Evaluation of Groundwater Quality for Irrigation in Deep Aquifers Using Multiple Graphical and Indexing Approaches Supported with Machine Learning Models and GIS Techniques, Souf Valley, Algeria
Abstract
:1. Introduction
2. Materials and Methods
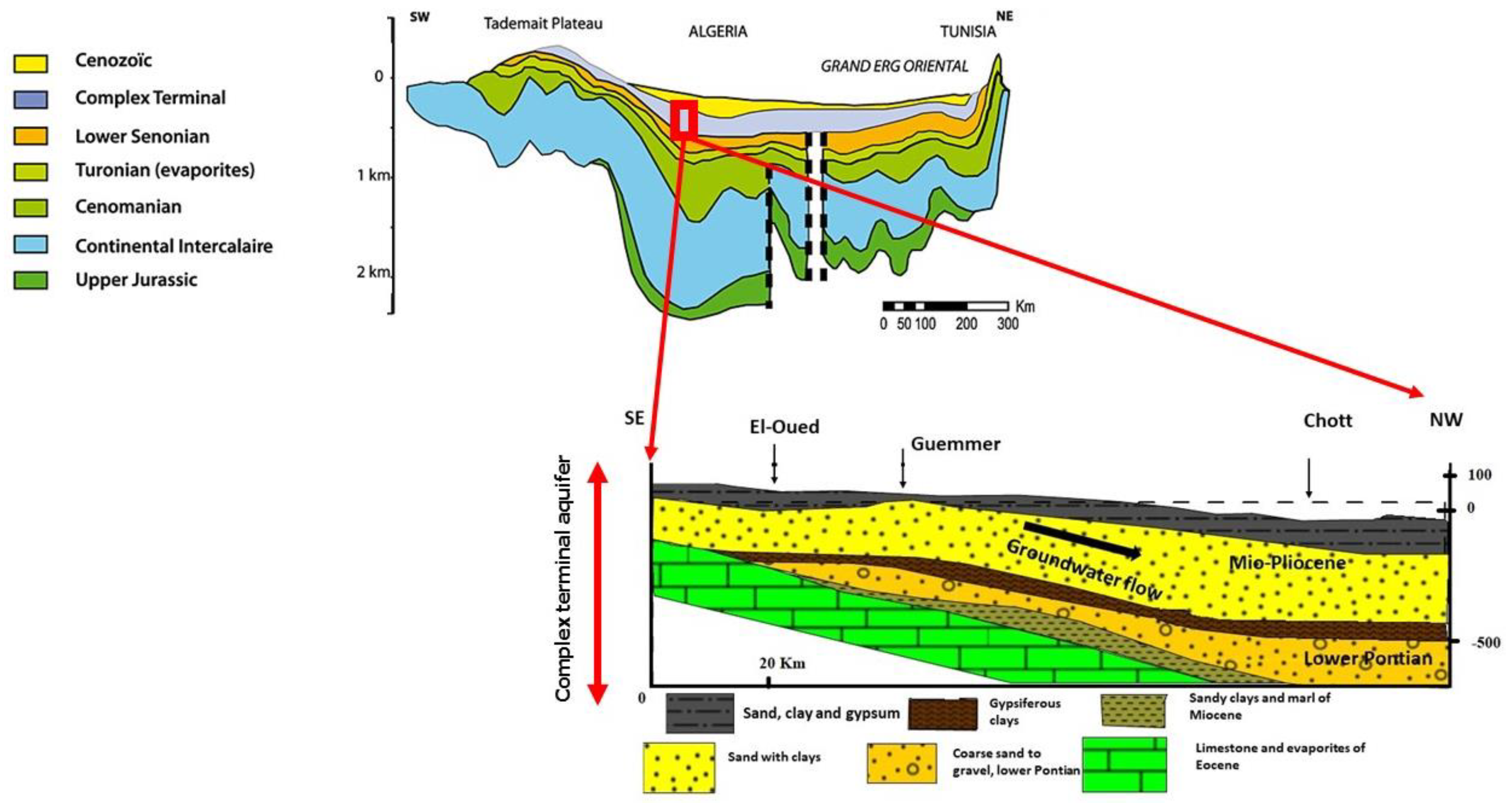
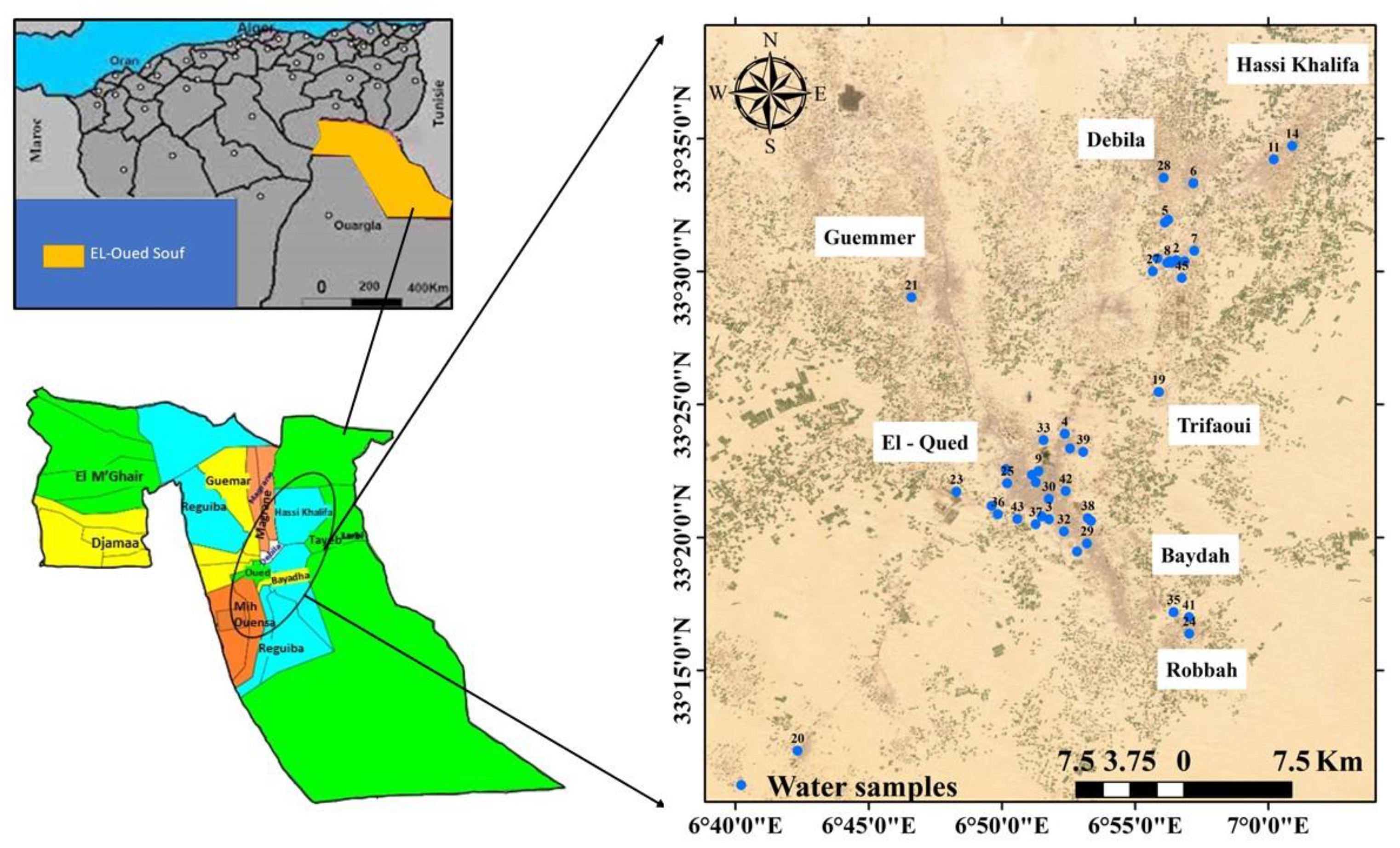
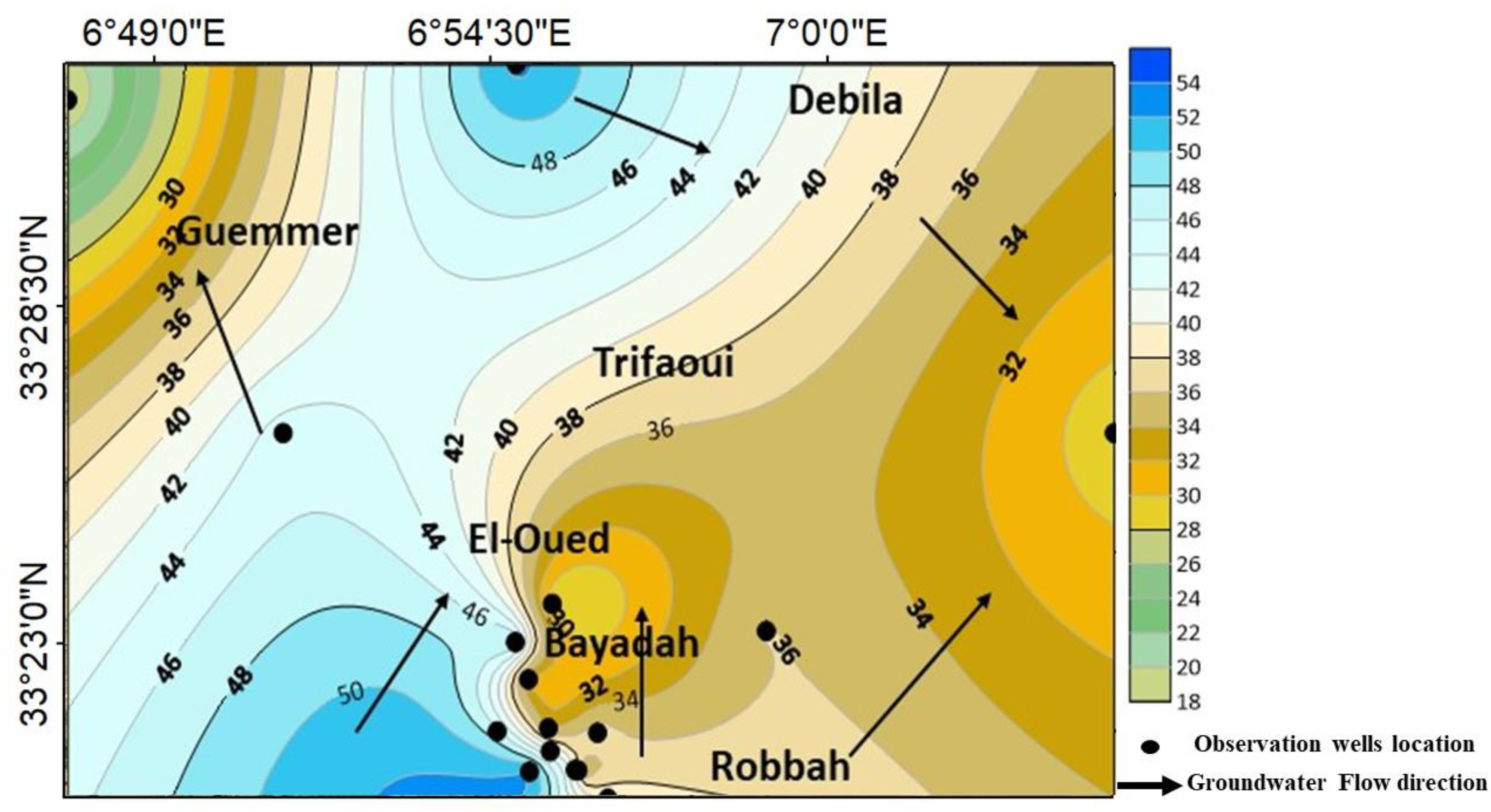
2.1. Site Descriptions and Hydrogeological Settings
2.2. Sampling and Analysis
2.3. Indexing Approach
2.3.1. Irrigation Water Quality Indices (IWQIs)
2.3.2. Irrigation Water Quality Index (IWQI)
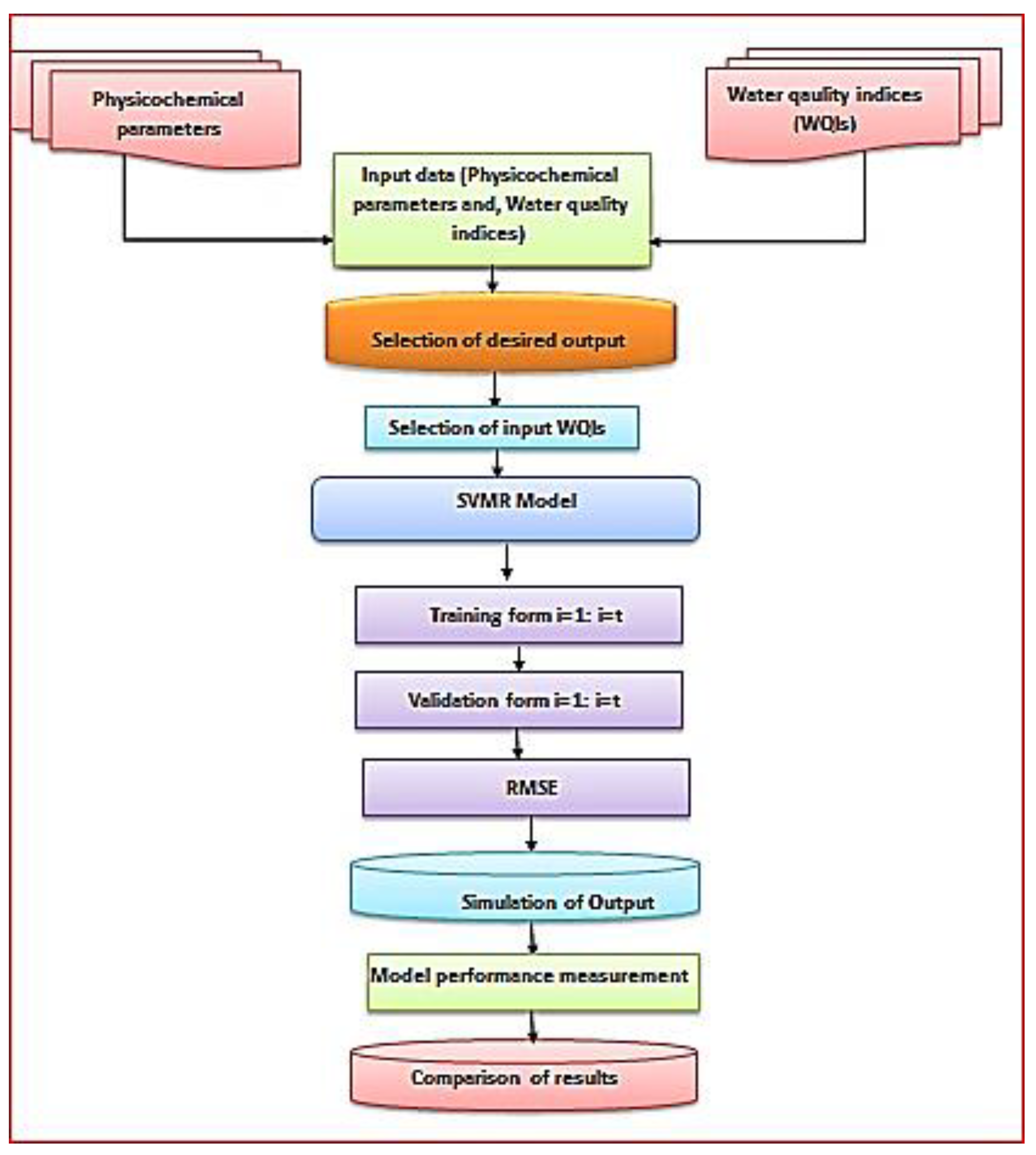
2.3.3. Support Vector Machine Regression (SVMR)
Data Analysis and Processing
3. Results and Discussion
3.1. Physicochemical Parameters of Groundwater
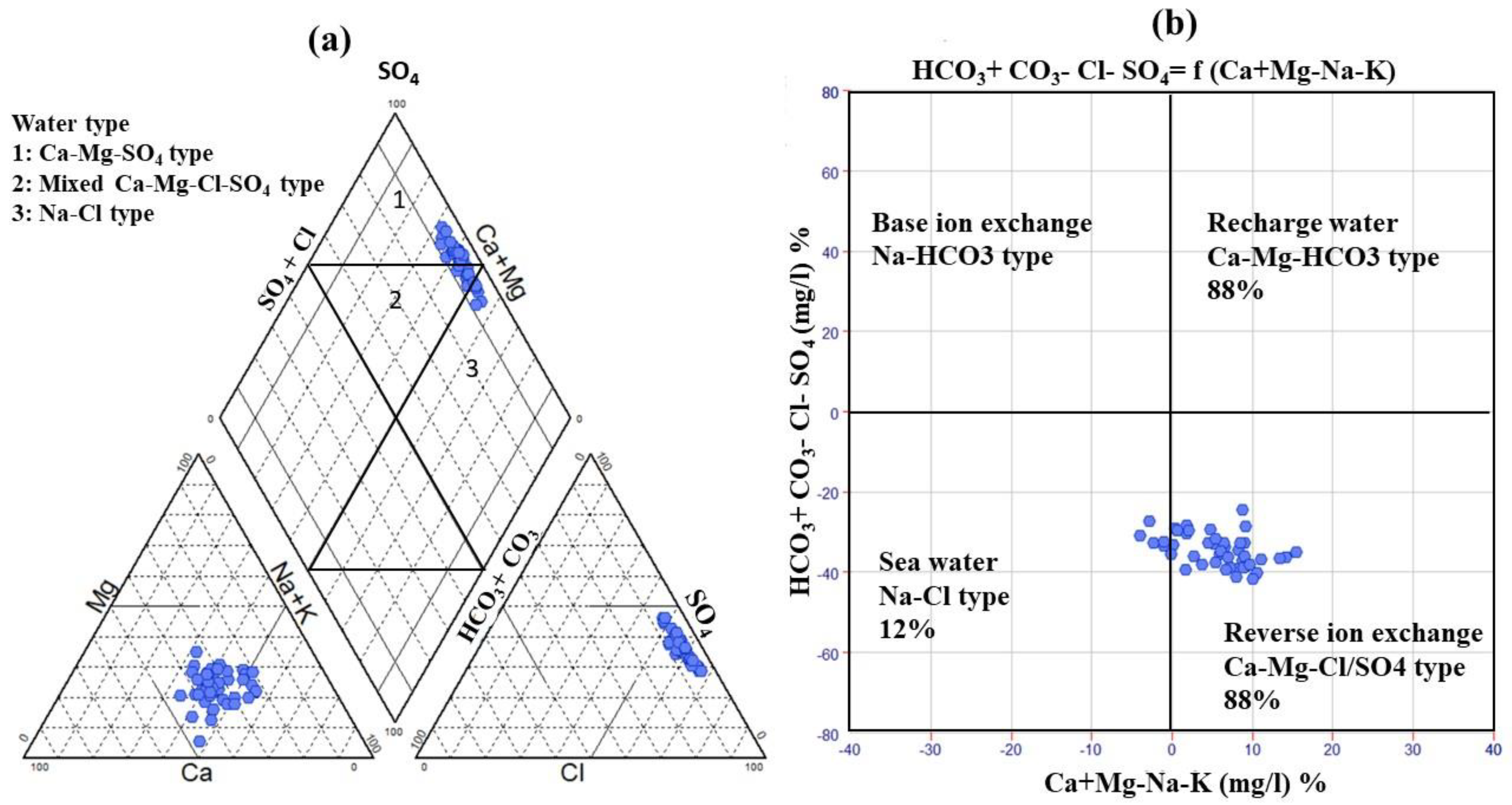
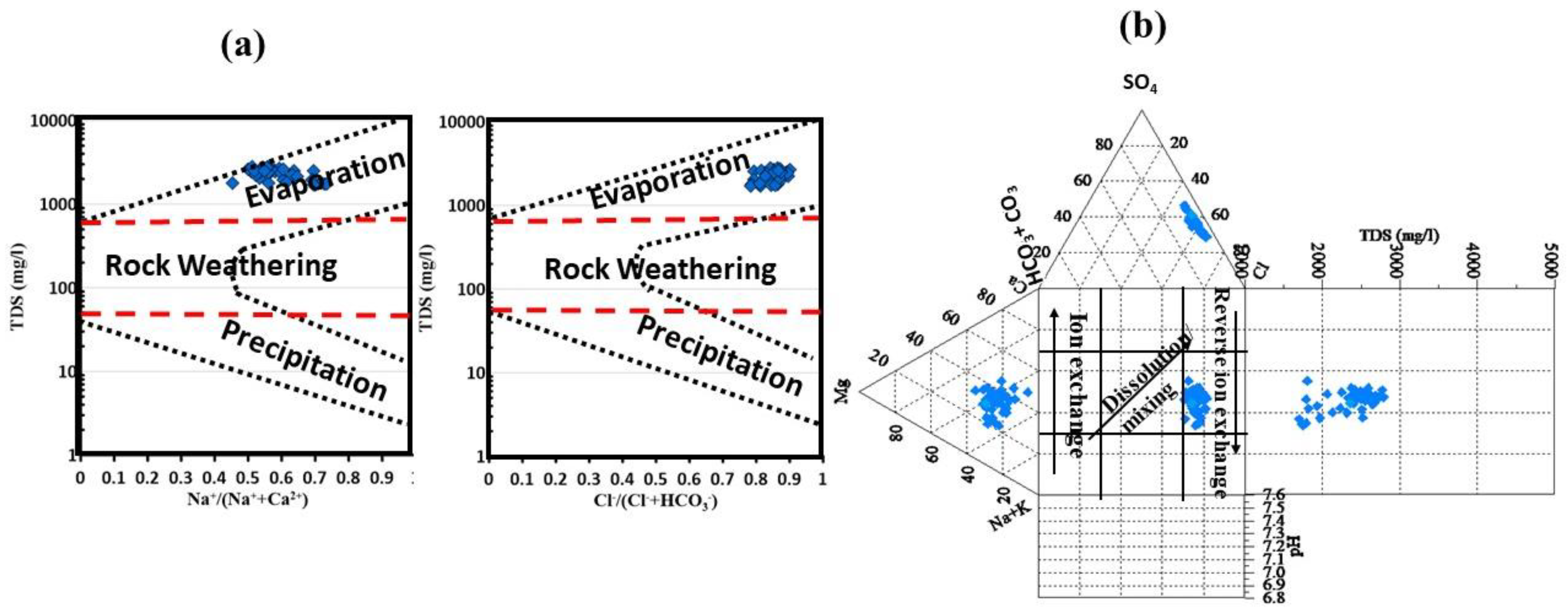
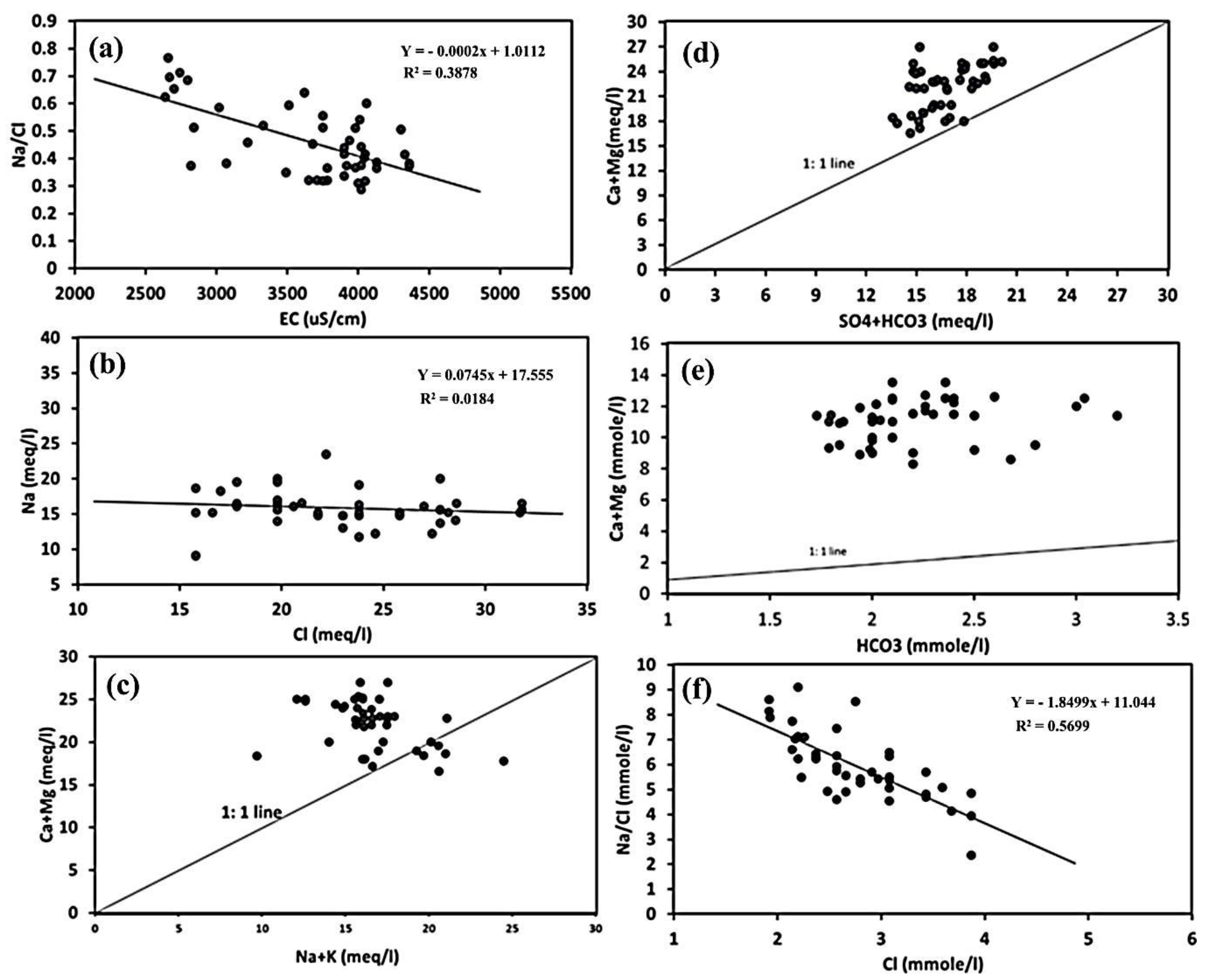
3.2. Groundwater Facies and Controlling Geochemical Processes
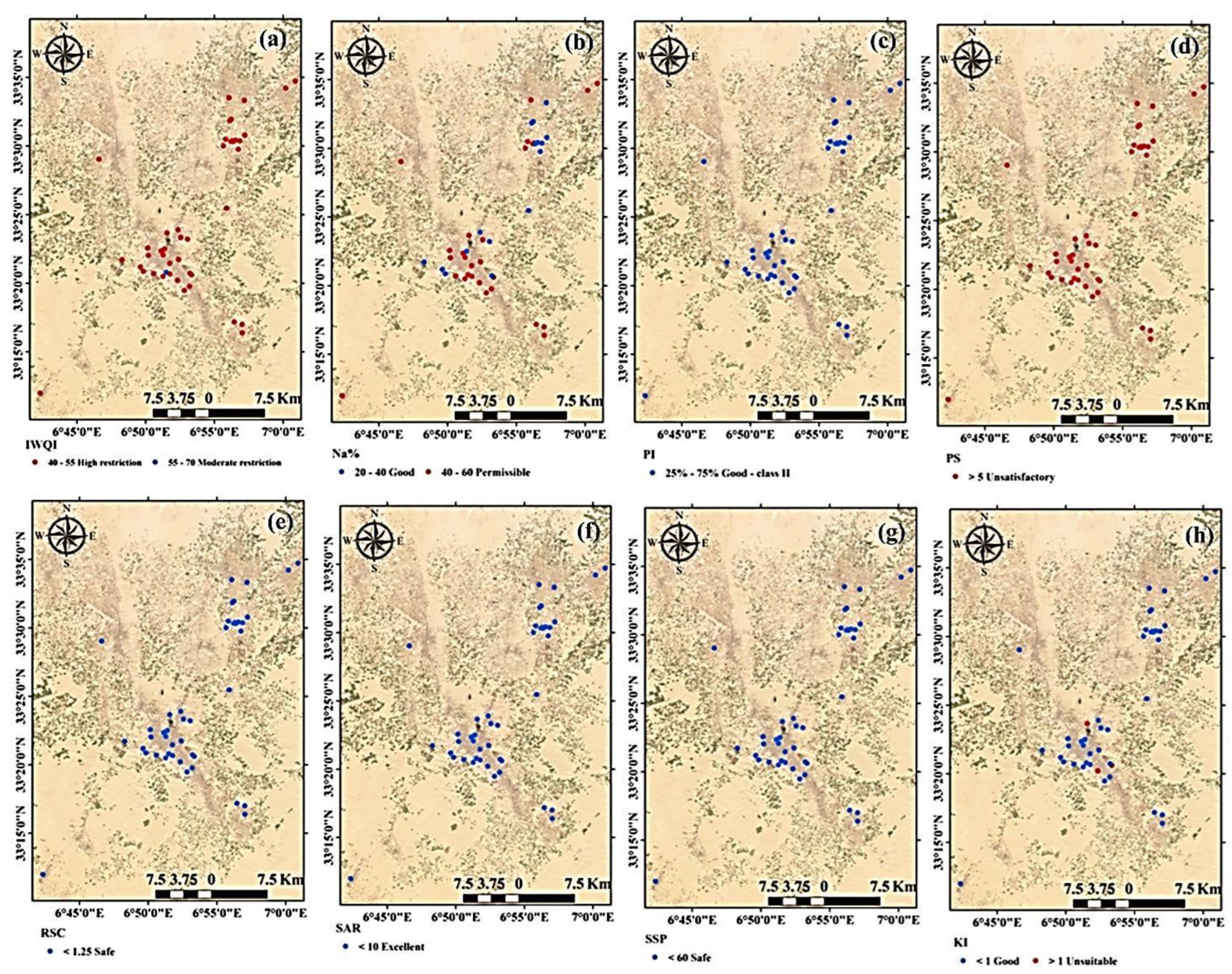
3.3. Water Quality Indices for Agricultural Purposes
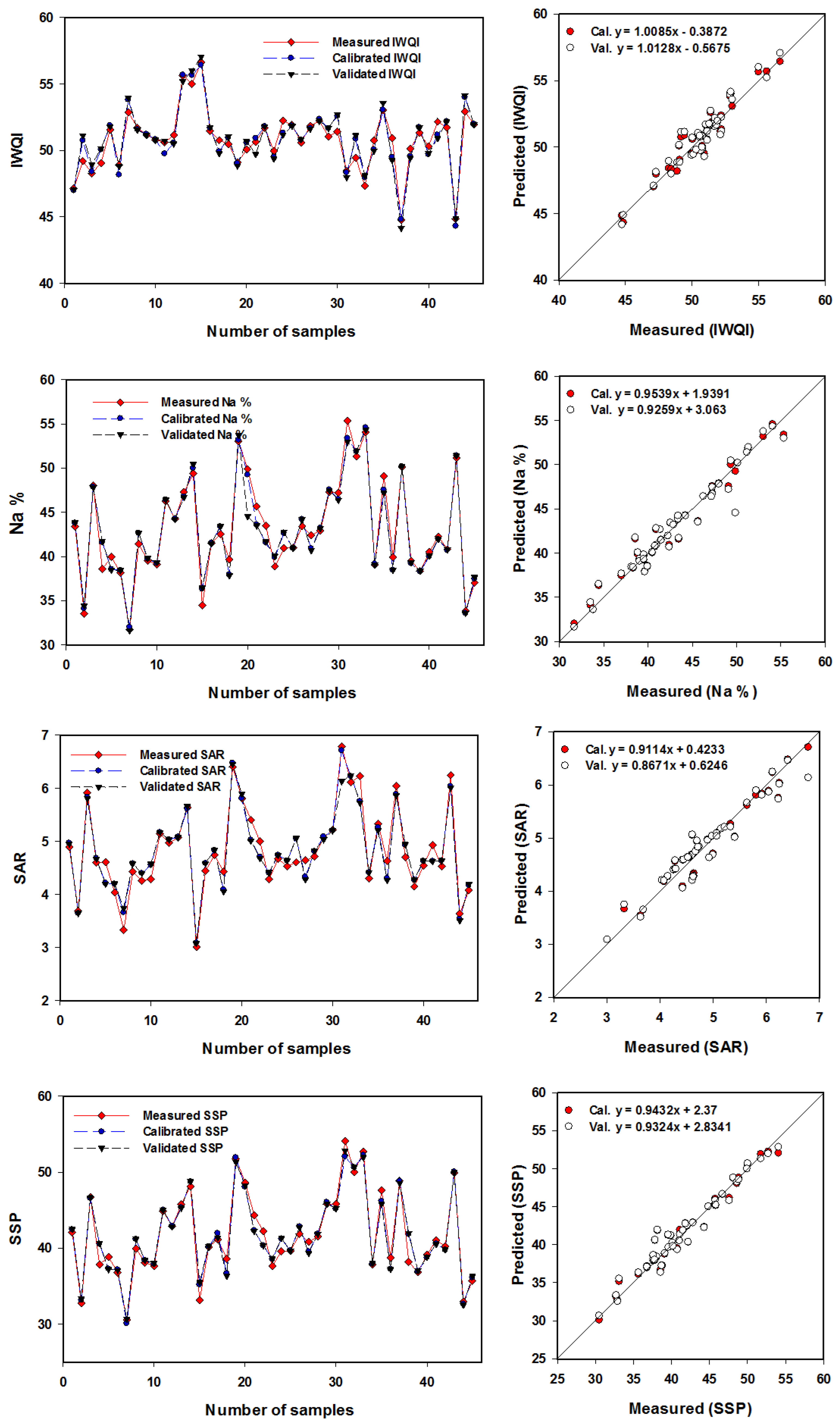
3.4. Irrigation Water Quality Index (IWQI)
3.5. Sodium Percentage (Na%)
3.6. Sodium Adsorption Ratio (SAR)
3.7. Soluble Sodium Percentage (SSP)
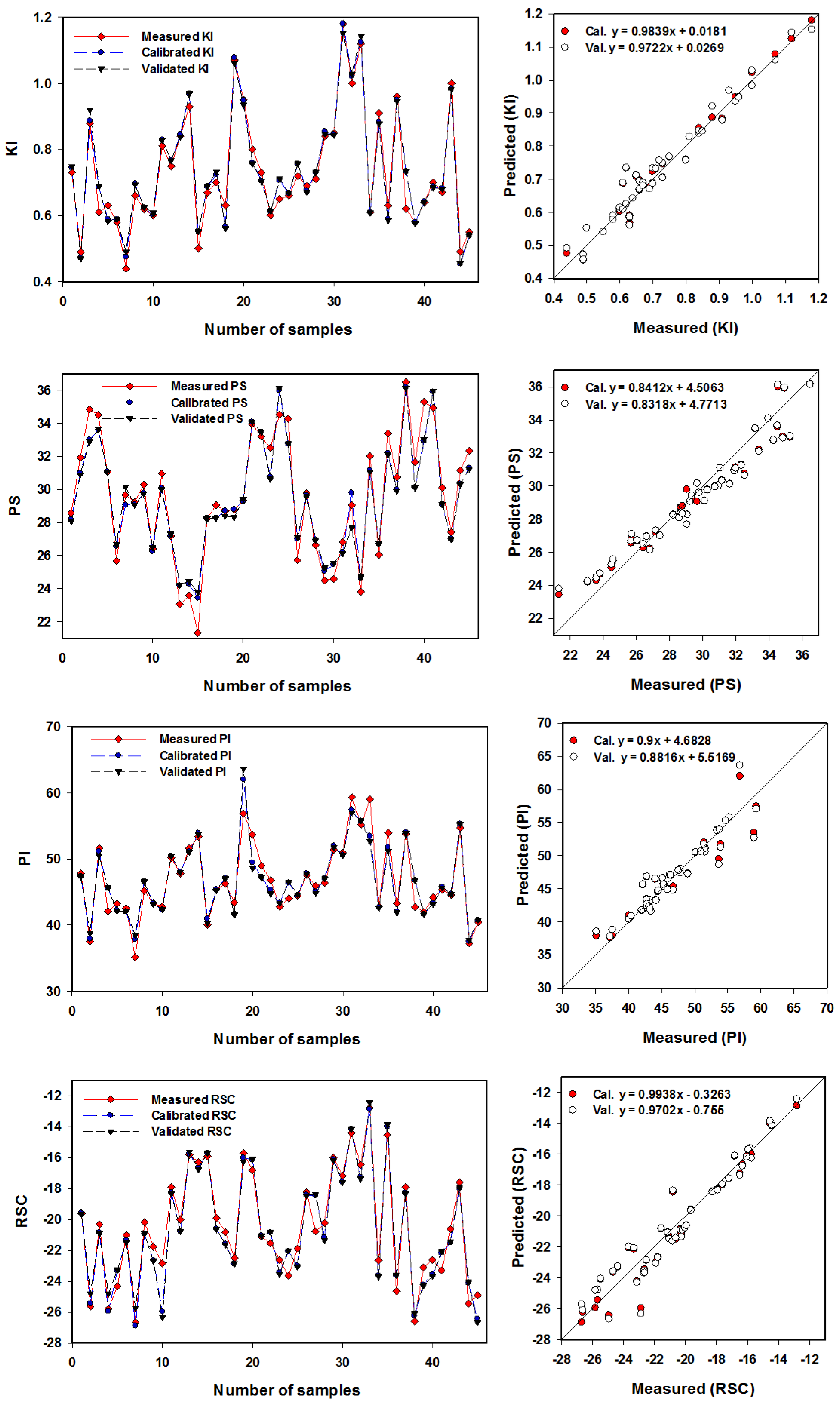
3.8. Kelly Index (KI)
3.9. Potential Salinity (PS)
3.10. Permeability Index (PI)
3.11. Residual Sodium Carbonate (RSC)
