Removal of Ibuprofen and Diclofenac in Batch Nitrifying Reactors: Effect of Natural Zeolite on the Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nitrifying Inoculum, Substrate, and Zeolite Characteristics
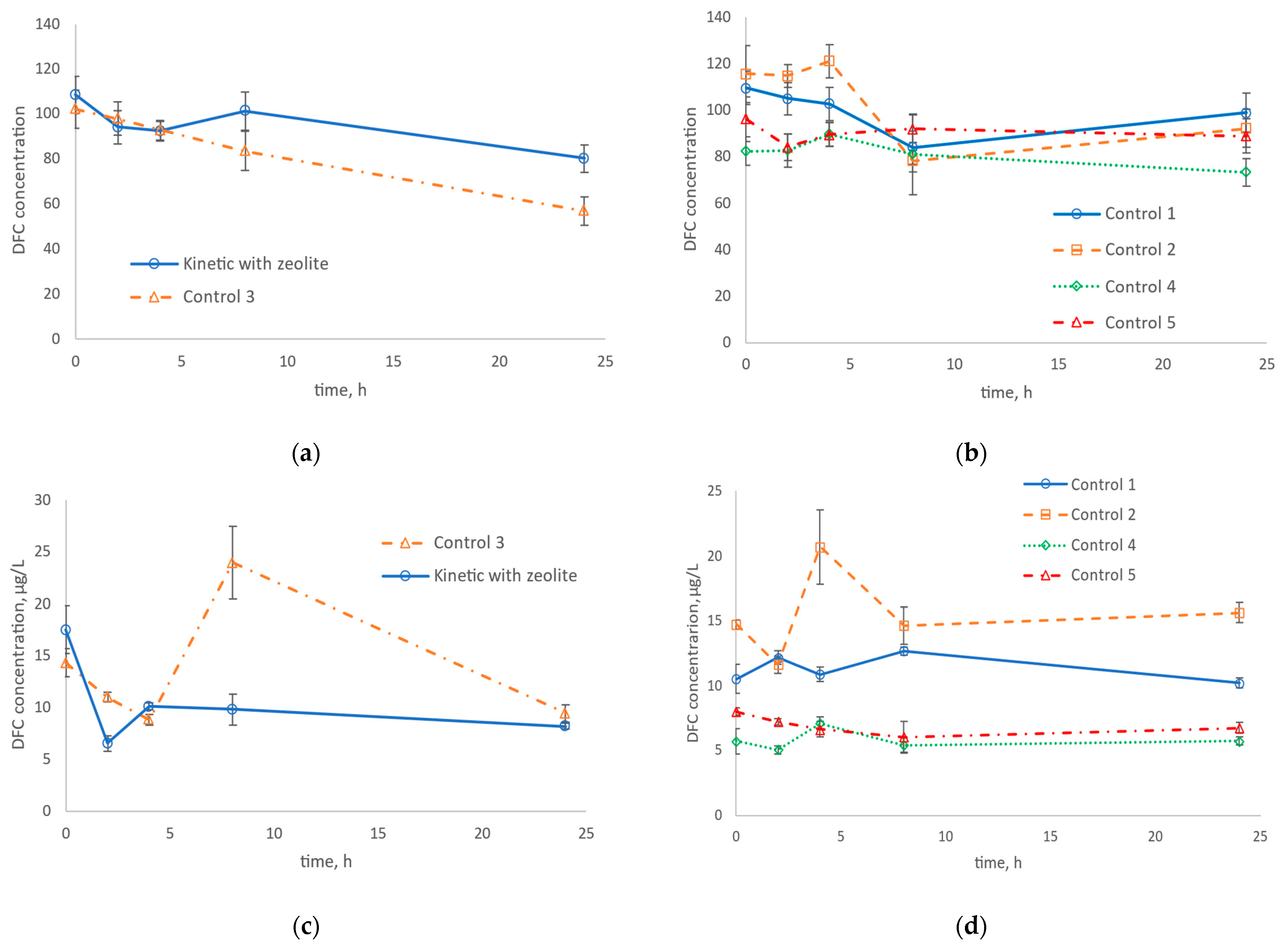
2.2. Experimental Setup and Experimental Design
- Control 1: without zeolite and without biomass;
- Control 2: with zeolite and without biomass;
- Control 3: with nitrifying biomass and without zeolite;
- Control 4: with allylthiourea (ATU, 98%), nitrifying biomass, and without zeolite;
- Control 5: with ATU, nitrifying biomass, and zeolite.
