Dynamics of the Seawater Carbonate System in the East Siberian Sea: The Diversity of Driving Forces
Abstract
:1. Introduction
2. Materials and Methods
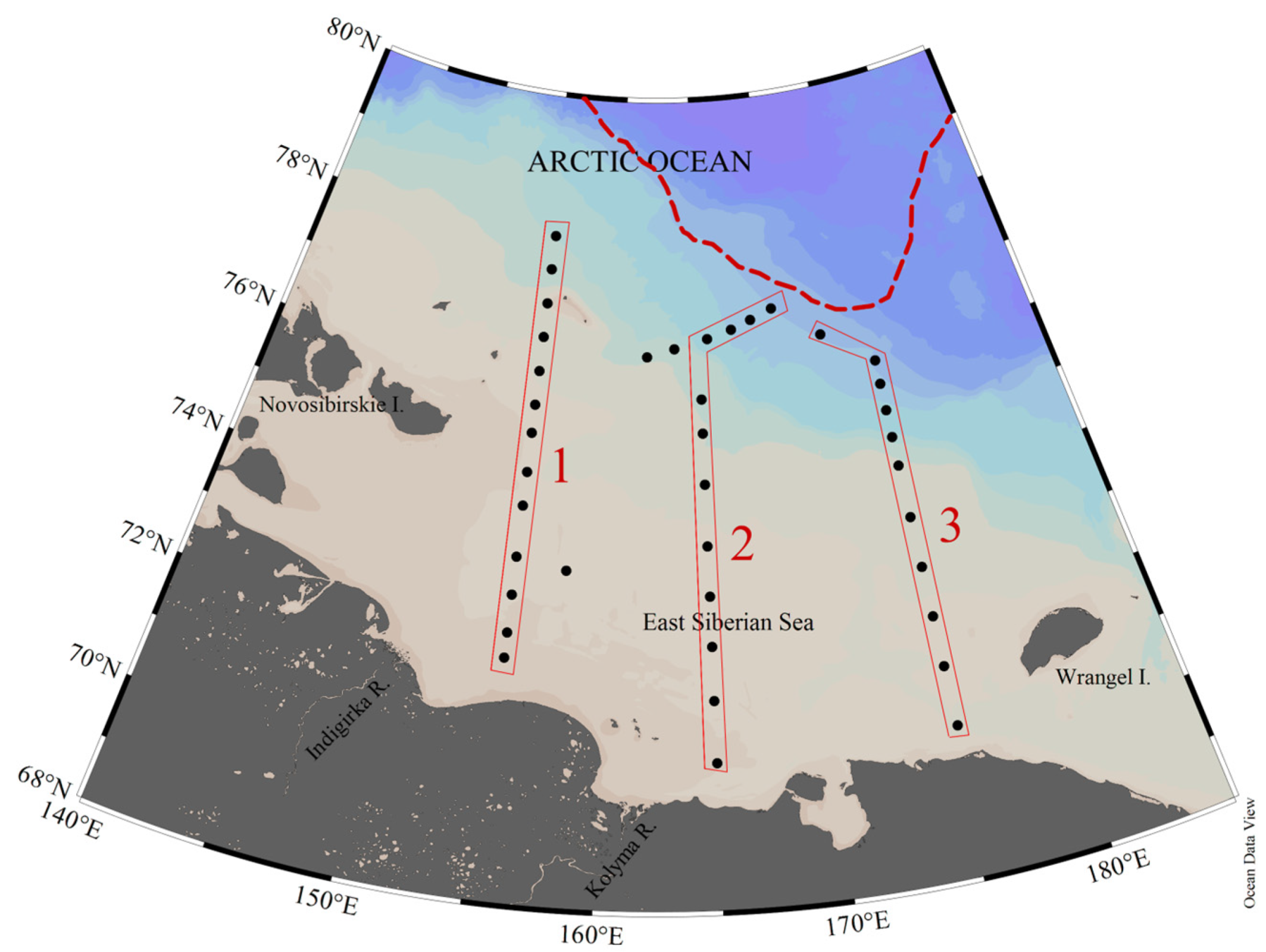
2.1. Study Area
2.2. Methods of Measurements and Calculations
3. Results
3.1. Hydrometeorological Situation
3.1.1. Air Temperature
3.1.2. Ice Cover
3.1.3. River Discharge
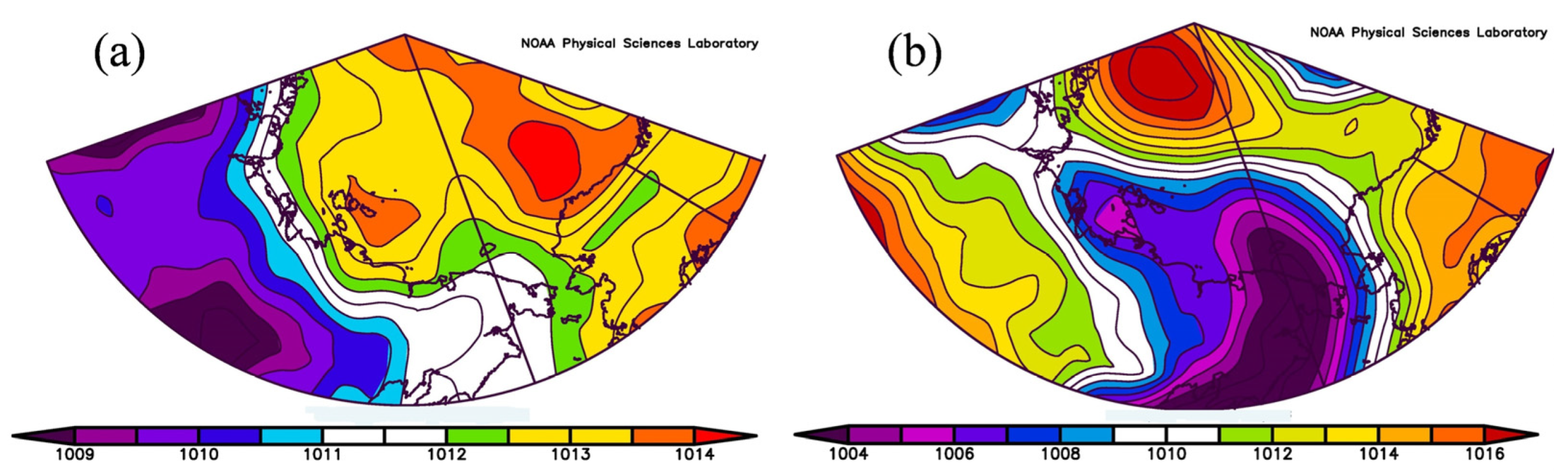
3.1.4. Sea Level Pressure
3.2. Spatial Distribution of Studied Parameters
3.2.1. Lateral Distribution
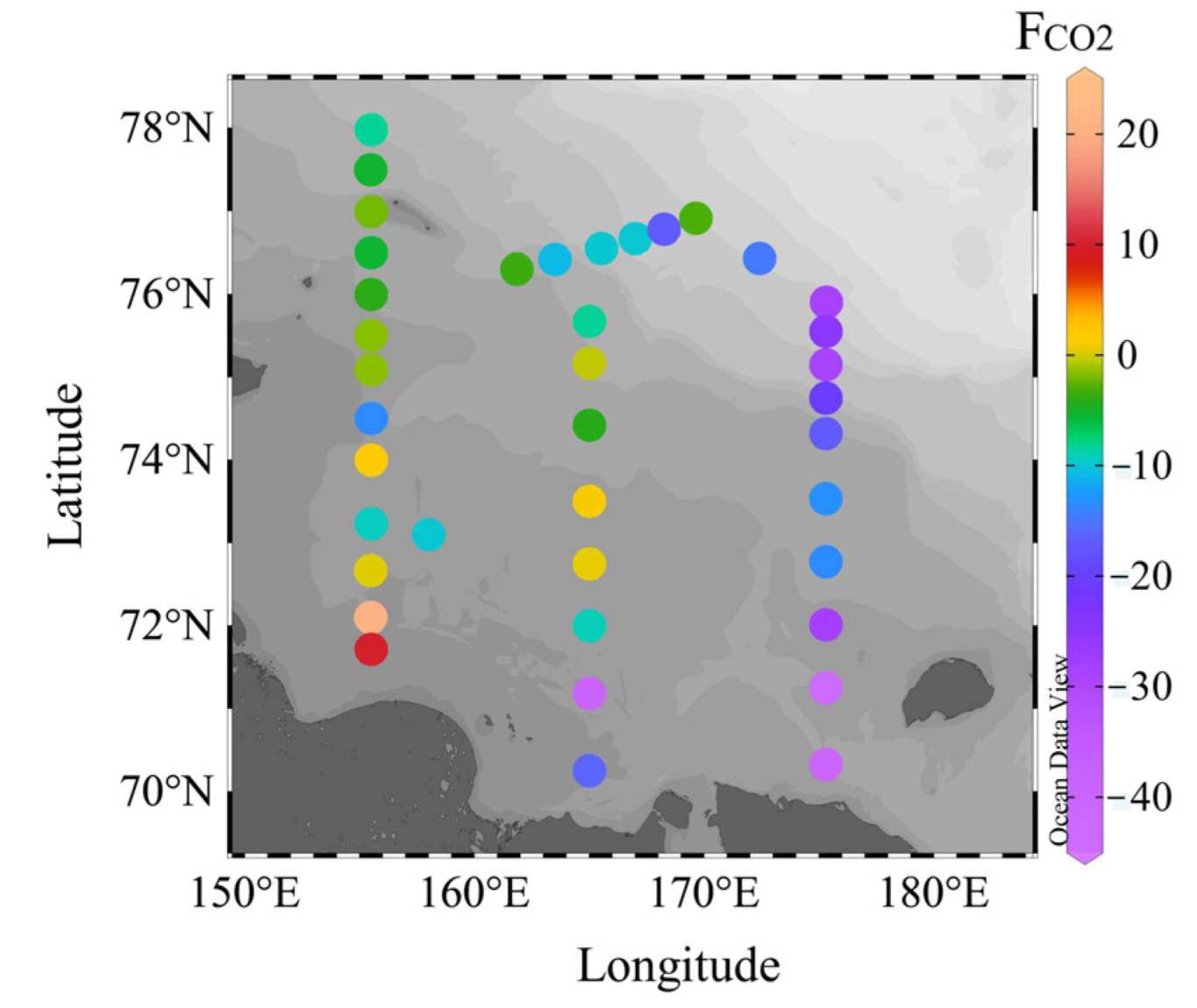
