Kinetic Study of the Anaerobic Digestion of Recycled Paper Mill Effluent (RPME) by Using a Novel Modified Anaerobic Hybrid Baffled (MAHB) Reactor
Abstract
1. Introduction
2. Materials and Methods
2.1. Equipment
2.2. Inoculum and Wastewater Preparation
2.3. Kinetic Study
2.4. Analytical Method
3. Results
3.1. Kinetic Study of Anaerobic Digestion by Using an MAHB Reactor
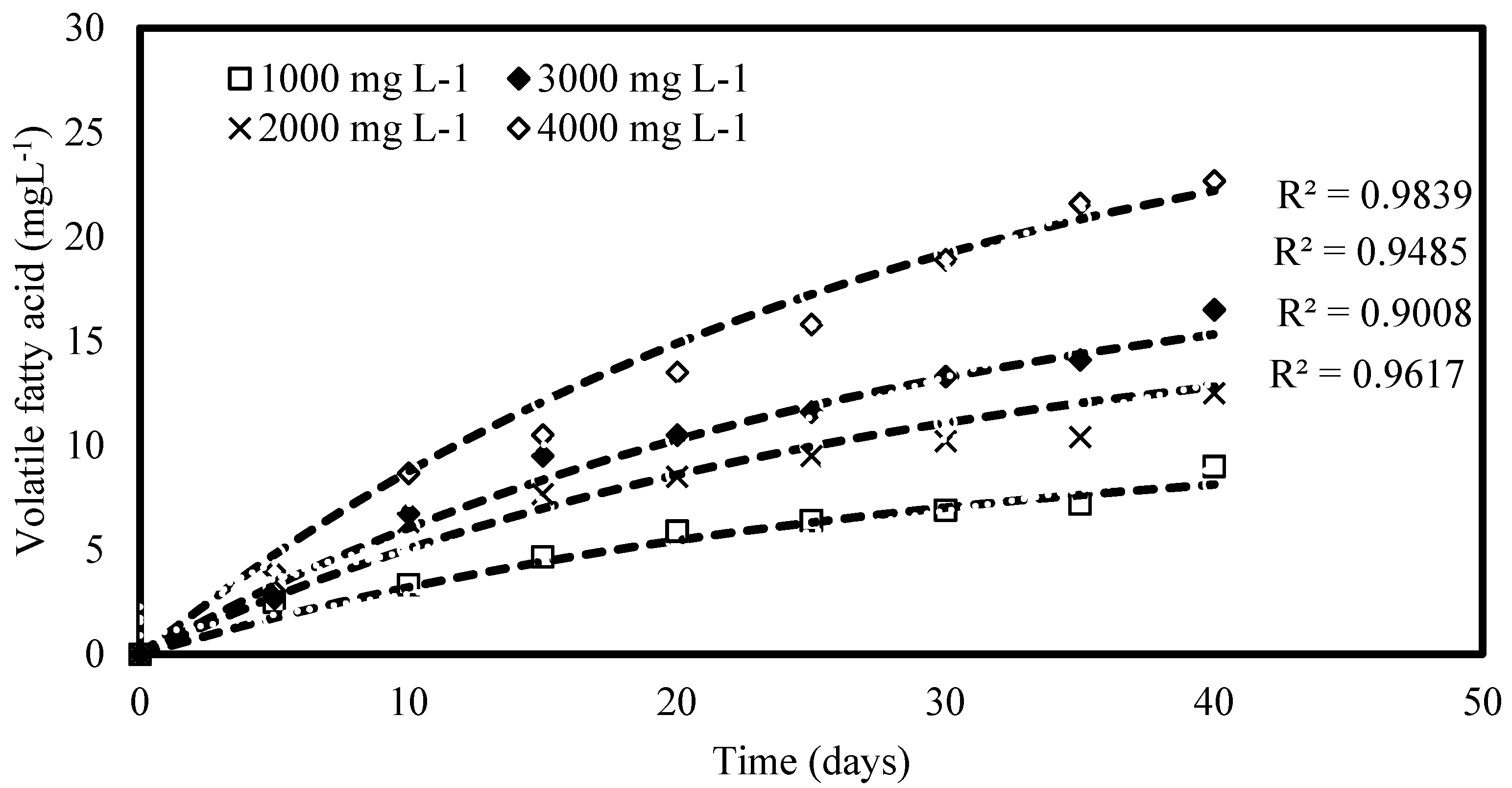
3.1.1. Hydrolysis Kinetics
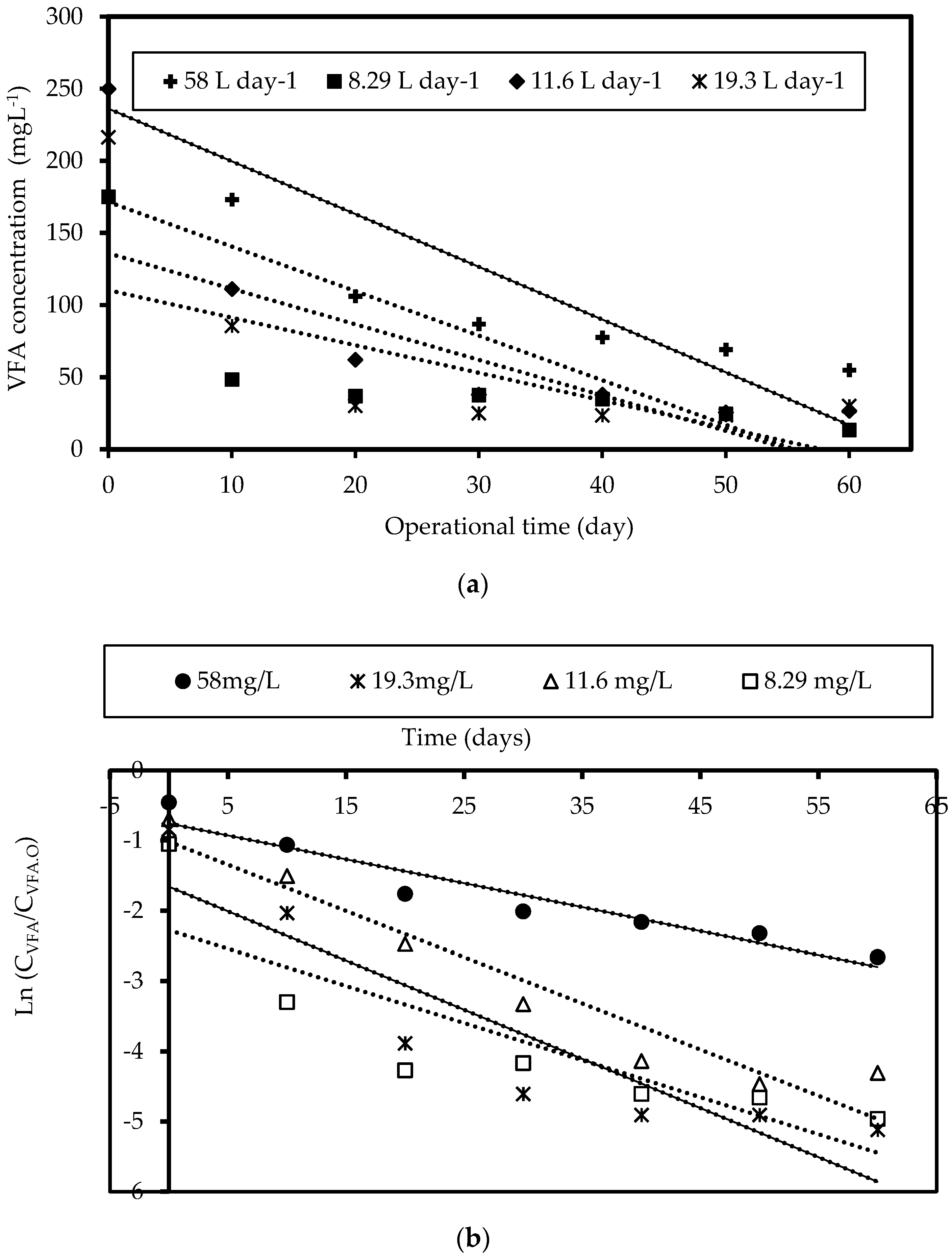
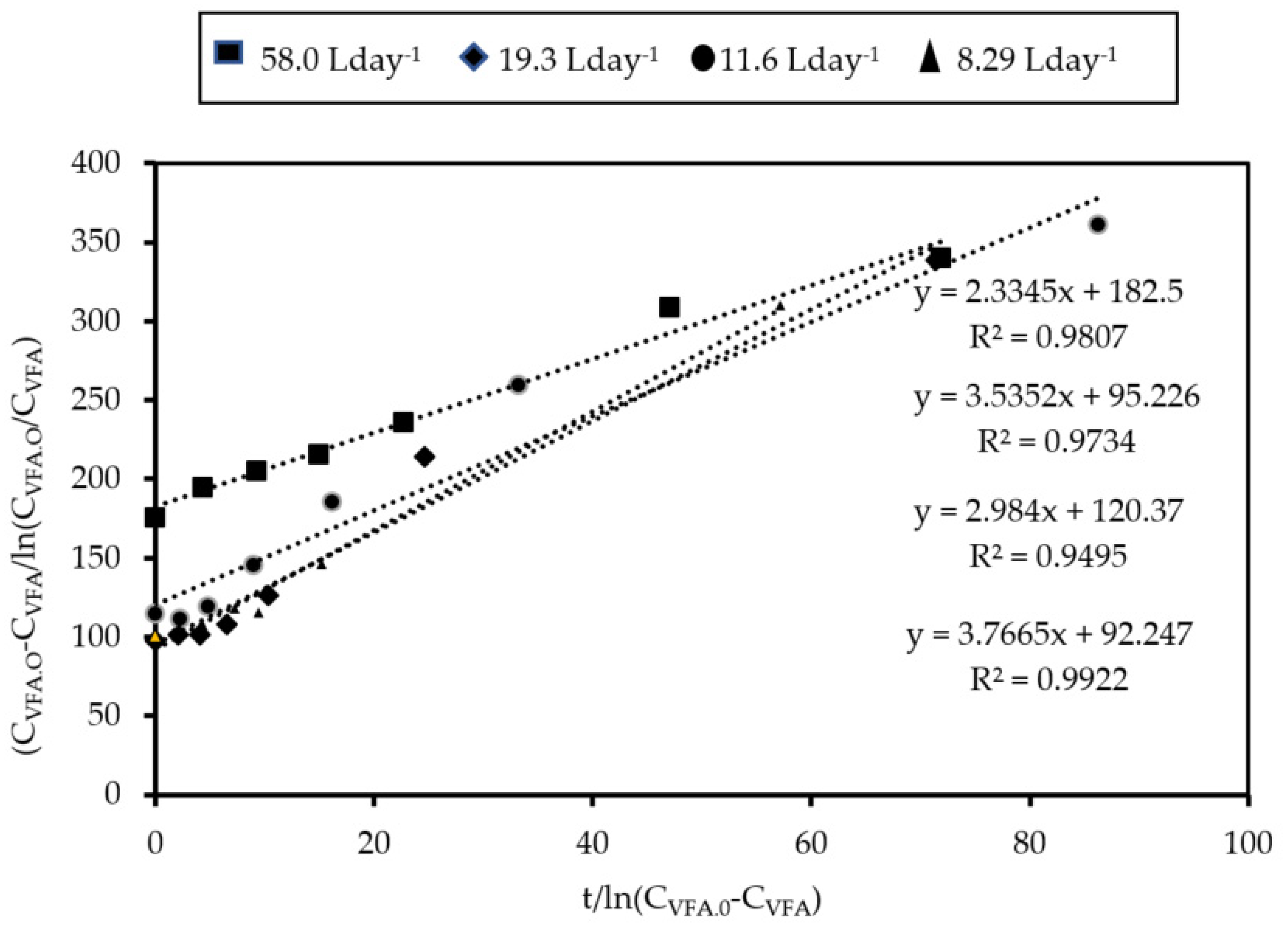
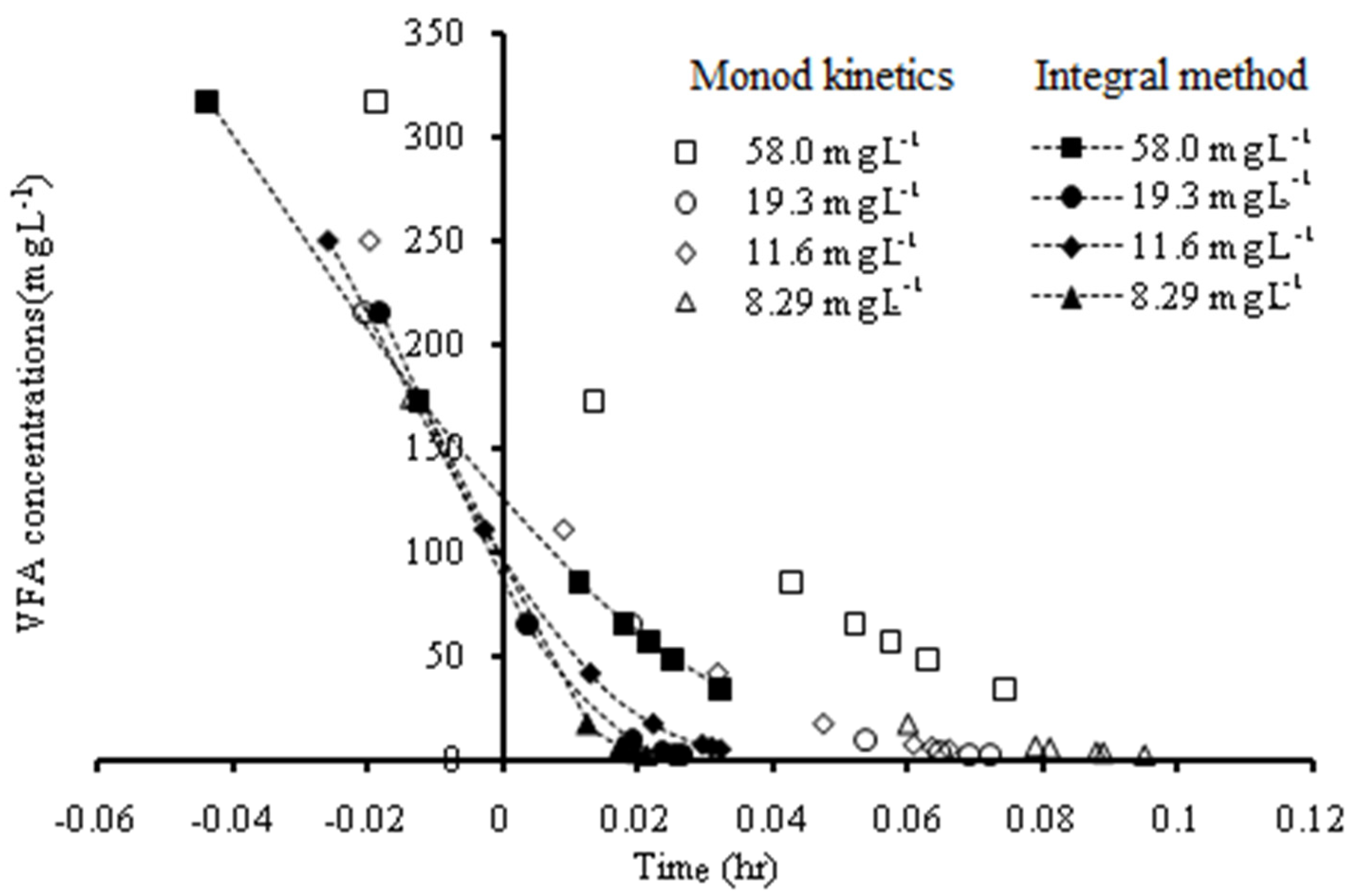
3.1.2. Kinetics of Acetogenesis
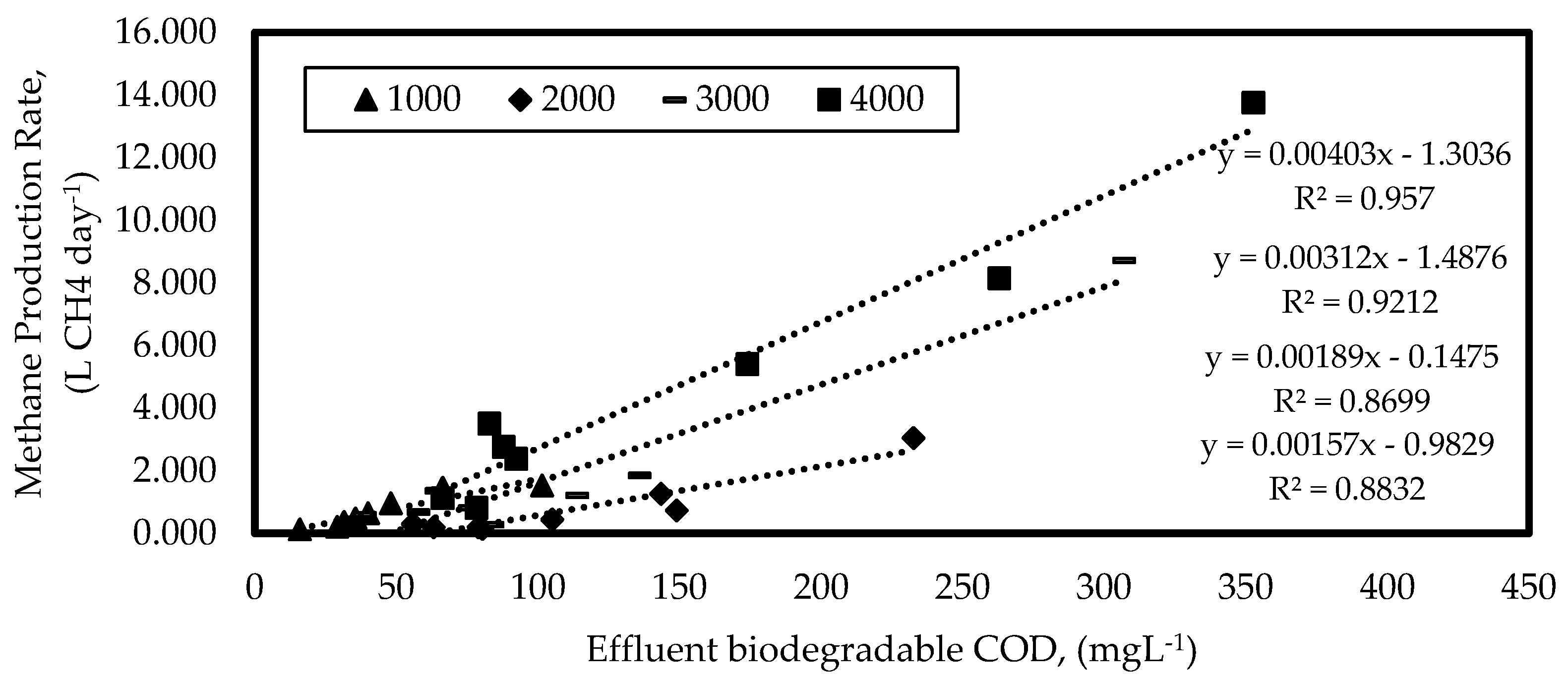
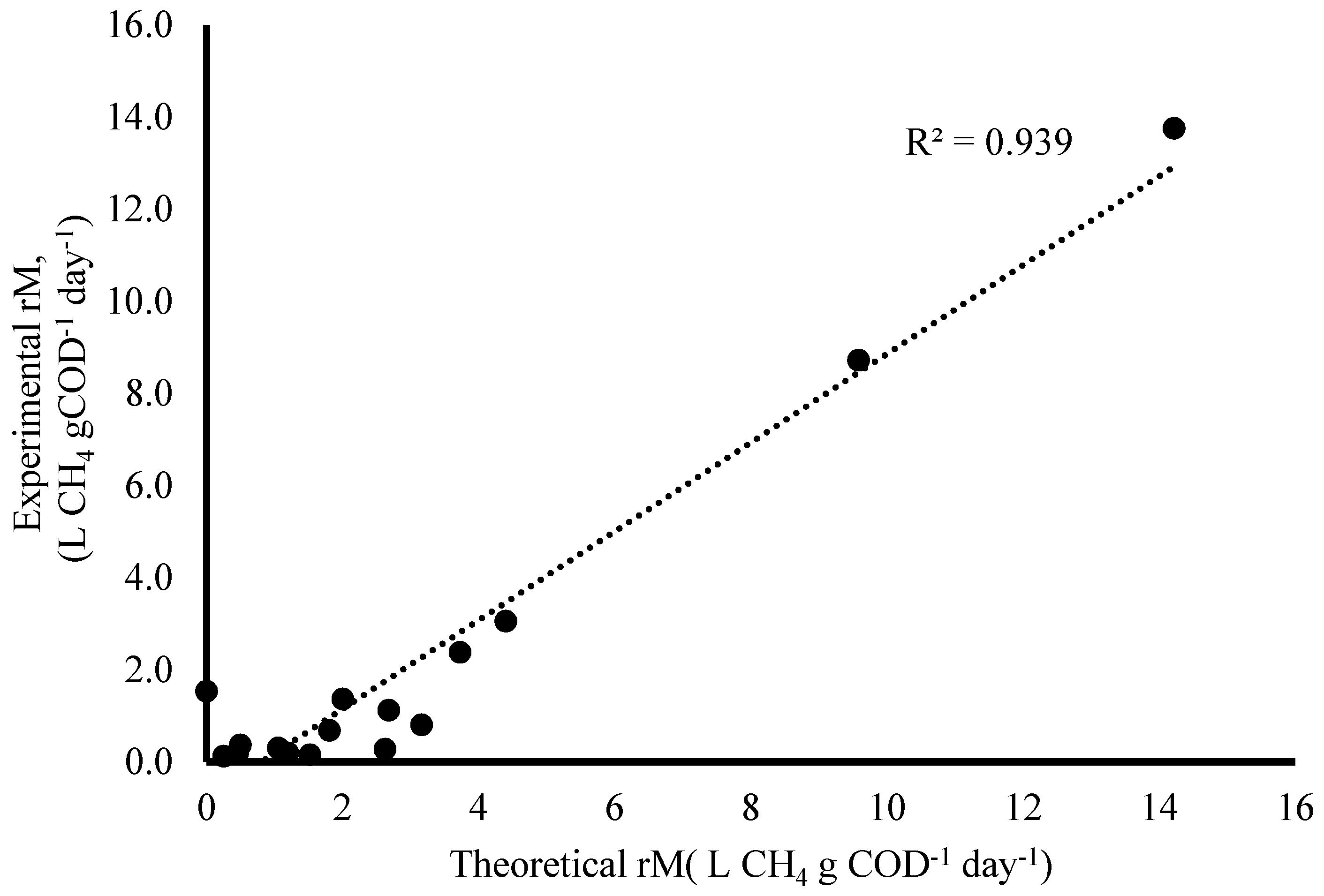
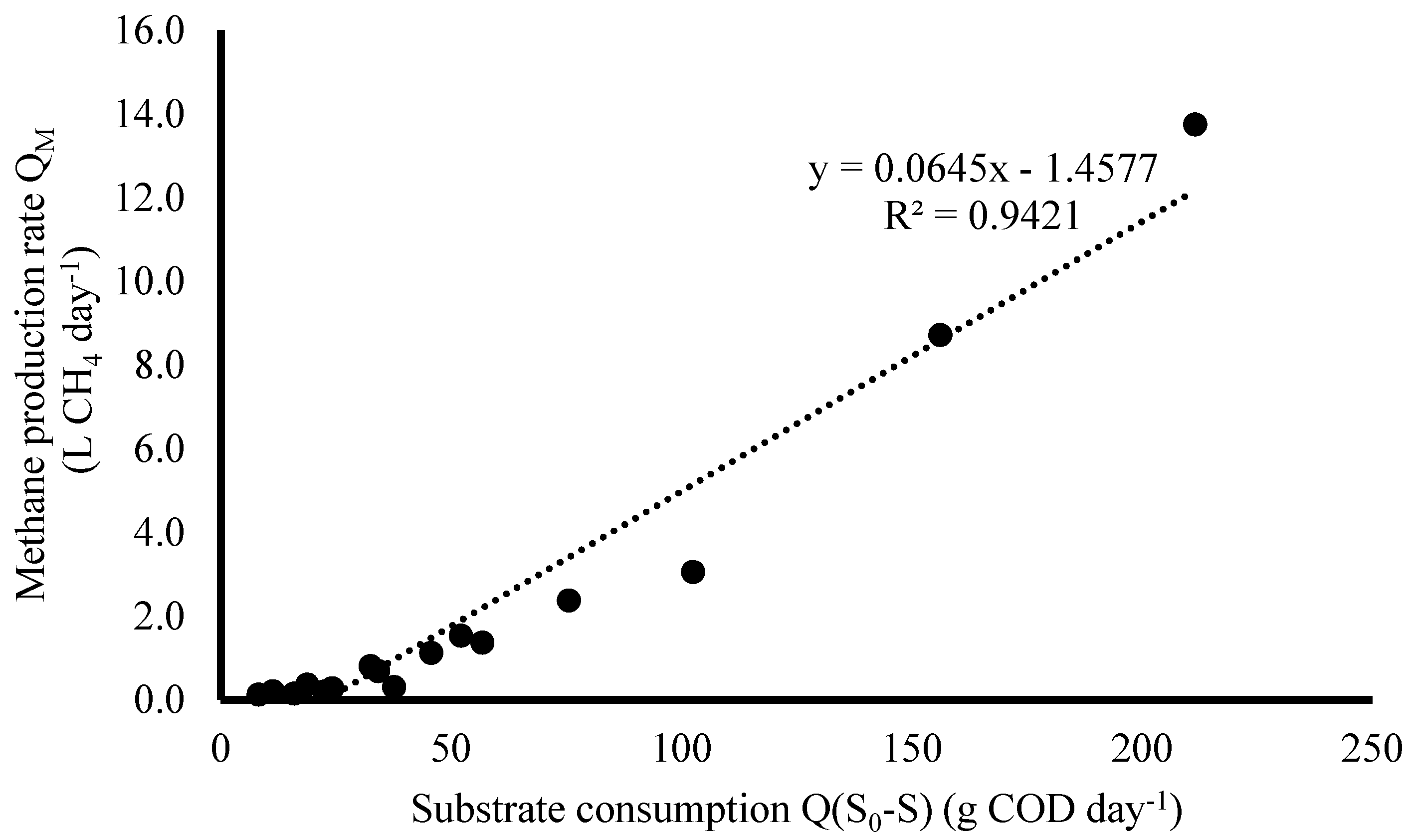
3.1.3. Kinetics of Methanogenesis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, U.S.; Panwar, S.; Jain, R.K.; Tripathi, Y.C. Assessment of Physicochemical Characteristics of Effluents from Paper Mill in the State of Uttar Pradesh, India. Int. J. Eng. Res. Technol. IJERT 2020, 9, 313–318. [Google Scholar]
- Hanum, F.; Yuan, L.C.; Kamahara, H.; Aziz, H.B.; Atsuta, Y.; Yamada, T.; Daimon, H. Treatment of Sewage Sludge Using Anaerobic Digestion in Malaysia: Current State and Challenges. Bioenergy Biofuel 2019, 7, 19. [Google Scholar] [CrossRef]
