The Effect of Mineral Ions Present in Tap Water on Photodegradation of Organic Pollutants: Future Perspectives
Abstract
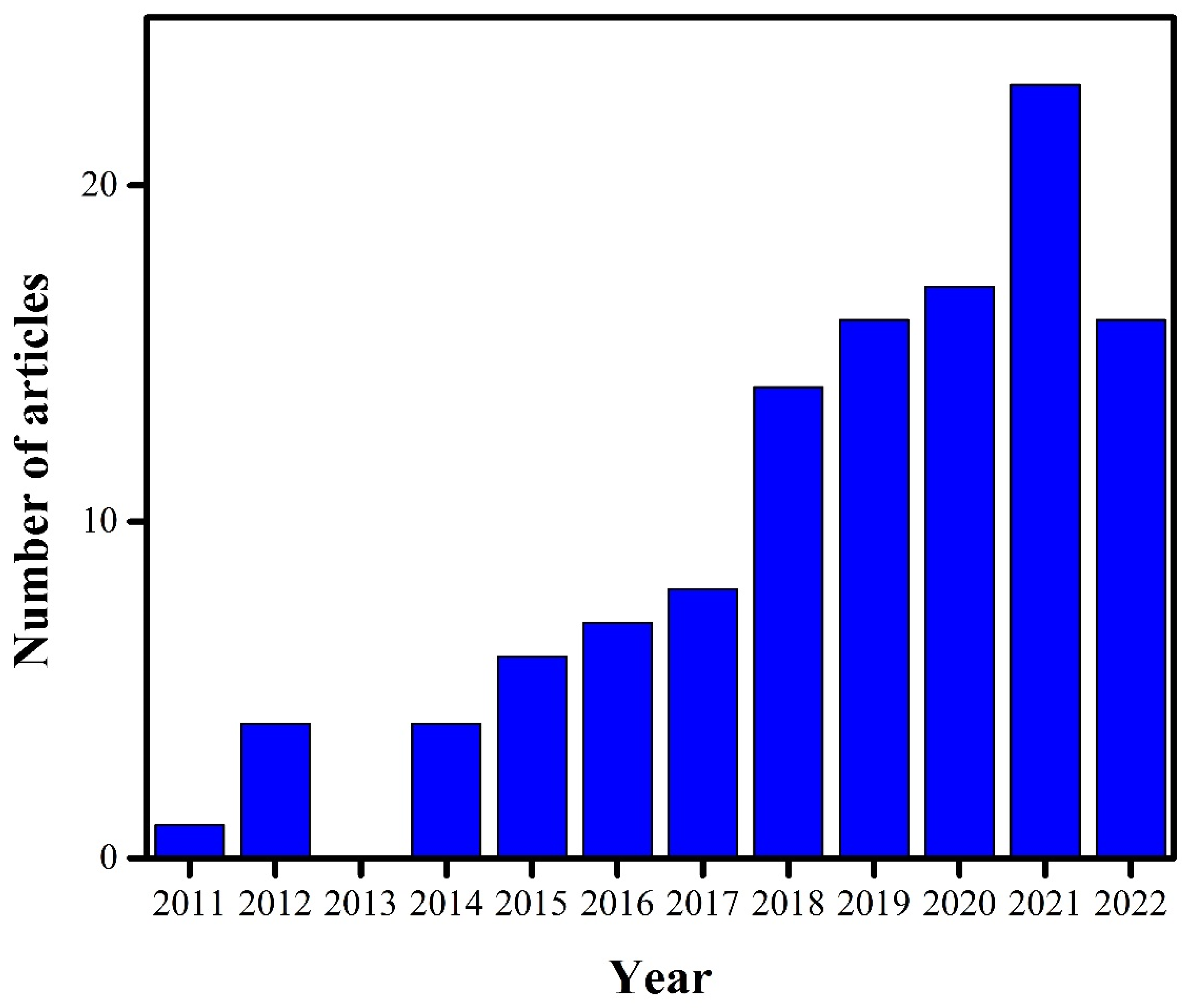
:1. Introduction
2. Effect of TW on Photodegradation of Dyes
3. Effect of TW on Photodegradation of Pharmaceutical Products
4. Effect of TW on Photodegradation of Pesticides
5. Effect of TW on Photodegradation of Organic Solvents
6. Future Perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Geetha, D.; Nagarajan, E.R. Impact and Issues of Organic Pollutants. In Management of Contaminants of Emerging Concern (CEC) in Environment; Singh, P., Hussain, C.M., Rajkhowa, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 93–126. [Google Scholar] [CrossRef]
- Mishra, D.; Srivastava, M. Low-dimensional nanomaterials for the photocatalytic degradation of organic pollutants. In Nano-Materials as Photocatalysts for Degradation of Environmental Pollutants; Elsevier: Amsterdam, The Netherlands, 2020; pp. 15–38. [Google Scholar] [CrossRef]
- You, X.; Zhou, R.; Zhu, Y.; Bu, D.; Cheng, D. Adsorption of dyes methyl violet and malachite green from aqueous solution on multi-step modified rice husk powder in single and binary systems: Characterization, adsorption behavior and physical interpretations. J. Hazard. Mater. 2022, 430, 128445. [Google Scholar] [CrossRef]
- Zhuo, S.N.; Ren, H.Y.; Liu, B.F. In situ utilization of biomass pretreatment liquor as a novel flocculant for anion dyes removal: Performance and mechanism. J. Hazard. Mater. 2022, 424, 127737. [Google Scholar] [CrossRef]
- Mcyotto, F.; Wei, Q.; Macharia, D.K.; Huang, M.; Shen, C.; Chow, C.W.K. Effect of dye structure on color removal efficiency by coagulation. Chem. Eng. J. 2021, 405, 126674. [Google Scholar] [CrossRef]
- Mohanty, S.S.; Kumar, A. Biodegradation of Indanthrene Blue RS dye in immobilized continuous upflow packed bed bioreactor using corncob biochar. Sci. Rep. 2021, 11, 13390. [Google Scholar] [CrossRef]
- Kaushal, J.; Mahajan, P. Kinetic Evaluation for Removal of an Anionic Diazo Direct Red 28 by Using Phytoremediation Potential of Salvinia molesta Mitchell. Bull. Environ. Contam. Toxicol. 2022, 108, 437–442. [Google Scholar] [CrossRef]
- Belal, R.M.; Zayed, M.A.; El-Sherif, R.M.; Ghany, N.A.A. Electrochemical Degradation and Degree of Mineralization of the BY28 Dye in a Supporting Electrolyte Mixture Using an Expanded Dimensionally Stable Anode. Electrocatalysis 2022, 13, 26–36. [Google Scholar] [CrossRef]
- Pourshirband, N.; Nezamzadeh-Ejhieh, A. A Z-scheme AgI/BiOI binary nanophotocatalyst for the Eriochrome Black T photodegradation: A scavenging agents study. Mater. Res. Bull. 2022, 148, 111689. [Google Scholar] [CrossRef]
- Rafiq, A.; Ikram, M.; Ali, S.; Niaz, F.; Khan, M.; Khan, Q.; Maqbool, M. Photocatalytic degradation of dyes using semiconductor photocatalysts to clean industrial water pollution. J. Ind. Eng. Chem. 2021, 97, 111–128. [Google Scholar] [CrossRef]
- Karim, A.V.; Hassani, A.; Eghbali, P.; Nidheesh, P.V. Nanostructured modified layered double hydroxides (LDHs)-based catalysts: A review on synthesis, characterization, and applications in water remediation by advanced oxidation processes. Curr. Opin. Solid State Mater. Sci. 2022, 26, 100965. [Google Scholar] [CrossRef]
- Lim, H.; Yusuf, M.; Song, S.; Park, S.; Park, K.H. Efficient photocatalytic degradation of dyes using photo-deposited Ag nanoparticles on ZnO structures: Simple morphological control of ZnO. RSC Adv. 2021, 11, 8709–8717. [Google Scholar] [CrossRef]
- Panwar, S.; Upadhyay, G.K.; Purohit, L.P. Gd-doped ZnO:TiO2 heterogenous nanocomposites for advance oxidation process. Mater. Res. Bull. 2022, 145, 111534. [Google Scholar] [CrossRef]
- Khan, N.A.; Saeed, K.; Khan, I.; Gul, T.; Sadiq, M.; Uddin, A.; Zekker, I. Efficient photodegradation of orange II dye by nickel oxide nanoparticles and nanoclay supported nickel oxide nanocomposite. Appl. Water Sci. 2022, 12, 131. [Google Scholar] [CrossRef]
- Benz, D.; Van Bui, H.; Hintzen, H.T.; Kreutzer, M.T.; van Ommen, J.R. Mechanistic insight into the improved photocatalytic degradation of dyes for an ultrathin coating of SiO2 on TiO2 (P25) nanoparticles. Chem. Eng. J. Adv. 2022, 10, 100288. [Google Scholar] [CrossRef]
- Jana, S.; Konar, S.; Mitra, B.C.; Mondal, A.; Mukhopadhyay, S. Fabrication of a new heterostructure Au/Pt/SnO2: An excellent catalyst for fast reduction of para-nitrophenol and visible light assisted photodegradation of dyes. Mater. Res. Bull. 2021, 141, 111351. [Google Scholar] [CrossRef]
- Saeed, M.; Muneer, M.; Haq, A.U.; Akram, N. Photocatalysis: An effective tool for photodegradation of dyes—A review. Environ. Sci. Pollut. Res. 2021, 29, 293–311. [Google Scholar] [CrossRef]
- Cui, Y.; Lin, C.; Li, M.; Zhu, N.; Meng, J.; Zhao, J. CuWO4/CuS heterojunction photocatalyst for the application of visible-light-driven photodegradation of dye pollutions. J. Alloys Compd. 2022, 893, 162181. [Google Scholar] [CrossRef]
- Yang, M.; Ren, D.; Sun, S.; Cui, J.; Yang, Q.; Luo, Y.; Liang, S. One-pot construction of unprecedented direct Z-scheme ZnS/GaOOH heterojunction for photodegradation of antibiotics. Appl. Surf. Sci. 2022, 576, 151742. [Google Scholar] [CrossRef]
