Cellulose Acetate-Based Materials for Water Treatment in the Context of Circular Economy
Abstract
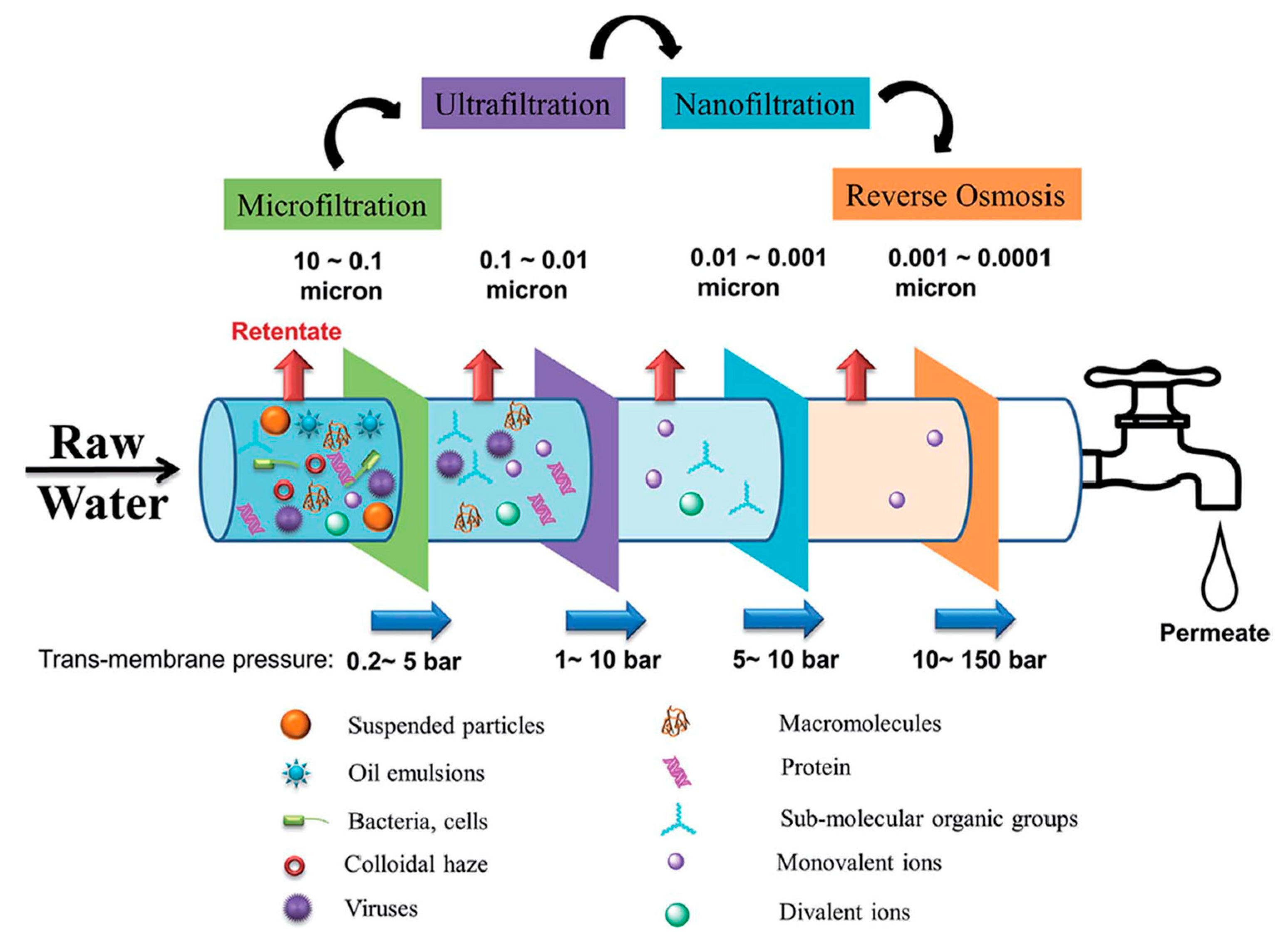
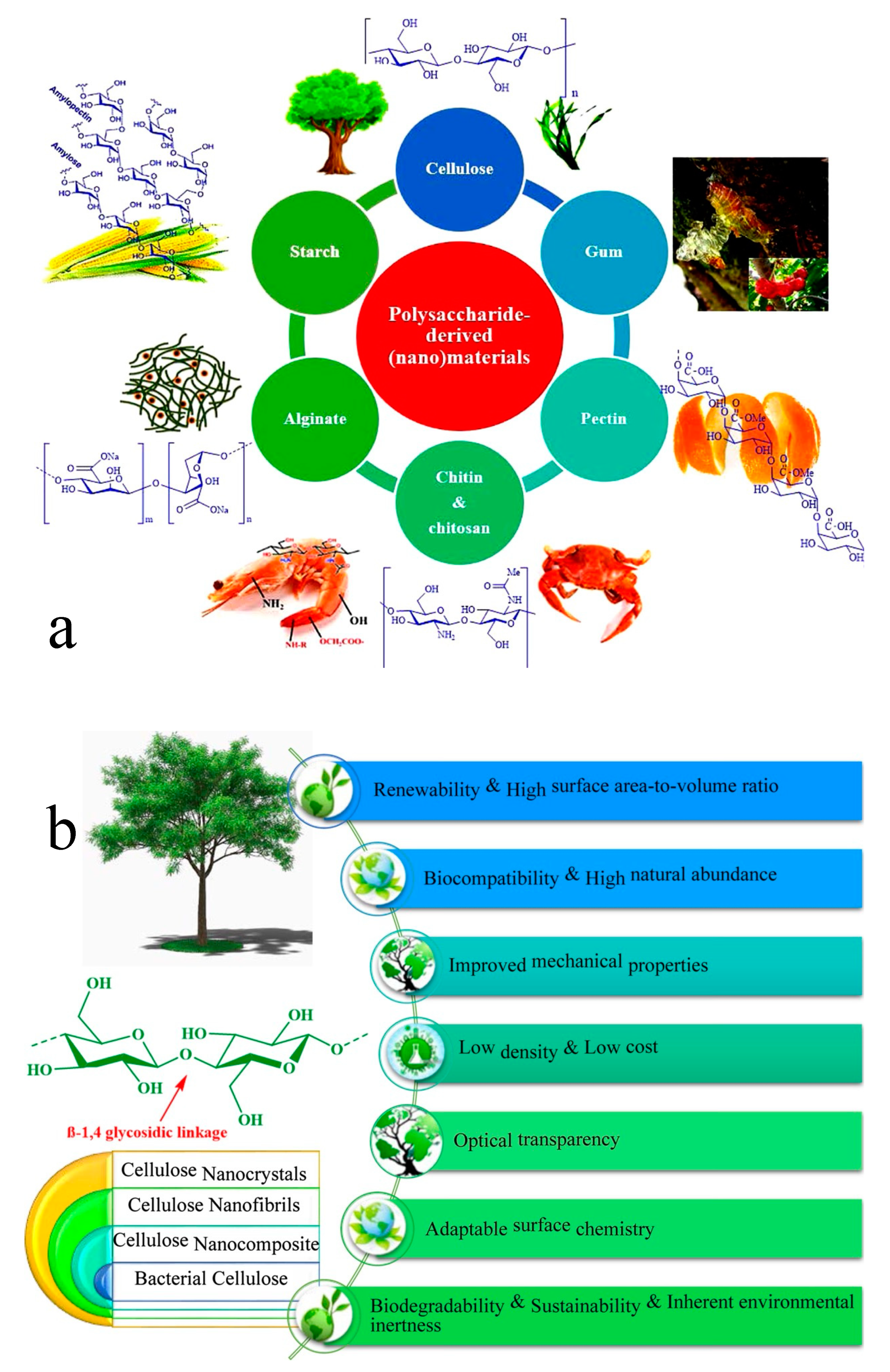
1. Introduction
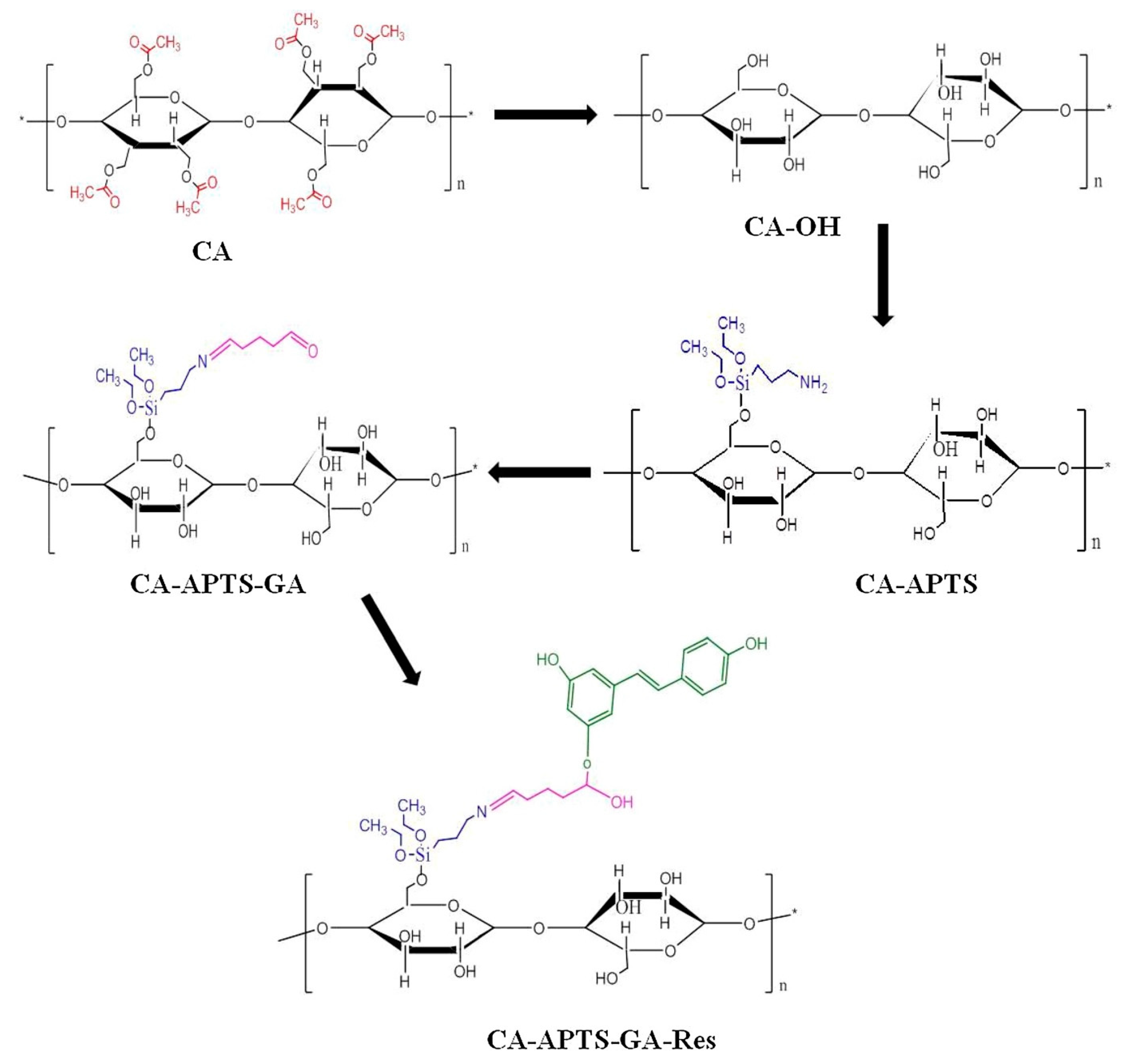
2. Cellulose Acetate Membranes in the Context of Circular Economy
3. Cellulose Acetate Membranes and Materials for Removal of Heavy Metals, Pharmaceutics and Dyes from Water
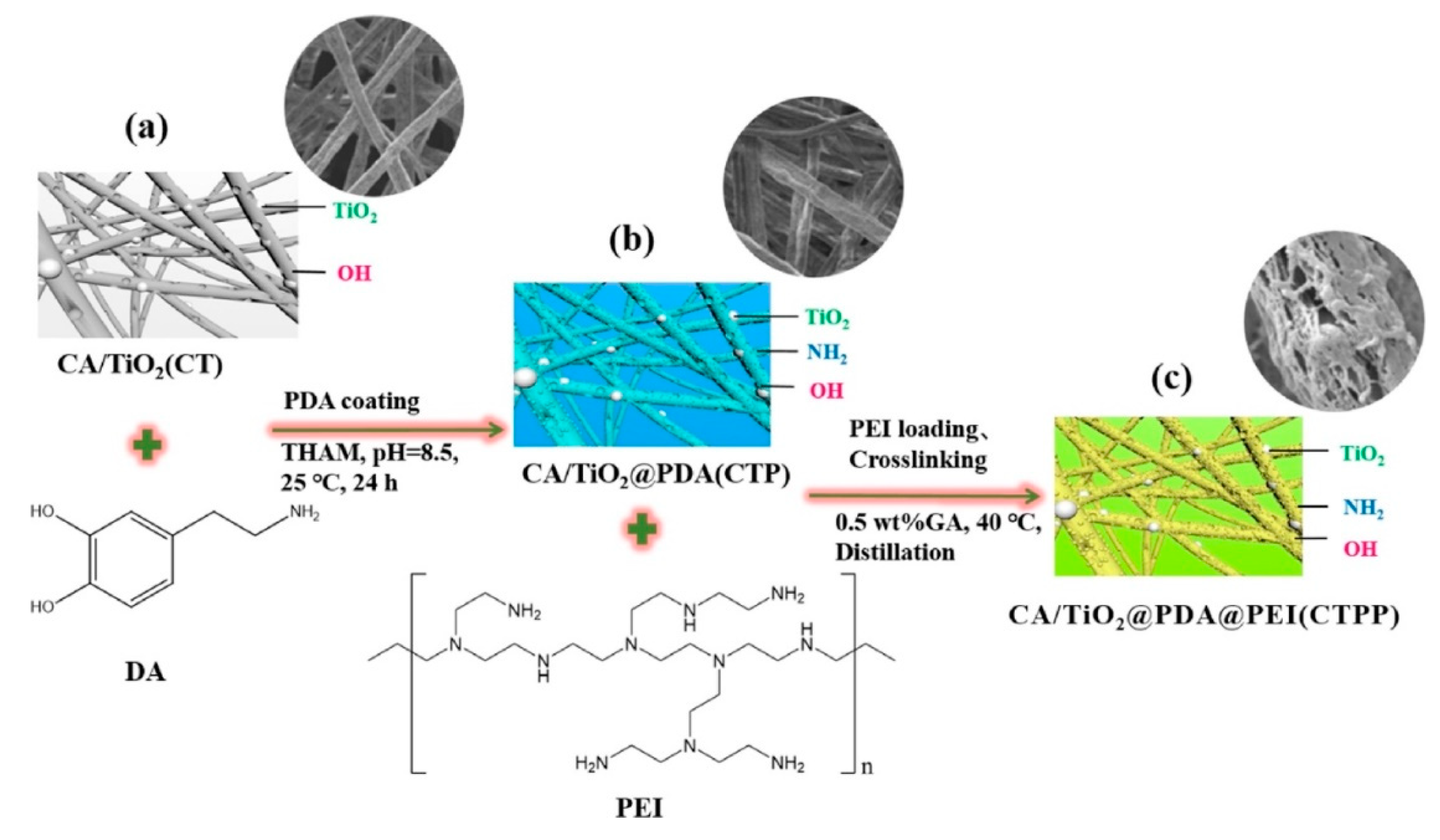
3.1. Removal of Heavy Metals from Water Using Cellulose Acetate Membranes
3.2. Removal of Pharmaceutics from Water Using Cellulose Acetate Membranes
3.3. Removal of Dyes from Water Using Cellulose Acetate Membranes
4. Conclusions
5. Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baker, R.W. Membrane Technology and Applications; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Ahmed, M.; Mavukkandy, M.O.; Giwa, A.; Elektorowicz, M.; Katsou, E.; Khelifi, O.; Naddeo, V.; Hasan, S.W. Recent developments in hazardous pollutants removal from wastewater and water reuse within a circular economy. NPJ Clean Water 2022, 5, 12. [Google Scholar] [CrossRef]
- Eke, J.; Yusuf, A.; Giwa, A.; Sodiq, A. The global status of desalination: An assessment of current desalination technologies, plants and capacity. Desalination 2020, 495, 114633. [Google Scholar] [CrossRef]
- Palit, S.J. Microfiltration, groundwater remediation and environmental engineering science-a scientific perspective and a far-reaching review. Nat. Environ. Pollut. Technol. 2015, 14, 817. [Google Scholar]
- Tsiourtis, N. Desalination and the environment. Desalination 2001, 138, 1. [Google Scholar] [CrossRef]
- Kumar, M.; Borah, P.; Devi, P. Chapter 3—Priority and emerging pollutants in water. In Inorganic Pollutants in Water; Devi, P., Singh, P., Kansal, S.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 33–49. [Google Scholar]
- Van der Bruggen, B. Chapter 2—Microfiltration, ultrafiltration, nanofiltration, reverse osmosis, and forward osmosis. In Fundamental Modelling of Membrane Systems; Luis, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 25–70. [Google Scholar]
- Charcosset, C. Ultrafiltration, microfiltration, nanofiltration and reverse osmosis in integrated membrane processes. In Integrated Membrane Systems and Processes; Wiley Online Library: Hoboken, NJ, USA, 2016; pp. 1–22. [Google Scholar]
- Miller, D.J.; Dreyer, D.R.; Bielawski, C.W.; Paul, D.R.; Freeman, B.D. Surface modification of water purification membranes. Angew. Chem. Int. Ed. 2017, 56, 4662–4711. [Google Scholar]
- Selatile, M.K.; Ray, S.S.; Ojijo, V.; Sadiku, R.J. Recent developments in polymeric electrospun nanofibrous membranes for seawater desalination. RSC Adv. 2018, 8, 37915–37938. [Google Scholar] [CrossRef]
- Liao, Y.; Loh, C.-H.; Tian, M.; Wang, R.; Fane, A.G. Progress in electrospun polymeric nanofibrous membranes for water treatment: Fabrication, modification and applications. Prog. Polym. Sci. 2018, 77, 69–94. [Google Scholar] [CrossRef]
- Oprea, M.; Voicu, S.I. Recent advances in applications of cellulose derivatives-based composite membranes with hydroxyapatite. Materials 2020, 13, 2481. [Google Scholar] [CrossRef]
- Mamah, S.C.; Goh, P.S.; Ismail, A.F.; Suzaimi, N.D.; Yogarathinam, L.T.; Raji, Y.O.; El-badawy, T.H. Recent development in modification of polysulfone membrane for water treatment application. J. Water Process Eng. 2021, 40, 101835. [Google Scholar] [CrossRef]
- Gohil, J.M.; Ray, P. A review on semi-aromatic polyamide TFC membranes prepared by interfacial polymerization: Potential for water treatment and desalination. Sep. Purif. Technol. 2017, 181, 159–182. [Google Scholar] [CrossRef]
- Johari, A.; Razmjouei, M.; Mansourizadeh, A.; Emadzadeh, D. Fabrication of blend hydrophilic polyamide imide (Torlon®)-sulfonated poly (ether ether ketone) hollow fiber membranes for oily wastewater treatment. Polym. Test. 2020, 91, 106733. [Google Scholar] [CrossRef]
