Influence of Salinity on Copper Toxicity in Paracentrotus lividus and Arbacia lixula Embryos
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Solutions
2.2. Chemical Analysis of Test Solutions
2.3. Sea Urchin Collection and Bioassays
2.4. Toxicity Criteria
2.5. Data Analysis
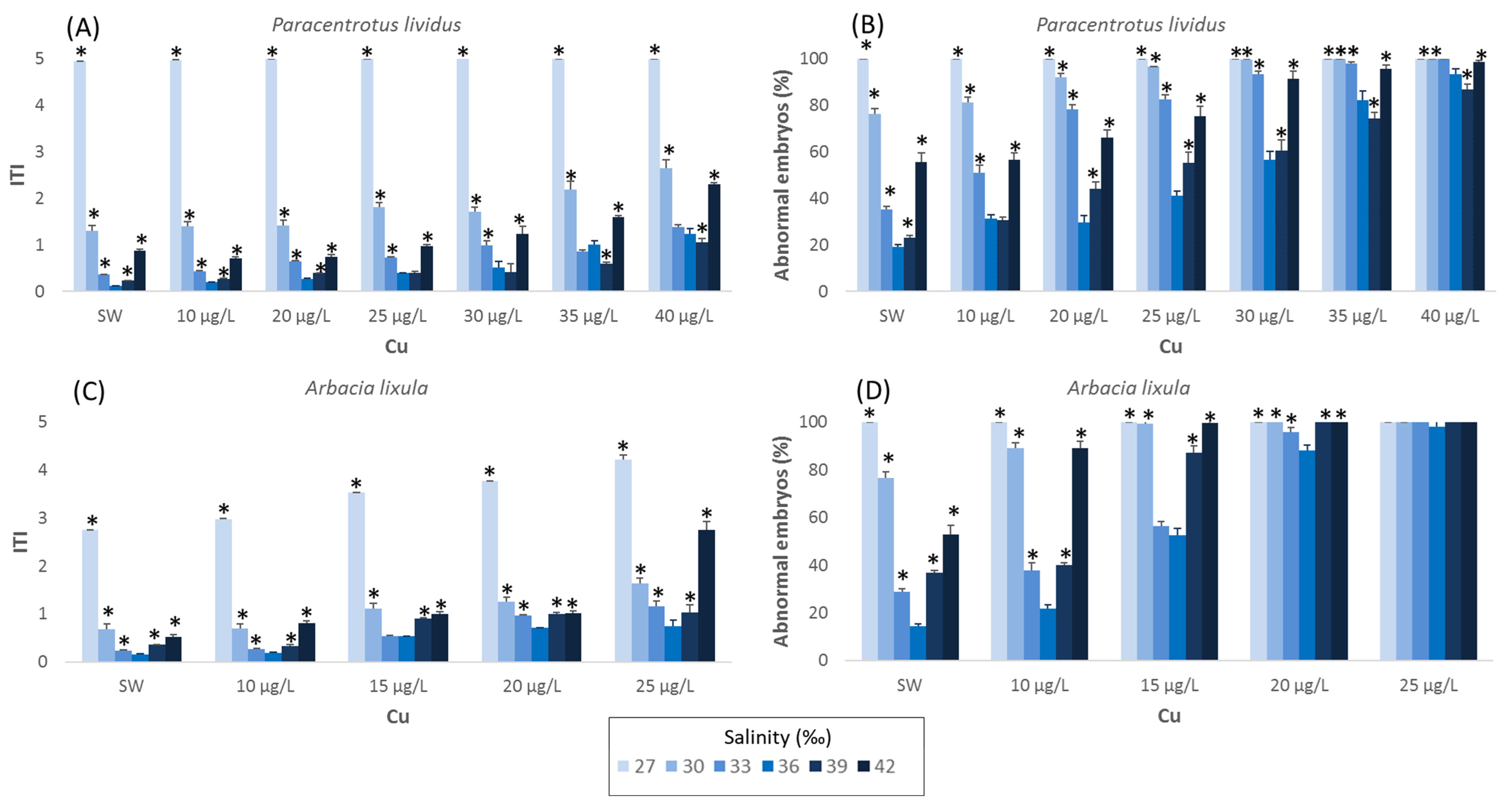
3. Results
3.1. Paracentrotus lividus
3.2. Arbacia lixula
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Boudoureque, C.F.; Verlaque, M. Paracentrotus lividus. In Developments in Aquaculture and Fisheries Science; Elsevier: Amsterdam, The Netherlands, 2020; pp. 447–485. [Google Scholar]
- Pagano, G.; Thomas, P.J.; Guida, M.; Palumbo, A.; Romano, G.; Trifuoggi, M.; Oral, R.; Trifuoggi, M. Sea urchin bioassays in toxicity testing: II. Sediment evaluation. Expert Opin. Environ. Biol. 2017, 6, 1. [Google Scholar] [CrossRef]
- Tuya, F.; Cisneros-Aguirre, J.; Ortega-Borges, L.; Haroun, R.J. Bathymetric segregation of sea urchins on reefs of the Canarian Archipelago: Role of flow-induced forces. Estuar. Coast. Shelf Sci. 2007, 73, 481–488. [Google Scholar] [CrossRef]
- Régis, M.B. Particularités microstructurales du squelette de Paracentrotus lividus et Arbacia lixula: Rapports avec l’écologie et l’éthologie de ces échinoïdes. Mar. Biol. 1979, 54, 373–382. [Google Scholar] [CrossRef]
- Lawrence, J.M. The effect of temperature-salinity combinations on the functional well- being of adult Lytechinus variegatus (Lamarck) (Echinodermata, Echinoidea). J. Exp. Mar. Biol. Ecol. 1975, 18, 271–275. [Google Scholar] [CrossRef]
- Trowbridge, C.D. Life at the edge: Population dynamics and salinity tolerance of a high intertidal, pool-dwelling ascoglossan opisthobranch on New Zealand rocky shores. J. Exp. Mar. Biol. Ecol. 1994, 182, 65–84. [Google Scholar] [CrossRef]
- Russell, M.P. Echinoderm responses to variation in salinity. Adv. Mar. Biol. 2013, 66, 171–212. [Google Scholar] [PubMed]
- Delorme, N.J.; Sewell, M.A. Temperature and salinity: Two climate change stressors affecting early development of the New Zealand sea urchin Evechinus chloroticus. Mar. Biol. 2014, 161, 1999–2009. [Google Scholar] [CrossRef]
- Bindoff, N.L.; Cheung, J.G.; Kairo, J.; Arístegui, V.A.; Guinder, R.; Hallberg, N.; Hilmi, N.; Jiao, M.S.; Karim, L.; Levin, S.; et al. Changing Ocean, Marine Ecosystems, and Dependent Communities. In IPCC Special Report on the Ocean and Cryosphere in a Changing Climate; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2019; pp. 447–587. [Google Scholar]
- IPCC (2014) Climate Change. Impact, Adaptation and vulnerability. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change, 1132. Available online: https://www.ipcc.ch/report/ar6/wg2/ (accessed on 16 November 2022).
- Allen, J.D.; Pechenik, J.A. Understanding the Effects of Low Salinity on Fertilization Success and Early Development in the Sand Dollar Echinarachnius parma. Biol. Bull. 2010, 218, 189–199. [Google Scholar] [CrossRef]