3.12. Performance of the Support Vector Machine Regression Based on Physicochemical Parameters for Predicting the Irrigation Water Quality Indices
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siebert, S.; Burke, J.; Faures, J.M.; Frenken, K.; Hoogeveen, J.; Döll, P.; Portmann, F.T. Groundwater Use for Irrigation—A Global Inventory. Hydrol. Earth Syst. Sci. 2010, 14, 1863–1880. [Google Scholar] [CrossRef] [Green Version]
- Baghvand, A.; Nasrabadi, T.; Bidhendi, G.N.; Vosoogh, A.; Karbassi, A.; Mehrdadi, N. Groundwater Quality Degradation of an Aquifer in Iran Central Desert. Desalination 2010, 260, 264–275. [Google Scholar] [CrossRef]
- Burri, N.M.; Weatherl, R.; Moeck, C.; Schirmer, M. A Review of Threats to Groundwater Quality in the Anthropocene. Sci. Total Environ. 2019, 684, 136–154. [Google Scholar] [CrossRef]
- El Asri, H.; Larabi, A.; Faouzi, M. Climate Change Projections in the Ghis-Nekkor Region of Morocco and Potential Impact on Groundwater Recharge. Appl Clim. 2019, 138, 713–727. [Google Scholar] [CrossRef]
- Houéménou, H.; Tweed, S.; Dobigny, G.; Mama, D.; Alassane, A.; Silmer, R.; Babic, M.; Ruy, S.; Chaigneau, A.; Gauthier, P.; et al. Degradation of Groundwater Quality in Expanding Cities in West Africa. A Case Study of the Unregulated Shallow Aquifer in Cotonou. J. Hydrol. 2020, 582, 124438. [Google Scholar] [CrossRef]
