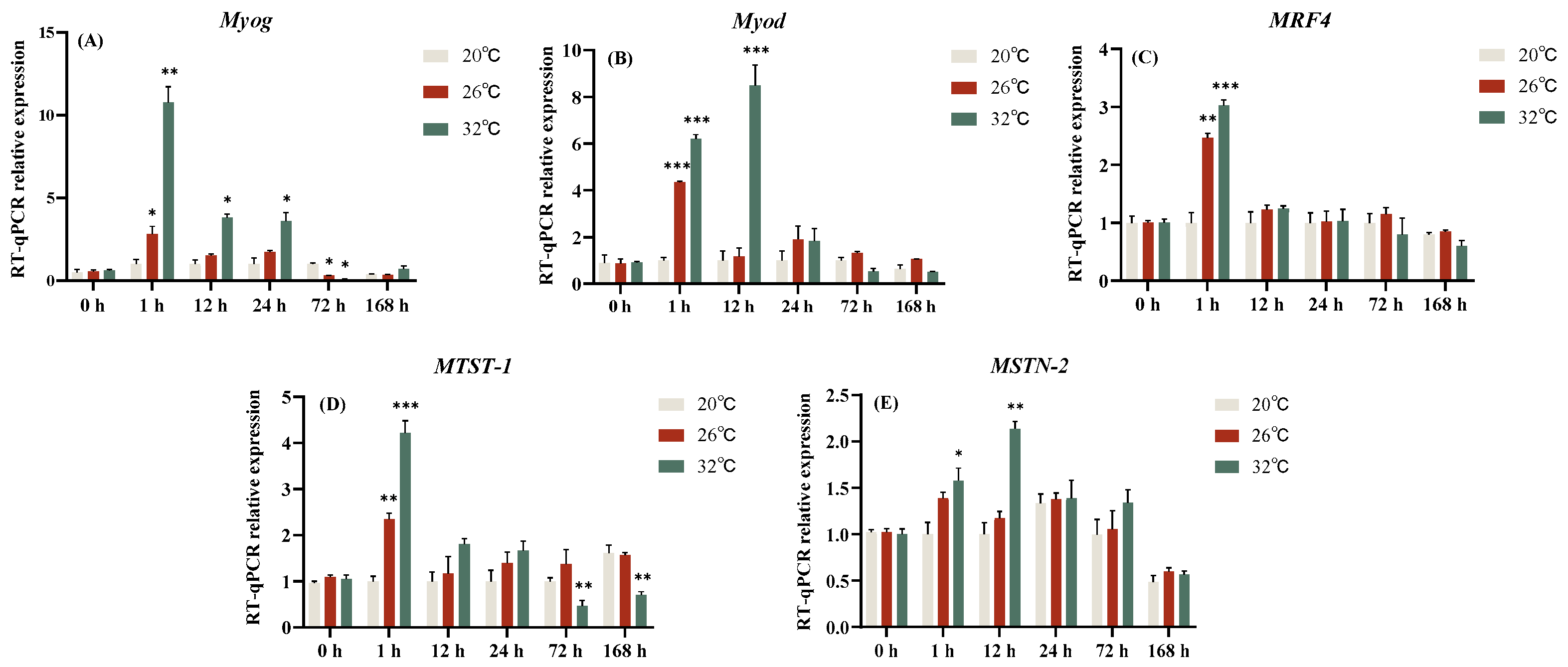
3.4. Effect of Acute Thermal Stress on MRFs Family Related Genes Expression Level of C. gibelio
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raval, A.; Ramanathan, V. Observational determination of the greenhouse effect. Nature 1989, 342, 758–761. [Google Scholar] [CrossRef]
- Peng, X.K.; Zhang, Y.; Huang, X.Y.; Zhou, G.C.; Xing, X.N.; Zhang, E.P. Effects of acute cold stress on immune function and expression of heat shock protein 70 family genes in different tissues in sheep. Acta. Vet. Zootec. Sin. 2019, 50, 1625–1634. [Google Scholar]
- Bolzan, L.P.; Barroso, D.C.; Souza, C.F.; Oliveira, F.C.; Wagner, R.; Baldisserotto, B.; Val, A.L.; Baldissera, M.D. Dietary supplementation with nerolidol improves the antioxidant capacity and muscle fatty acid profile of Brycon amazonicus exposed to acute heat stress. J. Therm. Biol. 2021, 99, 103003. [Google Scholar] [CrossRef]
- Wu, X.Y.; Lai, J.S.; Chen, Y.Y.; Liu, Y.; Song, M.J.; Li, F.Y.; Gong, Q. Characterization of MRF genes and their tissue distributions and analysis of the effects of starvation and refeeding on the expression of these genes in Acipenser dabryanus muscle. Comp. Biochem. Phys. B 2021, 256, 110648. [Google Scholar] [CrossRef]
- Habeeb, A.A.; Marai, I.F.; Kamal, T.H. Farm Animals and the Environment, 1st ed.; C.A.B. International: Örebro, Sweden, 1992; pp. 27–47. [Google Scholar]
- Sealey, W.M.; Gaylord, T.G.; Toner, M.; Ilgen, J.; Fraser, W.C.; Hooley, C.G.; Barrows, F.T. Growth and acute temperature tolerance of June Sucker juveniles fed varying dietary protein and lipid levels with and without supplemental dicalcium phosphate. N. Am. J. Aquac. 2013, 75, 124–132. [Google Scholar] [CrossRef]
- Doke, S.N.; Warrier, S.B.; Ninjoor, V.; Nadkarni, G.B. Role of hydrolytic enzymes in the spoilage of fish. J. Food. Sci. Technol. 1979, 16, 223–226. [Google Scholar]
- Paneru, B.D.; Al-Tobasei, R.; Kenney, B.; Kenney, B.; Leeds, T.D.; Salem, M. RNA-Seq reveals MicroRNA expression signature and genetic polymorphism associated with growth and muscle quality traits in rainbow trout. Sci. Rep. 2017, 7, 9078. [Google Scholar]
- Barbosa, V.; Maulvault, A.L.; Alves, R.N.; Anacleto, P.; Pousao-Ferreira, P.; Carvalho, M.; Nunes, M.L.; Rosa, R.; Marques, A. Will seabass (Dicentrarchus labrax) quality change in a warmer ocean? Food. Res. Int. 2017, 97, 27–36. [Google Scholar] [CrossRef]
- Schirone, R.C.; Gross, L. Effect of temperature on early embryological development of the zebra fish, Brachydanio rerio. J. Exp. Zool. 1968, 169, 43–52. [Google Scholar] [CrossRef]
