Development of a Risk Characterization Tool for Harmful Cyanobacteria Blooms on the Ohio River
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ohio River State and Water Data Sources
2.2. Compiling River Discharge Time Series
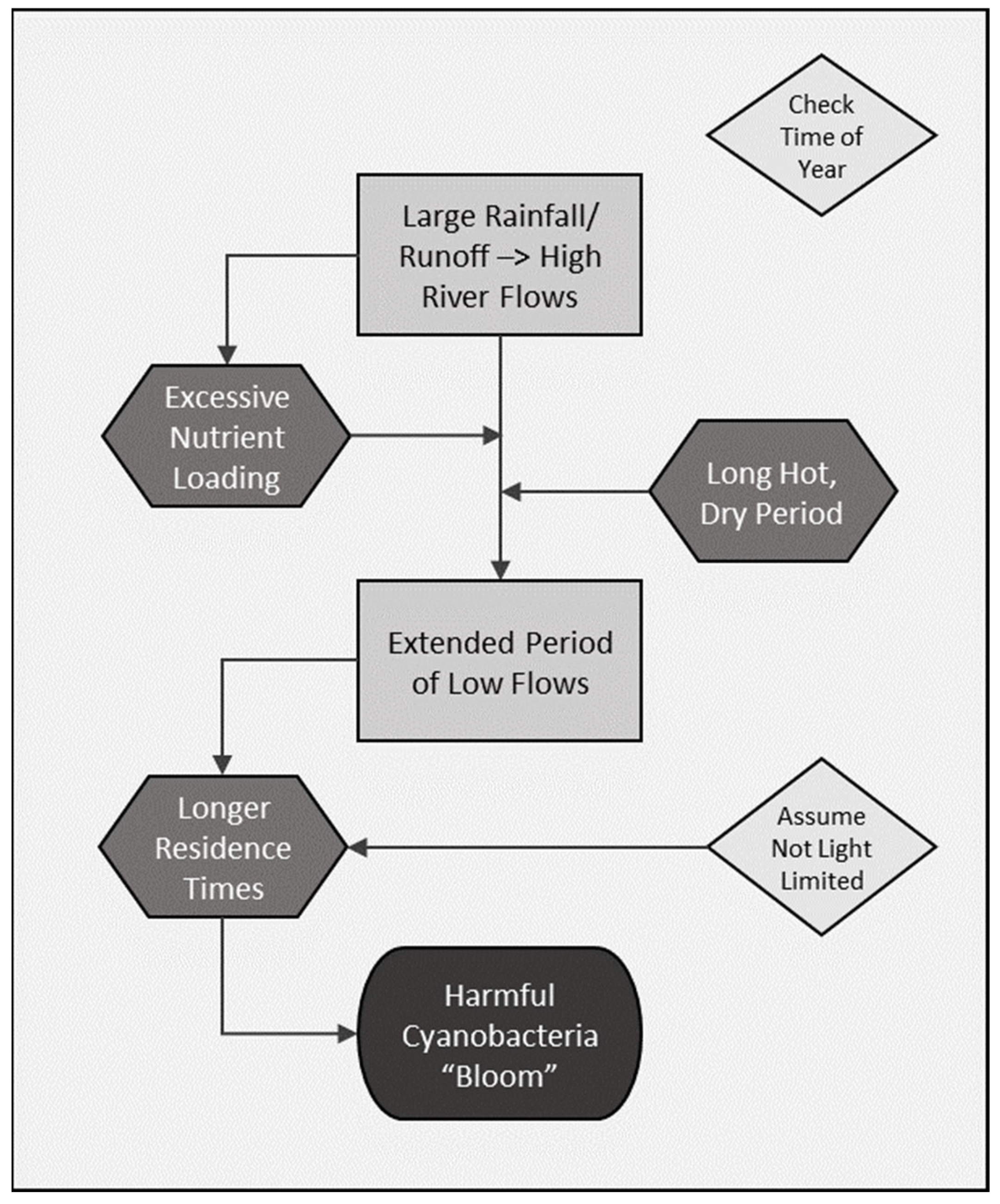
2.3. Guiding Conceptual Model of cyanoHAB Ecology
2.4. Model Development
2.4.1. Predictor Variables
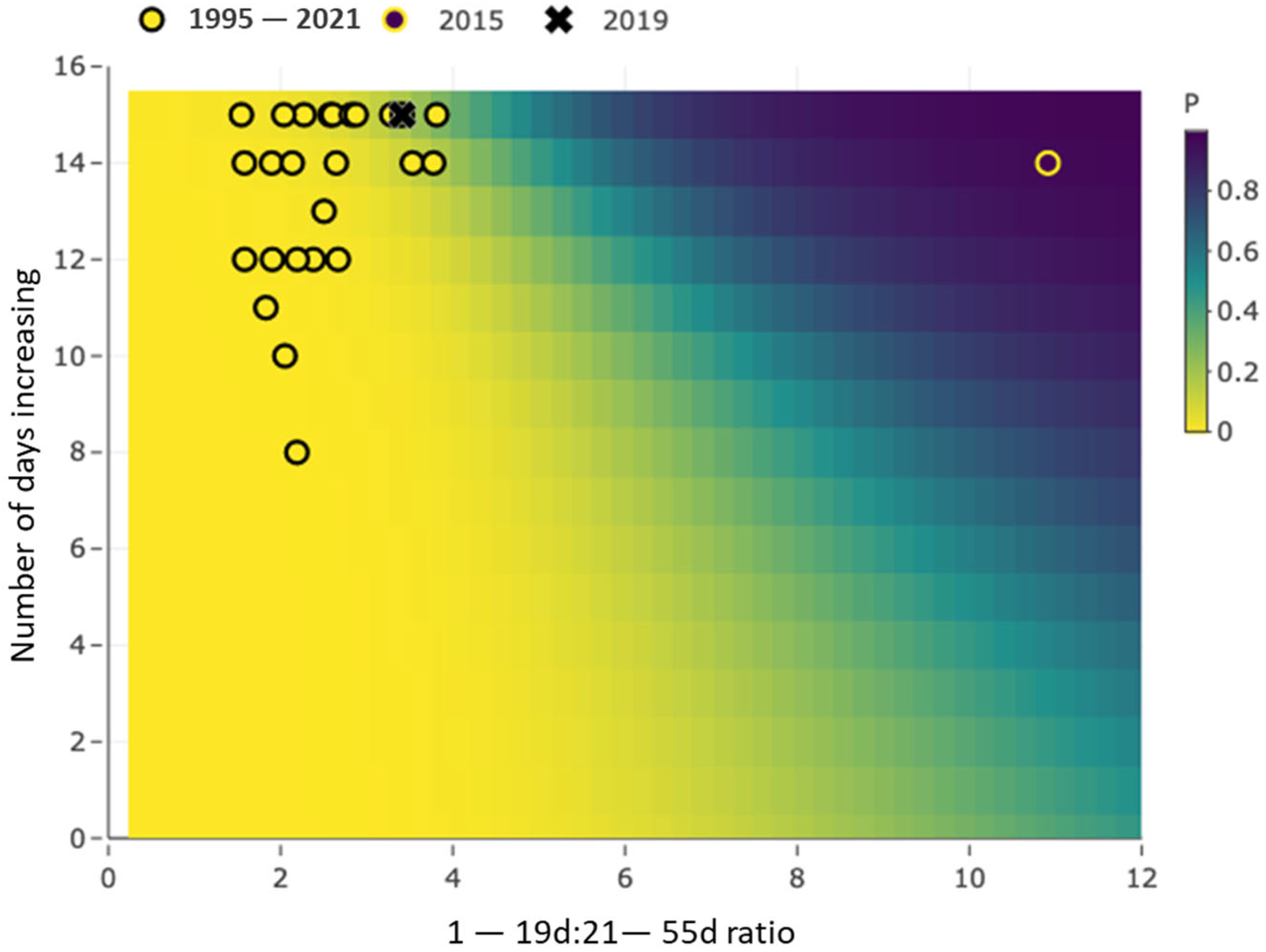
2.4.2. Occurrence Model
2.4.3. Persistence Model
2.5. Water Quality Data Visualization
2.6. R Programming and Risk Characterization Tool
3. Results
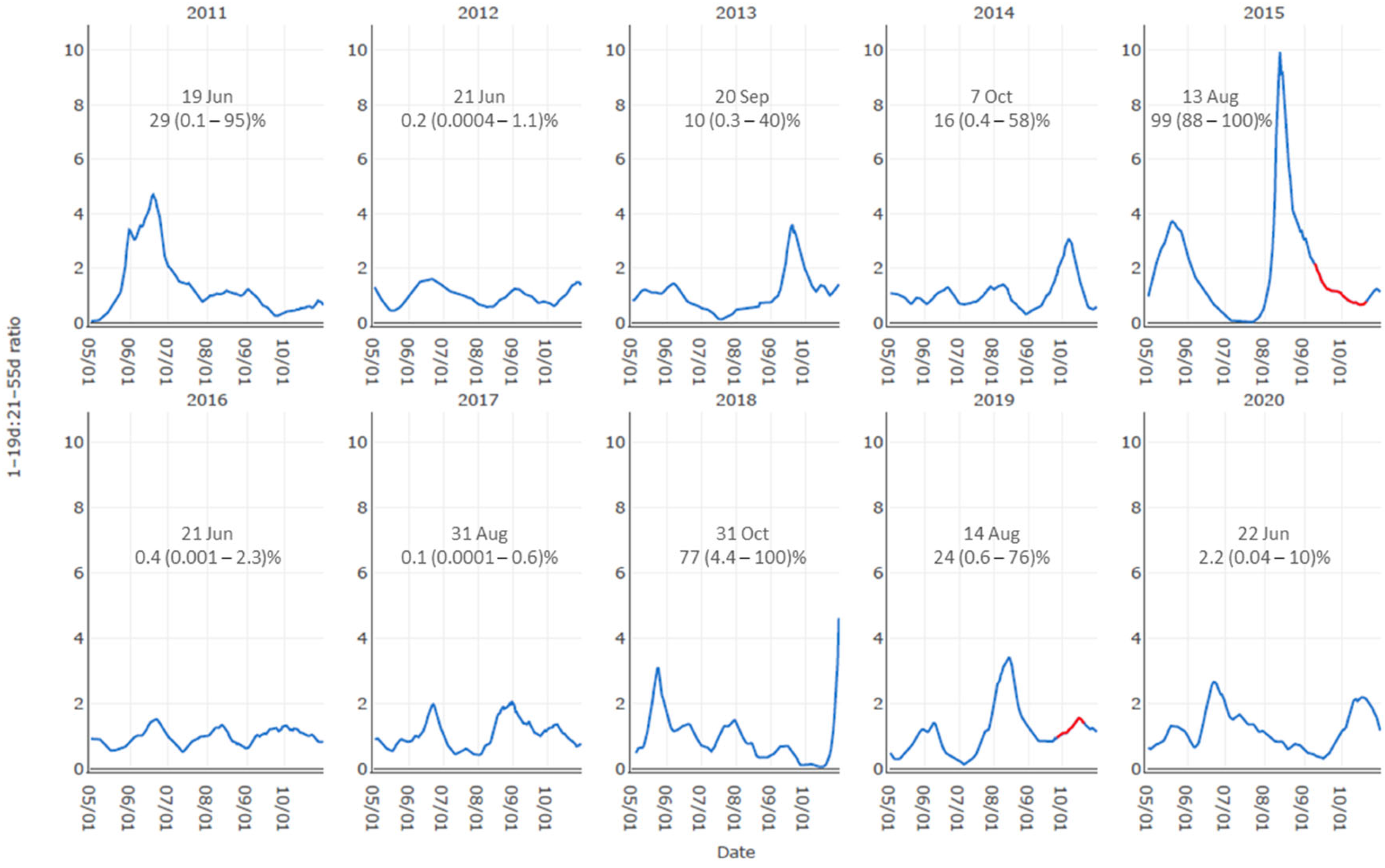
3.1. Supporting Evidence for Key Drivers in the Conceptual Model
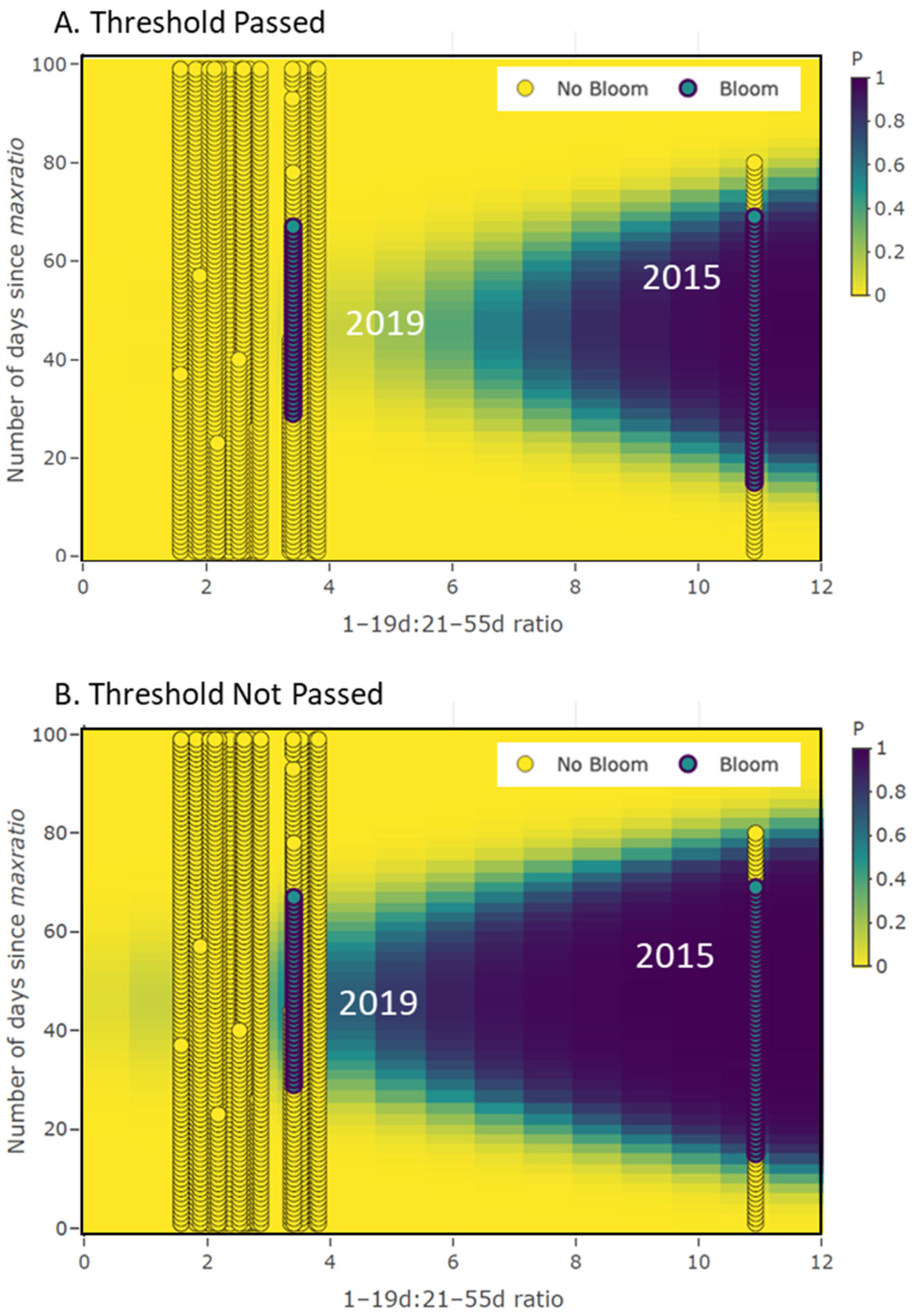
3.2. Occurrence Model Results Demonstration
