A Preliminary Approximation to Microbiological Beach Sand Quality along the Coast of the Department of Atlántico (Caribbean Sea of Colombia): Influence of the Magdalena River
Abstract
:1. Introduction
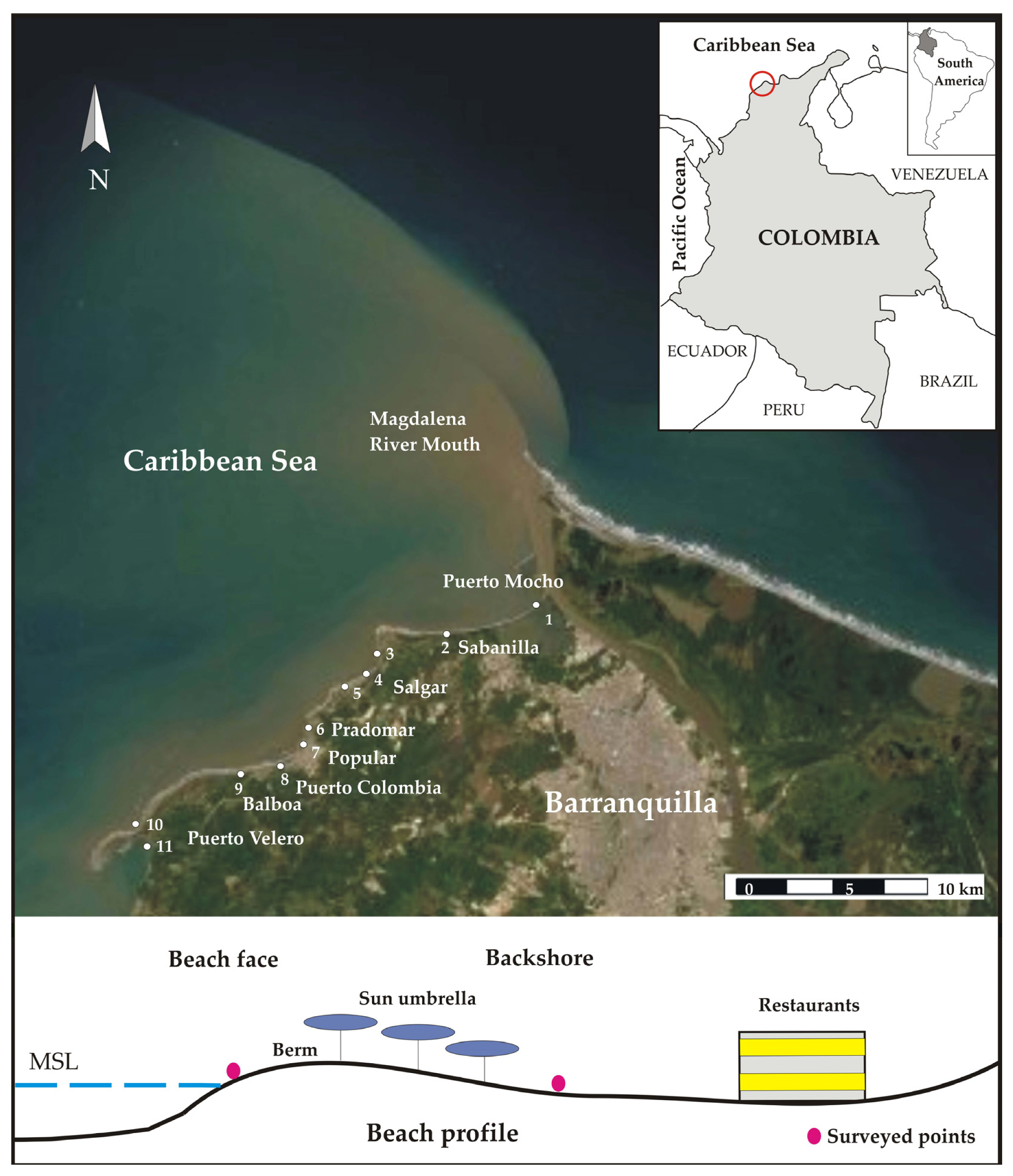
2. Study Area
3. Materials and Methods
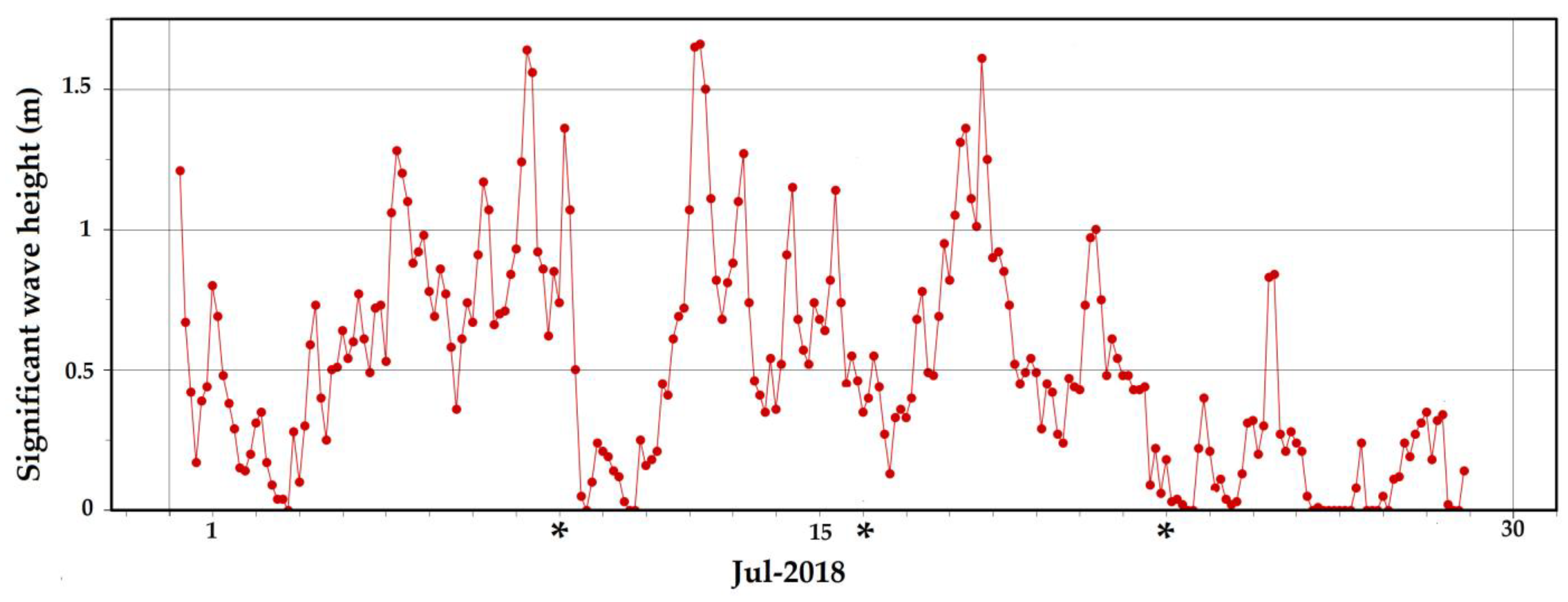
3.1. Marine Climate Characterization
3.2. Sand Sampling
3.3. Microbial Analysis
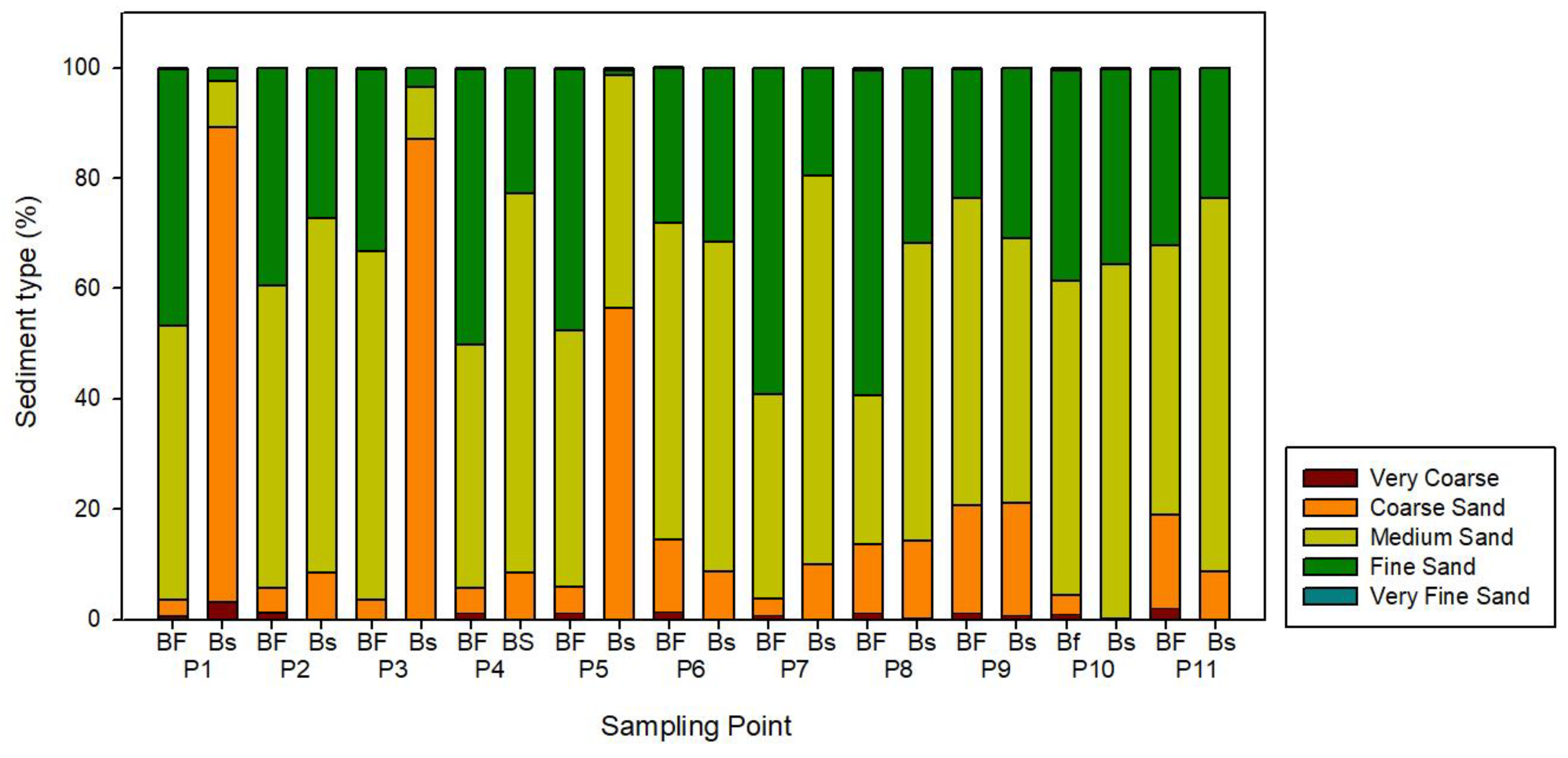
3.4. Granulometric Analysis
3.5. Statistical Data Analysis
4. Results
4.1. Marine Climate Characterization
4.2. Beach Characterization
4.3. Sedimentological Characteristics
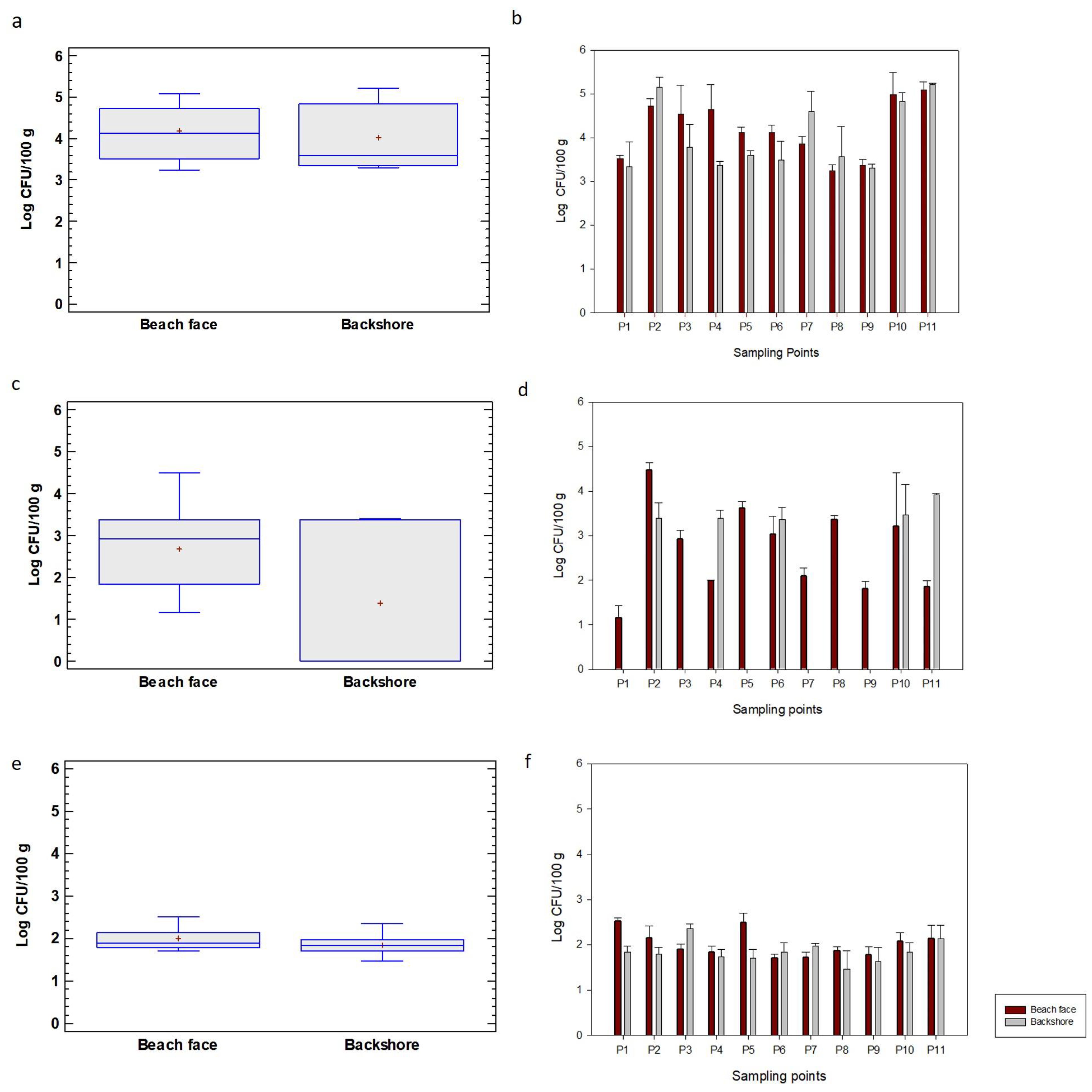
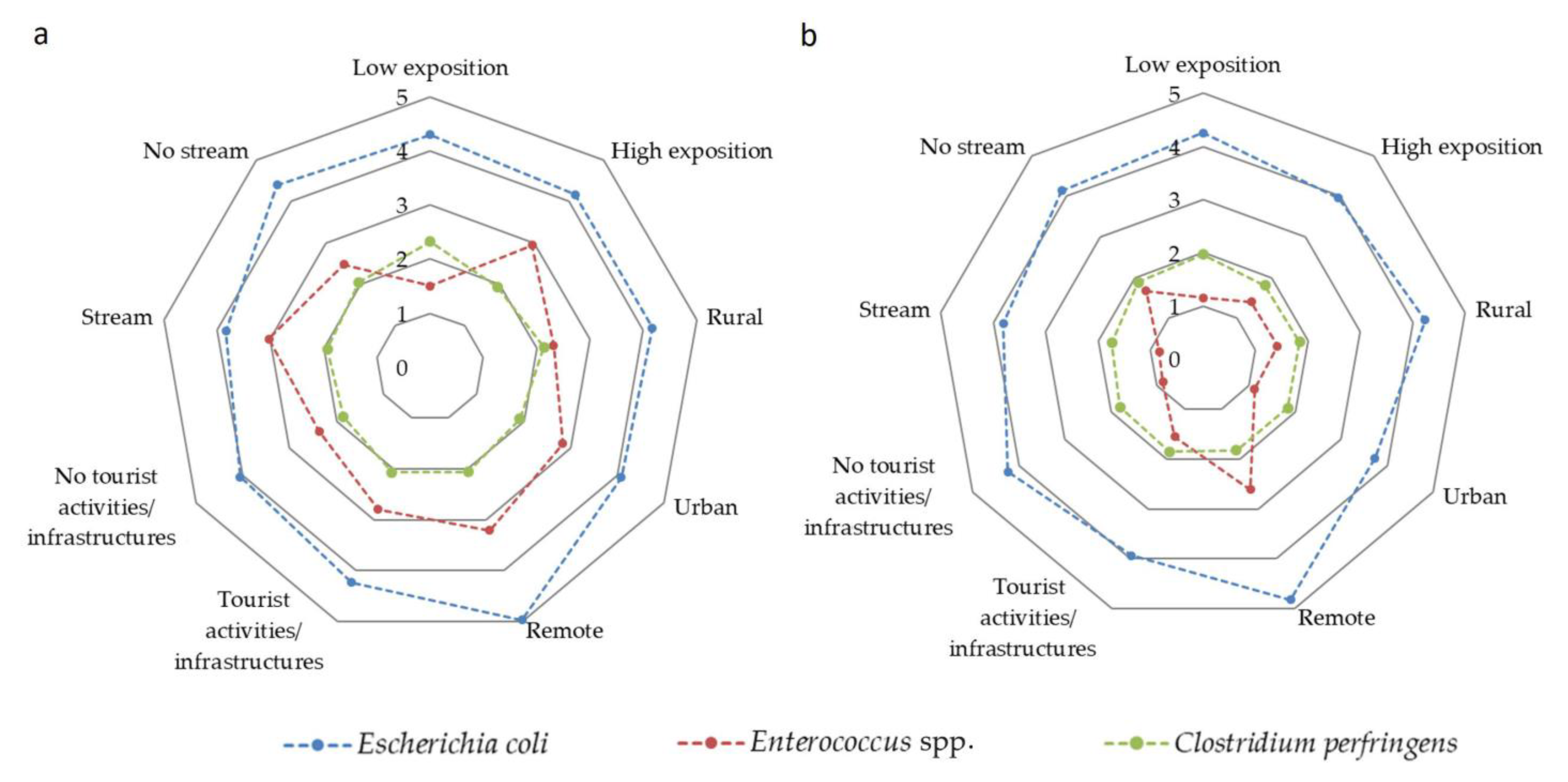