- Björnsson, B.; Tryggvadóttir, S.V. Effects of size on optimal temperature for growth and growth efficiency of immature Atlantic halibut (Hippoglossus hippoglossus L.). Aquaculture 1996, 142, 33–42. [Google Scholar] [CrossRef]
- Mahanty, A.; Purohit, G.K.; Mohanty, S.; Mohanty, B.P. Heat stress–induced alterations in the expression of genes associated with gonadal integrity of the teleost Puntius sophore. Fish. Physiol. Biochem. 2019, 45, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wu, Z.; Song, Z.; Xiao, P.; Liu, Y.; Zhang, P.; You, F. Insight into the heat resistance of fish via blood: Effects of heat stress on metabolism, oxidative stress and antioxidant response of olive flounder Paralichthys olivaceus and turbot Scophthalmus maximus. Fish Shellfish. Immunol. 2016, 58, 125–135. [Google Scholar] [PubMed]
- López-Olmeda, J.F.; Sánchez-Vázquez, F.J. Thermal biology of zebrafish (Danio rerio). J. Therm. Biol. 2011, 36, 91–104. [Google Scholar] [CrossRef]
- Luo, S.W.; Cai, L.; Liu, Y.; Wang, W.N. Functional analysis of a dietary recombinant Fatty acid binding protein 10 (FABP10) on the Epinephelus coioides in response to acute low temperature challenge. Fish Shellfish. Immunol. 2014, 36, 475–484. [Google Scholar] [PubMed]
- Scott, G.R.; Johnston, I.A. Temperature during embryonic development has persistent effects on thermal acclimation capacity in zebrafish. Proc. Natl. Acad. Sci. USA 2012, 109, 14247–14252. [Google Scholar]
- Malek, R.L.; Sajadi, H.; Abraham, J.; Grundy, M.A.; Gerhard, G.S. The effects of temperature reduction on gene expression and oxidative stress in skeletal muscle from adult zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2004, 138, 363–373. [Google Scholar]
- Armobin, K.; Ahmadifar, E.; Adineh, H.; Samani, M.N.; Kalhor, N.; Yilmaz, S.; Van, D.H. Quercetin application for common carp (Cyprinus carpio): I. effects on growth performance, humoral immunity, antioxidant status, immune-related genes, and resistance against heat stress. Aquac. Nutr. 2023, 2023, 1168262. [Google Scholar]
- Moffitt, B.P.; Crawshaw, L.I. Effects of acute temperature changes on metabolism, heart rate, and ventilation frequency in carp Cyprinus carpio L. Physiol. Biochem. Zool. 1983, 56, 397–403. [Google Scholar]
- Johnston, I.A.; Alderson, R.; Sandham, C.; Dingwall, A.; Mitchell, D.; Selkirk, C.; Nickell, D.; Baker, R.; Robertson, B.; Whyte, D.; et al. Muscle fibre density in relation to the colour and texture of smoked Atlantic salmon (Salmo salar L.). Aquaculture 2000, 189, 335–349. [Google Scholar]