3.3. Persistence Model Results Demonstration
3.4. Shiny App Real-Time Reporting
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ho, J.C.; Michalak, A.M.; Pahlevan, N. Widespread global increase in intense lake phytoplankton blooms since the 1980s. Nature 2019, 574, 667–670. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, M.; Pick, F.; Gregory-Eaves, I. Nutrients and water temperature are significant predictors of cyanobacterial biomass in a 1147 lakes data set. Limnol. Oceanogr. 2013, 58, 1736–1746. [Google Scholar] [CrossRef]
- Adams, C.M.; Larkin, S.L.; Hoagland, P.; Sancewich, B. Assessing the Economic Consequences of Harmful Algal Blooms. In Harmful Algal Blooms: A Compendium Desk Reference; Shumway, S.E., Burkholder, J., Morton, S.L., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 337–354. [Google Scholar]
- Hilborn, E.D.; Beasley, V.R. One health and cyanobacteria in freshwater systems: Animal illnesses and deaths are sentinel events for human health risks. Toxins 2015, 7, 1374–1395. [Google Scholar] [CrossRef] [Green Version]
- Backer, L.C.; Manassaram-Baptiste, D.; LePrell, R.; Bolton, B. Cyanobacteria and Algae Blooms: Review of Health and Environmental Data from the Harmful Algal Bloom-Related Illness Surveillance System (HABISS) 2007–2011. Toxins 2015, 7, 1048–1064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- USEPA. Cyanobacteria and Cyanotoxins: Information for Drinking Water Systems; EPA-810F11001; USEPA, Office of Water: Washington, DC, USA, 2012; pp. 1–9.
