Removal of Clostridium perfringens and Staphylococcus spp. in Microalgal–Bacterial Systems: Influence of Microalgal Inoculum and CO2/O2 Addition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Algal-Bacterial Inoculum
2.2. Gas and Domestic Wastewater
2.3. Experimental Setup and Sampling Procedure
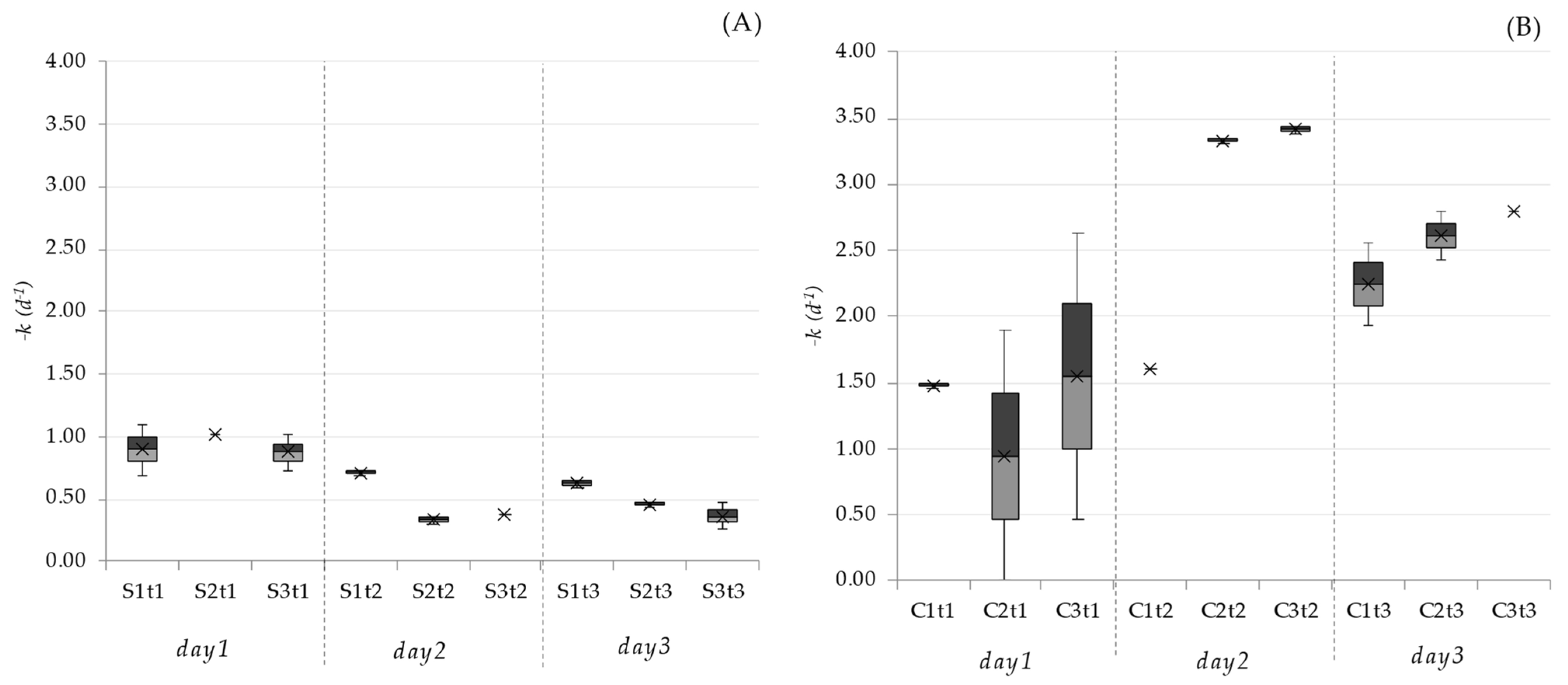
2.4. Inactivation Rates
2.5. Analytical Procedures