- Mountadar, S.; Younsi, A.; Hayani, A.; Siniti, M.; Tahiri, S. Groundwater Salinization Process in the Coastal Aquifer Sidi Abed-Ouled Ghanem (Province of El Jadida, Morocco). J. Afr. Earth Sci. 2018, 147, 169–177. [Google Scholar] [CrossRef]
- Lezzaik, K.; Milewski, A.; Mullen, J. The Groundwater Risk Index: Development and Application in the Middle East and North Africa Region. Sci. Total Environ. 2018, 628–629, 1149–1164. [Google Scholar] [CrossRef]
- Besbes, M.; Abdous, B.; Abidi, B.; Ayed, A.; Bachta, M.; Babasy, M.; Ben Baccar, B.; El Batti, D.; Ben Salah, Y.; Biet Charreton, M.; et al. Système Aquifère Du Sahara Septentrional Gestion Commune d’un Bassin Transfrontière. La Houille Blanche 2003, 89, 128–133. [Google Scholar] [CrossRef]
- Bouselsal, B.; Zeddouri, A.; Belksier, M.; Fenazi, B. Contribution de La Méthode de Vulnérabilité Intrinsèque GOD à l’Etude de La Pollution de La Nappe Libre d’Ouargla (SE Algérie). Int. J. Environ. Glob. Clim. Change 2015, 3, 92–99. [Google Scholar]
- Valley, O.-S.; Khechana, S.; Derradji, F.; Mega, N. Caractéristiques Hydrochimiques Des Eaux De La Nappe Phréatique Du Vallée d’Oued-Souf (SE Algérien). Eur. J. Sci. Res. 2011, 62, 207–215. [Google Scholar]
- Bouselsal, B.; Kherici, N. Effets de La Remontée Des Eaux de La Nappe Phréatique Sur l’homme et l’environnement: Cas de La Région d’El-Oued (SE Algérie). Afr. Sci. Rev. Int. Des Sci. Et Technol. 2014, 10, 161–170. [Google Scholar]