4.4. Distribution of Microbiological Indicators
4.5. Microbiological Indicators versus Beach and Sedimentological Characteristics
5. Discussion
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Palazzo, M.; Gigauri, I.; Panait, M.C.; Apostu, S.A.; Siano, A. Sustainable tourism issues in European countries during the global pandemic crisis. Sustainability 2022, 14, 3844. [Google Scholar] [CrossRef]
- Bunghez, C.L. The importance of tourism to a destination’s economy. J. East. Eur. Res. Bus. Econ. 2016, 2016, 240–247. [Google Scholar] [CrossRef]
- Mendoza-González, G.; Martínez, M.L.; Guevara, R.; Pérez-Maqueo, O.; Garza-Lagler, M.C.; Howard, A. Towards a sustainable sun, sea, and sand tourism: The value of ocean view and proximity to the coast. Sustainability 2018, 10, 1012. [Google Scholar] [CrossRef] [Green Version]
- Mestanza-Ramón, C.; Pranzini, E.; Anfuso, G.; Botero, C.M.; Chica-Ruiz, J.A.; Mooser, A. An Attempt to characterize the "3S" (sea, sun, and sand) parameters: Application to the Galapagos islands and continental Ecuadorian beaches. Sustainability 2020, 12, 3468. [Google Scholar] [CrossRef] [Green Version]
- World Tourism Organization UNWTO 2021: A Year in Review. Available online: https://www.unwto.org/news/tourism-grows-4-in-2021-but-remains-far-below-pre-pandemic-levels (accessed on 10 October 2022).