2.6. Statistical Analysis
3. Results and Discussion
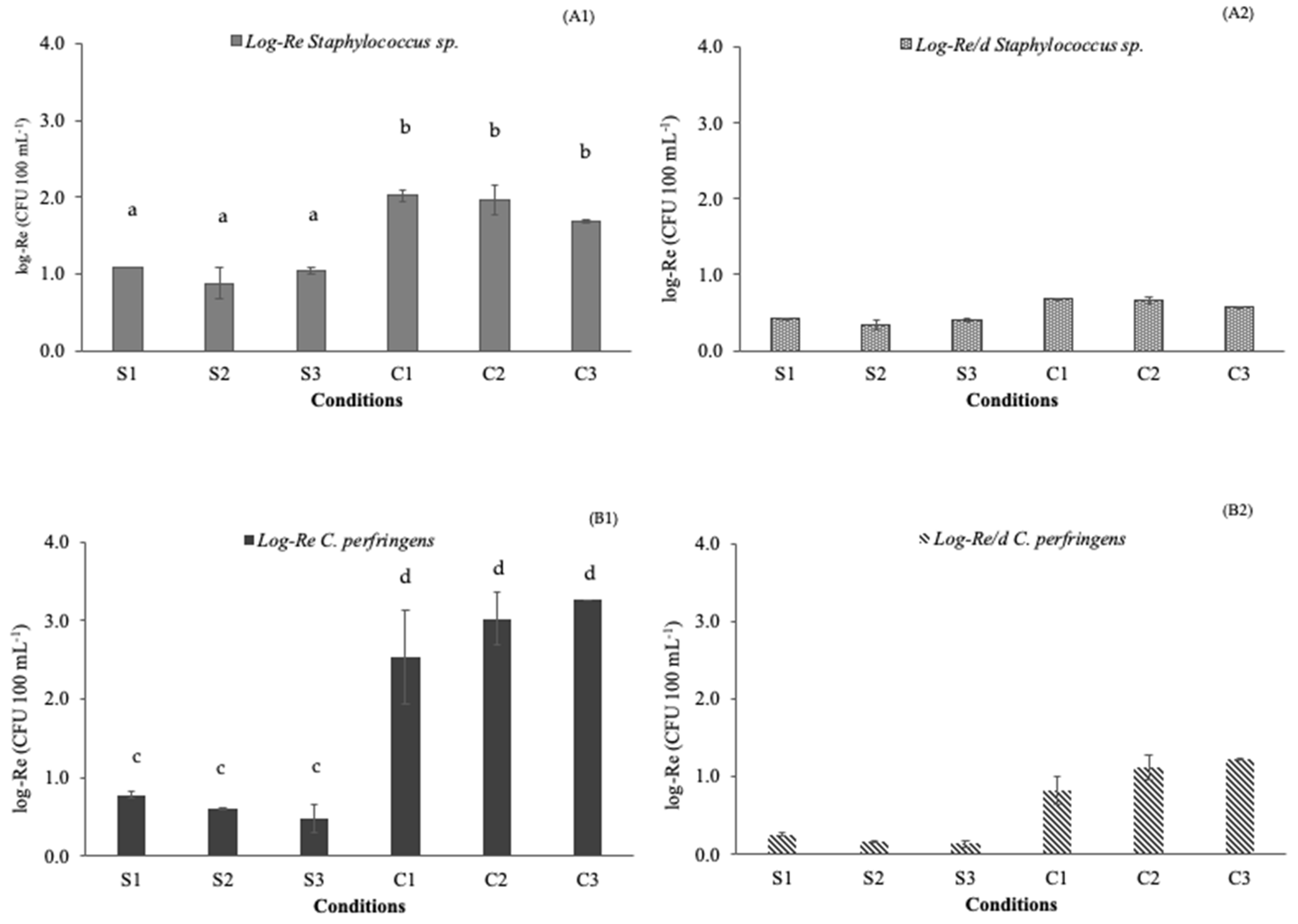
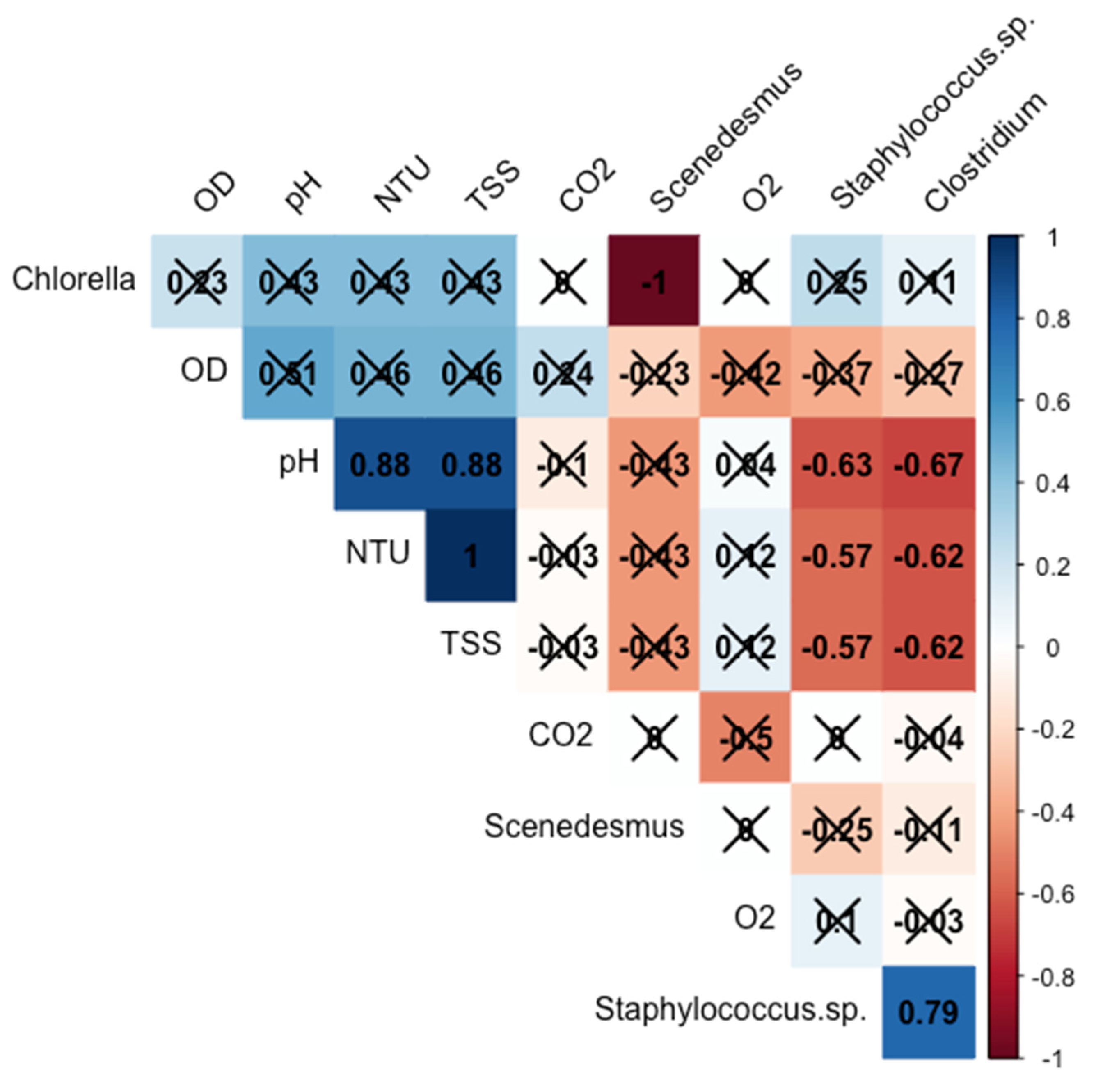
3.1. Pathogen Removal Efficiencies
3.2. COD and N-NH4+ Removal Efficiency, Productivity, and Specific Growth Rate (µ)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Guidelines on Sanitation and Health; WHO: Geneva, Switzerland, 2018; ISBN 9789241514705. Available online: https://apps.who.int/iris/bitstream/handle/10665/274939/9789241514705-eng.pdf (accessed on 17 November 2022).
- UNICEF and WHO. Progress on Household Drinking Water, Sanitation and Hygiene 2000–2017: Special Focus on Inequalities. 2019; ISBN 978-92-806-5036-5. Available online: https://washdata.org/sites/default/files/documents/reports/2019-07/jmp-2019-wash-households.pdf (accessed on 17 November 2022).