- ALLIA, Z. Le Système Aquifère Mio-Pliocène Du Bassin de Chott Melrhir-Zab Chergui: Caractérisation, Géométrie et Hydrochimie. Ph.D. Thesis, Université Mohamed Khider–Biskra, Biskra, Algeria, 2018. [Google Scholar]
- Saibi, H.; Mesbah, M.; Moulla, A.S.; Guendouz, A.H.; Ehara, S. Principal Component, Chemical, Bacteriological, and Isotopic Analyses of Oued-Souf Groundwaters (Revised). Env. Earth Sci 2016, 75, 272. [Google Scholar] [CrossRef]
- Zaiz, I.; Zine, B.; Boutoutaou, D.; Khechana, S. Contribution to the Study of the Quality Physicochemical of the Waters of the Water of the Complex Terminal in the Valley of Oued Souf (South-East Algerian). J. Fundam. Appl. Sci. 2017, 9, 1559. [Google Scholar] [CrossRef]
- Alqarawy, A.; El Osta, M.; Masoud, M.; Elsayed, S.; Gad, M. Use of Hyperspectral Reflectance and Water Quality Indices to AssessGroundwater Quality for Drinking in Arid Regions, Saudi Arabia. Water 2022, 14, 2311. [Google Scholar] [CrossRef]
- Moulla, A.S.; Guendouz, A.; Cherchali, M.E.-H.; Chaid, Z.; Ouarezki, S. Updated Geochemical and Isotopic Data from the Continental Intercalaire Aquifer in the Great Occidental Erg Sub-Basin (South-Western Algeria). Quat. Int. 2012, 257, 64–73. [Google Scholar] [CrossRef]
- Guendouz, A.; Moulla, A.S.; Edmunds, W.M.; Zouari, K.; Shand, P.; Mamou, A. Hydrogeochemical and Isotopic Evolution of Water in the Complexe Terminal Aquifer in the Algerian Sahara. Hydrogeol. J. 2003, 11, 483–495. [Google Scholar] [CrossRef]
