Functional Responses and Additive Multiple Predator Effects of Two Common Wetland Fish
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Collection
2.2. Experimental Design
2.3. Data Analysis
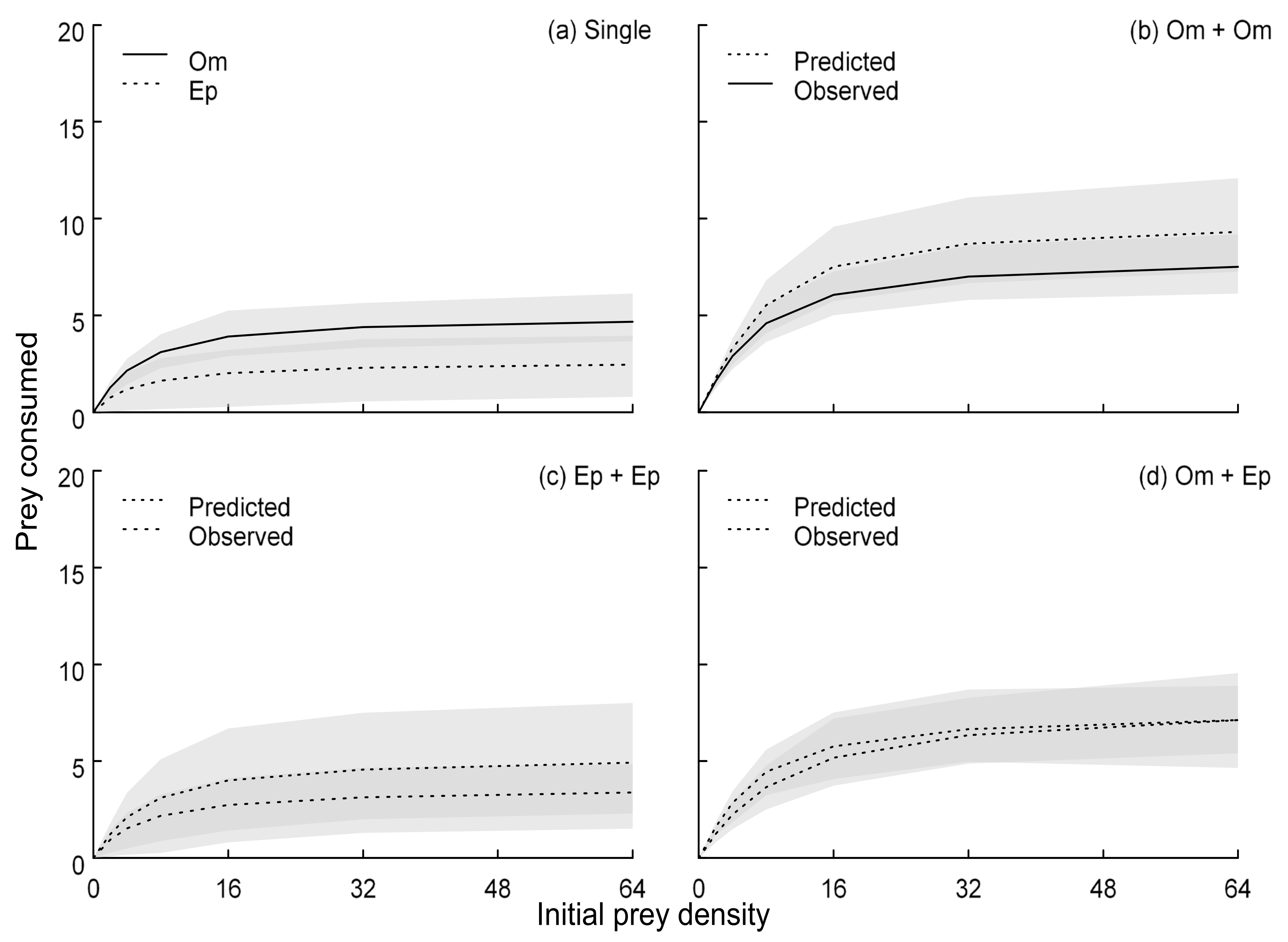
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sih, A.; Englund, G.; Wooster, D. Emergent Impacts of Multiple Predators on Prey. Trends Ecol. Evol. 1998, 13, 350–355. [Google Scholar] [CrossRef]
- Barrios-O’Neill, D.; Dick, J.T.A.; Ricciardi, A.; MacIsaac, H.J.; Emmerson, M.C. Deep Impact: In Situ Functional Responses Reveal Context-Dependent Interactions between Vertically Migrating Invasive and Native Mesopredators and Shared Prey. Freshw. Biol. 2014, 59, 2194–2203. [Google Scholar] [CrossRef]
- Kumar, R. Impacts of Predation by the Copepod, Mesocyclops Pehpeiensis, on Life Table Demographics and Population Dynamics of Four Cladoceran Species: A Comparative Laboratory Study. Zool. Stud. 2009, 48, 738–752. [Google Scholar]
- Cuthbert, R.N.; Dalu, T.; Wasserman, R.J.; Sentis, A.; Weyl, O.L.F.; Froneman, P.W.; Callaghan, A.; Dick, J.T.A. Prey and Predator Density-Dependent Interactions under Different Water Volumes. Ecol. Evol. 2021, 11, 6504–6512. [Google Scholar] [CrossRef]
- Dalu, T.; Wasserman, R.J. Fundamentals of Tropical Freshwater Wetlands: From Ecology to Conservation Management; Elsevier: Amsterdam, The Netherlands, 2022; ISBN 978-0-12-822362-8. [Google Scholar] [CrossRef]
- Daniel, J.; Rooney, R.C. Wetland Hydroperiod Predicts Community Structure, but Not the Magnitude of Cross-Community Congruence. Sci. Rep. 2021, 11, 429. [Google Scholar] [CrossRef]
- Sheaves, M.; Johnston, R.; Abrantes, K.; Sheaves, M.; Johnston, R.; Abrantes, K. Fish Fauna of Dry Tropical and Subtropical Estuarine Floodplain Wetlands. Mar. Freshw. Res. 2007, 58, 931–943. [Google Scholar] [CrossRef]
- Carvalho, D.A.; Williner, V.; Giri, F.; Vaccari, C.; Collins, P.A.; Carvalho, D.A.; Williner, V.; Giri, F.; Vaccari, C.; Collins, P.A. Quantitative Food Webs and Invertebrate Assemblages of a Large River: A Spatiotemporal Approach in Floodplain Shallow Lakes. Mar. Freshw. Res. 2016, 68, 293–307. [Google Scholar] [CrossRef] [Green Version]
- Middelburg, J.J. Stable Isotopes Dissect Aquatic Food Webs from the Top to the Bottom. Biogeosciences 2014, 11, 2357–2371. [Google Scholar] [CrossRef] [Green Version]