- Sharafi, K.; Moradi, M.; Karami, A.; Khosravi, T. Comparison of the Efficiency of Extended Aeration Activated Sludge System and Stabilization Ponds in Real Scale in the Removal of Protozoan Cysts and Parasite Ova from Domestic Wastewater Using Bailenger Method: A Case Study, Kermanshah, Iran. Desalin. Water Treat. 2015, 55, 1135–1141. [Google Scholar] [CrossRef]
- Nirmalakhandan, N.; Selvaratnam, T.; Henkanatte-Gedera, S.M.; Tchinda, D.; Abeysiriwardana-Arachchige, I.S.A.; Delanka-Pedige, H.M.K.; Munasinghe-Arachchige, S.P.; Zhang, Y.; Holguin, F.O.; Lammers, P.J. Algal Wastewater Treatment: Photoautotrophic vs. Mixotrophic Processes. Algal Res. 2019, 41, 101569. [Google Scholar] [CrossRef]
- Posadas, E.; del Mar Morales, M.; Gomez, C.; Acién, F.G.; Muñoz, R. Influence of PH and CO2 Source on the Performance of Microalgae-Based Secondary Domestic Wastewater Treatment in Outdoors Pilot Raceways. Chem. Eng. J. 2015, 265, 239–248. [Google Scholar] [CrossRef] [Green Version]
- Ruas, G.; Serejo, M.L.; Paulo, P.L.; Boncz, M.Á. Evaluation of Domestic Wastewater Treatment Using Microalgal-Bacterial Processes: Effect of CO2 Addition on Pathogen Removal. J. Appl. Phycol. 2018, 30, 921–929. [Google Scholar] [CrossRef]
- Ruas, G.; Farias, S.L.; Scarcelli, P.G.; Serejo, M.L.; Boncz, M.A. The Effect of CO2 Addition and Hydraulic Retention Time on Pathogens Removal in HRAPs. Water Sci. Technol. 2020, 1–9. [Google Scholar] [CrossRef]
- Venczel, L.V.; Arrowood, M.; Hurd, M.; Sobsey, M. Inactation of Cryptosporidium Parvum Oocyts and Clostridium Perfringens Spores by a Mixed-Oxidant Disinfectant and by Free Chlorine. Appl. Environ. Microbiol. 1997, 63, 1598–1601. [Google Scholar] [CrossRef] [Green Version]
- Lanao, M.; Ormad, M.P.; Goñi, P.; Miguel, N.; Mosteo, R.; Ovelleiro, J.L. Inactivation of Clostridium Perfringens Spores and Vegetative Cells by Photolysis and TiO2 Photocatalysis with H2O2. Sol. Energy 2010, 84, 703–709. [Google Scholar] [CrossRef]
- Abreu-Acosta, N.; Vera, L. Occurrence and Removal of Parasites, Enteric Bacteria and Faecal Contamination Indicators in Wastewater Natural Reclamation Systems in Tenerife-Canary Islands, Spain. Ecol. Eng. 2011, 37, 496–503. [Google Scholar] [CrossRef]
- Al-Tameemi, F.A.H.; Kadhim, T.A. Significance of Algae in Eliminating and Deactivating Pathological Index Arganisms in Wastewater Stabilization Pond Systems. E3S Web Conf. 2019, 96, 4–10. [Google Scholar] [CrossRef] [Green Version]
- Payment, P.; Franco, E. Clostridium Perfringens and Somatic Coliphages as Indicators of the Efficiency of Drinking Water Treatment for Viruses and Protozoan Cysts. Appl. Environ. Microbiol. 1993, 59, 2418–2424. [Google Scholar] [CrossRef]
- Bitton, G. Wastewater Microbiology, 3rd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; Volume 53, ISBN 978-0-471-71791-1. [Google Scholar]
- WHO. Evaluating Household Water Treatment Options: Health-Based Targets and Microbiological Performance Specifications; WHO: Geneva, Switzerland, 2011; Available online: https://apps.who.int/iris/bitstream/handle/10665/44693/9789241548229_eng.pdf (accessed on 17 November 2022).
- European Commisssion Council. Directive 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption; 1998. Available online: https://agris.fao.org/agris-search/search.do?recordID=CS2005000008 (accessed on 17 November 2022).