2.3. Determination of Reaction Rates and Removal Efficiencies
2.4. Chemical Analyses
3. Results
3.1. Characteristics of Natural Zeolites (NZ) and Its Effect on Batch Nitrification
3.2. Nitrification in the Presence of DFC, IBP, and Zeolite
3.3. IBP and DFC Removal in Nitrifying Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumwimba, M.N.; Meng, F. Roles of ammonia-oxidizing bacteria in improving metabolism and cometabolism of trace organic chemicals in biological wastewater treatment processes: A review. Sci. Total. Environ. 2018, 659, 419–441. [Google Scholar] [CrossRef]
- Feng, L.; van Hullebusch, E.D.; Rodrigo, M.A.; Esposito, G.; Oturan, M.A. Removal of residual anti-inflammatory and analgesic pharmaceuticals from aqueous systems by electrochemical advanced oxidation processes. A review. Chem. Eng. J. 2013, 228, 944–964. [Google Scholar] [CrossRef]
- González-Pérez, D.; Pérez, J.; Gómez, M. Behaviour of the main nonsteroidal anti-inflammatory drugs in a membrane bioreactor treating urban wastewater at high hydraulic- and sludge-retention time. J. Hazard. Mater. 2017, 336, 128–138. [Google Scholar] [CrossRef]
- Kennes-Veiga, D.M.; Gónzalez-Gil, L.; Carballa, M.; Lema, J.M. Enzymatic cometabolic biotransformation of organic micropollutants in wastewater treatment plants: A review. Bioresour. Technol. 2022, 344, 126291. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Xin, M.; Zhang, J.; Sun, Y.; Luo, S.; Wang, H.; Wang, Y.; Bi, X. Diclofenac inhibited the biological phosphorus removal: Performance and mechanism. Chemosphere 2020, 243, 125380. [Google Scholar] [CrossRef]
- Mansouri, F.; Chouchene, K.; Roche, N.; Ksibi, M. Removal of Pharmaceuticals from Water by Adsorption and Advanced Oxidation Processes: State of the Art and Trends. Appl. Sci. 2021, 11, 6659. [Google Scholar] [CrossRef]