- Siergrist, H.; Renggli, D.; Gujer, W. Mathematical modelling of anaerobic mesophilic sewage sludge treatment. Water Sci. Technol. 1993, 27, 25–36. [Google Scholar] [CrossRef]
- Anderson, G.K.; Kasapgil, B.; Ince, O. Microbial kinetics of a membrane anaerobic reactor system. Environ. Technol. 1996, 17, 449–464. [Google Scholar] [CrossRef]
- Grady, C.P.L.; Harlow, J.L.J.; Riesing, R.R. Effects of growth rate and influent substrate concentration on effluent quality from chemostats containing bacteria in pure and mixed culture. Biotechnol. Bioeng. 1972, 14, 391–410. [Google Scholar] [CrossRef]
- Fadzil, F.; Norazman, A.F.; Seswoya, R. Pilot scale anaerobic digestion of food waste: Evaluation on the stability, methane production and kinetic analysis. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Hu, W.C.; Thayanithy, K.; Forster, C.F. Kinetic study of anaerobic digestion of sulphate-rich wastewaters from manufacturing food industries. In Proceedings of the 7th International Conference on Environmental Science and Technology, Syros Island, Greece, 3 –6 September 2001. [Google Scholar]
- Vavilin, V.; Lokshina, L.; Rytov, S. Using kinetic isotope effect to evaluate the significance of the sequential and parallel steps: Formation of microbial consortium during reversible anaerobic methane oxidation coupled with sulfate reduction. Water Sci. Technol. 2019, 79, 2056–2067. [Google Scholar] [CrossRef]
- Chen, Y.R.; Hashimoto, A.G. Substrate utilisation kinetic model for biological treatment processes. Biotechnol. Bioeng. 1980, 22, 2081–2095. [Google Scholar] [CrossRef]
- Abu-Reesh, I.M. Kinetics of anaerobic digestion of labaneh whey in a batch reactor. Afr. J. Biotechnol. 2014, 13, 1745–1755. [Google Scholar]
- Vavilin, V.A.; Rytov, S.V.; Lokshina, L.Y.; Rintala, J.A.; Lyberatos, G. Simplified hydrolysis models for the optimal design of two-stage anaerobic digestion. Water Res. 2001, 35, 4247–4251. [Google Scholar] [CrossRef]
- Veeken, A.H.M.; Hamelers, B.V.M. Effect of substrate-seed mixing and the leachate recirculation on solid state digestion of biowaste. Water Sci. Technol. 2000, 41, 255–262. [Google Scholar] [CrossRef]
- Sukphun, P.; Sittijunda, S.; Reungsang, A. Volatile Fatty Acid Production from Organic Waste with the Emphasis on Membrane-Based Recovery. Fermentation 2021, 7, 159. [Google Scholar] [CrossRef]
