Abstract
The use of agricultural drainage water (ADW) in irrigation is a great challenge, improving water use efficiency, nutrient circularity, and avoiding surface and ground-water contamination. The objective of this study was to evaluate the chemical and ecotoxicological characteristics of an ADW to analyze the safety of its reuse. An irrigated area with maize crops was selected (Tejo Basin, Portugal), where a subsurface structure for the recovery of ADW was installed, collecting the drainage in a pond and recycling it for crop irrigation. Water was collected monthly during the irrigation campaign of 2021 (April to August). Three herbicides and two metabolites were quantified, reaching a maximum concentration of 0.74 µg L−1 for S-metolachlor and 0.48 µg L−1 for terbuthylazine. The lethal bioassays did not detect toxicity, except for the sample collected in August toward Vibrio fisheri (EC50 = 25.2%). The samples were not toxic to Pseudokirchneriella subcapitata, with a growth inhibition rate of less than 10%. The low lethal and sublethal effects may be ascribed to the high nutrient concentration (e.g., 1.76 mg P L−1 and 98.9 mg NO3− L−1, in July) that could have masked toxic effects. Ecotoxicological responses support the option of ADW reuse in irrigation, offering a safe and sustainable solution for water and nutrient management.
1. Introduction
To meet the world’s future food security and sustainable needs, food production must rise substantially while, at the same time, agriculture’s environmental footprint must decrease drastically [1]. The footprint of agriculture ranges from the contamination of soil, water, and food to the excessive use of water and the imbalance of ecosystem services. One of the routes for agricultural contamination of natural water resources is through runoff processes, which represent an important non-point source (NPS) of pollutants, such as nutrients and pesticides [2,3]. Excessive amounts of nitrogen and phosphorus speed up natural eutrophication processes, leading to the formation of algae blooms in surface waters [2]. Further, the runoff of these nutrients also represents a loss of valuable resources from the agricultural system. All these facts justify the importance of studying options to recycle agricultural drainage water (ADW), and one option is its use in the irrigation of other crops, avoiding runoff and improving the efficiency of the fertilization processes. Currently, smart technological solutions are being adopted, which allow the capture, storage, and reuse of ADW for irrigation purposes, increasing the efficiency of water management in a scenario of water scarcity [4,5]. In fact, subsurface drainage systems (e.g., with perforated plastic pipes) [6] are one of the most used water table management practices in floodplain soils to remove excess water in the soil profile and maintain a sufficient water level in the root zone, to maximize crop production [7].
This practice responds to the global challenges of increasing climate variability, the objectives of the European Green Deal, and the United Nations Sustainable Development Goals Agenda, providing an important solution that could cost-effectively recover nutrients from ADW, based on a circular economy point of view [8,9].
With this ADW recycling strategy, it is possible to reduce the entrance of a set of substances and chemicals into the downstream waterways. These substances can be mineral and organic suspended solids, nutrients, pesticides, and metal(loid)s, which may have a direct ecotoxicological effect on aquatic and benthic organics [3,9], compromising the ecosystem’s balance. On the other hand, the toxicity of the recycled ADW may also affect the organisms in the agroecosystem, which is important to assess. Notwithstanding, the literature is scarce on this type of risk assessment, using real ADW samples and standardized bioassays with aquatic organisms to evaluate their ecotoxicity. Therefore, it would be important to make this type of assessment, confronting the expected toxicity from the ADW chemical composition [10].
The use of ADW as irrigation water, recycling their nutrients back to the crop during the growing season, will only be environmentally sustainable if this type of water does not present environmental risk. This issue can only be properly addressed by linking chemical and ecotoxicological assessment, allowing cause–effect relationships, improving risk assessment results, and designing specific recovery strategies [11,12]. Hence, to obtain pertinent outcomes at the ecosystem level, the assessed responses should have consequences on the biological health of the species (e.g., mortality, growth, reproduction, and feeding rates) [13]. In fact, the use of the Ecotoxicological Line of Evidence (LoE) with a group of organisms of taxonomic representative species sensitive to chemical substances will allow to assess the impact of ADW in the terrestrial and aquatic ecosystems. The following steps are essential: (i) to analyze the impact of ADW on the abiotic and biotic compartments (soil, water, plants, organisms) when it enters the receptor medium; or (ii) to direct, whenever possible, its reuse as a source of nutrients for local agricultural crops or for cultural growth media for microalgae [12,14].
The objective of this study was to evaluate the chemical composition and the aquatic ecotoxicological effects of an ADW from a maize crop area, collected during a complete growing season, to assess its impact on the natural waters and the safety of its reuse in the irrigation of crops. The case study selected is in Rio Maior, sub-Basin (Tejo Basin, Portugal), where this type of drainage collection/irrigation system is installed, providing a unique opportunity to evaluate the potential risk to the aquatic system if this water was simply discharged, as happens in most situations.
2. Materials and Methods
2.1. Site Description and Sampling Characterization
This study was performed in a maize crop field located near São João da Ribeira (Rio Maior, Portugal) (Figure 1), in an area integrated with the Tejo Basin, sub-basin Maior (Maior River). This sub-basin presents an area of 923 km2 and is the delimitation of the municipalities of Alcobaça, Azambuja, Cadaval, Cartaxo, Rio Maior, and Santarém. The Tejo Basin is an international hydrographic region with a total area in Portuguese territory of 30,502 km2. This Basin signs the climate transition in the national territory between the North, temperate and humid, and the South, hot and dry. In general, the average annual temperature in the basin is 14.9 °C, with air temperatures to the North of the Tejo River being lower than those in the South. The average annual precipitation value over the Tejo Basin is approximately 870 mm and 600 mm in areas located South of the river. Further, the climate of Continental Portugal is marked by a seasonal irregularity, with about 75% of precipitation occurring in the wet season from October to March and only 25% in the dry season (from April to September) [15]. The irrigation in the area is mainly performed with surface water collected from Maior River or with groundwater, using wells and catchment holes. This specific area was selected because it has a drainage water recycling system comprising the capture, storage, and reuse of surface and subsurface drainage water, which is used in the irrigation of the crop during part of the growing season.