- Rani, M.; Yadav, J.; Shanker, U. Green synthesis of sunlight responsive zinc oxide coupled cadmium sulfide nanostructures for efficient photodegradation of pesticides. J. Colloid Interface Sci. 2021, 601, 689–703. [Google Scholar] [CrossRef]
- Pham, T.H.; Jung, S.H.; Kim, T.Y. Enhanced photodegradation of toxic volatile organic pollutants using Ni-doped graphitic carbon nitride under natural solar light. Sol. Energy 2021, 224, 18–26. [Google Scholar] [CrossRef]
- Dharwadkar, S.; Yu, L.; Achari, G. Enhancement of LED based photocatalytic degradation of sulfolane by integration with oxidants and nanomaterials. Chemosphere 2021, 263, 128124. [Google Scholar] [CrossRef]
- McQueen, A.D.; Ballentine, M.L.; May, L.R.; Laber, C.H.; Das, A.; Bortner, M.J.; Kennedy, A.J. Photocatalytic Degradation of Polycyclic Aromatic Hydrocarbons in Water by 3D Printed TiO2Composites. ACS Environ. Sci. Technol. Water 2022, 2, 137–147. [Google Scholar] [CrossRef]
- Di Mauro, A.; Natile, M.M.; Landström, A.; Concina, I.; Ferroni, M.; Privitera, V.; Impellizzeri, G.; Epifani, M. Visible light photodegradation of dyes and paracetamol by direct sensitization mechanism onto metallic MoO2 nanocrystals. J. Photochem. Photobiol. A Chem. 2021, 413, 113258. [Google Scholar] [CrossRef]
- Senasu, T.; Chankhanittha, T.; Hemavibool, K.; Nanan, S. Visible-light-responsive photocatalyst based on ZnO/CdS nanocomposite for photodegradation of reactive red azo dye and ofloxacin antibiotic. Mater. Sci. Semicond. Process. 2021, 123, 105558. [Google Scholar] [CrossRef]
- Ghasemi, I.; Haghighi, M.; Talati, A.; Abbasi Asl, E. Facile sono-design of 3D flower-like NiO–CuFe2O4 nano-heterostructure as an efficient and magnetically separable catalyst for photodegradation of organic dyes. J. Clean. Prod. 2022, 335, 130355. [Google Scholar] [CrossRef]
- Asadzadeh-Khaneghah, S.; Habibi-Yangjeh, A.; Seifzadeh, D.; Chand, H.; Krishnan, V. Visible-light-activated g-C3N4 nanosheet/carbon dot/FeOCl nanocomposites: Photodegradation of dye pollutants and tetracycline hydrochloride. Colloids Surf. A Physicochem. Eng. Asp. 2021, 617, 126424. [Google Scholar] [CrossRef]
- Ejeta, S.Y.; Imae, T. Photodegradation of pollutant pesticide by oxidized graphitic carbon nitride catalysts. J. Photochem. Photobiol. A Chem. 2021, 404, 112955. [Google Scholar] [CrossRef]
- Tian, J.; Wei, L.; Ren, Z.; Lu, J.; Ma, J. The facile fabrication of Z-scheme Bi2WO6-P25 heterojunction with enhanced photodegradation of antibiotics under visible light. J. Environ. Chem. Eng. 2021, 9, 106167. [Google Scholar] [CrossRef]
- Madihi-Bidgoli, S.; Asadnezhad, S.; Yaghoot-Nezhad, A.; Hassani, A. Azurobine degradation using Fe2O3@multi-walled carbon nanotube activated peroxymonosulfate (PMS) under UVA-LED irradiation: Performance, mechanism and environmental application. J. Environ. Chem. Eng. 2021, 9, 106660. [Google Scholar] [CrossRef]
- Chen, Z.; Chu, X.; Huang, X.; Sun, H.; Chen, L.; Guo, F. Fabrication of visible-light driven CoP/ZnSnO3 composite photocatalyst for high-efficient photodegradation of antibiotic pollutant. Sep. Purif. Technol. 2021, 257, 117900. [Google Scholar] [CrossRef]
- Bharati, R.; Sundaramurthy, S.; Thakur, C. Nanomaterials and food-processing wastewater. Water Purif. 2017, 479–516. [Google Scholar] [CrossRef]
- Zada, N.; Saeed, K.; Khan, I. Decolorization of Rhodamine B dye by using multiwalled carbon nanotubes/Co–Ti oxides nanocomposite and Co–Ti oxides as photocatalysts. Appl. Water Sci. 2020, 10, 40. [Google Scholar] [CrossRef] [Green Version]
- Hussain, M.K.; Khalid, N.R. Surfactant-assisted synthesis of MoO3 nanorods and its application in photocatalytic degradation of different dyes in aqueous environment. J. Mol. Liq. 2022, 346, 117871. [Google Scholar] [CrossRef]
- Viet, T.Q.Q.; Nhu, T.H.; Thinh, D.B.; Trinh, D.N.; Giang, N.T.H.; Dat, N.M.; Hai, N.D.; Nam, H.M.; Phong, M.T.; Hieu, N.H. Optimization of TiO2 immobilized–Reduce graphene oxide photocatalyst toward organic compounds in aqueous medium. Synth. Met. 2021, 280, 116867. [Google Scholar] [CrossRef]
- Mahar, Z.A.; Shar, G.Q.; Balouch, A.; Pato, A.H.; Shaikh, A.R. Effective and viable photocatalytic degradation of rhodamine B dye in aqueous media using CuO/PVA nanocomposites. New J. Chem. 2021, 45, 16500–16510. [Google Scholar] [CrossRef]
- Kumar, A.P.; Bilehal, D.; Tadesse, A.; Kumar, D. Photocatalytic degradation of organic dyes: Pd-γ-Al2O3and PdO-γ-Al2O3 as potential photocatalysts. RSC Adv. 2021, 11, 6396–6406. [Google Scholar] [CrossRef]
- Mahmoodi, N.M. Binary catalyst system dye degradation using photocatalysis. Fibers Polym. 2014, 15, 273–280. [Google Scholar] [CrossRef]
- Sakthivel, S.; Neppolian, B.; Shankar, M.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. Solar photocatalytic degradation of azo dye: Comparison of photocatalytic efficiency of ZnO and TiO2. Sol. Energy Mater. Sol. Cells 2003, 77, 65–82. [Google Scholar] [CrossRef]
- Ribeiro, M.A.; Murgu, M.; Silva, V.D.M.; Sawaya, A.C.; Ribeiro, L.F.; Justi, A.; Meurer, E.C. The screening of organic matter in mineral and tap water by UHPLC-HRMS. Talanta 2017, 174, 581–586. [Google Scholar] [CrossRef]