- Taoufik, N.; Boumya, W.; Achak, M.; Sillanpää, M.; Barka, N. Comparative overview of advanced oxidation processes and biological approaches for the removal pharmaceuticals. J. Environ. Manag. 2021, 288, 112404. [Google Scholar] [CrossRef] [PubMed]
- Iori, V.; Zacchini, M.; Pietrini, F. Growth, physiological response and phytoremoval capability of two willow clones exposed to ibuprofen under hydroponic culture. J. Hazard. Mater. 2013, 262, 796–804. [Google Scholar] [CrossRef]
- Madikizela, L.M.; Botha, T.L.; Kamika, I.; Msagati, T.A.M. Uptake, Occurrence, and Effects of Nonsteroidal Anti-Inflammatory Drugs and Analgesics in Plants and Edible Crops. J. Agric. Food Chem. 2022, 70, 34–45. [Google Scholar] [CrossRef]
- Bigott, Y.; Chowdhury, S.P.; Pérez, S.; Montemurro, N.; Manasfi, R.; Schröder, P. Effect of the pharmaceuticals diclofenac and lamotrigine on stress responses and stress gene expression in lettuce (Lactuca sativa) at environmentally relevant concentrations. J. Hazard. Mater. 2021, 403, 123881. [Google Scholar] [CrossRef]
- Li, S.; Peng, L.; Yang, C.; Song, S.; Xu, Y. Cometabolic biodegradation of antibiotics by ammonia oxidizing microorganisms during wastewater treatment processes. J. Environ. Manag. 2022, 305, 114336. [Google Scholar] [CrossRef]
- Alvarino, T.; Suarez, S.; Lema, J.; Omil, F. Understanding the removal mechanisms of PPCPs and the influence of main technological parameters in anaerobic UASB and aerobic CAS reactors. J. Hazard. Mater. 2014, 278, 506–513. [Google Scholar] [CrossRef]
- Alvarino, T.; Komesli, O.; Suarez, S.; Lema, J.; Omil, F. The potential of the innovative SeMPAC process for enhancing the removal of recalcitrant organic micropollutants. J. Hazard. Mater. 2016, 308, 29–36. [Google Scholar] [CrossRef]
- Jia, Y.; Yin, L.; Khanal, S.K.; Zhang, H.; Oberoi, A.S.; Lu, H. Biotransformation of ibuprofen in biological sludge systems: Investigation of performance and mechanisms. Water Res. 2020, 170, 115303. [Google Scholar] [CrossRef] [PubMed]