3.2.2. CO2 Fluxes in the Ocean–Atmosphere System
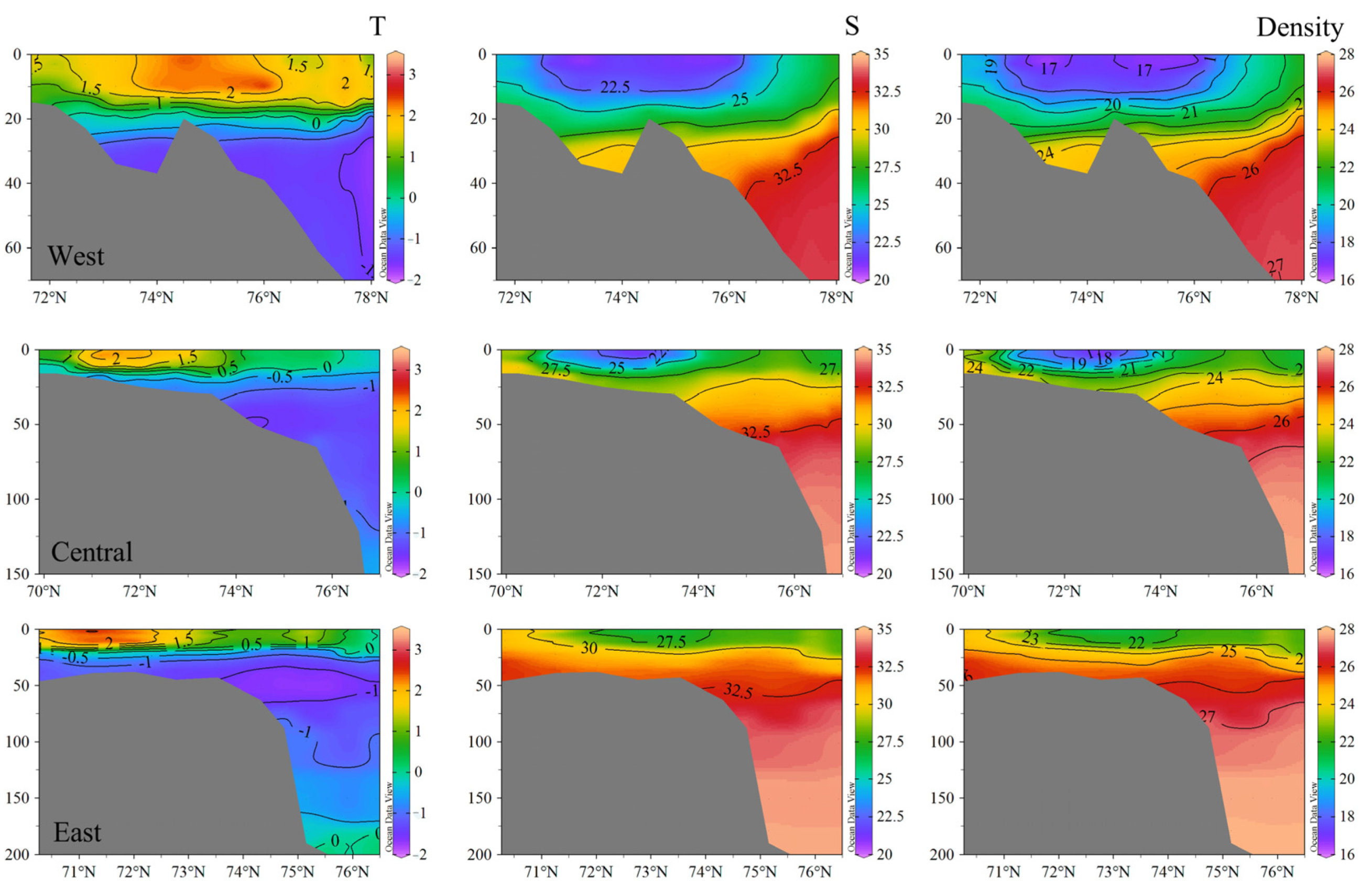
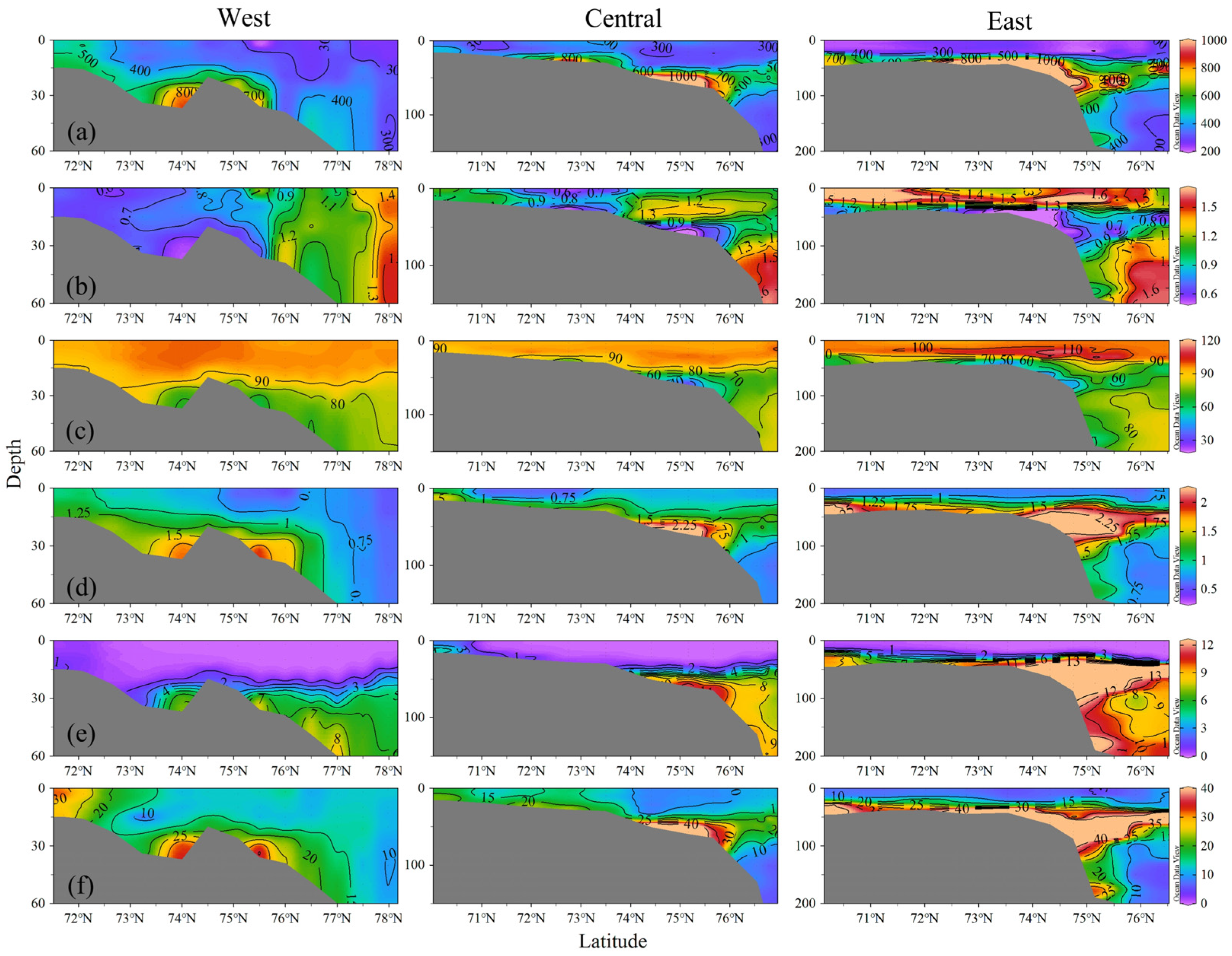
3.2.3. Vertical Structure of the Water Column
4. Discussion
4.1. Spatial Dynamics of Carbonate Parameters on the Shelf
4.2. CO2 Fluxes between the Seawater and the Atmosphere
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jorgenson, M.T.; Brown, J. Classification of the Alaskan Beaufort Sea Coast and estimation of sediment and carbon inputs from coastal erosion. Geo Mar. Lett. 2005, 482, 32–45. [Google Scholar] [CrossRef] [Green Version]
- Manson, G.K.; Solomon, S.M. Past and future forcing of Beaufort Sea coastal change. Atmos. Ocean 2007, 45, 107–122. [Google Scholar] [CrossRef]
- Stroeve, J.; Notz, D. Changing state of Arctic sea ice across all seasons. Environ. Res. Lett. 2018, 13, 103001. [Google Scholar] [CrossRef]
- Timmermans, M.-L.; Labe, Z. Sea Surface Temperature. In Arctic Report Card; Update to 2020; National Oceanic and Atmospheric Administration (NOAA): Washington, DC, USA, 2020. [Google Scholar] [CrossRef]
- AMAP Assessment 2013: Arctic Ocean Acidification; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2013; Available online: https://www.amap.no/documents/doc/amap-assessment-2013-arctic-ocean-acidification/881 (accessed on 1 December 2013).
- AMAP Assessment 2018: Arctic Ocean Acidification; Arctic Monitoring and Assessment Programme (AMAP): Tromsø, Norway, 2019; Available online: https://www.amap.no/documents/doc/amap-assessment-2018-arctic-ocean-acidification/1659 (accessed on 10 October 2018).