Figure 1.
Map evidencing the sampling location, São João da Ribeira (39°17′13.6″ N; 8°53′37.6″ W), close to Rio Maior (Portugal).
Considering the Portuguese Soil Classification System (1:25,000 (South); [16]), describing the soil morphology in the area, the Explanatory Note to the Portuguese Soil Classification System [17], and selecting the correspondent classes of the WRB classification [18], the soils in the area, affecting the draining system, are mainly Cambisols, Luvisols, and Regosols. Soil samples (0–30 cm) were also collected in the center-pivots areas, and they are characterized by circum-neutral to slightly alkaline pH (1:2.5 soil:water), ranging from 7.2 to 7.9, low organic matter content (14 to 18 g kg−1), very high level of fertility regarding extractable P content (Egner-Rhiem; 272.2 to >500 mg P2O5 kg−1), and high level of fertility regarding extractable K content (Egner-Rhiem; 120 to 187 mg K2O kg−1). Considering these soils analysis, the expected maize corn yield in this area is about 16 t ha−1, above the reference production in Portugal [19].
An area of approximately 40 ha has a subsurface drainage system with perforated pipes, which collects the subsurface drainage during the wet season. This water, plus the surface runoff, are both conducted by gravity to the same discharge point in a drainage pond located at a lower topographic level (39°17′13.6″ N 8°53′37.6″ W, São João da Ribeira, Rio Maior (Portugal)). The center-pivot irrigation uses water directly pumped from the drainage pond, which has an additional outlet (it can drain the surplus water to the river, in periods of heavy precipitation, during part of the wet season), and inlet (the pond is recharged with water pumped from the river when there is a water deficit). This system improves water reuse, mitigating drought conditions, further allowing the recycling of nutrients, and reducing non-point source pollution of the river with nutrients and, very importantly, pesticides.
This system was selected as a case study because it is an example of a subsurface water recycling system being used for irrigation and allowing the characterization of the ADW that would be discharged into the receiving surface bodies if the system was not installed. Therefore, this system provides a unique opportunity for the characterization of the subsurface drainage and runoff from an intensive irrigated area, both for their physicochemical parameters, chemical substances, and ecotoxicological responses, allowing the assessment of the risk of its discharge. In fact, to our best knowledge, this is the first time that this type of water is fully characterized, considering these groups of parameters.
The production factors applied to the maize crop are displayed in Table 1.

Table 1.
Production factors (fertilizers and pesticides) applied in the maize crop during the 2021 irrigation campaign.
Five samples (5 L each) were collected directly from the drainage pond, all at the same sampling location (Figure 1; São João da Ribeira, 39°17′13.6″ N; 8°53′37.6″ W), monthly, during the 2021 agricultural irrigation campaign, starting on 19 April, before maize sowing, and ending in August. Table 2 presents the meteorological conditions registered during that period in the nearest meteorological station (Fonte Boa e Estação Zootécnica, Santarém), available online on the site of the Portuguese Meteorological Institute [20]. The water samples were collected from the pond at a point close to the water inlet, stored in polyethylene bottles, and transported to the lab in a portable cooler with ice packs, where they were stored and preserved, following the indications for water conservation [21]. The sample used for physicochemical characterization was kept refrigerated at 4 °C until analyses (maximum one-month storage), while two subsamples were kept deep frozen at −20 °C to be used for the ecotoxicological characterization and the pesticides analysis.

Table 2.
Temperatures (minimum and maximum) and accumulated precipitation registered in the nearest meteorological station (Fonte Boa e Estação Zootécnica, Santarém). Data collected at IPMA, Instituto Português do Mar e da Atmosfera [20].
2.2. Physicochemical Parameters and Pesticides Analysis
The ADW characterization considered (i) physicochemical parameters: pH (by potentiometry, with a Thermo ScientificTM OrionTM 3-STAR Benchtop pH Meter); electrical conductivity (ECw; by conductimetry, with a Thermo ScientificTM Orion STARTM A212 Benchtop Conductivity Meter); nitrates (NO3−) and ammonium (NH4+), which sum represents the mineral N, with a Continuous Flow Analyzer with a UV detector (Skalar, San plus System); total concentrations of important elements as macro and micronutrients (P, K, S, Na, Ca, Mg, B, Fe, Mn, Cu, and Zn), by inductively coupled plasma—optical emission spectrometry, ICP-OES, using an iCAP 7000 Series ICP Spectrometer (Thermo Fisher Scientific, Waltham, MA, USA), on samples digested in a hot plate with concentrated nitric acid; chemical oxygen demand (COD; by acid digestion with potassium dichromate and titration with ammonium iron (II) sulfate); chloride (Cl−; by titration with silver nitrate, using Mohr’s method). Analytical-grade chemicals were used for general procedures, while calibrations were made with standards with Reagent-grade specifications. Standard methods were used, following the indications of the American Public Health Association [21]. All the analyses were performed in triplicate, and quality control procedures were adopted, which are routinely used in the laboratory, analyzing blanks, independent standard solutions, and calculating limits of detection (LOD) and quantification (LOQ). Results were only reported if above LOQ thresholds.
Pesticides approved for use in maize in Portugal [22] and applied in the studied area were selected, along with other pesticides detected in a large screening performed during a preliminary ADW assessment. Concentrations of these pesticides were measured through LC-MS/MS (liquid chromatography–tandem mass spectrometry) by Eurofins SOFIA GmbH, an external accredited laboratory (DIN EN ISO/IEC 17025:2018 DAkkS D-PL-19579-02-00), following a German standard method (DIN 38407-F36, 2014-09) [23], and an internal method for glyphosate and AMPA. All calibrations were performed with pesticide Analytical Standard Grade chemicals, and quality control procedures were performed routinely. The lowest concentration of the analyzed pesticides that can be measured with certainty (LOQ) is 0.05 µg/L.