- Paix, P. Les Nappes Artésiennes de l’Oued Rhir (Artesian Aquifers of the Wadi Rhir Valley). Ph.D. Thesis, Algiers University, Algiers, Algeria, 1956. [Google Scholar]
- Cornet, A. Introduction à l’hydrogéologie Saharienne. Géog. Phys. Et Géol. Dyn 1964, 6, 5–72. [Google Scholar]
- Noori, R.; Maghrebi, M.; Mirchi, A.; Tang, Q.; Bhattarai, R.; Sadegh, M.; Noury, M.; Torabi Haghighi, A.; Kløve, B.; Madani, K. Anthropogenic Depletion of Iran’s Aquifers. Proc. Natl. Acad. Sci. USA 2021, 118, e2024221118. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Ghosh, N.C.; Gurjar, S.; Krishan, G.; Kumar, S.; Berwal, P. Index-Based Assessment of Suitability of Water Quality for Irrigation Purpose under Indian Conditions. Env. Monit Assess 2018, 190, 29. [Google Scholar] [CrossRef]
- Regional Salinity Laboratory (US). Diagnosis and Improvement of Saline and Alkali Soils; US Department of Agriculture: Washington, DC, USA, 1954. Available online: https://acsess.onlinelibrary.wiley.com/doi/abs/10.2136/sssaj1954.03615995001800030032x (accessed on 15 November 2022).
- Wilcox, L.V. The Quality of Water for Irrigation Use; US Department of Agriculture: Washington, DC, USA, 1948. Available online: https://www.scirp.org/(S(i43dyn45teexjx455qlt3d2q))/reference/ReferencesPapers.aspx?ReferenceID=54035 (accessed on 15 November 2022).