- Kheirieh, S.; Asghari, M.; Afsari, M. Application and modification of polysulfone membranes. Rev. Chem. Eng. 2018, 34, 657–693. [Google Scholar] [CrossRef]
- Prihandana, G.S.; Sriani, T.; Muthi’ah, A.D.; Machmudah, A.; Mahardika, M.; Miki, N. Study effect of nAg particle size on the properties and antibacterial characteristics of polysulfone membranes. Nanomaterials 2022, 12, 388. [Google Scholar] [CrossRef]
- Lakshmi, D.S.; Saxena, M.; Radha, K.; Dass, L.A. Effect of sulfated seaweed polysaccharide on flat sheet polymer (Polysulfone) membrane properties. Chem. Eng. J. Adv. 2022, 11, 100314. [Google Scholar] [CrossRef]
- Serbanescu, O.; Voicu, S.; Thakur, V. Polysulfone functionalized membranes: Properties and challenges. Mater. Today Chem. 2020, 17, 100302. [Google Scholar] [CrossRef]
- Huang, H.-L.; Yang, S. Filtration characteristics of polysulfone membrane filters. J. Aerosol Sci. 2006, 37, 1198–1208. [Google Scholar] [CrossRef]
- Ionita, M.; Pandele, A.M.; Crica, L.; Pilan, L. Improving the thermal and mechanical properties of polysulfone by incorporation of graphene oxide. Compos. Part B Eng. 2014, 59, 133–139. [Google Scholar] [CrossRef]
- Dhanraj, N.D.; Hatha, A.A.M.; Jisha, M.S. Biodegradation of petroleum based and bio-based plastics: Approaches to increase the rate of biodegradation. Arch. Microbiol. 2022, 204, 258. [Google Scholar] [CrossRef]
- Bandehali, S.; Sanaeepur, H.; Amooghin, A.E.; Shirazian, S.; Ramakrishna, S.J.S.; Technology, P. Biodegradable polymers for membrane separation. Sep. Purif. Technol. 2021, 269, 118731. [Google Scholar] [CrossRef]
- Thiagamani, S.M.K.; Krishnasamy, S.; Siengchin, S. Challenges of biodegradable polymers: An environmental perspective. Appl. Sci. Eng. Prog. 2019, 12, 149. [Google Scholar] [CrossRef]
- Rai, P.; Mehrotra, S.; Priya, S.; Gnansounou, E.; Sharma, S.K. Recent advances in the sustainable design and applications of biodegradable polymers. Bioresour. Technol. 2021, 325, 124739. [Google Scholar] [CrossRef] [PubMed]
- Salgot, M.; Folch, M. Wastewater treatment and water reuse. Curr. Opin. Environ. Sci. Health 2018, 2, 64–74. [Google Scholar] [CrossRef]
- Biswas, A.K. Integrated Water Resources Management: Is It Working? Int. J. Water Resour. Dev. 2008, 24, 5–22. [Google Scholar] [CrossRef]
- Aryal, J.; Gautam, B.; Sapkota, N. Drinking water quality assessment. J. Nepal Health Res. Counc. 2012, 10. [Google Scholar] [CrossRef]
- Sgroi, M.; Vagliasindi, F.G.; Roccaro, P. Feasibility, sustainability and circular economy concepts in water reuse. Curr. Opin. Environ. Sci. Health 2018, 2, 20–25. [Google Scholar] [CrossRef]
- Bellver-Domingo, Á.; Hernández-Sancho, F. Circular economy and payment for ecosystem services: A framework proposal based on water reuse. J. Environ. Manag. 2022, 305, 114416. [Google Scholar] [CrossRef]
- Poff, N.L.; Brown, C.M.; Grantham, T.E.; Matthews, J.H.; Palmer, M.A.; Spence, C.M.; Wilby, R.L.; Haasnoot, M.; Mendoza, G.F.; Dominique, K.C.; et al. Sustainable water management under future uncertainty with eco-engineering decision scaling. Nat. Clim. Chang. 2016, 6, 25–34. [Google Scholar] [CrossRef]
- Li, S.; Wang, X.; Guo, Y.; Hu, J.; Lin, S.; Tu, Y.; Chen, L.; Ni, Y.; Huang, L. Recent advances on cellulose-based nanofiltration membranes and their applications in drinking water purification: A review. J. Clean. Prod. 2022, 333, 130171. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Sajjadi, M.; Iravani, S.; Varma, R.S. Starch, cellulose, pectin, gum, alginate, chitin and chitosan derived (nano) materials for sustainable water treatment: A review. Carbohydr. Polym. 2021, 251, 116986. [Google Scholar] [CrossRef]
- Vlasie, A.; Trifu, S.C.; Lupuleac, C.; Kohn, B.; Cristea, M.B. Restless legs syndrome: An overview of pathophysiology, comorbidities and therapeutic approaches (Review). Exp. Ther. Med. 2022, 23, 185. [Google Scholar] [CrossRef]