- Lübken, M.; Gehring, T.; Wichern, M. Microbiological fermentation of lignocellulosic biomass: Current state and prospects of mathematical modeling. Appl. Microbiol. Biotechnol. 2010, 85, 1643–1652. [Google Scholar] [CrossRef]
- Hassan, S.R.; Zwain, H.M.; Zaman, N.Q.; Dahlan, I. Recycled paper mill effluent treatment in a modified anaerobic baffled reactor: Start-up and steady-state performance. Environ. Technol. 2013, 35, 294–299. [Google Scholar] [CrossRef]
- Batstone, D.J.; Keller, J.; Blackall, L.L. The influence of substrate kinetics on the microbial community structure in granular anaerobic biomass. Water Res. 2004, 38, 1390–1404. [Google Scholar] [CrossRef]
- Huilinir, C.; Roa, E.; Vargas, D.; Roeckel, M.; Aspe, E. Kinetics of syntrophic acetogenesis in a saline medium. J. Chem. Technol. Biotechnol. 2008, 83, 1433–1440. [Google Scholar] [CrossRef]
- Clescerl, L.S.; Greenberg, A.E.; Eaton, A.D. (Eds.) Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association; American Water Works Association; Water Environment Federation: Washington, DC, USA, 1998. [Google Scholar]