- Ling, M.; Jing, W.; Marwanis Anua, S.; Mazlan, N. Concentrations of Magnesium, Calcium and Potassium in Drinking Water; A Comparison between Tap Water and Bore Water. J. Energy Saf. Technol. 2019, 2. [Google Scholar] [CrossRef]
- Khan, A.; Naeem, M.; Zekker, I.; Arian, M.B.; Michalski, G.; Khan, A.; Shah, N.; Zeeshan, S.; ul Haq, H.; Subhan, F.; et al. Evaluating groundwater nitrate and other physicochemical parameters of the arid and semi-arid district of DI Khan by multivariate statistical analysis. Environ. Technol. (UK) 2021. [Google Scholar] [CrossRef]
- Azlan, A.; Khoo, H.E.; Idris, M.A.; Ismail, A.; Razman, M.R. Evaluation of minerals content of drinking water in malaysia. Sci. World J. 2012, 2012, 403574. [Google Scholar] [CrossRef] [PubMed]
- Bathla, A.; Singla, D.; Pal, B. Highly efficient CaCO3-CaO extracted from tap water distillation for effective adsorption and photocatalytic degradation of malachite green dye. Mater. Res. Bull. 2019, 116, 1–7. [Google Scholar] [CrossRef]
- Barkatt, A.; Pulvirenti, A.L.; Adel-Hadadi, M.A.; Viragh, C.; Senftle, F.E.; Thorpe, A.N.; Grant, J.R. Composition and particle size of superparamagnetic corrosion products in tap water. Water Res. 2009, 43, 3319–3325. [Google Scholar] [CrossRef] [PubMed]
- Mehrvar, M.; Anderson, W.A.; Moo-Young, M. Photocatalytic degradation of aqueous organic solvents in the presence of hydroxyl radical scavengers. Int. J. Photoenergy 2002, 3, 187–191. [Google Scholar] [CrossRef] [Green Version]
- Platikanov, S.; Garcia, V.; Fonseca, I.; Rullán, E.; Devesa, R.; Tauler, R. Influence of minerals on the taste of bottled and tap water: A chemometric approach. Water Res. 2013, 47, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Burlingame, G.A.; Dietrich, A.M.; Whelton, A.J. Understanding the basics of tap water taste. J. Am. Water Works Assoc. 2007, 99, 100–111. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Ali, N.; Khan, I.; Zhang, B.; Sadiq, M. Heterogeneous photodegradation of industrial dyes: An insight to different mechanisms and rate affecting parameters. J. Environ. Chem. Eng. 2020, 8, 104364. [Google Scholar] [CrossRef]
- Mahasneh, B.Z. Assessment of Replacing Wastewater and treated water with Tap water in making Concrete Mix Soil stabilization and seismic load analysis View project Soil remediation View project Assessment of Replacing Wastewater and treated water with Tap water in making Concrete Mix. Artic. Electron. J. Geotech. Eng. 2014, 19, 2379–2386. [Google Scholar] [CrossRef]
- Chen, S.Q.; Li, M.; Ma, X.Y.; Zhou, M.J.; Wang, D.; Yan, M.Y.; Li, Z.; Yao, K.F. Influence of inorganic ions on degradation capability of Fe-based metallic glass towards dyeing wastewater remediation. Chemosphere 2021, 264, 128392. [Google Scholar] [CrossRef]
- Deng, H.; He, H.; Sun, S.; Zhu, X.; Zhou, D.; Han, F.; Huang, B.; Pan, X. Photocatalytic degradation of dye by Ag/TiO2 nanoparticles prepared with different sol–gel crystallization in the presence of effluent organic matter. Environ. Sci. Pollut. Res. 2019, 26, 35900–35912. [Google Scholar] [CrossRef]
- Brame, J.; Long, M.; Li, Q.; Alvarez, P. Inhibitory effect of natural organic matter or other background constituents on photocatalytic advanced oxidation processes: Mechanistic model development and validation. Water Res. 2015, 84, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.H.; Chang, T.F.M.; Chen, C.Y.; Sone, M.; Hsu, Y.J. Mechanistic Insights into Photodegradation of Organic Dyes Using Heterostructure Photocatalysts. Catalysts 2019, 9, 430. [Google Scholar] [CrossRef] [Green Version]
- Ahmadi, A.; Hajilou, M.; Zavari, S.; Yaghmaei, S. A comparative review on adsorption and photocatalytic degradation of classified dyes with metal/non-metal-based modification of graphitic carbon nitride nanocomposites: Synthesis, mechanism, and affecting parameters. J. Clean. Prod. 2023, 382, 134967. [Google Scholar] [CrossRef]
- Rauf, M.A.; Ashraf, S.S. Fundamental principles and application of heterogeneous photocatalytic degradation of dyes in solution. Chem. Eng. J. 2009, 151, 10–18. [Google Scholar] [CrossRef]
- Anwer, H.; Mahmood, A.; Lee, J.; Kim, K.H.; Park, J.W.; Yip, A.C.K. Photocatalysts for degradation of dyes in industrial effluents: Opportunities and challenges. Nano Res. 2019, 12, 955–972. [Google Scholar] [CrossRef]
- Akpan, U.G.; Hameed, B.H. Parameters affecting the photocatalytic degradation of dyes using TiO2-based photocatalysts: A review. J. Hazard. Mater. 2009, 170, 520–529. [Google Scholar] [CrossRef]
- Reza, K.M.; Kurny, A.; Gulshan, F. Parameters affecting the photocatalytic degradation of dyes using TiO2: A review. Appl. Water Sci. 2017, 7, 1569–1578. [Google Scholar] [CrossRef] [Green Version]
- Bekkouche, S.; Merouani, S.; Hamdaoui, O.; Bouhelassa, M. Efficient photocatalytic degradation of Safranin O by integrating solar-UV/TiO2/persulfate treatment: Implication of sulfate radical in the oxidation process and effect of various water matrix components. J. Photochem. Photobiol. A Chem. 2017, 345, 80–91. [Google Scholar] [CrossRef]
- Peng, Y.H.; Kashale, A.A.; Lai, Y.; Hsu, F.C.; Chen, I.W.P. Exfoliation of 2D materials by saponin in water: Aerogel adsorption/photodegradation organic dye. Chemosphere 2021, 274, 129795. [Google Scholar] [CrossRef]