- Rettig, J.E.; Smith, G.R. Relative Strength of Top-down Effects of an Invasive Fish and Bottom-up Effects of Nutrient Addition in a Simple Aquatic Food Web. Environ. Sci. Pollut. Res. 2021, 28, 5845–5853. [Google Scholar] [CrossRef]
- Křivan, V.; Schmitz, O.J. Trait and Density Mediated Indirect Interactions in Simple Food Webs. Oikos 2004, 107, 239–250. [Google Scholar] [CrossRef]
- Alexander, M.E.; Dick, J.T.A.; O’Connor, N.E. Trait-Mediated Indirect Interactions in a Marine Intertidal System as Quantified by Functional Responses. Oikos 2013, 122, 1521–1531. [Google Scholar] [CrossRef]
- Buxton, M.; Cuthbert, R.N.; Dalu, T.; Nyamukondiwa, C.; Wasserman, R.J. Predator Density Modifies Mosquito Regulation in Increasingly Complex Environments. Pest Manag. Sci. 2020, 76, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Soluk, D.A. Multiple Predator Effects: Predicting Combined Functional Response of Stream Fish and Invertebrate Predators. Ecology 1993, 74, 219–225. [Google Scholar] [CrossRef]
- Holling, C.S. Some Characteristics of Simple Types of Predation and Parasitism 1. Can. Entomol. 1959, 91, 385–398. [Google Scholar] [CrossRef]
- Holling, C.S. The Components of Predation as Revealed by a Study of Small-Mammal Predation of the European Pine Sawfly 1. Can. Entomol. 1959, 91, 293–320. [Google Scholar] [CrossRef]
- Dick, J.T.A.; Alexander, M.E.; Jeschke, J.M.; Ricciardi, A.; MacIsaac, H.J.; Robinson, T.B.; Kumschick, S.; Weyl, O.L.F.; Dunn, A.M.; Hatcher, M.J.; et al. Advancing Impact Prediction and Hypothesis Testing in Invasion Ecology Using a Comparative Functional Response Approach. Biol. Invasions 2014, 16, 735–753. [Google Scholar] [CrossRef] [Green Version]
- Holling, C.S. The Functional Response of Invertebrate Predators to Prey Density. Mem. Entomol. Soc. Can. 1966, 98, 5–86. [Google Scholar] [CrossRef]
- Abrams, P. The Functional Responses of Adaptive Consumers of Two Resources. Theor. Popul. Biol. 1987, 32, 262–288. [Google Scholar] [CrossRef]
- DeLong, J.P. Predator Ecology: Evolutionary Ecology of the Functional Response; Oxford University Press: Oxford, UK, 2021; ISBN 9780192895509. Available onlinehttps://books.google.co.za/books?id=ytg9EAAAQBAJ (accessed on 14 December 2021).
- Hermoso, V.; Clavero, M.; Blanco-Garrido, F.; Prenda, J. Invasive Species and Habitat Degradation in Iberian Streams: An Analysis of Their Role in Freshwater Fish Diversity Loss. Ecol. Appl. 2011, 21, 175–188. [Google Scholar] [CrossRef]