- Sibanda, T.; Ramganesh, S. Taxonomic and functional analyses reveal existence of virulence and antibiotic resistance genes in beach sand bacterial populations. Arch. Microbiol. 2021, 203, 1753–1766. [Google Scholar] [CrossRef]
- Anfuso, G.; Bolivar-Anillo, H.J.; Sanchez, H.; Villate Daza, D.A.; Lopez Daza, O.L. Coastal Tourism Importance and Beach Users’ Preferences: The "Big Fives" Criterions and Related Management Aspects. J. Tour. Hosp. 2018, 7, 7–8. [Google Scholar] [CrossRef]
- Asensio-Montesino, F.; Molina-Gil, R.; Bolívar-Anillo, H.; Botero, C.-M.; Pranzini, E.; Anfuso, G. Las preferencias de los usuarios en la elección de una playa: Los “Big Five” y su alcance en la gestión costera. In Estudios de Caso Sobre Manejo Integrado de Zonas Costeras en Iberoamérica: Gestión, Riesgo y Buenas Prácticas; Milanes, C., Lastra, R., Sierra-Correa, P., Eds.; Corporación Universidad de la Costa: Barranquilla, Colombia, 2019; pp. 380–406. ISBN 9789588921914. [Google Scholar]
- Williams, A. Definitions and typologies of coastal tourism beach destinations. In Disappearing Destinations: Climate Change and Future Challenges for Coastal Tourism; Jones, A., Phillips, M., Eds.; CABI: Chippenham, United Kingdom, 2011; pp. 47–66. [Google Scholar]
- Sánchez, M.M. Colombia in post-conflict: Tourism for peace or peace for tourism? Araucaria 2018, 20, 415–438. [Google Scholar] [CrossRef]
- World Tourism Organization UNWTO 2021: Tourism doing business investing in Colombia. Available online: https://www.unwto.org/investment/tourism-doing-business-investing-in-colombia (accessed on 10 October 2022).
- Aguilera-Díaz, M.M.; Reina-Aranza, Y.C.; Orozco-Gallo, A.J.; Yabrudy-Vega, J.; Barcos-Robles, R. Composición de la economía de la Región Caribe de Colombia. Ensayos Sobre Econ. Reg. 2013, 53, 1–66. [Google Scholar]
- Sánchez Moreno, H.; Bolívar-Anillo, H.J.; Soto-Varela, Z.E.; Aranguren, Y.; Gonzaléz, C.P.; Villate Daza, D.A.; Anfuso, G. Microbiological water quality and sources of contamination along the coast of the Department of Atlántico (Caribbean Sea of Colombia). Preliminary results. Mar. Pollut. Bull. 2019, 142, 303–308. [Google Scholar] [CrossRef]
- Soto-Varela, Z.E.; Rosado-Porto, D.; Bolívar-Anillo, H.J.; González, C.P.; Pantoja, B.G.; Alvarado, D.E.; Anfuso, G. Preliminary microbiological coastal water quality determination along the Department of Atlántico (Colombia): Relationships with beach characteristics. J. Mar. Sci. Eng. 2021, 9, 122. [Google Scholar] [CrossRef]
- Botero, C.M.; Tamayo, D.; Zielinski, S.; Anfuso, G. Qualitative and quantitative beach cleanliness assessment to support marine litter management in tropical destinations. Water 2021, 13, 3455. [Google Scholar] [CrossRef]
- Shah, A.H.; Abdelzaher, A.M.; Phillips, M.; Hernandez, R.; Solo-Gabriele, H.M.; Kish, J.; Scorzetti, G.; Fell, J.W.; Diaz, M.R.; Scott, T.M. Indicator microbes correlate with pathogenic bacteria, yeasts and helminthes in sand at a subtropical recreational beach site. J. Appl. Microbiol. 2011, 110, 1571–1583. [Google Scholar] [CrossRef]
- Fujioka, R.S.; Solo-Gabriele, H.M.; Byappanahalli, M.N.; Kirs, M.U.S. Recreational water quality criteria: A vision for the future. Int. J. Environ. Res. Public Health 2015, 12, 7752–7776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brinkmeyer, R.; Amon, R.M.W.; Schwarz, J.R.; Saxton, T.; Roberts, D.; Harrison, S.; Ellis, N.; Fox, J.; Di Guardi, K.; Hochman, M.; et al. Distribution and persistence of Escherichia coli and Enterococci in stream bed and bank sediments from two urban streams in Houston, TX. Sci. Total Environ. 2015, 502, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Perkins, T.L.; Clements, K.; Baas, J.H.; Jago, C.F.; Jones, D.L.; Malham, S.K.; McDonald, J.E. Sediment composition influences spatial variation in the abundance of human pathogen indicator bacteria within an estuarine environment. PLoS ONE 2014, 9, e112951. [Google Scholar] [CrossRef]
- Larrea-Murrell, J.A.; Rojas-Badía, M.M.; Romeu-Álvarez, B.; Rojas-Hernández, N.; Heydrich-Pérez, M. Bacterias indicadoras de contaminación fecal en la evaluación de la calidad de las aguas: Revisión de la literatura. Rev. CENIC Cienc. Biol. 2013, 44, 24–34. [Google Scholar]
- Şanlıtürk, G.; Güran, M. Monitoring of microbiological dynamics in beach sand and seawater samples from recreational and non-recreational beaches over a two-year period. Int. J. Environ. Health Res. 2021, 32, 9, 1–13. [Google Scholar] [CrossRef]
- Sassi, H.P.; van Ogtrop, F.; Morrison, C.M.; Zhou, K.; Duan, J.G.; Gerba, C.P. Sediment re-suspension as a potential mechanism for viral and bacterial contaminants. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2020, 55, 1398–1405. [Google Scholar] [CrossRef]
- European Community Commission (ECC). Directive 2006/7/EC Concerning the Management of Bathing Water Quality and Repealing; ECC: Brussels, Belgium, 2006; pp. 37–51. [Google Scholar]
- United States Environmental Protection Agency. Recreational Water Quality Criteria; United States Environmental Protection Agency: Washington, DC, USA, 2012; pp. 1–69.
