Fish Diversity Monitoring Using Environmental DNA Techniques in the Clarion–Clipperton Zone of the Pacific Ocean
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Sampling
2.2. eDNA Collection, Filtration, and Extraction
2.3. Metabarcoding of eDNA Samples
2.4. Bioinformatics
2.5. Statistical Analysis
3. Results
3.1. eDNA Metabarcoding Sequencing Results
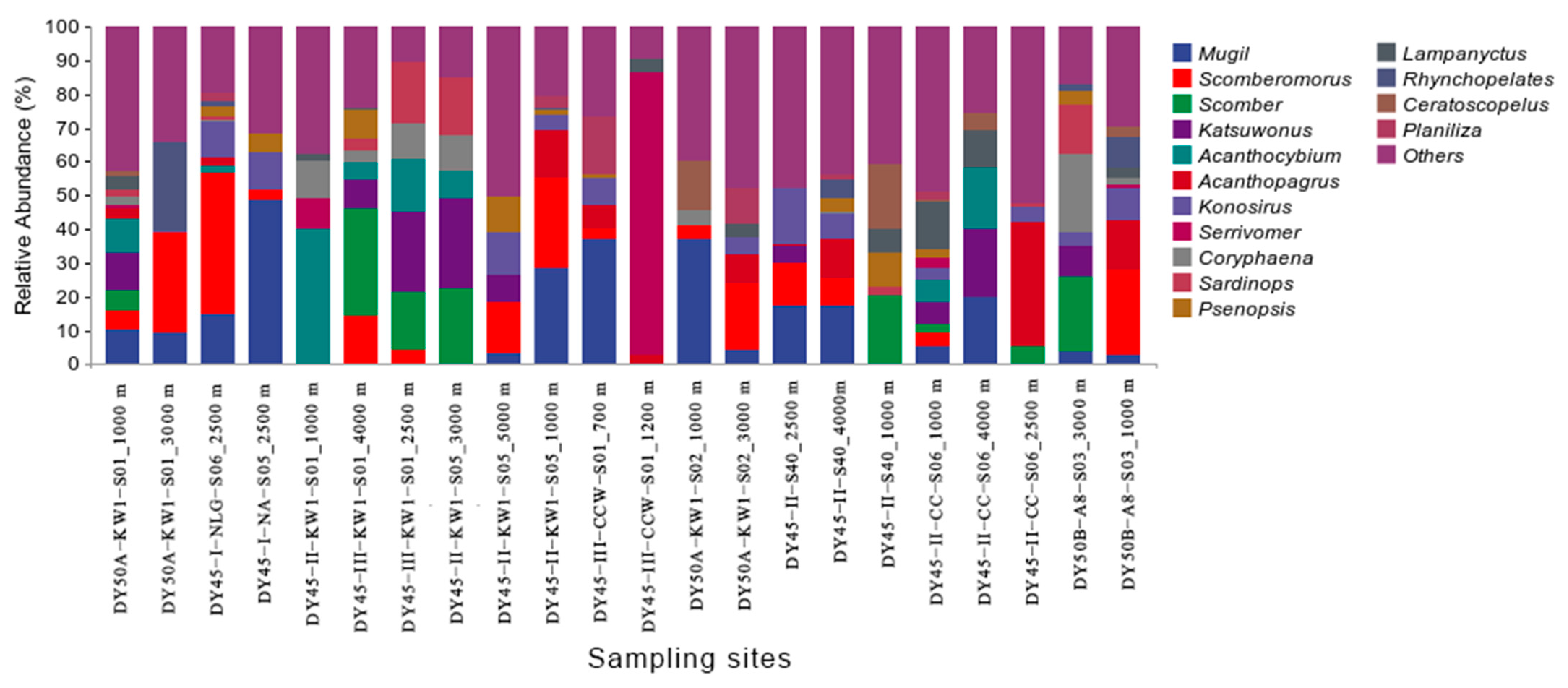
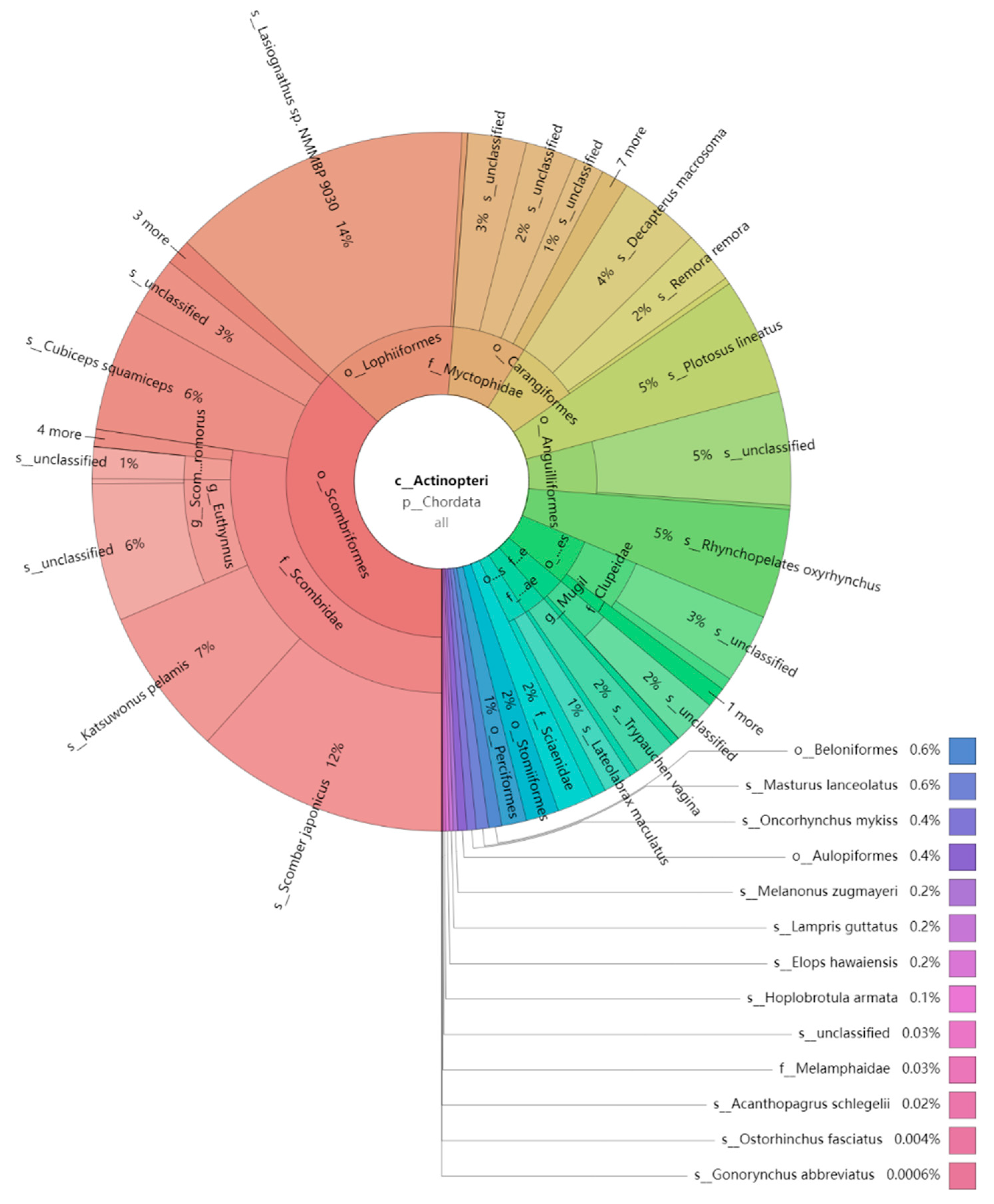
3.2. Species Composition and Diversity
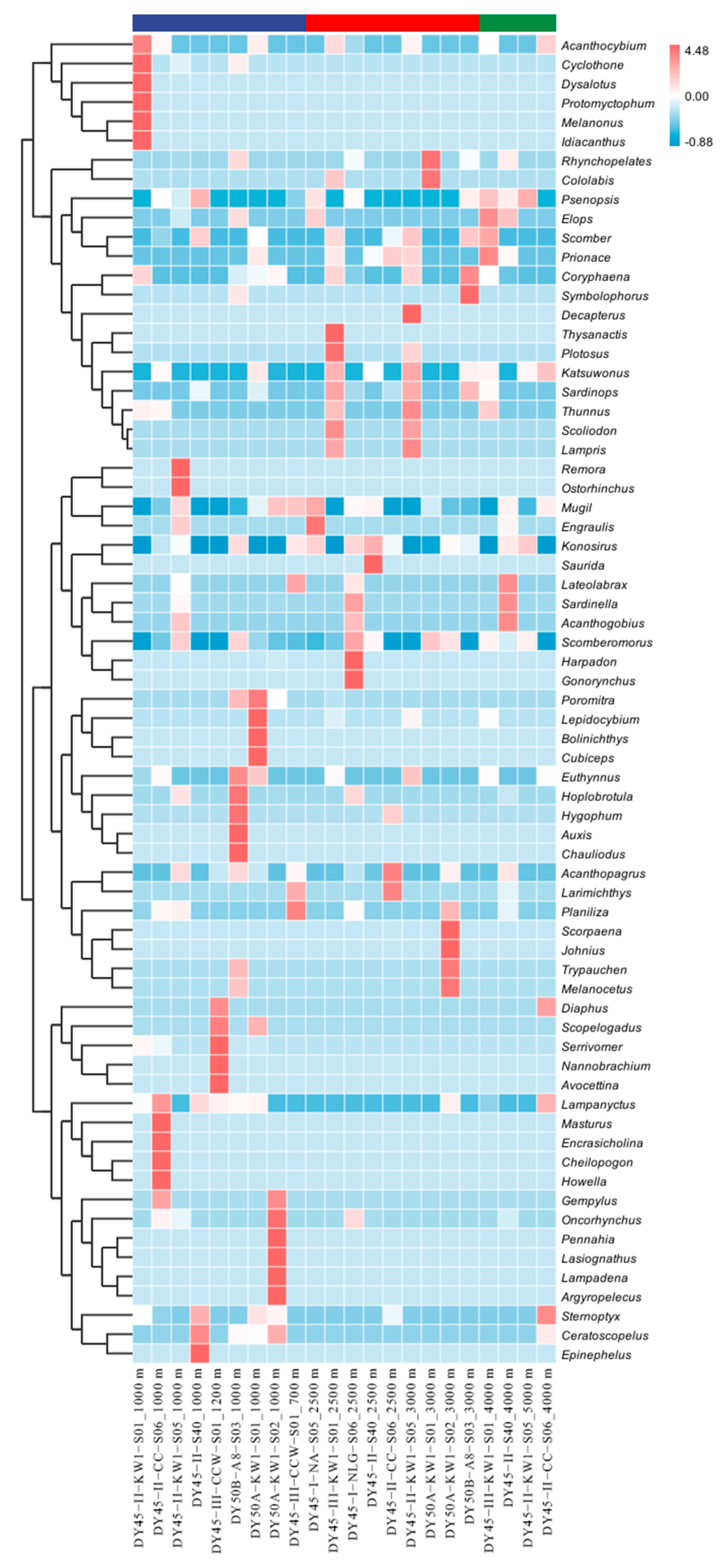
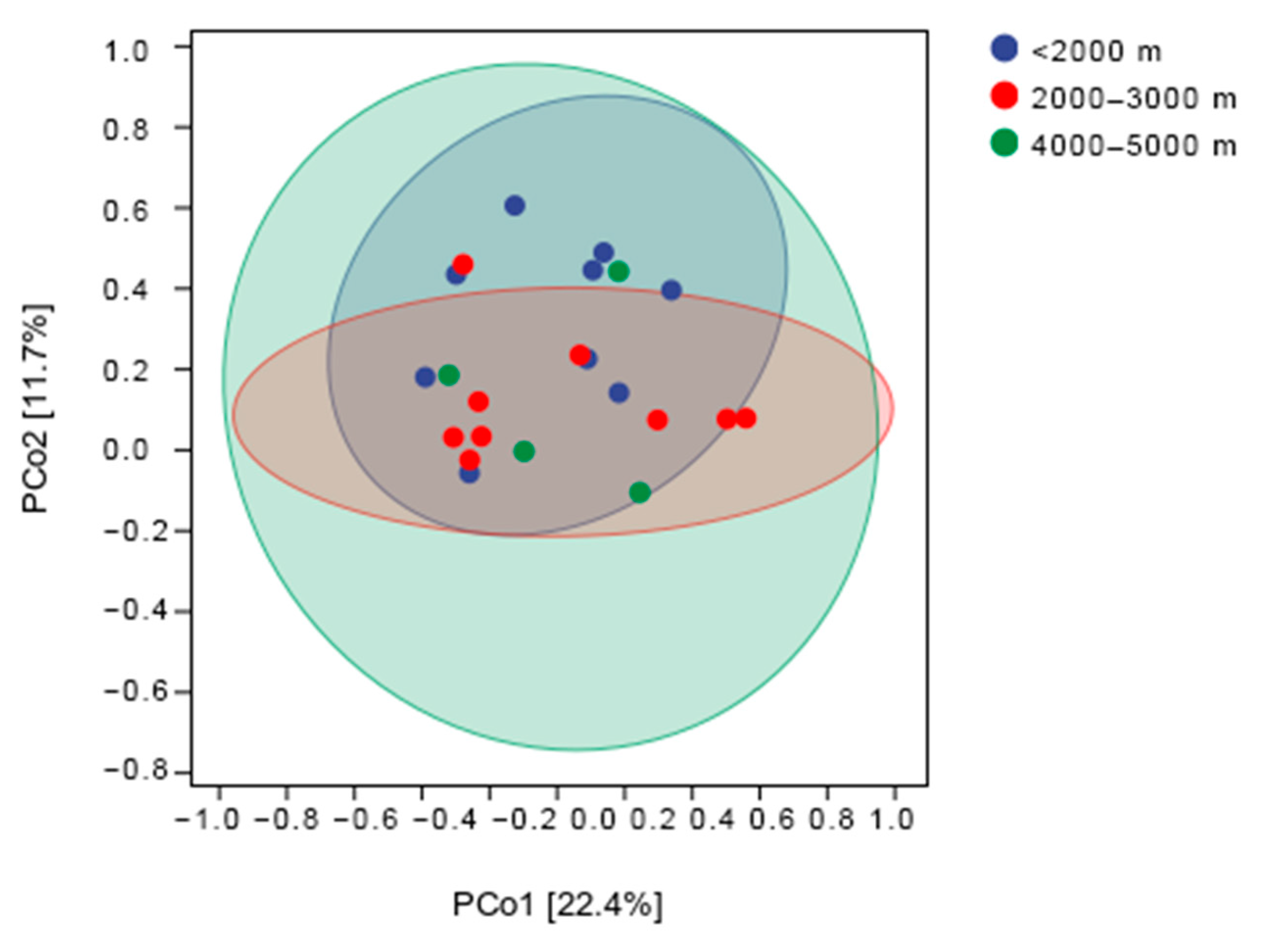
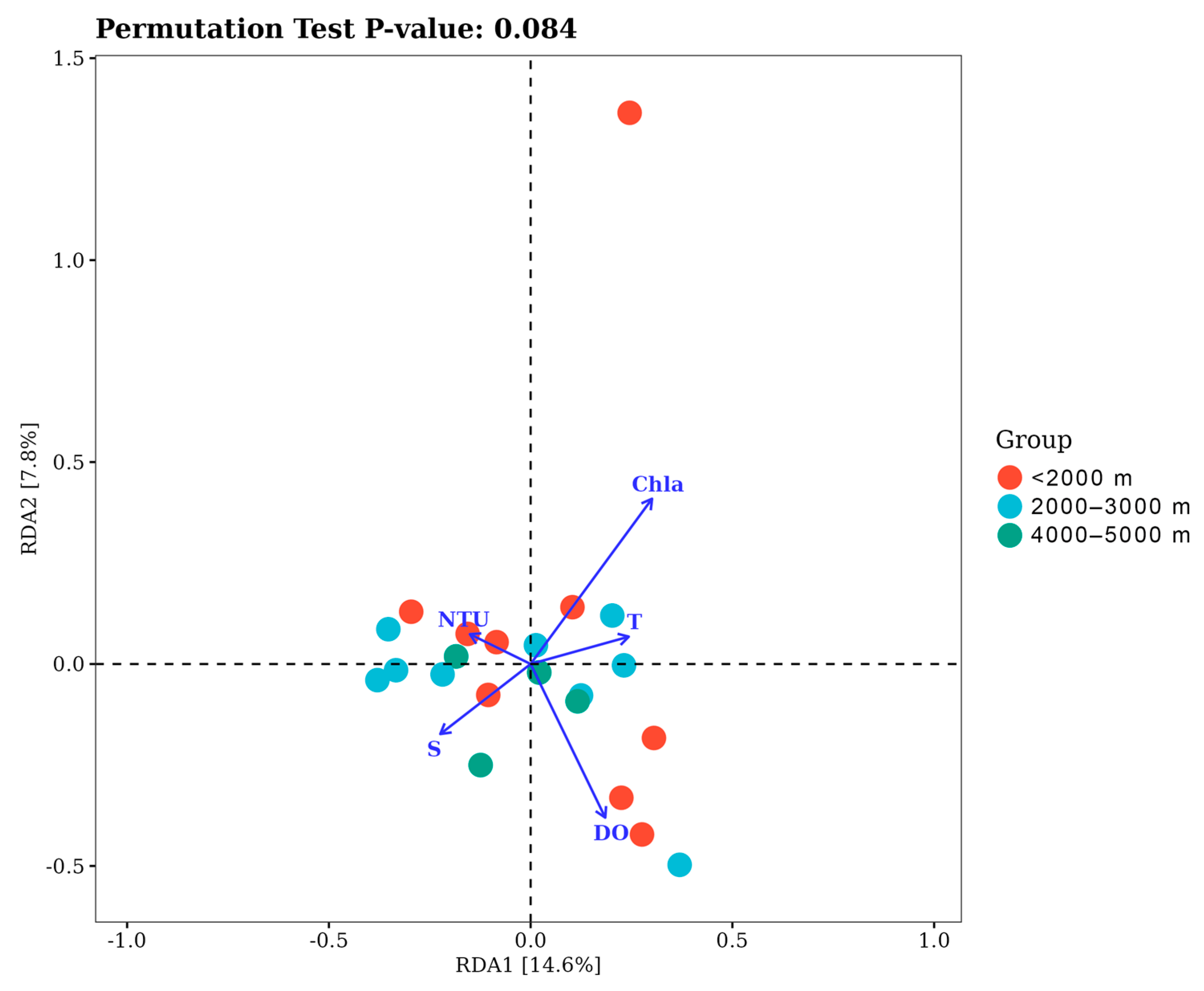
3.3. Community Diversity
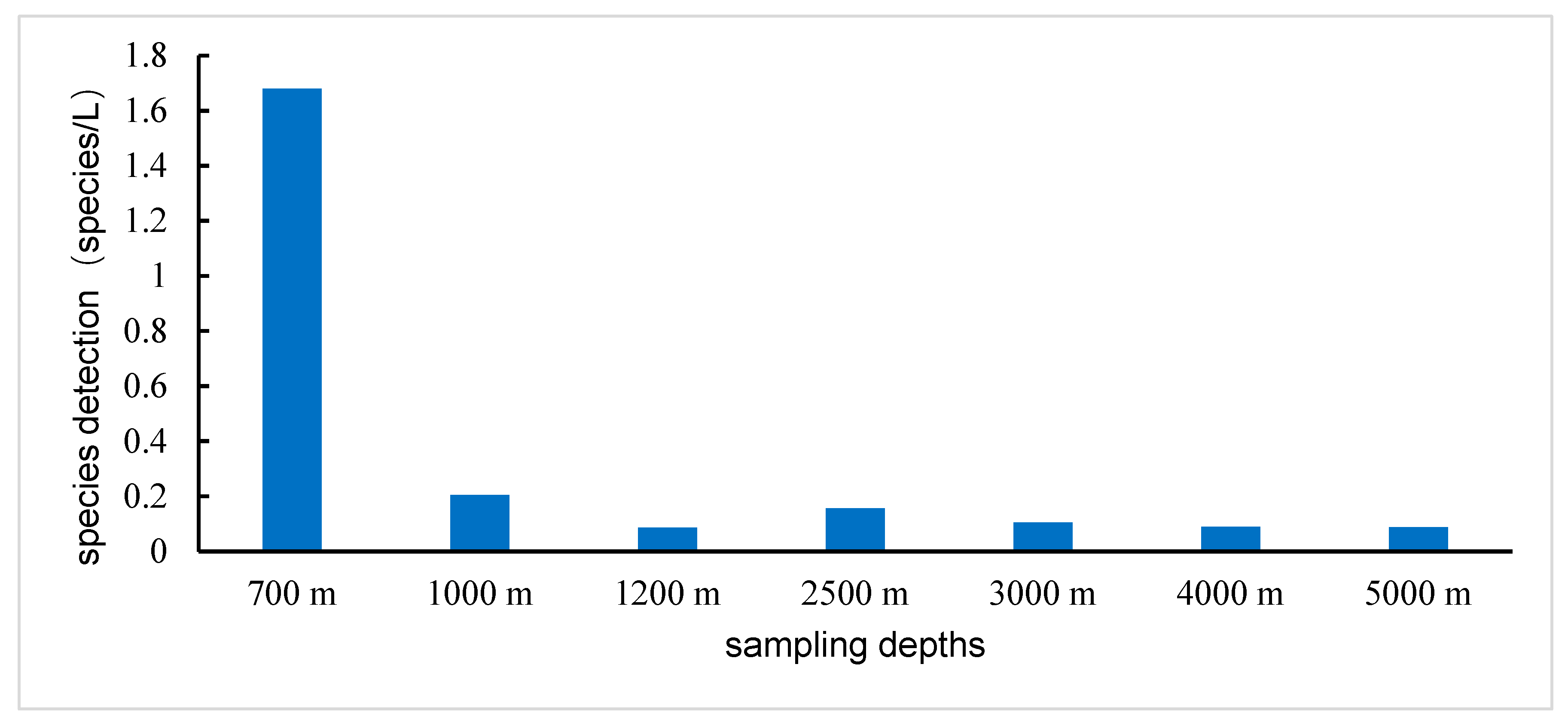
3.4. Species Distribution by Depth
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cairns, S.D. New abyssal Primnoidae (Anthozoa: Octocorallia) from the Clarion-Clipperton Fracture Zone, equatorial northeastern Pacific. Mar. Biodivers. 2016, 46, 141–150. [Google Scholar] [CrossRef]
- Jones, D.O.B.; Simon-Lledo, E.; Amon, D.J.; Bett, B.J.; Caulle, C.; Clement, L.; Connelly, D.P.; Dahlgren, T.G.; Durden, J.M.; Drazen, J.C.; et al. Environment, ecology, and potential effectiveness of an area protected from deep-sea mining (Clarion Clipperton Zone, abyssal Pacific). Prog. Oceanogr. 2021, 197, 102653. [Google Scholar] [CrossRef]
- Simon-Lledo, E.; Bett, B.J.; Huvenne, V.A.I.; Schoening, T.; Benoist, N.M.A.; Jeffreys, R.M.; Durden, J.M.; Jones, D.O.B. Megafaunal variation in the abyssal landscape of the Clarion Clipperton Zone. Prog. Oceanogr. 2019, 170, 119–133. [Google Scholar] [CrossRef]
- Smith, C.R.; Clark, M.R.; Goetze, E.; Glover, A.G.; Howell, K.L. Editorial: Biodiversity, Connectivity and Ecosystem Function Across the Clarion-Clipperton Zone: A Regional Synthesis for an Area Targeted for Nodule Mining. Front. Mar. Sci. 2021, 8, 797516. [Google Scholar] [CrossRef]
- Jakiel, A.; Palero, F.; Błażewicz, M. Deep ocean seascape and Pseudotanaidae (Crustacea: Tanaidacea) diversity at the Clarion-Clipperton Fracture Zone. Sci. Rep. 2019, 9, 17305. [Google Scholar] [CrossRef]
- Zinssmeister, C.; Wilke, T.; Hoppenrath, M. Species diversity of dinophysoid dinoflagellates in the Clarion-Clipperton Fracture Zone, eastern Pacific. Mar. Biodivers. 2017, 47, 271–287. [Google Scholar] [CrossRef]
- Taboada, S.; Riesgo, A.; Wiklund, H.; Paterson, G.L.J.; Koutsouveli, V.; Santodomingo, N.; Dale, A.C.; Smith, C.R.; Jones, D.O.B.; Dahlgren, T.G.; et al. Implications of population connectivity studies for the design of marine protected areas in the deep sea: An example of a demosponge from the Clarion-Clipperton Zone. Mol. Ecol. 2018, 27, 4657–4679. [Google Scholar] [CrossRef]
- Wang, C.-S.; Liao, L.; Xu, H.-X.; Xu, X.-W.; Wu, M.; Zhu, L.-Z. Bacterial diversity in the sediment from polymetallic nodule fields of the Clarion-Clipperton Fracture Zone. J. Microbiol. 2010, 48, 573–585. [Google Scholar] [CrossRef]
