Reliability Treatment of Silicon in Oilfield Wastewater by Electrocoagulation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material and Reagent
2.2. Experimental Setup
2.3. Experimental Method
2.4. Analysis Method
3. Results and Discussions
3.1. Single Factor Experiment
3.2. Orthogonal Experiment
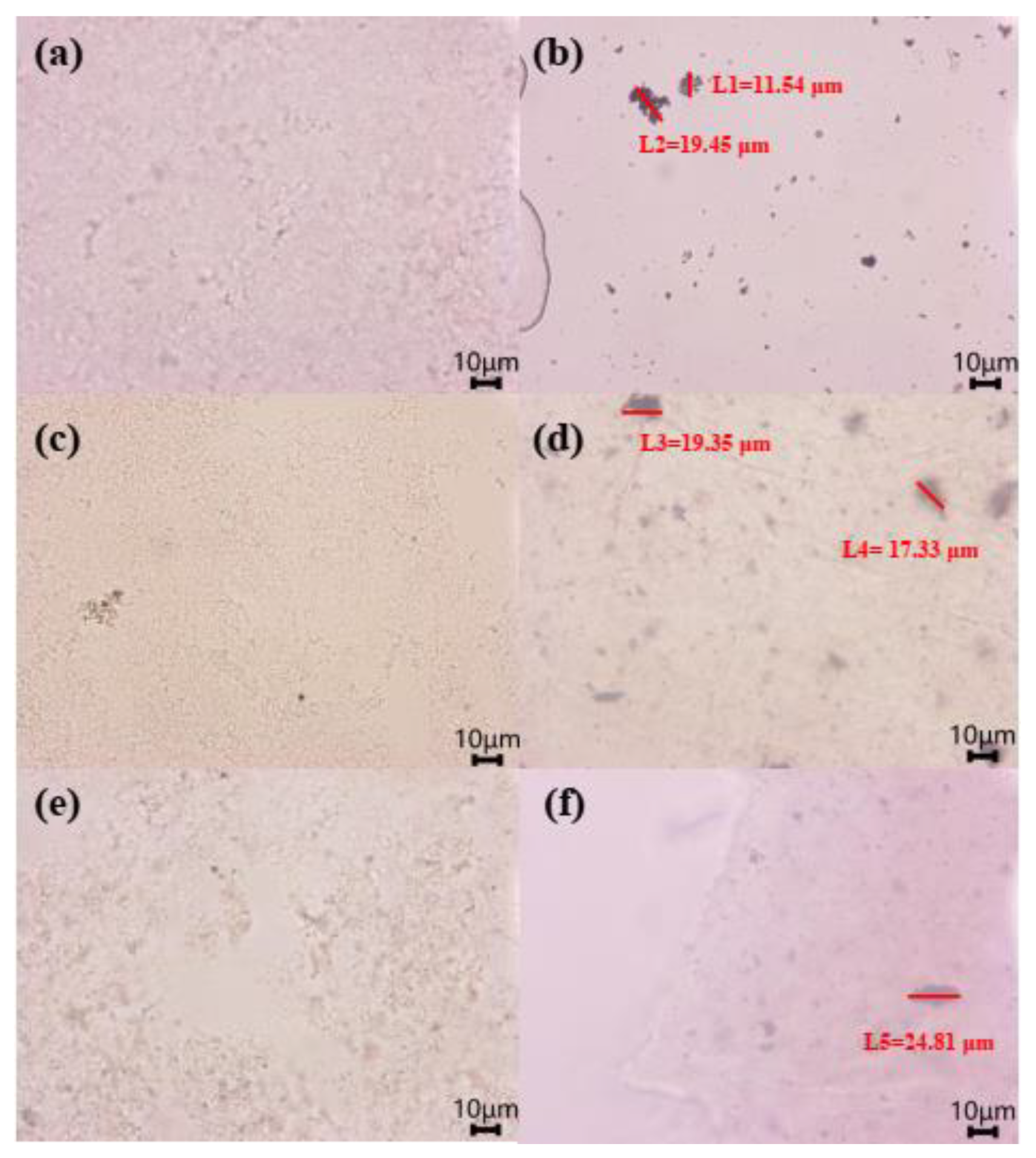
3.3. Changes in Silicon Scale during Electrocoagulation
3.4. Experiment on Wastewater from the Hongshan Oilfield
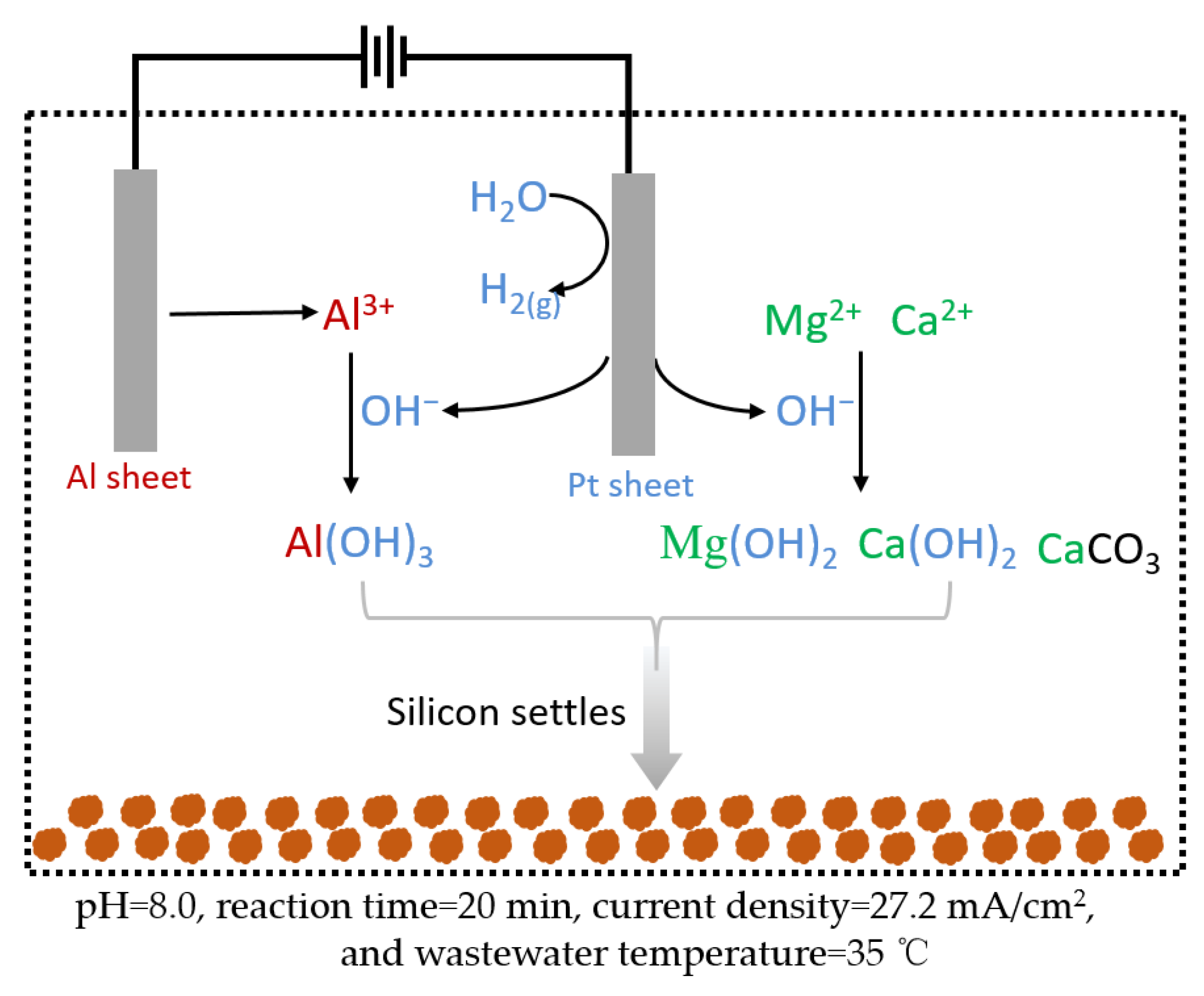
3.5. Mechanism Investigation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ghafoori, S.; Omar, M.; Koutahzadeh, N.; Zendehboudi, S.; Malhas, R.N.; Mohamed, M.; Al-Zubaidi, S.; Redha, K.; Baraki, F.; Mehrvar, M. New advancements, challenges, and future needs on treatment of oilfield produced water: A state-of-the-art review. Sep. Purif. Technol. 2022, 289, 120652. [Google Scholar] [CrossRef]
- Koo, T.; Lee, Y.J.; Sheikholeslami, R. Silica fouling and cleaning of reverse osmosis membranes. Desalination 2001, 139, 43–56. [Google Scholar] [CrossRef]
- Wang, X.J.; Goual, L.; Colberg, P.J.S. Characterization and treatment of dissolved organic matter from oilfield produced waters. J. Hazard. Mater. 2012, 217, 164–170. [Google Scholar] [CrossRef]
- Lu, K.-G.; Li, M.; Huang, H. Silica scaling of reverse osmosis membranes preconditioned by natural organic matter. Sci. Total Environ. 2020, 746, 141178. [Google Scholar] [CrossRef] [PubMed]
- Sheikholeslami, R.; Tan, S. Effects of water quality on silica fouling of desalination plants. Desalination 1999, 126, 267–280. [Google Scholar] [CrossRef]
- Allen, E.W. Process water treatment in Canada’s oil sands industry: I. Target pollutants and treatment objectives. J. Environ. Eng. Sci. 2008, 7, 123–138. [Google Scholar] [CrossRef] [Green Version]