- Hassard, F.; Gwyther, C.L.; Farkas, K.; Andrews, A.; Jones, V.; Cox, B.; Brett, H.; Jones, D.L.; McDonald, J.E.; Malham, S.K. Abundance and distribution of enteric bacteria and viruses in coastal and estuarine sediments—A review. Front. Microbiol. 2016, 7, 1692. [Google Scholar] [CrossRef] [Green Version]
- Karbasdehi, V.N.; Dobaradaran, S.; Nabipour, I.; Ostovar, A.; Arfaeinia, H.; Vazirizadeh, A.; Mirahmadi, R.; Keshtkar, M.; Ghasemi, F.F.; Khalifei, F. Indicator bacteria community in seawater and coastal sediment: The Persian Gulf as a case. J. Environ. Health Sci. Eng. 2017, 15, 6. [Google Scholar] [CrossRef] [Green Version]
- Jamieson, R.; Joy, D.M.; Lee, H.; Kostaschuk, R.; Gordon, R. Transport and deposition of sediment-associated Escherichia coli in natural streams. Water Res. 2005, 39, 2665–2675. [Google Scholar] [CrossRef] [PubMed]
- Nevers, M.B.; Byappanahalli, M.N.; Nakatsu, C.H.; Kinzelman, J.L.; Phanikumar, M.S.; Shively, D.A.; Spoljaric, A.M. Interaction of bacterial communities and indicators of water quality in shoreline sand, sediment, and water of Lake Michigan. Water Res. 2020, 178, 115671. [Google Scholar] [CrossRef] [PubMed]
- Abia, A.L.K.; Ubomba-Jaswa, E.; du Preez, M.; Momba, M.N.B. Riverbed sediments in the Apies River, South Africa: Recommending the use of both Clostridium perfringens and Escherichia coli as indicators of faecal pollution. J. Soils Sediments 2015, 15, 2412–2424. [Google Scholar] [CrossRef]
- Brandão, J.; Albergaria, I.; Albuquerque, J.; José, S.; Grossinho, J.; Ferreira, F.C.; Raposo, A.; Rodrigues, R.; Silva, C.; Jordao, L.; et al. Untreated sewage contamination of beach sand from a leaking underground sewage system. Sci. Total Environ. 2020, 740, 140237. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.J.; Droppo, I.G.; Liss, S.N.; Warren, L.; Wolfaardt, G. Influence of wave action on the partitioning and transport of unattached and floc-associated bacteria in fresh water. Can. J. Microbiol. 2015, 61, 584–596. [Google Scholar] [CrossRef] [Green Version]
- Garrido-Pérez, M.C.; Anfuso, E.; Acevedo, A.; Perales-Vargas-Machuca, J.A. Microbial indicators of faecal contamination in waters and sediments of beach bathing zones. Int. J. Hyg. Environ. Health 2008, 211, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, R.L.; Echeverry, A.; Stinson, C.M.; Green, M.; Bonilla, T.D.; Hartz, A.; McCorquodale, D.S.; Rogerson, A.; Esiobu, N. Survival trends of Staphylococcus aureus, Pseudomonas aeruginosa, and Clostridium perfringens in a sandy South Florida beach. Mar. Pollut. Bull. 2012, 64, 1201–1209. [Google Scholar] [CrossRef]
- Shibata, T.; Solo-Gabriele, H.M.; Fleming, L.E.; Elmir, S. Monitoring marine recreational water quality using multiple microbial indicators in an urban tropical environment. Water Res. 2004, 38, 3119–3131. [Google Scholar] [CrossRef] [Green Version]
- INVEMAR (Instituto de Investigaciones Marinas y Costeras). Ordenamiento Ambiental de la Zona Costera del Departamento del Atlántico; INVEMAR: Santa Marta, Colombia, 2007; pp. 1–563. [Google Scholar]
- Alcaldía Municipal de Puerto Colombia. Available online: https://www.puertocolombia-atlantico.gov.co/MiMunicipio/Paginas/Sitios-de-Interes.aspx (accessed on 12 October 2022).
- Anfuso, G.; Rangel-Buitrago, N.; Correa, I. Evolution of sandspits along the Caribbean coast of Colombia: Natural and human influences. In Sand and Gravel Spits; Randazzo, G., Jackson, D.W.T., Cooper, J.A.G., Eds.; Springer: New York, NY, USA, 2015; pp. 1–19. ISBN 9783319137162. [Google Scholar]
- Flórez, A.; Robertson, K.G. Morfodinámica del litoral caribe y amenazas naturales. Cuad. Geogr. Rev. Colomb. Geogr. 2001, 10, 1–35. [Google Scholar]
- Molina, A.; Molina, C.; Giraldo, L.; Barrera, R. Características estratigráficas y morfodinámicas de la franja de litoral Caribe colombiano (sector Barranquilla (Bocas de Ceniza)—Flecha de Galerazamba). Bol. Investig. Mar. Costeras 1999, 28, 61–94. [Google Scholar]
- Andrade, C. Cambios Recientes del Nivel del Mar en Colombia. In Deltas de Colombia: Morfodinámica y Vulnerabilidad Ante el Cambio Global; Restrepo, J., Ed.; EAFIT University Press: Medellin, Colombia, 2008; pp. 103–122. [Google Scholar]
- INVEMAR (Instituto de Investigaciones Marinas y Costeras). Climatologie de la Vitesse et la Direction des Vent Pour le Mar Territoriale Sous Juridiction Colombianne 8° a 19° N e 69° a 84° W. In Atlas ERS 1 et 2 et Quickscat, Colombie; INVEMAR: Santa Marta, Colombia, 2006. [Google Scholar]
- Osorio, A.F.; Mesa, J.C.; Bernal, G.R.; Montoya, R.D. Reconstrucción de cuarenta años de datos de oleaje en el mar Caribe colombiano empleando el modelo WWIIITM y diferentes fuentes de datos. Boletín Científico CIOH 2009, 56, 37–56. [Google Scholar] [CrossRef]
- García, L. Clima marítimo, procesos de erosión/acreción y amenazas/vulnerabilidades por erosión: Caso de estudio de la barrera costera de Puerto Velero. Master’s Thesis, Departamento del Atlántico, Universidad del Norte, Barranquilla, Colombia, 2021. [Google Scholar]
- Restrepo, J.D.; Zapata, P.; Díaz, J.M.; Garzón-Ferreira, J.; García, C.B. Fluvial fluxes into the Caribbean Sea and their impact on coastal ecosystems: The Magdalena River, Colombia. Glob. Planet. Change 2006, 50, 33–49. [Google Scholar] [CrossRef]
- Higgins, A.; Restrepo, J.C.; Ortiz, J.C.; Pierini, J.; Otero, L. Suspended sediment transport in the Magdalena River (Colombia, South America): Hydrologic regime, rating parameters and effective discharge variability. Int. J. Sediment Res. 2016, 31, 25–35. [Google Scholar] [CrossRef]
- Rangel-Buitrago, N.; Williams, A.; Anfuso, G.; Arias, M.; Gracia, A. Magnitudes, sources, and management of beach litter along the Atlántico department coastline, Caribbean coast of Colombia. Ocean Coast. Manag. 2017, 138, 142–157. [Google Scholar] [CrossRef]
- Rangel-Buitrago, N.; Velez, A.; Gracia, A.; Mantilla-Barbosa, E.; Arana, V.A.; Trilleras, J.; Arroyo-Olarte, H. Litter impacts on cleanliness and environmental status of Atlántico department beaches, Colombian Caribbean coast. Ocean Coast. Manag. 2019, 179, 104835. [Google Scholar] [CrossRef]
- MyOCEAN. Global Ocean Waves Reanalysis. Available online: https://myocean.marine.copernicus.eu/data?view=viewer&crs=epsg:4326&t (accessed on 1 November 2022).