- Lindh, M.V.; Maillot, B.M.; Smith, C.R.; Church, M.J. Habitat filtering of bacterioplankton communities above polymetallic nodule fields and sediments in the Clarion-Clipperton zone of the Pacific Ocean. Environ. Microbiol. Rep. 2018, 10, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Lambshead, P.J.; Brown, C.J.; Ferrero, T.J.; Hawkins, L.E.; Smith, C.R.; Mitchell, N.J. Biodiversity of nematode assemblages from the region of the Clarion-Clipperton Fracture Zone, an area of commercial mining interest. BMC Ecol. 2003, 3, 1. [Google Scholar] [CrossRef]
- Li, Q.; Lei, Y.; Liu, J.; Shen, Y.; Huang, H.; Wang, C.; Li, H.; Li, T. Characteristics of foraminiferal communities in the western Clarion–Clipperton Zone revealed by eDNA metabarcoding. J. Sea Res. 2022, 189, 102286. [Google Scholar] [CrossRef]
- Mora, C.; Tittensor, D.P.; Myers, R.A. The completeness of taxonomic inventories for describing the global diversity and distribution of marine fishes. Proc. R. Soc. B Biol. Sci. 2008, 275, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Costello, M.J.; Wilson, S.; Houlding, B. Predicting total global species richness using rates of species description and estimates of taxonomic effort. Syst. Biol. 2012, 61, 871–883. [Google Scholar] [CrossRef]
- Stuart-Smith, R.D.; Bates, A.E.; Lefcheck, J.S.; Duffy, J.E.; Baker, S.C.; Thomson, R.J.; Stuart-Smith, J.F.; Hill, N.A.; Kininmonth, S.J.; Airoldi, L.; et al. Integrating abundance and functional traits reveals new global hotspots of fish diversity. Nature 2013, 501, 539–542. [Google Scholar] [CrossRef]
- Wang, S.P.; Yan, Z.G.; Hanfling, B.; Zheng, X.; Wang, P.Y.; Fan, J.T.; Li, J.L. Methodology of fish eDNA and its applications in ecology and environment. Sci. Total Environ. 2021, 755, 142622. [Google Scholar] [CrossRef]
- Myers, R.A.; Worm, B. Rapid worldwide depletion of predatory fish communities. Nature 2003, 423, 280–283. [Google Scholar] [CrossRef] [PubMed]
- Frank, K.T.; Petrie, B.; Choi, J.S.; Leggett, W.C. Trophic cascades in a formerly cod-dominated ecosystem. Science 2005, 308, 1621–1623. [Google Scholar] [CrossRef] [PubMed]
- Genner, M.J.; Sims, D.W.; Wearmouth, V.J.; Southall, E.J.; Southward, A.J.; Henderson, P.A.; Hawkins, S.J. Regional climatic warming drives long-term community changes of British marine fish. Proc. R. Soc. B-Biol. Sci. 2004, 271, 655–661. [Google Scholar] [CrossRef]
- Thomsen, P.F.; Willerslev, E. Environmental DNA—An emerging tool in conservation for monitoring past and present biodiversity. Biol. Conserv. 2015, 183, 4–18. [Google Scholar] [CrossRef]
- Costello, M.J.; Chaudhary, C. Marine Biodiversity, Biogeography, Deep-Sea Gradients, and Conservation. Curr. Biol. 2017, 27, R511–R527. [Google Scholar] [CrossRef]
- Zou, K.; Chen, J.; Ruan, H.; Li, Z.; Guo, W.; Li, M.; Liu, L. eDNA metabarcoding as a promising conservation tool for monitoring fish diversity in a coastal wetland of the Pearl River Estuary compared to bottom trawling. Sci. Total Environ. 2020, 702, 134704. [Google Scholar] [CrossRef]
- Pont, D.; Rocle, M.; Valentini, A.; Civade, R.; Jean, P.; Maire, A.; Roset, N.; Schabuss, M.; Zornig, H.; Dejean, T. Environmental DNA reveals quantitative patterns of fish biodiversity in large rivers despite its downstream transportation. Sci. Rep. 2018, 8, 10361. [Google Scholar] [CrossRef] [PubMed]