- Nariyan, E.; Sillanpää, M.; Wolkersdorfer, C. Electrocoagulation treatment of mine water from the deepest working European metal mine–performance, isotherm and kinetic studies. Sep. Purif. Technol. 2017, 177, 363–373. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Piekutin, J. The Identification of Fouling in Reverse Osmosis in the Treatment of Water with Petroleum Substances. Water 2021, 13, 1092. [Google Scholar] [CrossRef]
- Lunevich, L.; Sanciolo, P.; Dumée, L.F.; Gray, S.R. Silica fouling in high salinity waters in reverse osmosis desalination (sodium–silica system). Environ. Sci. Water Res. Technol. 2016, 2, 539–548. [Google Scholar] [CrossRef]
- Shemer, H.; Melki-Dabush, N.; Semiat, R. Removal of silica from brackish water by integrated adsorption/ultrafiltration process. Environ. Sci. Pollut. Res. 2019, 26, 31623–31631. [Google Scholar] [CrossRef] [PubMed]
- Tu, K.L.; Chivas, A.R.; Nghiem, L.D. Effects of membrane fouling and scaling on boron rejection by nanofiltration and reverse osmosis membranes. Desalination 2011, 279, 269–277. [Google Scholar] [CrossRef] [Green Version]
- Sik Ali, M.B.; Hamrouni, B.; Bouguecha, S.; Dhahbi, M. Silica removal using ion-exchange resins. Desalination 2004, 167, 273–279. [Google Scholar] [CrossRef]
- Pedregal Montes, A.I.; Abeywickrama, J.; Hoth, N.; Grimmer, M.; Drebenstedt, C. Modeling of Ion Exchange Processes to Optimize Metal Removal from Complex Mine Water Matrices. Water 2021, 13, 3109. [Google Scholar] [CrossRef]
- Den, W.; Wang, C.J. Removal of silica from brackish water by electrocoagulation pretreatment to prevent fouling of reverse osmosis membranes. Sep. Purif. Technol. 2008, 59, 318–325. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, M.; Idrus, M.A.M.; Crombie, C.; Jegatheesan, V. Performance of precipitation and electrocoagulation as pretreatment of silica removal in brackish water and seawater. Process Saf. Environ. Prot. 2019, 126, 18–24. [Google Scholar] [CrossRef]
- Gutiérrez, Á.; Rodríguez, J.F.; Castañeda, L.F.; Nava, J.L.; Coreño, O.; Carreño, G. Abatement of As and hydrated silica from natural groundwater by electrocoagulation in a continuous plant having an electrolyzer and a flocculator-settler. Sep. Purif. Technol. 2022, 281, 119895. [Google Scholar] [CrossRef]
