Effects of Extracts Containing Metabolites of Different Cyanobacteria from an Ambient Spring (Central Europe) on Zooplankters Daphnia magna and Duckweed Spirodela polyrhiza
Abstract
1. Introduction
2. Materials and Methods
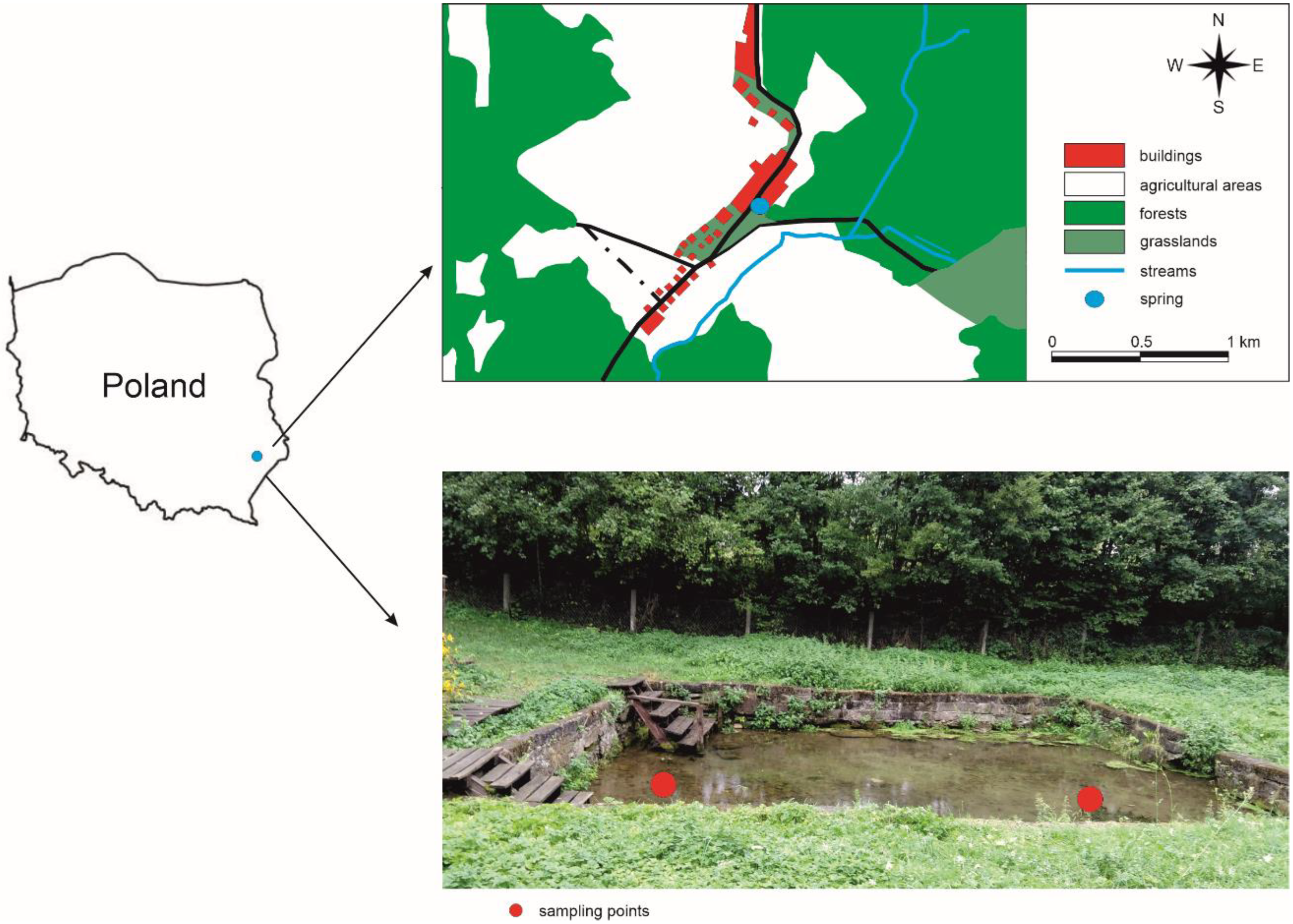
2.1. Study Area, Field Study, and Physical-Chemical Analysis of Spring Water
2.2. Collection and Microscopic Analyses of Microbial Mats
2.3. Extraction of Phycoflora Biomass
2.4. HPLC-PDA Analysis of Microcystins
2.5. HPLC-FLD Analysis of Anatoxin-a
2.6. Toxicity Bioassays
2.6.1. S. polyrhiza Bioassay
2.6.2. Analyses of Growth and Development Parameters
2.6.3. Analysis of Pigments
2.6.4. Acute Toxicity Bioassays on Daphnia Magna
2.7. Data Analysis
3. Results
3.1. Hydrological and Physicochemical Characteristics of Górecko Spring
3.2. Characteristics of Microbial Mats and Extracts
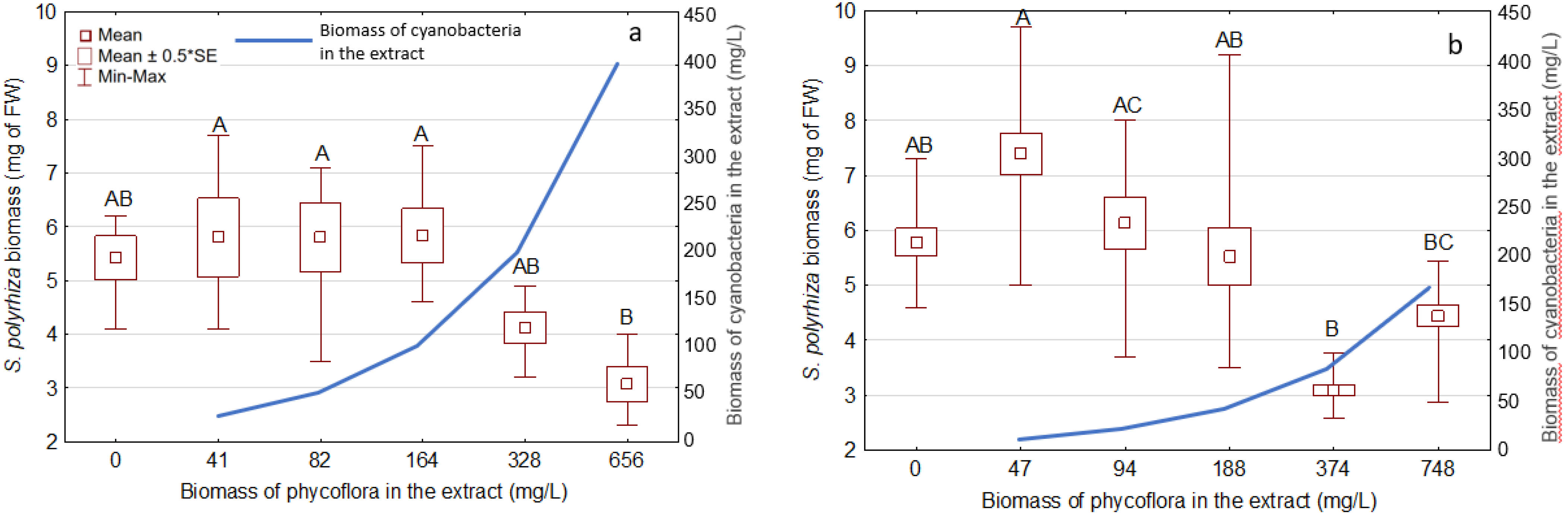
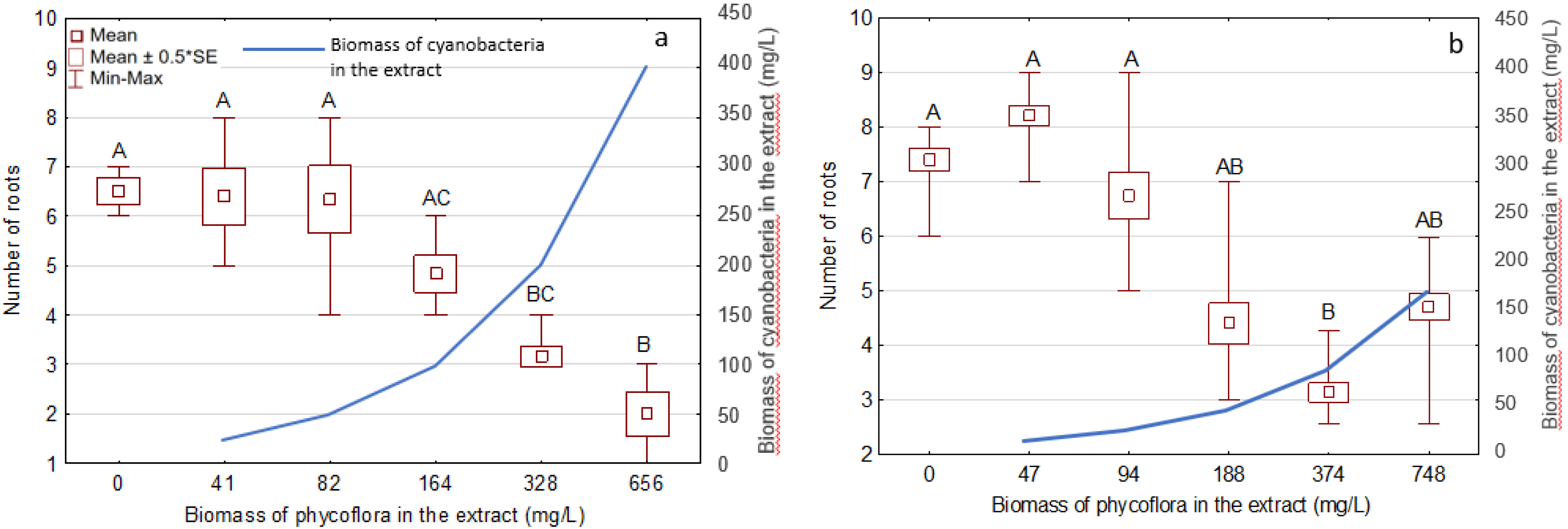
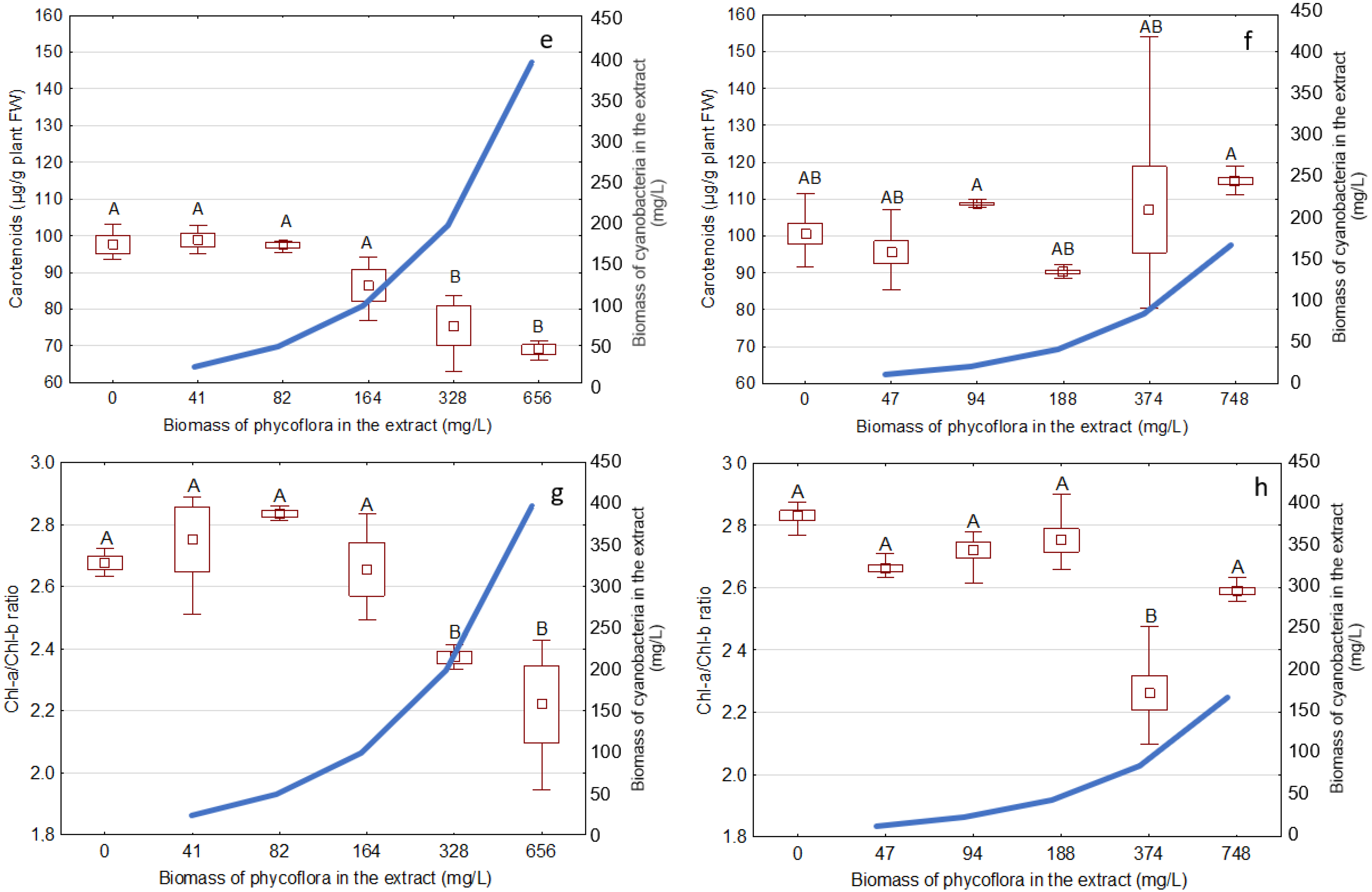
3.3. Effects of the Extracts on the Growth and Development of S. Polyrhiza
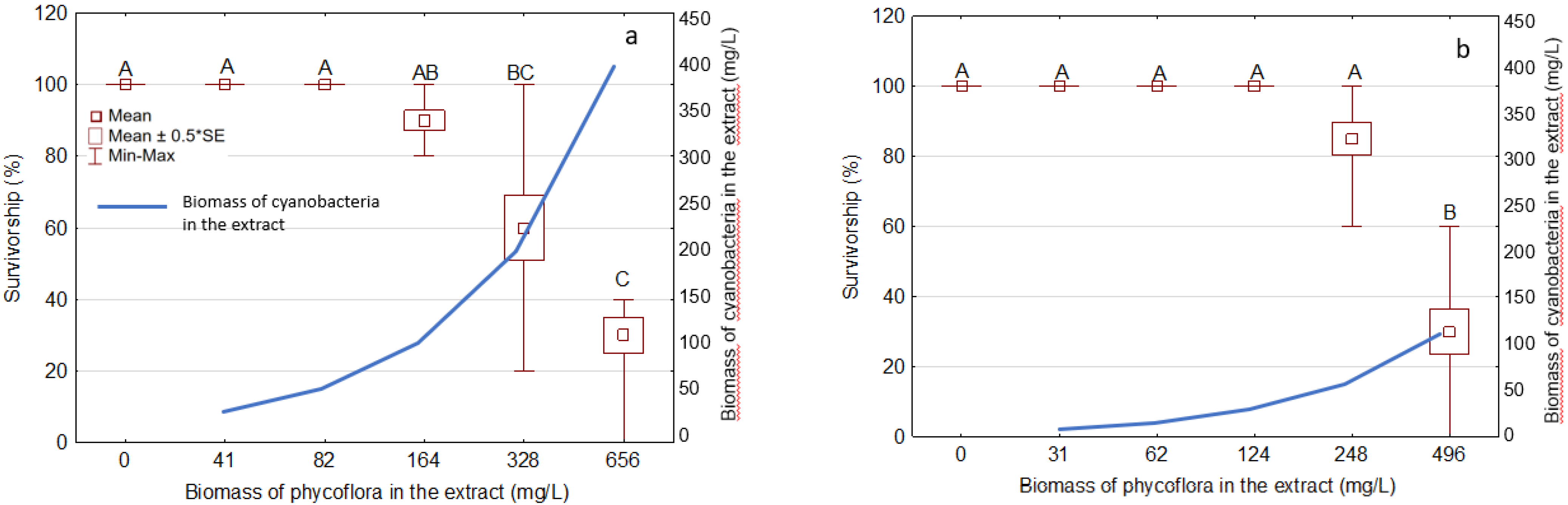
3.4. Effects of the Extracts on D. magna Survivorship
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Whitton, B.A.; Potts, M. The Ecology of Cyanobacteria: Their Diversity in Time and Space; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Hudnell, H.K. Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs; Springer: New York, NY, USA, 2008. [Google Scholar]
