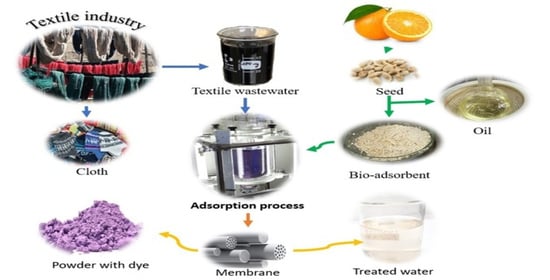
Efficient Dye Removal from Real Textile Wastewater Using Orange Seed Powder as Suitable Bio-Adsorbent and Membrane Technology
Abstract
1. Introduction
2. Material and Methods
2.1. Textile Wastewater
2.2. Bio-Adsorbent Obtention from Orange Seeds
2.3. Evaluation of More Influential Variables through the Screening Step
2.4. Dye Removal Optimization Using a Response Surface Methodology
2.5. Dye Adsorption under Optimized Conditions Using 10 L of TW
2.6. Bio-Adsorbent Removal by Membranes
3. Results and Discussion
3.1. Orange Seeds and Textile Wastewater Composition
3.2. Variable Identification Using Fractionated Experimental Design
3.3. Optimization of Dye Removal Using a Response Surface Methodology
3.4. Textile Wastewater Treatment in a 10 L Reactor
3.5. Bio-Adsorbent Removal by Ultrafiltration and pH Effect
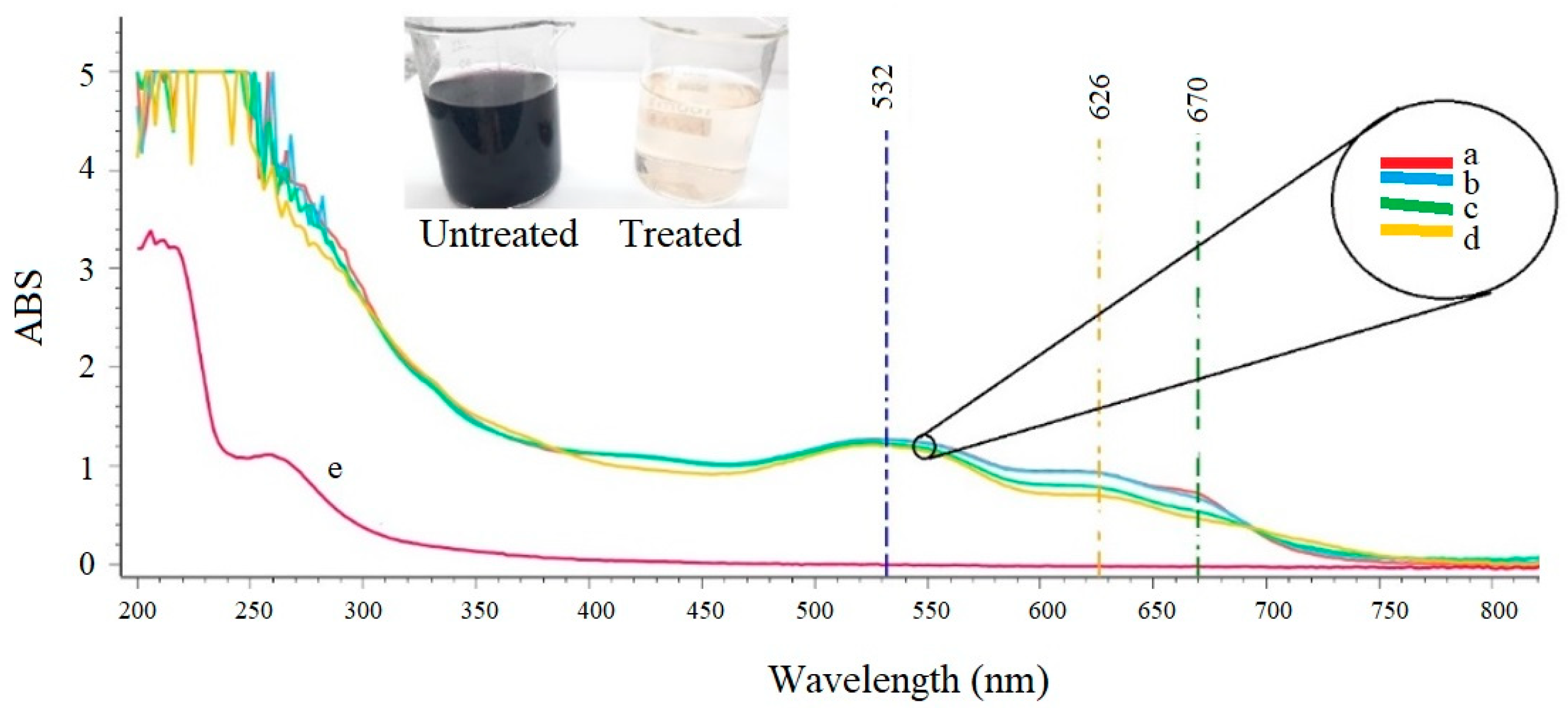
3.6. Characteristic of the Wastewater after the Sequential Treatment
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yaseen, D.A.; Scholz, M. Textile dye wastewater characteristics and constituents of synthetic effluents: A critical review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef]
- Oke, N.; Mohan, S. Development of nanoporous textile sludge based adsorbent for the dye removal from industrial textile effluent. J. Hazard. Mater. 2022, 422, 126864. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghouti, M.A.; Li, J.; Salamh, Y.; Al-Laqtah, N.; Walker, G.; Ahmad, M.N.M. Adsorption mechanisms of removing heavy metals and dyes from aqueous solution using date pits solid adsorbent. J. Hazard. Mater. 2010, 176, 510–520. [Google Scholar] [CrossRef]
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile finishing dyes and their impact on aquatic environs. Heliyon 2019, 5, e02711. [Google Scholar] [CrossRef] [PubMed]
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Opinion on Risk of Cancer Caused by Textiles and Leather Goods Coloured with Azo-Dyes|Scientific Committees, (n.d.). Available online: https://ec.europa.eu/health/scientific_committees/environmental_risks/opinions/sctee/sct_out27_en.htm (accessed on 19 August 2022).
- Deng, D.; Lamssali, M.; Aryal, N.; Ofori-Boadu, A.; Jha, M.K.; Samuel, R.E. Textiles wastewater treatment technology: A review. Water Environ. Res. 2020, 92, 1805–1810. [Google Scholar] [CrossRef]
- Genck, W.J. Section 18, Liquid-Solid Operations and Equipment. In Perry’s Chemical Engineers’ Handbook; McGraw-Hill: New York, NY, USA, 2008. [Google Scholar] [CrossRef]