- Radulescu, I.; Dragoi, A.M.; Trifu, S.C.; Cristea, M.B. Neuroplasticity and depression: Rewiring the brain's networks through pharmacological therapy (Review). Exp. Ther. Med. 2021, 22, 131. [Google Scholar] [CrossRef]
- Voicu, S.I.; Condruz, R.M.; Mitran, V.; Cimpean, A.; Miculescu, F.; Andronescu, C.; Miculescu, M.; Thakur, V.K. Sericin Covalent Immobilization onto Cellulose Acetate Membrane for Biomedical Applications. ACS Sustain. Chem. Eng. 2016, 4, 1765–1774. [Google Scholar] [CrossRef]
- Pandele, A.M.; Neacsu, P.; Cimpean, A.; Staras, A.I.; Miculescu, F.; Iordache, A.; Voicu, S.I.; Thakur, V.K.; Toader, O.D. Cellulose acetate membranes functionalized with resveratrol by covalent immobilization for improved osseointegration. Appl. Surf. Sci. 2018, 438, 2–13. [Google Scholar] [CrossRef]
- Pandele, A.M.; Comanici, F.E.; Carp, C.A.; Miculescu, F.; Voicu, S.I.; Thakur, V.K.; Serban, B.C. Synthesis and characterization of cellulose acetate-hydroxyapatite micro and nano composites membranes for water purification and biomedical applications. Vacuum 2017, 146, 599–605. [Google Scholar] [CrossRef]
- Elsayed, M.T.; Hassan, A.A.; Abdelaal, S.A.; Taher, M.M.; Ahmed, M.K.; Shoueir, K.R. Morphological, antibacterial, and cell attachment of cellulose acetate nanofibers containing modified hydroxyapatite for wound healing utilizations. J. Mater. Res. Technol. 2020, 9, 13927–13936. [Google Scholar] [CrossRef]
- Sofi, H.S.; Akram, T.; Shabir, N.; Vasita, R.; Jadhav, A.H.; Sheikh, F.A. Regenerated cellulose nanofibers from cellulose acetate: Incorporating hydroxyapatite (HAp) and silver (Ag) nanoparticles (NPs), as a scaffold for tissue engineering applications. Mater. Sci. Eng. C 2021, 118, 111547. [Google Scholar] [CrossRef]
- Landaburu-Aguirre, J.; García-Pacheco, R.; Molina, S.; Rodríguez-Sáez, L.; Rabadán, J.; García-Calvo, E. Fouling prevention, preparing for re-use and membrane recycling. Towards circular economy in RO desalination. Desalination 2016, 393, 16–30. [Google Scholar] [CrossRef]
- Tavares, T.; Leon, F.; Vaswani, J.; Peñate, B.; Ramos-Martín, A. Study for recycling water treatment membranes and components towards a circular economy—Case of Macaronesia area. Membranes 2022, 12, 970. [Google Scholar] [CrossRef]
- Jjagwe, J.; Olupot, P.W.; Menya, E.H.; Kalibbala, M. Synthesis and application of granular activated carbon from biomass waste materials for water treatment: A review. J. Bioresour. Bioprod. 2021, 6, 92–322. [Google Scholar] [CrossRef]
- Obey, G.; Adelaide, M.; Ramaraj, R. Biochar derived from non-customized matamba fruit shell as an adsorbent for wastewater treatment. J. Bioresour. Bioprod. 2022, 7, 109–115. [Google Scholar] [CrossRef]
- El-Aswar, E.I.; Ramadan, H.; Elkik, H.; Taha, A.G. A comprehensive review on preparation, functionalization and recent applications of nanofiber membranes in wastewater treatment. J. Environ. Manag. 2022, 301, 113908. [Google Scholar] [CrossRef]
- Yang, S.; Tang, R.; Dai, Y.; Wang, T.; Zeng, Z.; Zhang, L. Fabrication of cellulose acetate membrane with advanced ultrafiltration performances and antibacterial properties by blending with HKUST-1@ LCNFs. Sep. Purif. Technol. 2022, 279, 119524. [Google Scholar] [CrossRef]
- Ho, N.A.D.; Leo, C.P. A review on the emerging applications of cellulose, cellulose derivatives and nanocellulose in carbon capture. Environ. Res. 2021, 197, 111100. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.; Hakkarainen, M. Degradable or not? Cellulose acetate as a model for complicated interplay between structure, environment and degradation. Chemosphere 2021, 265, 128731. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Voicu, S.I. Recent advances in cellulose and chitosan based membranes for water purification: A concise review. Carbohydr. Polym. 2016, 146, 148–165. [Google Scholar] [CrossRef]
- Vatanpour, V.; Pasaoglu, M.E.; Barzegar, H.; Teber, O.O.; Kaya, R.; Bastug, M.; Koyuncu, I. Cellulose acetate in fabrication of polymeric membranes: A review. Chemosphere 2022, 295, 133914. [Google Scholar] [CrossRef]