- Carballeira, C.; Martín-Díaza, L.; DelVallsa, T.A. Influence of salinity on fertilization and larval development toxicity tests with two species of sea urchin. Mar. Environ. Res. 2011, 72, 196–203. [Google Scholar] [CrossRef]
- Sewell, M.A.; Young, C.M. Temperature limits to fertilization and early development in the tropical sea urchin Echinometra lucunter. J. Exp. Mar. Biol. Ecol. 1999, 236, 291–305. [Google Scholar] [CrossRef]
- Li, L.; Li, Q.; Sun, X.; Kong, L. Effects of temperature and salinity on larval growth, survival, and development of the sea cucumber Apostichopus japonicus. N. Am. J. Aquacult. 2011, 73, 296–303. [Google Scholar] [CrossRef]
- Pia, T.S.; Johnson, T.; George, S.B. Salinity-induced morphological changes in Pisaster ochraceus (Echinodermata: Asteroidea) larvae. J. Plankton. Res. 2012, 34, 590–601. [Google Scholar] [CrossRef]
- Mak, K.K.Y.; Chan, K.Y.K. Interactive effects of temperature and salinity on early life stages of the sea urchin Heliocidaris crassispina. Mar. Bio. 2018, 165, 57. [Google Scholar] [CrossRef]
- Bressan, M.; Marin, M.; Brunetti, R. Influence of temperature and salinity on embryonic development of Paracentrotus lividus (Lmk, 1816). Hydrobiologia 1995, 304, 175–184. [Google Scholar] [CrossRef]
- His, E.; Seaman, M.N.L.; Beiras, R. A simplification the bivalve embryogenesis and larval development bioassay method for water quality assessment. Wat Res. 1997, 31, 351–355. [Google Scholar] [CrossRef]
- Volpi Ghirardini, A.V.; Arizzi Novelli, A.; Likar, B.; Pojana, G.; Ghetti, P.F.; Marcomini, A. Sperm cell toxicity test using sea Urchin Paracentrotus lividus Lamarck (Echinodermata: Echinoidea): Sensitivity and discriminatory ability toward anionic and nonionic surfactants. Environ. Toxicol. Chem. 2001, 20, 644–651. [Google Scholar] [CrossRef]
- Marín, M.G.; Da Ros, L.; Moschino, V.; Campesan, G. Sediment elutriate toxicity testing with embryos of sea urchin (Paracentrotus lividus). Aquat. Ecosyst. Health Manag. 2001, 4, 215–221. [Google Scholar] [CrossRef]
- Fernández, N. Evaluación Biológica de la Contaminación Marina Costera Mediante Bioensayos con Embriones del Erizo de mar Paracentrotus lividus; Universidad de Vigo: Vigo, Spain, 2002. [Google Scholar]
- Pétinay, S.; Chataigner, C.; Basuyaux, O. Standardisation du développement larvaire de l’oursin, Paracentrotus lividus, pour l’évaluation de la qualité d’une eau de mer. Comptes Rendus Biol. 2009, 332, 1104–1114. [Google Scholar] [CrossRef]
- Saco-Álvarez, L.; Durán, I.; Ignacio Lorenzo, J.; Beiras, R. Methodologicalbasis for the optimization of amarine sea-urchinembryo test (SET) for the ecological assessment of coastal water quality. Ecotox. Environ. Safe 2010, 73, 491–499. [Google Scholar] [CrossRef]