- American Public Health Association. Standard Methods for Examination of Water and Wastewater, 22nd ed.; American Public Health Association: Washington, DC, USA, 2012; ISBN 978-087553-013-0. [Google Scholar]
- Zimmer-Faust, A.G.; Thulsiraj, V.; Marambio-Jones, C.; Cao, Y.; Griffith, J.F.; Holden, P.A.; Jay, J.A. Effect of freshwater sediment characteristics on the persistence of fecal indicator bacteria and genetic markers within a Southern California watershed. Water Res. 2017, 119, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Giró, S.; Maldonado, A. Análisis granulométrico por métodos automáticos: Tubo de sedimentación y sedigraph. Acta Geol. Hisp. 1985, 20, 95–102. [Google Scholar]
- Das, B.M. Fundamentos de Ingeniería Geotécnica, 4th ed.; Learning, C., Ed.; Cengafe Learning: Boston, MA, USA, 2013; ISBN 9781111576752. [Google Scholar]
- Wentworth, C.K. A scale of grade and class terms for clastic sediments. J. Geol. 1922, 30, 377–392. [Google Scholar] [CrossRef]
- Folk, R.L.; Ward, W.C. Brazos River bar: A study in the significance of grain size parameters. J. Sediment. Res. 1957, 27, 3–26. [Google Scholar] [CrossRef]
- Sánchez-Moreno, H.; Bolívar-Anillo1, H.J.; Villate-Daza, D.A.; Escobar-Olaya, G.; Anfuso, G. Influencia de los impactos antrópicos sobre la evolución del bosque de manglar en Puerto Colombia (Mar Caribe colombiano). Rev. Latinoam. Recur. Nat. 2019, 15, 1–16. [Google Scholar]
- Torres-Bejarano, F.; González-Márquez, L.C.; Díaz-Solano, B.; Torregroza-Espinosa, A.C.; Cantero-Rodelo, R. Effects of beach tourists on bathing water and sand quality at Puerto Velero, Colombia. Environ. Dev. Sustain. 2018, 20, 255–269. [Google Scholar] [CrossRef]
- Williams, A.T.; Micallef, A. Beach Management, Principles & Practice; Earthscan: London, UK, 2009; ISBN 978-1-84407-435-8. [Google Scholar]
- Gavio, B.; Palmer-Cantillo, S.; Mancera, J.E. Historical analysis (2000-2005) of the coastal water quality in San Andrés Island, SeaFlower Biosphere Reserve, Caribbean Colombia. Mar. Pollut. Bull. 2010, 60, 1018–1030. [Google Scholar] [CrossRef]
- Wear, S.L.; Acuña, V.; McDonald, R.; Font, C. Sewage pollution, declining ecosystem health, and cross-sector collaboration. Biol. Conserv. 2021, 255, 109010. [Google Scholar] [CrossRef]
- Fernández-Nóvoa, D.; Mendes, R.; Dias, J.M.; Sánchez-Arcilla, A.; Gómez-Gesteira, M. Analysis of the influence of river discharge and wind on the Ebro turbid plume using MODIS-Aqua and MODIS-Terra data. J. Mar. Syst. 2015, 142, 40–46. [Google Scholar] [CrossRef]
- Torregroza-Espinosa, A.C.; Restrepo, J.C.; Correa-Metrio, A.; Hoyos, N.; Escobar, J.; Pierini, J.; Martínez, J.M. Fluvial and oceanographic influences on suspended sediment dispersal in the Magdalena River Estuary. J. Mar. Syst. 2020, 204, 103282. [Google Scholar] [CrossRef]
- INVEMAR (Instituto de Investigaciones Marinas y Costeras). Diagnóstico y Evaluación de la Calidad de las Aguas Marinas y Costeras del Caribe y Pacifico Colombianos; INVEMAR: Santa Marta, Colombia, 2016; pp. 1–260. [Google Scholar]
- INVEMAR (Instituto de Investigaciones Marinas y Costeras). Diagnóstico y Evaluación de la Calidad de las Aguas Marinas y Costeras del Caribe y Pacifico Colombianos; INVEMAR: Santa Marta, Colombia, 2017; pp. 1–337. [Google Scholar]
- INVEMAR (Instituto de Investigaciones Marinas y Costeras). Diagnóstico y Evaluacion de la Calidad de las Aguas Marinas y Costeras del Caribe y Pacifico Colombianos; INVEMAR: Santa Marta, Colombia, 2018; pp. 1–213. [Google Scholar]
- INVEMAR (Instituto de Investigaciones Marinas y Costeras). Diagnóstico y Evaluación de la Calidad de las Aguas Marinas y Costeras en el Caribe y Pacífico Colombianos; INVEMAR: Santa Marta, Colombia, 2019; pp. 1–164. [Google Scholar]
- Gast, R.J.; Gorrell, L.; Raubenheimer, B.; Elgar, S. Impact of erosion and accretion on the distribution of enterococci in beach sands. Cont. Shelf Res. 2011, 31, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Buitrago, N.; Castro-Barros, J.D.; Gracia, A.; Villadiego, J.D.V.; Williams, A.T. Litter impacts on beach/dune systems along the Atlántico Department, the Caribbean Coastline of Colombia. Mar. Pollut. Bull. 2018, 137, 35–44. [Google Scholar] [CrossRef]
- Rangel-Buitrago, N.; Gracia, C.A.; Vélez-Mendoza, A.; Mantilla-Barbosa, E.; Arana, V.A.; Trilleras, J.; Arroyo-Olarte, H. Abundance and distribution of beach litter along the Atlántico Department, Caribbean coast of Colombia. Mar. Pollut. Bull 2018, 136, 435–447. [Google Scholar] [CrossRef]
- Williams, A.T.; Rangel-Buitrago, N.G.; Anfuso, G.; Cervantes, O.; Botero, C.M. Litter impacts on scenery and tourism on the Colombian north Caribbean coast. Tour. Manag. 2016, 55, 209–224. [Google Scholar] [CrossRef]
- Huang, G.; Falconer, R.A.; Lin, B. Integrated hydro-bacterial modelling for predicting bathing water quality. Estuar. Coast. Shelf Sci. 2017, 188, 145–155. [Google Scholar] [CrossRef]