- Johnston, I.A.; Lee, H.T.; Macqueen, D.J.; Paranthaman, K.; Kawashima, C.; Anwar, A.; Kinghorn, J.R.; Dalmay, T. Embryonic temperature affects muscle fibre recruitment in adult zebrafish: Genome-wide changes in gene and microRNA expression associated with the transition from hyperplastic to hypertrophic growth phenotypes. J. Exp. Biol. 2009, 212, 1781–1793. [Google Scholar]
- López-Albors, O.; Ayala, M.D.; Gil, F.; Garcıa-Alcázar, A.; Abellán, E.; Latorre, R.; Ramírez-Zarzosa, G.; Vázquez, J.M. Early temperature effects on muscle growth dynamics and histochemical profile of muscle fibres of sea bass Dicentrarchus labrax L., during larval and juvenile stages. Aquaculture 2003, 220, 385–406. [Google Scholar]
- Refaey, M.M.; Li, D.; Tian, X.; Zhang, Z.; Zhang, X.; Li, L.; Tang, R. High stocking density alters growth performance, blood biochemistry, intestinal histology, and muscle quality of channel catfish Ictalurus punctatus. Aquaculture 2018, 492, 73–81. [Google Scholar]
- Bugeon, J.; Lefevre, F.; Fauconneau, B. Fillet texture and muscle structure in brown trout (Salmo trutta) subjected to long-term exercise. Aquac. Res. 2003, 34, 1287–1295. [Google Scholar]
- Zammit, P.S. Function of the myogenic regulatory factors Myf5, MyoD, Myogenin and MRF4 in skeletal muscle, satellite cells and regenerative myogenesis. Semin. Cell Dev. Biol. 2017, 72, 19–32. [Google Scholar] [CrossRef]
- Hernández, J.M.; García-González, E.G.; Brun, C.E.; Rudnicki, M.A. The myogenic regulatory factors, determinants of muscle development, cell identity and regeneration. Semin. Cell Dev. Biol. 2017, 72, 10–18. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lawler, A.M.; Lee, S.J. Regulation of skeletal muscle mass in mice by a new TGF-p superfamily member. Nature 1997, 387, 83–90. [Google Scholar]
- Wu, Y.; You, X.; Sun, W.; Xiong, G.Q.; Shi, L.; Qiao, Y.; Wu, W.J.; Xin, L.; Jun, W.; Anzi, D.; et al. Insight into acute heat stress on meat qualities of rainbow trout (Oncorhynchus mykiss) during short-time transportation. Aquaculture 2021, 543, 737013. [Google Scholar]
- Rudnicki, M.A.; Schnegelsberg, P.N.; Stead, R.H.; Braun, T.; Arnold, H.H.; Jaenisch, R. MyoD or Myf-5 is required for the formation of skeletal muscle. Cell 1993, 75, 1351–1359. [Google Scholar] [CrossRef]
- Tajbakhsh, S.; Rocancourt, D.; Cossu, G.; Buckingham, M. Redefining the genetic hierarchies controlling skeletal myogenesis: Pax-3 and Myf-5 act upstream of MyoD. Cell 1997, 89, 127–138. [Google Scholar] [PubMed] [Green Version]
- Campos, C.; Valente, L.; Conceicao, L.; Engrola, S.; Fernandes, J. Temperature affects methylation of the myogenin putative promoter, its expression and muscle cellularity in Senegalese sole larvae. Epigenetics 2013, 8, 389–397. [Google Scholar] [PubMed] [Green Version]
