Bioremediation Treatment of Polyaromatic Hydrocarbons for Environmental Sustainability
Abstract
:1. Introduction
2. PAHS Aromatic Hydrocarbons
3. The Effects of Oil Pollution on Ecosystems and Human Health
4. Bioremediation of Petroleum Hydrocarbons
Genetically Modified Organisms
5. The Function of Microorganisms
6. Aerobic Decomposition
Anaerobic Decomposition
7. Reactions Involved in the Breakdown of Hydrocarbons
Decomposition Reaction and Energy Requirements
8. Enzyme-Based Decomposition of Hydrocarbons
8.1. Factors Affecting the Decomposition of Petroleum Hydrocarbons
8.1.1. Temperature
8.1.2. Nutrients
8.1.3. pH
8.1.4. Oxygen
8.2. Kinetics for the Biodegradation of PAHs
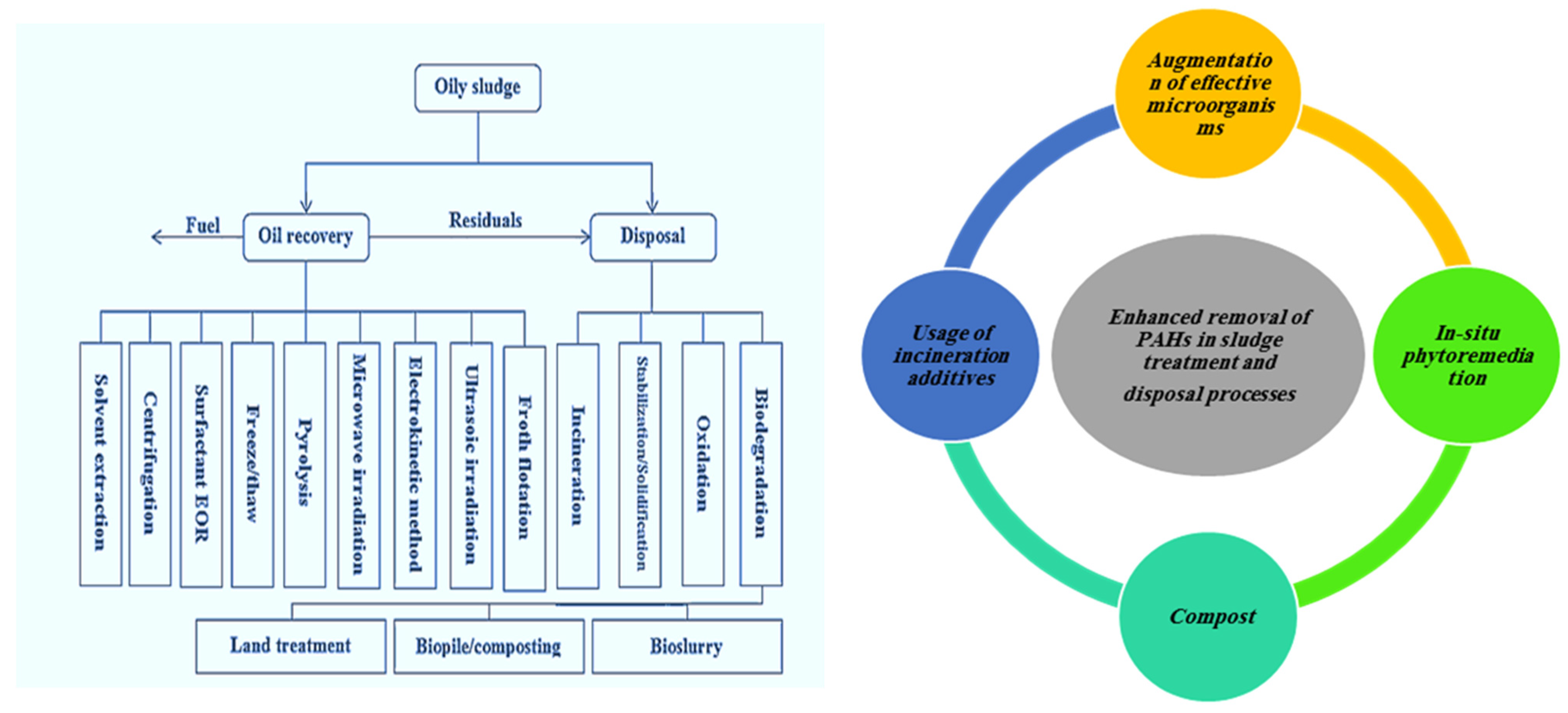
8.3. Overview of Sludge Management Methods PAHs
8.4. Challenges, Limitations, and Future Perspective
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lawal, A.T. Polycyclic aromatic hydrocarbons. A review. Cogent Environ. Sci. 2017, 3, 1339841. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, L.; Zhang, H.; Xing, W.; Wang, Y.; Bai, P.; Zhang, L.; Hayakawa, K.; Toriba, A.; Wei, Y. Assessing approaches of human inhalation exposure to polycyclic aromatic hydrocarbons: A review. Int. J. Environ. Res. Public Health 2021, 18, 3124. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafy, H.I.; Mansour, M.S. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef] [Green Version]
- Mallah, M.A.; Changxing, L.; Mallah, M.A.; Noreen, S.; Liu, Y.; Saeed, M.; Xi, H.; Ahmed, B.; Feng, F.; Mirjat, A.A. Polycyclic aromatic hydrocarbon and its effects on human health: An updated review. Chemosphere 2022, 296, 133948. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D.; Ferguson, M.; Datta, R.; Birnbaum, S. Bioremediation of petroleum hydrocarbons in contaminated soils: Comparison of biosolids addition, carbon supplementation, and monitored natural attenuation. Environ. Pollut. 2005, 136, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Sniegowski, K.; Vanhecke, M.; D’Huys, P.-J.; Braeken, L. Potential of activated carbon to recover Randomly-methylated-B-cyclodextrin solution from waste water originating from in situ soil flushing. In Proceedings of the Aquaconsoil, Barcelona, Spain, 16–19 April 2013. [Google Scholar]
- Xu, X.; Liu, W.; Tian, S.; Wang, W.; Qi, Q.; Jiang, P.; Gao, X.; Li, F.; Li, H.; Yu, H. Petroleum hydrocarbon-degrading bacteria for the remediation of oil pollution under aerobic conditions: A perspective analysis. Front. Microbiol. 2018, 9, 2885. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Vo, D.-V.N.; Jeevanantham, S.; Karishma, S.; Yaashikaa, P. A review on catalytic-enzyme degradation of toxic environmental pollutants: Microbial enzymes. J. Hazard. Mater. 2021, 419, 126451. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Gholami, A.; Omidifar, N.; Zarei, M.; Bahrani, S.; Yousefi, K.; Chiang, W.-H.; Babapoor, A. Bioinorganic synthesis of polyrhodanine stabilized Fe3O4/Graphene oxide in microbial supernatant media for anticancer and antibacterial applications. Bioinorg. Chem. Appl. 2021, 2021, 9972664. [Google Scholar] [CrossRef]
- Ossai, I.C.; Ahmed, A.; Hassan, A.; Hamid, F.S. Remediation of soil and water contaminated with petroleum hydrocarbon: A review. Environ. Technol. Innov. 2020, 17, 100526. [Google Scholar]
- Eze, M.O.; Hose, G.C.; George, S.C.; Daniel, R. Diversity and metagenome analysis of a hydrocarbon-degrading bacterial consortium from asphalt lakes located in Wietze, Germany. AMB Express 2021, 11, 89. [Google Scholar] [CrossRef]
- Azhdari, R.; Mousavi, S.M.; Hashemi, S.A.; Bahrani, S.; Ramakrishna, S. Decorated graphene with aluminum fumarate metal organic framework as a superior non-toxic agent for efficient removal of Congo Red dye from wastewater. J. Environ. Chem. Eng. 2019, 7, 103437. [Google Scholar] [CrossRef]