- Flores Alarcón, M.A.D.; Arenas Jarro, R.Y.; Ahmed, M.A.; García Bustos, K.A.; Pacheco Tanaka, D.A.; Terán Hilares, R. Intensification of Red-G dye degradation used in the dyeing of alpaca wool by advanced oxidation processes assisted by hydrodynamic cavitation. Ultrason. Sonochem. 2022, 89, 106144. [Google Scholar] [CrossRef]
- Li, W.; Mu, B.; Yang, Y. Feasibility of industrial-scale treatment of dye wastewater via bio-adsorption technology. Bioresour. Technol. 2019, 277, 157–170. [Google Scholar] [CrossRef]
- Suteu, D.; Badeanu, M.; Malutan, T.; Chirculescu, A.-I. Valorization of food wastes (orange seeds) as adsorbent for dye retention from aqueous medium. Desalination Water Treat. 2016, 57, 29070–29081. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Bhatnagar, A.; Hameed, B.H.; Ok, Y.S.; Omirou, M. A review on waste-derived adsorbents from sugar industry for pollutant removal in water and wastewater. J. Mol. Liq. 2017, 240, 179–188. [Google Scholar] [CrossRef]
- Bhattacharjee, C.; Dutta, S.; Saxena, V.K. A review on biosorptive removal of dyes and heavy metals from wastewater using watermelon rind as biosorbent. Environ. Adv. 2020, 2, 100007. [Google Scholar] [CrossRef]
- Ali, S.M. Fabrication of a nanocomposite from an agricultural waste and its application as a biosorbent for organic pollutants. Int. J. Environ. Sci. Technol. 2018, 15, 1169–1178. [Google Scholar] [CrossRef]
- Rezzadori, K.; Benedetti, S.; Amante, E.R. Proposals for the residues recovery: Orange waste as raw material for new products. Food Bioprod. Process. 2012, 90, 606–614. [Google Scholar] [CrossRef]
- Akpata, M.I.; Akubor, P.I. Chemical composition and selected functional properties of sweet orange (Citrus sinensis) seed flour. Plant Foods Hum. Nutr. 1999, 54, 353–362. [Google Scholar] [CrossRef]
- Ahmad, M.A.; Eusoff, M.A.; Oladoye, P.O.; Adegoke, K.A.; Bello, O.S. Optimization and batch studies on adsorption of Methylene blue dye using pomegranate fruit peel based adsorbent. Chem. Data Collect. 2021, 32, 100676. [Google Scholar] [CrossRef]
- Prasad, N.; Kumar, P.; Pal, D.B. Cadmium removal from aqueous solution by jackfruit seed bio-adsorbent. SN Appl. Sci. 2020, 2, 1018. [Google Scholar] [CrossRef]
- Ali, N.S.; Jabbar, N.M.; Alardhi, S.M.; Majdi, H.S.; Albayati, T.M. Adsorption of methyl violet dye onto a prepared bio-adsorbent from date seeds: Isotherm, kinetics, and thermodynamic studies. Heliyon 2022, 8, e10276. [Google Scholar] [CrossRef] [PubMed]
- Keskin, B.; Ağtaş, M.; Ormancı-Acar, T.; Türken, T.; Imer, D.Y.; Ünal, S.; Menceloğlu, Y.Z.; Uçar-Demir, T.; Koyuncu, I. Halloysite nanotube blended nanocomposite ultrafiltration membranes for reactive dye removal. Water Sci. Technol. 2021, 83, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Babu, B.R.; Parande, A.K.; Raghu, S.; Prem Kumar, T. Textile Technology Cotton Textile Processing: Waste Generation and Effluent Treatment. J. Cotton Sci. 2007, 11, 141–153. Available online: http://journal.cotton.org (accessed on 20 August 2022).