- Konwarh, R.; Karak, N.; Misra, M. Electrospun cellulose acetate nanofibers: The present status and gamut of biotechnological applications. Biotechnol. Adv. 2013, 31, 421–437. [Google Scholar] [CrossRef]
- Mu, K.; Zhang, D.; Shao, Z.; Qin, D.; Wang, Y.; Wang, S. Enhanced permeability and antifouling performance of cellulose acetate ultrafiltration membrane assisted by l-DOPA functionalized halloysite nanotubes. Carbohydr. Polym. 2016, 174, 688–696. [Google Scholar] [CrossRef]
- Bifari, E.N.; Khan, S.B.; Alamry, K.A.; Asiri, M.A.; Akhtar, K. Cellulose acetate based nanocomposites for biomedical applications: A review. Curr. Pharm. Des. 2016, 22, 3007–3019. [Google Scholar] [CrossRef]
- Sanoja-López, K.A.; Quiroz-Suárez, K.A.; Dueñas-Rivadeneira, A.A.; Maddela, N.R.; Montenegro, M.C.B.; Luque, R.; Rodríguez-Díaz, J.M. Polymeric membranes functionalized with nanomaterials (MP@ NMs): A review of advances in pesticide removal. Environ. Res. 2022, 217, 114776. [Google Scholar] [CrossRef]
- Ach, A. Biodegradable plastics based on cellulose acetate. J. Macromol. Sci. Part A Pure Appl. Chem. 1993, 30, 733–740. [Google Scholar] [CrossRef]
- Puls, J.; Wilson, S.A.; Hölter, D. Degradation of cellulose acetate-based materials: A review. J. Polym. Environ. 2011, 19, 152–165. [Google Scholar] [CrossRef]
- Zhang, M.; Liao, B.-q.; Zhou, X.; He, Y.; Hong, H.; Lin, H.; Chen, J. Effects of hydrophilicity/hydrophobicity of membrane on membrane fouling in a submerged membrane bioreactor. Bioresour. Technol. 2015, 175, 59–67. [Google Scholar] [CrossRef]
- Abu-Zurayk, R.; Alnairat, N.; Khalaf, A.; Ibrahim, A.A.; Halaweh, G. Cellulose acetate membranes: Fouling types and antifouling strategies—A brief review. Processes 2023, 11, 489. [Google Scholar] [CrossRef]
- Shami, R.; Sabir, A.; Iqbal, S.S.; Gull, N.; Zohra, R.; Khan, S.M. Synergistic effect of GO/ZnO loading on the performance of cellulose acetate/chitosan blended reverse osmosis membranes for NOM rejection. Heliyon 2023, 9, e13736. [Google Scholar] [CrossRef]
- Han, B.; Zhang, D.; Shao, Z.; Kong, L.; Lv, S. Preparation and characterization of cellulose acetate/carboxymethyl cellulose acetate blend ultrafiltration membranes. Desalination 2013, 311, 80–89. [Google Scholar] [CrossRef]
- Athira, V.B.; Mohanty, S.; Nayak, S.K. Preparation and characterization of porous polyethersulfone (PES) membranes with improved biocompatibility by blending sulfonated polyethersulfone (SPES) and cellulose acetate (CA)—A comparative study. Mater. Today Commun. 2020, 25, 101544. [Google Scholar] [CrossRef]
- Azhar, O.; Jahan, Z.; Sher, F.; Niazi, M.B.K.; Kakar, S.J.; Shahid, M. Cellulose acetate-polyvinyl alcohol blend hemodialysis membranes integrated with dialysis performance and high biocompatibility. Mat. Sci. Eng. C 2021, 126, 112127. [Google Scholar] [CrossRef]
- Vinodhini, P.A.; Sangeetha, K.; Thandapani, G.; Sudha, P.N.; Jayachandran, V.; Sukumaran, A. FTIR, XRD and DSC studies of nanochitosan, cellulose acetate and polyethylene glycol blend ultrafiltration membranes. Int. J. Biol. Macromol. 2017, 104, 1721–1729. [Google Scholar] [CrossRef]
- Zavastin, D.; Cretescu, I.; Bezdadea, M.; Bourceanu, M.; Drăgan, M.; Lisa, G.; Mangalagiu, I.; Vasić, V.; Savić, J. Preparation, characterization and applicability of cellulose acetate–polyurethane blend membrane in separation techniques. Colloids Surf. A Physicochem. Eng. Asp. 2010, 370, 120–128. [Google Scholar] [CrossRef]
- Tyrka, M.; Nowak, M.; Misic, D.; Półbrat, T.; Koter, S.; Trusek, A.; Zizovic, I. Cellulose acetate membranes modification by aminosilane grafting in supercritical carbon dioxide towards antibiofilm properties. Membranes 2022, 12, 33. [Google Scholar] [CrossRef] [PubMed]