- Leitão, R.P.; Zuanon, J.; Mouillot, D.; Leal, C.G.; Hughes, R.M.; Kaufmann, P.R.; Villéger, S.; Pompeu, P.S.; Kasper, D.; de Paula, F.R.; et al. Disentangling the Pathways of Land Use Impacts on the Functional Structure of Fish Assemblages in Amazon Streams. Ecography 2018, 41, 219–232. [Google Scholar] [CrossRef]
- Hurd, L.E.; Sousa, R.G.C.; Siqueira-Souza, F.K.; Cooper, G.J.; Kahn, J.R.; Freitas, C.E.C. Amazon Floodplain Fish Communities: Habitat Connectivity and Conservation in a Rapidly Deteriorating Environment. Biol. Conserv. 2016, 195, 118–127. [Google Scholar] [CrossRef]
- McCoy, M.W.; Stier, A.C.; Osenberg, C.W. Emergent Effects of Multiple Predators on Prey Survival: The Importance of Depletion and the Functional Response. Ecol. Lett. 2012, 15, 1449–1456. [Google Scholar] [CrossRef] [PubMed]
- Sentis, A.; Boukal, D.S. On the Use of Functional Responses to Quantify Emergent Multiple Predator Effects. Sci. Rep. 2018, 8, 11787. [Google Scholar] [CrossRef] [PubMed]
- Sentis, A.; Gémard, C.; Jaugeon, B.; Boukal, D.S. Predator Diversity and Environmental Change Modify the Strengths of Trophic and Nontrophic Interactions. Glob. Change Biol. 2017, 23, 2629–2640. [Google Scholar] [CrossRef]
- Alsakaji, H.J.; Kundu, S.; Rihan, F.A. Delay Differential Model of One-Predator Two-Prey System with Monod-Haldane and Holling Type II Functional Responses. Appl. Math. Comput. 2021, 397, 125919. [Google Scholar] [CrossRef]
- Calbet, A.; Landry, M.R. Mesozooplankton Influences on the Microbial Food Web: Direct and Indirect Trophic Interactions in the Oligotrophic Open Ocean. Limnol. Oceanogr. 1999, 44, 1370–1380. [Google Scholar] [CrossRef]
- Schmitz, O.J.; Suttle, K.B. Effects of Top Predator Species on Direct and Indirect Interactions in a Food Web. Ecology 2001, 82, 2072–2081. [Google Scholar] [CrossRef]
- Brooks, A.C.; Gaskell, P.N.; Maltby, L.L. Sublethal Effects and Predator-Prey Interactions: Implications for Ecological Risk Assessment. Environ. Toxicol. Chem. 2009, 28, 2449–2457. [Google Scholar] [CrossRef]
- Klecka, J.; Boukal, D.S. Foraging and Vulnerability Traits Modify Predator–Prey Body Mass Allometry: Freshwater Macroinvertebrates as a Case Study. J. Anim. Ecol. 2013, 82, 1031–1041. [Google Scholar] [CrossRef]