- Doneen, L.D. Water Quality for Agriculture; Department of Irrigation, University of California: Davis, CA, USA, 1964; 48p, Available online: https://www.scirp.org/(S(vtj3fa45qm1ean45vvffcz55))/reference/ReferencesPapers.aspx?ReferenceID=1742078 (accessed on 15 November 2022).
- Gad, M.; Abou El-Safa, M.M.; Farouk, M.; Hussein, H.; Alnemari, A.M.; Elsayed, S.; Khalifa, M.M.; Moghanm, F.S.; Eid, E.M.; Saleh, A.H. Integration of Water Quality Indices and Multivariate Modeling for Assessing Surface Water Quality in Qaroun Lake, Egypt. Water 2021, 13, 2258. [Google Scholar] [CrossRef]
- El Osta, M.; Masoud, M.; Alqarawy, A.; Elsayed, S.; Gad, M. Groundwater Suitability for Drinking and Irrigation Using Water Quality Indices and Multivariate Modeling in Makkah Al-Mukarramah Province, Saudi Arabia. Water 2022, 14, 483. [Google Scholar] [CrossRef]
- Gad, M.; Saleh, A.H.; Hussein, H.; Farouk, M.; Elsayed, S. Appraisal of Surface Water Quality of Nile River Using Water Quality Indices, Spectral Signature and Multivariate Modeling. Water 2022, 14, 1131. [Google Scholar] [CrossRef]
- Ocampo-Duque, W.; Osorio, C.; Piamba, C.; Schuhmacher, M.; Domingo, J.L. Water Quality Analysis in Rivers with Non-Parametric Probability Distributions and Fuzzy Inference Systems: Application to the Cauca River, Colombia. Environ. Int. 2013, 52, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Sutadian, A.D.; Muttil, N.; Yilmaz, A.G.; Perera, B.J.C. Development of River Water Quality Indices—A Review. Env. Monit Assess 2016, 188, 58. [Google Scholar] [CrossRef] [PubMed]
- Ayers, R.; Westcott, D. Water Quality for Agriculture; FAO Irrigation and Drainage Paper 29 Rev. 1; Food and Agricultural Organisation of the United Nations: California, USA, 1994; Available online: https://www.fao.org/3/t0234e/t0234e00.htm (accessed on 15 November 2022).
- Meireles, A.C.M.; de Andrade, E.M.; Chaves, L.C.G.; Frischkorn, H.; Crisostomo, L.A. A New Proposal of the Classification of Irrigation Water. Rev. Ciênc. Agron. 2010, 41, 349–357. [Google Scholar] [CrossRef] [Green Version]
- Todd, D.K.; Mays, L.W. Groundwater Hydrology; John Wiley & Sons: Hoboken, NJ, USA, 2004; ISBN 0-471-05937-4. [Google Scholar]
- Richards, L.A. Diagnosis and Improvement of Saline and Alkali Soils; LWW: Washington, DC, USA, 1954; Volume 78, ISBN 0038-075X. Available online: https://www.ars.usda.gov/ARSUserFiles/20360500/hb60_pdf/hb60complete.pdf (accessed on 15 November 2022).
- Kelley, W.P. Permissible Composition and Concentration of Irrigation Water. In Proceedings of the American society of civil engineers; 1940; Volume 66, pp. 607–613. Available online: https://www.scirp.org/(S(351jmbntvnsjt1aadkposzje))/reference/ReferencesPapers.aspx?ReferenceID=2517629 (accessed on 15 November 2022).
- Eaton, F.M. Significance of Carbonates in Irrigation Waters. Soil Sci. 1950, 69, 123–134. [Google Scholar] [CrossRef]
- Ahmed, M.T.; Hasan, M.Y.; Monir, M.U.; Samad, M.A.; Rahman, M.M.; Islam Rifat, M.S.; Islam, M.N.; Khan, A.A.S.; Biswas, P.K.; Jamil, A.H.M.N. Evaluation of Hydrochemical Properties and Groundwater Suitability for Irrigation Uses in Southwestern Zones of Jashore, Bangladesh. Groundw. Sustain. Dev. 2020, 11, 100441. [Google Scholar] [CrossRef]
- Bhunia, G.S.; Keshavarzi, A.; Shit, P.K.; Omran, E.-S.E.; Bagherzadeh, A. Evaluation of Groundwater Quality and Its Suitability for Drinking and Irrigation Using GIS and Geostatistics Techniques in Semiarid Region of Neyshabur, Iran. Appl. Water Sci. 2018, 8, 168. [Google Scholar] [CrossRef] [Green Version]
- Thapa, R.; Gupta, S.; Reddy, D.V.; Kaur, H. An Evaluation of Irrigation Water Suitability in the Dwarka River Basin through the Use of GIS-Based Modelling. Env. Earth Sci. 2017, 76, 471. [Google Scholar] [CrossRef]
- Masoud, M.; El Osta, M.; Alqarawy, A.; Elsayed, S.; Gad, M. Evaluation of groundwater quality for agricultural under different conditions using water quality indices, partial least squares regression models, and GIS approaches. Appl. Water Sci. 2022, 12, 244. [Google Scholar] [CrossRef]
- Noori, R.; Ghiasi, B.; Salehi, S.; Esmaeili Bidhendi, M.; Raeisi, A.; Partani, S.; Meysami, R.; Mahdian, M.; Hosseinzadeh, M.; Abolfathi, S. An Efficient Data Driven-Based Model for Prediction of the Total Sediment Load in Rivers. Hydrology 2022, 9, 36. [Google Scholar] [CrossRef]
- Sarkar, A.; Pandey, P. River Water Quality Modelling Using Artificial Neural Network Technique. Aquat. Procedia 2015, 4, 1070–1077. [Google Scholar] [CrossRef]
- Noori, R.; Karbassi, A.R.; Mehdizadeh, H.; Vesali-Naseh, M.; Sabahi, M.S. A Framework Development for Predicting the Longitudinal Dispersion Coefficient in Natural Streams Using an Artificial Neural Network. Environ. Prog. Sustain. Energy 2011, 30, 439–449. [Google Scholar] [CrossRef]
- Boulifa, K. Synthèse Hydrogéologique Sur La Region d’El-Oued Sahara Nord Oriental–Est Algérien. Magister en Géologie Option Hydrogéologie, Algérie 2012. Available online: http://www.secheresse.info/spip.php?article80362 (accessed on 15 November 2022).
- Bouselsal, B. Etude Hydrogéologique et Hydrochimique de l’aquifère Libre d’El Oued Souf (SE Algérie). Th. Dr. Univ. D’annaba. 2016. Available online: https://biblio.univ-annaba.dz/wp-content/uploads/2019/07/These-Bouselsal-Boualem.pdf (accessed on 15 November 2022).
- Busson, G. Le Mesozoique Sarahien, Deuxieme Partie: Essai de Syntheses Des Donnees de Sondages Algero-Tuniiens, Centre de Recherche Sur Le Zones Arides. Ser. Geol. 1970. Available online: https://www.abebooks.com/M%C3%A9sozo%C3%AFque-Saharien-essai-synth%C3%A8se-donn%C3%A9es-sondages/1418091373/bd (accessed on 15 November 2022).
- Chebbah, M.; Allia, Z. Geochemistry and Hydrogeochemical Process of Groundwater in the Souf Valley of Low Septentrional Sahara, Algeria. Afr. J. Environ. Sci. Technol. 2015, 9, 261–273. [Google Scholar] [CrossRef] [Green Version]
- Gonçalvès, J.; Petersen, J.; Deschamps, P.; Hamelin, B.; Baba-Sy, O. Quantifying the Modern Recharge of the “Fossil” Sahara Aquifers. Geophys. Res. Lett. 2013, 40, 2673–2678. [Google Scholar] [CrossRef]
- Domenico, P.A.; Schwartz, F.W. Physical and Chemical Hydrogeology; Wiley: New York, NY, USA, 1998; Volume 506. [Google Scholar]
- Abbasnia, A.; Yousefi, N.; Mahvi, A.H.; Nabizadeh, R.; Radfard, M.; Yousefi, M.; Alimohammadi, M. Evaluation of Groundwater Quality Using Water Quality Index and Its Suitability for Assessing Water for Drinking and Irrigation Purposes: Case Study of Sistan and Baluchistan Province (Iran). Hum. Ecol. Risk Assess. Int. J. 2019, 25, 988–1005. [Google Scholar] [CrossRef]
- Piper, A.M. A Graphic Procedure in the Geochemical Interpretation of Water-Analyses. Trans. AGU 1944, 25, 914. [Google Scholar] [CrossRef]
- Gibbs, R.J. Mechanisms Controlling World Water Chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef]
- Vasanthavigar, M.; Srinivasamoorthy, K.; Prasanna, M.V. Evaluation of Groundwater Suitability for Domestic, Irrigational, and Industrial Purposes: A Case Study from Thirumanimuttar River Basin, Tamilnadu, India. Env. Monit Assess 2012, 184, 405–420. [Google Scholar] [CrossRef]
- Freeze, R.A.; Cherry, J. Groundwater; Prentice-Hall Inc.: Englewood Cliffs, NJ, USA, 1979; Available online: https://www.scirp.org/(S(lz5mqp453edsnp55rrgjct55))/reference/ReferencesPapers.aspx?ReferenceID=1894372 (accessed on 15 November 2022).