- Sivonen, K.; Börner, T. Bioactive compounds produced by cyanobacteria. In The Cyanobacteria; Caister Academic Press: Poole, UK, 2008; pp. 159–197. [Google Scholar]
- Toporowska, M.; Mazur-Marzec, H.; Pawlik-Skowrońska, B. The effects of cyanobacterial bloom extracts on the biomass, Chl-a, MC and other oligopeptides contents in a natural Planktothrix agardhii population. Int. J. Environ. Res. Public Health 2020, 17, 2881. [Google Scholar] [CrossRef]
- Mantzouki, E.; Lürling, M.; Fastner, J.; de Senerpont Domis, L.; Wilk-Woźniak, E.; Koreivienė, J.; Warming, T.P. Temperature effects explain continental scale distribution of cyanobacterial toxins. Toxins 2018, 10, 156. [Google Scholar] [CrossRef] [PubMed]
- Chorus, I.; Welker, M. Toxic Cyanobacteria in Water: A Guide to their Public Health Consequences, Monitoring and Management; Taylor & Francis: Abingdon-on-Thames, UK, 2021; p. 858. [Google Scholar]
- Glazier, D.S. Springs. In Encyclopedia of Inland Waters; Likens, G.E., Ed.; Academic Press: San Diego, CA, USA, 2009; Volume 1, pp. 734–755. [Google Scholar]
- Cantonati, M.; Komárek, J.; Montejano, G. Cyanobacteria in ambient springs. Biodivers. Conserv. 2015, 24, 865–888. [Google Scholar] [CrossRef]