- Shoults, D.C.; Ashbolt, N.J. Decreased Efficacy of UV Inactivation of Staphylococcus Aureus after Multiple Exposure and Growth Cycles. Int. J. Hyg. Environ. Health 2019, 222, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Bassetti, M.; Nicco, E.; Mikulska, M. Why Is Community-Associated MRSA Spreading across the World and How Will It Change Clinical Practice? Int. J. Antimicrob. Agents 2009, 34, S15–S19. [Google Scholar] [CrossRef] [PubMed]
- Duckworth, G. Revised Guidelines for the Control of Epidemic Methicillin-Resistant Staphylococcus Aureus. J. Hosp. Infect. 1990, 16, 351–377. [Google Scholar] [CrossRef]
- Börjesson, S.; Matussek, A.; Melin, S.; Löfgren, S.; Lindgren, P.E. Methicillin-Resistant Staphylococcus Aureus (MRSA) in Municipal Wastewater: An Uncharted Threat? J. Appl. Microbiol. 2010, 108, 1244–1251. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg Goldstein, R.E.; Micallef, S.A.; Gibbs, S.G.; Davis, J.A.; He, X.; George, A.; Kleinfelter, L.M.; Schreiber, N.A.; Mukherjee, S.; Sapkota, A. Methicillin-Resistant Staphylococcus Aureus (MRSA) Detected at Four, U.S. Wastewater Treatment Plants. Environ. Health Perspect. 2012, 120, 1551–1558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García, M.; Soto, F.; González, J.M.; Bécares, E. A Comparison of Bacterial Removal Efficiencies in Constructed Wetlands and Algae-Based Systems. Ecol. Eng. 2008, 32, 238–243. [Google Scholar] [CrossRef]
- Sutherland, D.L.; Howard-Williams, C.; Turnbull, M.H.; Broady, P.A.; Craggs, R.J. The Effects of CO2 Addition along a PH Gradient on Wastewater Microalgal Photo-Physiology, Biomass Production and Nutrient Removal. Water Res. 2014, 70C, 9–26. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, L.; Geeraerd, A.H.; Spilimbergo, S.; Elst, K.; Van Ginneken, L.; Debevere, J.; Van Impe, J.F.; Devlieghere, F. High Pressure Carbon Dioxide Inactivation of Microorganisms in Foods: The Past, the Present and the Future. Int. J. Food Microbiol. 2007, 117, 1–28. [Google Scholar] [CrossRef]
- Serejo, M.L.; Farias, S.L.; Ruas, G.; Paulo, P.L.; Boncz, M.A. Surfactant Removal and Biomass Production in a Microalgal-Bacterial Process: Effect of Feeding Regime. Water Sci. Technol. 2020, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Hall, G.; Champagne, P. The Role of Algae in the Removal and Inactivation of Pathogenic Indicator Organisms in Wastewater Stabilization Pond Systems. Algal Res. 2020, 46, 101777. [Google Scholar] [CrossRef]
- Colwell, R.R.; Grimes, D.J. Nonculturable Microorganisms in the Environment; Colweel, R.R., Grimes, D.J., Eds.; American Society for Microbiology: Washington, DC, USA, 2000; ISBN 9781475702736. [Google Scholar]
- APHA. Standard Methods for the Examination of Water, 22nd ed.; American Public Health Association, American Water Works Association, Water Environment Federation: New York, NY, USA, 2012; ISBN 9780875530130. [Google Scholar]
- Sournia, A. Phytoplankton Manual; UNESCO, Ed.; Muséum National d’Histoire Naturelle: Paris, France, 1978; ISBN 92-3-101572-9. [Google Scholar]
- Zaiontz, C. The Data Analysis for This Paper Was Generated Using the Real Statistics Resource Pack Software (Release 7.6). 2020. [Google Scholar]
- R Core Team R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org (accessed on 17 November 2022).