- De, P.T.G.; De, A.F.L.A.; Carani, F.R.; Vechetti-Júnior, I.J.; Padovani, C.R.; Salomao, R.A.S.; Dal-Pai-Silva, M.; Vander, B.; Maeli, D. Rearing temperature induces changes in muscle growth and gene expression in juvenile pacu (Piaractus mesopotamicus). Comp. Biochem. Phys. B 2014, 169, 31–37. [Google Scholar]
- Wilkes, D.; Xie, S.Q.; Stickland, N.C.; Alami-Durante, H.; Kentouri, M.; Sterioti, A.; Goldspink, G. Temperature and myogenic factor transcript levels during early development determines muscle growth potential in rainbow trout (Oncorhynchus mykiss) and sea bass (Dicentrarchus labrax). J. Exp. Biol. 2001, 204, 2763–2771. [Google Scholar]
- Zhou, L.; Wang, Y.; Gui, J.F. Analysis of genetic heterogeneity among five gynogenetic clones of silver crucian carp, Carassius auratus gibelio Bloch, based on detection of RAPD molecular markers. Cytogenet. Genome. Res. 2000, 88, 133–139. [Google Scholar]
- Li, Z.; Liang, H.; Wang, Z.; Zou, G.; Gui, J. Comparative analysis on the flesh quality and nutrient component of tetraploid gibel carp “changfeng” variety. Acta. Hydrobiol. Sin. 2016, 40, 853–858. [Google Scholar]
- Cang, P.; Zhang, M.; Qiao, G.; Sun, Q.; Xu, D.; Li, Q.; Yuan, X.; Liu, W. Analysis of Growth, Nutrition and Economic Profitability of Gibel Carp (Carassius auratus gibelio♀ × C. carpio♂) Cultured in Zero-water Exchange System. Pak. J. Zool. 2019, 51, 619. [Google Scholar] [CrossRef]
- Gui, J.; Zhu, Z. Molecular basis and genetic improvement of economically important traits in aquaculture animals. Chin. Sci. Bull. 2012, 57, 1751–1760. [Google Scholar] [CrossRef] [Green Version]
- Valente, L.M.; Moutou, K.A.; Conceicao, L.E.; Engrola, S.; Fernandes, J.M.; Johnston, I.A. What determines growth potential and juvenile quality of farmed fish species? Rev. Aquac. 2013, 1, 168–193. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Xia, J.; Zhang, X.; He, X.; Li, L.; Tang, R.; Li, D. Diet affects muscle quality and growth traits of grass carp (Ctenopharyngodon idellus): A comparison between grass and artificial feed. Front. Physiol. 2018, 9, 283. [Google Scholar] [PubMed]
- Zhang, L.; Xu, N.; Liu, X.; Onxayvieng, K.; Liu, L.; Tang, R.; Li, D. Exercise training accelerates UPS-and mTOR-mediated protein turnover of grass carp Ctenopharyngodon idella. Aquaculture 2021, 545, 737252. [Google Scholar]
- Lu, J.; Li, S.; He, X.; Tang, R.; Li, D. An in-pond tank culture system for high-intensive fish production: Effect of stocking density on growth of grass carp (Ctenopharyngodon idella Valenciennes, 1844) and blunt snout bream (Megalobrama amblycephala Yih, 1955). Aquaculture 2022, 549, 737808. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar]