- Nowicka-Krawczyk, P.B.; Żelazna-Wieczorek, J. Cyanobacteria microflora in a limestone spring (Troniny spring, Central Poland). ASBP 2013, 82, 221–224. [Google Scholar] [CrossRef][Green Version]
- Nowicka-Krawczyk, P.; Żelazna-Wieczorek, J. Dynamics in cyanobacterial communities from a relatively stable environment in an urbanised area (ambient springs in Central Poland). Stoten 2017, 579, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Uher, B.; Skácelová, O.; Kováčik, L. Sinice několika studánek v okolí Brna (Cyanobacteria of several wells in Brno surroundings). Czech Phycol. 2001, 1, 21–30. (In Czech) [Google Scholar]
- Cantonati, M. Cyanoprokaryotes and algae other than diatoms in springs and streams of the Dolomiti Bellunesi National Park (Northern Italy). Algol. Stud. 2008, 126, 113–136. [Google Scholar] [CrossRef]
- Ferencz, B.; Dawidek, J.; Bronowicka-Mielniczuk, U. Alteration of yield and springs number as an indicator of climate changes. Case study of Eastern Poland. Ecol. Indic. 2022, 138, 108798. [Google Scholar] [CrossRef]
- McGregor, G.B.; Sendall, B.C. Iningainema pulvinus gen nov., sp nov. (Cyanobacteria, Scytonemataceae) a new nodularin producer from Edgbaston Reserve, north-eastern Australia. Harmful Algae 2017, 62, 10–19. [Google Scholar] [CrossRef]
- Cantoral Uriza, E.A.; Asencio, A.D.; Aboal, M. Are we underestimating benthic cyanotoxins? Extensive sampling results from Spain. Toxins 2017, 9, 385. [Google Scholar] [CrossRef]