- Asiri, A.M.; Petrosino, F.; Pugliese, V.; Khan, S.B.; Alamry, K.A.; Alfifi, S.Y.; Marwani, H.M.; Alotaibi, M.M.; Algieri, C.; Chakraborty, S. Synthesis and characterization of blended cellulose acetate membranes. Polymers 2022, 14, 4. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.A.; Hilliou, L.; de Amorim, M.T.P. Fabrication of pristine-multiwalled carbon nanotubes/cellulose acetate composites for removal of methylene blue. Polym. Bull. 2020, 77, 623–653. [Google Scholar] [CrossRef]
- Voicu, S.I.; Thakur, V.K. Aminopropyltriethoxysilane as a linker for cellulose-based functional materials: New horizons and future challenges. Curr. Opin. Green Sustain. Chem. 2021, 30, 100480. [Google Scholar] [CrossRef]
- Voicu, S.; Dobrica, A.; Sava, S.; Ivan, A.; Naftanaila, L. Cationic surfactants-controlled geometry and dimensions of polymeric membrane pores. J. Optoelectron. Adv. Mater. 2012, 14, 923–928. [Google Scholar]
- Voicu, S.I.; Ninciuleanu, C.M.; Muhulet, O.; Miculescu, M. Cellulose acetate membranes with controlled porosity and their use for the separation of aminoacids and proteins. J. Optoelectron. Adv. Mater. 2014, 16, 903–908. [Google Scholar]
- Halleb, A.; Nakajima, M.; Yokoyama, F.; Neves, M.A. Effect of Surfactants on Reverse Osmosis Membrane Performance. Separations 2023, 10, 168. [Google Scholar] [CrossRef]
- Wang, J.; Song, H.; Ren, L.; Talukder, M.E.; Chen, S.; Shao, J. Study on the Preparation of Cellulose Acetate Separation Membrane and New Adjusting Method of Pore Size. Membranes 2022, 12, 9. [Google Scholar] [CrossRef]
- Figueiredo, A.S.; Ferraria, A.M.; Botelho do Rego, A.M.; Monteiro, S.; Santos, R.; Minhalma, M.; Sánchez-Loredo, M.G.; Tovar-Tovar, R.L.; de Pinho, M.N. Bactericide activity of cellulose acetate/silver nanoparticles asymmetric membranes: Surfaces and porous structures role. Membranes 2023, 13, 4. [Google Scholar] [CrossRef]
- Peng, K.; Huang, Y.; Peng, N.; Chang, C. Antibacterial nanocellulose membranes coated with silver nanoparticles for oil/water emulsions separation. Carbohydr. Polym. 2022, 278, 118929. [Google Scholar] [CrossRef]
- Rusen, E.; Mocanu, A.; Nistor, L.C.; Dinescu, A.; Călinescu, I.; Mustăţea, G.; Voicu, Ş.I.; Andronescu, C.; Diacon, A. Design of antimicrobial membrane based on polymer colloids/multiwall carbon nanotubes hybrid material with silver nanoparticles. ACS Appl. Mater. Interfaces 2014, 6, 17384–17393. [Google Scholar] [CrossRef]
- Tomczak, W.; Gryta, M. The application of cellulose acetate membranes for separation of fermentation broths by the reverse osmosis: A feasibility study. Int. J. Mol. Sci. 2022, 23, 11738. [Google Scholar] [CrossRef]
- Elbadawi, N.A.; Ramadan, A.R.; Esawi, A.M.K. Studying the effect of shortening carbon nanotubes via ball milling on cellulose acetate nanocomposite membranes for desalination applications. Membranes 2022, 12, 474. [Google Scholar] [CrossRef]
- Wang, R.; Chen, D.; Wang, Q.; Ying, Y.; Gao, W.; Xie, L. Recent advances in applications of carbon nanotubes for desalination: A review. Nanomaterials 2020, 10, 1203. [Google Scholar] [CrossRef]
- Krystyjan, M.; Khachatryan, G.; Khachatryan, K.; Krzan, M.; Ciesielski, W.; Zarska, S.; Szczepankowska, J. Polysaccharides composite materials as carbon nanoparticles carrier. Polymers 2022, 14, 948. [Google Scholar] [CrossRef]
- Jamil, T.S.; Nasr, R.A.; Abbas, H.A.; Ragab, T.I.M.; Xabela, S.; Moutloali, R. Low-cost high performance polyamide thin film composite (cellulose triacetate/graphene oxide) membranes for forward osmosis desalination from palm fronds. Membranes 2022, 12, 6. [Google Scholar] [CrossRef]
- Li, T.; Wang, Y.; Wang, X.; Cheng, C.; Zhang, K.; Yang, J.; Han, G.; Wang, Z.; Wang, X.; Wang, L. Desalination characteristics of cellulose acetate fo membrane incorporated with zif-8 nanoparticles. Membranes 2022, 12, 122. [Google Scholar] [CrossRef]
- Batool, M.; Shafeeq, A.; Haider, B.; Ahmad, N.M. TiO2 nanoparticle filler-based mixed-matrix PES/CA nanofiltration membranes for enhanced desalination. Membranes 2021, 11, 433. [Google Scholar] [CrossRef]