- Roberts, V.A.; Vigar, M.; Backer, L.; Veytsel, G.E.; Hilborn, E.D.; Hamilton, E.I.; Vanden Esschert, K.L.; Lively, J.Y.; Cope, J.R.; Hlavsa, M.C.; et al. Surveillance for Harmful Algal Bloom Events and Associated Human and Animal Illnesses—One Health Harmful Algal Bloom System, United States, 2016–2018. Morb. Mortal. Wkly. Rep. 2020, 1889–1894. [Google Scholar] [CrossRef]
- Pelaez, M.; Antoniou, M.G.; He, X.; Dionysiou, D.D.; de la Cruz, A.A.; Tsimeli, K.; Triantis, T.; Hiskia, A.; Kaloudis, T.; Williams, C.; et al. Sources and Occurrence of Cyanotoxins Worldwide. In Xenobiotics in the Urban Water Cycle: Mass Flows, Environmental Processes, Mitigation and Treatment Strategies; Fatta-Kassinos, D., Bester, K., Kümmerer, K., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 101–127. [Google Scholar]
- Bullerjahn, G.S.; McKay, R.M.; Davis, T.W.; Baker, D.B.; Boyer, G.L.; D’Anglada, L.V.; Doucette, G.J.; Ho, J.C.; Irwin, E.G.; Kling, C.L.; et al. Global solutions to regional problems: Collecting global expertise to address the problem of harmful cyanobacterial blooms. A Lake Erie case study. Harmful Algae 2016, 54, 223–238. [Google Scholar] [CrossRef] [Green Version]
- Dolman, A.M.; Rücker, J.; Pick, F.R.; Fastner, J.; Rohrlack, T.; Mischke, U.; Wiedner, C. Cyanobacteria and Cyanotoxins: The Influence of Nitrogen versus Phosphorus. PLoS ONE 2012, 7, e38757. [Google Scholar] [CrossRef]
- Watson, S.B.; Miller, C.; Arhonditsis, G.; Boyer, G.L.; Carmichael, W.; Charlton, M.N.; Confesor, R.; Depew, D.C.; Höök, T.O.; Ludsin, S.A.; et al. The re-eutrophication of Lake Erie: Harmful algal blooms and hypoxia. Harmful Algae 2016, 56, 44–66. [Google Scholar] [CrossRef]
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Newell, S.E.; Davis, T.W.; Johengen, T.H.; Gossiaux, D.; Burtner, A.; Palladino, D.; McCarthy, M.J. Reduced forms of nitrogen are a driver of non-nitrogen-fixing harmful cyanobacterial blooms and toxicity in Lake Erie. Harmful Algae 2019, 81, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; King, K.W.; Williams, M.R. What is causing the harmful algal blooms in Lake Erie. J. Soil Water Conserv. 2015, 70, 27A–29A. [Google Scholar] [CrossRef] [Green Version]
- Gobler, C.J. Climate Change and Harmful Algal Blooms: Insights and perspective. Harmful Algae 2020, 91, 101731. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Wan, L.; Chen, X.; Deng, Q.; Yang, L.; Li, X.; Zhang, J.; Song, C.; Zhou, Y.; Cao, X. Phosphorus strategy in bloom-forming cyanobacteria (Dolichospermum and Microcystis) and its role in their succession. Harmful Algae 2019, 84, 46–55. [Google Scholar] [CrossRef]
- Lürling, M.; van Oosterhout, F.; Faassen, E. Eutrophication and Warming Boost Cyanobacterial Biomass and Microcystins. Toxins 2017, 9, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brooks, B.W.; Lazorchak, J.M.; Howard, M.D.; Johnson, M.V.; Morton, S.L.; Perkins, D.A.; Reavie, E.D.; Scott, G.I.; Smith, S.A.; Steevens, J.A. Are harmful algal blooms becoming the greatest inland water quality threat to public health and aquatic ecosystems? Environ. Toxicol. Chem. 2016, 35, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Chapra, S.C.; Boehlert, B.; Fant, C.; Bierman, V.J.; Henderson, J.; Mills, D.; Mas, D.M.L.; Rennels, L.; Jantarasami, L.; Martinich, J.; et al. Climate Change Impacts on Harmful Algal Blooms in U.S. Freshwaters: A Screening-Level Assessment. Environ. Sci. Technol. 2017, 51, 8933–8943. [Google Scholar] [CrossRef]
- Wiedner, C.; Rücker, J.; Brüggemann, R.; Nixdorf, B. Climate change affects timing and size of populations of an invasive cyanobacterium in temperate regions. Oecologia 2007, 152, 473–484. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef]
- Kosten, S.; Huszar, V.L.M.; Bécares, E.; Costa, L.S.; van Donk, E.; Hansson, L.-A.; Jeppesen, E.; Kruk, C.; Lacerot, G.; Mazzeo, N.; et al. Warmer climates boost cyanobacterial dominance in shallow lakes. Glob. Chang. Biol. 2012, 18, 118–126. [Google Scholar] [CrossRef]
- Moss, B.; Kosten, S.; Meerhoff, M.; Battarbee, R.W.; Jeppesen, E.; Mazzeo, N.; Havens, K.; Lacerot, G.; Liu, Z.; De Meester, L.; et al. Allied attack: Climate change and eutrophication. Inland Waters 2011, 1, 101–105. [Google Scholar] [CrossRef] [Green Version]
- Carey, C.C.; Ibelings, B.W.; Hoffmann, E.P.; Hamilton, D.P.; Brookes, J.D. Eco-physiological adaptations that favour freshwater cyanobacteria in a changing climate. Water Res. 2012, 46, 1394–1407. [Google Scholar] [CrossRef]
- Lurling, M.; Eshetu, F.; Faassen, E.J.; Kosten, S.; Huszar, V.L.M. Comparison of cyanobacterial and green algal growth rates at different temperatures. Freshw. Biol. 2013, 58, 552–559. [Google Scholar] [CrossRef]
- Rigosi, A.; Carey, C.C.; Ibelings, B.W.; Brookes, J.D. The interaction between climate warming and eutrophication to promote cyanobacteria is dependent on trophic state and varies among taxa. Limnol. Oceanogr. 2014, 59, 99–114. [Google Scholar] [CrossRef] [Green Version]
- Smucker, N.J.; Beaulieu, J.J.; Nietch, C.T.; Young, J.L. Increasingly severe cyanobacterial blooms and deep water hypoxia coincide with warming water temperatures in reservoirs. Glob. Chang. Biol. 2021, 27, 2507–2519. [Google Scholar] [CrossRef]