- Jeffries, K.M.; Hinch, S.G.; Sierocinski, T.; Clark, T.D.; Eliason, E.J.; Donaldson, M.R.; Li, S.R.; Pavlidis, P.; Miller, K.M. Consequences of high temperatures and premature mortality on the transcriptome and blood physiology of wild adult sockeye salmon (Oncorhynchus nerka). Ecol. Evol. 2012, 2, 1747–1764. [Google Scholar] [CrossRef]
- Kim, K.H.; Hwang, Y.J.; Kwon, S.R. Influence of daily water temperature changes on the chemiluminescent response and mortality of cultured rockfish (Sebastes schlegeli). Aquaculture 2001, 192, 93–99. [Google Scholar] [CrossRef]
- Dulger, N.; Kumlu, M.; Turkmen, S.; Olculu, A.; Yilmaz, H.A.; Ocal, N. Thermal tolerance of European Sea Bass (Dicentrarchus labrax) juveniles acclimated to three temperature levels. J. Therm. Biol. 2012, 37, 79–82. [Google Scholar] [CrossRef]
- Dunajski, E. Texture of fish muscle. J. Texture. Stud. 1980, 10, 301–318. [Google Scholar]
- Hyldig, G.; Nielsen, D. A review of sensory and instrumental methods used to evaluate the texture of fish muscle. J. Texture Stud. 2001, 32, 219–242. [Google Scholar]
- Taylor, R.G.; Fjaera, S.O.; Skjervold, P.O. Salmon fillet texture is determined by myofiber-myofiber and myofiber-myocommata attachment. J. Food Sci. 2002, 67, 2067–2071. [Google Scholar] [CrossRef]
- Hultmann, L.; Turid, R. Iced storage of Atlantic salmon (Salmo salar)–effects on endogenous enzymes and their impact on muscle proteins and texture. Food Chem. 2004, 87, 31–41. [Google Scholar]
- Periago, M.J.; Ayala, M.D.; López-Albors, O.; Abdel, I.; Martínez, C.; García-Alcázar, A.; Ros, G.; Gil, F. Muscle cellularity and flesh quality of wild and farmed sea bass, Dicentrarchus labrax L. Aquaculture 2005, 249, 175–188. [Google Scholar] [CrossRef]
- Cheng, J.H.; Sun, D.W.; Han, Z.; Zeng, X.A. Texture and structure measurements and analyses for evaluation of fish and fillet freshness quality: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 52–61. [Google Scholar] [PubMed]
- Lee, J.S.; Cook, M.A.; Luckenbach, J.A.; Berejikian, B.A.; Simchick, C.A.; Oden, S.M.; Goetz, F.W. Investigation of long-term effects of larval rearing temperature on growth, deformities, flesh quality, and phenotypic sex of cultured sablefish (Anoplopoma fimbria). Aquaculture 2017, 479, 91–99. [Google Scholar]
- Hatae, K.; Yoshimatsu, F.; Matsumoto, J.J. Role of muscle fibers in contributing firmness of cooked fish. J. Food Sci. 1990, 55, 693–696. [Google Scholar] [CrossRef]
- Johnston, I.A.; Bower, N.I.; Macqueen, D.J. Growth and the regulation of myotomal muscle mass in teleost fish. J. Exp. Biol. 2011, 214, 1617–1628. [Google Scholar] [CrossRef] [Green Version]