- Steiner, N.S.; Bowman, J.; Campbell, K.; Chierici, M.; Eronen-Rasimus, E.; Falardeau, M.; Flores, H.; Fransson, A.; Herr, H.; Insley, S.J.; et al. Climate change impacts on sea-ice ecosystems and associated ecosystem services. Elem. Sci. Anth. 2021, 9, 00007. [Google Scholar] [CrossRef]
- Aagaard, K.; Coachman, L.K.; Carmack, E. On the halocline of the Arctic Ocean. Deep-Sea Res. 1981, 28A, 529–545. [Google Scholar] [CrossRef]
- Whitefield, J.; Winsor, P.; McClelland, J.; Menemenlis, D. A new river discharge and river temperature climatology data set for the pan-Arctic region. Ocean Model. 2015, 88, 1–15. [Google Scholar] [CrossRef]
- Carmack, E.C.; Yamamoto-Kawai, M.; Haine, T.W.N.; Bacon, S.; Bluhm, B.A.; Lique, C.; Melling, H.; Polyakov, I.V.; Straneo, F.; Timmermans, M.L.; et al. Freshwater and its role in the Arctic Marine System: Sources, disposition, storage, export, and physical and biogeochemical consequences in the Arctic and global oceans. J. Geophys. Res. Biogeosci. 2016, 121, 675–717. [Google Scholar] [CrossRef] [Green Version]
- Carmack, E.C. The alpha/beta ocean distinction: A perspective on freshwater fluxes, convection, nutrients and productivity in high-latitude seas. Deep Sea Res. Part II Top. Stud. Oceanogr. 2007, 54, 2578–2598. [Google Scholar] [CrossRef]
- McClelland, J.W.; Holmes, R.M.; Dunton, K.H.; Macdonald, R.W. The Arctic Ocean estuary. Estuaries Coasts 2012, 35, 353–368. [Google Scholar] [CrossRef] [Green Version]
- Proshutinsky, A.; Krishfield, R.; Timmermans, M.-L.; Toole, J.; Carmack, E.; McLaughlin, F.; Williams, W.J.; Zimmermann, S.; Itoh, M.; Shimada, K. Beaufort Gyre freshwater reservoir: State and variability from observations. J. Geophys. Res. 2009, 114, C00A10. [Google Scholar] [CrossRef] [Green Version]
- Haine, T.W.N.; Curry, B.; Rildiger, G.; Hansen, E.; Karcher, M.; Lee, C.; Rudels, B.; de Steur, L.; Woodgate, R. Arctic freshwater export: Status, mechanisms, and prospects. Glob. Planet. Change 2015, 125, 13–35. [Google Scholar] [CrossRef] [Green Version]
- Holmes, R.M.; Coe, M.T.; Fiske, G.J.; Gurtovaya, T.; McClelland, J.W.; Shiklomanov, A.I.; Spencer, R.G.; Tank, S.E.; Zhulidov, A.V. Climate Change Impacts on the Hydrology and Biogeochemistry of Arctic Rivers. In Climatic Change and Global Warming of Inland Waters; Goldman, C.R., Kumagai, M., Robarts, R.D., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; pp. 1–26. [Google Scholar] [CrossRef]
- Carmack, E.C.; Wassmann, P. Food-webs and physical biological coupling on pan-arctic shelves: Perspectives, unifying concepts and future research. Prog. Oceanogr. 2006, 71, 446–477. [Google Scholar] [CrossRef]
- Wassmann, P.; Carmack, E.C.; Bluhm, B.A.; Duarte, C.M.; Berge, J.; Brown, K.; Grebmeier, J.M.; Holding, J.; Kosobokova, K.; Kwok, R.; et al. Towards a unifying pan-arctic perspective: A conceptual modelling toolkit. Prog. Oceanogr. 2020, 189, 102455. [Google Scholar] [CrossRef]
- Nikiforov, Y.G.; Shpaikher, A.O. Features of the formation of Hydrological Regime Large-Scale Variations in the Arctic Ocean; Hydrometeoizdat: Leningrad, Russia, 1980. (In Russian) [Google Scholar]
- Johnson, M.A.; Polyakov, I. The Laptev Sea as a source for recent Arctic Ocean salinity change. Geophys. Res. Lett. 2001, 28, 2017–2020. [Google Scholar] [CrossRef]
- Dmitrenko, I.; Kirillov, S.; Eicken, H.; Markova, N. Wind-driven summer surface hydrography of the eastern Siberian shelf. Geophys. Res. Lett. 2005, 32, L14613. [Google Scholar] [CrossRef] [Green Version]
- Polyakov, I.V.; Alexeev, V.A.; Belchansky, G.I.; Dmitrenko, I.A.; Ivanov, V.V.; Krillov, S.A.; Korablev, A.A.; Steele, M.L.; Timokhov, A.; Yashayaev, I. Arctic Ocean freshwater changes over the past 100 years and their causes. J. Climate 2008, 21, 364–384. [Google Scholar] [CrossRef] [Green Version]
- Anderson, L.G.; Björk, G.; Jutterström, S.; Pipko, I.; Shakhova, N.; Semiletov, I.; Wåhlström, I. East Siberian Sea, an Arctic region of very high biogeochemical activity. Biogeosciences 2011, 8, 1745–1754. [Google Scholar] [CrossRef] [Green Version]
- Pipko, I.I.; Semiletov, I.P.; Pugach, S.P. The carbonate system of the East Siberian Sea waters. Dokl. Earth Sci. 2005, 402, 624–627. [Google Scholar]
- Pipko, I.I.; Pugach, S.P.; Semiletov, I.P. The autumn distribution of the CO2 partial pressure in bottom waters of the East Siberian Sea. Dokl. Earth Sci. 2009, 425, 345–349. [Google Scholar] [CrossRef]
- Pipko, I.I.; Semiletov, I.P.; Pugach, S.P.; Wáhlström, I.; Anderson, L.G. Interannual variability of air-sea CO2 fluxes and carbon system in the East Siberian Sea. Biogeosciences 2011, 8, 1987–2007. [Google Scholar] [CrossRef] [Green Version]
- Pugach, S.P.; Pipko, I.I.; Shakhova, N.E.; Shirshin, E.A.; Perminova, I.V.; Gustafsson, Ö.; Bondur, V.G.; Ruban, A.S.; Semiletov, I.P. Dissolved organic matter and its optical characteristics in the Laptev and East Siberian seas: Spatial distribution and interannual variability (2003–2011). Ocean Sci. 2018, 14, 87–103. [Google Scholar] [CrossRef] [Green Version]
- Semiletov, I.; Dudarev, O.; Luchin, V.; Charkin, A.; Shin, K.-H.; Tanaka, N. The East Siberian Sea as a transition zone between Pacific-derived waters and Arctic shelf waters. Geophys. Res. Lett. 2005, 32, L10614. [Google Scholar] [CrossRef]
- Semiletov, I.; Pipko, I.; Gustafsson, Ö.; Anderson, L.G.; Sergienko, V.; Pugach, S.; Dudarev, O.; Charkin, A.; Gukov, A.; Bröder, L.; et al. Acidification of East Siberian Arctic Shelf waters through addition of freshwater and terrestrial carbon. Nat. Geosci. 2016, 9, 361–365. [Google Scholar] [CrossRef]
- Tesi, T.; Geibel, M.C.; Pearce, C.; Panova, E.; Vonk, J.E.; Karlsson, E.; Salvado, J.A.; Kruså, M.; Bröder, L.; Humborg, C.; et al. Carbon geochemistry of plankton-dominated supra-micron samples in the Laptev and East Siberian shelves: Contrasts in suspended particle composition. Ocean Sci. 2017, 13, 735–748. [Google Scholar] [CrossRef] [Green Version]
- Vonk, J.E.; Sánchez-García, L.; van Dongen, B.E.; Alling, V.; Kosmach, D.; Charkin, A.; Semiletov, I.P.; Dudarev, O.V.; Shakhova, N.; Roos, P.; et al. Activation of old carbon by erosion of coastal and subsea permafrost in Arctic Siberia. Nature 2012, 489, 137–140. [Google Scholar] [CrossRef]
- Shiklomanov, A.I.; Holmes, R.M.; McClelland, J.W.; Tank, S.E.; Spencer, R.G.M. Arctic Great Rivers Observatory. Discharge Dataset, 2021. Version 20211118. Available online: https://www.arcticrivers.org/data (accessed on 18 November 2021).