- Aouni, A.; Fersi, C.; Cuartas-Uribe, B.; Bes-Pía, A.; Alcaina-Miranda, M.I.; Dhahbi, M. Reactive dyes rejection and textile effluent treatment study using ultrafiltration and nanofiltration processes. Desalination 2012, 297, 87–96. [Google Scholar] [CrossRef]
- Nadeem, K.; Guyer, G.T.; Keskinler, B.; Dizge, N. Investigation of segregated wastewater streams reusability with membrane process for textile industry. J. Clean. Prod. 2019, 228, 1437–1445. [Google Scholar] [CrossRef]
- American Public Health Association; American Water Works Association; Water Environment Association. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar] [CrossRef]
- Zygler, A.; Słomińska, M.; Namieśnik, J. Soxhlet Extraction and New Developments Such as Soxtec. Compr. Sampl. Sample Prep. 2012, 2, 65–82. [Google Scholar] [CrossRef]
- da Silva, M.D.; da Boit Martinello, K.; Knani, S.; Lütke, S.F.; Machado, L.M.M.; Manera, C.; Perondi, D.; Godinho, M.; Collazzo, G.C.; Silva, L.F.O.; et al. Pyrolysis of citrus wastes for the simultaneous production of adsorbents for Cu(II), H2, and d-limonene. Waste Manag. 2022, 152, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Brik, M.; Schoeberl, P.; Chamam, B.; Braun, R.; Fuchs, W. Advanced treatment of textile wastewater towards reuse using a membrane bioreactor. Process. Biochem. 2006, 41, 1751–1757. [Google Scholar] [CrossRef]
- Kehinde, F.; Aziz, H.A. Textile Waste Water and the advanced Oxidative Treatment Process, an Overview. Int. J. Innov. Res. Sci. Eng. Technol. 2014, 3, 15310–15317. [Google Scholar] [CrossRef]
- Avlonitis, S.A.; Poulios, I.; Sotiriou, D.; Pappas, M.; Moutesidis, K. Simulated cotton dye effluents treatment and reuse by nanofiltration. Desalination 2008, 221, 259–267. [Google Scholar] [CrossRef]
- Benkaddour, S.; Slimani, R.; Hiyane, H.; El Ouahabi, I.; Hachoumi, I.; El Antri, S.; Lazar, S. Removal of reactive yellow 145 by adsorption onto treated watermelon seeds: Kinetic and isotherm studies. Sustain. Chem. Pharm. 2018, 10, 16–21. [Google Scholar] [CrossRef]
- Asgher, M.; Bhatti, H.N. Removal of reactive blue 19 and reactive blue 49 textile dyes by citrus waste biomass from aqueous solution: Equilibrium and kinetic study. Can. J. Chem. Eng. 2012, 90, 412–419. [Google Scholar] [CrossRef]
- Safa, Y.; Bhatti, H.N.; Bhatti, I.A.; Asgher, M. Removal of direct Red-31 and direct Orange-26 by low cost rice husk: Influence of immobilisation and pretreatments. Can. J. Chem. Eng. 2011, 89, 1554–1565. [Google Scholar] [CrossRef]