Table 3 presents data on the chemical and physical properties of the pesticides analyzed in the ADW samples collected from the Pesticide Properties Database [24].

Table 3.
Selected information on the physical and chemical properties of the pesticides analyzed in the ADW samples [24].
2.3. Assessment of AWD Quality for Irrigation
As this water is recycled for irrigation, it is important to evaluate its quality considering that specific use. In Portugal, the quality of water for irrigation is regulated by Law Decree No. 236/98 from 1 August [25] (except the treated urban wastewater, which is specifically regulated by more recent legislation Law Decree No. 119/2019, 21 August [26]), which establishes quality standards, criteria, and quality objectives, with the aim of protecting the aquatic environment and improving the quality of water according to its main uses. This legislation provides Maximum Recommended Values (MRV) and Maximum Permissible Values (MPV) or intervals, in the case of pH, for some parameters, which are critical to evaluating its quality for irrigation. On the other hand, internationally accepted guidelines can be used for assessing the quality of water for irrigation, such as those from the Food and Agriculture Organization of the United Nations (FAO), based on the quality standards reported by Ayers and Westcot [27]. In these guidelines, specific water characteristics are addressed, classifying the restrictions to its use into three levels: no restrictions; slight to moderate restrictions; and severe restrictions [27,28]. The main characteristics to be evaluated, considering the risks to the soil or to the crop, are as follows: (i) water salinity, assessed from the water electrical conductivity (ECw); (ii) problems with the infiltration rate of water in the soil, assessed using, simultaneously, ECw and the sodium adsorption rate (SAR; calculated using Equation (1) and concentrations in meq L−1); (iii) specific ions, which frequently achieve concentrations that may induce toxicity problems in sensitive crops, namely, sodium (Na+), chloride (Cl−), and boron (B); and (iv) other problems, such as an excess of nitrates, which may cause adverse effects on the environment [27].
2.4. Ecotoxicological Assessment
Ecotoxicological assays were performed with a set of representative bioindicators at different trophic levels. The responses analyzed are essential for the life cycle of species whose dysfunction can deal with a decrease in population density and, in the long term, disturb the aquatic ecosystem. Hence, the bioassays developed, and the responses analyzed were as follows: (i) light inhibition of the bacteria Vibrio fisheri; (ii) growth inhibition of the green microalgae Pseudokirchneriella subcapitata; (iii) mortality of the crustacean Thamnocephalus platyurus; and (iv) feeding rate of the crustacean Daphnia magna. For all bioassays, a test with a reference substance was performed as a positive control. The sensitivity of the organisms was in accordance with the followed protocols.
2.4.1. Luminescence Inhibition of V. fisheri
The luminescence inhibition of the bacteria V. fisheri (NRRL B-11177) was assessed following the protocol of the “DR LANGE luminescent bacteria test” [29]. Tests were carried out using the dilutions of ADW with a 2% NaCl solution (50, 25, 12.5, 6.25, and 3.125%; v/v), with two replicates per treatment. The light emission was measured against a nontoxic control (2% NaCl solution), at a temperature of 15 ± 0.5 °C, with a period of exposure of 30 min. The concentration that induced 50% of bacterial light inhibition (30 min-EC50; % v/v) was determined, comparing with the control non-toxic.
2.4.2. Growth Inhibition of the Green Microalgae P. subcapitata
Green microalgae were exposed to five dilutions of ADW samples (12.5, 25, 50, 75, 100%; v/v, with negative control of MBL medium) over a 72 h period, following the OCDE protocol [30]. The tests started with a biomass of algae inoculum of 3–5 × 104 cells ml−1; six replicates per dilution were incubated in an orbital shaker at 21 ± 2 °C and with a constant luminous intensity (60–120 μE m−2 s−1, equivalent to 6000–10,000 lx). The concentration of the algae biomass was determined by counting the number of cells in a Neubauer chamber.
The average specific growth rate, determined during a specific period, was calculated using Equation (2):
where μi−j is an average specific growth rate for the specific time interval, i to j; ti is the time for the start of the exposure period; tj is the time for the end of the exposure period; Bi is the biomass concentration at the time I, and Bj is a biomass concentration at time j.
μi−j = (ln Bj − ln Bi)/tj − ti
The inhibition of the algal growth was estimated from Equation (3):
where % I is the mean percentage of inhibition for specific growth rate; μc is the mean value for the growth rate in control; μt is the mean value for the growth rate in the ADW samples.
% I= [(µc − µt)/µc] × 100
2.4.3. Mortality Test with T. platyurus
The test with T. platyurus was developed using the THAMNOTOXKIT FTM kit, adapted from Persoone [31]. Larvae of shrimp T. platyurus (<24 h, obtained by the hatching of cysts) were exposed to different dilutions of ADW samples (12.5, 25, 50, 75, and 100%, v/v), diluted with synthetic freshwater–water included in the test kit, which was also used as a nontoxic control. Four replicates for each of the five concentrations were incubated at 25 °C for 24 h in the dark. The number of dead shrimps after 24 h of exposure was used as the toxic response, and the 24 h-EC50 (%) was calculated.