- Wagner, C.; Adrian, R. Cyanobacteria dominance: Quantifying the effects of climate change. Limnol. Oceanogr. 2009, 54, 2460–2468. [Google Scholar] [CrossRef]
- Huber, V.; Wagner, C.; Gerten, D.; Adrian, R. To bloom or not to bloom: Contrasting responses of cyanobacteria to recent heat waves explained by critical thresholds of abiotic drivers. Oecologia 2012, 169, 245–256. [Google Scholar] [CrossRef]
- Cottingham, K.L.; Ewing, H.A.; Greer, M.L.; Carey, C.C.; Weathers, K.C. Cyanobacteria as biological drivers of lake nitrogen and phosphorus cycling. Ecosphere 2015, 6, 1–19. [Google Scholar] [CrossRef]
- Posch, T.; Köster, O.; Salcher, M.M.; Pernthaler, J. Harmful filamentous cyanobacteria favoured by reduced water turnover with lake warming. Nat. Clim. Chang. 2012, 2, 809–813. [Google Scholar] [CrossRef] [Green Version]
- Graham, J.L.; Dubrovsky, N.M.; Foster, G.M.; King, L.R.; Loftin, K.A.; Rosen, B.H.; Stelzer, E.A. Cyanotoxin occurrence in large rivers of the United States. Inland Waters 2020, 10, 109–117. [Google Scholar] [CrossRef]
- Loftin, K.A.; Clark, J.M.; Journey, C.A.; Kolpin, D.W.; Van Metre, P.C.; Bradley, P.M. Spatial and temporal variation in microcystins occurrence in wadeable streams in the southeastern USA. Environ. Toxicol. Chem. 2016, 35, 2281–2287. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Oliver, R.L.; Rees, C.; Bowling, L.C.; Buckney, R.T. Critical flow velocities for the growth and dominance of Anabaena circinalis in some turbid freshwater rivers. Freshw. Biol. 2003, 48, 164–174. [Google Scholar] [CrossRef] [Green Version]
- Quiblier, C.; Wood, S.; Echenique-Subiabre, I.; Heath, M.W.; Villeneuve, A.; Humbert, J.-F. A review of current knowledge on toxic benthic freshwater cyanobacteria—Ecology, toxin production and risk management. Water Res. 2013, 47, 5464–5479. [Google Scholar] [CrossRef]
- Wood, S.A.; Kelly, L.T.; Bouma-Gregson, K.; Humbert, J.-F.; Laughinghouse IV, H.D.; Lazorchak, J.; McAllister, T.G.; McQueen, A.; Pokrzywinski, K.; Puddick, J.; et al. Toxic benthic freshwater cyanobacterial proliferations: Challenges and solutions for enhancing knowledge and improving monitoring and mitigation. Freshw. Biol. 2020, 65, 1824–1842. [Google Scholar] [CrossRef]
- Cha, Y.K.; Cho, K.H.; Lee, H.; Kang, T.; Kim, J.H. The relative importance of water temperature and residence time in predicting cyanobacteria abundance in regulated rivers. Water Res. 2017, 124, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Seo, I.W.; Baek, D. Seasonally varying effects of environmental factors on phytoplankton abundance in the regulated rivers. Sci. Rep. 2019, 9, 9266. [Google Scholar] [CrossRef]
- Youngstrom, G. ORSANCO Harmful Algae Bloom Monitoring, Response and Communication Plan—Draft; Ohio River Water Sanitation Commission: Cincinnati, OH, USA, 2020; Available online: https://www.orsanco.org/wp-content/uploads/2017/2003/2020HABMonitoringandResponsePlanDraft.pdf (accessed on 29 December 2021).
- Ohio River Valley Water Sanitation Commission. Ohio River Harmful Algae Blooms. Available online: http://www.orsanco.org/data/harmful-algae-bloom-data/ (accessed on 1 January 2021).
- Wines, M. Toxic Algae Outbreak Overwhelms a Polluted Ohio River; The New York Times: New York, NY, USA, 2015. [Google Scholar]
- Beck, R.; Xu, M.; Zhan, S.; Johansen, R.; Liu, H.; Tong, S.; Yang, B.; Shu, S.; Wu, Q.; Wang, S.; et al. Comparison of satellite reflectance algorithms for estimating turbidity and cyanobacterial concentrations in productive freshwaters using hyperspectral aircraft imagery and dense coincident surface observations. J. Great Lakes Res. 2019, 45, 413–433. [Google Scholar] [CrossRef]
- Johansen, R.; Beck, R.; Nowosad, J.; Nietch, C.; Xu, M.; Shu, S.; Yang, B.; Liu, H.; Emery, E.; Reif, M.; et al. Evaluating the portability of satellite derived chlorophyll-a algorithms for temperate inland lakes using airborne hyperspectral imagery and dense surface observations. Harmful Algae 2018, 76, 35–46. [Google Scholar] [CrossRef]
- Papenfus, M.; Schaeffer, B.; Pollard, A.I.; Loftin, K. Exploring the potential value of satellite remote sensing to monitor chlorophyll-a for US lakes and reservoirs. Environ. Monit. Assess. 2020, 192, 808. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, B.A.; Bailey, S.W.; Conmy, R.N.; Galvin, M.; Ignatius, A.R.; Johnston, J.M.; Keith, D.J.; Lunetta, R.S.; Parmar, R.; Stumpf, R.P.; et al. Mobile device application for monitoring cyanobacteria harmful algal blooms using Sentinel-3 satellite Ocean and Land Colour Instruments. Environ. Model. Softw. 2018, 109, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Ohio River Valley Water Sanitation Commission. The State of the Ohio River; Ohio River Valley Water Sanitation Commission: Cincinnati, OH, USA, 2017; p. 8. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Gelman, A.; Carlin, J.B.; Stern, H.S.S.; Dunson, D.B.; Vehtari, A.; Rubin, D.B. Bayesian Data Analysis, 3rd ed.; Chapman & Hall/CRC Texts in Statistical Science: Boca Raton, FL, USA, 2013. [Google Scholar]
- Stan Development Team. “RStan: The R Interface to Stan”. R Package Version 2.21.3. 2021. Available online: https://mc-stan.org/ (accessed on 29 December 2021).