- Wasserman, R.J.; Alexander, M.E.; Dalu, T.; Ellender, B.R.; Kaiser, H.; Weyl, O.L.F. Using Functional Responses to Quantify Interaction Effects among Predators. Funct. Ecol. 2016, 30, 1988–1998. [Google Scholar] [CrossRef]
- Mofu, L.; South, J.; Wasserman, R.J.; Dalu, T.; Woodford, D.J.; Dick, J.T.A.; Weyl, O.L.F. Inter-Specific Differences in Invader and Native Fish Functional Responses Illustrate Neutral Effects on Prey but Superior Invader Competitive Ability. Freshw. Biol. 2019, 64, 1655–1663. [Google Scholar] [CrossRef]
- Barson, M.; Nhiwatiwa, T. Influence of Drought and Flooding on the Colonisation of Floodplain Pans by Riverine Fish in the Zimbabwean Lowveld. Afr. J. Aquat. Sci. 2010, 35, 205–208. [Google Scholar] [CrossRef]
- Nhiwatiwa, T.; Maseko, Z.; Dalu, T. Fish Communities in Small Subtropical Reservoirs Subject to Extensive Drawdowns, with Focus on the Biology of Enteromius Paludinosus (Peters, 1852) and Clarias Gariepinus (Burchell, 1822). Ecol. Res. 2017, 32, 971–982. [Google Scholar] [CrossRef]
- De Moor, I.J.; Bruton, M.N. Atlas of Alien and Translocated Indigenous Aquatic Animals in Southern Africa; National Scientific Programmes Unit: CSIR, 1988; Available online: http://hdl.handle.net/10204/2416 (accessed on 22 December 2021).
- Mattson, N.S. Trophic Interactions between Two Tropical Omnivorous Fishes, Oreochromis Shiranus and Barbus Paludinosus: Feeding Selectivities and Food Web Responses. Hydrobiologia 1998, 380, 195. [Google Scholar] [CrossRef]
- Rapatsa, M.M.; Moyo, N.A.G. The Functional Response of Two Aquatic Predatory Insects and Its Implications for the Biological Control of Coptodon Rendalli Fry. Ecohydrol. Hydrobiol. 2021, 21, 106–113. [Google Scholar] [CrossRef]
- Boets, P.; Laverty, C.; Fukuda, S.; Verreycken, H.; Green, K.; Britton, R.J.; Caffrey, J.; Goethals, P.L.M.; Pegg, J.; Médoc, V.; et al. Intra- and Intercontinental Variation in the Functional Responses of a High Impact Alien Invasive Fish. Biol. Invasions 2019, 21, 1751–1762. [Google Scholar] [CrossRef]
- Weyl, O.L.F.; Pattrick, P.; Ellender, B.R.; Miya, T.; Woodford, D.J.; Bennett, R.H.; Wasserman, R.J.; Äkinen, T.M. Ethical Considerations for Field Research on Fishes. Koedoe Afr. Prot. Area Conserv. Sci. 2016, 58, a1353. [Google Scholar] [CrossRef] [Green Version]
- Length, R. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. R Package Version 1.4.8. 2020. Available online: https://cran.r-project.org/web/packages/emmeans/emmeans.pdf (accessed on 27 December 2021).