2.4.4. Feeding Assay with D. magna
The feeding rate assay with D. magna was adapted from the methodology reported by McWilliam and Baird [32]. Groups of five neonates (4 or 5 days old born between the 3rd to 5th broods) were exposed to the solutions of ADW samples (100 mL of total volume). To each solution, a volume of algae, corresponding to a density of 3.0 × 105 cells mL−1 Daphnia−1 (equivalent to 2.65 mg C mL−1), was added. At the beginning of the test, the algae biomass was determined in each solution using a Neubauer chamber, after which the organisms were added to each vessel. Five replicates per dilution and a negative control test (with ASTM) were prepared. The test took place for 24 h at a temperature of 20 °C in the dark to avoid algae growth. At the end of the test, the organisms were removed from each vessel, the solution was shaken to resuspend the algae, and the number of algae cells was counted. The feeding rate was calculated according to Equation (4) [33]:
where F is a feeding rate (cells × animal−1 × h−1); V is a volume of medium in the test vessel (mL); C0 is an initial cell concentration (number × mL−1); C24 is a final cell concentration (number × mL−1); t is a duration of the experiment (h).
F = V × (C0 − C24)/t
2.5. Statistical Analysis
The results of the physicochemical parameters were analyzed using descriptive statistics (mean and standard deviation (SD), n = 3). Differences among results obtained in different months were evaluated using a one-way ANOVA analysis of variance. Whenever significant differences were found, a post hoc Tukey Honest Significant Difference (HSD) test was used to further elucidate differences among means at a p-level of 0.05.
The 30 min-EC50 (% v/v) of V. fischeri bioluminescence inhibition test was determined using LUMISsoft 4 Software™. The EC50 (% v/v) values for the T. platyurus mortality were determined using the probit analysis [34]. Results of sublethal endpoints (growth inhibition and feeding rate) were checked for homogeneity of variance by the Kolmogorov–Smirnov test and, when possible, subjected to a one-way analysis of variance (ANOVA). Data that do not satisfy the assumption for ANOVA were analyzed non-parametrically using Kruskal–Wallis ANOVA by ranks test. Whenever significant differences were found (p < 0.05), a post hoc Dunnett’s test was used to compare treatments with the control, for a p-value of 0.05 as the significant level [35].
The statistical analyses were performed with the STATISTICA 7.0 (Software™ Inc., Peachtree Corners, PA, USA, 2007).
3. Results and Discussion
3.1. Physicochemical Characterization, Pesticides Analysis, and Quality of ADW Samples for Irrigation
Physicochemical characteristics of the drainage water were influenced by different factors, which is important to refer to understand the chemical variability observed during this study. The 20/21 winter was characterized by scarce precipitation until March, and even in March, the accumulated precipitation was low (Table 2). April registered higher precipitation and, therefore, a higher volume of drainage collected at the pond, which occurred after the sampling (about 70% of the April precipitation occurred after the 25th, and the sample was collected on the 19th). In addition, the evaporation process from the pond due to the increase in the temperatures from April to July (Table 2) may also be responsible for the observed rise in the concentration of physical–chemical parameters over the months of this study. Thus, the sample collected in April was not so influenced by the drainage of the soil resulting from the runoff (one of the most important ways of water contamination by pesticides and fertilizers) as the sample collected in May [36]. On the other hand, the sample collected in August was influenced by a dilution effect from the water pumped from river Maior to recharge the pond, whose level was lowering.
All these factors had an impact on the physicochemical characteristics of the samples: the sample collected in April had statistically lower values for some parameters (e.g., ECw, Na, K, Mg, P, S, Cl−, and NO3−), which increased during the campaign to reach a maximum in July, with the highest concentrations for some nutrients (e.g., 1.76 mg P L−1 and 98.9 mg NO3− L−1), while the August sample presented lower concentration for some parameters (e.g., ECw, K, P, and NO3−), due to the referred dilution effect (Table 4).

Table 4.
Physicochemical characterization of the agricultural drainage water (mean ± standard deviation, n = 3). Values in a raw marked with the same letter are not significantly different (p > 0.05); Tukey HSD test.
Relatively to the quality of the ADW for irrigation, and taking into account the ECw values, all water samples are classified as having a “slight to moderate degree of restriction” for their use, with values ranging in the interval 0.7–3.0 mS cm−1, and two of the samples (May and June) surpassed the MRV recommended in the Portuguese Legislation (1 mS cm−1) (Table 4). Considering the infiltration rate, although the SAR values also surpass the MRV of the Portuguese Legislation (MRV (SAR) = 1) in all the samples (Table 4), FAO guidelines allow its classification in the lower class of risk (SAR in the range 0–3 and ECw > 0.7 dS m−1). Therefore, the use of this type of water, stored in the drainage pond, should reflect that its salinity may not allow its recycling to irrigate all types of crops, only those with moderate resistance to salinity [37], but that practice will not contribute to infiltration problems of the soil in the future.
The analysis of specific ions that may lead to toxicity problems in the irrigated crops showed that all the samples have a Cl− content that surpasses the MRV of the Portuguese Legislation (70 mg L−1), however, with no risk to irrigation, according to the FAO guideline values [27].
As for NO3−, it is a critical parameter in irrigation water, mostly because of the risk of surface waters’ eutrophication. The NO3− concentration in the July sample almost doubled the MRV of the Portuguese Legislation, which is set at 50 mg L−1. Of course, that does not mean the water should not be used for irrigation; rather, it should be used with caution, accounting for the total N carried out by the irrigation water, which must be estimated and considered in the crop fertilization plan. FAO is more precautionary, establishing at 30 mg NO3− L−1 of the cut-off value that, when surpassed, implies that the water should be used with “severe” restrictions. That value was surpassed in May, June, and July samples, those with higher NO3− concentrations, which indicates a high risk of contamination to the receiving surface waters with this type of drainage/irrigation water. Its recycling is, therefore, strongly recommended, but the N provided in the irrigation water must be accounted for to avoid excess fertilization and NO3− NPS pollution.