- Máximo, M.V.; Mottola, L.S.M.; Resgalla, C., Jr. Sensibilidade do ouriço Arbacia lixula (Echinodermata: Echinoidea) em testes de toxicidade. J. Braz. Soc. Ecotoxicol. 2008, 3, 47–52. [Google Scholar] [CrossRef]
- His, E.; Heyvang, I.; Geffard, O.; de Montaudouin, X. A comparison between oyster (Crassostrea gigas) and sea urchin (Paracentrotus lividus) larval bioassays for toxicological studies. Water Res. 1999, 33, 1706–1718. [Google Scholar] [CrossRef]
- Carballeira, C.; De Orte, M.R.; Viana, I.G.; Delvalls, T.A.; Carballeira, A. Assessing the toxicity of chemical compounds associated with land-based marine fish farms: The sea urchin embryo bioassay with Paracentrotus lividus and Arbacia lixula. Arch. Environ. Contam. Toxicol. 2012, 63, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Maisano, M.; Cappello, T.; Catanese, E.; Vitale, V.; Natalotto, A.; Giannetto, A.; Barreca, D.; Brunelli, E.; Mauceri, A.; Fasulo, S. Developmental abnormalities and neurotoxicological effects of CuO NPs on the black sea urchin Arbacia lixula by embryotoxicity assay. Mar. Environ. Res. 2015, 111, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Morroni, L.; Pinsino, A.; Pellegrini, D.; Regoli, F.; Matranga, V. Development of a new integrative toxicity index based on an improvement of the sea urchin embryo toxicity test. Ecotoxicol. Environ. Saf. 2016, 123, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Morroni, L.; Pinsino, A.; Pellegrini, D.; Regoli, F. Reversibility of metal induced malformations in sea urchin embryos. Ecotoxicol. Environ. Saf. 2018, 148, 923–929. [Google Scholar] [CrossRef]
- Gambardella, C.; Marcellini, F.; Falugi, C.; Varrella, S.; Corinaldesi, C. Early-stage anomalies in the sea urchin (Paracentrotus lividus) as bioindicators of multiple stressors in the marine environment: Overview and future perspectives. Environ. Pollut. 2021, 287, 117608. [Google Scholar] [CrossRef]
- Du Laing, G.; De Vos, R.; Vandecasteele, B.; Lesage, E.; Tack, F.M.; Verloo, M.G. Effect of salinity on heavy metal mobility and availability in intertidal sediments of the Scheldt estuary. Estuar. Coast. Shelf Sci. 2008, 77, 589–602. [Google Scholar] [CrossRef]
- Lewis, C.; Ellis, R.; Vernon, E.; Elliot, K.; Newbatt, S.; Wilson, R.W. Ocean acidification increases copper toxicity differentially in two key marine invertebrates with distinct acid-base responses. Sci. Rep. 2016, 6, 21554. [Google Scholar] [CrossRef]
- Grosell, M.; Blanchard, J.; Brix, K.V.; Gerdes, R. Physiology is pivotal for interactions between salinity and acute copper toxicity to fish and invertebrates. Aquatic. Toxicol. 2007, 84, 162–172. [Google Scholar] [CrossRef]
- Nielson, C.; Hird, C.; Lewis, C. Ocean acidification buffers the physiological responses of the king ragworm Alitta virens to the common pollutant copper. Aquat. Toxixol. 2019, 212, 120–127. [Google Scholar] [CrossRef]