- Burian, A.; Mauvisseau, Q.; Bulling, M.; Domisch, S.; Qian, S.; Sweet, M. Improving the reliability of eDNA data interpretation. Mol. Ecol. Resour. 2021, 21, 1422–1433. [Google Scholar] [CrossRef] [PubMed]
- Lacoursière-Roussel, A.; Rosabal, M.; Bernatchez, L. Estimating fish abundance and biomass from eDNA concentrations: Variability among capture methods and environmental conditions. Mol. Ecol. Resour. 2016, 16, 1401–1414. [Google Scholar] [CrossRef]
- Hervé, A.; Domaizon, I.; Baudoin, J.M.; Dejean, T.; Gibert, P.; Jean, P.; Peroux, T.; Raymond, J.C.; Valentini, A.; Vautier, M.; et al. Spatio-temporal variability of eDNA signal and its implication for fish monitoring in lakes. PLoS ONE 2022, 17, e0272660. [Google Scholar] [CrossRef]
- Takeuchi, A.; Iijima, T.; Kakuzen, W.; Watanabe, S.; Yamada, Y.; Okamura, A.; Horie, N.; Mikawa, N.; Miller, M.J.; Kojima, T.; et al. Release of eDNA by different life history stages and during spawning activities of laboratory-reared Japanese eels for interpretation of oceanic survey data. Sci. Rep. 2019, 9, 6074. [Google Scholar] [CrossRef]
- Rey, A.; Carney, K.J.; Quinones, L.E.; Pagenkopp Lohan, K.M.; Ruiz, G.M.; Basurko, O.C.; Rodriguez-Ezpeleta, N. Environmental DNA Metabarcoding: A Promising Tool for Ballast Water Monitoring. Environ. Sci. Technol. 2019, 53, 11849–11859. [Google Scholar] [CrossRef]
- Miya, M.; Sato, Y.; Fukunaga, T.; Sado, T.; Poulsen, J.Y.; Sato, K.; Minamoto, T.; Yamamoto, S.; Yamanaka, H.; Araki, H.; et al. MiFish, a set of universal PCR primers for metabarcoding environmental DNA from fishes: Detection of more than 230 subtropical marine species. R. Soc. Open Sci. 2015, 2, 150088. [Google Scholar] [CrossRef]
- Diao, C.; Jia, H.; Guo, S.; Hou, G.; Xian, W.; Zhang, H. Biodiversity exploration in autumn using environmental DNA in the South China sea. Environ. Res. 2022, 204, 112357. [Google Scholar] [CrossRef]
- Fujii, K.; Doi, H.; Matsuoka, S.; Nagano, M.; Sato, H.; Yamanaka, H. Environmental DNA metabarcoding for fish community analysis in backwater lakes: A comparison of capture methods. PLoS ONE 2019, 14, e0210357. [Google Scholar] [CrossRef] [PubMed]
- Monuki, K.; Barber, P.H.; Gold, Z. eDNA captures depth partitioning in a kelp forest ecosystem. PLoS ONE 2021, 16, e0253104. [Google Scholar] [CrossRef]
- Keck, F.; Blackman, R.C.; Bossart, R.; Brantschen, J.; Couton, M.; Hürlemann, S.; Kirschner, D.; Locher, N.; Zhang, H.; Altermatt, F. Meta-analysis shows both congruence and complementarity of DNA and eDNA metabarcoding to traditional methods for biological community assessment. Mol. Ecol. 2022, 31, 1820–1835. [Google Scholar] [CrossRef]
- Nguyen, B.N.; Shen, E.W.; Seemann, J.; Correa, A.M.S.; O’Donnell, J.L.; Altieri, A.H.; Knowlton, N.; Crandall, K.A.; Egan, S.P.; McMillan, W.O.; et al. Environmental DNA survey captures patterns of fish and invertebrate diversity across a tropical seascape. Sci. Rep. 2020, 10, 6729. [Google Scholar] [CrossRef]