- Sugashini, S.; Gomathi, T.; Devi, R.A.; Sudha, P.N.; Rambabu, K.; Banat, F. Nanochitosan/carboxymethyl cellulose/TiO2 biocomposite for visible-light-induced photocatalytic degradation of crystal violet dye. Environ. Res. 2022, 204, 112047. [Google Scholar] [CrossRef]
- Si, Y.; Li, J.; Cui, B.; Tang, D.; Yang, L.; Murugadoss, V.; Maganti, S.; Huang, M.; Guo, Z. Janus phenol–formaldehyde resin and periodic mesoporous organic silica nanoadsorbent for the removal of heavy metal ions and organic dyes from polluted water. Adv. Compos. Hybrid Mater. 2022, 5, 1180–1195. [Google Scholar] [CrossRef]
- Othman, Z.; Sinopoli, A.; MacKey, H.R.; Mahmoud, K.A. Efficient Photocatalytic Degradation of Organic Dyes by AgNPs/TiO2/Ti3C2Tx MXene Composites under UV and Solar Light. ACS Omega 2021, 6, 33325–33338. [Google Scholar] [CrossRef] [PubMed]
- Agócs, T.Z.; Puskás, I.; Varga, E.; Molnár, M.; Fenyvesi, É. Stabilization of nanosized titanium dioxide by cyclodextrin polymers and its photocatalytic effect on the degradation of wastewater pollutants. Beilstein J. Org. Chem. 2016, 12, 2873–2882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karim, M.A.H.; Aziz, K.H.H.; Omer, K.M.; Salih, Y.M.; Mustafa, F.; Rahman, K.O.; Mohammad, Y. Degradation of aqueous organic dye pollutants by heterogeneous photo-assisted Fenton-like process using natural mineral activator: Parameter optimization and degradation kinetics. IOP Conf. Ser. Earth Environ. Sci. 2021, 958, 012011. [Google Scholar] [CrossRef]
- Saeed, K.; Khan, I.; Gul, T.; Sadiq, M. Efficient photodegradation of methyl violet dye using TiO2/Pt and TiO2/Pd photocatalysts. Appl. Water Sci. 2017, 7, 3841–3848. [Google Scholar] [CrossRef]
- Zazouli, M.A.; Ghanbari, F.; Yousefi, M.; Madihi-Bidgoli, S. Photocatalytic degradation of food dye by Fe3O4–TiO2 nanoparticles in presence of peroxymonosulfate: The effect of UV sources. J. Environ. Chem. Eng. 2017, 5, 2459–2468. [Google Scholar] [CrossRef]
- Pugazhenthiran, N.; Mangalaraja, R.V.; Sathishkumar, P.; Murugesan, S.; Muneeswaran, T.; Pandiyarajan, T.; Naveenraj, S.; Contreras, D.; Anandan, S. Green synthesis of porous Au–Nx-TiO2 nanospheres for solar light induced photocatalytic degradation of diazo and triazo dyes and their eco-toxic effects. New J. Chem. 2018, 42, 18717–18728. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.S.; Hadavifar, M.; Ghasemi, S.S.; Arab Chamjangali, M. Synthesis of ZnO nanostructure using activated carbon for photocatalytic degradation of methyl orange from aqueous solutions. Appl. Water Sci. 2018, 8, 104. [Google Scholar] [CrossRef]
- Khan, I.; Zada, N.; Khan, I.; Sadiq, M.; Saeed, K. Enhancement of photocatalytic potential and recoverability of Fe3O4 nanoparticles by decorating over monoclinic zirconia. J. Environ. Heal. Sci. Eng. 2020, 18, 1473–1489. [Google Scholar] [CrossRef]
- Rincón, A.G.; Pulgarin, C. Effect of pH, inorganic ions, organic matter and H2O2 on E. coli K12 photocatalytic inactivation by TiO2: Implications in solar water disinfection. Appl. Catal. B Environ. 2004, 51, 283–302. [Google Scholar] [CrossRef]
- Khan, I.; Sadiq, M.; Khan, I.; Saeed, K. Manganese dioxide nanoparticles/activated carbon composite as efficient UV and visible-light photocatalyst. Environ. Sci. Pollut. Res. 2019, 26, 5140–5154. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Khan, A.A.; Khan, I.; Usman, M.; Sadiq, M.; Ali, F.; Saeed, K. Investigation of the photocatalytic potential enhancement of silica monolith decorated tin oxide nanoparticles through experimental and theoretical studies. New J. Chem. 2020, 44, 13330–13343. [Google Scholar] [CrossRef]
- Khan, I.; Khan, I.; Usman, M.; Imran, M.; Saeed, K. Nanoclay-mediated photocatalytic activity enhancement of copper oxide nanoparticles for enhanced methyl orange photodegradation. J. Mater. Sci. Mater. Electron. 2020, 31, 8971–8985. [Google Scholar] [CrossRef]
- Zafar, Z.; Fatima, R.; Kim, J.O. Experimental studies on water matrix and influence of textile effluents on photocatalytic degradation of organic wastewater using Fe–TiO2 nanotubes: Towards commercial application. Environ. Res. 2021, 197, 111120. [Google Scholar] [CrossRef]
- Mu, Y.; Yang, S.; Li, Y.; Zhang, J.; Ma, M.; Wang, J.; Yu, Z.; Ren, Z.; Liu, J. Highly efficient adsorptive and photocatalytic degradation of dye pollutants over biomass-derived carbon-supported Ag composites under visible light. J. Environ. Chem. Eng. 2021, 9, 106580. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A.; et al. Review on Methylene Blue: Its Properties, Uses, Toxicity and Photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Sheng, H.; Li, Q.; Ma, W.; Ji, H.; Chen, C.; Zhao, J. Photocatalytic degradation of organic pollutants on surface anionized TiO2: Common effect of anions for high hole-availability by water. Appl. Catal. B Environ. 2013, 138–139, 212–218. [Google Scholar] [CrossRef]
- Meghlaoui, F.Z.; Merouani, S.; Hamdaoui, O.; Alghyamah, A.; Bouhelassa, M.; Ashokkumar, M. Fe(III)-catalyzed degradation of persistent textile dyes by chlorine at slightly acidic conditions: The crucial role of Cl2●− radical in the degradation process and impacts of mineral and organic competitors. Asia-Pac. J. Chem. Eng. 2021, 16, e2553. [Google Scholar] [CrossRef]