- Pipko, I.I.; Semiletov, I.P.; Tishchenko, P.Y.; Pugach, S.P.; Savel’eva, N.I. Variability of the carbonate system parameters in the coast-shelf zone of the East Siberian Sea during the autumn season. Oceanology 2008, 48, 54–67. [Google Scholar] [CrossRef]
- Pipko, I.I.; Pugach, S.P.; Semiletov, I.P.; Anderson, L.G.; Shakhova, N.E.; Gustafsson, Ö.; Repina, I.A.; Spivak, E.A.; Charkin, A.N.; Salyuk, A.N.; et al. The spatial and interannual dynamics of the surface water carbonate system and air–sea CO2 fluxes in the outer shelf and slope of the Eurasian Arctic Ocean. Ocean Sci. 2017, 13, 997–1016. [Google Scholar] [CrossRef] [Green Version]
- Anderson, L.G.; Ek, J.; Ericson, Y.; Humborg, C.; Semiletov, I.; Sundbom, M.; Ulfsbo, A. Export of calcium carbonate corrosive waters from the East Siberian Sea. Biogeosciences 2017, 14, 1811–1823. [Google Scholar] [CrossRef] [Green Version]
- Humborg, C.; Geibel, M.C.; Anderson, L.G.; Björk, G.; Mörth, C.-M.; Sundbom, M.; Thornton, B.F.; Deutsch, B.; Gustafsson, E.; Gustafsson, B.; et al. Sea-air exchange patterns along the central and outer East Siberian Arctic Shelf as inferred from continuous CO2, stable isotope, and bulk chemistry measurements. Glob. Biogeochem. Cy. 2017, 31, 1173–1191. [Google Scholar] [CrossRef]
- Semiletov, I.P.; Pipko, I.I.; Repina, I.A.; Shakhova, N.E. Carbonate chemistry dynamics and carbon dioxide fluxes across the atmosphere-ice-water interfaces in the Arctic Ocean: Pacific sector of the Arctic. J. Mar. Syst. 2007, 66, 204–226. [Google Scholar] [CrossRef]
- Anderson, L.G.; Jutterström, S.; Hjalmarsson, S.; Wahlström, I.; Semiletov, I. Out-gassing of CO2 from Siberian Shelf seas by terrestrial organic matter decomposition. Geophys. Res. Lett. 2009, 36, L20601. [Google Scholar] [CrossRef] [Green Version]
- Dudarev, O.; Charkin, A.; Shakhova, N.; Ruban, A.; Chernykh, D.; Vonk, J.; Tesi, T.; Martens, J.; Pipko, I.; Pugach, S.; et al. East Siberian Sea: Interannual heterogeneity of the suspended particulate matter and its biogeochemical signature. Prog. Oceanogr. 2022, 208, 102903. [Google Scholar] [CrossRef]
- Bellerby, R.G.J. Ocean Acidification without borders. Nat. Clim. Chang. 2017, 7, 241–242. [Google Scholar] [CrossRef]
- Anderson, L.G.; Björk, G.; Holby, O.; Jutterström, S.; Mörth, C.M.; O’Regan, M.; Pearce, C.; Semiletov, I.; Stranne, C.; Stöven, T.; et al. Shelf–Basin interaction along the East Siberian Sea. Ocean Sci. 2017, 13, 349–363. [Google Scholar] [CrossRef] [Green Version]
- Stein, R.; Macdonald, R.W. (Eds.) The Organic Carbon Cycle in the Arctic Ocean; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Jakobsson, M. Hypsometry and volume of the Arctic Ocean and its constituent seas. Geochem. Geophys. Geosyst. 2002, 3, 1028. [Google Scholar] [CrossRef] [Green Version]
- Dickson, A.G.; Sabine, C.L.; Christian, J.R. (Eds.) Guide to Best Practices for Ocean CO2 Measurements; PICES Special Publication 3; North Pacific Marine Science Organization: Sidney, BC, Canada, 2007. Available online: http://www.nodc.noaa.gov/ocads/oceans/Handbook_2007.html (accessed on 10 June 2019).