- R-Core-Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- National Oceanic Atmospheric Association. Lake Erie Harmful Algal Bloom Forecast. Available online: https://tidesandcurrents.noaa.gov/hab/lakeerie.html (accessed on 28 December 2021).
- Rowe, M.D.; Anderson, E.J.; Wynne, T.T.; Stumpf, R.P.; Fanslow, D.L.; Kijanka, K.; Vanderploeg, H.A.; Strickler, J.R.; Davis, T.W. Vertical distribution of buoyant Microcystis blooms in a Lagrangian particle tracking model for short-term forecasts in Lake Erie. J. Geophys. Res. Oceans 2016, 121, 5296–5314. [Google Scholar] [CrossRef]
- Myer, M.H.; Urquhart, E.; Schaeffer, B.A.; Johnston, J.M. Spatio-Temporal Modeling for Forecasting High-Risk Freshwater Cyanobacterial Harmful Algal Blooms in Florida. Front. Environ. Sci. 2020, 8, 581091. [Google Scholar] [CrossRef]
- Ohio River Water Sanitation Commission. Lower Wabash River Nutrients and Continuous Monitoring Project; Ohio River Valley Water Sanitation Commission: Cincinnati, OH, USA, 2014. [Google Scholar]
- Sellers, T.; Bukaveckas, P.A. Phytoplankton production in a large, regulated river: A modeling and mass balance assessment. Limnol. Oceanogr. 2003, 48, 1476–1487. [Google Scholar] [CrossRef]
- Graham, J.L.; Ziegler, A.C.; Loving, B.L.; Loftin, K.A. Fate and Transport of Cyanobacteria and Associated Toxins and Taste and-Odor Compounds from Upstream Reservoir Releases in the Kansas River, Kansas September and October 2011; sir2012–5129; US Geological Survey: Reston, VA, USA, 2012.
- Wilkinson, A.A.; Hondzo, M.; Guala, M. Investigating Abiotic Drivers for Vertical and Temporal Heterogeneities of Cyanobacteria Concentrations in Lakes Using a Seasonal In-situ Monitoring Station. Water Resour. Res. 2019, 55, 954–972. [Google Scholar] [CrossRef]
- Francy, D.S.; Brady, A.M.G.; Ecker, C.D.; Graham, J.L.; Stelzer, E.A.; Struffolino, P.; Dwyer, D.F.; Loftin, K.A. Estimating microcystin levels at recreational sites in western Lake Erie and Ohio. Harmful Algae 2016, 58, 23–34. [Google Scholar] [CrossRef]
- Francy, D.S.; Graham, J.L.; Stelzer, E.A.; Ecker, C.D.; Brady, A.M.G.; Pam, S.; Loftin, K.A. Water Quality, Cyanobacteria, and Environmental Factors and Their Relations to Microcystin Concentrations for Use in Predictive Models at Ohio Lake Erie and Inland Lake Recreational Sites, 2013–2014; sir2015–5120; US Geological Survey: Reston, VA, USA, 2015; p. 70.
- Marion, J.W.; Lee, J.; Wilkins, J.R.; Lemeshow, S.; Lee, C.; Waletzko, E.J.; Buckley, T.J. In Vivo Phycocyanin Flourometry as a Potential Rapid Screening Tool for Predicting Elevated Microcystin Concentrations at Eutrophic Lakes. Environ. Sci. Technol. 2012, 46, 4523–4531. [Google Scholar] [CrossRef]
- Francy, D.S.; Brady, A.M.G.; Zimmerman, T.M. Real-Time Assessments of Water Quality—A Nowcast for Escherichia coli and Cyanobacterial Toxins; sir 2019–3061; US Geological Survey: Reston, VA, USA, 2019; p. 4.
- Pace, M.L.; Batt, R.D.; Buelo, C.D.; Carpenter, S.R.; Cole, J.J.; Kurtzweil, J.T.; Wilkinson, G.M. Reversal of a cyanobacterial bloom in response to early warnings. Proc. Natl. Acad. Sci. USA 2017, 114, 352–357. [Google Scholar] [CrossRef] [Green Version]
- Chaffin, J.D.; Kane, D.D.; Johnson, A. Effectiveness of a fixed-depth sensor deployed from a buoy to estimate water-column cyanobacterial biomass depends on wind speed. J. Environ. Sci. 2020, 93, 23–29. [Google Scholar] [CrossRef]
- Chaffin, J.D.; Kane, D.D.; Stanislawczyk, K.; Parker, E.M. Accuracy of data buoys for measurement of cyanobacteria, chlorophyll, and turbidity in a large lake (Lake Erie, North America): Implications for estimation of cyanobacterial bloom parameters from water quality sonde measurements. Environ. Sci. Pollut. Res. 2018, 25, 25175–25189. [Google Scholar] [CrossRef] [PubMed]
- Jeong, K.-S.; Joo, G.-J.; Kim, H.-W.; Ha, K.; Recknagel, F. Prediction and elucidation of phytoplankton dynamics in the Nakdong River (Korea) by means of a recurrent artificial neural network. Ecol. Model. 2001, 146, 115–129. [Google Scholar] [CrossRef]
- Bae, S.; Seo, D. Analysis and modeling of algal blooms in the Nakdong River, Korea. Ecol. Model. 2018, 372, 53–63. [Google Scholar] [CrossRef]
- Kim, K.; Park, M.; Min, J.-H.; Ryu, I.; Kang, M.-R.; Park, L.J. Simulation of algal bloom dynamics in a river with the ensemble Kalman filter. J. Hydrol. 2014, 519, 2810–2821. [Google Scholar] [CrossRef]
- Kim, K.B.; Jung, M.-K.; Tsang, Y.F.; Kwon, H.-H. Stochastic modeling of chlorophyll-a for probabilistic assessment and monitoring of algae blooms in the Lower Nakdong River, South Korea. J. Hazard. Mater. 2020, 400, 123066. [Google Scholar] [CrossRef]
- Kim, S.; Kim, S.; Mehrotra, R.; Sharma, A. Predicting cyanobacteria occurrence using climatological and environmental controls. Water Res. 2020, 175, 115639. [Google Scholar] [CrossRef]
- Kim, Y.W.; Lee, J.H.; Park, T.J.; Byun, I.G. Changes in the water environment and algae generation characteristics in Mulgeum area downstream of the Nakdong River after construction. J. Korean Soc. Hazard Mitig. 2017, 17, 383–392. [Google Scholar] [CrossRef]
- Kim, S.; Mehrotra, R.; Kim, S.; Sharma, A. Probabilistic forecasting of cyanobacterial concentration in riverine systems using environmental drivers. J. Hydrol. 2021, 593, 125626. [Google Scholar] [CrossRef]
- Hampel, J.J.; McCarthy, M.J.; Neudeck, M.; Bullerjahn, G.S.; McKay, R.M.L.; Newell, S.E. Ammonium recycling supports toxic Planktothrix blooms in Sandusky Bay, Lake Erie: Evidence from stable isotope and metatranscriptome data. Harmful Algae 2019, 81, 42–52. [Google Scholar] [CrossRef] [Green Version]
- Jobson, H.E.; Schoellhamer, D.H. Users Manual for a Branched Lagrangian Transport Model; 87-4163; U.S. Geological Survey: Washington, DC, USA, 1993.