- Morroni, L.; Sartori, D.; Costantini, M.; Genovesi, L.; Magliocco, T.; Ruocco, N.; Buttino, I. First molecular evidence of the toxicogenetic effects of copper on sea urchin Paracentrotus lividus embryo development. Water Res. 2019, 160, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Sartori, D.; Pellegrini, D.; Gaion, A. Analysis of variability in Embryological response of two sea urchin species to spatial and temporal features-can these factors influence responses in standardized Ecotoxicological assays? Expert Opin. Environ. Biol. 2016, S1, 2. [Google Scholar] [CrossRef]
- Fernández, N.; Beiras, R. Combined toxicity of dissolved mercury with copper, lead and cadmium on embryogenesis and early larval growth of the Paracentrotus lividus sea-urchin. Ecotoxicology 2001, 10, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Arizzi Novelli, A.; Losso, C.; Ghetti, P.F.; Ghirardini, A.V. Toxicity of heavy metals using sperm cell and embryo toxicity bioassays with Paracentrotus lividus (Echinodermata: Echinoidea): Comparisons with exposure concentrations in the lagoon of Venice, Italy. Environ. Toxicol. Chem. Int. J. 2003, 22, 1295–1301. [Google Scholar] [CrossRef]
- Manzo, S.; Buono, S.; Cresimini, C. Predictability of copper, Irgarol, and diuron combined effects on Sea Urchin Paracentrotus lividus. Arch. Environ. Contam. Toxicol. 2008, 54, 57–68. [Google Scholar] [CrossRef]
- Manzo, S.; Buono, S.; Cresimini, C. Cadmium, lead and their mixtures with copper: Paracentrotus lividus embryotoxicity assessment, prediction, and offspring quality evaluation. Ecotoxicology 2010, 19, 1209–1223. [Google Scholar] [CrossRef]
- Gharred, T.; Ezzine, I.K.; Naija, A.; Bouali, R.R.; Jebali, J. Assessment of toxic interactions between deltamethrin and copper on the fertility and developmental events in the Mediterranean sea urchin, Paracentrotus lividus. Environ. Monit. Assess. 2015, 187, 193. [Google Scholar] [CrossRef]
- Giannetto, A.; Cappello, T.; Oliva, S.; Parrino, V.; De Marco, G.; Fasulo, S.; Mauceri, A.; Maisano, M. Copper oxide nanoparticles induce the transcriptional modulation of oxidative stress-related genes in Arbacia lixula embryos. Aquatic. Toxicol. 2018, 201, 187–197. [Google Scholar] [CrossRef]
- Bonaventura, R.; Zito, F.; Morroni, L.; Pellegrini, D.; Regoli, F.; Pinsino, A. Development and validation of new analytical methods using sea urchin embryo bioassay to evaluate dredged marine sediments. J. Environ. Manag. 2021, 281, 111862. [Google Scholar] [CrossRef]
- Charles, C.; Veber, P.; Delignette-Muller, M.L. MOSAIC: A web-interface for statistical analyses in ecotoxicology. Environ. Sci. Pollut. Res. 2018, 25, 11295–11302. [Google Scholar] [CrossRef]
- Delignette-Muller, M.L.; Ruiz, P.; Charles, S.; Duchemin, W.; Lopes, C.; Kon-Kam-King, G.; Veber, P. Morse: Modelling Tools for Reproduction and Survival Data in Ecotoxicology. R Package Version 2.2.0. 2016. Available online: https://cran.r-project.org/web/packages/morse/index.html (accessed on 16 November 2022).