- Salleh, M.A.M.; Mahmoud, D.K.; Karim, W.A.W.A.; Idris, A. Cationic and anionic dye adsorption by agricultural solid wastes: A comprehensive review. Desalination 2011, 280, 1–13. [Google Scholar] [CrossRef]
- Akar, T.; Tosun, I.; Kaynak, Z.; Ozkara, E.; Yeni, O.; Sahin, E.N.; Akar, S.T. An attractive agro-industrial by-product in environmental cleanup: Dye biosorption potential of untreated olive pomace. J. Hazard. Mater. 2009, 166, 1217–1225. [Google Scholar] [CrossRef]
- Yadav, J.; Sahu, O.; Marwah, H. Bioadsorption of dye from textile effluent with surface response methodology. Mater. Today Proc. 2022, 68, 937–942. [Google Scholar] [CrossRef]
- Nielsen, B.V.; Maneein, S.; Anghan, J.D.; Anghan, R.M.; Al Farid, M.; Milledge, J.J. Biosorption Potential of Sargassum for Removal of Aqueous Dye Solutions. Appl. Sci. 2022, 12, 4173. [Google Scholar] [CrossRef]
- Terán Hilares, R.; Sánchez Vera, F.P.; Colina Andrade, G.J.; Tejada Meza, K.; García, J.C.; Pacheco Tanaka, D.A. Continuous Cultivation of Microalgae in Cattle Slaughterhouse Wastewater Treated with Hydrodynamic Cavitation. Water 2022, 14, 1288. [Google Scholar] [CrossRef]
- Iaich, S.; Miyah, Y.; Elazhar, F.; Lagdali, S.; El-Habacha, M. Low-cost ceramic microfiltration membranes made from Moroccan clay for domestic wastewater and Congo Red dye treatment. Desalination Water Treat. 2021, 235, 251–271. [Google Scholar] [CrossRef]
- Luogo, B.D.P.; Salim, T.; Zhang, W.; Hartmann, N.B.; Malpei, F.; Candelario, V.M. Reuse of Water in Laundry Applications with Micro- and Ultrafiltration Ceramic Membrane. Membranes 2022, 12, 223. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Wu, X.; Ji, Y.; Hao, Y.; Liao, S.; Cui, Z.; Li, J.; Younas, M.; He, B. pH-responsive nanofiltration membrane containing chitosan for dye separation. J. Membr. Sci. 2021, 635, 119445. [Google Scholar] [CrossRef]
- Saini, P.; Bulasara, V.K.; Reddy, A.S. Performance of a new ceramic microfiltration membrane based on kaolin in textile industry wastewater treatment. Chem. Eng. Commun. 2019, 206, 227–236. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, J.; Gao, W. Reviewing textile wastewater produced by industries: Characteristics, environmental impacts, and treatment strategies. Water Sci. Technol. 2022, 85, 2076–2096. [Google Scholar] [CrossRef]
- Shrivastava, K.; Pokhriyal, S.; Dahiya, H. Application of Peanut Shell Bio Adsorbent to Improve Water Quality Parameters of Formazine and Clay Suspension. Nveo—Nat. Volatiles Essent. Oils J. Nveo 2021, 8, 3946–3957. Available online: https://www.nveo.org/index.php/journal/article/view/4166 (accessed on 4 October 2022).