- Hussain, A.; Al-Yaari, M. Development of polymeric membranes for oil/water separation. Membranes 2021, 11, 42. [Google Scholar] [CrossRef]
- Yu, X.; Zhang, X.; Xing, Y.; Zhang, H.; Jiang, W.; Zhou, K.; Li, Y. Development of Janus cellulose acetate fiber (CA) membranes for highly efficient oil–water separation. Materials 2021, 14, 5916. [Google Scholar] [CrossRef]
- Ang, M.B.M.Y.; Devanadera, K.P.O.; Duena, A.N.R.; Luo, Z.-Y.; Chiao, Y.-H.; Millare, J.C.; Aquino, R.R.; Huang, S.-H.; Lee, K.-R. Modifying cellulose acetate mixed-matrix membranes for improved oil–water separation: Comparison between sodium and organo-montmorillonite as particle additives. Membranes 2021, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Mousa, H.M.; Alfadhel, H.; Abouel Nasr, E. Engineering and characterization of antibacterial coaxial nanofiber membranes for oil/water separation. Polymers 2020, 12, 2597. [Google Scholar] [CrossRef] [PubMed]
- Kapahi, M.; Sachdeva, S. Bioremediation options for heavy metal pollution. J. Health Pollut. 2019, 9, 191203. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Liu, L.; Yang, F. Removal of aqueous Hg(II) and Cr(VI) using phytic acid doped polyaniline/cellulose acetate composite membrane. J. Hazard. Mater. 2014, 280, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Luo, D.; Song, W.; Lu, Y.; Cheng, N.; Zhang, J.; Huang, T.; Wang, Y. Polydopamine-assisted polyethyleneimine grafting on electrospun cellulose acetate/TiO2 fibers towards highly efficient removal of Cr(VI). Eur. Polym. J. 2022, 180, 111632. [Google Scholar] [CrossRef]
- Costa, A.F.; Van Der Pol, C.B.; Maralani, P.J.; McInnes, M.D.; Shewchuk, J.R.; Verma, R.; Hurrell, C.; Schieda, N. Gadolinium deposition in the brain: A systematic review of existing guidelines and policy statement issued by the canadian association of radiologists. Can. Assoc. Radiol. J. 2018, 69, 373–382. [Google Scholar] [CrossRef]
- Abujudeh, H.H.; Kosaraju, V.K.; Kaewlai, R. Acute adverse reactions to gadopentetate dimeglumine and gadobenate dimeglumine: Experience with 32,659 injections. Am. J. Roentgenol. 2010, 194, 430–434. [Google Scholar] [CrossRef]
- Elsofany, E. Removal of lanthanum and gadolinium from nitrate medium using Aliquat-336 impregnated onto Amberlite XAD-4. J. Hazard. Mater. 2008, 153, 948–954. [Google Scholar] [CrossRef]
- Serbanescu, O.S.; Pandele, A.M.; Oprea, M.; Semenescu, A.; Thakur, V.K.; Voicu, S.I. Crown ether-immobilized cellulose acetate membranes for the retention of Gd (III). Polymers 2021, 13, 3978. [Google Scholar] [CrossRef]
- Radu, E.R.; Voicu, S.I.; Thakur, V.K. Polymeric membranes for biomedical applications. Polymers 2023, 15, 619. [Google Scholar] [CrossRef]
- Pandele, A.M.; Oprea, M.; Dutu, A.A.; Miculescu, F.; Voicu, S.I. A novel generation of polysulfone/crown ether-functionalized reduced graphene oxide membranes with potential applications in hemodialysis. Polymers 2021, 14, 148. [Google Scholar] [CrossRef]
- Oprea, M.; Pandele, A.M.; Nicoara, A.I.; Nicolescu, A.; Deleanu, C.; Voicu, S.I. Crown ether-functionalized cellulose acetate membranes with potential applications in osseointegration. Int. J. Biol. Macromol. 2023, 230, 123162. [Google Scholar] [CrossRef]
- Serbanescu, O.; Pandele, A.; Miculescu, F.; Voicu, Ş.I. Synthesis and characterization of cellulose acetate membranes with self-indicating properties by changing the membrane surface color for separation of Gd(III). Coatings 2020, 10, 468. [Google Scholar] [CrossRef]
- Pandele, A.M.; Serbanescu, O.S.; Voicu, S.I. Polysulfone Composite membranes with carbonaceous structure. Synthesis and applications. Coatings 2020, 10, 609. [Google Scholar] [CrossRef]
- Ionita, M.; Crica, L.E.; Voicu, S.I.; Pandele, A.M.; Iovu, H. Fabrication of cellulose triacetate/graphene oxide porous membrane. Polym. Adv. Technol. 2016, 27, 350–357. [Google Scholar] [CrossRef]