- Underwood, A.J. Experiments in Ecology. Their Logical Design and Interpretation Using Analysis of Variance; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Murado, M.A.; Prieto, M.A. NOEC and LOEC as merely concessive expedients: Two unambiguous alternatives and some criteria to maximize the efficiency of dose–response experimental designs. Sci. Total Environ. 2013, 461, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Hammer, O.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Morroni, L.; Rakaj, A.; Grosso, L.; Fianchini, A.; Pellegrini, D.; Regoli, F. Sea cucumber Holothuria polii (Delle Chiaje, 1823) as new model for embryo bioassays in ecotoxicological studies. Chemosphere 2020, 240, 124819. [Google Scholar] [CrossRef] [PubMed]
- Rakaj, A.; Morroni, L.; Grosso, L.; Fianchini, A.; Pensa, D.; Pellegrini, D.; Regoli, F. Towards sea cucumbers as a new model in embryo-larval bioassays: Holothuria tubulosa as test species for the assessment of marine pollution. Sci. Total Environ. 2021, 787, 147593. [Google Scholar] [CrossRef]
- Carreras, C.; García-Cisneros, A.; Wangensteen, O.S.; Ordóñez, V.; Palacín, C.; Pascual, M.; Turon, X. East is East and West is West: Population genomics and hierarchical analyses reveal genetic structure and adaptation footprints in the keystone species Paracentrotus lividus (Echinoidea). Divers. Distrib. 2020, 26, 382–398. [Google Scholar] [CrossRef]
- Sarifudin, M.; Aminur Rahman, M.; Yu, S.H.; Yusoff, F.M.; Arshad, A. Effects of Salinity on Embryonic and Early Larval Development of a Tropical Sea Urchin, Salmacis sphaeroides. Iran. J. Fish. Sci. 2014, 13, 456–468. [Google Scholar]
- Sarifudin, M.; Aminur Rahman, M.; Yusoff, F.M.; Arshad, A.; Tans, S.G. Influence of salinity variations on the embryonic and early larval development of long-spined black sea urchin (Diadema setosum). J. Anim. Plant Sci. 2017, 27, 316–324. [Google Scholar]
- Shamim Parvez, M.D.; Aminur Rahman, M.; Yusoff, F.M.; Arshad, A.; Lee, S.-G. Salinity effects on the development of embryos and larvae of a high-valued sea urchin, Tripneustes gratilla (Linnaeus, 1758). J. Environ. Biol. 2018, 39, 785–794. [Google Scholar] [CrossRef]
- Greenwood, P.J.; Bennett, T. Some effects of temperature salinity combinations on the early development of the sea urchin Parechinus angulosus (leske). Fertilization. J. Exp. Mar. Biol. 1981, 51, 119–131. [Google Scholar] [CrossRef]
- Metaxas, A. The effect of salinity on larval survival and development in the sea urchin Echinometra lucunter. Invertebr. Reprod. Dev. 1998, 34, 323–330. [Google Scholar] [CrossRef]
- Dinnel, P.A.; Link, J.M.; Stober, Q.J.; Letourneau, M.W.; Roberts, W.E. Comparative sensitivity of sea urchin sperm bioassays to metals and pesticides. Arch. Environ. Contam. Toxicol. 1989, 18, 748–755. [Google Scholar] [CrossRef] [PubMed]
- Carr, S.R.; Chapman, D.C.; Long, E.R.; Windom, H.L.; Thursby, G.; Sloane, G.M.; Wolfe, D.A. Sediment quality assessment studies of Tampa Bay, Florida. Environ. Toxicol. Chem. 1996, 15, 1218–1231. [Google Scholar] [CrossRef]
- Warnau, M.; Temara, A.; Jangoux, M.; Dubois, P.; Iaccarino, M.; De Biase, A.; Pagano, G. Spermiotoxicity and embryotoxicity of heavy metals in the echinoid Paracentrotus lividus. Environ. Toxicol. Chem. 1996, 15, 1931–1936. [Google Scholar] [CrossRef]