Additionally, an analysis of the possible effects of this AWD on the ecological status of the waters of Rio Maior was carried out, comparing the measured concentrations with the limit values proposed for the water classification as with reasonable or good status [38]. The results showed that this type of water could contribute to worsening the classification of the waters of the Tejo Basin due to the concentrations of P, NO3− and NH4+, which surpassed the limits proposed for a reasonable status classification for all the samples (P = 0.13 mg L−1; NO3− = 10 mg L−1; NH4+ = 0.5 mg L−1 [38]). Thus, these results support the proposal to reuse this type of water, improving the efficiency of water management, contributing to solving the problem of water scarcity, and avoiding further contamination and degradation of the ecological and chemical status of surface waters.
The concentrations of pesticides in the ADW sampled during the irrigation campaign of 2021 are displayed in Table 5. The herbicides S-metolachlor, tembotrione, terbuthylazine, and the metabolites AMPA and pyridafol were detected in the ADW samples at different times during the maize crop season (Table 5). Although the herbicides glyphosate and nicosulfuron were detected in the screening made in one ADW sample, which justified its monitoring during this study, none of the samples subsequently collected and analyzed in the irrigation campaign showed detectable concentrations for these herbicides (concentrations < 0.05 μg L−1). The insecticide lambda-cyhalothrin, despite being applied to the maize crop, was not detected in these samples (concentrations < 0.05 μg L−1), which can be explained by its low solubility in water and high partition coefficient organic carbon for water (Koc), indicating its possible adsorption to soil particles (Table 3).

Table 5.
Pesticides quantified in the ADW samples (mean ± standard deviation, n = 3) during the irrigation campaign of 2021.
S-Metolachlor was the herbicide quantified in the highest total concentration, reaching a value of 1.964 µg L−1. This herbicide was quantified in all analyzed AWD samples, being one of the most used herbicides in maize, which, combined with its high stability in water (DT50_water: 88 days; Table 3), may be the reason for the observed results. Further, the octanol–water partition coefficient higher than 3 indicated that this herbicide presents a tendency to be bioaccumulated. Terbuthylazine was quantified in June and July ADW samples, with a maximum concentration of 0.48 µg L−1. Concerning the metabolites, AMPA was quantified in most of the samples, with the highest total concentration observed in the July sample, 0.36 µg L−1. As glyphosate was not applied to the maize crop (Table 1), AMPA concentrations in the ADW may be explained by the high persistence of AMPA in the soil compartment (DT50_soil = 234 days). The June 2021 ADW sample showed the highest total concentration of herbicides of 1.56 µg L−1. Measured concentrations were compared with environmental quality standards for specific pollutants in river basins [38] and priority substances applicable to surface water under the European Water Framework Directive [39]. The herbicides quantified in the ADW are not considered priority substances in the surface waters of the European Union. However, terbuthylazine and its metabolite, desethyl-terbuthylazine, are classified as river basin-specific pollutants in Portugal by the National Environmental Protection Agency [38].
For the reported pesticides, limit of quantification (LOQ) = 0.05 μg L−1).
3.2. Ecotoxicological Analysis
The ecotoxicological analysis provided information on the effects of reusing this type of water, ensuring environmental sustainability, and improving chemical results, being the bridge to the risk assessment at the ecosystem level. The ecotoxicological results are displayed in Table 6 and Figure 2 and Figure 3. In general, the acute ecotoxicological bioassays showed that the ADWs were not toxic to the bioindicators used; only the sample from August 2021 induced a considerable decrease in luminescence of the bacterial V. fisheri with a 30 min-EC50 of 25%. The V. fisheri bioassay is mainly sensitive to wastewater, samples contaminated with potentially toxic metals (PTM) [40], or with chlorides and ammonia [41,42]; but in the present study, the very low PTM concentrations, and the ammonia and chloride concentrations similar to the other samples, do not justify this observed toxicity. In fact, the August ADW sample had the highest concentrations of pyridafol (0.22 µg L−1; pyridate metabolite) and s-metolachlor (0.65 µg L−1), which may indicate that these herbicides, singly or in the mixture, may have influenced the observed toxic effect. There is no literature on the toxicity of pyridafol to the luminescent bacteria (this being the first study with results about it), but regarding s-metolachlor, studies indicated toxicity toward V. fisheri only at higher concentrations (178.4 mg L−1) [43].

Table 6.
EC50 (%) values calculated for V. fisheri (mean ± SD; n = 2) and T. platyurus (mean ± SD; n = 4) bioassays after exposure to ADW samples.
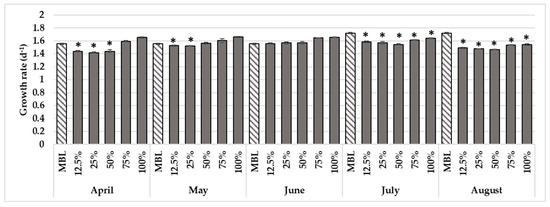
Figure 2.
Growth rate (d−1) of algae P. subcapitata after 72-h exposure to several dilutions of AWD samples during the irrigation campaign of 2021. Mean ± standard deviation (n = 6), * p < 0.05, Dunnett’s post hoc comparison test with the control MBL.
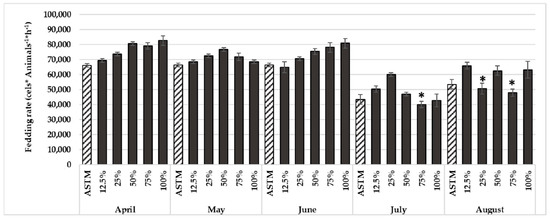
Figure 3.
Results of feeding rate of D. magna, after 24 h exposed to ADW samples. Mean ± standard deviation (n = 6), * p < 0.05, Dunnett’s post hoc comparison test with the control ASTM.