- Jirka, A.M.; Carter, M.J. Micro semiautomated analysis of surface and waste waters for chemical oxygen demand. Anal. Chem. 1975, 47, 1397–1402. [Google Scholar] [CrossRef]
- Shuizhou, K.; Shi, Z. Applications of two phase anaerobic degradation in industrial wastewater treatment. Int. J. Environ. Pollut. 2005, 23, 65–80. [Google Scholar]
- Vavilin, V.A.; Fernandez, B.; Palatsi, J.; Flotats, X. Hydrolysis kinetics in anaerobic degradation of particulate organic material: An overview. Waste Manag. 2008, 28, 939–951. [Google Scholar] [CrossRef]
- Momoh, O.L.Y.; Anyata, B.U.; Saroj, D.P. Development of simplified anaerobic digestion models (SADM’s) for studying anaerobic biodegradability and kinetics of complex biomass. Biochem. Eng. J. 2013, 79, 84–93. [Google Scholar] [CrossRef][Green Version]
- Skiadas, I.V.; Gavala, H.N.; Lyberatos, G. Modelling of the periodic anaerobic baffled reactor (PABR) based on the retaining factor concept. Water Res. 2000, 34, 3725–3736. [Google Scholar] [CrossRef]
- Romli, M.; Keller, J.; Lee, P.J.; Greenfield, P.F. Model prediction and verification of a two-stage highrate anaerobic wastewater treatment system subjected to shock loads. Process Saf. Environ. Prot. 1995, 73, 151–154. [Google Scholar]