- Mahvi, A.H.; Ghanbarian, M.; Nasseri, S.; Khairi, A. Mineralization and discoloration of textile wastewater by TiO2 nanoparticles. Desalination 2009, 239, 309–316. [Google Scholar] [CrossRef]
- Rayaroth, M.P.; Aravind, U.K.; Aravindakumar, C.T. Effect of inorganic ions on the ultrasound initiated degradation and product formation of triphenylmethane dyes. Ultrason. Sonochem. 2018, 48, 482–491. [Google Scholar] [CrossRef]
- Habibi, M.H.; Hassanzadeh, A.; Mahdavi, S. The effect of operational parameters on the photocatalytic degradation of three textile azo dyes in aqueous TiO2 suspensions. J. Photochem. Photobiol. A Chem. 2005, 172, 89–96. [Google Scholar] [CrossRef]
- Hu, C.; Jimmy, C.Y.; Hao, Z.; Wong, P. Effects of acidity and inorganic ions on the photocatalytic degradation of different azo dyes. Appl. Catal. B Environ. 2003, 46, 35–47. [Google Scholar] [CrossRef]
- Saleh, R.; Taufik, A. Photo-Fenton degradation of methylene blue in the presence of Au-Fe3O4/graphene composites under UV and visible light at near neutral pH: Effect of coexisting inorganic anion. Environ. Nanotechnol. Monit. Manag. 2019, 11, 100221. [Google Scholar] [CrossRef]
- Cheng, X.; Chong, R.; Cao, Y.; Li, D.; Chang, Z.; Zhang, L. Influence of inorganic anions on photocatalytic degeneration of methylene blue on Ag3PO4. J. Nanosci. Nanotechnol. 2016, 16, 12489–12497. [Google Scholar] [CrossRef]
- Joorabi, F.T.; Kamali, M.; Sheibani, S. Effect of aqueous inorganic anions on the photocatalytic activity of CuO–Cu2O nanocomposite on MB and MO dyes degradation. Mater. Sci. Semicond. Process. 2022, 139, 106335. [Google Scholar] [CrossRef]
- Gupta, N.K.; Ghaffari, Y.; Kim, S.; Bae, J.; Kim, K.S.; Saifuddin, M. Photocatalytic degradation of organic pollutants over MFe2O4 (M= Co, Ni, Cu, Zn) nanoparticles at neutral pH. Sci. Rep. 2020, 10, 4942. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.; Dong, Y.; Liu, T. Simultaneous photodegradation of dyes by NiS/CuS-CdS composites in visible light region. Colloids Surf. A: Physicochem. Eng. Asp. 2020, 598, 124854. [Google Scholar] [CrossRef]
- Rizal, M.Y.; Saleh, R.; Taufik, A.; Yin, S. Photocatalytic decomposition of methylene blue by persulfate-assisted Ag/Mn3O4 and Ag/Mn3O4/graphene composites and the inhibition effect of inorganic ions. Environ. Nanotechnol. Monit. Manag. 2021, 15, 100408. [Google Scholar] [CrossRef]
- Sahoo, C.; Gupta, A.K.; Sasidharan Pillai, I.M. Photocatalytic degradation of methylene blue dye from aqueous solution using silver ion-doped TiO2 and its application to the degradation of real textile wastewater. J. Environ. Sci. Health Part A 2012, 47, 1428–1438. [Google Scholar] [CrossRef]
- Mohammed, R.; Ali, M.E.M.; Gomaa, E.; Mohsen, M. Green ZnO nanorod material for dye degradation and detoxification of pharmaceutical wastes in water. J. Environ. Chem. Eng. 2020, 8, 104295. [Google Scholar] [CrossRef]
- Krishnakumar, B.; Swaminathan, M. Influence of operational parameters on photocatalytic degradation of a genotoxic azo dye Acid Violet 7 in aqueous ZnO suspensions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 81, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Bathla, A.; Kaur, D.; Pal, B. Impact of metal ions (Cr +6 /Mn +7) loaded CaCO3 extracted from tap water for adsorption/ degradation of toxic pollutants under sunlight. Mater. Express 2022, 12, 106–113. [Google Scholar] [CrossRef]
- Chang, C.; Yang, H.; Kan, L.; Mu, W.; Wang, Q.; Lu, S.Y.; Deng, B. Mechanism and impacts of inorganic ion addition on photocatalytic degradation of triclosan catalyzed by heterostructured Bi7O9I3/Bi. J. Taiwan Inst. Chem. Eng. 2021, 125, 176–185. [Google Scholar] [CrossRef]
- Wang, C.; Zhu, Q.; Gu, C.; Luo, X.; Yu, C.; Wu, M. Photocatalytic degradation of two different types of dyes by synthesized La/Bi2WO6. RSC Adv. 2016, 6, 85852–85859. [Google Scholar] [CrossRef]
- Zainal, Z.; Lee, C.Y.; Hussein, M.Z.; Kassim, A.; Yusof, N.A. Effect of supporting electrolytes in electrochemically-assisted photodegradation of an azo dye. J. Photochem. Photobiol. A Chem. 2005, 172, 316–321. [Google Scholar] [CrossRef]
- Sohrabi, M.R.; Ghavami, M. Photocatalytic degradation of Direct Red 23 dye using UV/TiO2: Effect of operational parameters. J. Hazard. Mater. 2008, 153, 1235–1239. [Google Scholar] [CrossRef]
- Borthakur, P.; Das, M.R. Hydrothermal assisted decoration of NiS2 and CoS nanoparticles on the reduced graphene oxide nanosheets for sunlight driven photocatalytic degradation of azo dye: Effect of background electrolyte and surface charge. J. Colloid Interface Sci. 2018, 516, 342–354. [Google Scholar] [CrossRef]
- Borthakur, P.; Boruah, P.K.; Hussain, N.; Silla, Y.; Das, M.R. Specific ion effect on the surface properties of Ag/reduced graphene oxide nanocomposite and its influence on photocatalytic efficiency towards azo dye degradation. Appl. Surf. Sci. 2017, 423, 752–761. [Google Scholar] [CrossRef]
- Nyankson, E.; Kumar, R.V. Removal of water-soluble dyes and pharmaceutical wastes by combining the photocatalytic properties of Ag3PO4 with the adsorption properties of halloysite nanotubes. Mater. Today Adv. 2019, 4, 100025. [Google Scholar] [CrossRef]