- Bolade, O.O.; Sangodoyin, A.Y. Adsorption and Equilibrium Studies of Textile Effluent Treatment with Activated Snail Shell Carbon. IOSR J. Environ. Sci. 2018, 12, 26–33. [Google Scholar] [CrossRef]
- El Gaayda, J.; Titchou, F.E.; Barra, I.; Karmal, I.; Afanga, H.; Zazou, H.; Yap, P.-S.; Abidin, Z.Z.; Hamdani, M.; Akbour, R.A. Optimization of turbidity and dye removal from synthetic wastewater using response surface methodology: Effectiveness of Moringa oleifera seed powder as a green coagulant. J. Environ. Chem. Eng. 2022, 10, 106988. [Google Scholar] [CrossRef]
- Gautam, S.; Arora, A.S.; Singh, A.K.; Ekka, P.; Daniel, H.; Gokul, B.; Toppo, S.; Chockalingam, P.; Kumar, H.; Lyngdoh, J.F. Coa-gulation influencing parameters investigation on textile industry discharge using Strychnos potatorum seed powders. Environ. Dev. Sustain. 2021, 23, 5666–5673. [Google Scholar] [CrossRef]
- Yang, Z.-Y.; Gao, F.; Liu, J.-Z.; Yang, J.-S.; Liu, M.; Ge, Y.-M.; Chen, D.-Z.; Chen, J.-M. Improving sedimentation and lipid production of microalgae in the photobioreactor using saline wastewater. Bioresour. Technol. 2021, 347, 126392. [Google Scholar] [CrossRef]
- Li, B.; Bao, M.; Liu, Y.; Cheng, L.; Cui, B.; Hu, Z. Novel shortcut biological nitrogen removal using activated sludge-biofilm coupled with symbiotic algae. J. Water Process. Eng. 2021, 43, 102275. [Google Scholar] [CrossRef]




| Parameters | This Study | Kehinde et al. [28] | Avlonitis et al. [29] |
|---|---|---|---|
| pH | 8.84 | 6.95–11.8 | - |
| COD (mg O2/L) | 159 | 150–30,000 | 100 |
| Total phosphorous (mg P/L) | 2 | - | - |
| Total nitrogen (mg N/L) | 653 | - | - |
| Detergents (mg MBAS/L) | <0.061 | - | - |
| Salinity (PSU) | 2.19 | - | - |
| Total dissolved solids (mg/L) | 2048 | 2900–3100 | - |
| Turbidity (NTU) | 7–15 | - | - |
| Conductivity (µS/cm) | 4096 | - | 1000 |
| Run | Variables | Dye Adsorption (%) | ||||
|---|---|---|---|---|---|---|
| pH | Bio-Adsorbent (g/L) | Stirring Speed (rpm) | Temperature (°C) | Time (h) | ||
| 1 | 6 | 0.50 | 160 | 35 | 1 | 0.00 |
| 2 | 6 | 2.50 | 160 | 35 | 2 | 17.81 |
| 3 | 6 | 2.50 | 80 | 25 | 2 | 17.97 |
| 4 | 2 | 2.50 | 160 | 25 | 2 | 83.99 |
| 5 | 6 | 0.50 | 80 | 25 | 1 | 2.45 |
| 6 | 6 | 0.50 | 80 | 35 | 2 | 0.00 |
| 7 | 2 | 0.50 | 160 | 35 | 2 | 83.33 |
| 8 | 2 | 2.50 | 80 | 35 | 2 | 84.48 |
| 9 | 2 | 0.50 | 80 | 25 | 2 | 91.99 |
| 10 | 6 | 2.50 | 80 | 35 | 1 | 5.23 |
| 11 | 6 | 2.50 | 160 | 25 | 1 | 30.88 |
| 12 | 6 | 0.50 | 160 | 25 | 2 | 0.98 |
| 13 | 2 | 2.50 | 80 | 25 | 1 | 89.54 |
| 14 | 2 | 0.50 | 80 | 35 | 1 | 87.25 |
| 15 | 2 | 0.50 | 160 | 25 | 1 | 88.40 |
| 16 | 2 | 2.50 | 160 | 35 | 1 | 78.59 |
| Run | Variables | Dye Adsorption (%) | ||
|---|---|---|---|---|
| pH | Bio-Adsorbent Dosage (g/L) | Temperature (°C) | ||
| 1 | 4 | 0.20 | 30 | 10.78 |
| 2 | 3 | 1.00 | 25 | 96.22 |
| 3 | 3 | 0.20 | 35 | 46.11 |
| 4 | 2 | 1.00 | 30 | 81.20 |
| 5 | 3 | 0.60 | 30 | 89.21 |
| 6 | 4 | 1.00 | 30 | 57.07 |
| 7 | 3 | 0.60 | 30 | 87.23 |
| 8 | 3 | 0.20 | 25 | 79.80 |
| 9 | 4 | 0.60 | 35 | 43.21 |