- Vavilin, V.A.; Lockshina, L.Y. Modelling of volatile fatty acids degradation kinetics and evaluation of microorganism activity. Bioresoure Technol. 1996, 57, 69–80. [Google Scholar] [CrossRef]
- Hvala, N.; Strmcnik, S.; Sel, D.; Milanic, S.; Banko, B. Influence of model validation on proper selection of process models—An industrial case study. Comput. Chem. Eng. 2005, 29, 1507–1522. [Google Scholar] [CrossRef]
- Zinatizadeh, A.A.L.; Mohamed, A.R.; Najafpour, G.D.; Hasnain, I.M.; Nasrollahzadeh, H. Kinetic evaluation of palm oil mill effluent digestion in a high rate up-flow anaerobic sludge fixed film bioreactor. Process Biochem. 2006, 41, 1038–1046. [Google Scholar] [CrossRef]
- Belhadj, S.; El Bari, H.; Karouach, F.; Joute, Y.; Chica, A.F.; Santos, M.A.M. Production of methane from mesophilic anaerobic digestion of sewage sludge in morocco. Am. J. Adv. Sci. Res. 2014, 2, 81–91. [Google Scholar]




| Anaerobic Phase | Parameters | Model Used |
|---|---|---|
| Hydrolysis | Data Feeding Concentration: 1000, 2000, 3000 and 4000 mg COD L−1 at HRT of 7 days | First order kinetics model |
| Acetogenesis | Feed flow rates: 58, 19.3,11.6 and 8.29 L day−1 | Monod kinetic and integral method |