- Kolakovic, S.; Salgado, R.; Freitas, E.B.; Bronze, M.R.; Sekulic, M.T.; Carvalho, G.; Reis, M.A.; Oehmen, A. Diclofenac biotransformation in the enhanced biological phosphorus removal process. Sci. Total. Environ. 2022, 806, 151232. [Google Scholar] [CrossRef]
- Deng, Y.; Li, B.; Yu, K.; Zhang, T. Biotransformation and adsorption of pharmaceutical and personal care products by activated sludge after correcting matrix effects. Sci. Total. Environ. 2016, 544, 980–986. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Geng, J.; Li, S.; Li, J.; Fu, Y.; Xu, K.; Ren, H.; Zhang, X. Abiotic and biotic processes of diclofenac in enriched nitrifying sludge: Kinetics, transformation products and reactions. Sci. Total. Environ. 2019, 683, 80–88. [Google Scholar] [CrossRef]
- Montalvo, S.; Huiliñir, C.; Borja, R.; Sánchez, E.; Herrmann, C. Application of zeolites for biological treatment processes of solid wastes and wastewaters—A review. Bioresour. Technol. 2020, 301, 122808. [Google Scholar] [CrossRef]
- Adegoke, K.A.; Olagunju, A.O.; Alagbada, T.C.; Alao, O.C.; Adesina, M.O.; Afolabi, I.C.; Adegoke, R.O.; Bello, O.S. Adsorptive Removal of Endocrine-Disrupting Chemicals from Aqueous Solutions: A Review. Water Air Soil Pollut. 2022, 233, 38. [Google Scholar] [CrossRef]
- Vasilachi, I.C.; Asiminicesei, D.M.; Fertu, D.I.; Gavrilescu, M. Occurrence and Fate of Emerging Pollutants in Water Environment and Options for Their Removal. Water 2021, 13, 181. [Google Scholar] [CrossRef]
- Al-Rimawi, F.; Daana, M.; Khamis, M.; Karaman, R.; Khoury, H.; Qurie, M. Removal of Selected Pharmaceuticals from Aqueous Solutions Using Natural Jordanian Zeolite. Arab. J. Sci. Eng. 2019, 44, 209–215. [Google Scholar] [CrossRef]
- Karthik, R.M.; Philip, L. Sorption of pharmaceutical compounds and nutrients by various porous low cost adsorbents. J. Environ. Chem. Eng. 2021, 9, 104916. [Google Scholar] [CrossRef]