- Chen, L.; Giesy, J.P.; Adamovsky, O.; Svirčev, Z.; Meriluoto, J.; Codd, G.A.; Xie, P. Challenges of using blooms of Microcystis spp. in animal feeds: A comprehensive review of nutritional, toxicological and microbial health evaluation. Stoten 2021, 764, 142319. [Google Scholar] [CrossRef]
- Pawlik-Skowrońska, B.; Toporowska, M.; Mazur-Marzec, H. Effects of secondary metabolites produced by different cyanobacterial populations on the freshwater zooplankters Brachionus calyciflorus and Daphnia pulex. ESPR 2019, 26, 11793–11804. [Google Scholar] [CrossRef]
- Aboal, M.; Angeles Puig, M.; Mateo, P.; Perona, E. Implications of cyanophyte toxicity on biological monitoring of calcareous streams in north-east Spain. J. Appl. Phycol. 2002, 14, 49–56. [Google Scholar] [CrossRef]
- Aboal, M.; Puig, M.A.; Asencio, A.D. Production of microcystins in calcareous Mediterranean streams: The Alharabe River, Segura River basin in south-east Spain. J. Appl. Phycol. 2005, 17, 231–243. [Google Scholar] [CrossRef]
- Fetscher, A.E.; Howard, M.D.; Stancheva, R.; Kudela, R.M.; Stein, E.D.; Sutula, M.A.; Sheath, R.G. Wadeable streams as widespread sources of benthic cyanotoxins in California, USA. Harmful Algae 2015, 49, 105–116. [Google Scholar] [CrossRef]