The sublethal bioassay with the green microalgae did not reveal toxicity. The results indicated a slight decrease in the microalgae growth rate when exposed to the July (statistically validated by the One-Way ANOVA when comparing with the control MBL: F5,32 = 187.71; p < 0.00005) and to the August AWDs (statistically validated by the One-Way ANOVA when comparing with the control MBL: F5,32 = 429.55; p < 0.00002; Figure 2). The herbicides present in these samples were in much lower concentrations than those that induce the inhibition of algal growth (AMPA: 191 mg L−1; pyridafol: 4.93 mg L−1; s-metolachlor: 0.017 mg L−1; terbuthylazine: 0.012 mg L−1 [24]), justifying the obtained results. In fact, the possible toxicity of the samples may have been masked, due to the high concentrations of nutrients, mainly phosphorus (limiting nutrient for microalgae growth), such as those from April and May, stimulating the growth rate of the microalgae [44]. In fact, as demonstrated in the study of Polazzo et al. [45], this could be a key factor influencing the resilience of freshwater ecosystems to multiple stressors.
The results of the behavioral bioassay with D. magna showed no relevant toxicity, with an increase in the feeding rate of the crustacean when exposed to most of the ADW samples and a significant decrease in that performance only in the August samples (Figure 3). Previous studies carried out in surface waters highlighted the loss of sensitivity of this bioassay with the increment of nutrients and organic matter [46,47]. In fact, this behavior may also be ascribed to the high concentration of total phosphorus present in the ADWs.
The reuse of ADW as agricultural irrigation water has several advantages, such as the improvement of water use efficiency, the reduction in excess water in the downstream watercourses, and the reduction in the amount of phosphorus, suspended solids, and nitrogen carried out by the runoff to the freshwater systems [4,48]. At the same time, this water recycling system helps to close the nutrient cycles, putting them back where they belong, in the growing crop [49], avoiding their loss from the system. Results indicated that the ADW from the maize field showed low toxicity, a condition that must exist to allow its reuse in multiple uses. As for the use of ADW for crop irrigation, non-toxicity guarantees the protection of the agroecosystems’ organisms, protecting biodiversity, and evidences the low risk to the aquatic organisms of this type of irrigation water in case of a runoff.
4. Conclusions
The reuse of AWD for irrigation can be one important water management technique to cope with the growing water scarcity problems and the quantification of hazardous substances, namely, pesticides, plus the results from ecotoxicological bioassays, makes it possible to determine the potential risk to agroecosystem’s organisms. This set of results also makes it possible to assess the impact on the ecological and chemical status of receiving surface waters if drainage should occur.
In this study, the AWD samples showed some parameters above the proposed limit value for irrigation purposes, namely, ECw, NO3−, and Cl−, which indicated that this water must be used with caution, and a regular monitoring process must be implemented. Notwithstanding, the controlled reuse of this water in agricultural activities is probably a better option than its return to the hydrographic basin, as it could contribute to the reduction in the ecological status of river Tejo. The ecotoxicological evaluation, using a battery of bioassays, indicated that these ADW presented low toxicity, which is important for its reuse. However, these results must be considered with caution, and further analyses must be carried out to determine the real influence of the nutrient concentrations on the ecotoxicological results obtained. Furthermore, attention should always be paid to heavy metals, disinfection by-products, and pharmaceuticals, considered as requirements for water quality, which should be monitored when necessary and appropriate to ensure adequate protection of the environment and of human and animal health.
The low toxicity of the ADW, including toward the microalgae, evidenced other possibilities for its reuse, allowing water and nutrient recycling.
Author Contributions
Conceptualization, P.P. and P.A.; investigation, P.P., A.C., E.S. and P.A.; resources, P.P. and P.A.; writing—original draft preparation, P.P. and P.A.; writing—review and editing, P.P., P.A. and E.S.; supervision, P.A.; project administration, P.A.; funding acquisition, P.P. and P.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded through the Project AgriWW2Fertilizer, from LEAF/ISA, FCT—Fundação para a Ciência e a Tecnologia, I.P., in the scope of the Project LEAF (Ref. UIDB/04129/2020 and UIDP/04129/2020), and by the European Union through the European Regional Development Fund, included in the COMPETE 2020 (Operational Program Competitiveness and Internationalization) through the ICT project (UIDB/04683/2020) and through the FitoFarmGest Operational Group (PDR2020-101-030926).
Data Availability Statement
Data will be made available on request.
Acknowledgments
The authors are thankful to Marta Baioneta Martins for the map preparation.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C.; et al. Solutions for a cultivated planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef]
- Cameira, M.; Mota, M. Nitrogen Related Diffuse Pollution from Horticulture Production—Mitigation Practices and Assessment Strategies. Horticulturae 2017, 3, 25. [Google Scholar] [CrossRef]
- Silva, E.; Daam, M.A.; Cerejeira, M.J. Aquatic risk assessment of priority and other river basin specific pesticides in surface waters of Mediterranean river basins. Chemosphere 2015, 135, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Willison, R.S.; Nelson, K.A.; Abendroth, L.J.; Chighladze, G.; Hay, C.H.; Jia, X.; Kjaersgaard, J.; Reinhart, B.D.; Strock, J.S.; Wikle, C.K. Corn yield response to subsurface drainage water recycling in the midwestern United States. Agron. J. 2021, 113, 1865–1881. [Google Scholar] [CrossRef]
- Abdelraouf, R.E.; Marwa, M.A.; Dewedar, O.M.; El-Shafie, A.F. Smart Techniques for Improving Water Use Under the Conditions of Arid and Semi-Arid Environmental Areas: A Review. J. Chem. 2022, 65, 835–844. [Google Scholar] [CrossRef]
- Hama, J.R.; Jorgensen, D.B.G.; Diamantopoulos, E. Bucheli, T.D. Hansen, H.C.B.; Strobel, B.W. Indole and quinolizidine alkaloids from blue lupin leach to agricultural drainage water. Sci. Total Environ. 2022, 834, 155283. [Google Scholar] [CrossRef]
- Kaur, H.; Nelson, K.A.; Singh, G.; Veum, K.S.; Davis, M.P.; Udawatta, R.P.; Kaur, G. Drainage water management impacts soil properties in floodplain soils in the midwestern, USA. Agric. Water Manag. 2023, 279, 108193. [Google Scholar] [CrossRef]
- UN. United Nations Decade on Ecosystem Restoration (2021–2030). 2019, pp. 1–6. Available online: undocs.org/A/RES/73/284 (accessed on 15 May 2023).