- Antonakos, A.; Lambrakis, N. Hydrodynamic Characteristics and Nitrate Propagation in Sparta Aquifer. Water Res. 2000, 34, 3977–3986. [Google Scholar] [CrossRef]
- Adimalla, N. Spatial Distribution, Exposure, and Potential Health Risk Assessment from Nitrate in Drinking Water from Semi-Arid Region of South India. Hum. Ecol. Risk Assess. Int. J. 2020, 26, 310–334. [Google Scholar] [CrossRef]
- Chadha, D.K. A Proposed New Diagram for Geochemical Classification of Natural Waters and Interpretation of Chemical Data. Hydrogeol. J. 1999, 7, 431–439. [Google Scholar] [CrossRef]
- Durov, S.A. Natural Waters and Graphic Representation of Their Composition. In Proceedings of the Dokl Akad Nauk SSSR; 1948; Volume 59, pp. 87–90. Available online: https://www.scirp.org/(S(lz5mqp453edsnp55rrgjct55))/reference/ReferencesPapers.aspx?ReferenceID=1742074 (accessed on 15 November 2022).
- Fisher, R.S.; Mullican, W.F., III. Hydrochemical Evolution of Sodium-Sulfate and Sodium-Chloride Groundwater Beneath the Northern Chihuahuan Desert, Trans-Pecos, Texas, USA. HYJO 1997, 5, 4–16. [Google Scholar] [CrossRef]
- Srinivasamoorthy, K.; Chidambaram, S.; Prasanna, M.V.; Vasanthavihar, M.; Peter, J.; Anandhan, P. Identification of Major Sources Controlling Groundwater Chemistry from a Hard Rock Terrain—A Case Study from Mettur Taluk, Salem District, Tamil Nadu, India. J. Earth Syst. Sci. 2008, 117, 49–58. [Google Scholar] [CrossRef]
- Jacks, G.; Sefe, F.; Carling, M.; Hammar, M.; Letsamao, P. Tentative Nitrogen Budget for Pit Latrines-Eastern Botswana. Environ. Geol. 1999, 38, 199–203. [Google Scholar] [CrossRef]
- Biswas, A.; Nath, B.; Bhattacharya, P.; Halder, D.; Kundu, A.K.; Mandal, U.; Mukherjee, A.; Chatterjee, D.; Mörth, C.-M.; Jacks, G. Hydrogeochemical Contrast between Brown and Grey Sand Aquifers in Shallow Depth of Bengal Basin: Consequences for Sustainable Drinking Water Supply. Sci. Total Environ. 2012, 431, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Meybeck, M. Global Chemical Weathering of Surficial Rocks Estimated from River Dissolved Loads. Am. J. Sci. 1987, 287, 401–428. [Google Scholar] [CrossRef]
- Jankowski, J.; Acworth, R.I. Impact of Debris-Flow Deposits on Hydrogeochemical Processes and the Developement of Dryland Salinity in the Yass River Catchment, New South Wales, Australia. HYJO 1997, 5, 71–88. [Google Scholar] [CrossRef]
- Rajmohan, N.; Elango, L. Identification and Evolution of Hydrogeochemical Processes in the Groundwater Environment in an Area of the Palar and Cheyyar River Basins, Southern India. Environ. Geol. 2003, 1, 47–61. [Google Scholar] [CrossRef]
- Gad, M.; El Osta, M. Geochemical Controlling Mechanisms and Quality of the Groundwater Resources in El Fayoum Depression, Egypt. Arab. J. Geosci. 2020, 13, 861. [Google Scholar] [CrossRef]
- Ma, Q.; Ge, W.; Tian, F. Geochemical Characteristics and Controlling Factors of Chemical Composition of Groundwater in aPart of the Nanchang Section of Ganfu Plain. Sustainability 2022, 14, 7976. [Google Scholar] [CrossRef]
- Nazzal, Y.; Ahmed, I.; Al-Arifi, N.S.N.; Ghrefat, H.; Zaidi, F.K.; El-Waheidi, M.M.; Batayneh, A.; Zumlot, T. A Pragmatic Approach to Study the Groundwater Quality Suitability for Domestic and Agricultural Usage, Saq Aquifer, Northwest of Saudi Arabia. Env. Monit Assess 2014, 186, 4655–4667. [Google Scholar] [CrossRef]
- Gad, M.; El-Hendawy, S.; Al-Suhaibani, N.; Tahir, M.U.; Mubushar, M.; Elsayed, S. Combining Hydrogeochemical Characterization and a Hyperspectral Reflectance Tool for Assessing Quality and Suitability of Two Groundwater Resources for Irrigation in Egypt. Water 2020, 12, 2169. [Google Scholar] [CrossRef]
- Kaka, E.A.; Akiti, T.T.; Nartey, V.K.; Bam, E.K.P.; Adomako, D. Hydrochemistry and Evaluation of Groundwater Suitability for Irrigation and Drinking Purposes in the Southeastern Volta River Basin: Manyakrobo Area, Ghana. Elixir Agric. 2011, 39, 4793–4807. [Google Scholar]
- Kawo, N.S.; Karuppannan, S. Groundwater Quality Assessment Using Water Quality Index and GIS Technique in Modjo River Basin, Central Ethiopia. J. Afr. Earth Sci. 2018, 147, 300–311. [Google Scholar] [CrossRef]
- Li, P.; Wu, J.; Qian, H. Assessment of Groundwater Quality for Irrigation Purposes and Identification of Hydrogeochemical Evolution Mechanisms in Pengyang County, China. Env. Earth Sci. 2013, 69, 2211–2225. [Google Scholar] [CrossRef]
- RamyaPriya, R.; Elango, L. Evaluation of Geogenic and Anthropogenic Impacts on Spatio-Temporal Variation in Quality of Surface Water and Groundwater along Cauvery River, India. Env. Earth Sci. 2018, 77, 2. [Google Scholar] [CrossRef]
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; FAO Irrigation and Drainage Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 1985; ISBN 978-92-5-102263-4. [Google Scholar]
- Saleh, A.; Al-Ruwaih, F.; Shehata, M. Hydrogeochemical Processes Operating within the Main Aquifers of Kuwait. J. Arid Environ. 1999, 42, 195–209. [Google Scholar] [CrossRef]
- Wang, X.; Ozdemir, O.; Hampton, M.A.; Nguyen, A.V.; Do, D.D. The Effect of Zeolite Treatment by Acids on Sodium Adsorption Ratio of Coal Seam Gas Water. Water Res. 2012, 46, 5247–5254. [Google Scholar] [CrossRef]
- Hanson, B.; Grattan, S.R.; Fulton, A. Agricultural Salinity and Drainage; University of California Irrigation Program, University of California, Davis: Davis, CA, USA, 1999. [Google Scholar]
- Bhat, M.A.; Grewal, M.S.; Rajpaul, R.; Wani, S.A.; Dar, E.A. Assessment of Groundwater Quality for Irrigation Purposes Using Chemical Indices. Indian J. Ecol. 2016, 43, 574–579. [Google Scholar]
- Sudhakar, A.; Narsimha, A. Suitability and Assessment of Groundwater for Irrigation Purpose: A Case Study of Kushaiguda Area, Ranga Reddy District, Andhra Pradesh, India. Adv. Appl. Sci. Res. 2013, 4, 75–81. [Google Scholar]
- Sundaray, S.K.; Nayak, B.B.; Bhatta, D. Environmental Studies on River Water Quality with Reference to Suitability for Agricultural Purposes: Mahanadi River Estuarine System, India—A Case Study. Env. Monit Assess 2009, 155, 227–243. [Google Scholar] [CrossRef]
- Srinivasamoorthy, K.; Gopinath, M.; Chidambaram, S.; Vasanthavigar, M.; Sarma, V.S. Hydrochemical Characterization and Quality Appraisal of Groundwater from Pungar Sub Basin, Tamilnadu, India. J. King Saud Univ.-Sci. 2014, 26, 37–52. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.; Kumari, K.; Ramanathan, A.; Saxena, R. A Comparative Evaluation of Groundwater Suitability for Irrigation and Drinking Purposes in Two Intensively Cultivated Districts of Punjab, India. Environ. Geol. 2007, 53, 553–574. [Google Scholar] [CrossRef]
- Prasad, A.; Kumar, D.; Singh, D.V. Effect of Residual Sodium Carbonate in Irrigation Water on the Soil Sodication and Yield of Palmarosa (Cymbopogon Martinni) and Lemongrass (Cymbopogon Flexuosus). Agric. Water Manag. 2001, 50, 161–172. [Google Scholar] [CrossRef]