| Methanogenesis | Feeding Concentration: 1000, 2000, 3000 and 4000 mg COD L−1 at HRT in a range of 1–7 days | Monod kinetic model |
| Feeding Concentration, (mg COD L−1) | Substrate Conversions Coefficient, α So (mL) | First Order Rate Coefficient, k (Day−1) |
|---|---|---|
| 1000 | 8.682 | 0.1040 |
| 2000 | 51.564 | 0.0440 |
| 3000 | 29.974 | 0.0578 |
| 4000 | 24.210 | 0.0643 |
| Feed Flow Rate (L Day−1) | Monod Model | Integral Method | ||
|---|---|---|---|---|
(g VFA L−1) | (mg VFA mg−1 VSS Day−1) | (g VFA L−1) | (mg VFA mg−1 VSS Day−1) | |
| 58.0 | 0.29 | 13.35 | 0.18 | 12.66 |
| 19.3 | 0.15 | 19.83 | 0.10 | 19.17 |
| 11.6 | 0.15 | 16.75 | 0.12 | 16.18 |
| 8.29 | 0.10 | 10.36 | 0.090 | 10.43 |
| Feed Flow Rate (L Day−1) | TIC | |
|---|---|---|
| Monod | Integral | |
| 58.0 | 0.203 | 0.201 |
| 19.3 | 0.036 | 0.035 |
| 11.6 | 0.059 | 0.057 |
| 8.29 | 0.025 | 0.026 |
| AD Phase | Kinetic Parameters | |
|---|---|---|
| Hydrolysis | α So | 7315 mL |
| k | 0.0117 day−1 | |
| Acetogenesis | Monod Model Integral Method | 0.10–0.29 g VFA L−1 10.36–19.83 mg VFA mg-1 VSS day−1 0.090–0.18 g VFA L−1 10.43–19.17 mg VFA mg−1 VSS day−1 |
| Methanogenesis | YM | 0.0645 L CH4 g COD−1 |