- Davies-Colley, R.J.; Craggs, R.J.; Park, J.; Sukias, J.P.S.; Nagels, J.W.; Stott, R. Virus Removal in a Pilot-Scale “advanced” Pond System as Indicated by Somatic and F-RNA Bacteriophages. Water Sci. Technol. 2005, 51, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Maghembe, R.; Damian, D.; Makaranga, A.; Nyandoro, S.S.; Lyantagaye, S.L.; Kusari, S.; Hatti-Kaul, R. Omics for Bioprospecting and Drug Discovery from Bacteria and Microalgae. Antibiotics 2020, 9, 229. [Google Scholar] [CrossRef] [PubMed]
- Mezrioui, N.; Oudra, B.; Oufdou, K.; Hassani, L.; Loudiki, M.; Darley, J. Effect of Microalgae Growing on Wastewater Batch Culture on Escherichia Coli and Vibrio Cholerae Survival. Water Sci. Technol. 1994, 30, 295–302. [Google Scholar] [CrossRef]
- Oufdou, K.; Mezrioui, N.; Oudra, B.; Loudiki, M.; Barakate, M.; Sbiyyâa, B. Bioactive Compounds from Pseudanabaena Species (Cyanobacteria). Microbios 2001, 106, 21–29. [Google Scholar]
- Krustok, I.; Truu, J.; Odlare, M.; Truu, M.; Ligi, T.; Tiirik, K.; Nehrenheim, E. Effect of Lake Water on Algal Biomass and Microbial Community Structure in Municipal Wastewater-Based Lab-Scale Photobioreactors. Appl. Microbiol. Biotechnol. 2015, 99, 6537–6549. [Google Scholar] [CrossRef]
- Lucena, F.; Duran, A.E.; Morón, A.; Calderón, E.; Campos, C.; Gantzer, C.; Skraber, S.; Jofre, J. Reduction of Bacterial Indicators and Bacteriophages Infecting Faecal Bacteria in Primary and Secondary Wastewater Treatments. J. Appl. Microbiol. 2004, 97, 1069–1076. [Google Scholar] [CrossRef]
- del Rosario Rodero, M.; Posadas, E.; Toledo-Cervantes, A.; Lebrero, R.; Muñoz, R. Influence of Alkalinity and Temperature on Photosynthetic Biogas Upgrading Efficiency in High Rate Algal Ponds. Algal Res. 2018, 33, 284–290. [Google Scholar] [CrossRef]
- Mosteo, R.; Ormad, M.P.; Goñi, P.; Rodríguez-Chueca, J.; García, A.; Clavel, A. Identification of Pathogen Bacteria and Protozoa in Treated Urban Wastewaters Discharged in the Ebro River (Spain): Water Reuse Possibilities. Water Sci. Technol. 2013, 68, 575–583. [Google Scholar] [CrossRef]
- Scheurer, M.; Heß, S.; Lüddeke, F.; Sacher, F.; Güde, H.; Löffler, H.; Gallert, C. Removal of Micropollutants, Facultative Pathogenic and Antibiotic Resistant Bacteria in a Full-Scale Retention Soil Filter Receiving Combined Sewer Overflow. Environ. Sci. Process. Impacts 2015, 17, 186–196. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, R.E.; Hirsch, C.F.; Sesin, D.F.; Flor, J.E.; Chartrain, M.; Fromtling, R.E.; Harris, G.H.; Salvatore, M.J.; Liesch, J.M.; Yudin, K. Pharmaceuticals from Cultured Algae. J. Ind. Microbiol. 1990, 5, 113–123. [Google Scholar] [CrossRef]
- Sánchez-Saavedra, M.D.P.; Licea-Navarro, A.; Bernáldez-Sarabia, J. Evaluation of the Antibacterial Activity of Different Species of Phytoplankton. Rev. Biol. Mar. Oceanogr. 2010, 45, 531–536. [Google Scholar] [CrossRef]
- Syed, S.; Arasu, A.; Ponnuswamy, I. The Uses of Chlorella Vulgaris as Antimicrobial Agent and as a Diet: The Presence of Bio-Active Compounds Which Caters the Vitamins, Minerals in General. Int. J. Bio-Sci. Bio-Technol. 2015, 7, 185–190. [Google Scholar] [CrossRef]