- Chow, H.; Pham, A.L.-T. Effective removal of silica and sulfide from oil sands thermal in-situ produced water by electrocoagulation. J. Hazard. Mater. 2019, 380, 120880. [Google Scholar] [CrossRef]
- Shokri, A.; Fard, M.S. A critical review in electrocoagulation technology applied for oil removal in industrial wastewater. Chemosphere 2022, 288, 132355. [Google Scholar] [CrossRef]
- Wan, W.; Pepping, T.J.; Banerji, T.; Chaudhari, S.; Giammar, D.E. Effects of water chemistry on arsenic removal from drinking water by electrocoagulation. Water Res. 2011, 45, 384–392. [Google Scholar] [CrossRef]
- Khalifa, O.; Banat, F.; Srinivasakannan, C.; Radjenovic, J.; Hasan, S.W. Performance tests and removal mechanisms of aerated electrocoagulation in the treatment of oily wastewater. J. Water Process Eng. 2020, 36, 101290. [Google Scholar] [CrossRef]
- Tao, T.; Liu, S.; Li, S. Advances in management model of urban water resources. J. Water Resour. Water Eng. 2005, 16, 60. [Google Scholar]
- Harif, T.; Khai, M.; Adin, A. Electrocoagulation versus chemical coagulation: Coagulation/flocculation mechanisms and resulting floc characteristics. Water Res. 2012, 46, 3177–3188. [Google Scholar] [CrossRef] [PubMed]
- Eskibalci, M.F.; Ozkan, M.F. Comparison of conventional coagulation and electrocoagulation methods for dewatering of coal preparation plant. Miner. Eng. 2018, 122, 106–112. [Google Scholar] [CrossRef]
- Martin-Dominguez, A.; Rivera-Huerta, M.L.; Perez-Castrejon, S.; Garrido-Hoyos, S.E.; Villegas-Mendoza, I.E.; Gelover-Santiago, S.L.; Drogui, P.; Buelna, G. Chromium removal from drinking water by redox-assisted coagulation: Chemical versus electrocoagulation. Sep. Purif. Technol. 2018, 200, 266–272. [Google Scholar] [CrossRef] [Green Version]