- Gärtner, G.; Stoyneva-Gärtner, M.; Uzunov, B. Algal toxic compounds and their aeroterrestrial, airborne and other extremophilic producers with attention to soil and plant contamination: A review. Toxins 2021, 13, 322. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, Z.A. Toxic cyanobacteria and cyanotoxins in public hot springs in Saudi Arabia. Toxicon 2008, 51, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Flores, C.; Caixach, J.; Barca, S.; Vieira-Lanero, R.; Cobo, F. Occurrence of Cyanotoxins in Mineral Water Sources and Hot Springs from NW Iberian Peninsula. In Biology and Life Sciences Forum 14 (1); Multidisciplinary Digital Publishing Institute: Basel, Switzerland, 2022; p. 26. [Google Scholar]
- Plaas, H.E.; Paerl, H.W. Toxic cyanobacteria: A growing threat to water and air quality. Environ. Sci. Technol. 2020, 55, 44–64. [Google Scholar] [CrossRef]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Jessup, D.A. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from land to sea otters. PLoS ONE 2010, 5, e12576. [Google Scholar] [CrossRef]
- Gaget, V.; Almuhtaram, H.; Kibuye, F.; Hobson, P.; Zamyadi, A.; Wert, E.; Brookes, J.D. Benthic cyanobacteria: A utility centred field study. Harmful Algae 2022, 113, 102185. [Google Scholar] [CrossRef]
- ISO 20079:2005; Water Quality—Determination of the Toxic Effect of Water Constituents and Wastewater on Duckweed (Lemna Minor)—Duckweed Growth Inhibition Test. ISO: Geneva, Switzerland, 2005.