- Chen, Y.; Qin, B.; Teubner, K.; Dokulil, M.T. Long-term dynamics of phytoplankton assemblages: Microcystis-domination in Lake Taihu, a large shallow lake in China. J. Plankton Res. 2003, 25, 445–453. [Google Scholar] [CrossRef]
- Deng, J.; Qin, B.; Paerl, H.W.; Zhang, Y.; Ma, J.; Chen, Y. Earlier and warmer springs increase cyanobacterial (Microcystis spp.) blooms in subtropical Lake Taihu, China. Freshw. Biol. 2014, 59, 1076–1085. [Google Scholar] [CrossRef]
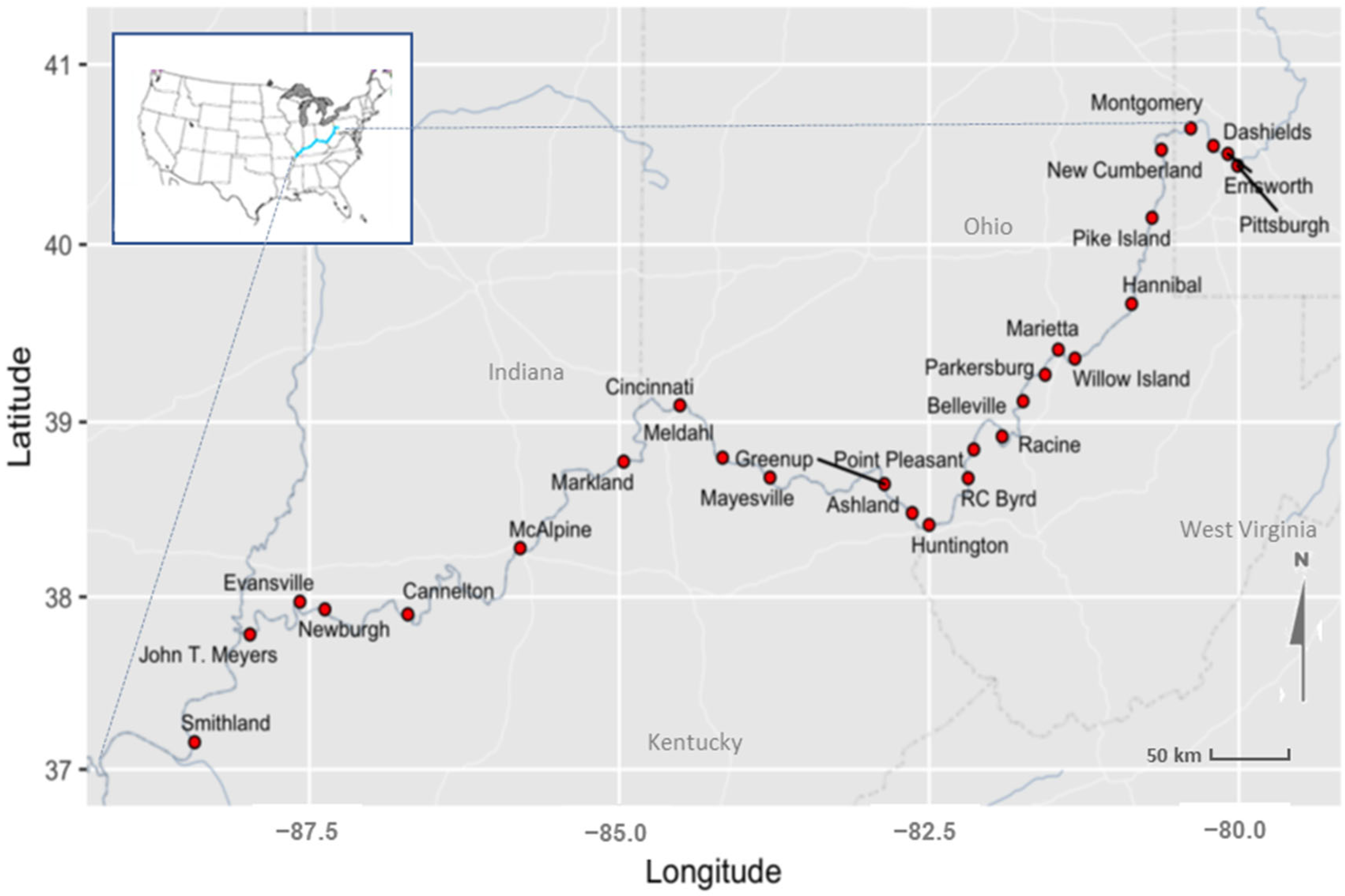



| # | Site Name | Latitude | Longitude | Type | River Miles below Pittsburgh, PA | 2015 HAB (Date First Observed) | 2019 HAB (Date First Observed) | Used for Modeling (+)| Model Results Reported (✓) |
|---|---|---|---|---|---|---|---|---|
| 1 | Pittsburgh | 40.43944 | −80.01083 | Mid-Pool | 0 | No Bloom | No Bloom | NA |
| 2 | Emsworth | 40.50500 | −80.08972 | L&D | 6.2 | No Bloom | No Bloom | ✓ |
| 3 | Dashields | 40.54972 | −80.20694 | L&D | 13.3 | No Bloom | No Bloom | ✓ |
| 4 | Montgomery | 40.64722 | −80.38889 | L&D | 31.7 | No Bloom | No Bloom | ✓ |
| 5 | New Cumberland | 40.52806 | −80.62583 | L&D | 54.4 | No Bloom | No Bloom | ✓ |
| 6 | Pike Island | 40.14972 | −80.70167 | L&D | 84.2 | 19 August 2015 | No Bloom | ✓ |
| 7 | Hannibal | 39.66722 | −80.86611 | L&D | 126.4 | 21 August 2015 | No Bloom | ✓ |
| 8 | Willow Island | 39.35900 | −81.32400 | L&D | 161.7 | 24 August 2015 | No Bloom | ✓ |
| 9 | Marietta | 39.40944 | −81.45778 | Mid-Pool | 172 | 24 August 2015 | No Bloom | + |
| 10 | Parkersburg | 39.26806 | −81.56389 | Mid-Pool | 185 | 24 August 2015 | No Bloom | + |
| 11 | Belleville | 39.11800 | −81.74200 | L&D | 203.9 | 24 August 2015 | No Bloom | ✓ |
| 12 | Racine | 38.91800 | −81.91100 | L&D | 237.5 | 25 August 2015 | No Bloom | ✓ |
| 13 | Point Pleasant | 38.84389 | −82.13972 | Mid-Pool | 265 | 26 August 2015 | No Bloom | + |
| 14 | RC Byrd | 38.68000 | −82.18500 | L&D | 279.2 | 27 August 2015 | No Bloom | ✓ |
| 15 | Huntington | 38.41333 | −82.50056 | Mid-Pool | 312 | 27 August 2015 | 12 September 2019 | ✓ |
| 16 | Ashland | 38.48111 | −82.63667 | Mid-Pool | 322 | 27 August 2015 | 11 September 2019 | + |
| 17 | Greenup | 38.64667 | −82.86056 | L&D | 341 | 27 August 2015 | 12 September 2019 | ✓ |
| 18 | Mayesville | 38.68389 | −83.78389 | Mid-Pool | 409 | 28 August 2015 | 12 September 2019 | + |
| 19 | Meldahl | 38.79722 | −84.16667 | L&D | 436.2 | 1 September 2015 | 17 September 2019 | ✓ |
| 20 | Cincinnati | 39.09444 | −84.51056 | Mid-Pool | 471 | 9 September 2015 | 19 September 2019 | ✓ |