| K | 4.03 L CH4 g−1 COD day−1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassan, S.R.; Hung, Y.-T.; Dahlan, I.; Abdul Aziz, H. Kinetic Study of the Anaerobic Digestion of Recycled Paper Mill Effluent (RPME) by Using a Novel Modified Anaerobic Hybrid Baffled (MAHB) Reactor. Water 2022, 14, 390. https://doi.org/10.3390/w14030390
Hassan SR, Hung Y-T, Dahlan I, Abdul Aziz H. Kinetic Study of the Anaerobic Digestion of Recycled Paper Mill Effluent (RPME) by Using a Novel Modified Anaerobic Hybrid Baffled (MAHB) Reactor. Water. 2022; 14(3):390. https://doi.org/10.3390/w14030390
Chicago/Turabian StyleHassan, Siti Roshayu, Yung-Tse Hung, Irvan Dahlan, and Hamidi Abdul Aziz. 2022. "Kinetic Study of the Anaerobic Digestion of Recycled Paper Mill Effluent (RPME) by Using a Novel Modified Anaerobic Hybrid Baffled (MAHB) Reactor" Water 14, no. 3: 390. https://doi.org/10.3390/w14030390
APA StyleHassan, S. R., Hung, Y.-T., Dahlan, I., & Abdul Aziz, H. (2022). Kinetic Study of the Anaerobic Digestion of Recycled Paper Mill Effluent (RPME) by Using a Novel Modified Anaerobic Hybrid Baffled (MAHB) Reactor. Water, 14(3), 390. https://doi.org/10.3390/w14030390