- Philips, B.M.; Anderson, B.S.; Hunt, J.W. Spatial and temporal variation in results of purple urchin (Strongylocentrotus purpuratus) toxicity tests with zinc. Environ. Toxicol. Chem. 1998, 17, 453–459. [Google Scholar] [CrossRef]
- Radenac, G.; Fichet, D.; Miramand, P. Bioaccumulation and toxicity of four dissolved metals in Paracentrotus lividus sea-urchin embryo. Mar. Environ. Res. 2001, 51, 151–166. [Google Scholar] [CrossRef] [PubMed]
- Gaion, A.; Scuderi, A.; Pellegrini, D.; Sartori, D. Arsenic exposure affects embryo development of sea urchin, Paracentrotus lividus (Lamarck, 1816). Bull. Environ. Contam. Toxicol. 2013, 91, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Sartori, D.; Scuderi, A.; Sansone, G.; Gaion, A. Echinoculture: The rearing of Paracentrotus lividus in a recirculating aquaculture system. Experiments of artificial diets for the maintenance of sexual maturation. Aquacult. Int. 2015, 23, 111–125. [Google Scholar] [CrossRef]
- Florence, T.M. Trace element speciation and aquatic toxicology. Trends Anal. Chem. 1983, 2, 162–166. [Google Scholar] [CrossRef]
- Blust, R.; Fontaine, A.; Decleir, W. Effect of hydrogen ions and inorganic complexing on the uptake of copper by the brine shrimp Artemia franciscana. Mar. Ecol. Prog. Ser. 1991, 76, 273–282. [Google Scholar] [CrossRef]
- Vercauteren, K.; Blust, R. Bioavailability of dissolved zinc to the common mussel Mytilus edulis in complexing environments. Mar. Ecol. Prog. Ser. 1996, 137, 123–132. [Google Scholar] [CrossRef]
- Lewis, A.G.; Whitfield, P.; Ramnarine, A. Some particulate and soluble agents affecting the relationship between metal toxicity and organism survival in the calanoid copepod Euchaeta japonica. Mar. Biol. 1972, 17, 215–221. [Google Scholar] [CrossRef]
- Lewis, A.G.; Whitfield, P.; Ramnarine, A. The reduction of copper toxicity in a marine copepod by sediment extract. Limnol. Oceanogr. 1973, 18, 324–326. [Google Scholar] [CrossRef]
- Davey, E.W.; Morgan, M.J.; Erickson, S.J. A biological measurement of the copper complexation capacity of sea water. Limnol. Oceanogr. 1973, 18, 993–997. [Google Scholar] [CrossRef]
- Morris, O.P.; Russel, G. Effect ofchelation on the toxicity of copper. Mar. Pollut. Bull. 1973, 4, 159–160. [Google Scholar] [CrossRef]
- Turner, D.R.; Whitfield, M.; Dickson, A.G. The equilibrium speciation of dissolved components in freshwater and seawater at 25 °C and 1 atm pressure. Geochim. Cosmochim. Acta 1981, 45, 855–881. [Google Scholar] [CrossRef]
- Newman, M.C.; Jagoe, C.H. Ligands and the bioavailability of metals in aquatic environments. In Bioavailability. Physical, Chemical and Biological Interactions; Hamelink, S.L., Landrum, P.F., Bergman, H.L., Benson, W.H., Eds.; CRC Press: New York, NY, USA, 1994; pp. 39–61. [Google Scholar]
- Coglianese, M.P. The effects of salinity on copper and silver toxicity to embryos of the Pacific oyster. Arch. Environ. Contam. Toxicol. 1983, 11, 297. [Google Scholar] [CrossRef]
- Hall, L.W.; Anderson, R.D. The Influence of Salinity on the Toxicity of Various Classes of Chemicals to Aquatic Biota. Crit. Rev. Toxicol. 1995, 2, 281–346. [Google Scholar] [CrossRef]