- Gayathiri, E.; Prakash, P.; Karmegam, N.; Varjani, S.; Awasthi, M.K.; Ravindran, B. Biosurfactants: Potential and eco-friendly material for sustainable agriculture and environmental safety—A review. Agronomy 2022, 12, 662. [Google Scholar] [CrossRef]
- Fathepure, B.Z. Recent studies in microbial degradation of petroleum hydrocarbons in hypersaline environments. Front. Microbiol. 2014, 5, 173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mousavi, S.M.; Zarei, M.; Hashemi, S.A.; Ramakrishna, S.; Chiang, W.-H.; Lai, C.W.; Gholami, A. Gold nanostars-diagnosis, bioimaging and biomedical applications. Drug Metab. Rev. 2020, 52, 299–318. [Google Scholar] [CrossRef]
- Lin, C.; Gan, L.; Chen, Z.-L. Biodegradation of naphthalene by strain Bacillus fusiformis (BFN). J. Hazard. Mater. 2010, 182, 771–777. [Google Scholar] [CrossRef]
- Abootalebi, S.N.; Saeed, A.; Gholami, A.; Mohkam, M.; Kazemi, A.; Nezafat, N.; Mousavi, S.M.; Hashemi, S.A.; Shorafa, E. Screening, characterization and production of thermostable alpha-amylase produced by a novel thermophilic Bacillus megaterium isolated from pediatric intensive care unit. J. Environ. Treat. Tech. 2020, 8, 952–960. [Google Scholar]
- Mohkam, M.; Rasoul-Amini, S.; Shokri, D.; Berenjian, A.; Rahimi, F.; Sadraeian, M.; Khalvati, B.; Gholami, A.; Ghasemi, Y. Characterization and in vitro probiotic assessment of potential indigenous Bacillus strains isolated from soil rhizosphere. Minerva Biotecnol. 2016, 28, 19–28. [Google Scholar]
- Ahmed, S.; Aitani, A.; Rahman, F.; Al-Dawood, A.; Al-Muhaish, F. Decomposition of hydrocarbons to hydrogen and carbon. Appl. Catal. A Gen. 2009, 359, 1–24. [Google Scholar] [CrossRef]
- Varjani, S.J.; Upasani, V.N. A new look on factors affecting microbial degradation of petroleum hydrocarbon pollutants. Int. Biodeterior. Biodegrad. 2017, 120, 71–83. [Google Scholar] [CrossRef]
- Megharaj, M.; Ramakrishnan, B.; Venkateswarlu, K.; Sethunathan, N.; Naidu, R. Bioremediation approaches for organic pollutants: A critical perspective. Environ. Int. 2011, 37, 1362–1375. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Yari Kalashgrani, M.; Omidifar, N.; Lai, C.W.; Vijayakameswara Rao, N.; Gholami, A.; Chiang, W.-H. The Pivotal Role of Quantum Dots-Based Biomarkers Integrated with Ultra-Sensitive Probes for Multiplex Detection of Human Viral Infections. Pharmaceuticals 2022, 15, 880. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.-M.; Nejad, Z.M.; Hashemi, S.A.; Salari, M.; Gholami, A.; Ramakrishna, S.; Chiang, W.-H.; Lai, C.W. Bioactive agent-loaded electrospun nanofiber membranes for accelerating healing process: A review. Membranes 2021, 11, 702. [Google Scholar] [CrossRef]
- Gholami, A.; Mohammadi, F.; Ghasemi, Y.; Omidifar, N.; Ebrahiminezhad, A. Antibacterial activity of SPIONs versus ferrous and ferric ions under aerobic and anaerobic conditions: A preliminary mechanism study. IET Nanobiotechnol. 2020, 14, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Muradov, N. Emission-free fuel reformers for mobile and portable fuel cell applications. J. Power Sources 2003, 118, 320–324. [Google Scholar] [CrossRef]
- Atlas, R.M.; Hazen, T.C. Oil Biodegradation and Bioremediation: A Tale of the Two Worst Spills in US History; ACS Publications: Columbus, OH, USA, 2011. [Google Scholar]
- Mousavi, S.M.; Hashemi, S.A.; Ghahramani, Y.; Azhdari, R.; Yousefi, K.; Gholami, A.; Fallahi Nezhad, F.; Vijayakameswara Rao, N.; Omidifar, N.; Chiang, W.-H. Antiproliferative and Apoptotic Effects of Graphene Oxide@ AlFu MOF Based Saponin Natural Product on OSCC Line. Pharmaceuticals 2022, 15, 1137. [Google Scholar] [CrossRef] [PubMed]
- Vidonish, J.E.; Zygourakis, K.; Masiello, C.A.; Sabadell, G.; Alvarez, P.J. Thermal treatment of hydrocarbon-impacted soils: A review of technology innovation for sustainable remediation. Engineering 2016, 2, 426–437. [Google Scholar] [CrossRef]