- Ferguson, D.M.; Moore, D.F.; Getrich, M.A.; Zhowandai, M.H. Enumeration and speciation of enterococci found in marine and intertidal sediments and coastal water in southern California. J. Appl. Microbiol. 2005, 99, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Hur, H.G.; Sadowsky, M.J.; Byappanahalli, M.N.; Yan, T.; Ishii, S. Environmental Escherichia coli: Ecology and public health implications—A review. J. Appl. Microbiol. 2017, 123, 570–581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piggot, A.M.; Klaus, J.S.; Johnson, S.; Phillips, M.C.; Helena, M.S.G. Relationship between enterococcal levels and sediment biofilms at recreational beaches in South Florida. Appl. Environ. Microbiol. 2012, 78, 5973–5982. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, C.M.; Lin, T.Y.; Lin, C.C.; Kohbodi, G.N.A.; Bhatt, A.; Lee, R.; Jay, J.A. Persistence of fecal indicator bacteria in Santa Monica Bay beach sediments. Water Res. 2006, 40, 2593–2602. [Google Scholar] [CrossRef] [PubMed]
- Sabino, R.; Rodrigues, R.; Costa, I.; Carneiro, C.; Cunha, M.; Duarte, A.; Faria, N.; Ferreira, F.C.; Gargaté, M.J.; Júlio, C.; et al. Routine screening of harmful microorganisms in beach sands: Implications to public health. Sci. Total Environ. 2014, 472, 1062–1069. [Google Scholar] [CrossRef]
- Papadakis, J.A.; Mavridou, A.; Richardson, S.C.; Lampiri, M.; Marcelou, U. Bather-related microbial and yeast populations in sand and seawater. Water Res. 1997, 31, 799–804. [Google Scholar] [CrossRef]
- Bonilla, T.D.; Nowosielski, K.; Cuvelier, M.; Hartz, A.; Green, M.; Esiobu, N.; McCorquodale, D.S.; Fleisher, J.M.; Rogerson, A. Prevalence and distribution of fecal indicator organisms in South Florida beach sand and preliminary assessment of health effects associated with beach sand exposure. Mar. Pollut. Bull. 2007, 54, 1472–1482. [Google Scholar] [CrossRef]
- Ishii, S.; Yan, T.; Vu, H.; Hansen, D.L.; Hicks, R.E.; Sadowsky, M.J. Factors controlling long-term survival and growth of naturalized Escherichia coli populations in temperate field soils. Microbes Environ. 2010, 25, 8–14. [Google Scholar] [CrossRef] [Green Version]
- Abdelzaher, A.M.; Wright, M.E.; Ortega, C.; Solo-Gabriele, H.M.; Miller, G.; Elmir, S.; Newman, X.; Shih, P.; Alfredo Bonilla, J.; Bonilla, T.D.; et al. Presence of pathogens and indicator microbes at a non-point source subtropical recreational marine beach. Appl. Environ. Microbiol. 2010, 76, 724–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavez, L.D.; Gulloso, E.M.; Murgas, Y.G.; Vega, J.R. A baseline study of fecal indicator bacteria on Caribbean beach sand from Riohacha, La Guajira, Colombia. Contemp. Eng. Sci. 2018, 11, 5093–5107. [Google Scholar] [CrossRef]
- Byappanahalli, M.N.; Nevers, M.B.; Korajkic, A.; Staley, Z.R.; Harwood, V.J. Enterococci in the Environment. Microbiol. Mol. Biol. Rev. 2012, 76, 685–706. [Google Scholar] [CrossRef] [Green Version]
- Alm, E.W.; Burke, J.; Spain, A. Fecal indicator bacteria are abundant in wet sand at freshwater beaches. Water Res. 2003, 37, 3978–3982. [Google Scholar] [CrossRef]
- Stelma, G.N. Use of bacterial spores in monitoring water quality and treatment. J. Water Health 2018, 16, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Heaney, C.D.; Exum, N.G.; Dufour, A.P.; Brenner, K.P.; Haugland, R.A.; Chern, E.; Schwab, K.J.; Love, D.C.; Serre, M.L.; Noble, R.; et al. Water quality, weather and environmental factors associated with fecal indicator organism density in beach sand at two recreational marine beaches. Sci. Total Environ. 2014, 497–498, 440–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lisle, J.T.; Smith, J.J.; Edwards, D.D.; McFeters, G.A. Occurrence of microbial indicators and Clostridium perfringens in wastewater, water column samples, sediments, drinking water, and Weddell seal feces collected at McMurdo Station, Antarctica. Appl. Environ. Microbiol. 2004, 70, 7269–7276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davies, C.M.; Long, J.A.H.; Donald, M.; Ashbolt, N.J. Survival of fecal microorganisms in marine and freshwater sediments. Appl. Environ. Microbiol. 1995, 61, 1888–1896. [Google Scholar] [CrossRef]
- Wang, A.; Lin, B.; Sleep, B.E.; Liss, S.N. The impact of biofilm growth on transport of Escherichia coli O157:H7 in sand. Ground Water 2011, 49, 20–31. [Google Scholar] [CrossRef]
- Tymensen, L.D.; Pyrdok, F.; Coles, D.; Koning, W.; Mcallister, T.A.; Jokinen, C.C.; Dowd, S.E.; Neumann, N.F. Comparative accessory gene fingerprinting of surface water Escherichia coli reveals genetically diverse naturalized population. J. Appl. Microbiol. 2015, 119, 263–277. [Google Scholar] [CrossRef]