| 21 | Markland | 38.77472 | −84.96444 | L&D | 531.5 | 9 September 2015 | 26 September 2019 | ✓ |
| 22 | McAlpine | 38.28028 | −85.79917 | L&D | 606.8 | 11 September 2015 | 24 September 2019 | ✓ |
| 23 | Cannelton | 37.89944 | −86.70556 | L&D | 720.7 | 15 September 2015 | No Bloom | ✓ |
| 24 | Newburgh | 37.92833 | −87.37500 | L&D | 776.1 | 16 September 2015 | No Bloom | ✓ |
| 25 | Evansville | 37.97222 | −87.57639 | Mid-Pool | 792 | 17 September 2015 | No Bloom | ✓ |
| 26 | John T. Meyers | 37.78333 | −87.97944 | L&D | 846 | 18 September 2015 | No Bloom | NA |
| 27 | Smithland | 37.15833 | −88.42611 | L&D | 918.5 | 19 September 2015 | No Bloom | ✓ |
| Parameter | Variable Name | Effect | Description |
|---|---|---|---|
| Occurrence Model | |||
| X1 | maxratio | fixed | maximum 1–19:21–55 day ratio |
| X2 | inc15 | fixed | Number of days increasing in 15 days prior to maxratio day |
| X3 | meanrt | fixed | Site’s mean residence time |
| X4 | maxratio × meanrt | fixed | Interaction term |
| α1 | na | random | maxratio slope adjustment |
| α0 | na | random | maxratio intercept adjustment |
| β0, …, β4 | na | na | regression coefficients |
| Persistence Model | |||
| X1 | maxratio | fixed | maximum 1–19:21–55 day ratio |
| X2 | days | fixed | Number of days after maxratio |
| X3 | binary indicator (1 or 0) | fixed | indicator of increase in flow |
| X4 | binary indicator × maxratio | fixed | interaction term |
| X5 | meanrt | fixed | site’s mean residence time |
| X6 | maxratio × meanrt | fixed | interaction term |
| α1 | na | random | maxratio slope adjustment |
| α0 | na | random | maxratio intercept adjustment |
| β0, …, β6 | na | na | regression coefficients |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nietch, C.T.; Gains-Germain, L.; Lazorchak, J.; Keely, S.P.; Youngstrom, G.; Urichich, E.M.; Astifan, B.; DaSilva, A.; Mayfield, H. Development of a Risk Characterization Tool for Harmful Cyanobacteria Blooms on the Ohio River. Water 2022, 14, 644. https://doi.org/10.3390/w14040644
Nietch CT, Gains-Germain L, Lazorchak J, Keely SP, Youngstrom G, Urichich EM, Astifan B, DaSilva A, Mayfield H. Development of a Risk Characterization Tool for Harmful Cyanobacteria Blooms on the Ohio River. Water. 2022; 14(4):644. https://doi.org/10.3390/w14040644
Chicago/Turabian StyleNietch, Christopher T., Leslie Gains-Germain, James Lazorchak, Scott P. Keely, Gregory Youngstrom, Emilee M. Urichich, Brian Astifan, Abram DaSilva, and Heather Mayfield. 2022. "Development of a Risk Characterization Tool for Harmful Cyanobacteria Blooms on the Ohio River" Water 14, no. 4: 644. https://doi.org/10.3390/w14040644