- Premnath, N.; Mohanrasu, K.; Rao, R.G.R.; Dinesh, G.; Prakash, G.S.; Ananthi, V.; Ponnuchamy, K.; Muthusamy, G.; Arun, A. A crucial review on polycyclic aromatic Hydrocarbons-Environmental occurrence and strategies for microbial degradation. Chemosphere 2021, 280, 130608. [Google Scholar]
- Eldos, H.I.; Zouari, N.; Saeed, S.; Al-Ghouti, M.A. Recent advances in the treatment of PAHs in the environment: Application of nanomaterial-based technologies. Arab. J. Chem. 2022, 15, 103918. [Google Scholar] [CrossRef]
- Johnston, J.E.; Lim, E.; Roh, H. Impact of upstream oil extraction and environmental public health: A review of the evidence. Sci. Total Environ. 2019, 657, 187–199. [Google Scholar] [CrossRef]
- Bautista, O.; Méndez, F.; Trevino, C. Theoretical analysis of the direct decomposition of methane gas in a laminar stagnation-point flow: CO2-free production of hydrogen. Int. J. Hydrogen Energy 2008, 33, 7419–7426. [Google Scholar] [CrossRef]
- Luo, L.; Wang, P.; Lin, L.; Luan, T.; Ke, L.; Tam, N.F.Y. Removal and transformation of high molecular weight polycyclic aromatic hydrocarbons in water by live and dead microalgae. Process Biochem. 2014, 49, 1723–1732. [Google Scholar] [CrossRef]
- Loyeh, E.; Mohsenpour, R. Investigation of oil pollution on aquatic animals and methods of its prevention. J. Aquac. Mar. Biol. 2020, 9, 160–165. [Google Scholar]
- Mohanty, M. Potential Applications of Biosurfactant from Marine Bacteria in Bioremediation. Master’s Thesis, National Institute of Technology, Rourkela, India, 2013. [Google Scholar]
- Alireza Hashemi, S.; Bahrani, S.; Mojtaba Mousavi, S.; Mojoudi, F.; Omidifar, N.; Bagheri Lankarani, K.; Arjmand, M.; Ramakrishna, S. Development of sulfurized Polythiophene-Silver Iodide-Diethyldithiocarbamate nanoflakes toward Record-High and selective absorption and detection of mercury derivatives in aquatic substrates. Chem. Eng. J. 2022, 440, 135896. [Google Scholar] [CrossRef]
- Islam, M.S.; Tanaka, M. Impacts of pollution on coastal and marine ecosystems including coastal and marine fisheries and approach for management: A review and synthesis. Mar. Pollut. Bull. 2004, 48, 624–649. [Google Scholar] [CrossRef]
- Kalashgarani, M.Y.; Babapoor, A. Application of nano-antibiotics in the diagnosis and treatment of infectious diseases. Adv. Appl. NanoBio-Technol. 2022, 3, 22–35. [Google Scholar]
- Mousavi, S.M.; Hashemi, S.A.; Kalashgrani, M.Y.; Omidifar, N.; Bahrani, S.; Vijayakameswara Rao, N.; Babapoor, A.; Gholami, A.; Chiang, W.-H. Bioactive Graphene Quantum Dots Based Polymer Composite for Biomedical Applications. Polymers 2022, 14, 617. [Google Scholar] [CrossRef] [PubMed]
- Tam, N.F.; Wong, T.W.; Wong, Y. A case study on fuel oil contamination in a mangrove swamp in Hong Kong. Mar. Pollut. Bull. 2005, 51, 1092–1100. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.M.; Hashemi, S.A.; Iman Moezzi, S.M.; Ravan, N.; Gholami, A.; Lai, C.W.; Chiang, W.-H.; Omidifar, N.; Yousefi, K.; Behbudi, G. Recent advances in enzymes for the bioremediation of pollutants. Biochem. Res. Int. 2021, 2021, 5599204. [Google Scholar] [CrossRef]
- Hall, J.; Matos, S.; Bachor, V. From green technology development to green innovation: Inducing regulatory adoption of pathogen detection technology for sustainable forestry. Small Bus. Econ. 2019, 52, 877–889. [Google Scholar] [CrossRef] [Green Version]
- Hamouda, R.A.; Daassi, D.; Hassan, H.A.; Hussein, M.H.; El-Sheekh, M.M. Use of live microbes for oil degradation in situ. In Advances in Oil-Water Separation; Elsevier: Berlin/Heidelberg, Germany, 2022; pp. 297–317. [Google Scholar]
- Hosseini, H.; Mousavi, S.M.; Wurm, F.R.; Goodarzi, V. Display of hidden properties of flexible aerogel based on bacterial cellulose/polyaniline nanocomposites with helping of multiscale modeling. Eur. Polym. J. 2021, 146, 110251. [Google Scholar] [CrossRef]