- Lin, F.; Lin, J.; Liu, X.; Yuan, Y.; Liu, G.; Ye, X. Effects of temperature on muscle growth and collagen deposition in zebrafish (Danio rerio). Aquacult. Rep. 2022, 22, 100952. [Google Scholar]
- Baziz, H.A.; Geraert, P.A.; Padilha, J.C.; Guillaumin, S. Chronic heat exposure enhances fat deposition and modifies muscle and fat partition in broiler carcasses. Poult. Sci. 1996, 75, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Jia, G.Q.; Zuo, J.J.; Zhang, Y.; Lei, J.; Ren, L.; Feng, D.Y. Effects of constant and cyclic heat stress on muscle metabolism and meat quality of broiler breast fillet and thigh meat. Poult. Sci. 2012, 91, 2931–2937. [Google Scholar] [CrossRef]
- Lefaucheur, L.; Le, D.J.; Mourot, J.; Monin, G.; Ecolan, P.; Krauss, D. Influence of environmental temperature on growth, muscle and adipose tissue metabolism, and meat quality in swine. J. Anim. Sci. 1991, 69, 2844–2854. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.; Goodband, R.D.; Allee, G.L.; Jesse, G.W.; Nelssen, J.L.; Tokach, M.D.; Becker, B.A. The effects of diets formulated on an ideal protein basis on growth performance, carcass characteristics, and thermal balance of finishing giltshoused in a hot, diurnal environment. J. Anim. Sci. 1994, 72, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.M.; Yeunsu, S.; Sangsu, S.; Kichoon, L. Skeletal muscle characterization of Japanese quail line selectively bred for lower body weight as an avian model of delayed muscle growth with hypoplasia. PLoS ONE 2014, 9, e95932. [Google Scholar]
- De, A.; Carvalho, R.; Pinhal, D.; Padovani, C.; Martins, C.; Dal, P. Differential expression of myogenic regulatory factor MyoD in pacu skeletal muscle (Piaractus mesopotamicus Holmberg 1887: Serrasalminae, Characidae, Teleostei) during juvenile and adult growth phases. Micron 2008, 39, 1306–1311. [Google Scholar]
- Lee, B.; Lee, B.J.; Lee, Y.; Hur, S.W.; Kim, K.D.; Kim, K.W.; Kim, H.S.; Han, S.S.; Jeehwan, C.; Kichoon, L.; et al. Muscle fiber growth in olive flounder, Paralichthys olivaceus: Fiber hyperplasia at a specific body weight period and continuous hypertrophy. J. World Aquac. Soc. 2019, 50, 593–603. [Google Scholar]
- Zammit, P.S.; Relaix, F.; Nagata, Y.; Ruiz, A.P.; Collins, C.A.; Partridge, T.A.; Beauchamp, J.R. Pax7 and myogenic progression in skeletal muscle satellite cells. J. Cell Sci. 2006, 119, 1824–1832. [Google Scholar] [PubMed] [Green Version]
- Zhu, X.; Hu, J.; Zhang, J.; Liu, J.; Bao, L.; Pan, Y.; Chu, W. Effect of short-term fasting and glucocorticoids on KLF15 expression and branched-chain amino acids metabolism in Chinese perch. Aquacult. Rep. 2021, 19, 100617. [Google Scholar]