- He, S.; Wu, J. Relationships of Groundwater Quality and Associated Health Risks with Land Use/Land Cover Patterns: A Case Study in a Loess Area, Northwest China. Hum. Ecol. Risk Assess. Int. J. 2019, 25, 354–373. [Google Scholar] [CrossRef]
- Tiyasha; Tung, T.M.; Yaseen, Z.M. A Survey on River Water Quality Modelling Using Artificial Intelligence Models: 2000–2020. J. Hydrol. 2020, 585, 124670. [Google Scholar] [CrossRef]
- Beltran, N.H.; Duarte-Mermoud, M.A.; Soto Vicencio, V.A.; Salah, S.A.; Bustos, M.A. Chilean Wine Classification Using Volatile Organic Compounds Data Obtained With a Fast GC Analyzer. IEEE Trans. Instrum. Meas. 2008, 57, 2421–2436. [Google Scholar] [CrossRef] [Green Version]
- Guyon, I.; Elisseeff, A. An Introduction to Variable and Feature Selection. J. Mach. Learn. Res. 2003, 3, 1157–1182. [Google Scholar]
- Schulze, F.H.; Wolf, H.; Jansen, H.W.; van der Veer, P. Applications of Artificial Neural Networks in Integrated Water Management: Fiction or Future? Water Sci. Technol. 2005, 52, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, S.; Hussein, H.; Moghanm, F.S.; Khedher, K.M.; Eid, E.M.; Gad, M. Application of Irrigation Water Quality Indices and Multivariate Statistical Techniques for Surface Water Quality Assessments in the Northern Nile Delta, Egypt. Water 2020, 12, 3300. [Google Scholar] [CrossRef]
- Ahmed, U.; Mumtaz, R.; Anwar, H.; Shah, A.A.; Irfan, R.; García-Nieto, J. Efficient water quality prediction using supervised machine learning. Water 2019, 11, 2210. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.B.; Liu, W.C. Water quality modeling in reservoirs using multivariate linear regression and two neural network models. Adv. Artif. Neural Syst. 2015, 2015, 521721. [Google Scholar] [CrossRef]

| Index | Formula | Reference |
|---|---|---|
| IWQI | [45] | |
| Na% | [(Na2+ + K+)/(Ca2+ + Mg2+ + Na2+ + K+)] × 100 | [46] |
| SAR | ) × 100 | [47] |
| SSP | [Na2+/(Ca2+ + Mg2+ + Na2+)] × 100 | [46] |
| KI | KI = Na+/(Ca2+ + Mg2+) | [48] |
| PS | Cl− + (SO42−/2) | [49] |
| PI | [(Na2+ +√ HCO3−)/(Ca2+ + Mg2+ + Na2+)] × 100 | [49] |
| RSC | (HCO32− + CO3−)-(Ca2+ + Mg2+) | [50] |
| Qi | EC (μs/cm) | SAR | Na+ (emp) | Cl− (emp) | HCO32− (epm) |
|---|---|---|---|---|---|
| 85–100 | 200 ≤ EC < 750 | 2 ≤ EC < 3 | 2 ≤ Na < 3 | 1 ≤ Cl < 4 | 1 ≤ HCO3 < 1.5 |
| 60–85 | 750 ≤ EC < 1500 | 3 ≤ EC < 6 | 3 ≤ Na < 6 | 4 ≤ Cl < 7 | 1.5 ≤ HCO3 < 4.5 |
| 35–60 | 1500 ≤ EC < 3000 | 6 ≤ EC < 12 | 6 ≤ Na < 9 | 7 ≤ Cl < 10 | 4.5 ≤ HCO3 < 8.5 |
| 0–35 | EC < 200 or EC ≥ 3000 | SAR > 2 or SAR ≥ 12 | Na < 2 or SAR ≥ 9 | Cl < 1 or Cl ≥ 10 | HCO3 < 1 or HCO3 ≥ 8.5 |
| Parameter | T °C | pH | EC | TDS | K+ | Na+ | Mg2+ | Ca2+ | Cl− | SO42− | HCO3− | CO32− | NO3− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Deep Groundwater Aquifers, Algeria (n = 45) | |||||||||||||
| Min. | 13.9 | 7.0 | 2640 | 1702 | 12.00 | 210.00 | 24.30 | 168.33 | 560.15 | 532.20 | 105.80 | 0.00 | 0.83 |
| Max. | 38.8 | 7.8 | 4360 | 2790 | 42.00 | 540.00 | 157.90 | 340.68 | 1127.40 | 840.27 | 195.20 | 1.03 | 31.53 |
| Mean | 26.6 | 7.4 | 3646 | 2342 | 33.30 | 364.38 | 109.70 | 258.98 | 822.25 | 697.53 | 135.50 | 0.12 | 15.82 |
| SD | 5.6 | 0.2 | 524 | 337 | 7.67 | 56.51 | 28.52 | 43.80 | 158.73 | 82.25 | 20.79 | 0.21 | 10.66 |
| Parameters | IWQI | Na% | SAR | SSP | KI | PS | PI | RSC |
|---|---|---|---|---|---|---|---|---|
| Min. | 44.76 | 31.71 | 3.01 | 30.53 | 0.44 | 21.33 | 34.02 | −26.69 |
| Max. | 56.63 | 55.35 | 6.79 | 54.08 | 1.18 | 36.49 | 58.17 | −12.80 |
| Mean | 50.78 | 43.07 | 4.85 | 41.78 | 0.74 | 29.60 | 45.65 | −20.44 |
| SD | 2.28 | 5.62 | 0.82 | 5.56 | 0.17 | 3.73 | 5.68 | 3.58 |
| Index | Range | Water Category | Number of Samples (%) |
|---|---|---|---|
| IWQI | 0–40 | Severe restriction | 0 (0%) |
| 40–55 | High restriction | 43 (93.6%) | |
| 55–70 | Moderate restriction | 2 (4.4%) | |
| 70–85 | Low restriction | 0 (0.0%) | |
| 85–100 | No restriction | 0 (0.0%) | |
| Na% | 40–60 | Permissible | 29 (64.4%) |
| 20–40 | Good | 16 (35.6%) | |
| <20 | Excellent | 0 (0.0%) | |
| SAR | >26 | Unsuitable | 0 (0.0%) |
| 18–26 | Doubtful or fairly poor | 0 (0.0%) | |
| 10–18 | Good | 0 (0.0%) | |
| <10 | Excellent | 45 (100%) | |
| SSP | >60 | Unsafe | 0 (0.0%) |
| <60 | Safe | 45 (100%) | |
| KI | >1 | Unsuitable | 41 (91.2%) |
| <1 | Good | 4 (8.8%) | |
| PS | >5 | Injurious to unsatisfactory | 45 (100%) |
| 3–5 | Good to injurious | 0 (0.0%) | |
| <3 | Excellent to good | 0 (0.0%) | |
| PI | <25% | Unsuitable—Class III | 0 (0.0%) |
| 25–75% | Good—Class II | 45 (100%) | |
| >75% | Good—Class I | 0 (0.0%) | |
| RSC | >2.5 | Unsuitable | 0 (0.0%) |
| 1.25–2.5 | Marginal | 0 (0.0%) | |
| <1.25 | Safe | 45 (100.0%) |
| Variable | Calibration | Validation | ||
|---|---|---|---|---|
| R2 | RMSE | R2 | RMSE | |
| IWQI | 0.93 *** | 0.85 | 0.90 *** | 0.92 |
| Na% | 0.97 *** | 1.25 | 0.92 *** | 1.37 |
| SAR | 0.94 *** | 0.22 | 0.92 *** | 0.27 |
| SSP | 0.96 *** | 1.21 | 0.95 *** | 1.42 |
| KI | 0.97 *** | 0.04 | 0.96 *** | 0.05 |
| PS | 0.95 *** | 1.13 | 0.94 *** | 1.20 |
| PI | 0.90 *** | 1.79 | 0.88 *** | 2.09 |
| RSC | 0.94 *** | 1.02 | 0.92 *** | 1.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eid, M.H.; Elbagory, M.; Tamma, A.A.; Gad, M.; Elsayed, S.; Hussein, H.; Moghanm, F.S.; Omara, A.E.-D.; Kovács, A.; Péter, S. Evaluation of Groundwater Quality for Irrigation in Deep Aquifers Using Multiple Graphical and Indexing Approaches Supported with Machine Learning Models and GIS Techniques, Souf Valley, Algeria. Water 2023, 15, 182. https://doi.org/10.3390/w15010182
Eid MH, Elbagory M, Tamma AA, Gad M, Elsayed S, Hussein H, Moghanm FS, Omara AE-D, Kovács A, Péter S. Evaluation of Groundwater Quality for Irrigation in Deep Aquifers Using Multiple Graphical and Indexing Approaches Supported with Machine Learning Models and GIS Techniques, Souf Valley, Algeria. Water. 2023; 15(1):182. https://doi.org/10.3390/w15010182
Chicago/Turabian StyleEid, Mohamed Hamdy, Mohssen Elbagory, Ahmed A. Tamma, Mohamed Gad, Salah Elsayed, Hend Hussein, Farahat S. Moghanm, Alaa El-Dein Omara, Attila Kovács, and Szűcs Péter. 2023. "Evaluation of Groundwater Quality for Irrigation in Deep Aquifers Using Multiple Graphical and Indexing Approaches Supported with Machine Learning Models and GIS Techniques, Souf Valley, Algeria" Water 15, no. 1: 182. https://doi.org/10.3390/w15010182
APA StyleEid, M. H., Elbagory, M., Tamma, A. A., Gad, M., Elsayed, S., Hussein, H., Moghanm, F. S., Omara, A. E.-D., Kovács, A., & Péter, S. (2023). Evaluation of Groundwater Quality for Irrigation in Deep Aquifers Using Multiple Graphical and Indexing Approaches Supported with Machine Learning Models and GIS Techniques, Souf Valley, Algeria. Water, 15(1), 182. https://doi.org/10.3390/w15010182