- Ioniță, M.; Crică, L.E.; Voicu, S.I.; Dinescu, S.; Miculescu, F.; Costache, M.; Iovu, H. Synergistic effect of carbon nanotubes and graphene for high performance cellulose acetate membranes in biomedical applications. Carbohydr. Polym. 2018, 183, 50–61. [Google Scholar] [CrossRef]
- Voicu, S.I.; Pandele, M.; Vasile, E.; Rughinis, R.; Crica, L.; Pilan, L.; Ionita, M. The impact of sonication time through polysulfone-graphene oxide composite films properties. Dig. J. Nanomater. Biostruct. 2013, 8, 1389–1394. [Google Scholar]
- Pai, S.; Kini, M.S.; Selvaraj, R. A review on adsorptive removal of dyes from wastewater by hydroxyapatite nanocomposites. Environ. Sci. Pollut. Res. 2021, 28, 11835–11849. [Google Scholar] [CrossRef]
- Corobea, M.C.; Muhulet, O.; Miculescu, F.; Antoniac, I.V.; Vuluga, Z.; Florea, D.; Vuluga, D.M.; Butnaru, M.; Ivanov, D.; Voicu, S.I. Novel nanocomposite membranes from cellulose acetate and clay-silica nanowires. Polym. Adv. Technol. 2016, 27, 1586–1595. [Google Scholar] [CrossRef]
- Zhang, H.; Mu, Q.; Yu, X.; Zhou, K.; Chen, X.; Hao, H.; Li, Y. Preparation of Copper Ion Adsorbed Modified Montmorillonite/Cellulose Acetate Porous Composite Fiber Membrane by Centrifugal Spinning. Polymers 2022, 14, 5458. [Google Scholar] [CrossRef]
- Pérez-Silva, I.; Páez-Hernández, M.E.; Ibarra, I.S.; Camacho-Mendoza, R.L. Evaluation of the hybrid membrane of ZnO particles supported in cellulose acetate for the removal of lead. Membranes 2023, 13, 123. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, S.; Subbaiya, R.; Saravanan, M.; Ponraj, M.; Selvam, M.; Pugazhendhi, A. A critical review of advanced nanotechnology and hybrid membrane-based water recycling, reuse, and wastewater treatment processes. Chemosphere 2022, 289, 132867. [Google Scholar] [CrossRef] [PubMed]
- Couto, C.F.; Lange, L.C.; Amaral, M.C. Occurrence, fate and removal of pharmaceutically active compounds (PhACs) in water and wastewater treatment plants—A review. J. Water Process. Eng. 2019, 32, 100927. [Google Scholar] [CrossRef]
- Pandele, A.M.; Iovu, H.; Orbeci, C.; Tuncel, C.; Miculescu, F.; Nicolescu, A.; Deleanu, C.; Voicu, S.I. Surface modified cellulose acetate membranes for the reactive retention of tetracycline. Sep. Purif. Technol. 2020, 249, 117145. [Google Scholar] [CrossRef]
- Varga, B.; Meiczinger, M.; Jakab, M.; Somogyi, V. Design and optimization of laccase immobilization in cellulose acetate microfiltration membrane for micropollutant remediation. Catalysts 2023, 13, 222. [Google Scholar] [CrossRef]
- Raicopol, M.D.; Andronescu, C.; Voicu, S.I.; Vasile, E.; Pandele, A.M. Cellulose acetate/layered double hydroxide adsorptive membranes for efficient removal of pharmaceutical environmental contaminants. Carbohydr. Polym. 2019, 214, 204–212. [Google Scholar] [CrossRef]
- Muhulet, A.; Tuncel, C.; Miculescu, F.; Pandele, A.M.; Bobirica, C.; Orbeci, C.; Bobirica, L.; Palla-Papavlu, A.; Voicu, S.I. Synthesis and characterization of polysulfone-TiO2 decorated MWCNT composite membranes by sonochemical method. Appl. Phys. A 2020, 126, 233. [Google Scholar] [CrossRef]
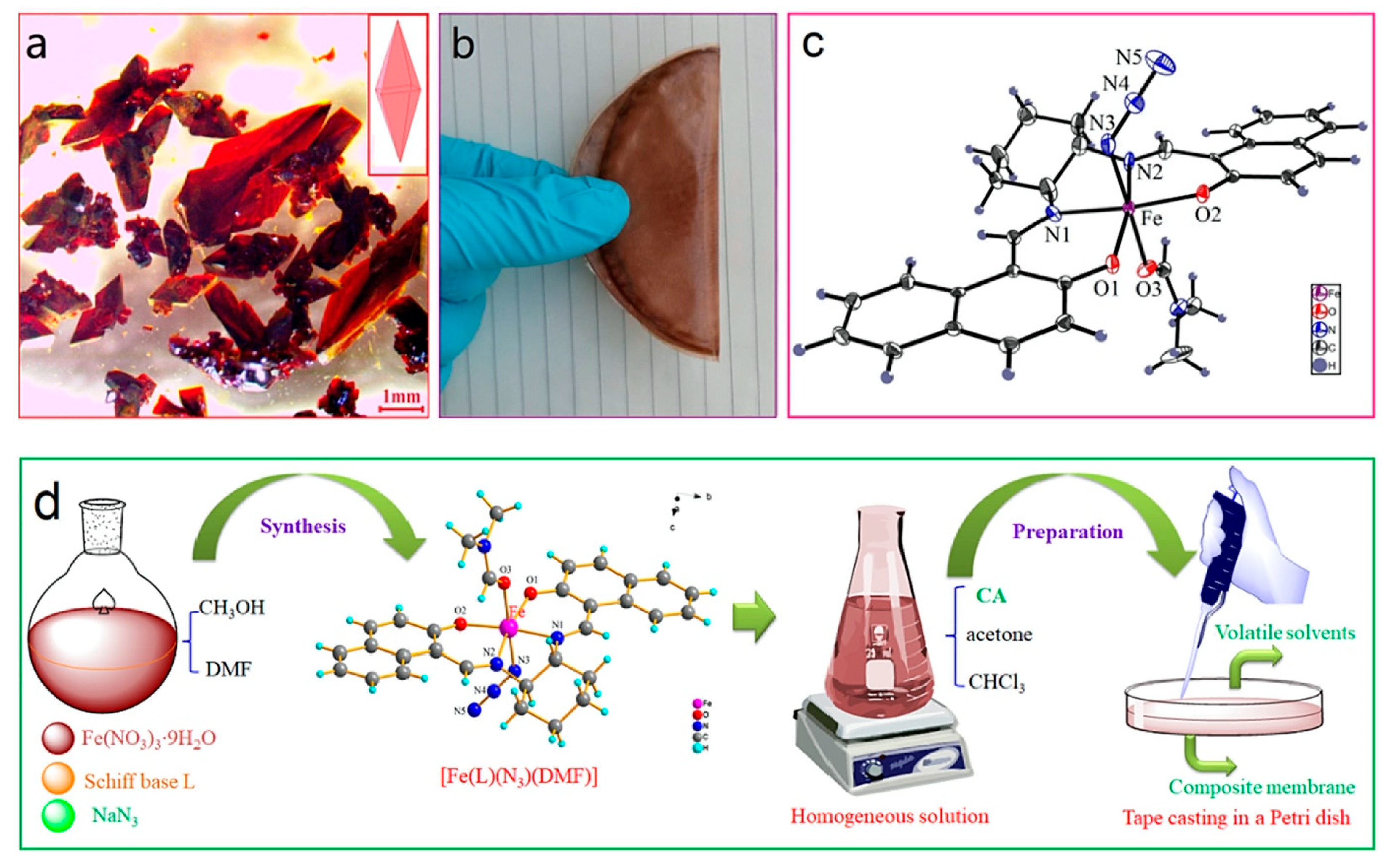
- Wang, D.; Yang, J.; Yang, H.; Zhao, P.; Shi, Z. Fe-complex modified cellulose acetate composite membrane with excellent photo-Fenton catalytic activity. Carbohydr. Polym. 2022, 296, 119960. [Google Scholar] [CrossRef]
- Mofokeng, L.E.; Hlekelele, L.; Moma, J.; Tetana, Z.N.; Chauke, V.P. Energy-efficient CuO/TiO2@GCn cellulose acetate-based membrane for concurrent filtration and photodegradation of ketoprofen in drinking and groundwater. Appl. Sci. 2022, 12, 1649. [Google Scholar] [CrossRef]
- Tavangar, T.; Jalali, K.; Alaei Shahmirzadi, M.A.; Karimi, M. Toward real textile wastewater treatment: Membrane fouling control and effective fractionation of dyes/inorganic salts using a hybrid electrocoagulation—Nanofiltration Process. Sep. Purif. Technol. 2019, 216, 115–125. [Google Scholar] [CrossRef]