- Juliano, S. Non-Linear Curve Fitting: Predation and Functional Response Curve. Des. Anal. Ecol. Exp. 2001, 178–196. [Google Scholar] [CrossRef]
- Pritchard, D.W.; Paterson, R.A.; Bovy, H.C.; Barrios-O’Neill, D. FRAIR: An R Package for Fitting and Comparing Consumer. Funct. Responses 2017, 8, 1528–1534. [Google Scholar]
- Rogers, D. Random Search and Insect Population Models. J. Anim. Ecol. 1972, 41, 369–383. [Google Scholar] [CrossRef]
- Bolker, B.M. Ecological Models and Data in R; Princeton University Press: Vienna, Austria, 2008; ISBN 9781400840908. [Google Scholar]
- Cuthbert, R.N.; Wasserman, R.J.; Dalu, T.; Kaiser, H.; Weyl, O.L.F.; Dick, J.T.A.; Sentis, A.; McCoy, M.W.; Alexander, M.E. Influence of Intra- and Interspecific Variation in Predator–Prey Body Size Ratios on Trophic Interaction Strengths. Ecol. Evol. 2020, 10, 5946–5962. [Google Scholar] [CrossRef] [PubMed]
- Soetaert, K.; Petzoldt, T. Inverse Modelling, Sensitivity and Monte Carlo Analysis in R Using Package FME. J. Stat. Softw. 2010, 33, 1–28. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2020. Available online: https://www.R-project.org/ (accessed on 27 December 2021).
- Cuthbert, R.N.; Dalu, T.; Wasserman, R.J.; Callaghan, A.; Weyl, O.L.F.; Dick, J.T.A. Using Functional Responses to Quantify Notonectid Predatory Impacts across Increasingly Complex Environments. Acta Oecologica 2019, 95, 116–119. [Google Scholar] [CrossRef] [Green Version]
- Heck, K.L.; Crowder, L.B. Habitat Structure and Predator—Prey Interactions in Vegetated Aquatic Systems. In Habitat Structure: The Physical Arrangement of Objects in Space; Bell, S.S., McCoy, E.D., Mushinsky, H.R., Eds.; Population and Community Biology Series; Springer: Dordrecht, The Netherlands, 1991; pp. 281–299. ISBN 9789401130769. [Google Scholar]
- Wasserman, R.J.; Noyon, M.; Avery, T.S.; Froneman, P.W. Trophic Level Stability-Inducing Effects of Predaceous Early Juvenile Fish in an Estuarine Mesocosm Study. PLoS ONE 2013, 8, e61019. [Google Scholar] [CrossRef] [Green Version]
- Palacios, M.M.; Malerba, M.E.; McCormick, M.I. Multiple Predator Effects on Juvenile Prey Survival. Oecologia 2018, 188, 417–427. [Google Scholar] [CrossRef]
- Metcalfe, N.B.; Huntingford, F.A.; Thorpe, J.E. The Influence of Predation Risk on the Feeding Motivation and Foraging Strategy of Juvenile Atlantic Salmon. Anim. Behav. 1987, 35, 901–911. [Google Scholar] [CrossRef]
- Labropoulou, M.; Eleftheriou, A. The Foraging Ecology of Two Pairs of Congeneric Demersal Fish Species: Importance of Morphological Characteristics in Prey Selection. J. Fish Biol. 1997, 50, 324–340. [Google Scholar] [CrossRef]
- Wilbur, H.M.; Fauth, J.E. Experimental Aquatic Food Webs: Interactions between Two Predators and Two Prey. Am. Nat. 1990, 135, 176–204. [Google Scholar] [CrossRef]
- Vucetich, J.A.; Hebblewhite, M.; Smith, D.W.; Peterson, R.O. Predicting Prey Population Dynamics from Kill Rate, Predation Rate and Predator–Prey Ratios in Three Wolf-Ungulate Systems. J. Anim. Ecol. 2011, 80, 1236–1245. [Google Scholar] [CrossRef]