- Huiliñir, C.; Fuentes, V.; Estuardo, C.; Antileo, C.; Pino-Cortés, E. Partial Nitrification in a Sequencing Moving Bed Biofilm Reactor (SMBBR) with Zeolite as Biomass Carrier: Effect of Sulfide Pulses and Organic Matter Presence. Water 2021, 13, 2484. [Google Scholar] [CrossRef]
- Huiliñir, C.; Fuentes, V.; Esposito, G.; Montalvo, S.; Guerrero, L. Nitrification in the presence of sulfide and organic matter in a sequencing moving bed biofilm reactor (SMBBR) with zeolite as biomass carrier. J. Chem. Technol. Biotechnol. 2020, 95, 1614. [Google Scholar] [CrossRef] [Green Version]
- Ghampson, I.T.; Sepúlveda, C.; Garcia, R.; Fierro, J.G.; Escalona, N.; DeSisto, W.J. Comparison of alumina- and SBA-15-supported molybdenum nitride catalysts for hydrodeoxygenation of guaiacol. Appl. Catal. A Gen. 2012, 435–436, 51–60. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Huiliñir, C.; Medina, R.; Montalvo, S.; Castillo, A.; Guerrero, L. Biological nitrification in the presence of sulfide and organic matter: Effect of zeolite on the process in a batch system. J. Chem. Technol. Biotechnol. 2018, 93, 2390–2398. [Google Scholar] [CrossRef]
- Dawas-Massalha, A.; Gur-Reznik, S.; Lerman, S.; Sabbah, I.; Dosoretz, C.G. Co-metabolic oxidation of pharmaceutical compounds by a nitrifying bacterial enrichment. Bioresour. Technol. 2014, 167, 336–342. [Google Scholar] [CrossRef]
- Fernandez-Fontaina, E.; Gomes, I.; Aga, D.; Omil, F.; Lema, J.; Carballa, M. Biotransformation of pharmaceuticals under nitrification, nitratation and heterotrophic conditions. Sci. Total. Environ. 2016, 541, 1439–1447. [Google Scholar] [CrossRef]
- Association, A.P.H.; Association, A.W.W.; Federation, W.E. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; Routledge: London, UK, 2017. [Google Scholar]
- Candia-Onfray, C.; Irikura, K.; Calzadilla, W.; Rojas, S.; Zanoni, M.V.B.; Salazar, R. Degradation of contaminants of emerging concern in a secondary effluent using synthesized MOF-derived photoanodes: A comparative study between photo-, electro- and photoelectrocatalysis. Chemosphere 2023, 315, 137683. [Google Scholar] [CrossRef]