- Maruyama, A.; Sugatani, K.; Watanabe, K.; Yamanaka, H.; Imamura, A. Environmental DNA analysis as a non-invasive quantitative tool for reproductive migration of a threatened endemic fish in rivers. Ecol. Evol. 2018, 8, 11964–11974. [Google Scholar] [CrossRef]
- Sato, Y.; Miya, M.; Fukunaga, T.; Sado, T.; Iwasaki, W. MitoFish and MiFish Pipeline: A Mitochondrial Genome Database of Fish with an Analysis Pipeline for Environmental DNA Metabarcoding. Mol. Biol. Evol. 2018, 35, 1553–1555. [Google Scholar] [CrossRef]
- Bessey, C.; Jarman, S.N.; Simpson, T.; Miller, H.; Stewart, T.; Keesing, J.K.; Berry, O. Passive eDNA collection enhances aquatic biodiversity analysis. Commun. Biol. 2021, 4, 236. [Google Scholar] [CrossRef] [PubMed]
- Deutschmann, B.; Mueller, A.-K.; Hollert, H.; Brinkmann, M. Assessing the fate of brown trout (Salmo trutta) environmental DNA in a natural stream using a sensitive and specific dual-labelled probe. Sci. Total Environ. 2019, 655, 321–327. [Google Scholar] [CrossRef]
- Mondal, R.; Bhat, A. Temporal and environmental drivers of fish-community structure in tropical streams from two contrasting regions in India. PLoS ONE 2020, 15, e0227354. [Google Scholar] [CrossRef]
- Li, J.; Convertino, M. Temperature increase drives critical slowing down of fish ecosystems. PLoS ONE 2021, 16, e0246222. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Harrison, D.P.; Hinton, M.G.; Siegrist, Z.C.; Kiefer, D.A.J.F.O. Habitat analysis of the commercial tuna of the Eastern Tropical Pacific Ocean. Fish. Oceanogr. 2018, 27, 417–434. [Google Scholar] [CrossRef]
- DiBattista, J.D.; Fowler, A.M.; Riley, I.J.; Reader, S.; Hay, A.; Parkinson, K.; Hobbs, J.A. The use of environmental DNA to monitor impacted coastal estuaries. Mar. Pollut. Bull. 2022, 181, 113860. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Reaume, A.M.; Farrell, E.; Gaither, M.R. Comparing eDNA metabarcoding primers for assessing fish communities in a biodiverse estuary. PLoS ONE 2022, 17, e0266720. [Google Scholar] [CrossRef]
- Shen, M.; Xiao, N.; Zhao, Z.; Guo, N.; Luo, Z.; Sun, G.; Li, J. eDNA metabarcoding as a promising conservation tool to monitor fish diversity in Beijing water systems compared with ground cages. Sci. Rep. 2022, 12, 11113. [Google Scholar] [CrossRef]
- Miya, M.; Gotoh, R.O.; Sado, T. MiFish metabarcoding: A high-throughput approach for simultaneous detection of multiple fish species from environmental DNA and other samples. Fish. Sci. 2020, 86, 939–970. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J..; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Holmes, S.P. Exact sequence variants should replace operational taxonomic units in marker-gene data analysis. ISME J. 2017, 11, 2639–2643. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Misawa, K.; Kuma, K.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef]
- McDonald, D.; Price, M.N.; Goodrich, J.; Nawrocki, E.P.; DeSantis, T.Z.; Probst, A.; Andersen, G.L.; Knight, R.; Hugenholtz, P. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J. 2012, 6, 610–618. [Google Scholar] [CrossRef]
- Ondov, B.D.; Bergman, N.H.; Phillippy, A.M. Interactive metagenomic visualization in a Web browser. BMC Bioinform. 2011, 12, 385. [Google Scholar] [CrossRef]