- Riba, I.; DelValls, T.A.; Forja, J.M.; Gómez-Parra, A. The influence of ph and salinity on the toxicity of heavy metals in sediment to the estuarine clam Ruditapes philippinarum. Environ. Toxicol. Chem. 2004, 23, 1100–1107. [Google Scholar] [CrossRef]
- Martins, C.D.M.G.; Barcarolli, I.F.; de Menezes, E.J.; Giacomin, M.M.; Wood, C.M.; Bianchini, A. Acute toxicity, accumulation and tissue distribution of copper in the blue crab Callinectes sapidus acclimated to different salinities: In vivo and in vitro studies. Aquat. Toxicol. 2011, 101, 88–99. [Google Scholar] [CrossRef]
- Park, J.; Kim, S.; Yoo, J.; Lee, J.S.; Park, J.W.; Jung, J. Effect of salinity on acute copper and zinc toxicity to Tigriopus japonicus: The difference between metal ions and nanoparticles. Mar. Pollut. Bull. 2014, 85, 526–531. [Google Scholar] [CrossRef]
- Piazza, V.; Gambardella, C.; Canepa, S.; Costa, E.; Faimali, M.; Garaventa, F. Temperature and salinity effects on cadmium toxicity on lethal and sublethal responses of Amphibalanus amphitrite nauplii. Ecotox. Environ. Saf. 2016, 123, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.; Figueira, E.; Libralato, G.; Soares, A.M.V.M.; Guida, M.; Freitas, R. Comparative sensitivity of Crassostrea angulata and Crassostrea gigas embryo-larval development to As under varying salinity and temperature. Mar. Environ. Res. 2018, 140, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Thurberg, F.P.; Dawson, M.A.; Collier, R.S. Effects of copper and cadmium on osmoregulation and oxygen consumption in two species of estuarine crabs. Mar. Biol. 1973, 23, 171–175. [Google Scholar] [CrossRef]
- MacInnes, J.R.; Calabrese, A. Combined effects of salinity, temperature and copper on embryos and early larvae of the American oyster, Crassostrea virginica. Arch. Environ. Contam. Toxicol. 1979, 8, 553. [Google Scholar] [CrossRef]
- Webber, R.E.; Swaan, A.; Bang, A. Interactive effects of ambient copper and anoxia, temperature and salinity stress on survival and hemolymph and muscle tissue osmotic effectors in Mytilus edulis. J. Exp. Mar. Biol. Ecol. 1992, 159, 135. [Google Scholar] [CrossRef]
- Mcleese, D.W. Toxicity of copper at 2 temperatures and 3 salinities to American lobster (Homarus americanus). J. Fish. Res. Bd. Can. 1974, 31, 1949–1952. [Google Scholar] [CrossRef]
- Reardon, I.S.; Harrell, R.M. Acute toxicity of formalin and coppersulfate to striped bass fingerlings held in varying salinities. Aquaculture 1990, 87, 255–270. [Google Scholar] [CrossRef]
- Fernandez, T.V.; Jones, N.V. Studies on the toxicity of zinc and copper applied singly and jointly to Neries diversicolor at different salinities and temperatures. Trop. Ecol. 1990, 31, 47–55. [Google Scholar]
- Jones, L.H.; Jones, N.V.; Radlett, A.J. Some effects of salinity on toxicity of copper to polychaete Nereis diversicolor. Estuar. Coast. Mar. Sci. 1976, 4, 107–111. [Google Scholar] [CrossRef]
- Olson, K.R.; Harrel, R.C. Effect of salinity on acute toxicity of mercury, copper, and chromium for Rangia cuneata (Pelecypoda, Mactridae). Contrib. Mar. Sci. 1973, 17, 9–13. [Google Scholar]