- Mengistu, L.R.; Samuel, Z.A.; Kitila, C.D.; Bayu, A.B. Comparison Study on Sonodirect and Sonoalternate Current Electrocoagulation Process for Domestic Wastewater Treatment. Int. J. Anal. Chem. 2022, 2022, 3477995. [Google Scholar] [CrossRef]
- Das, P.P.; Sharma, M.; Purkait, M.K. Recent progress on electrocoagulation process for wastewater treatment: A review. Sep. Purif. Technol. 2022, 292, 121058. [Google Scholar] [CrossRef]
- Mousazadeh, M.; Niaragh, E.K.; Usman, M.; Khan, S.U.; Sandoval, M.A.; Al-Qodah, Z.; Khalid, Z.B.; Gilhotra, V.; Emamjomeh, M.M. A critical review of state-of-the-art electrocoagulation technique applied to COD-rich industrial wastewaters. Environ. Sci. Pollut. Res. 2021, 28, 43143–43172. [Google Scholar] [CrossRef]
- Cho, W.C.; Poo, K.M.; Mohamed, H.O.; Kim, T.N.; Kim, Y.S.; Hwang, M.H.; Jung, D.W.; Chae, K.J. Non-selective rapid electro-oxidation of persistent, refractory VOCs in industrial wastewater using a highly catalytic and dimensionally stable Ir-Pd/Ti composite electrode. Chemosphere 2018, 206, 483–490. [Google Scholar] [CrossRef]
- Emamjomeh, M.M.; Jamali, H.A.; Naghdali, Z.; Mousazadeh, M. Carwash wastewater treatment by the application of an environmentally friendly hybrid system: An experimental design approach. Desalination Water Treat. 2019, 160, 171–177. [Google Scholar] [CrossRef] [Green Version]
- Shams, M.; Alam, I.; Chowdhury, I. Interactions of nanoscale plastics with natural organic matter and silica surfaces using a quartz crystal microbalance. Water Res. 2021, 197, 117066. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.Y.; Ma, L.P.; Dai, Q.X.; Yang, X.B.; Ao, R.; Yang, J.; Yang, J.; Li, W.G. Modified corn-core powder for enhancing sludge dewaterability: Synthesis, characterization and sludge dewatering performance. Chin. J. Chem. Eng. 2021, 32, 368–377. [Google Scholar] [CrossRef]
- Yasri, N.G.; Ingelsson, M.; Nightingale, M.; Jaggi, A.; Dejak, M.; Kryst, K.; Oldenburg, T.B.P.; Roberts, E.P.L. Investigation of electrode passivation during electrocoagulation treatment with aluminum electrodes for high silica content produced water. Water Sci. Technol. 2022, 85, 925–942. [Google Scholar] [CrossRef] [PubMed]