- Liang, C.Z.; Sun, S.P.; Zhao, B.W.; Chung, T.S. Integration of Nanofiltration hollow fiber membranes with coagulation-flocculation to treat colored wastewater from a dyestuff manufacturer: A pilot-scale study. Ind. Eng. Chem. Res. 2015, 54, 11159–11166. [Google Scholar] [CrossRef]
- Voicu, S.I.; Thakur, V.K. Graphene-based composite membranes for nanofiltration: Performances and future perspectives. Emergent Mater. 2022, 5, 1429–1441. [Google Scholar] [CrossRef]
- Karadag, D.; Tok, S.; Akgul, E.; Ulucan, K.; Evden, H.; Kaya, M.A. Combining adsorption and coagulation for the treatment of azo and anthraquinone dyes from aqueous solution. Ind. Eng. Chem. Res. 2006, 45, 3969–3973. [Google Scholar] [CrossRef]
- Ong, M.D.; Vasquez, I.; Alvarez, B.; Cho, D.R.; Williams, M.B.; Vincent, D., Jr.; Ali, M.A.; Aich, N.; Pinto, A.H.; Choudhury, M.R. Modification of cellulose acetate microfiltration membranes using graphene oxide–polyethyleneimine for enhanced dye rejection. Membranes 2023, 13, 143. [Google Scholar] [CrossRef]
- Praveen, S.; Jegan, J.; Pushpa, T.B.; Gokulan, R.; Bulgariu, L. Biochar for removal of dyes in contaminated water: An overview. Biochar 2022, 4, 10. [Google Scholar] [CrossRef]
- Mcyotto, F.; Wei, Q.; Macharia, D.K.; Huang, M.; Shen, C.; Chow, C. Effect of dye structure on color removal efficiency by coagulation. Chem. Eng. J. 2021, 405, 126674. [Google Scholar] [CrossRef]
- Koriem, O.A.; Kamel, A.M.; Shaaban, W.; Elkady, M.F. Enhancement of dye separation performance of eco-friendly cellulose acetate-based membranes. Sustainability 2022, 14, 14665. [Google Scholar] [CrossRef]
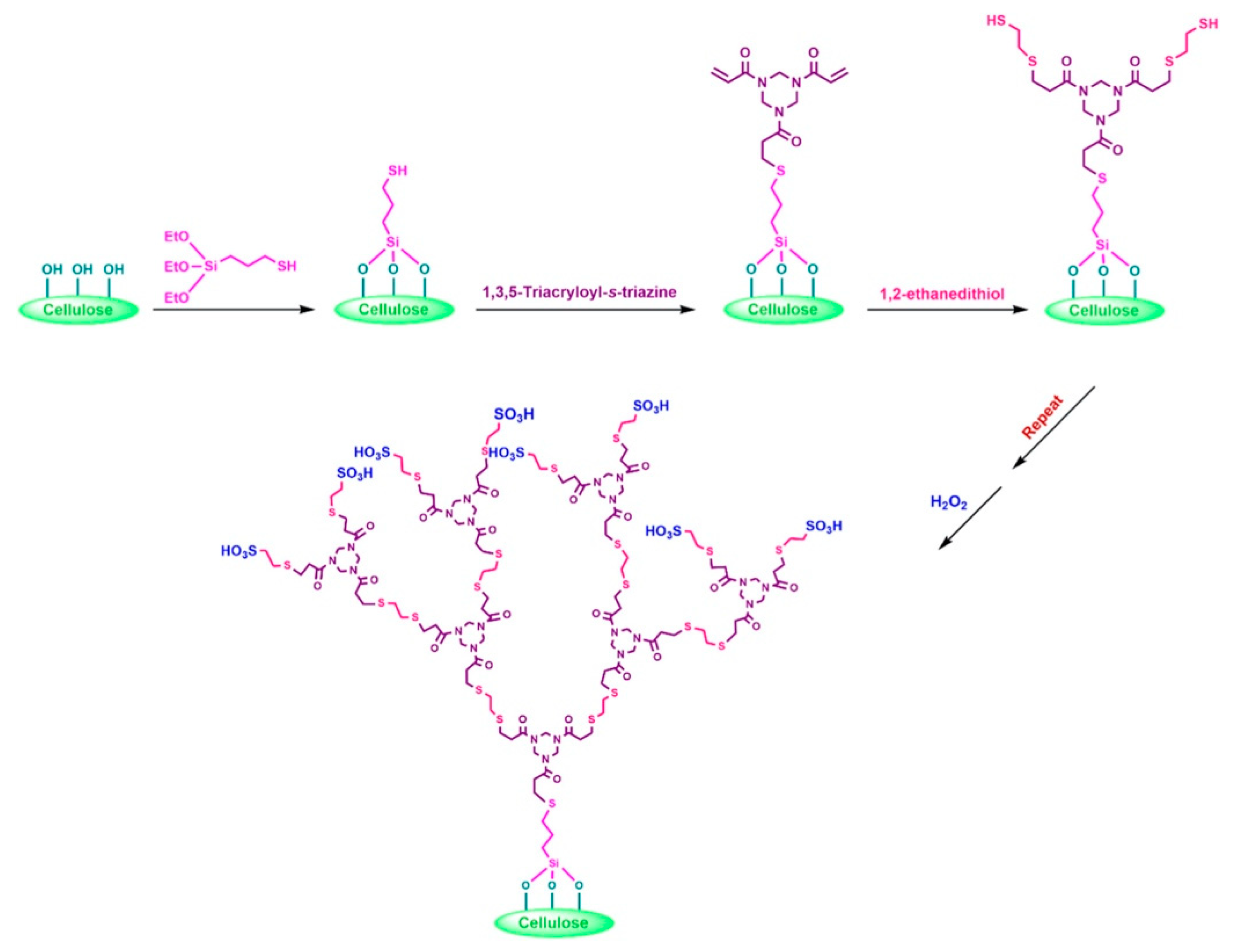
- Vatanpour, V.; Mehrabi, M.; Masteri-Farahani, M.; Behroozi, A.H.; Niakan, M.; Koyuncu, I. Sulfonic acid functionalized dendrimer-grafted cellulose as a charge and hydrophilic modifier of cellulose acetate membranes in removal of inorganic and organic pollutants. J. Water Process Eng. 2022, 50, 103307. [Google Scholar] [CrossRef]
- Abu-Dalo, M.A.; Al-Rosan, S.A.; Albiss, B.A. Photocatalytic Degradation of methylene blue using polymeric membranes based on cellulose acetate impregnated with ZnO nanostructures. Polymers 2021, 13, 3451. [Google Scholar] [CrossRef]
- Yu, D.; Li, L.; Wu, M.; Crittenden, J. Enhanced photocatalytic ozonation of organic pollutants using an iron-based metal-organic framework. Appl. Catal. B Environ. 2019, 251, 66–75. [Google Scholar] [CrossRef]
- Asiri, A.M.; Pugliese, V.; Petrosino, F.; Khan, S.B.; Alamry, K.A.; Alfifi, S.Y.; Marwani, H.M.; Alotaibi, M.M.; Mukherjee, D.; Chakraborty, S. Photocatalytic degradation of textile dye on blended cellulose acetate membranes. Polymers 2022, 14, 636. [Google Scholar] [CrossRef] [PubMed]
- Kumar Gupta, P.; Kumar Srivastava, A.; Hiremath, L.; Kumar, N.S.; Kumar Srivastava, A.; Narayan, A.V.; Rajendran, S.; Ravi, K.M.; Narasanagi, M. Functionalized cellulose nanofiber-composite membranes for wastewater treatment—A Review. J. Nanotechnol. Mater. Sci. 2018, 5, 35–43. [Google Scholar]
- Silva, M.A.; Belmonte-Reche, E.; de Amorim, M.T.P. Combining experimental data with statistical methods to evaluate hydrolyzed reactive dye removal by α-fe2o3 in a cellulose-based membrane. Fibers 2021, 9, 61. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oprea, M.; Voicu, S.I. Cellulose Acetate-Based Materials for Water Treatment in the Context of Circular Economy. Water 2023, 15, 1860. https://doi.org/10.3390/w15101860
Oprea M, Voicu SI. Cellulose Acetate-Based Materials for Water Treatment in the Context of Circular Economy. Water. 2023; 15(10):1860. https://doi.org/10.3390/w15101860
Chicago/Turabian StyleOprea, Madalina, and Stefan Ioan Voicu. 2023. "Cellulose Acetate-Based Materials for Water Treatment in the Context of Circular Economy" Water 15, no. 10: 1860. https://doi.org/10.3390/w15101860
APA StyleOprea, M., & Voicu, S. I. (2023). Cellulose Acetate-Based Materials for Water Treatment in the Context of Circular Economy. Water, 15(10), 1860. https://doi.org/10.3390/w15101860