- Chao, A. Nonparametric-Estimation of the Number of Classes in a Population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Shannon, C.E. A mathematical theory of communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Simpson, E.H. Measurement of Diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Yan, T.; He, J.; Yang, D.; Ma, Z.; Chen, H.; Zhang, Q.; Deng, F.; Ye, L.; Pu, Y.; Zhang, M.; et al. Fish Community Structure and Biomass Particle-Size Spectrum in the Upper Reaches of the Jinsha River (China). Animals 2022, 12, 3412. [Google Scholar] [CrossRef] [PubMed]
- Moushomi, R.; Wilgar, G.; Carvalho, G.; Creer, S.; Seymour, M. Environmental DNA size sorting and degradation experiment indicates the state of Daphnia magna mitochondrial and nuclear eDNA is subcellular. Sci. Rep. 2019, 9, 12500. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Gao, W.; Shen, Z.; Zhang, Y.; Bai, J.; Cai, X.; Ouyang, J.; Zhao, Y. A Review of Environmental DNA Field and Laboratory Protocols Applied in Fish Ecology and Environmental Health. Front. Environ. Sci. 2022, 10, 725360. [Google Scholar] [CrossRef]
- Simon-Lledó, E.; Bett, B.J.; Huvenne, V.A.I.; Schoening, T.; Benoist, N.M.A.; Jones, D.O.B. Ecology of a polymetallic nodule occurrence gradient: Implications for deep-sea mining. Limnol. Oceanogr. 2019, 64, 1883–1894. [Google Scholar] [CrossRef]
- Stratmann, T.; Soetaert, K.; Kersken, D.; van Oevelen, D. Polymetallic nodules are essential for food-web integrity of a prospective deep-seabed mining area in Pacific abyssal plains. Sci. Rep. 2021, 11, 12238. [Google Scholar] [CrossRef]
- Pérez-Burillo, J.; Trobajo, R.; Leira, M.; Keck, F.; Rimet, F.; Sigró, J.; Mann, D.G. DNA metabarcoding reveals differences in distribution patterns and ecological preferences among genetic variants within some key freshwater diatom species. Sci. Total Environ. 2021, 798, 149029. [Google Scholar] [CrossRef]
- Marques, V.; Castagné, P.; Polanco, A.F.; Borrero-Pérez, G.H.; Hocdé, R.; Guérin, P.; Juhel, J.B.; Velez, L.; Loiseau, N.; Letessier, T.B.; et al. Use of environmental DNA in assessment of fish functional and phylogenetic diversity. Conserv. Biol. 2021, 35, 1944–1956. [Google Scholar] [CrossRef]
- Valdivia-Carrillo, T.; Rocha-Olivares, A.; Reyes-Bonilla, H.; Domínguez-Contreras, J.F.; Munguia-Vega, A. Integrating eDNA metabarcoding and simultaneous underwater visual surveys to describe complex fish communities in a marine biodiversity hotspot. Mol. Ecol. Resour. 2021, 21, 1558–1574. [Google Scholar] [CrossRef]
- Aglieri, G.; Baillie, C.; Mariani, S.; Cattano, C.; Calò, A.; Turco, G.; Spatafora, D.; Di Franco, A.; Di Lorenzo, M.; Guidetti, P.; et al. Environmental DNA effectively captures functional diversity of coastal fish communities. Mol. Ecol. 2021, 30, 3127–3139. [Google Scholar] [CrossRef] [PubMed]
- Treberg, J.R.; Speers-Roesch, B. Does the physiology of chondrichthyan fishes constrain their distribution in the deep sea? J. Exp. Biol. 2016, 219, 615–625. [Google Scholar] [CrossRef]