- Safdel, M.; Anbaz, M.A.; Daryasafar, A.; Jamialahmadi, M. Microbial enhanced oil recovery, a critical review on worldwide implemented field trials in different countries. Renew. Sustain. Energy Rev. 2017, 74, 159–172. [Google Scholar] [CrossRef]
- Abbasian, F.; Lockington, R.; Mallavarapu, M.; Naidu, R. A comprehensive review of aliphatic hydrocarbon biodegradation by bacteria. Appl. Biochem. Biotechnol. 2015, 176, 670–699. [Google Scholar] [CrossRef] [PubMed]
- Karigar, C.S.; Rao, S.S. Role of microbial enzymes in the bioremediation of pollutants: A review. Enzym. Res. 2011, 2011, 805187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarsaiya, S.; Awasthi, S.K.; Jain, A.; Mishra, S.; Jia, Q.; Shu, F.; Li, J.; Duan, Y.; Singh, R.; Awasthi, M.K. Recent Developments in the Treatment of Petroleum Hydrocarbon and Oily Sludge from the Petroleum Industry. In Biological Processing of Solid Waste; CRC Press: Boca Raton, FL, USA, 2019; Volume 277. [Google Scholar]
- Varner, P.M.; Gunsch, C.K. Properties affecting transfer and expression of degradative plasmids for the purpose of bioremediation. Biodegradation 2021, 32, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Obayori, O.S.; Salam, L.B. Degradation of polycyclic aromatic hydrocarbons: Role of plasmids. Sci. Res. Essays 2010, 5, 4093–4106. [Google Scholar]
- Das, N.; Chandran, P. Microbial degradation of petroleum hydrocarbon contaminants: An overview. Biotechnol. Res. Int. 2011, 2011, 941810. [Google Scholar] [CrossRef] [Green Version]
- Al-Hawash, A.B.; Dragh, M.A.; Li, S.; Alhujaily, A.; Abbood, H.A.; Zhang, X.; Ma, F. Principles of microbial degradation of petroleum hydrocarbons in the environment. Egypt. J. Aquat. Res. 2018, 44, 71–76. [Google Scholar] [CrossRef]
- Liu, X.; Li, Z.; Zhang, C.; Tan, X.; Yang, X.; Wan, C.; Lee, D.-J. Enhancement of anaerobic degradation of petroleum hydrocarbons by electron intermediate: Performance and mechanism. Bioresour. Technol. 2020, 295, 122305. [Google Scholar] [CrossRef]
- Wilkes, H.; Buckel, W.; Golding, B.T.; Rabus, R. Metabolism of hydrocarbons in n-alkane-utilizing anaerobic bacteria. Microb. Physiol. 2016, 26, 138–151. [Google Scholar] [CrossRef]
- Bahrani, S.; Hashemi, S.A.; Mousavi, S.M.; Azhdari, R. Zinc-based metal–organic frameworks as nontoxic and biodegradable platforms for biomedical applications: Review study. Drug Metab. Rev. 2019, 51, 356–377. [Google Scholar] [CrossRef]
- Truskewycz, A.; Gundry, T.D.; Khudur, L.S.; Kolobaric, A.; Taha, M.; Aburto-Medina, A.; Ball, A.S.; Shahsavari, E. Petroleum hydrocarbon contamination in terrestrial ecosystems—Fate and microbial responses. Molecules 2019, 24, 3400. [Google Scholar] [CrossRef] [Green Version]
- Mousavi, S.M.; Hashemi, S.A.; Kalashgrani, M.Y.; Gholami, A.; Omidifar, N.; Babapoor, A.; Vijayakameswara Rao, N.; Chiang, W.-H. Recent Advances in Plasma-Engineered Polymers for Biomarker-Based Viral Detection and Highly Multiplexed Analysis. Biosensors 2022, 12, 286. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, D.; Ghosh, S.; Dutta, T.K.; Ahn, Y. Current state of knowledge in microbial degradation of polycyclic aromatic hydrocarbons (PAHs): A review. Front. Microbiol. 2016, 7, 1369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, B.; Shukla, P. Futuristic avenues of metabolic engineering techniques in bioremediation. Biotechnol. Appl. Biochem. 2022, 69, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, B.S.; Esmaeili, H.; Foroutan, R.; Mousavi, S.M.; Hashemi, S.A. Removal of Cd2+ from aqueous solution using eucalyptus sawdust as a bio-adsorbent: Kinetic and equilibrium studies. J. Environ. Treat. Tech. 2020, 8, 112–118. [Google Scholar]