- Bruevich, S.V.; Demenchenok, S.K. Instruction for Chemical Investigation of Seawater; Glavsevmorput: Moscow, Russia, 1944. (In Russian) [Google Scholar]
- Pavlova, G.Y.; Tishchenko, P.Y.; Volkova, T.I.; Dickson, A.; Wallmann, K. Intercalibration of Bruevich’s method to determine the total alkalinity in seawater. Oceanology 2008, 48, 438–443. [Google Scholar] [CrossRef]
- Andreev, A.G.; Pipko, I.I.; Pugach, S.P. Impact of the paleo-river valleys on the chemical parameter distributions in the East Siberian Sea. Reg. Stud. Mar. Sci. 2023, 57, 102763. [Google Scholar] [CrossRef]
- Pierrot, D.; Lewis, E.; Wallace, D.W.R. MS Excel Program Developed for CO2 System Calculations. ORNL/CDIAC-105a; Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, U.S. Department of Energy: Oak Ridge, TN, USA, 2006. [CrossRef]
- Mehrbach, C.; Culberson, C.H.; Hawley, J.E.; Pytkowicz, R.M. Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnol. Oceanogr. 1973, 18, 897–907. [Google Scholar] [CrossRef]
- Dickson, A.G.; Millero, F.J. A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep-Sea Res. 1987, 34, 1733–1743. [Google Scholar] [CrossRef]
- Weiss, R. The solubility of nitrogen, oxygen, and argon in water and seawater. Deep-Sea Res. Oceanogr. Abstr. 1970, 17, 721–735. [Google Scholar] [CrossRef]
- Ivanenkov, V.N.; Bordovsky, O.K. Methods of Hydrochemical Investigation of Seawater; Nauka: Moscow, Russia, 1978. (In Russian) [Google Scholar]
- Codispoti, L.A.; Flagg, C.; Kelly, V.; Swift, J.H. Hydrographic conditions during the 2002 SBI process experiments. Deep Sea Res. Part II Top. Stud. Oceanogr. 2005, 52, 3199–3226. [Google Scholar] [CrossRef]
- Frey, K.E.; Sobczak, W.V.; Mann, P.J.; Holmes, R.M. Optical properties and bioavailability of dissolved organic matter along a flow-path continuum from soil pore waters to the Kolyma River mainstem, East Siberia. Biogeosciences 2016, 13, 2279–2290. [Google Scholar] [CrossRef] [Green Version]
- Wanninkhof, R. Relationship between wind speed and gas exchange over the ocean revisited. Limnol. Oceanogr. Methods 2014, 12, 351–362. [Google Scholar] [CrossRef]
- Thoning, K.W.; Crotwell, A.M.; Mund, J.W. Atmospheric Carbon Dioxide Dry Air Mole Fractions from Continuous Measurements at Mauna Loa, Hawaii, Barrow, Alaska, American Samoa and South Pole. 1973–2021, Version 2022-05 National Oceanic and Atmospheric Administration (NOAA). Global Monitoring Laboratory (GML): Boulder, CO, USA. [CrossRef]
- Fransson, A.; Chierici, M.; Nojiri, Y. New insights into the spatial variability of the surface water carbon dioxide in varying sea-ice conditions in the Arctic Ocean. Cont. Shelf Res. 2009, 29, 1317–1328. [Google Scholar] [CrossRef]
- Nishino, S.; Itoh, M.; Williams, W.J.; Semiletov, I. Shoaling of the nutricline with an increase in near-freezing temperature water in the Makarov Basin. J. Geophys. Res. Oceans 2013, 118, 635–649. [Google Scholar] [CrossRef] [Green Version]
- Schlitzer, R. Ocean Data View. 2022. Available online: www.odv.awi.de (accessed on 21 January 2023).
- RF Service for Hydrometeorology and Environmental Monitoring (Roshydromet); A Report on Climate Features on the Territory of the Russian Federation in 2019; Roshydromet: Moscow, Russia, 2020; p. 97. ISBN 978-5-906099-58-7.