- Evangelista, V.; Barsanti, L.; Coltelli, P.; Frassanito, A.M.; Passarelli, V.; Vesentini, N.; Gualtieri, P. Algal Toxins: Nature, Occurrence, Effect and Detection; Springer Science & Business Media: Pisa, Italy, 2007; ISBN 978-1-4020-8480-5. [Google Scholar]
- Al-Gheethi, A.A.; Mohamed, R.M.; Jais, N.M.; Efaq, A.N.; Halid, A.A.; Wurochekke, A.A.; Amir-Hashim, M.K. Influence of Pathogenic Bacterial Activity on Growth of Scenedesmus Sp. and Removal of Nutrients from Public Market Wastewater. J. Water Health 2017, 15, 741–756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Curtis, T.P.; Mara, D.D.; Silva, S.A. The Effect of Sunlight on Faecal Coliforms in Ponds: Implications for Research and Design. Water Sci. Technol. 1992, 26, 1729–1738. [Google Scholar] [CrossRef]
- Bermejo, R.; Talavera, E.M.; Alvarez-Pez, J.M.; Orte, J.C. Chromatographic Purification of Biliproteins from Spirulina Platensis High-Performance Liquid Chromatographic Separation of Their α and β Subunits. J. Chromatogr. A 1997, 778, 441–450. [Google Scholar] [CrossRef]
- Nurachman, Z.; Hartini, H.; Rahmaniyah, W.R.; Kurnia, D.; Hidayat, R.; Prijamboedi, B.; Suendo, V.; Ratnaningsih, E.; Panggabean, L.M.G.; Nurbaiti, S. Tropical Marine Chlorella Sp. PP1 as a Source of Photosynthetic Pigments for Dye-Sensitized Solar Cells. Algal Res. 2015, 10, 25–32. [Google Scholar] [CrossRef]
- Sinton, L.W.; Davies-Colley, R.J.; Bell, R.G. Inactivation of Enterococci and Fecal Coliforms from Sewage and Meatworks Effluents in Seawater Chambers. Appl. Environ. Microbiol. 1994, 60, 2040–2048. [Google Scholar] [CrossRef] [Green Version]
- Sharmila, J.; Saravanan, P.; Sivasankar, S.; Chamundeeswari, M. A Novel and an Eco-Friendly Approach for Organic Dyes Degradation Using Spirulina Platensis Cultivated Water. Catal. Today 2018, 340, 245–252. [Google Scholar] [CrossRef]
- Maraccini, P.A.; Wenk, J.; Boehm, A.B. Photoinactivation of Eight Health-Relevant Bacterial Species: Determining the Importance of the Exogenous Indirect Mechanism. Environ. Sci. Technol. 2016, 50, 5050–5059. [Google Scholar] [CrossRef] [Green Version]
- Cheah, W.Y.; Show, P.L.; Chang, J.; Ling, T.C.; Juan, J.C. Biosequestration of Atmospheric CO2 and Flue Gas-Containing CO2 by Microalgae. Bioresour. Technol. 2015, 184, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Msanne, J.; Polle, J.; Starkenburg, S. An Assessment of Heterotrophy and Mixotrophy in Scenedesmus and Its Utilization in Wastewater Treatment. Algal Res. 2020, 48, 101911. [Google Scholar] [CrossRef]
- Ji, M.K.; Yun, H.S.; Park, Y.T.; Kabra, A.N.; Oh, I.H.; Choi, J. Mixotrophic Cultivation of a Microalga Scenedesmus Obliquus in Municipal Wastewater Supplemented with Food Wastewater and Flue Gas CO2 for Biomass Production. J. Environ. Manag. 2015, 159, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, R.; Guieysse, B. Algal-Bacterial Processes for the Treatment of Hazardous Contaminants: A Review. Water Res. 2006, 40, 2799–2815. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Min, M.; Li, Y.; Chen, P.; Chen, Y.; Liu, Y.; Wang, Y.; Ruan, R. Cultivation of Green Algae Chlorella Sp. in Different Wastewaters from Municipal Wastewater Treatment Plant. Appl. Biochem. Biotechnol. 2010, 162, 1174–1186. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, W.; Hu, B.; Min, M.; Chen, P.; Ruan, R.R. Integration of Algae Cultivation as Biodiesel Production Feedstock with Municipal Wastewater Treatment: Strains Screening and Significance Evaluation of Environmental Factors. Bioresour. Technol. 2011, 102, 10861–10867. [Google Scholar] [CrossRef]