- Li, X.; Li, H.; Qu, C. A review of the mechanism of microbial degradation of petroleum pollution. IOP Conf. Ser. Mater. Sci. Eng. 2019, 484, 012060. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Bahrani, S.; Yousefi, K.; Behbudi, G.; Babapoor, A.; Omidifar, N.; Lai, C.W.; Gholami, A.; Chiang, W.-H. Recent advancements in polythiophene-based materials and their biomedical, geno sensor and DNA detection. Int. J. Mol. Sci. 2021, 22, 6850. [Google Scholar] [CrossRef]
- Shen, T.; Pi, Y.; Bao, M.; Xu, N.; Li, Y.; Lu, J. Biodegradation of different petroleum hydrocarbons by free and immobilized microbial consortia. Environ. Sci. Process. Impacts 2015, 17, 2022–2033. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Bahrani, S.; Sadrmousavi-Dizaj, A.; Arjmand, O.; Omidifar, N.; Lai, C.W.; Chiang, W.-H.; Gholami, A. Bioinorganic Synthesis of Sodium Polytungstate/Polyoxometalate in Microbial Kombucha Media for Precise Detection of Doxorubicin. Bioinorg. Chem. Appl. 2022, 2022, 2265108. [Google Scholar] [CrossRef]
- Pinheiro Pires, A.P.; Arauzo, J.; Fonts, I.; Domine, M.E.; Fernández Arroyo, A.; Garcia-Perez, M.E.; Montoya, J.; Chejne, F.; Pfromm, P.; Garcia-Perez, M. Challenges and opportunities for bio-oil refining: A review. Energy Fuels 2019, 33, 4683–4720. [Google Scholar] [CrossRef]
- Thavasi, R.; Jayalakshmi, S.; Banat, I.M. Application of biosurfactant produced from peanut oil cake by Lactobacillus delbrueckii in biodegradation of crude oil. Bioresour. Technol. 2011, 102, 3366–3372. [Google Scholar] [CrossRef] [PubMed]
- Jafari, A.; Zamankhan, P.; Mousavi, S.M.; Henttinen, K. Multiscale modeling of fluid turbulence and flocculation in fiber suspensions. J. Appl. Phys. 2006, 100, 034901. [Google Scholar] [CrossRef]
- Xu, D.; Zhang, K.; Li, B.-G.; Mbadinga, S.M.; Zhou, L.; Liu, J.-F.; Yang, S.-Z.; Gu, J.-D.; Mu, B.-Z. Simulation of in situ oil reservoir conditions in a laboratory bioreactor testing for methanogenic conversion of crude oil and analysis of the microbial community. Int. Biodeterior. Biodegrad. 2019, 136, 24–33. [Google Scholar] [CrossRef]
- Kertesz, M.A.; Kawasaki, A.; Stolz, A. Aerobic hydrocarbon-degrading alphaproteobacteria: Sphingomonadales. In Taxonomy, Genomics and Ecophysiology of Hydrocarbon-Degrading Microbes; Springer: Berlin/Heidelberg, Germany, 2019; pp. 105–124. [Google Scholar]
- Niu, J.; Liu, Q.; Lv, J.; Peng, B. Review on microbial enhanced oil recovery: Mechanisms, modeling and field trials. J. Pet. Sci. Eng. 2020, 192, 107350. [Google Scholar] [CrossRef]
- Omidifar, N.; Nili-Ahmadabadi, A.; Nakhostin-Ansari, A.; Lankarani, K.B.; Moghadami, M.; Mousavi, S.M.; Hashemi, S.A.; Gholami, A.; Shokripour, M.; Ebrahimi, Z. The modulatory potential of herbal antioxidants against oxidative stress and heavy metal pollution: Plants against environmental oxidative stress. Environ. Sci. Pollut. Res. 2021, 28, 61908–61918. [Google Scholar] [CrossRef] [PubMed]
- Alegbeleye, O.O.; Opeolu, B.O.; Jackson, V.A. Polycyclic aromatic hydrocarbons: A critical review of environmental occurrence and bioremediation. Environ. Manag. 2017, 60, 758–783. [Google Scholar]
- Chandra, S.; Sharma, R.; Singh, K.; Sharma, A. Application of bioremediation technology in the environment contaminated with petroleum hydrocarbon. Ann. Microbiol. 2013, 63, 417–431. [Google Scholar] [CrossRef]
- Wartell, B.; Boufadel, M.; Rodriguez-Freire, L. An effort to understand and improve the anaerobic biodegradation of petroleum hydrocarbons: A literature review. Int. Biodeterior. Biodegrad. 2021, 157, 105156. [Google Scholar]
- Meckenstock, R.U.; Boll, M.; Mouttaki, H.; Koelschbach, J.S.; Tarouco, P.C.; Weyrauch, P.; Dong, X.; Himmelberg, A.M. Anaerobic degradation of benzene and polycyclic aromatic hydrocarbons. Microb. Physiol. 2016, 26, 92–118. [Google Scholar] [CrossRef]