- Semmens, M.J.; Goodrich, R.R. Biological regeneration of ammonium-saturated clinoptilolite. I. Initial observations. Environ. Sci. Technol. 1977, 11, 255–259. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, Y.; Wu, D.; Wang, Z.; Wang, Y.; Wang, G.; Zhou, X.; Sun, H. Effect of free nitrous acid on nitritation process: Microbial community, inhibitory kinetics, and functional biomarker. Bioresour. Technol. 2023, 371, 128595. [Google Scholar] [CrossRef]
- Xu, Y.; Yuan, Z.; Ni, B.-J. Biotransformation of pharmaceuticals by ammonia oxidizing bacteria in wastewater treatment processes. Sci. Total. Environ. 2016, 566, 796–805. [Google Scholar] [CrossRef]
- Keener, W.K.; Arp, D.J. Transformations of Aromatic Compounds by Nitrosomonas europaea. Appl. Environ. Microbiol. 1994, 60, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Roh, H.; Subramanya, N.; Zhao, F.; Yu, C.-P.; Sandt, J.; Chu, K.-H. Biodegradation potential of wastewater micropollutants by ammonia-oxidizing bacteria. Chemosphere 2009, 77, 1084–1089. [Google Scholar] [CrossRef]
- Melo, A.; Costa, J.; Quintelas, C.; Ferreira, E.C.; Mesquita, D.P. Effect of ibuprofen on extracellular polymeric substances (EPS) production and composition, and assessment of microbial structure by quantitative image analysis. J. Environ. Manag. 2021, 293, 112852. [Google Scholar] [CrossRef] [PubMed]
- Kraigher, B.; Mandic-Mulec, I. Influence of Diclofenac on Activated Sludge Bacterial Communities in Fed-Batch Reactors. Food Technol. Biotechnol. 2020, 58, 402–410. [Google Scholar] [CrossRef]
- Zheng, X.; Jiang, N.; Zheng, H.; Wu, Y.; Heijman, S.G. Predicting adsorption isotherms of organic micropollutants by high-silica zeolite mixtures. Sep. Purif. Technol. 2022, 282, 120009. [Google Scholar] [CrossRef]