- Baudo, R.; Foudoulakis, M.; Arapis, G.; Perdaen, K.; Lanneau, W.; Paxinou, A.C.; Kouvdou, S.; Persoone, G. History andsensitivity comparison of the Spirodela polyrhiza microbiotest and Lemna toxicity tests. KMAE 2015, 416–432, 23. [Google Scholar] [CrossRef]
- Pietrini, F.; Iannilli, V.; Passatore, L.; Carloni, S.; Sciacca, G.; Cerasa, M.; Zacchini, M. Ecotoxicological and genotoxic effects of dimethyl phthalate (DMP) on Lemna minor L. and Spirodela polyrhiza (L.) Schleid. Plants under a short-term laboratory assay. STOTEN 2022, 806, 150972. [Google Scholar] [CrossRef]
- Connors, K.A.; Brill, J.L.; Norberg-King, T.; Barron, M.G.; Carr, G.; Belanger, S.E. Daphnia magna and Ceriodaphnia dubia have similar sensitivity in standard acute and chronic toxicity tests. Environ. Toxicol. Chem. 2022, 41, 134–147. [Google Scholar] [CrossRef]
- Reyes, V.P.; Ventura, M.A.; Amarillo, P.B. Ecotoxicological assessment of water and sediment in areas of Taal Lake with heavy aquaculture practices using Allium cepa and Daphnia magna assay. Philipp J. Sci. 2022, 151, 969–974. [Google Scholar] [CrossRef]
- Tanaka, A.; Tanaka, R. Chlorophyll metabolism. Curr. Opin. Plant Biol. 2006, 9, 248–255. [Google Scholar] [CrossRef]
- Komárek, J.; Anagnostidis, K. Oscillatoriales. T 2. In Süßwasserflora von Mitteleuropa; Ettl, H., Gärtner, G., Gerloff, J., Heyning, H., Mollenhauer, D., Eds.; Elsevier GmbH, Spektrum Akademischer Verlag: München, Germany, 2005. [Google Scholar]
- Cox, E.J. Identification of Freshwater Diatoms from Live Material; Chapman and Hall: London, UK, 1996. [Google Scholar]
- Starmach, K. Freshwater Phytoplankton. Study Methods, Key to Freshwater Species of Central Europe; PWN: Warszawa-Kraków, Poland, 1989; p. 496. (In Polish) [Google Scholar]
- Hillebrand, H.; Dürselen, C.D.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- James, K.J.; Furey, A.; Sherlock, I.R.; Stack, M.A.; Twohing, M. Liquid chromatography with fluorometric detection involves sensitive determination of anatoxin-a and homoanatoxin-a and their degradation products. J. Chromatogr. 1998, 798, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Janiec, B. Ion Transformations and Translocations in Natural Waters of the West Roztocze; UMCS: Lublin, Poland, 1997. (In Polish) [Google Scholar]
- Chmiel, S. Physico-chemical properties of spring and river waters. In Springs of Lublin Upland and Roztocze; Wydawnictwo UMCS: Lublin, Poland, 2001; pp. 219–252. (In Polish) [Google Scholar]
- Chmiel, S.; Maciejewska, E.; Michalczyk, Z. Hydrochemical characteristics of a spring snowmelt flood in the Upper Wieprz River basin (Roztocze region) in year 2006. J. Water Land Dev. 2009, 13, 57–67. [Google Scholar] [CrossRef]
- Hájek, M.; Roleček, J.; Cottenie, K.; Kintrová, K.; Horsák, M.; Poulíčková, A.; Dítě, D. Environmental and spatial controls of biotic assemblages in a discrete semi-terrestrial habitat: Comparison of organisms with different dispersal abilities sampled in the same plots. J. Biogeogr. 2011, 38, 1683–1693. [Google Scholar] [CrossRef]
- Poulícková, A.; Hašler, P.; Kitner, M. Cyanobacteria and algae. In Ecology and Palaeoecology of Spring Fens of the West Carpathians; Poulícková, A., Hájek, M., Rybnícek, K., Eds.; Palacký University Press: Olomouc, Czech Republic, 2005; pp. 105–130. [Google Scholar]
- Mazmouz, R.; Chapuis-Hugon, F.; Mann, S.; Pichon, V.; Méjean, A.; Ploux, O. Biosynthesis of cylindrospermopsin and 7-epicylindrospermopsin in Oscillatoria sp. strain PCC 6506: Identification of the cyr gene cluster and toxin analysis. Appl. Environ. Microbiol. 2010, 76, 4943–4949. [Google Scholar] [CrossRef]
- Méjean, A.; Mann, S.; Maldiney, T.; Vassiliadis, G.; Lequin, O.; Ploux, O. Evidence that biosynthesis of the neurotoxic alkaloids anatoxin-a and homoanatoxin-a in the cyanobacterium Oscillatoria PCC 6506 occurs on a modular polyketide synthase initiated by l-proline. J. Am. Chem. Soc. 2009, 131, 7512–7513. [Google Scholar] [CrossRef] [PubMed]