- Kalashgrani, M.Y.; Harzand, F.V.; Javanmardi, N.; Nejad, F.F.; Rahmanian, V. Recent Advances in Multifunctional magnetic nano platform for Biomedical Applications: A mini review. Adv. Appl. NanoBio-Technol. 2022, 3, 31–37. [Google Scholar]
- Hashemi, S.A.; Mousavi, S.M.; Bahrani, S.; Ramakrishna, S.; Hashemi, S.H. Picomolar-level detection of mercury within non-biological/biological aqueous media using ultra-sensitive polyaniline-Fe3O4-silver diethyldithiocarbamate nanostructure. Anal. Bioanal. Chem. 2020, 412, 5353–5365. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, E.; Liu, F.; Xu, M. Interactions of PAH-degradation and nitrate-/sulfate-reducing assemblages in anaerobic sediment microbial community. J. Hazard. Mater. 2020, 388, 122068. [Google Scholar] [CrossRef] [PubMed]
- Subashchandrabose, S.R.; Venkateswarlu, K.; Venkidusamy, K.; Palanisami, T.; Naidu, R.; Megharaj, M. Bioremediation of soil long-term contaminated with PAHs by algal–bacterial synergy of Chlorella sp. MM3 and Rhodococcus wratislaviensis strain 9 in slurry phase. Sci. Total Environ. 2019, 659, 724–731. [Google Scholar] [CrossRef] [PubMed]
- Ojha, N.; Mandal, S.K.; Das, N. Enhanced degradation of indeno(1,2,3-cd)pyrene using Candida tropicalis NN4 in presence of iron nanoparticles and produced biosurfactant: A statistical approach. 3 Biotech 2019, 9, 86. [Google Scholar] [CrossRef]
- Mehetre, G.T.; Dastager, S.G.; Dharne, M.S. Biodegradation of mixed polycyclic aromatic hydrocarbons by pure and mixed cultures of biosurfactant producing thermophilic and thermo-tolerant bacteria. Sci. Total Environ. 2019, 679, 52–60. [Google Scholar] [CrossRef]
- Elyamine, A.M.; Kan, J.; Meng, S.; Tao, P.; Wang, H.; Hu, Z. Aerobic and anaerobic bacterial and fungal degradation of pyrene: Mechanism pathway including biochemical reaction and catabolic genes. Int. J. Mol. Sci. 2021, 22, 8202. [Google Scholar] [CrossRef]
- Holladay, J.D.; Hu, J.; King, D.L.; Wang, Y. An overview of hydrogen production technologies. Catal. Today 2009, 139, 244–260. [Google Scholar] [CrossRef]
- Kalashgrani, M.Y.; Nejad, F.F.; Rahmanian, V. Carbon Quantum Dots Platforms: As nano therapeutic for Biomedical Applications. Adv. Appl. NanoBio-Technol. 2022, 3, 38–42. [Google Scholar]
- Hosseini, H.; Mousavi, S.M. Bacterial cellulose/polyaniline nanocomposite aerogels as novel bioadsorbents for removal of hexavalent chromium: Experimental and simulation study. J. Clean. Prod. 2021, 278, 123817. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Parvin, N.; Gholami, A.; Ramakrishna, S.; Omidifar, N.; Moghadami, M.; Chiang, W.-H.; Mazraedoost, S. Recent biotechnological approaches for treatment of novel COVID-19: From bench to clinical trial. Drug Metab. Rev. 2021, 53, 141–170. [Google Scholar] [CrossRef]
- Fulcheri, L.; Probst, N.; Flamant, G.; Fabry, F.; Grivei, E.; Bourrat, X. Plasma processing: A step towards the production of new grades of carbon black. Carbon 2002, 40, 169–176. [Google Scholar] [CrossRef]
- Muradov, N.; Smith, F.; Huang, C.; Ali, T. Autothermal catalytic pyrolysis of methane as a new route to hydrogen production with reduced CO2 emissions. Catal. Today 2006, 116, 281–288. [Google Scholar] [CrossRef]
- Choudhary, T.; Goodman, D. Methane decomposition: Production of hydrogen and carbon filaments. Catalysis 2006, 19, 164–183. [Google Scholar]
- Okolafor, F.; Ekhaise, F.O. Microbial Enzyme Remediation of Poly-Aromatic Hydrocarbon (PAH’s): A review. J. Int. Environ. Appl. Sci. 2022, 17, 10–21. [Google Scholar]
- Omidifar, N.; Nili-Ahmadabadi, A.; Gholami, A.; Dastan, D.; Ahmadimoghaddam, D.; Nili-Ahmadabadi, H. Biochemical and Histological Evidence on the Protective Effects of Allium hirtifolium Boiss (Persian Shallot) as an Herbal Supplement in Cadmium-Induced Hepatotoxicity. Evid Based Complement Altern. Med. 2020, 2020, 7457504. [Google Scholar] [CrossRef]