- Caipang, C.M.A.; Monica, F.B.; Viswanath, K. Short-term overcrowding of Atlantic cod, Gadus morhua: Effects on serum-mediated antibacterial activity and transcription of glucose transport and antioxidant defense related genes. Comp. Biochem. Phys. A 2008, 151, 560–565. [Google Scholar]
- Jun, Q.; Hong, Y.; Hui, W.; Didlyn, K.M.; Jie, H.; Pao, X. Physiological responses and HSP70 mRNA expression in GIFT tilapia juveniles, Oreochromisniloticus under short-term crowding. Aquac. Res. 2015, 46, 335–345. [Google Scholar]
- Zheng, G.D.; Sun, C.F.; Pu, J.W.; Chen, J.; Jiang, X.Y.; Zou, S.M. Two myostatin genes exhibit divergent and conserved functions in grass carp (Ctenopharyngodon idellus). Gen. Comp. Endocr. 2015, 214, 68–76. [Google Scholar]

| Primer | Forward Sequence (5′ to 3′) | Reverse Sequence (5′ to 3′) |
|---|---|---|
| Myog | GGACGCACTGCTCCACTCTG | AGGAACATCAGCAGGGAAACC |
| Myod | GAGAGCATCCAGAGGGCATC | AGTTCTACCTGGCCTCCAGT |
| MRF4 | CTGCGACGGTCAGTGTCTAATGT | CAGCCTCTGGTTCGGATTGG |
| MSTN-1 | TCAGTCCGAAGATCCAAGCG | TCCTGCGTTCACGTCGATTT |
| MSTN-2 | TGCATGCCATCAAGTCCCAA | TCATCCCCCAGAACGTCGTA |
| β-actin | CATTGACTCAGGATGCGGAAACT | CTGTGAGGGCAGAGTGGTAGACG |
| Parameters | 20 °C | 26 °C | 32 °C |
|---|---|---|---|
| Hardness (g) | 5049.92 ± 113.16 a | 4427.19 ± 72.00 b | 3953.51 ± 337.20 b |
| Adhesiveness | −21.16 ± 1.18 | −18.52 ± 1.45 | −17.00 ± 4.32 |
| Springiness | 0.57 ± 0.02 | 0.57 ± 0.02 | 0.55 ± 0.04 |
| Cohesiveness (g) | 0.31 ± 0.01 | 0.28 ± 0.01 | 0.30 ± 0.01 |
| Gumminess (g) | 1565.84 ± 37.16 a | 1205.91 ± 68.04 b | 1207.96 ± 40.45 b |
| Chewiness | 952.66 ± 65.41 a | 722.06 ± 14.17 b | 729.58 ± 47.04 b |
| Resilience | 0.15 ± 0.01 | 0.14 ± 0.01 | 0.15 ± 0.01 |
| Shear force | 558.96 ± 4.61 a | 626.76 ± 2.95 b | 704.63 ± 17.14 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Q.; Lu, J.; Yang, Y.; Li, D.; Liu, J. Acute Thermal Stress Reduces Skeletal Muscle Growth and Quality in Gibel Carp (Carassius gibelio). Water 2023, 15, 2706. https://doi.org/10.3390/w15152706
Hu Q, Lu J, Yang Y, Li D, Liu J. Acute Thermal Stress Reduces Skeletal Muscle Growth and Quality in Gibel Carp (Carassius gibelio). Water. 2023; 15(15):2706. https://doi.org/10.3390/w15152706
Chicago/Turabian StyleHu, Qixin, Jiamin Lu, Yu Yang, Dapeng Li, and Jieya Liu. 2023. "Acute Thermal Stress Reduces Skeletal Muscle Growth and Quality in Gibel Carp (Carassius gibelio)" Water 15, no. 15: 2706. https://doi.org/10.3390/w15152706
APA StyleHu, Q., Lu, J., Yang, Y., Li, D., & Liu, J. (2023). Acute Thermal Stress Reduces Skeletal Muscle Growth and Quality in Gibel Carp (Carassius gibelio). Water, 15(15), 2706. https://doi.org/10.3390/w15152706