- Priede, I.G.; Froese, R.; Bailey, D.M.; Bergstad, O.A.; Collins, M.A.; Dyb, J.E.; Henriques, C.; Jones, E.G.; King, N. The absence of sharks from abyssal regions of the world’s oceans. Proc. R. Soc. B Biol. Sci. 2006, 273, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Forster, G.R. Line Fishing on the Continental Slope the Selective Effect of Different Hook Patterns. J. Mar. Biol. Assoc. United Kingd. 1973, 53, 749–751. [Google Scholar] [CrossRef]
- Molinos, J.G.; Halpern, B.S.; Schoeman, D.S.; Brown, C.J.; Kiessling, W.; Moore, P.J.; Pandolfi, J.M.; Poloczanska, E.S.; Richardson, A.J.; Burrows, M.T. Climate velocity and the future global redistribution of marine biodiversity. Nat. Clim. Chang. 2016, 28, 5849–5858. [Google Scholar] [CrossRef]
- Burrows, M.T.; Bates, A.E.; Costello, M.J.; Edwards, M.; Edgar, G.J.; Fox, C.J.; Halpern, B.S.; Hiddink, J.G.; Pinsky, M.L.; Batt, R.D.; et al. Ocean community warming responses explained by thermal affinities and temperature gradients. Nat. Clim. Chang. 2019, 9, 959. [Google Scholar] [CrossRef]
- Takahara, T.; Minamoto, T.; Yamanaka, H.; Doi, H.; Kawabata, Z. Estimation of fish biomass using environmental DNA. PLoS ONE 2012, 7, e35868. [Google Scholar] [CrossRef] [PubMed]
- Ravanat, J.L.; Douki, T.; Cadet, J. Direct and indirect effects of UV radiation on DNA and its components. J. Photochem. Photobiol. B Biol. 2001, 63, 88–102. [Google Scholar] [CrossRef]
- Collins, R.A.; Wangensteen, O.S.; O’Gorman, E.J.; Mariani, S.; Sims, D.W.; Genner, M.J. Persistence of environmental DNA in marine systems. Commun. Biol. 2018, 1, 185. [Google Scholar] [CrossRef]
- Cooke, S.J.; Bergman, J.N.; Twardek, W.M.; Piczak, M.L.; Casselberry, G.A.; Lutek, K.; Dahlmo, L.S.; Birnie-Gauvin, K.; Griffin, L.P.; Brownscombe, J.W.; et al. The movement ecology of fishes. J. Fish Biol. 2022, 101, 756–779. [Google Scholar] [CrossRef] [PubMed]
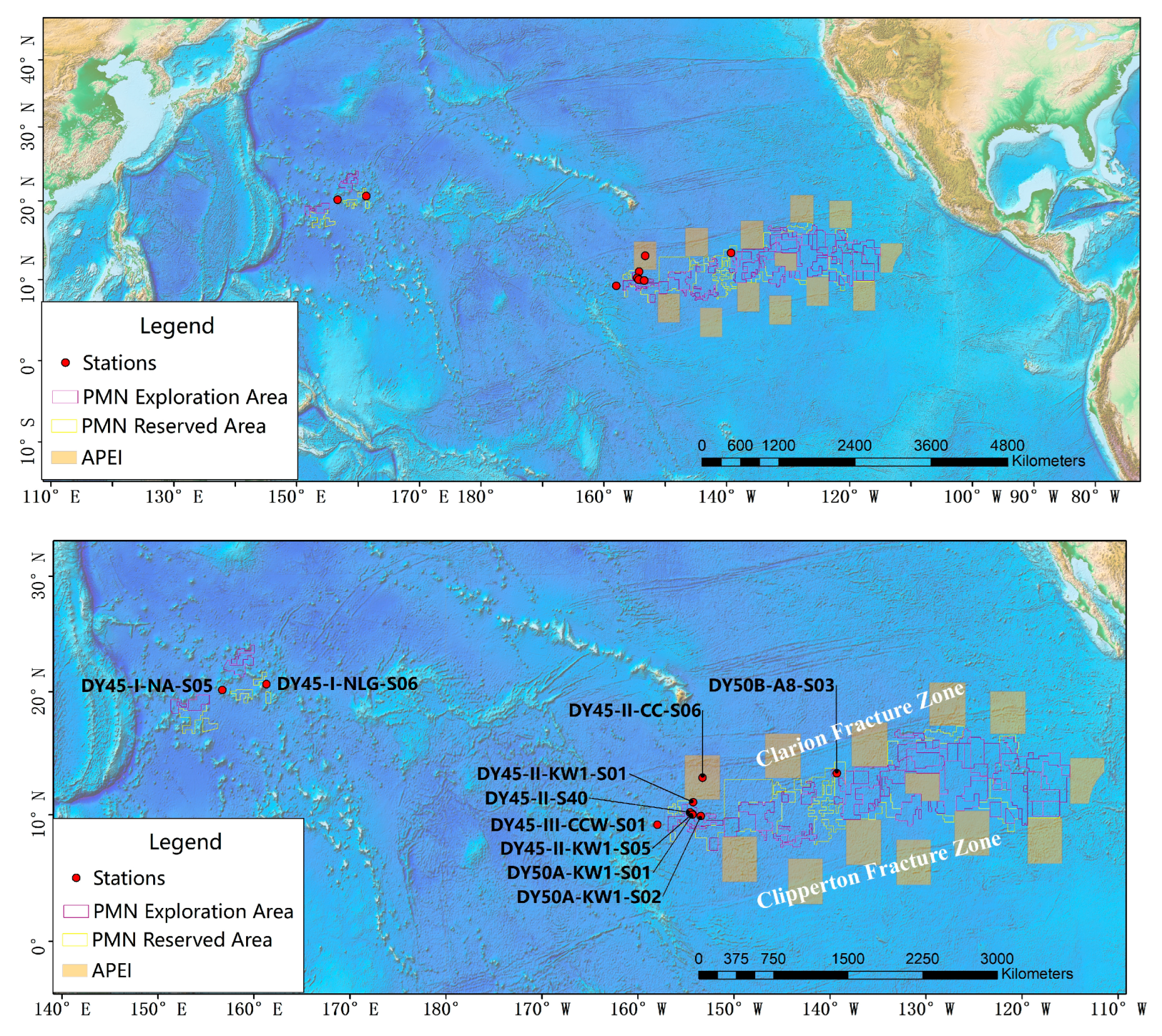

| Sample ID | Date | Latitude (N) | Longitude (E) | Depth (m) |
|---|---|---|---|---|
| DY50A-KW1-S01_1000 m | 12 August 2018 | 10.004 | 205.665 | 5198 |
| DY50A-KW1-S01_3000 m | 12 August 2018 | 10.004 | 205.665 | 5198 |
| DY45-I-NLG-S06_2500 m | 6 August 2017 | 20.637 | 161.299 | 4941 |
| DY45-I-NA-S05_2500 m | 26 August 2017 | 20.140 | 156.695 | 3584 |
| DY45-II-KW1-S01_1000 m | 18 September 2017 | 10.990 | 205.741 | 5234 |
| DY45-III-KW1-S01_4000 m | 10 October 2017 | 10.990 | 205.741 | 5236 |
| DY45-III-KW1-S01_2500 m | 10 October 2017 | 10.990 | 205.741 | 5236 |
| DY45-II-KW1-S05_3000 m | 11 September 2017 | 10.056 | 205.658 | 5169 |
| DY45-II-KW1-S05_5000 m | 11 August 2017 | 10.056 | 205.658 | 5169 |
| DY45-II-KW1-S05_1000 m | 11 July 2017 | 10.056 | 205.658 | 5169 |
| DY45-III-CCW-S01_700 m | 16 October 2017 | 9.207 | 202.015 | 1304 |
| DY45-III-CCW-S01_1200 m | 16 October 2017 | 9.207 | 202.015 | 1304 |
| DY50A-KW1-S02_1000 m | 14 August 2018 | 9.888 | 206.539 | 5083 |
| DY50A-KW1-S02_3000 m | 14 August 2018 | 9.888 | 206.539 | 5083 |
| DY45-II-S40_2500 m | 21 September 2017 | 10.187 | 205.407 | 5147 |
| DY45-II-S40_4000 m | 21 September 2017 | 10.187 | 205.407 | 5147 |