- Gugger, M.F.; Lenoir, S.; Berger, C.; Ledreux, A.; Druart, J.; Humbert, J.F.; Bernard, C. First report in a river in France of the benthic cyanobacterium Phormidium favosum producing anatoxin-a associated with dog neurotoxicosis. Toxicon 2005, 45, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.A.; Smith, F.M.; Heath, M.W.; Palfroy, T.; Gaw, S.; Young, R.G.; Ryan, K.G. Withinmat variability in anatoxin-a and homoanatoxin-a production among benthic Phormidium (cyanobacteria) strains. Toxins 2012, 4, 900–912. [Google Scholar] [CrossRef] [PubMed]
- Izaguirre, G.; Jungblut, A.D.; Neilan, B.A. Benthic cyanobacteria (Oscillatoriaceae) that produce microcystin-LR, isolated from four reservoirs in southern California. Water Res. 2007, 41, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Borges, H.L.F.; Branco, L.H.Z.; Martins, M.D.; Lima, C.S.; Barbosa, P.T.; Lira, G.A.S.T.; Molica, R.J.R. Cyanotoxin production and phylogeny of benthic cyanobacterial strains isolated from the northeast of Brazil. Harmful Algae 2015, 43, 46–57. [Google Scholar] [CrossRef]
- Anderson, B.; Voorhees, J.; Phillips, B.; Fadness, R.; Stancheva, R.; Nichols, J.; Wood, S.A. Extracts from benthic anatoxin-producing Phormidium are toxic to 3 macroinvertebrate taxa at environmentally relevant concentrations. Environ. Toxicol. Chem. 2018, 37, 2851–2859. [Google Scholar] [CrossRef]
- Abubakr, A. Sanative role of macrophytes in aquatic ecosystems. Nat. Environ. Pollut. 2010, 9, 657–662. [Google Scholar]
- Pawlik-Skowrońska, B.; Toporowska, M.; Mazur-Marzec, H. Toxic oligopeptides in the cyanobacterium Planktothrix agardhii-dominated blooms and their effects on duckweed (Lemnaceae) development. KMAE 2018, 419, 41–50. [Google Scholar] [CrossRef]
- Aron, D. Copper enzymes isolated chloroplasts, polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Rajalakshmi, K.; Banu, N. Extraction and estimation of chlorophyll from medicinal plants. IJSR 2015, 4, 209–212. [Google Scholar]
- Vogelmann, T.C.; Martin, G. The functional significance of palisade tissue: Penetration of directional versus diffuse light. Plant Cell Environ. 1993, 16, 65–72. [Google Scholar] [CrossRef]
- Maina, J.N.; Wang, Q. Seasonal response of chlorophyll a/b ratio to stress in a typical desert species: Haloxylon ammodendron. Arid. Land Res. Manag. 2015, 29, 321–334. [Google Scholar] [CrossRef]
- Souza, L.R.R.; Bernardes, L.E.; Barbetta, M.F.S.; da Veiga, M.A.M.S. Iron oxide nanoparticle phytotoxicity to the aquatic plant Lemna minor: Effect on reactive oxygen species (ROS) production and chlorophyll a/chlorophyll b ratio. Environ. Sci. Pollut. Res. 2019, 26, 24121–24131. [Google Scholar] [CrossRef] [PubMed]
- Bafford, R.A.; Seagull, R.W.; Chung, S.Y.; Millie, D.F. Intracellular localization of the taste/odor metabolite 2-methylisoborneol in Oscillatoria limosa (Cyanophyta). J. Phycol. 1993, 29, 91–95. [Google Scholar] [CrossRef]
- Gilbert, J.J. Succeptibility of planktonic rotifers to a toxic strain of Anabaena floes-aquae. Limnol. Oceanogr. 1994, 39, 1286–1297. [Google Scholar] [CrossRef]
- Hulot, F.D.; Carmignac, D.; Legendre, S.; Yepremian, C. Effects of microcystin-producing and microcystin-free strains of Planktothrix agardhii on long-term population dynamics of Daphnia magna. Ann. Limnol. Int. J. Limnol. 2012, 48, 337–347. [Google Scholar] [CrossRef]
- Smutná, M.; Babica, P.; Jarque, S.; Hilscherová, K.; Marsálek, B.; Haeba, M.; Bláha, L. Acute, chronic and reproductive toxicity of complex cyanobacterial blooms in Daphnia magna and the role of microcystins. Toxicon 2014, 79, 11–18. [Google Scholar] [CrossRef]
- Barrios, C.A.Z.; Nandini, S.; Sarma, S.S.S. Effect of crude extract of Dolichospermum planctonicum on the demography of Plationus platulus (Rotifera) and Ceriodaphnia cornuta (Cladocera). Ecotoxicology 2015, 24, 85–93. [Google Scholar] [CrossRef]
- Bittner, M.; Štern, A.; Smutná, M.; Hilscherová, K.; Žegura, B. Cytotoxic and genotoxic effects of cyanobacterial and algal extracts—Microcystin and retinoic acid content. Toxins 2021, 13, 107. [Google Scholar] [CrossRef]
- Ferrão-Filho, A.D.S.; Kozlowsky-Suzuki, B. Cyanotoxins: Bioaccumulation and effects on aquatic animals. Mar. Drugs 2011, 9, 2729–2772. [Google Scholar] [CrossRef]
- Shin, H.J.; Murakami, M.; Matsuda, H.; Yamaguchi, K. Microviridins D-F, serine protease inhibitors from the cyanobacterium Oscillatoria agardhii (NIES-204). Tetrahedron 1996, 52, 8159–8168. [Google Scholar] [CrossRef]