- European Commission. Commission Staff Working Document: European Overview–River Basin Management Plans 296; European Commission: Brussels, Belgium, 2019. [Google Scholar]
- Helmecke, M.; Fries, E.; Schulte, C. Regulating water reuse for agricultural irrigation: Risks related to organic micro-contaminants. Environ. Sci. Eur. 2020, 32, 4. [Google Scholar] [CrossRef]
- Suárez-Serrano, A.; Ibáñez, C.; Lacorte, S.; Barata, C. Ecotoxicological effects of rice field waters on selected planktonic species: Comparison between conventional and organic farming. Ecotoxicology 2010, 19, 1523–1535. [Google Scholar] [CrossRef]
- SCHEER. Scientific Committee on Health, Environmental and Emerging Risks. Scientific Advice on Proposed EU Minimum quality Requirements for Water Reuse in Agricultural Irrigation and Aquifer Recharge; European Union: Luxembourg, 2017; pp. 1–24. [Google Scholar]
- Rand, G.M. Fundamentals of Aquatic Toxicology: Effects, Environmental Fate, and Risk Assessment, 2nd ed.; Taylor and Francis: Bristol, UK, 1995. [Google Scholar]
- Alvarenga, P.; Martins, M.; Ribeiro, H.; Mota, M.; Guerra, I.; Cardoso, H.; Silva, J.L. Evaluation of the fertilizer potential of Chlorella vulgaris and Scenedesmus obliquus grown in agricultural drainage water from maize fields. Sci. Total Environ. 2023, 861, 160670. [Google Scholar] [CrossRef] [PubMed]
- APA (Agência Portuguesa do Ambiente). Plano de Gestão de Região Hidrográfica 2016/2021. Parte 2. Caracterização e Diagnóstico; Anexo IV; APA: Amadora, Portugal, 2016. [Google Scholar]
- SNISOLOS. Sistema Nacional de Informação dos Solos. Available online: https://snisolos.dgadr.gov.pt (accessed on 15 May 2023).
- DGADR. Direção-Geral de Agricultura e Desenvolvimento Rural. Nota Explicativa da Carta dos Solos de Portugal e da Carta de Capacidade de Uso do Solo. Available online: https://dgadr.gov.pt (accessed on 15 May 2023).
- IUSS Working Group WRB. World Reference Base for Soil Resources. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps, 4th ed.; International Union of Soil Sciences (IUSS): Vienna, Austria, 2022. [Google Scholar]
- INIAV (Instituto Nacional de Investigação Agrária e Veterinária, I.P.). Manual de Fertilização das Culturas; INIAV: Oeiras, Portugal, 2022; ISBN 978-972-579-063-2. [Google Scholar]
- IPMA. Instituto Português do Mar e da Atmosfera. Monitorização Diária. Available online: https://www.ipma.pt/pt/oclima/monitoriza.dia/ (accessed on 15 May 2022).
- APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association, American Water Works Association and Water Environmental Federation: Washington, DC, USA, 1998. [Google Scholar]
- DGAV. SIFITO—Sistema de Gestão das Autorizações de Produtos Fitofarmacêuticos. 2021. Available online: https://sifito.dgav.pt (accessed on 15 May 2022).
- DIN 38407-36:2014-09, German Standard Methods for the Examination of Water, Waste Water and Sludge—Jointly Determinable Substances (Group F)—Part 36: Determination of Selected Active Substances of Plant Protection Products and Other Organic Substances in Water—Method Using High Performance Liquid Chromatography and Mass Spectrometric Detection (HPLC-MS/MS or -HRMS) after Direct Injection (F 36). Deutsches Institut für Normung e. V. Available online: https://www.beuth.de/en/standard/din-38407-36/208008665 (accessed on 15 May 2022).
- PPDB. Pesticide Properties DataBase. Available online: https://sitem.herts.ac.uk/aeru/ppdb/en/index.htm (accessed on 15 April 2022).
- Decreto-Lei nº 236/98, de 1 Agosto. Diário da República no. 176/1998—I Série A. 1998, pp. 3676–3722. Available online: https://diariodarepublica.pt/dr/detalhe/decreto-lei/236-1998-430457 (accessed on 15 May 2023).
- Decreto-Lei n.o 119/2019, de 21 de agosto. Diário da República n.o 159/2019. 2019, pp. 21–44. Available online: https://dre.tretas.org/dre/3824634/decreto-lei-119-2019-de-21-de-agosto (accessed on 15 May 2023).
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; FAO Irrigation and Drainage Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 1985; ISBN 978-92-5-102263-4. [Google Scholar]
- Tomaz, A.; Palma, P.; Fialho, S.; Lima, A.; Alvarenga, P.; Potes, M.; Costa, M.J.; Salgado, R. Risk assessment of irrigation-related soil salinization and sodification in Mediterranean areas. Water 2020, 12, 3569. [Google Scholar] [CrossRef]
- ISO 11348-2; Determination of Inhibitory Effect of Water Samples on the Light Emission of Vibrio Fischeri (Luminescent Bacteria Test). Part 2: Method Using Liquid-dried Bacteria. International Organization for Standardisation: Geneve, Switzerland, 1998.