- Vatovec, C.; Kolodinsky, J.; Callas, P.; Hart, C.; Gallagher, K. Pharmaceutical pollution sources and solutions: Survey of human and veterinary medication purchasing, use, and disposal. J. Environ. Manag. 2021, 285, 112106. [Google Scholar] [CrossRef]
- Kaur, K.; Badru, R.; Singh, P.P.; Kaushal, S. Photodegradation of organic pollutants using heterojunctions: A review. J. Environ. Chem. Eng. 2020, 8, 103666. [Google Scholar] [CrossRef]
- De Oliveira Souza, H.; dos Santos Costa, R.; Quadra, G.R.; dos Santos Fernandez, M.A. Pharmaceutical pollution and sustainable development goals: Going the right way? Sustain. Chem. Pharm. 2021, 21, 100428. [Google Scholar] [CrossRef]
- Freitas, L.; Radis-Baptista, G. Pharmaceutical Pollution and Disposal of Expired, Unused, and Unwanted Medicines in the Brazilian Context. J. Xenobiotics 2021, 11, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Gaeta, M.; Sanfilippo, G.; Fraix, A.; Sortino, G.; Barcellona, M.; Conti, G.O.; Fragalà, M.E.; Ferrante, M.; Purrello, R.; D’urso, A. Photodegradation of Antibiotics by Noncovalent Porphyrin-Functionalized TiO2 in Water for the Bacterial Antibiotic Resistance Risk Management. Int. J. Mol. Sci. 2020, 21, 3775. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.J.; Mo, C.H.; Li, Y.W.; Gao, P.; Tai, Y.P.; Zhang, Y.; Ruan, Z.L.; Xu, J.W. Determination of four fluoroquinolone antibiotics in tap water in Guangzhou and Macao. Environ. Pollut. 2010, 158, 2350–2358. [Google Scholar] [CrossRef] [PubMed]
- Valcárcel, Y.; González Alonso, S.; Rodríguez-Gil, J.L.; Gil, A.; Catalá, M. Detection of pharmaceutically active compounds in the rivers and tap water of the Madrid Region (Spain) and potential ecotoxicological risk. Chemosphere 2011, 84, 1336–1348. [Google Scholar] [CrossRef]
- Askari, N.; Beheshti, M.; Mowla, D.; Farhadian, M. Facile construction of novel Z-scheme MnWO4/Bi2S3 heterojunction with enhanced photocatalytic degradation of antibiotics. Mater. Sci. Semicond. Process. 2021, 127, 105723. [Google Scholar] [CrossRef]
- Yang, X.; Chen, Z.; Zhao, W.; Liu, C.; Qian, X.; Zhang, M.; Wei, G.; Khan, E.; Hau Ng, Y.; Sik Ok, Y. Recent advances in photodegradation of antibiotic residues in water. Chem. Eng. J. 2021, 405, 126806. [Google Scholar] [CrossRef]
- Durán-Álvarez, J.C.; Martínez-Avelar, C.; González-Cervantes, E.; Gutiérrez-Márquez, R.A.; Rodríguez-Varela, M.; Varela, A.S.; Castillón, F.; Zanella, R. Degradation and mineralization of oxytetracycline in pure and tap water under visible light irradiation using bismuth oxyiodides and the effect of depositing Au nanoparticles. J. Photochem. Photobiol. A Chem. 2020, 388, 112163. [Google Scholar] [CrossRef]
- Zaviska, F.; Drogui, P.; El Hachemi, E.M.; Naffrechoux, E. Effect of nitrate ions on the efficiency of sonophotochemical phenol degradation. Ultrason. Sonochem. 2014, 21, 69–75. [Google Scholar] [CrossRef]
- Gul, T.; Khan, I.; Saeed, K. Synthesis of mica supported tungsten oxide nanoparticles and their photocatalytic, biological and antioxidant activity. Int. J. Environ. Anal. Chem. 2022, 1–18. [Google Scholar] [CrossRef]
- Ye, Y.; Feng, Y.; Bruning, H.; Yntema, D.; Rijnaarts, H.H.M. Photocatalytic degradation of metoprolol by TiO2 nanotube arrays and UV-LED: Effects of catalyst properties, operational parameters, commonly present water constituents, and photo-induced reactive species. Appl. Catal. B Environ. 2018, 220, 171–181. [Google Scholar] [CrossRef]
- Bianchi, C.L.; Sacchi, B.; Pirola, C.; Demartin, F.; Cerrato, G.; Morandi, S.; Capucci, V. Aspirin and paracetamol removal using a commercial micro-sized TiO2 catalyst in deionized and tap water. Environ. Sci. Pollut. Res. 2017, 24, 12646–12654. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Salcedo, M.; Monge, M.; Tena, M.T. The photocatalytic degradation of naproxen with g-C3N4 and visible light: Identification of primary by-products and mechanism in tap water and ultrapure water. J. Environ. Chem. Eng. 2022, 10, 106964. [Google Scholar] [CrossRef]
- Baran, W.; Cholewiński, M.; Sobczak, A.; Adamek, E. A new mechanism of the selective photodegradation of antibiotics in the catalytic system containing Tio2 and the inorganic cations. Int. J. Mol. Sci. 2021, 22, 8696. [Google Scholar] [CrossRef]
- Pereira, J.H.O.S.; Reis, A.C.; Queirós, D.; Nunes, O.C.; Borges, M.T.; Vilar, V.P.; Boaventura, R.A.R. Insights into solar TiO2-assisted photocatalytic oxidation of two antibiotics employed in aquatic animal production, oxolinic acid and oxytetracycline. Sci. Total Environ. 2013, 463–464, 274–283. [Google Scholar] [CrossRef]
- Hung, C.M.; Chen, C.W.; Huang, C.P.; Dong, C. Di N-doped metal-free biochar activation of peroxymonosulfate for enhancing the degradation of antibiotics sulfadiazine from aquaculture water and its associated bacterial community composition. J. Environ. Chem. Eng. 2022, 10, 107172. [Google Scholar] [CrossRef]
- Gao, L.; Zhou, B.; Wang, F.; Yuan, R.; Chen, H.; Han, X. Effect of dissolved organic matters and inorganic ions on TiO2 photocatalysis of diclofenac: Mechanistic study and degradation pathways. Environ. Sci. Pollut. Res. 2020, 27, 2044–2053. [Google Scholar] [CrossRef]