- Pipko, I.I.; Pugach, S.P.; Shcherbakova, K.P.; Semiletov, I.P. Optical signatures of dissolved organic matter in the Siberian Rivers during summer season. J. Hydrol. 2023, 620, 129468. [Google Scholar] [CrossRef]
- Matsubara, F.; Wild, B.; Martens, J.; Andersson, A.; Wennström, R.; Bröder, L.; Dudarev, O.; Semiletov, I.; Gustafsson, Ö. Molecular-Multiproxy Assessment of Land-Derived Organic Matter Degradation over Extensive Scales of the East Siberian Arctic Shelf Seas. Glob. Biogeochem. Cy. 2022, 36, e2022GB007428. [Google Scholar] [CrossRef]
- Wild, B.; Andersson, A.; Bröder, L.; Vonk, J.; Hugelius, G.; McClelland, J.W.; Song, W.; Raymond, P.A.; Gustafsson, Ö. Rivers across the Siberian Arctic unearth the patterns of carbon release from thawing permafrost. Proc. Natl. Acad. Sci. USA 2019, 116, 10280–10285. [Google Scholar] [CrossRef] [Green Version]
- Jones, E.P.; Anderson, L.G. On the Origin of the Chemical Properties of the Arctic Ocean Halocline. J. Geophys. Res. 1986, 91, 10759–10767. [Google Scholar] [CrossRef]
- Dmitrenko, I.A.; Kirillov, S.A.; Tremblay, L.B.; Kassens, H.; Anisimov, O.A.; Lavrov, S.A.; Razumov, S.O.; Grigoriev, M.N. Recent changes in shelf hydrography in the Siberian Arctic: Potential for subsea permafrost instability. J. Geophys. Res. 2011, 116, C10027. [Google Scholar] [CrossRef]
- Bertosio, C.; Provost, C.; Athanase, M.; Sennéchael, N.; Garric, G.; Lellouche, J.-M.; Kim, J.-H.; Cho, K.-H.; Park, T. Changes in Arctic halocline waters along the East Siberian slope and in the Makarov Basin from 2007 to 2020. J. Geophys. Res. Oceans 2022, 127, e2021JC018082. [Google Scholar] [CrossRef]
- Jung, J.; Cho, K.-H.; Park, T.; Yoshizawa, E.; Lee, Y.; Yang, E.J.; Gal, J.K.; Ha, S.Y.; Kim, S.; Kang, S.H.; et al. Atlantic-origin cold saline water intrusion and shoaling of the nutricline in the Pacific Arctic. Geophys. Res. Lett. 2021, 48, e2020GL090907. [Google Scholar] [CrossRef]
- Polyakov, I.V.; Pnyushkov, A.V.; Alkire, M.B.; Ashik, I.M.; Baumann, T.M.; Carmack, E.C.; Goszczko, I.; Guthrie, J.; Ivanov, V.V.; Kanzow, T.; et al. Greater role for Atlantic inflows on Eurasian Basin of the Arctic Ocean. Science 2017, 356, 285–291. [Google Scholar] [CrossRef] [Green Version]
- Carmack, E.; Chapman, D.C. Wind-driven shelf/basin exchange on an Arctic shelf: The joint roles of ice cover extent and shelf-break bathymetry. Geophys. Res. Lett. 2003, 30, 1778. [Google Scholar] [CrossRef]
- Spall, M.A.; Pickart, R.S.; Brugler, E.T.; Moore, G.W.K.; Thomas, L.; Arrigo, K.R. Role of shelfbreak upwelling in the formation of a massive under-ice bloom in the Chukchi Sea. Deep Sea Res. Part II Top. Stud. Oceanogr. 2014, 105, 17–19. [Google Scholar] [CrossRef] [Green Version]
- Niebauer, H.J.; Alexander, V. Oceanographic frontal structure and biological production at an ice edge. Cont. Shelf Res. 1985, 4, 367–388. [Google Scholar] [CrossRef]
- Polukhin, A. The role of river runoff in the Kara Sea surface layer acidification and carbonate system changes. Environ. Res. Lett. 2019, 14, 105007. [Google Scholar] [CrossRef] [Green Version]
- Pipko, I.I.; Pugach, S.P.; Semiletov, I.P. Characteristic features of the dynamics of carbonate parameters in the eastern part of the Laptev Sea. Oceanology 2015, 55, 68–81. [Google Scholar] [CrossRef]
- Pipko, I.I.; Pugach, S.P.; Semiletov, I.P. Dynamics of Carbonate Characteristics of the Kara Sea Waters in the Late Autumn Season of 2021. Dokl. Earth Sci. 2022, 506, 671–676. [Google Scholar] [CrossRef]





| Water Type | Salinity (‰) | Total Alkalinity (μmol kg−1) |
|---|---|---|
| MW | 4 | 263 |
| FW | 0 | 930 |
| AW | 34.87 | 2306 |
| Site | ΔpCO2 | U | k | FCO2 |
|---|---|---|---|---|
| Transect 1 n = 14 | −51 ± 79 | 5.27 ± 1.86 | 5.96 ± 3.38 | −2.2 ± 8.7 |
| Transect 2 n = 14 | −57 ± 48 | 7.64 ± 2.59 | 9.31 ± 6.66 | −9.4 ± 10.3 |
| Transect 3 n = 11 | −1658 ± 36 | 8.12 ± 1.36 | 9.94 ± 3.55 | −25.1 ± 10.4 |
| Date/Parameters | ΔpCO2 | U | FCO2 |
|---|---|---|---|
| 2008 (4–12 September), n = 37 | −111 ± 71 | 3.92 ± 1.43 | −3.9 ± 2.6 |
| 2019 (25 September–4 October), n = 33 | −108 ± 56 | 7.25 ± 2.27 | −14.3 ± 11.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pipko, I.; Pugach, S.; Semiletov, I.; Konstantinov, O. Dynamics of the Seawater Carbonate System in the East Siberian Sea: The Diversity of Driving Forces. Water 2023, 15, 2670. https://doi.org/10.3390/w15142670
Pipko I, Pugach S, Semiletov I, Konstantinov O. Dynamics of the Seawater Carbonate System in the East Siberian Sea: The Diversity of Driving Forces. Water. 2023; 15(14):2670. https://doi.org/10.3390/w15142670
Chicago/Turabian StylePipko, Irina, Svetlana Pugach, Igor Semiletov, and Oleg Konstantinov. 2023. "Dynamics of the Seawater Carbonate System in the East Siberian Sea: The Diversity of Driving Forces" Water 15, no. 14: 2670. https://doi.org/10.3390/w15142670
APA StylePipko, I., Pugach, S., Semiletov, I., & Konstantinov, O. (2023). Dynamics of the Seawater Carbonate System in the East Siberian Sea: The Diversity of Driving Forces. Water, 15(14), 2670. https://doi.org/10.3390/w15142670