- Uiterwaal, S.F.; DeLong, J.P. Multiple Factors, Including Arena Size, Shape the Functional Responses of Ladybird Beetles. J. Appl. Ecol. 2018, 55, 2429–2438. [Google Scholar] [CrossRef]
- Cuthbert, R.N.; Dalu, T.; Wasserman, R.J.; Weyl, O.L.F.; Froneman, P.W.; Callaghan, A.; Dick, J.T.A. Additive Multiple Predator Effects of Two Specialist Paradiaptomid Copepods towards Larval Mosquitoes. Limnologica 2019, 79, 125727. [Google Scholar] [CrossRef]
- Barrios-O’Neill, D.; Dick, J.T.A.; Emmerson, M.C.; Ricciardi, A.; MacIsaac, H.J. Predator-Free Space, Functional Responses and Biological Invasions. Funct. Ecol. 2015, 29, 377–384. [Google Scholar] [CrossRef]
- Uiterwaal, S.F.; Lagerstrom, I.T.; Lyon, S.R.; DeLong, J.P. Data Paper: FoRAGE (Functional Responses from Around the Globe in All Ecosystems) Database: A Compilation of Functional Responses for Consumers and Parasitoids. BioRxiv 2018. [Google Scholar] [CrossRef] [Green Version]
- Thorp, C.J.; Alexander, M.E.; Vonesh, J.R.; Measey, J. Size-Dependent Functional Response of Xenopus Laevis Feeding on Mosquito Larvae. PeerJ 2018, 6, e5813. [Google Scholar] [CrossRef] [Green Version]
- Cuthbert, R.N.; Dalu, T.; Wasserman, R.J.; Monaco, C.J.; Callaghan, A.; Weyl, O.L.F.; Dick, J.T.A. Assessing Multiple Predator, Diurnal and Search Area Effects on Predatory Impacts by Ephemeral Wetland Specialist Copepods. Aquat. Ecol. 2020, 54, 181–191. [Google Scholar] [CrossRef] [Green Version]
- Escalera-Vázquez, L.H.; Zambrano, L. The Effect of Seasonal Variation in Abiotic Factors on Fish Community Structure in Temporary and Permanent Pools in a Tropical Wetland. Freshw. Biol. 2010, 55, 2557–2569. [Google Scholar] [CrossRef]
- Barrios-O’Neill, D.; Kelly, R.; Dick, J.T.A.; Ricciardi, A.; MacIsaac, H.J.; Emmerson, M.C. On the Context-Dependent Scaling of Consumer Feeding Rates. Ecol. Lett. 2016, 19, 668–678. [Google Scholar] [CrossRef] [Green Version]
- Yam, R.S.; Huang, K.P.; Hsieh, H.L.; Lin, H.J.; Huang, S.C. An ecosystem-service approach to evaluate the role of non-native species in urbanized wetlands. Int. J. Environ. Res. Public Health 2015, 12, 3926–3943. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munyai, L.F.; Dalu, T.; Wasserman, R.J.; Mugwedi, L.; Dondofema, F.; O’Brien, G.; Cuthbert, R.N. Functional Responses and Additive Multiple Predator Effects of Two Common Wetland Fish. Water 2022, 14, 699. https://doi.org/10.3390/w14050699
Munyai LF, Dalu T, Wasserman RJ, Mugwedi L, Dondofema F, O’Brien G, Cuthbert RN. Functional Responses and Additive Multiple Predator Effects of Two Common Wetland Fish. Water. 2022; 14(5):699. https://doi.org/10.3390/w14050699
Chicago/Turabian StyleMunyai, Linton F., Tatenda Dalu, Ryan J. Wasserman, Lutendo Mugwedi, Farai Dondofema, Gordon O’Brien, and Ross N. Cuthbert. 2022. "Functional Responses and Additive Multiple Predator Effects of Two Common Wetland Fish" Water 14, no. 5: 699. https://doi.org/10.3390/w14050699
APA StyleMunyai, L. F., Dalu, T., Wasserman, R. J., Mugwedi, L., Dondofema, F., O’Brien, G., & Cuthbert, R. N. (2022). Functional Responses and Additive Multiple Predator Effects of Two Common Wetland Fish. Water, 14(5), 699. https://doi.org/10.3390/w14050699