- Damalas, C.A.; Koutroubas, S.D. Farmers’ Exposure to Pesticides: Toxicity Types and Ways of Prevention. Toxics 2016, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, R.K.; Kumar, V.; Roy, K. Ecotoxicological QSTR and QSTTR Modeling for the Prediction of Acute Oral Toxicity of Pesticides against Multiple Avian Species. Environ. Sci. Technol. 2022, 56, 335–348. [Google Scholar] [CrossRef]
- Alengebawy, A.; Abdelkhalek, S.T.; Qureshi, S.R.; Wang, M.Q. Heavy Metals and Pesticides Toxicity in Agricultural Soil and Plants: Ecological Risks and Human Health Implications. Toxics 2021, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Soler, J.; García-Ripoll, A.; Hayek, N.; Miró, P.; Vicente, R.; Arques, A.; Amat, A. Effect of inorganic ions on the solar detoxification of water polluted with pesticides. Water Res. 2009, 43, 4441–4450. [Google Scholar] [CrossRef] [PubMed]
- Orellana-García, F.; Álvarez, M.A.; López-Ramón, V.; Rivera-Utrilla, J.; Sánchez-Polo, M.; Mota, A.J. Photodegradation of herbicides with different chemical natures in aqueous solution by ultraviolet radiation. Effects of operational variables and solution chemistry. Chem. Eng. J. 2014, 255, 307–315. [Google Scholar] [CrossRef]
- Fadaei, A.; Dehghani, M.; Mahvi, A.; Nasseri, S.; Rastkari, N.; Shayeghi, M. Degradation of organophosphorus pesticides in water during UV/Htreatment: Role of sulphate and bicarbonate ions. E-J. Chem. 2012, 9, 2015–2022. [Google Scholar] [CrossRef] [Green Version]
- Liang, R.; Tang, F.; Wang, J.; Yue, Y. Photo-degradation dynamics of five neonicotinoids: Bamboo vinegar as a synergistic agent for improved functional duration. PLoS ONE 2019, 14, e0223708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahmoodi, N.M.; Arami, M.; Limaee, N.Y.; Gharanjig, K. Photocatalytic degradation of agricultural N-heterocyclic organic pollutants using immobilized nanoparticles of titania. J. Hazard. Mater. 2007, 145, 65–71. [Google Scholar] [CrossRef]
- Rabindranathan, S.; Devipriya, S.; Yesodharan, S. Photocatalytic degradation of phosphamidon on semiconductor oxides. J. Hazard. Mater. 2003, 102, 217–229. [Google Scholar] [CrossRef]
- Dugandžić, A.M.; Tomašević, A.V.; Radišić, M.M.; Šekuljica, N.; Mijin, D.; Petrović, S.D. Effect of inorganic ions, photosensitisers and scavengers on the photocatalytic degradation of nicosulfuron. J. Photochem. Photobiol. A Chem. 2017, 336, 146–155. [Google Scholar] [CrossRef]
- Wu, R.J.; Chen, C.C.; Chen, M.H.; Lu, C.S. Titanium dioxide-mediated heterogeneous photocatalytic degradation of terbufos: Parameter study and reaction pathways. J. Hazard. Mater. 2009, 162, 945–953. [Google Scholar] [CrossRef]
- Levet, A.; Bordes, C.; Clément, Y.; Mignon, P.; Morell, C.; Chermette, H.; Marote, P.; Lantéri, P. Acute aquatic toxicity of organic solvents modeled by QSARs. J. Mol. Model. 2016, 22, 288. [Google Scholar] [CrossRef]
- Oomen, W.W.; Begines, P.; Mustafa, N.R.; Wilson, E.G.; Verpoorte, R.; Choi, Y.H. Natural Deep Eutectic Solvent Extraction of Flavonoids of Scutellaria baicalensis as a Replacement for Conventional Organic Solvents. Molecules 2020, 25, 617. [Google Scholar] [CrossRef] [Green Version]
- Raj Joshi, D.; Adhikari, N.; Cruz-Olivares, J. An Overview on Common Organic Solvents and Their Toxicity. J. Pharm. Res. Int. 2019, 28, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Zhang, F.; Wang, C.; Mu, C.; Lin, W. A novel hydrophobic all-biomass aerogel reinforced by dialdehyde carboxymethyl cellulose for oil/organic solvent-water separation. Polymer (Guildf) 2022, 238, 124402. [Google Scholar] [CrossRef]
- Dharwadkar, S.; Yu, L.; Achari, G. Photocatalytic Degradation of Sulfolane Using a LED-Based Photocatalytic Treatment System. Catalysts 2021, 11, 624. [Google Scholar] [CrossRef]
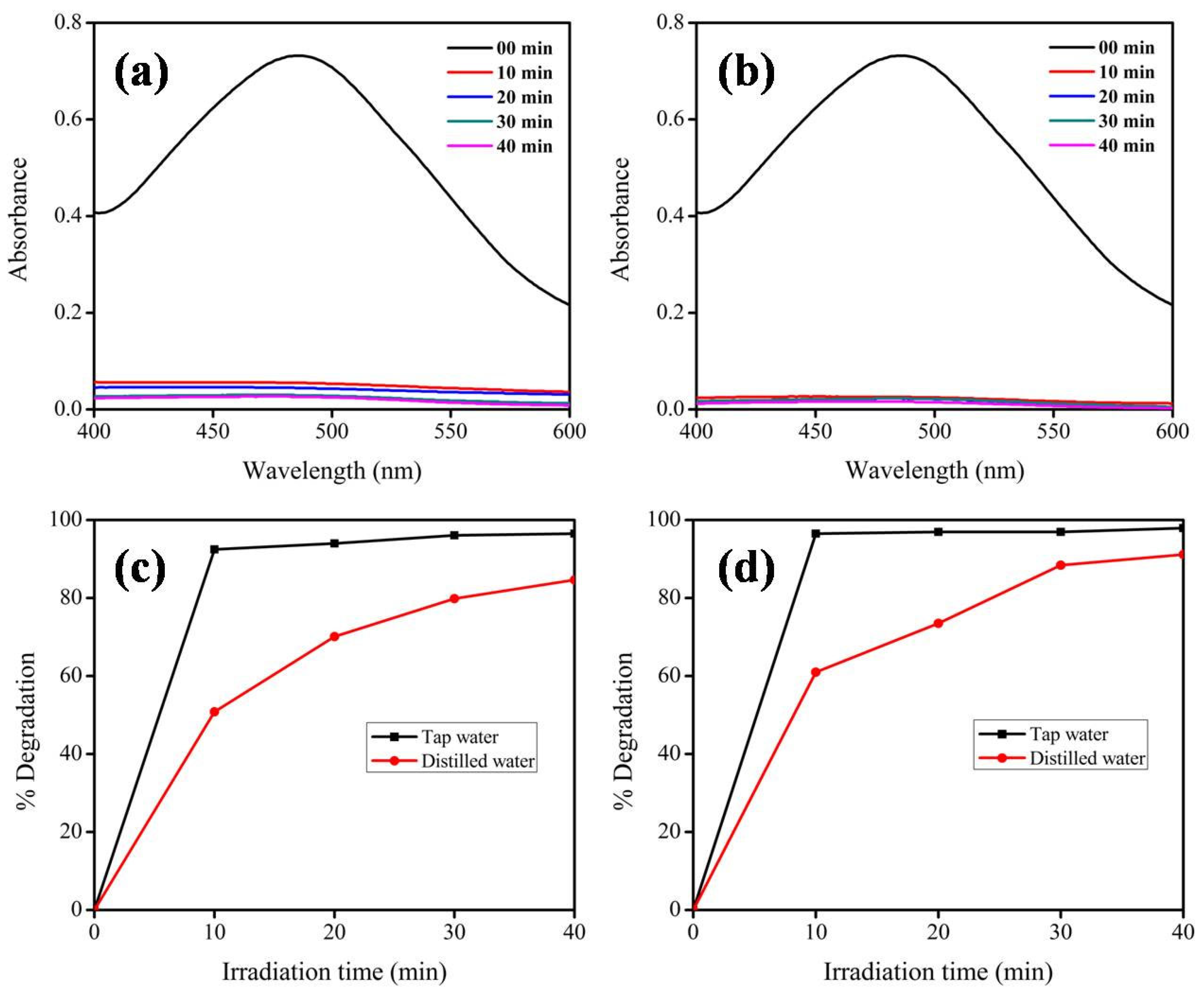


| Photocatalysts and Dye | %Degradation in Deionized Water | %Degradation in Tap Water | Conclusion and Reason | Ref |
|---|---|---|---|---|
| MnO2/AC nanocomposite and MnO2 NPs. Congo red dye | 98.53% by MnO2/AC and 66.57% by MnO2 in 5 min under UV-light. | Dye almost completely (99%) degraded in 1 min under UV and visible light. | Dye degraded efficiently in TW. The presence of various mineral ions in the tap water enhanced the photocatalytic activity. | [73] |
| SnO2/SiO2 NPs and SnO2 NPs | 94.58% by SnO2/SiO2 NPs and 65.93% by SnO2 NPs in 30 min under UV-light. | 8.92% by SnO2/SiO2 NPs and 22.81% by SnO2 NPs in 30 min under UV-light. | Dye degraded slowly in tap water. The presence of organic, inorganic and metallic ions present in TW serve as competing species for the active sites of photocatalysts and reduce their photocatalytic activity. | [74] |
| CuO/NC NPs and CuO NPs | 97.18% by CuO/NC NPs and 68.22% by CuO NPs in 4 min under UV-light. | 47.33% dye by CuO/NC NPs and 23.4% by CuO NPs in 4 min under UV-light. | Dye degraded slowly in TW. The presence of organic, inorganic and metallic ions in TW, serves as competitive species for the active sites of photocatalyst and reduce their photocatalytic activity. | [75] |
| Fe3O4/ZrO2 NPs and Fe3O4 NPs. Methyl red | 91% by Fe3O4/ZrO2 NPs and 84% by Fe3O4 NPs within 40 min under UV-light. | 97% by Fe3O4/ZrO2 NPs and 96% by Fe3O4 NPs within 40 min under UV-light. | Dye degraded efficiently in TW. The presence of mineral ions in the TW enhances photocatalyst activity. | [71] |