- Ishii, S.; Sadowsky, M.J. Escherichia coli in the environment: Implications for water quality and human health. Microbes Environ. 2008, 23, 101–108. [Google Scholar] [CrossRef] [Green Version]
- Solo-Gabriele, H.M.; Harwood, V.J.; Kay, D.; Fujioka, R.S.; Sadowsky, M.J.; Whitman, R.L.; Wither, A.; Caniça, M.; Da Fonseca, R.C.; Duarte, A.; et al. Beach sand and the potential for infectious disease transmission: Observations and recommendations. J. Mar. Biol. Assoc. UK 2016, 96, 101–120. [Google Scholar] [CrossRef]

| Sampling Point and Location | Wind Speed (m/s) * | Wave Height (m) * | Direction (°) | Beach Exposition Level | Streams/ Outflows | Beach Typology | Tourist Activities/ Infrastructures |
|---|---|---|---|---|---|---|---|
| P1. PUERTO MOCHO | 8.2 | 0.54 | 010° | Low | Absent | Rural | Present |
| P2. SABANILLA | 8.2 | 1.43 | 010° | High | Absent | Rural | Present |
| P3. SALGAR | 8.2 | 1.26 | 010° | High | Absent | Urban | Present |
| P4. SALGAR | 8.2 | 1.26 | 010° | High | Absent | Urban | Present |
| P5. SALGAR | 7.4 | 1.68 | 010° | High | Present | Urban | Present |
| P6. PRADOMAR | 4.6 | 1.41 | 010° | High | Present | Urban | Present |
| P7. POPULAR | 7.4 | 1.43 | 010° | High | Present | Urban | Present |
| P8. PUERTO COLOMBIA | 4.6 | 1.41 | 010° | High | Present | Urban | Present |
| P9. BALBOA | 4.6 | 1.41 | 010° | High | Absent | Rural | Absent |
| P10. PUERTO VELERO | 7.4 | 1.68 | 010° | High | Absent | Remote | Absent |
| P11. PUERTO VELERO | 7.4 | 0.20 | 220° | Low | Present | Rural | Present |
| Sampling Point | Sample Location | Mean (phi) | Sorting | ||
|---|---|---|---|---|---|
| P1. PUERTO MOCHO | Beach face | 1.63 | Medium sand | 0.51 | Moderately well sorted |
| Backshore | 0.56 | Coarse sand | 0.39 | Well sorted | |
| P2. SABANILLA | Beach face | 1.59 | Medium sand | 0.53 | Moderately well sorted |
| Backshore | 1.52 | Medium sand | 0.52 | Moderately well sorted | |
| P3. SALGAR | Beach face | 1.57 | Medium sand | 0.50 | Well sorted |
| Backshore | 0.55 | Coarse sand | 0.44 | Well sorted | |
| P4. SALGAR | Beach face | 1.81 | Medium sand | 0.54 | Moderately well sorted |
| Backshore | 1.50 | Medium sand | 0.51 | Moderately well sorted | |
| P5. SALGAR | Beach face | 1.63 | Medium sand | 0.54 | Moderately well sorted |
| Backshore | 0.96 | Coarse sand | 0.31 | Very well sorted | |
| P6. PRADOMAR | Beach face | 1.50 | Medium sand | 0.57 | Moderately well sorted |
| Backshore | 1.54 | Medium sand | 0.54 | Moderately well sorted | |
| P7. POPULAR | Beach face | 1.86 | Medium sand | 0.51 | Moderately well sorted |
| Backshore | 1.47 | Medium sand | 0.51 | Moderately well sorted | |
| P8. PUERTO COLOMBIA | Beach face | 1.82 | Medium sand | 0.61 | Moderately well sorted |
| Backshore | 1.52 | Medium sand | 0.57 | Moderately well sorted | |
| P9. BALBOA | Beach face | 1.43 | Medium sand | 0.59 | Moderately well sorted |
| Backshore | 1.45 | Medium sand | 0.66 | Moderately well sorted | |
| P10. PUERTO VELERO | Beach face | 1.59 | Medium sand | 0.61 | Moderately well sorted |
| Backshore | 1.59 | Medium sand | 0.49 | Well sorted | |
| P11. PUERTO VELERO | Beach face | 1.48 | Medium sand | 0.62 | Moderately well sorted |
| Backshore | 1.50 | Medium sand | 0.52 | Moderately well sorted | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bolívar-Anillo, H.J.; Soto-Varela, Z.E.; Sánchez Moreno, H.; Villate Daza, D.A.; Rosado-Porto, D.; Vega Benites, S.; Pichón González, C.; Anfuso, G. A Preliminary Approximation to Microbiological Beach Sand Quality along the Coast of the Department of Atlántico (Caribbean Sea of Colombia): Influence of the Magdalena River. Water 2023, 15, 48. https://doi.org/10.3390/w15010048
Bolívar-Anillo HJ, Soto-Varela ZE, Sánchez Moreno H, Villate Daza DA, Rosado-Porto D, Vega Benites S, Pichón González C, Anfuso G. A Preliminary Approximation to Microbiological Beach Sand Quality along the Coast of the Department of Atlántico (Caribbean Sea of Colombia): Influence of the Magdalena River. Water. 2023; 15(1):48. https://doi.org/10.3390/w15010048
Chicago/Turabian StyleBolívar-Anillo, Hernando José, Zamira E. Soto-Varela, Hernando Sánchez Moreno, Diego Andrés Villate Daza, David Rosado-Porto, Shersy Vega Benites, Camila Pichón González, and Giorgio Anfuso. 2023. "A Preliminary Approximation to Microbiological Beach Sand Quality along the Coast of the Department of Atlántico (Caribbean Sea of Colombia): Influence of the Magdalena River" Water 15, no. 1: 48. https://doi.org/10.3390/w15010048
APA StyleBolívar-Anillo, H. J., Soto-Varela, Z. E., Sánchez Moreno, H., Villate Daza, D. A., Rosado-Porto, D., Vega Benites, S., Pichón González, C., & Anfuso, G. (2023). A Preliminary Approximation to Microbiological Beach Sand Quality along the Coast of the Department of Atlántico (Caribbean Sea of Colombia): Influence of the Magdalena River. Water, 15(1), 48. https://doi.org/10.3390/w15010048