| DY45-II-S40_1000 m | 21 September 2017 | 10.187 | 205.407 | 5147 |
| DY45-II-CC-S06_1000 m | 22 August 2017 | 12.976 | 206.729 | 5524 |
| DY45-II-CC-S06_4000 m | 22 August 2017 | 12.976 | 206.729 | 5524 |
| DY45-II-CC-S06_2500 m | 22 August 2017 | 12.976 | 206.729 | 5524 |
| DY50B-A8-S03_3000 m | 6 September 2018 | 13.323 | 139.295 | 4987 |
| DY50B-A8-S03_1000 m | 6 September 2019 | 13.323 | 139.295 | 4987 |
| Order | Family | Genus/Species |
|---|---|---|
| Alepocephaliformes | Alepocephalidae | Alepocephalus sp. |
| Leptoderma spp. | ||
| Platytroctidae | ||
| Anguilliformes | Nemichthyidae | Avocettina sp |
| Serrivomeridae | Serrivomer beanii | |
| Serrivomer sector | ||
| Anguillidae | Anguilla spp. | |
| Aulopiformes | Evermannellidae | Coccorella sp. |
| Synodontidae | Saurida spp. | |
| Harpadon nehereus | ||
| Beloniformes | Exocoetidae | Cheilopogon sp. |
| Belonidae | Cololabis saira | |
| Beryciformes | Berycidae | Beryx spp. |
| Melamphaidae | Poromitra spp. | |
| Scopelogadus sp. | ||
| Carangiformes | Echeneidae | Remora remora |
| Coryphaenidae | Coryphaena hippurus | |
| Carangidae | Decapterus macrosoma | |
| Centrarchiformes | Terapontidae | Rhynchopelates oxyrhynchus |
| Kyphosidae | ||
| Clupeiformes | Clupeidae | Konosirus punctatus |
| Konosirus spp. | ||
| Sardinella spp. | ||
| Sardinops melanostictus | ||
| Sardinops sagax | ||
| Engraulidae | Encrasicholina punctifer | |
| Engraulis japonicus | ||
| Elopiformes | Elopidae | Elops hawaiensis |
| Megalopidae | Megalops spp. | |
| Gadiformes | Macrouridae | |
| Gadidae | Gadus spp. | |
| Melanonidae | Melanonus zugmayeri | |
| Gobiiformes | Gobiidae | Acanthogobius hasta |
| Exyrias spp. | ||
| Luciogobius sp. | ||
| Trypauchen vagina | ||
| Gonorynchiformes | Chanidae | Chanos sp. |
| Gonorynchidae | Gonorynchus breviatus | |
| Kurtiformes | Apogonidae | Ostorhinchus fasciatus |
| Lampriformes | Lampridae | Lampris guttatus |
| Lophiiformes | Melanocetidae | Melanocetus murrayi |
| Melanocetus johnsonii | ||
| Thaumatichthyidae | Lasiognathus sp. NMMBP 9030 | |
| Diceratiidae | Bufoceratias sp. | |
| Mugiliformes | Mugilidae | Planiliza spp. |
| Mugil cephalus | ||
| Mugil spp. | ||
| Myctophiformes | Myctophidae | Ceratoscopelus spp. |
| Diaphus aliciae | ||
| Diaphus spp. | ||
| Myctophum spp. | ||
| Nannobrachium sp. | ||
| Symbolophorus spp. | ||
| Lampanyctus spp. | ||
| Hygophum proximum | ||
| Protomyctophum thompsoni | ||
| Lampanyctus tenuiformis | ||
| Bolinichthys pyrsobolus | ||
| Bolinichthys spp. | ||
| Ophidiiformes | Ophidiidae | Hoplobrotula armata |
| Pempheriformes | Howellidae | Howella sp. |
| Lateolabracidae | Lateolabrax japonicus | |
| Lateolabrax maculatus | ||
| Perciformes | Scorpaenidae | Scorpaena spp. |
| Sebastidae | Helicolenus spp. | |
| Serranidae | Epinephelus fuscoguttatus | |
| Epinephelus sp. | ||
| Pleuronectiformes | Cynoglossidae | |
| Pomacentridae | Pomacentridae | Abudefduf spp. |
| Salmoniformes | Salmonidae | Salvelinus leucomaenis |
| Salvelinus sp. | ||
| Sciaenidae | Sciaenidae | Johnius spp. |