- Fernandez-Fontaina, E.; Omil, F.; Lema, J.; Carballa, M. Influence of nitrifying conditions on the biodegradation and sorption of emerging micropollutants. Water Res. 2012, 46, 5434–5444. [Google Scholar] [CrossRef] [PubMed]
- Quintelas, C.; Mesquita, D.P.; Torres, A.M.; Costa, I.; Ferreira, E.C. Degradation of widespread pharmaceuticals by activated sludge: Kinetic study, toxicity assessment, and comparison with adsorption processes. J. Water Process. Eng. 2020, 33, 101061. [Google Scholar] [CrossRef]
- Yu, T.-H.; Lin, A.Y.-C.; Panchangam, S.C.; Hong, P.-K.A.; Yang, P.-Y.; Lin, C.-F. Biodegradation and bio-sorption of antibiotics and non-steroidal anti-inflammatory drugs using immobilized cell process. Chemosphere 2011, 84, 1216–1222. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Langenhoff, A.A.; Comans, R.N.; Sutton, N.B.; Rijnaarts, H.H. Effects of dissolved organic matter and nitrification on biodegradation of pharmaceuticals in aerobic enrichment cultures. Sci. Total. Environ. 2018, 630, 1335–1342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, J.; Wang, X.; Yin, F.; Xu, G. Characterizing the removal routes of seven pharmaceuticals in the activated sludge process. Sci. Total. Environ. 2019, 650, 2437–2445. [Google Scholar] [CrossRef] [PubMed]
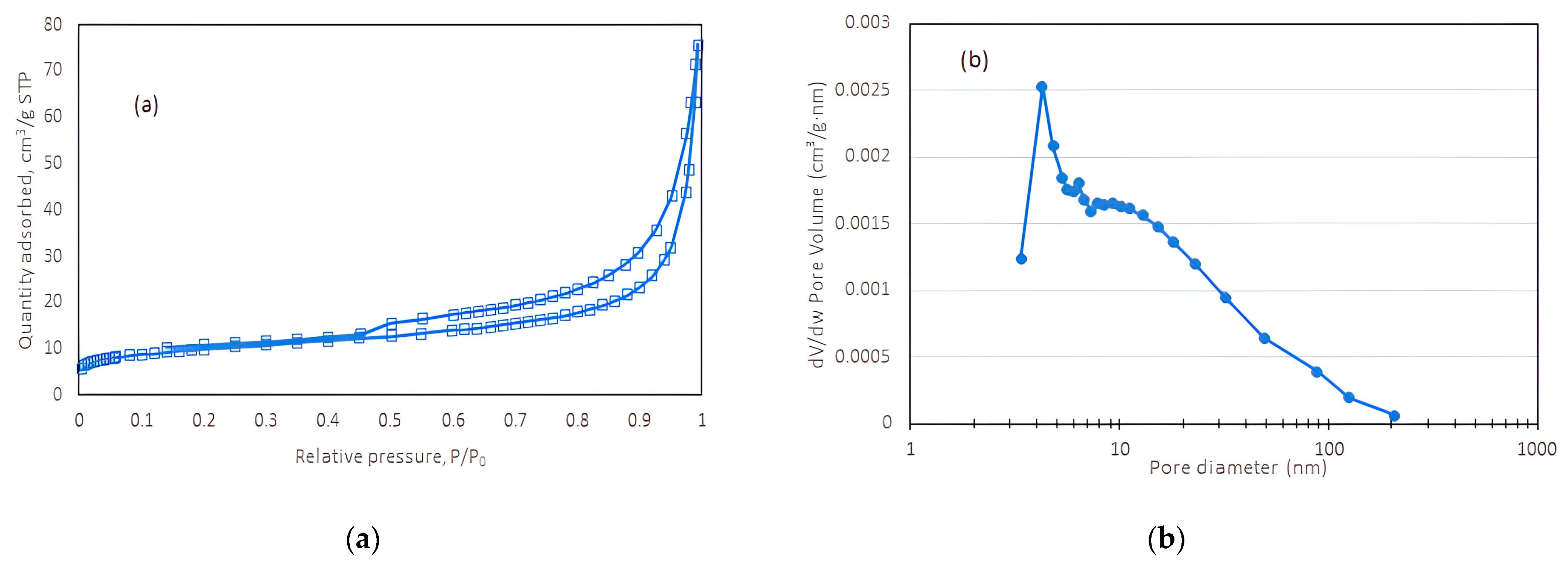

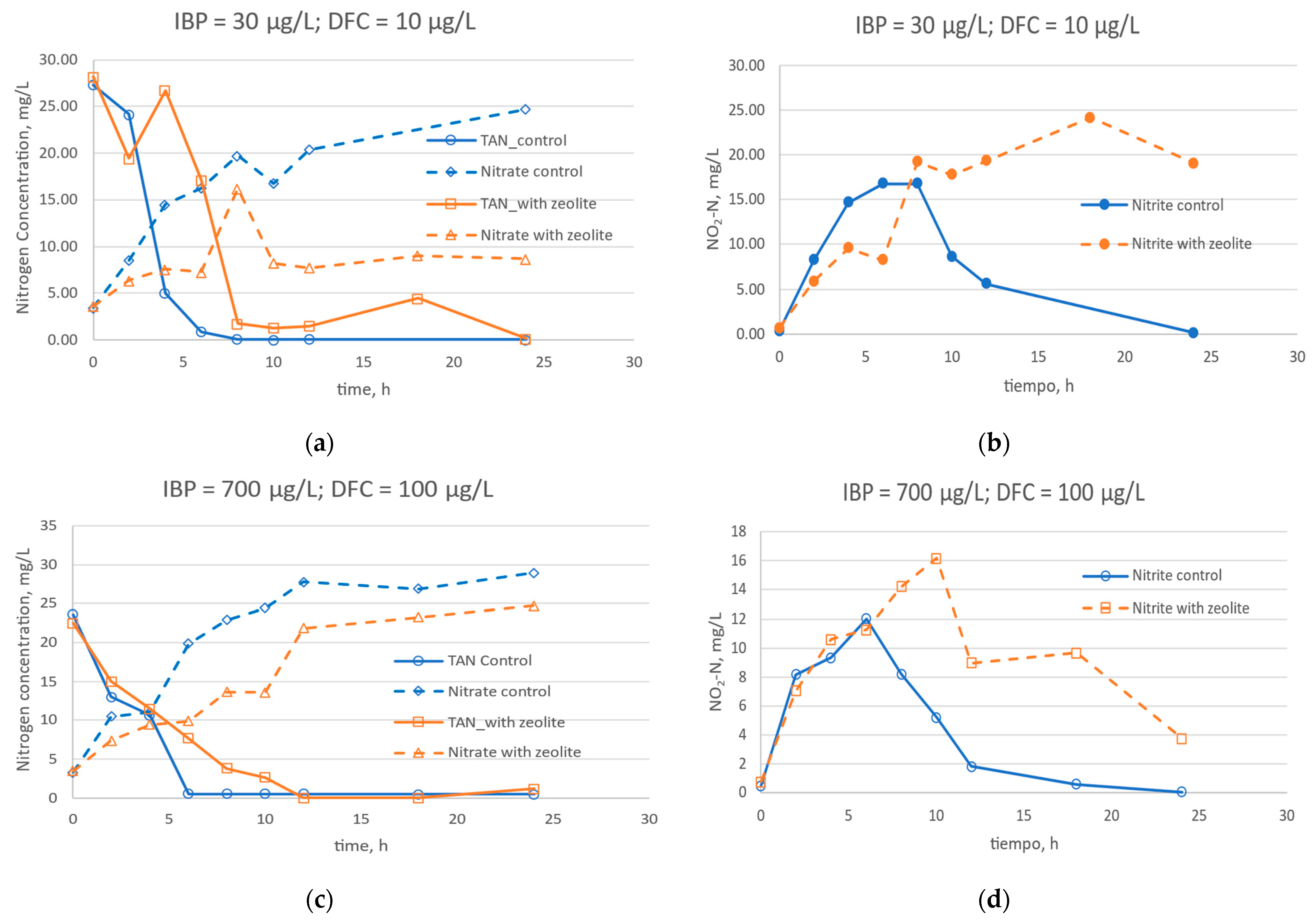
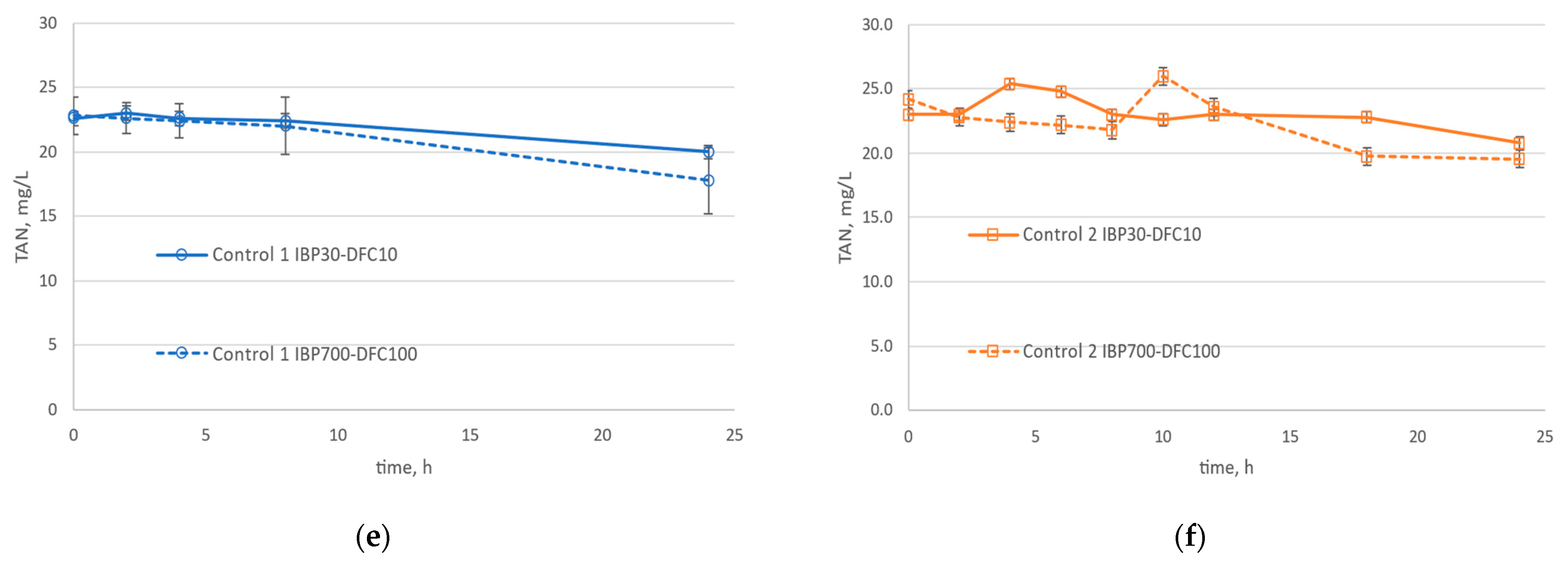
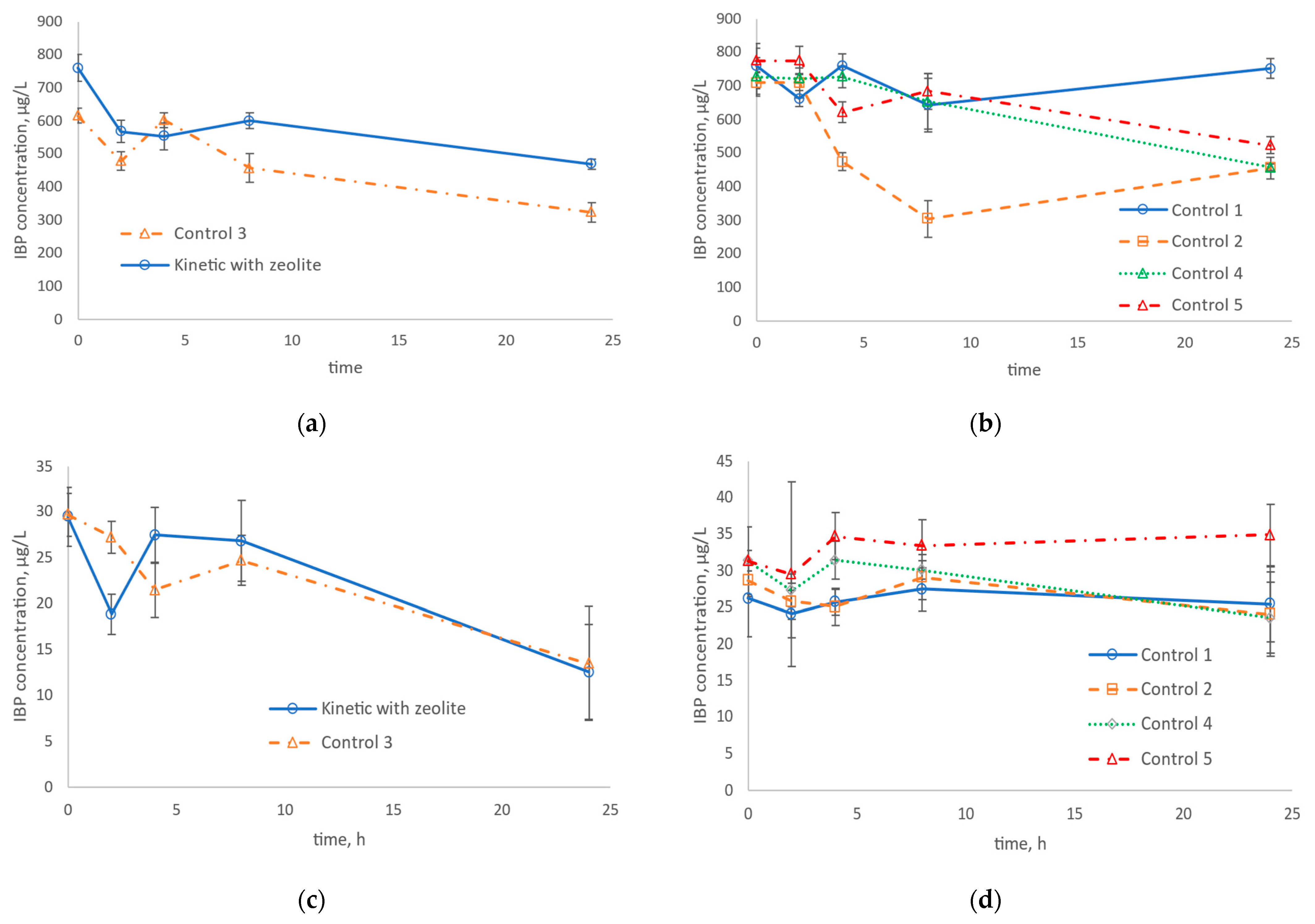
| Properties | Value |
|---|---|
| BET surface area (m2/g) | 35 |
| Total pore volume (cm3/g) | 0.11 |
| Average pore diameter (4V/A by BET) (nm) | 12 |
| Condition | (mg TAN/g VSS h) |
|---|---|
| Nitrification without MP and without zeolite | 4.78 ± 0.404 |
| Nitrification without MP and with zeolite | 1.48 ± 0.414 |
| Control 3, IBP30-DFC10 | 4.55 ± 1.105 |
| Assay with zeolite IBP30-DFC10 | 2.81 ± 1.083 |
| Control 3, IBP700-DFC100 | 3.69 ± 0.883 |
| Assay with zeolite IBP700-DFC100 | 2.12 ± 0.523 |