- Hassanshahian, M.; Emtiazi, G.; Caruso, G.; Cappello, S. Bioremediation (bioaugmentation/biostimulation) trials of oil polluted seawater: A mesocosm simulation study. Mar. Environ. Res. 2014, 95, 28–38. [Google Scholar] [CrossRef]
- van Beilen, J.B.; Funhoff, E.G. Expanding the alkane oxygenase toolbox: New enzymes and applications. Curr. Opin. Biotechnol. 2005, 16, 308–314. [Google Scholar] [CrossRef]
- Van Beilen, J.B.; Funhoff, E.G. Alkane hydroxylases involved in microbial alkane degradation. Appl. Microbiol. Biotechnol. 2007, 74, 13–21. [Google Scholar] [CrossRef] [Green Version]
- Zimmer, T.; Ohkuma, M.; Ohta, A.; Takagi, M.; Schunck, W.-H. The CYP52 Multigene Family of Candida maltose Encodes Functionally Diverse n-Alkane-Inducible Cytochromes P450. Biochem. Biophys. Res. Commun. 1996, 224, 784–789. [Google Scholar] [CrossRef]
- Gupte, A.; Tripathi, A.; Patel, H.; Rudakiya, D.; Gupte, S. Bioremediation of polycyclic aromatic hydrocarbon (PAHs): A perspective. Open Biotechnol. J. 2016, 10, 363–378. [Google Scholar] [CrossRef] [Green Version]
- Kadri, T.; Rouissi, T.; Brar, S.K.; Cledon, M.; Sarma, S.; Verma, M. Biodegradation of polycyclic aromatic hydrocarbons (PAHs) by fungal enzymes: A review. J. Environ. Sci. 2017, 51, 52–74. [Google Scholar] [CrossRef] [PubMed]
- Ekpo, M.; Udofia, U. Rate of biodegradation of crude oil by microorganisms isolated from oil sludge environment. Afr. J. Biotechnol. 2008, 7, 4495–4499. [Google Scholar]
- Iranzo, M.; Sainz-Pardo, I.; Boluda, R.; Sanchez, J.; Mormeneo, S. The use of microorganisms in environmental remediation. Ann. Microbiol. 2001, 51, 135–144. [Google Scholar]
- Adams, G.; Tawari-Fufeyin, P.; Igelenyah, E. Bioremediation of spent oil contaminated soils using poultry litter. Res. J. Eng. Appl. Sci. 2014, 3, 124–130. [Google Scholar]
- Graj, W.; Lisiecki, P.; Szulc, A.; Chrzanowski, Ł.; Wojtera-Kwiczor, J. Bioaugmentation with petroleum-degrading consortia has a selective growth-promoting impact on crop plants germinated in diesel oil-contaminated soil. Water Air Soil Pollut. 2013, 224, 1676. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.; Zhang, H.; Sun, L.; Qi, G.; Chen, S.; Zhao, X. Microbial community composition is related to soil biological and chemical properties and bacterial wilt outbreak. Sci. Rep. 2017, 7, 343. [Google Scholar] [CrossRef] [Green Version]
- Cutright, T.J. Polycyclic aromatic hydrocarbon biodegradation and kinetics using Cunninghamella echinulata var. elegans. Int. Biodeterior. Biodegrad. 1995, 35, 397–408. [Google Scholar] [CrossRef]
- Haritash, A.; Kaushik, C. Biodegradation aspects of polycyclic aromatic hydrocarbons (PAHs): A review. J. Hazard. Mater. 2009, 169, 1–15. [Google Scholar] [CrossRef]
- Hu, G.; Li, J.; Zeng, G. Recent development in the treatment of oily sludge from petroleum industry: A review. J. Hazard. Mater. 2013, 261, 470–490. [Google Scholar]
- Mailler, R.; Gasperi, J.; Patureau, D.; Vulliet, E.; Delgenes, N.; Danel, A.; Deshayes, S.; Eudes, V.; Guerin, S.; Moilleron, R. Fate of emerging and priority micropollutants during the sewage sludge treatment: Case study of Paris conurbation. Part 1: Contamination of the different types of sewage sludge. Waste Manag. 2017, 59, 379–393. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Yu, T.; Li, X.; Yao, J.; Liu, W.; Chang, S.; Chen, Y. The fate and enhanced removal of polycyclic aromatic hydrocarbons in wastewater and sludge treatment system: A review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 1425–1475. [Google Scholar] [CrossRef]
- Zhou, W.; Lu, Y.; Jiang, S.; Xiao, Y.; Zheng, G.; Zhou, L. Impact of sludge conditioning treatment on the bioavailability of pyrene in sewage sludge. Ecotoxicol. Environ. Saf. 2018, 163, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Siebielska, I. Comparison of changes in selected polycyclic aromatic hydrocarbons concentrations during the composting and anaerobic digestion processes of municipal waste and sewage sludge mixtures. Water Sci. Technol. 2014, 70, 1617–1624. [Google Scholar] [CrossRef]