| Larimichthys crocea | ||
| Pennahia argentata | ||
| Miichthys miiuy | ||
| Scombriformes | Bramidae | Eumegistus illustris |
| Chiasmodontidae | Dysalotus sp. | |
| Centrolophidae | Psenopsis anomala | |
| Gempylidae | Gempylus serpens | |
| Lepidocybium flavobrunneum obrunneum flavobrunneum flavobrunneum flavobrunneum | ||
| Scombridae | Euthynnus alletteratus | |
| Euthynnus affinis | ||
| Euthynnus spp. | ||
| Auxis thazard | ||
| Scomberomorus niphonius | ||
| Scomberomorus spp. | ||
| Katsuwonus pelamis | ||
| Thunnus alalunga | ||
| Thunnus spp. | ||
| Scomber japonicus | ||
| Scomber sp. | ||
| Acanthocybium solandri | ||
| Auxis rochei | ||
| Nomeidae | Nomeus gronovii | |
| Cubiceps squamiceps | ||
| Siganidae | Siganidae | |
| Siluriformes | Plotosidae | Plotosus lineatus |
| Ariidae | Netuma spp. | |
| Stomiiformes | Gonostomatidae | Cyclothone atraria |
| Cyclothone pallida | ||
| Cyclothone obscura | ||
| Phosichthyidae | Vinciguerria nimbaria | |
| Stomiidae | Chauliodus sp. | |
| Stomias sp. | ||
| Idiacanthus antrostomus | ||
| Thysanactis spp. | ||
| Sternoptyx obscura | ||
| Sternoptychidae | Argyropelecus sladeni | |
| Argyropelecus sp. | ||
| Spariformes | Sparidae | Acanthopagrus schlegelii |
| Sphyraenidae | Sphyraenidae | Sphyraena spp. |
| Syngnathiformes | Syngnathidae | |
| Tetraodontiformes | Molidae | Masturus lanceolatus |
| Uranoscopiformes | Ammodytidae | Ammodytes sp. |
| Carcharhiniformes | Carcharhinidae | Scoliodon laticaudus |
| Prionace glauca | ||
| Hexanchidae | ||
| Myliobatiformes | Myliobatidae |
| Depths | Chao1 | Observed_Species | Shannon | Simpson |
|---|---|---|---|---|
| DY45 < 2000 m | 24.13333 | 23.93333 | 3.08599 | 0.768111 |
| DY45 2000 m–3000 m | 43.243 | 42.06667 | 3.24167 | 0.815328 |
| DY45 4000 m–5000 m | 30.03543 | 29.2 | 3.629975 | 0.885577 |
| DY50 1000 m | 36.2611 | 35.46667 | 3.78488 | 0.883085 |
| DY50 3000 m | 17.63333 | 17.56667 | 3.1269 | 0.851768 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, W.; Wang, L.; Ou, D.; Li, W.; Huang, H.; Ou, R.; Qiu, J.; Cai, L.; Lin, L.; Zhang, Y. Fish Diversity Monitoring Using Environmental DNA Techniques in the Clarion–Clipperton Zone of the Pacific Ocean. Water 2023, 15, 2123. https://doi.org/10.3390/w15112123
He W, Wang L, Ou D, Li W, Huang H, Ou R, Qiu J, Cai L, Lin L, Zhang Y. Fish Diversity Monitoring Using Environmental DNA Techniques in the Clarion–Clipperton Zone of the Pacific Ocean. Water. 2023; 15(11):2123. https://doi.org/10.3390/w15112123
Chicago/Turabian StyleHe, Weiyi, Lei Wang, Danyun Ou, Weiwen Li, Hao Huang, Rimei Ou, Jinli Qiu, Lei Cai, Lianghua Lin, and Yanxu Zhang. 2023. "Fish Diversity Monitoring Using Environmental DNA Techniques in the Clarion–Clipperton Zone of the Pacific Ocean" Water 15, no. 11: 2123. https://doi.org/10.3390/w15112123
APA StyleHe, W., Wang, L., Ou, D., Li, W., Huang, H., Ou, R., Qiu, J., Cai, L., Lin, L., & Zhang, Y. (2023). Fish Diversity Monitoring Using Environmental DNA Techniques in the Clarion–Clipperton Zone of the Pacific Ocean. Water, 15(11), 2123. https://doi.org/10.3390/w15112123