| Condition | K DFC (h−1) | kT DFC (L/g VSS h) | R2 DFC | K IBP (h−1) | kT IBP (L/g VSS h) | R2 IBP |
|---|---|---|---|---|---|---|
| Control 4, IBP30-DFC10 | --- | --- | 0.0113 ± 2.64 × 10−3 | 0.0113 ± 2.64 × 10−3 | 0.8219 | |
| Control 5, IBP30-DFC10 | --- | --- | --- | --- | ||
| Control 3, IBP30-DFC10 | 0.0208 ± 0.011 | 0.0193 ± 0.0102 | 0.543 | 0.0332 ± 4.09 × 10−3 | 0.0307 ± 3.79 × 10−3 | 0.9429 |
| Assay with Zeolite IBP30-DFC10 | 0.0382 ± 0.0115 | 0.0402 ± 0.0116 | 0.7871 | 0.034 ± 8.29 × 10−3 | 0.0357 ± 8.73 × 10−3 | 0.8082 |
| Control 4, IBP700-DFC100 | --- | --- | 0.0182 ± 1.78 × 10−3 | 0.0214 ± 2.09 × 10−3 | 0.963 | |
| Control 5, IBP700-DFC100 | --- | --- | 0.0172 ± 3.08 × 10−3 | 0.0172 ± 3.08 × 10−3 | 0.8861 | |
| Control 3, IBP700-DFC100 | 0.0245 ± 1.59 × 10−4 | 0.0252 ± 1.64 × 10−4 | 0.9998 | 0.0279 ± 4.47 × 10−3 | 0.0288 ± 4.61 × 10−3 | 0.9068 |
| Assay with Zeolite IBP700-DFC100 | 0.0132 ± 3.16 × 10−3 | 0.0140 ± 3.36 × 10−3 | 0.8144 | 0.0232 ± 6.69 × 10−3 | 0.0247 ± 7.12 × 10−3 | 0.7508 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Escobar, J.; Hernández, L.; González, J.L.; Salazar-González, R.; Calzadilla, W.; Guerrero, L.; Escalona, N.; Huiliñir, C. Removal of Ibuprofen and Diclofenac in Batch Nitrifying Reactors: Effect of Natural Zeolite on the Process. Water 2023, 15, 2665. https://doi.org/10.3390/w15142665
Escobar J, Hernández L, González JL, Salazar-González R, Calzadilla W, Guerrero L, Escalona N, Huiliñir C. Removal of Ibuprofen and Diclofenac in Batch Nitrifying Reactors: Effect of Natural Zeolite on the Process. Water. 2023; 15(14):2665. https://doi.org/10.3390/w15142665
Chicago/Turabian StyleEscobar, Jenny, Leslie Hernández, Jorge Leiva González, Ricardo Salazar-González, Wendy Calzadilla, Lorna Guerrero, Néstor Escalona, and César Huiliñir. 2023. "Removal of Ibuprofen and Diclofenac in Batch Nitrifying Reactors: Effect of Natural Zeolite on the Process" Water 15, no. 14: 2665. https://doi.org/10.3390/w15142665