- Cirak, M. High-temperature electrocoagulation of colloidal calcareo-argillaceous suspension. Powder Technol. 2018, 328, 13–25. [Google Scholar] [CrossRef]
- Korbahti, B.K.; Artut, K. Electrochemical oil/water demulsification and purification of bilge water using Pt/Ir electrodes. Desalination 2010, 258, 219–228. [Google Scholar] [CrossRef]



| Chemicals | Value |
|---|---|
| pH | 8.08 |
| HCO3− (mg/L) | 466.09 |
| Cl− (mg/L) | 19,126.29 |
| Ca2+ (mg/L) | 453.04 |
| Mg2+ (mg/L) | 167.00 |
| SiO32− (mg/L) | 628.20 |
| K+ + Na+ (mg/L) | 12,048.84 |
| Mineralization degree(mg/L) | 32,889.46 |
| Density (g/cm3) | 1.02 |
| Silicon (mg/L) | 83.69 |
| pH | Current Density (mA/cm2) | Reaction Time (min) | Wastewater Temperature (°C) |
|---|---|---|---|
| 5 | 18 | 14 | 35 |
| 6 | 21 | 17 | 40 |
| 7 | 24 | 20 | 45 |
| 8 | 27 | 23 | 50 |
| Evaluation Indexes | Silica Removal Rate | Post-Experimental Silica Content | Anode Mass Loss | Theoretical Optimal Scheme | |
|---|---|---|---|---|---|
| Influencing Factor | |||||
| pH | 6 | 6 | 8 | 6 | |
| Current Density (mA/cm2) | 27 | 27 | 18 | 27 | |
| Reaction time (min) | 23 | 23 | 14 | 20 | |
| Temperature (°C) | 40 | 35 | 50 | 35 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, W.; Liu, S.; Zhang, X.; Zhang, F.; Yang, X.; Xu, M.; Hou, J. Reliability Treatment of Silicon in Oilfield Wastewater by Electrocoagulation. Water 2023, 15, 206. https://doi.org/10.3390/w15010206
Teng W, Liu S, Zhang X, Zhang F, Yang X, Xu M, Hou J. Reliability Treatment of Silicon in Oilfield Wastewater by Electrocoagulation. Water. 2023; 15(1):206. https://doi.org/10.3390/w15010206
Chicago/Turabian StyleTeng, Weiwei, Shijie Liu, Xin Zhang, Feng Zhang, Xianglu Yang, Mengxiao Xu, and Junwei Hou. 2023. "Reliability Treatment of Silicon in Oilfield Wastewater by Electrocoagulation" Water 15, no. 1: 206. https://doi.org/10.3390/w15010206