| Fe–TiO2 nanotubes. Congo red | 86% under visible light. | 74% under visible light. | Dye degraded slowly in TW. Lower activity in tap water is due to the deactivating effects of organic, inorganic, and salt compounds. | [76] |
| Ag/P@BC. Rhodamine B | 83.08% under visible light irradiation. | 75.85% under visible light irradiation. | Some inorganic salts in TW slightly affected the process of adsorption and photodegradation. | [77] |
| Ag NPs/TiO2/Ti3C2Tx | full degradation in 15 min under UV-light irradiation. | Lower photodegradation in tap water under UV-light irradiation. | Lower activity in TW. The presence of other competing organics in the wastewater. | [64] |
| TiO2/Pd and TiO2/Pt | 78% and 95% in 20 min under UV-light. | 62% and 47% in 20 min under UV-light. | Lower activity in TW. Presence of organic, inorganic, and metallic ions in TW, adsorbed on the catalyst active sites decreases its activity | [67] |
| Photocatalyst and Dye | Inorganic Anions | Positive Effect | Negative Effect | Negligible Effect | Reference |
|---|---|---|---|---|---|
| commercial TiO2. Direct 80, Direct Blue, Reactive Yellow 2 | SO42−, Cl− and NO3− | SO42−, Cl− and NO3− | [83] | ||
| TiO2 dispersions. Procion Red MX-5B and Cationic Blue X-GRL | SO42−, H2PO4−, ClO4− and F− | In acidic medium | In basic medium | [84] | |
| Au-Fe3O4/ graphene composites. Methylene blue | NaCl, Na2SO4, NaH2PO4, NaNO3, and Na2CO3 | SO42−, Cl−, H2PO4−, NO3−, CO32− | Na+ | [85] | |
| Ag3PO4. Methylene blue | NO3−, OH−, NO2−, HCO3−, Cl−, Br−, CO32−, SO42−, SO32−, S2− and PO43− | OH−, Cl−, Br−, HCO−, CO32−, SO42−, SO32−, S2− | NO2−, | NO3−, | [86] |
| CuO–Cu2O nanocomposite. Methylene blue (MB) and Methyl orange (MO). | SO42−, Cl− and NO3− | Cl− (0.5 mM) on MB | SO42− on MB. Cl− on MO | SO42− on MO. | [87] |
| ZnFe2O4 Methylene blue | SO42−, NO3−, Cl−, CO32− | SO42−, NO3−, Cl−, CO32− | [88] | ||
| cerium-doped SiO2/TiO2 Methylene blue | NO3−, SO42−, Cl− | NO3−, SO42−, Cl− | [89] | ||
| Ag/Mn3O4 and Ag/Mn3O4/graphene with persulfate. Methylene blue | Cl−, SO42−, NO3−, H2PO4−, CO3− | All ions have negative effect in the order H2PO4− > CO32− > SO42− > Cl− > NO3− | [90] | ||
| silver ion-doped TiO2 Methylene blue | Cl−, NO3−, SO42−, CO32− | Cl−, NO3−, SO42−, CO32− | [91] | ||
| ZnO nanorod. MB, Acid red, Remazol red, and Rhodamine B | PO43−, Cl−, SO42−, NO3−, | All ions have negative effect in the order PO43−, > Cl−, > SO42− ≈ NO3− | [92] | ||
| NiS/CuS-CdS composites. MB and MO | NaCl, K3PO4 and Na2CO3 | K3PO4 for MO | NaCl and Na2CO3 both MB and MO. K3PO4 for MB. | [89] | |
| TiO2 NPs | SO42−, NO3−, HCO3, CO3−2 | SO42−, NO3−, HCO3, CO3−2 | [81] |
| Photocatalyst and Dye | Inorganic Anions | Positive Effect | Negative Effect | Negligible Effect | Reference |
|---|---|---|---|---|---|
| La/Bi2WO6 composite. Reactive brilliant red X-3B (X-3B) and rhodamine B (RhB) | Na+, K+, Ca2+ and Mg2+ | All cations promoted the removal of RhB | All cations inhibited the removal of X-3B | [96] | |
| TiO2/Electrochemically-assisted photodegradation. Methyl orange | Na+, K+, Ca2+, NH4+ and Mg2+ | Na+, K+, Ca2+ and NH4+ | Mg2+ | [97] | |
| UV/TiO2 system. Direct Red 23 | Cu2+, Al3+, Cr3+, Sn4+ | Cu2+, Al3+, Cr3+, Sn4+ | [98] | ||
| NiS2-rGO and CoS-rGO nanocomposite | Na+, Mg2+ and Ca2+ | Positive effect at 0.1 M salt solution concentration | Negligible effect at 0.01 M salt solution concentration | [99] | |
| Ag/rGO nanocomposite. congo red and bismarck brow | Ca2+, Mg2+, Na+ and NH4+ | Ca2+, Mg2+, Na+ and NH4+ | [100] | ||
| persulfate-assisted Ag/Mn3O4 and Ag/Mn3O4/graphene composites. methylene blue | K+, Ca2+, and Mg2+ | Inhibition effect in the order Ca2+ > Mg2+ > K+ | [90] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, S.; Almehmadi, M.; Janjuhah, H.T.; Kontakiotis, G.; Abdulaziz, O.; Saeed, K.; Ahmad, H.; Allahyani, M.; Aljuaid, A.; Alsaiari, A.A.; et al. The Effect of Mineral Ions Present in Tap Water on Photodegradation of Organic Pollutants: Future Perspectives. Water 2023, 15, 175. https://doi.org/10.3390/w15010175
Ahmad S, Almehmadi M, Janjuhah HT, Kontakiotis G, Abdulaziz O, Saeed K, Ahmad H, Allahyani M, Aljuaid A, Alsaiari AA, et al. The Effect of Mineral Ions Present in Tap Water on Photodegradation of Organic Pollutants: Future Perspectives. Water. 2023; 15(1):175. https://doi.org/10.3390/w15010175
Chicago/Turabian StyleAhmad, Shujaat, Mazen Almehmadi, Hammad Tariq Janjuhah, George Kontakiotis, Osama Abdulaziz, Khalid Saeed, Hanif Ahmad, Mamdouh Allahyani, Abdulelah Aljuaid, Ahad Amer Alsaiari, and et al. 2023. "The Effect of Mineral Ions Present in Tap Water on Photodegradation of Organic Pollutants: Future Perspectives" Water 15, no. 1: 175. https://doi.org/10.3390/w15010175
APA StyleAhmad, S., Almehmadi, M., Janjuhah, H. T., Kontakiotis, G., Abdulaziz, O., Saeed, K., Ahmad, H., Allahyani, M., Aljuaid, A., Alsaiari, A. A., Muhammad, J., & Khan, I. (2023). The Effect of Mineral Ions Present in Tap Water on Photodegradation of Organic Pollutants: Future Perspectives. Water, 15(1), 175. https://doi.org/10.3390/w15010175