| Set | I | II | ||||
|---|---|---|---|---|---|---|
| Conditions | S1 | S2 | S3 | C1 | C2 | C3 |
| Scenedesmus Acutus Meyen | + | + | + | - | - | - |
| Chlorella vulgaris | - | - | - | + | + | + |
| CO2 | - | + | - | - | + | - |
| O2 | - | - | + | - | - | + |
| Parameters | Unit | Set I | Set II |
|---|---|---|---|
| Ambient temperature | °C | 30 ± 1 | 30 ± 1 |
| Light Intensity | µmol m−2 s−1 | 406 ± 19 | 446 ± 32 |
| pH | - | 7.87 ± 0.92 | 8.68 ± 1.02 |
| DO | mg O2 L−1 | 5.42 ± 2.42 | 7.42 ± 4.59 |
| Set | I | II | ||||
|---|---|---|---|---|---|---|
| Condition | S1 | S2 | S3 | C1 | C2 | C3 |
| C. perfringens day−1 | 0.60 | 0.46 | 0.35 | 1.79 | 2.24 | 2.47 |
| Staphylococcus spp. day−1 | 0.84 | 0.66 | 0.81 | 1.53 | 1.47 | 1.27 |
| Set | I | II | ||||
|---|---|---|---|---|---|---|
| Condition | S1 | S2 | S3 | C1 | C2 | C3 |
| COD-Re (%) | 57.64 ± 0.78 a,b | 79.00 ± 7.81 c | 38.12 ± 1.56 c,d,e,f | 82.47 ± 8.55 c,d | 94.96 ± 5.04 a,e | 92.97 ± 3.05 b,f |
| N-NH4+-Re (%) | 62.82 ± 9.5 | 98.64 ± 1.35 | 99.41 ± 0.51 | 81.66 ± 18.3 | 86.66 ± 13.3 | 99.36 ± 0.63 |
| pH (mean) | 8.02 ± 1.00 | 7.67 ± 1.11 | 7.91 ± 0.94 | 8.80 ± 1.12 | 8.53 ± 1.16 | 8.71 ± 1.17 |
| pH final | 9.22 ± 0.02 | 9.27 ± 0.01 | 9.20 ± 0.01 | 9.49 ± 0.05 | 9.73 ± 0.04 | 9.59 ± 0.07 |
| DO (mean) (mg O2 L−1) | 5.81 ± 3.36 | 5.90 ± 2.70 | 4.54 ± 1.47 | 9.48 ± 4.95 | 8.88 ± 5.08 | 3.89 ± 1.49 |
| DO final (mg O2 L−1) | 5.15 ± 0.07 | 6.15 ± 0.49 | 4.95 ± 0.07 | 8.98 ± 0.83 | 7.04 ± 2.57 | 5.43 ± 0.64 |
| Productivity (mg TSS L−1 d−1) | 114.6 ± 4.2 a,b,c,d | 153.0 ± 9.8 e | 201.7 ± 21.6 a,f | 216.0 ± 5.6 b,g | 273 ± 15.2 c,e,h | 384 ± 14.6 d,e,f,g,h |
| µ (d−1) | 0.30 | 0.40 | 0.49 | 0.49 | 0.56 | 0.68 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruas, G.; Farias, S.L.; dos Reis, B.A.B.; Serejo, M.L.; da Silva, G.H.R.; Boncz, M.Á. Removal of Clostridium perfringens and Staphylococcus spp. in Microalgal–Bacterial Systems: Influence of Microalgal Inoculum and CO2/O2 Addition. Water 2023, 15, 5. https://doi.org/10.3390/w15010005
Ruas G, Farias SL, dos Reis BAB, Serejo ML, da Silva GHR, Boncz MÁ. Removal of Clostridium perfringens and Staphylococcus spp. in Microalgal–Bacterial Systems: Influence of Microalgal Inoculum and CO2/O2 Addition. Water. 2023; 15(1):5. https://doi.org/10.3390/w15010005
Chicago/Turabian StyleRuas, Graziele, Sarah Lacerda Farias, Bruno A. B. dos Reis, Mayara Leite Serejo, Gustavo Henrique Ribeiro da Silva, and Marc Árpád Boncz. 2023. "Removal of Clostridium perfringens and Staphylococcus spp. in Microalgal–Bacterial Systems: Influence of Microalgal Inoculum and CO2/O2 Addition" Water 15, no. 1: 5. https://doi.org/10.3390/w15010005
APA StyleRuas, G., Farias, S. L., dos Reis, B. A. B., Serejo, M. L., da Silva, G. H. R., & Boncz, M. Á. (2023). Removal of Clostridium perfringens and Staphylococcus spp. in Microalgal–Bacterial Systems: Influence of Microalgal Inoculum and CO2/O2 Addition. Water, 15(1), 5. https://doi.org/10.3390/w15010005