- Mezzanotte, V.; Anzano, M.; Collina, E.; Marazzi, F.A.; Lasagni, M. Distribution and removal of polycyclic aromatic hydrocarbons in two Italian municipal wastewater treatment plants in 2011–2013. Polycycl. Aromat. Compd. 2016, 36, 213–228. [Google Scholar] [CrossRef]
- Singh, S.; Haritash, A. Polycyclic aromatic hydrocarbons: Soil pollution and remediation. Int. J. Environ. Sci. Technol. 2019, 16, 6489–6512. [Google Scholar]
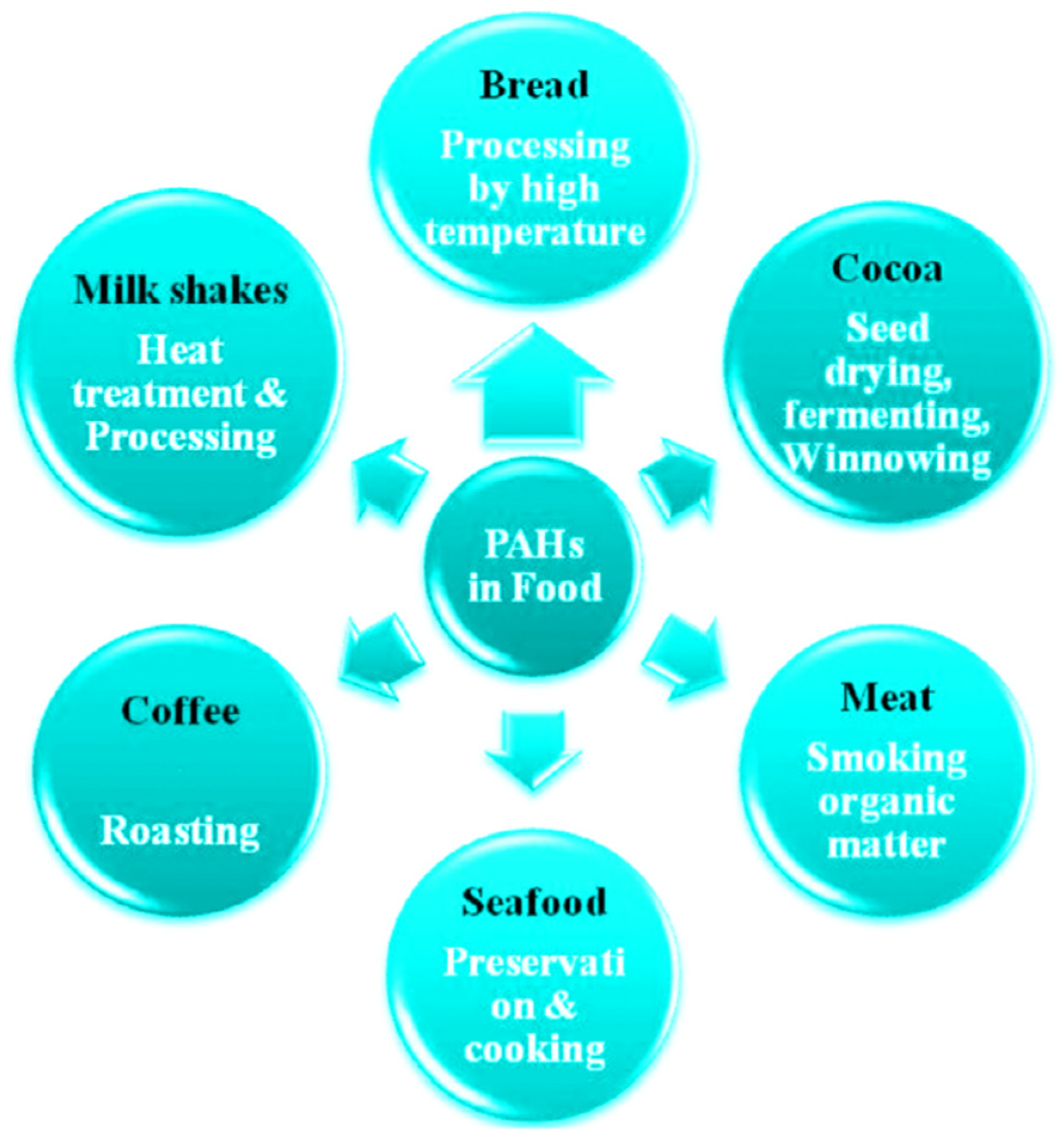
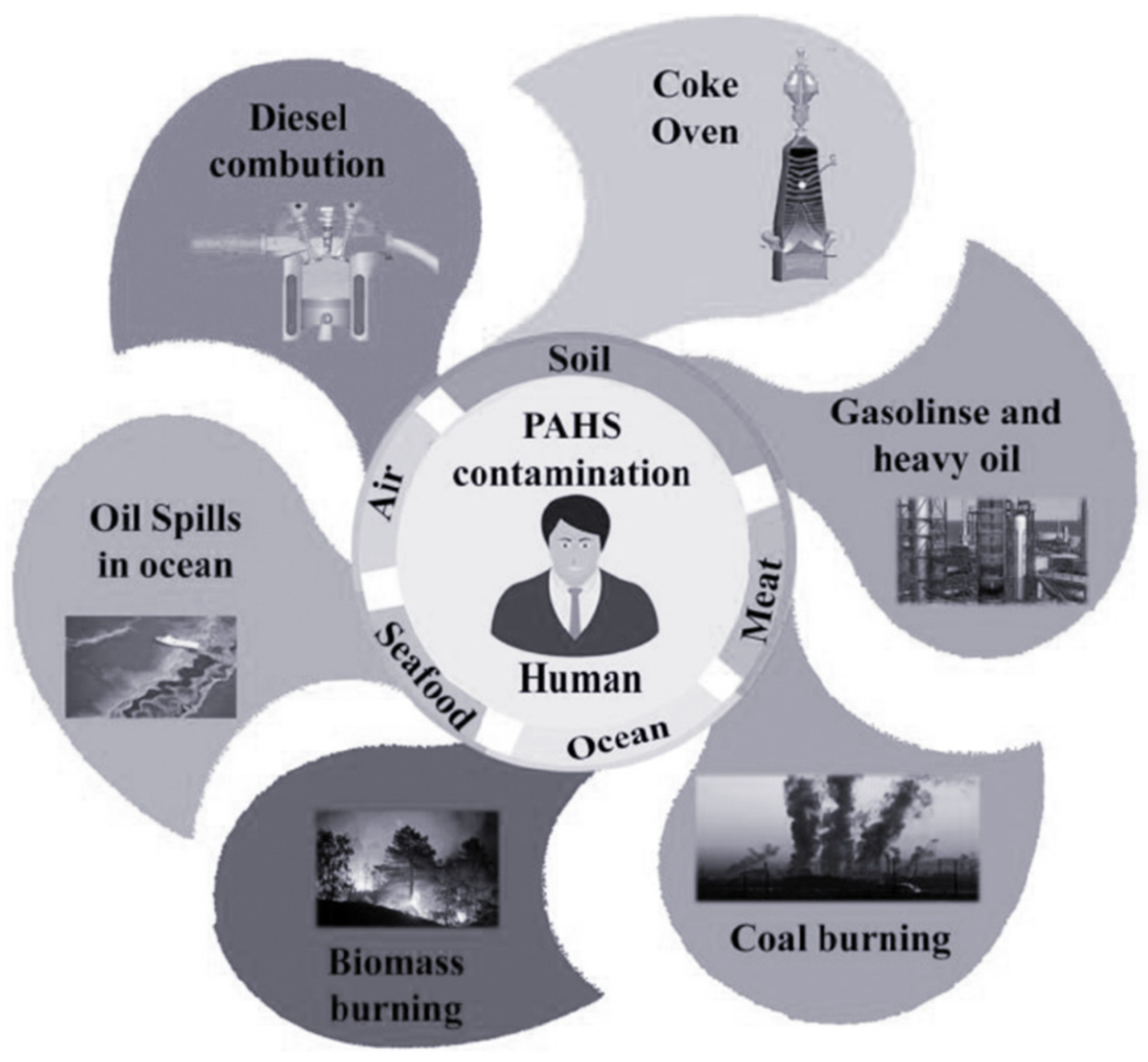
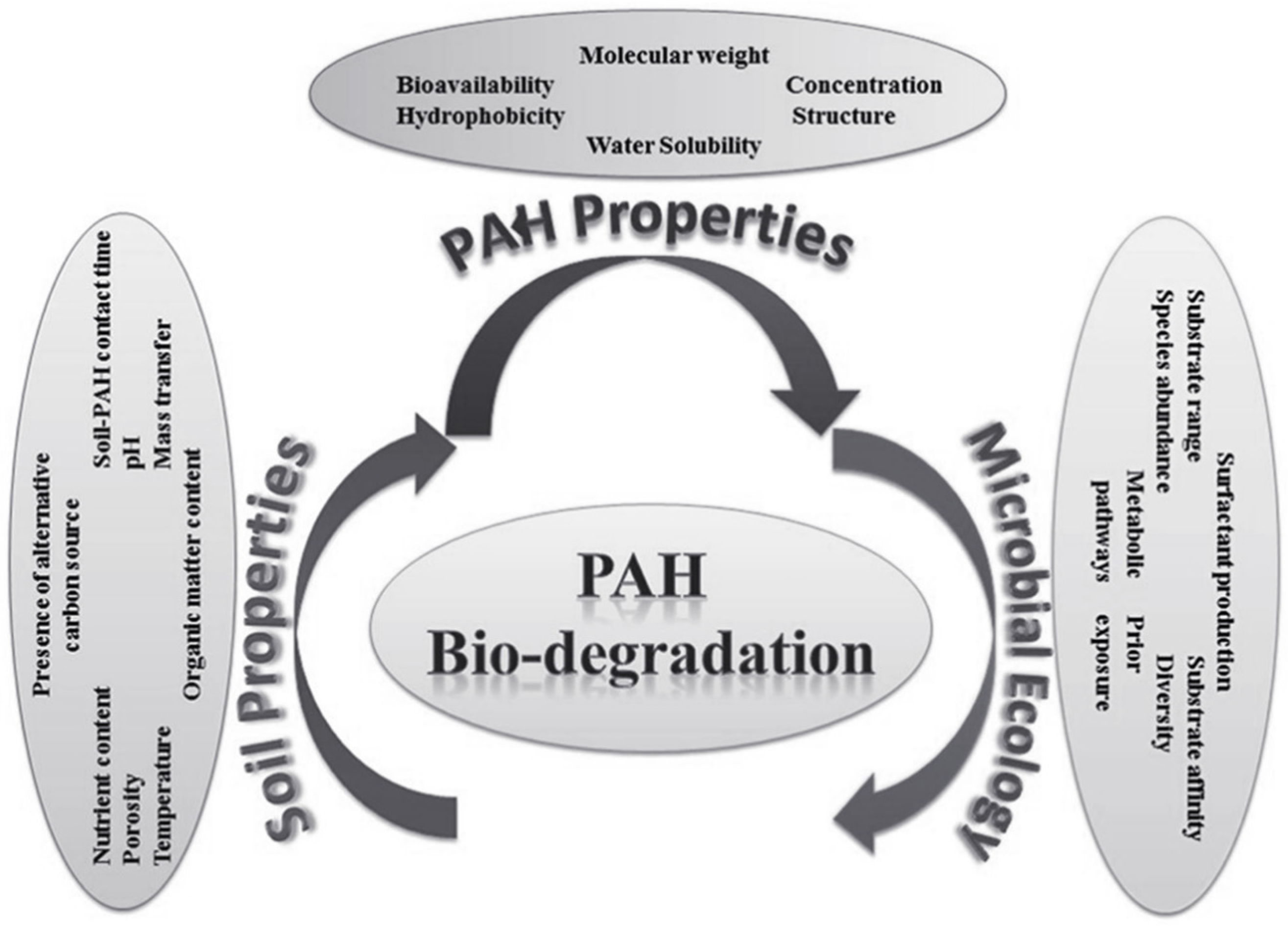
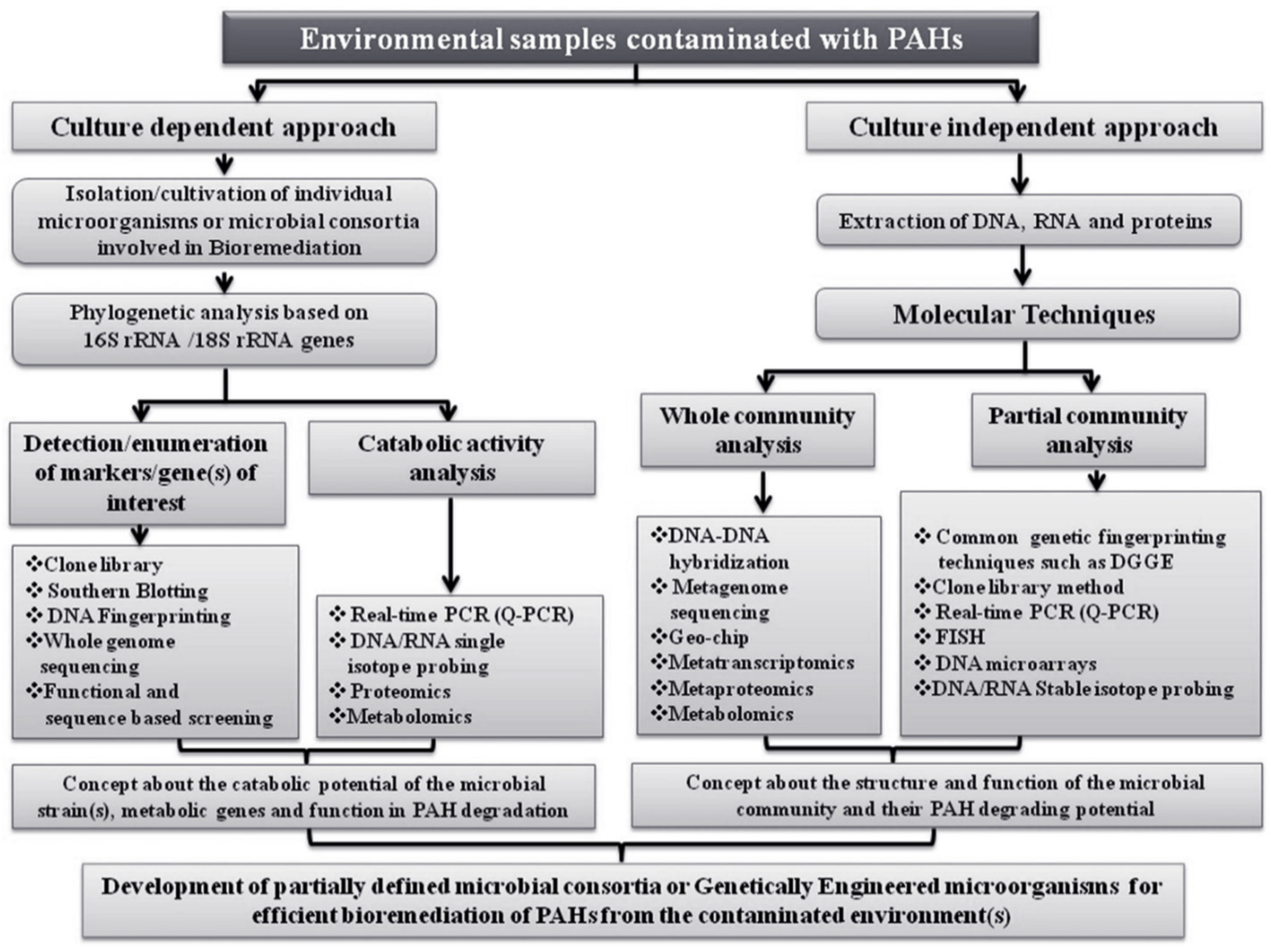


| PAH Name | Molecule Formula | Molecular Weight (g/mole) | Aqueous Solubility (mg/L) | Melting Point (°C) | Boiling Point (°C) | Vapor Pressure (Pa, 25 °C) | Log KOW |
|---|---|---|---|---|---|---|---|
| Naphthalene | C10H8 | 128 | 31.7 | 80.2 | 218 | 11.14 | 3.30 |
| Acenaphthene | C12H10 | 154 | 3.9 | 90–96 | 265–280 | 3.92 | 3.92 |
| Acenaphthylene | C12H8 | 152 | 16.1 | 92–93 | 265–280 | 3.87 | 3.94 |
| Fluorene | C13H10 | 166 | 1.9 | 116–118 | 293–295 | 1.66 | 4.18 |
| Phenanthrene | C14H10 | 178 | 1.15 | 96–101 | 339–340 | 1.06 × 10−1 | 4.46 |
| Pyrene | C16 H10 | 22 | 0.135 | 150–156 | 360–404 | 5.0 × 10−5 | 4.88 |
| Chrysene | C18H12 | 228 | 0.002 | 252–256 | 441–448 | 4.0 × 10−6 | 5.81 |
| Benzo(2)fluoranthene | C20H12 | 252 | 0.0015 | 198–217 | 480–471 | 5.0 × 10−7 | 5.78 |
| Benzofluoranthene | C20H12 | 252 | 0.0008 | 167–168 | 481 | 5.2 × 10−8 | 6.11 |
| Benzopyrene | C20H12 | 252 | 0.00162 | 177–179 | 493–496 | 6.0 × 10−8 | 6.13 |
| Pollutant Removal | An Example of Method | Advantages | Disadvantages |
|---|---|---|---|
| physical | Soil washing | Ease of doing | High cost |
| Chemical | Chemical inactivation | Affordable | Environmental pollution |
| Biologically | Bioremediation/microbial degradation | Environment lover | Preparation of microorganism |
| PAHs Used in the Study | Aerobic or Anaerobic Conditions | Microbial Remediation | Degradation Condition | Degradation (%) | References |
|---|---|---|---|---|---|
| 16 Priority PAHs (with nitrate and sulfate) | Anaerobic | Bacteria | Sediment Anaerobic | 37, 21, and 28% | [77] |
| Phenanthrene, Pyrene, and Benzo(a) pyrene | Aerobic | Bacterial–algal synergy | Soil slurry Aerobic | 100% | [78] |
| Indeno[1,2,3-cd]pyrene | Aerobic | fungi | Liquid medium Aerobic | 91% | [79] |
| Anthracene, Fluorene, Phenanthrene, and Pyrene | Aerobic | Extremophiles | Liquid medium At 50 C Aerobic | 96, 86, 54, and 71% | [80] |
| Pyrene | Anaerobic | facultative bacteria | Pseudomonas sp. JP1 and Klebsiella sp. LZ6 | - | [81] |
| Pyrene | Aerobic | Bacteria | Mycobacterium vanbaalenii PRY-1 and Mycobacterium sp. KMS | - | [81] |
| Anthracene, Acenaphthene, Fluorene, Phenanthrene, Fluoranthene, and Pyrene | Aerobic | Non-ligninolytic fungi | Liquid medium Aerobic | 71, 78, 70, 47, 52, and 62% | [82] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salari, M.; Rahmanian, V.; Hashemi, S.A.; Chiang, W.-H.; Lai, C.W.; Mousavi, S.M.; Gholami, A. Bioremediation Treatment of Polyaromatic Hydrocarbons for Environmental Sustainability. Water 2022, 14, 3980. https://doi.org/10.3390/w14233980
Salari M, Rahmanian V, Hashemi SA, Chiang W-H, Lai CW, Mousavi SM, Gholami A. Bioremediation Treatment of Polyaromatic Hydrocarbons for Environmental Sustainability. Water. 2022; 14(23):3980. https://doi.org/10.3390/w14233980
Chicago/Turabian StyleSalari, Marjan, Vahid Rahmanian, Seyyed Alireza Hashemi, Wei-Hung Chiang, Chin Wei Lai, Seyyed Mojtaba Mousavi, and Ahmad Gholami. 2022. "Bioremediation Treatment of Polyaromatic Hydrocarbons for Environmental Sustainability" Water 14, no. 23: 3980. https://doi.org/10.3390/w14233980