- Rohrlack, T.; Utkilen, H. Effects of nutrient and light availability on production of bioactive anabaenopeptins and microviridin by the cyanobacterium Planktothrix agardhii. Hydrobiologia 2007, 583, 231–240. [Google Scholar] [CrossRef]
- Jones, M.R.; Pinto, E.; Torres, M.A.; Dörr, F.; Mazur-Marzec, H.; Szubert, K.; Janssen, E.M.L. CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria. Water Res. 2021, 196, 117017. [Google Scholar] [CrossRef] [PubMed]
- Welker, M.; Döhren, H. Cyanobacterial peptides—Nature’s own combinatorial biosynthesis. FEMS Microbiol. Rev. 2006, 30, 530–563. [Google Scholar] [CrossRef]
- Janssen, E.M.-L. Cyanobacterial peptides beyond microcystins—A review on co-occurrence, toxicity, and challenges for risk assessment. Water Res. 2019, 151, 488–499. [Google Scholar] [CrossRef]
- Mejean, A.; Mazmouz, R.; Mann, S.; Calteau, A.; Medigue, C.; Ploux, O. The genome sequence of the cyanobacterium Oscillatoria sp. PCC 6506 reveals several gene clusters responsible for the biosynthesis of toxins and secondary metabolites. J. Bacteriol. 2010, 192, 5264–5265. [Google Scholar] [CrossRef]
- Kaya, K.; Sano, T. Cyanobacterial Retinoids. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 173–178. [Google Scholar]
- Peckol, P.; Putnam, A.B. Differential toxic effects of Ulva lactuca (Chlorophyta) on the herbivorous gastropods, Littorina littorea and L. obtusata (Mollusca). J. Phycol. 2017, 53, 361–367. [Google Scholar] [CrossRef]
- Dhar, B.C.; Cimarelli, L.; Singh, K.S.; Brandi, L.; Brandi, A.; Puccinelli, C.; Marcheggiani, S.; Spurio, R. Molecular detection of a potentially toxic diatom species. Int. J. Environ. Res. Public Health 2015, 12, 4921–4941. [Google Scholar] [CrossRef]

| Parameters | Phormidium Mat/Extract | Oscillatoria Mat/Extract |
|---|---|---|
| The total phycoflora biomass (mg FW/L) | 4634.0 ± 602.4 | 2625.3 ± 183.8 |
| The cyanobacterial biomass (mg FW/L) | 2799.5 ± 370.7 | 583.5 ± 70.0 |
| Cyanobacterial taxa and their contribution to the total cyanobacterial biomass (%) | Phormidium breve (95.8%) Leptolyngbya sp. 1 (3.3) Oscillatoria limosa (0.8) Leptolyngbya sp. 2 (0.1) | Oscillatoria limosa (95.1%) Phormidium breve (4.4) Leptolyngbya sp. (0.4) Pseudanabaena minima (0.1) |
| Biomass of eukaryotic algae (mg FW/L): | ||
| Bacillariophyceae | 1834.5 ± 302.7 | 1033.6 ± 98.2 |
| Chlorophyceae | n.f. | 0.7 ± 0.03 |
| Filamentous Ulvophyceae | n.f. | 1007.5 ± 6.5 |
| Microcystins (µg/L) | n.d. | n.d. |
| Anatoxin-a (µg/L) | n.d. | n.d. |
| Extract | Phycoflora Biomass (mg/L) | Cyanobacterial Biomass (mg/L) | Growth Inhibition (%) | |
|---|---|---|---|---|
| S. polyrhiza Biomass | Number of Roots | |||
| Phormidium | 328; 656 | 200; 400 | 24 ± 4.5; 43 ± 4.9 | 51 ± 2.6; 69 ± 5.6 |
| Oscillatoria | 374; 748 | 83; 166 | 47 ± 3.2; 23 ± 6.7 | 58 ± 4.9; 37 ± 6.5 |
| Extract | 48 h LC50 Expressed as mg/L | |
|---|---|---|
| Total Phycoflora Biomass | Cyanobacterial Biomass | |
| Phormidium | 381 ± 82 | 181 ± 61 |
| Oscillatoria | 369 ± 18 | 81 ± 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toporowska, M.; Ferencz, B.; Dawidek, J. Effects of Extracts Containing Metabolites of Different Cyanobacteria from an Ambient Spring (Central Europe) on Zooplankters Daphnia magna and Duckweed Spirodela polyrhiza. Water 2022, 14, 4107. https://doi.org/10.3390/w14244107
Toporowska M, Ferencz B, Dawidek J. Effects of Extracts Containing Metabolites of Different Cyanobacteria from an Ambient Spring (Central Europe) on Zooplankters Daphnia magna and Duckweed Spirodela polyrhiza. Water. 2022; 14(24):4107. https://doi.org/10.3390/w14244107
Chicago/Turabian StyleToporowska, Magdalena, Beata Ferencz, and Jarosław Dawidek. 2022. "Effects of Extracts Containing Metabolites of Different Cyanobacteria from an Ambient Spring (Central Europe) on Zooplankters Daphnia magna and Duckweed Spirodela polyrhiza" Water 14, no. 24: 4107. https://doi.org/10.3390/w14244107
APA StyleToporowska, M., Ferencz, B., & Dawidek, J. (2022). Effects of Extracts Containing Metabolites of Different Cyanobacteria from an Ambient Spring (Central Europe) on Zooplankters Daphnia magna and Duckweed Spirodela polyrhiza. Water, 14(24), 4107. https://doi.org/10.3390/w14244107