- OCDE (Organization for Economic Cooperation and Development). OCDE 211, Daphnia Magna Reproduction Test; OCDE: Paris, France, 1998. [Google Scholar]
- Persoone, G. THAMNOTOXKIT FTM—Crustacean Toxicity Screening Test for Fresh-water. Standard Operational Procedure. Belgium. 1999. Available online: https://www.microbiotests.com/wp-content/uploads/2019/07/thamnocephalus-toxicity-test_thamnotoxkit-f_standard-operating-procedure.pdf (accessed on 15 May 2023).
- McWilliam, R.A.; Baird, D.J. Postexposure feeding depression: A new toxicity endpoint for use in laboratory studies with Daphnia magna. Environ. Toxicol. Chem. 2002, 21, 1198–1205. [Google Scholar] [CrossRef]
- Allen, Y.; Calow, P.; Baird, D. A mechanistic model of contaminant-induced feeding inhibition in Daphnia magna. Environ. Toxicol. Chem. 1995, 14, 1625–1630. [Google Scholar] [CrossRef]
- Finney, D.J. Probit Analysis; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- Zar, J.H. Biostatistical Analysis; Prentice-Hall International: Englewood Cliff, NJ, USA, 1996. [Google Scholar]
- Liu, Z.; Cui, S.; Fu, Q.; Zhang, F.; Zhang, Z.; Hough, R.; An, L.; Li, Y.; Zhang, L. Transport of neonicotinoid insecticides in a wetland ecosystem: Has the cultivation of different crops become the major sources? J. Environ. Manag. 2023, 339, 117838. [Google Scholar] [CrossRef]
- Tomaz, A.; Palma, P.; Alvarenga, P.; Gonçalves, M.C. Soil salinity risk in a climate change scenario and its effect on crop yield, Chapter 13. In Climate Change and Soil Interactions; Prasad, M.N.V., Pietrzykowski, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 351–398. [Google Scholar] [CrossRef]
- APA, Agência Portuguesa do Ambiente. Critérios para a Classificação das Massas de Água—Ficha Técnica, DRH/DEQA. 2021. Available online: https://apambiente.pt/sites/default/files/_Agua/DRH/ParticipacaoPublica/PGRH/2022-2027/3_Fase/PGRH_3_SistemasClassificacao.pdf (accessed on 15 May 2023).
- EPCEU (European Parliament and the Council of the European Union). Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy. Off. J. Eur. Union 2013, 226, 1–17. [Google Scholar]
- Palma, P.; Penha, A.M.; Novais, M.H.; Fialho, S.; Lima, A.; Catarino, A.; Mourinha, C.; Alvarenga, P.; Iakunin, M.; Rodrigues, G.; et al. Integrative toolbox to assess the quality of freshwater sediments contaminated with potentially toxic metals. Environ. Res. 2022, 217, 114798. [Google Scholar] [CrossRef]
- Alvarenga, P.; Palma, P. Gonçalves, A.P. Fernandes, R.M. de Varennes, A. Vallini, G. Duarte, E. Cunha-Queda, A.C. Organic residues as immobilizing agents in aided phytostabilization: (II) Effects on soil biochemical and ecotoxicological characteristics. Chemosphere 2009, 74, 1301–1308. [Google Scholar] [CrossRef]
- Alzahrani, S.; Mohammad, A.W.; Hilal, N.; Abdullah, P.; Jaafar, O. Comparative study of NF and RO membranes in the treatment of produced water II: Toxicity removal efficiency. Desalination 2013, 315, 27–32. [Google Scholar] [CrossRef]
- Joly, P.; Bonnemoy, F.; Charvy, J.C.; Bohatier, J.; Mallet, C. Toxicity assessment of the maize herbicides S-metolachlor, benoxacor, mesotrione and nicosulfuron, and their corresponding commercial formulations, alone and in mixtures, using the Microtox(®) test. Chemosphere 2013, 93, 2444–2450. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Meng, F.L.; Hu, Y.Y.; Habibul, N.; Sheng, G.P. Concentration- and nutrient-dependent cellular responses of microalgae Chlorella pyrenoidosa to perfluorooctanoic acid. Water Res. 2020, 185, 116248. [Google Scholar] [CrossRef]
- Polazzo, F.; dos Anjos, T.B.O.; Arenas-Sánchez, A.; Romo, S.; Vighi, M.; Rico, A. Effect of multiple agricultural stressors on freshwater ecosystems: The role of community structure, trophic status, and biodiversity-functioning relationships on ecosystem responses. Sci. Total Environ. 2022, 807, 151052. [Google Scholar] [CrossRef] [PubMed]
- Diogo, B.S.; Rodrigues, S.; Silva, N.; Pinto, I.; Antunes, S.C. Evidence for Links between Feeding Behavior of Daphnia magna and Water Framework Directive Elements: Case Study of Crestuma-Lever Reservoir. Water 2022, 14, 3989. [Google Scholar] [CrossRef]
- Pinto, I.; Rodrigues, S.; Lage, O.M.; Antunes, S.C. Assessment of water quality in Aguieira reservoir: Ecotoxicological tools in addition to the Water Framework Directive. Ecotoxicol. Environ. Saf. 2021, 208, 111583. [Google Scholar] [CrossRef]
- Reinhart, B.D.; Frankenberger, J.R.; Hay, C.H.; Helmers, M.J. Simulated water quality and irrigation benefits from drainage water recycling at two tile-drained sites in the US Midwest. Agric. Water Manag. 2019, 223, 105699. [Google Scholar] [CrossRef]
- Frankenberger, J.; Reinhart, B.; Nelson, K.; Bowling, L.; Hay, C.; Youssef, M.; Allred, B. Questions and Answers about Drainage Water Recycling for the Midwest. 2017. Available online: https://www.extension.purdue.edu/extmedia/ABE/ABE-156-W.pdf (accessed on 15 June 2022).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).