- Birdsong, C.L.; Avault, J.W. Toxicity of certain chemicals to juvenile pompano. Prog. Fish Cult. 1971, 33, 76. [Google Scholar] [CrossRef]
- Anderson, B.S.; Hunt, J.W.; Piekarski, W.J.; Phillips, B.M.; Englund, M.A.; Tjeerdema, R.S.; Goetzl, J.D. Influence of salinity on copper and azide toxicity to larval topsmelt Atherinops affinis (Ayres). Arch. Environ. Contam. Physiol. 1995, 29, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Deruytter, D.; Vandegehuchte, M.B.; Garrevoet, J.; De Laender, F.; Vergucht, E.; Delbeke, K.; Blust, R.; De Schamphelaere, K.A.; Vincze, L.; Janssen, C.R. Salinity and dissolved organic carbon both affect copper toxicity in mussel larvae: Copper speciation or competition cannot explain everything. Environ. Toxicol. Chem. 2015, 34, 1330–1336. [Google Scholar] [CrossRef] [PubMed]
- Forbes, V.E. Response of Hydrobia ventrosa (Montagu) to environmental stress: Effects of salinity fluctuations and cadmium exposure on growth. Funct. Ecol. 1991, 5, 642–648. [Google Scholar] [CrossRef]
- Knezovich, J.P. Chemical and biological factors affecting bioavailability of contaminants in seawater. In Bioavailability: Physical, Chemical and Biological Interactions; Hamelink, J.L., Landrum, P.F., Bergman, H.L., Benson, W.H., Eds.; Lewis Publishers: London, UK, 1994; pp. 23–30. [Google Scholar]
- Blanchard, J.; Grosell, M. Effects of salinity on copper accumulation in the common killifish (Fundulus heteroclitus). Environ. Toxicol. Chem. 2005, 24, 1403–1413. [Google Scholar] [CrossRef]
| Paracentrotus lividus | Arbacia lixula | |||||||
|---|---|---|---|---|---|---|---|---|
| Source of Variation | df | MS | p | df | MS | p | ||
| Salinity | 6 | 34957 | 0.001 | 6 | 17131 | 0.001 | ||
| [Cu] | 4 | 21670 | 0.001 | 4 | 31848 | 0.001 | ||
| Salinity × Cu | 20 | 1734.3 | 0.001 | 20 | 3303.4 | 0.001 | ||
| Res | 333 | 5.7033 | 333 | 67.257 | ||||
| Total | 363 | 363 | ||||||
| Salinity (‰) | EC50 (µg/L) | |
|---|---|---|
| Paracentrotus lividus | Arbacia lixula | |
| 27 | n.c. | n.c. |
| 30 | 10.7 (8.3–12.9) | 9.82 (9.31–10.3) |
| 33 | 17.6 (16.3–18.9) | 15.8 (15.6–16.1) |
| 36 | 28.8 (28.4–29.3) | 15.5 (15.1–15.8) |
| 39 | 24.7 (23.6–25.7) | 13.6 (13.3–13.9) |
| 42 | 21.7 (19.3–23.8) | 9.0 (8.6–9.3) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morroni, L.; Gaion, A.; Broccoli, A.; Ferrari, S.; Pellegrini, D.; Sartori, D. Influence of Salinity on Copper Toxicity in Paracentrotus lividus and Arbacia lixula Embryos. Water 2023, 15, 65. https://doi.org/10.3390/w15010065
Morroni L, Gaion A, Broccoli A, Ferrari S, Pellegrini D, Sartori D. Influence of Salinity on Copper Toxicity in Paracentrotus lividus and Arbacia lixula Embryos. Water. 2023; 15(1):65. https://doi.org/10.3390/w15010065
Chicago/Turabian StyleMorroni, Lorenzo, Andrea Gaion, Andrea Broccoli, Stefano Ferrari, David Pellegrini, and Davide Sartori. 2023. "Influence of Salinity on Copper Toxicity in Paracentrotus lividus and Arbacia lixula Embryos" Water 15, no. 1: 65. https://doi.org/10.3390/w15010065
APA StyleMorroni, L., Gaion, A., Broccoli, A., Ferrari, S., Pellegrini, D., & Sartori, D. (2023). Influence of Salinity on Copper Toxicity in Paracentrotus lividus and Arbacia lixula Embryos. Water, 15(1), 65. https://doi.org/10.3390/w15010065