| 10 | 4 | 0.60 | 25 | 79.99 |
| 11 | 2 | 0.20 | 30 | 68.80 |
| 12 | 3 | 0.60 | 30 | 88.31 |
| 13 | 2 | 0.60 | 35 | 90.19 |
| 14 | 3 | 1.00 | 35 | 90.11 |
| 15 | 2 | 0.60 | 25 | 95.05 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 8170.98 | 9 | 907.89 | 85.14 | <0.0001 * |
| A—pH | 2598.84 | 1 | 2598.84 | 243.73 | <0.0001 * |
| B—Dosage | 1773.40 | 1 | 1773.40 | 166.32 | <0.0001 * |
| C—Temperature | 829.06 | 1 | 829.06 | 77.75 | 0.0003 * |
| AB | 287.13 | 1 | 287.13 | 26.93 | 0.0035 * |
| AC | 254.72 | 1 | 254.72 | 23.89 | 0.0045 * |
| BC | 190.16 | 1 | 190.16 | 17.83 | 0.0083 * |
| A2 | 1113.87 | 1 | 1113.87 | 104.46 | 0.0002 * |
| B2 | 995.36 | 1 | 995.36 | 93.35 | 0.0002 * |
| C2 | 143.25 | 1 | 143.25 | 13.43 | 0.0145 * |
| Residual | 53.31 | 5 | 10.66 | ||
| Lack of fit | 51.35 | 3 | 17.12 | 17.42 | 0.0548 |
| Pure error | 1.97 | 2 | 0.9828 | ||
| Total | 8224.29 | 14 |
| Analysis | Crude TW | TW after Treatment with Bio-Adsorbent | Treated TW after Microfiltration (3 µm) |
|---|---|---|---|
| Absorbance unit (AU560nm) | 1.19 | 0.05 | 0.02 |
| Chemical oxygen demand (mg O2/L) | 159.3 ± 16.5 | 163.7 ± 16.6 | 165.4 ± 16.6 |
| Total phosphorus (mg P/L) | 2.35 ± 0.19 | 4.07 ± 0.33 | 4.71 ± 0.38 |
| Total nitrogen (mg N/L) | 653.7 ± 16.3 | 306.5 ± 7.7 | 411.3 ± 10.3 |
| Detergents (mg MBAS/L) | <0.061 | 0.366 ± 0.09 | 0.398 ± 0.1 |
| Salinity (PSU) | 2.19 | 2.32 | 2.24 |
| TDS (ppm) | 2048 | 2173 | 2104 |
| Turbidity (NTU) | 15.4 | 10.5 | 0.66 |
| Conductivity (µS/cm) | 4096 | 4345 | 4204 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores Alarcón, M.A.D.; Revilla Pacheco, C.; Garcia Bustos, K.; Tejada Meza, K.; Terán-Hilares, F.; Pacheco Tanaka, D.A.; Colina Andrade, G.J.; Terán-Hilares, R. Efficient Dye Removal from Real Textile Wastewater Using Orange Seed Powder as Suitable Bio-Adsorbent and Membrane Technology. Water 2022, 14, 4104. https://doi.org/10.3390/w14244104
Flores Alarcón MAD, Revilla Pacheco C, Garcia Bustos K, Tejada Meza K, Terán-Hilares F, Pacheco Tanaka DA, Colina Andrade GJ, Terán-Hilares R. Efficient Dye Removal from Real Textile Wastewater Using Orange Seed Powder as Suitable Bio-Adsorbent and Membrane Technology. Water. 2022; 14(24):4104. https://doi.org/10.3390/w14244104
Chicago/Turabian StyleFlores Alarcón, Miguel A. D., Claudia Revilla Pacheco, Kiara Garcia Bustos, Kevin Tejada Meza, Felix Terán-Hilares, David. A. Pacheco Tanaka, Gilberto J. Colina Andrade, and Ruly Terán-Hilares. 2022. "Efficient Dye Removal from Real Textile Wastewater Using Orange Seed Powder as Suitable Bio-Adsorbent and Membrane Technology" Water 14, no. 24: 4104. https://doi.org/10.3390/w14244104
APA StyleFlores Alarcón, M. A. D., Revilla Pacheco, C., Garcia Bustos, K., Tejada Meza, K., Terán-Hilares, F., Pacheco Tanaka, D. A., Colina Andrade, G. J., & Terán-Hilares, R. (2022). Efficient Dye Removal from Real Textile Wastewater Using Orange Seed Powder as Suitable Bio-Adsorbent and Membrane Technology. Water, 14(24), 4104. https://doi.org/10.3390/w14244104