Enhanced Swine Wastewater Treatment by Constructed Wetland—Microbial Fuel Cell Systems
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Setup and Operating Conditions
2.2. Swine Wastewater and Inoculation
2.3. Chemical Analysis
2.4. Electrochemical and Other Calculations
2.5. Microbial Community Analysis
3. Results and Discussions
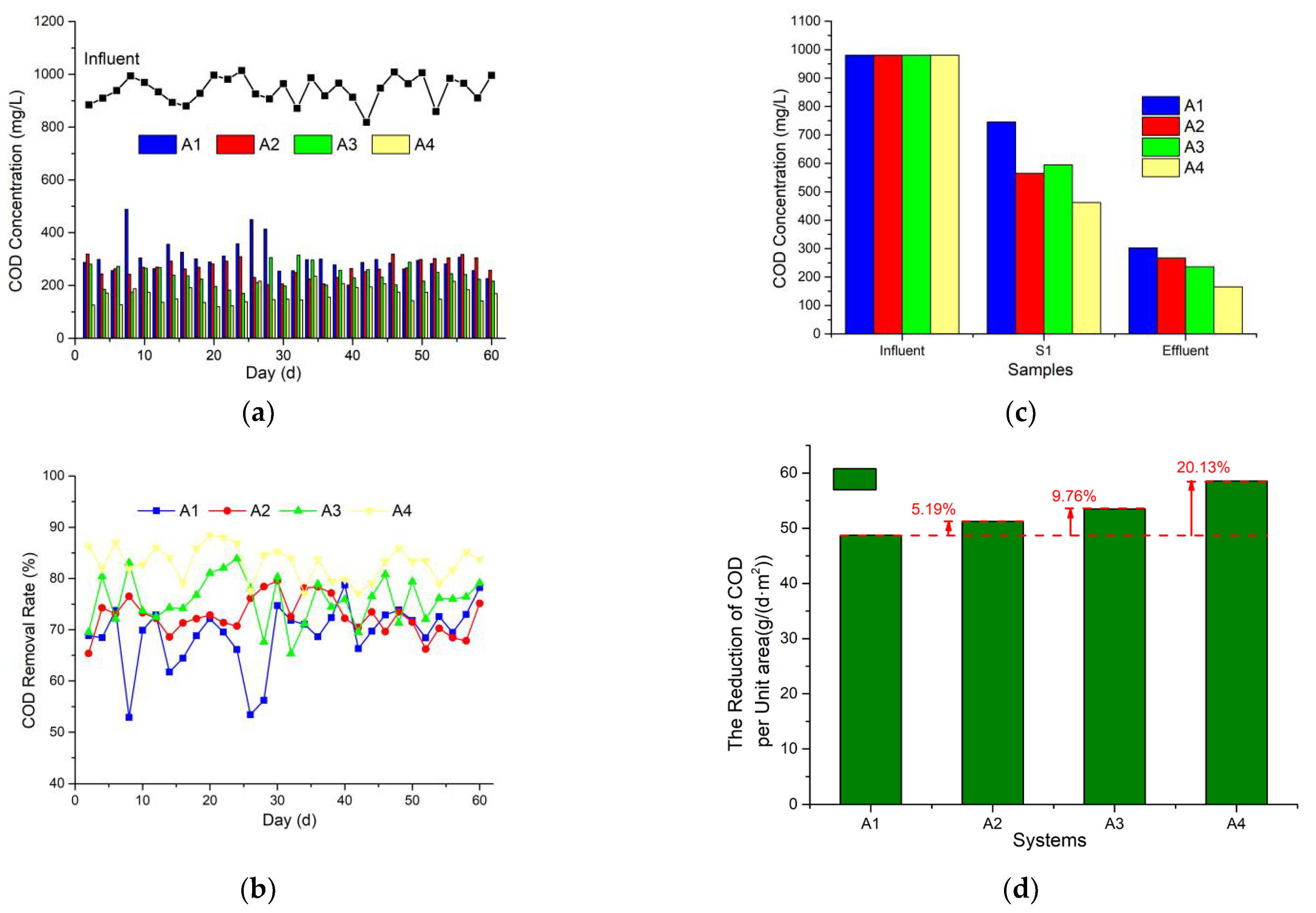
3.1. Organics/COD Removal
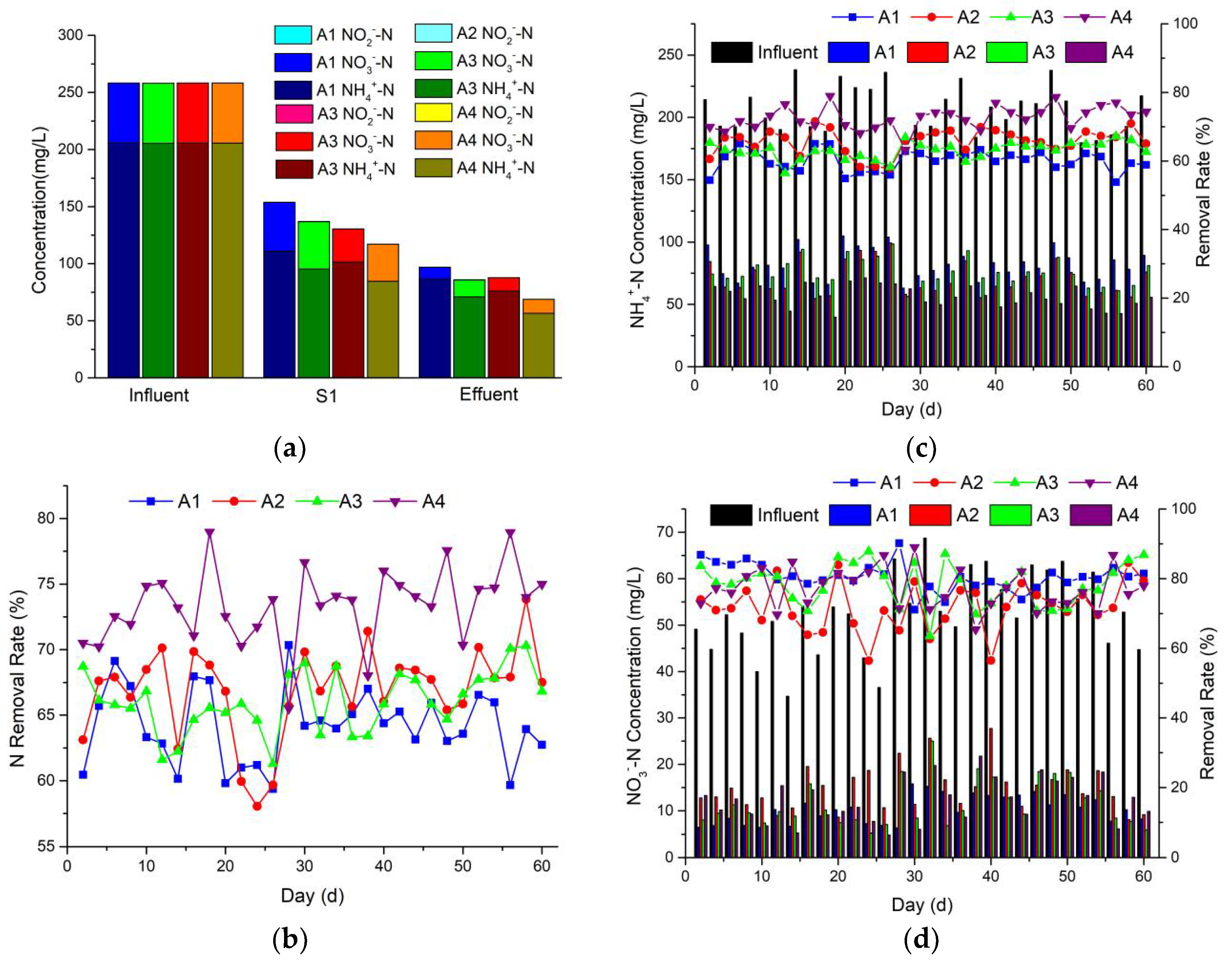
3.2. Nitrogen Removal
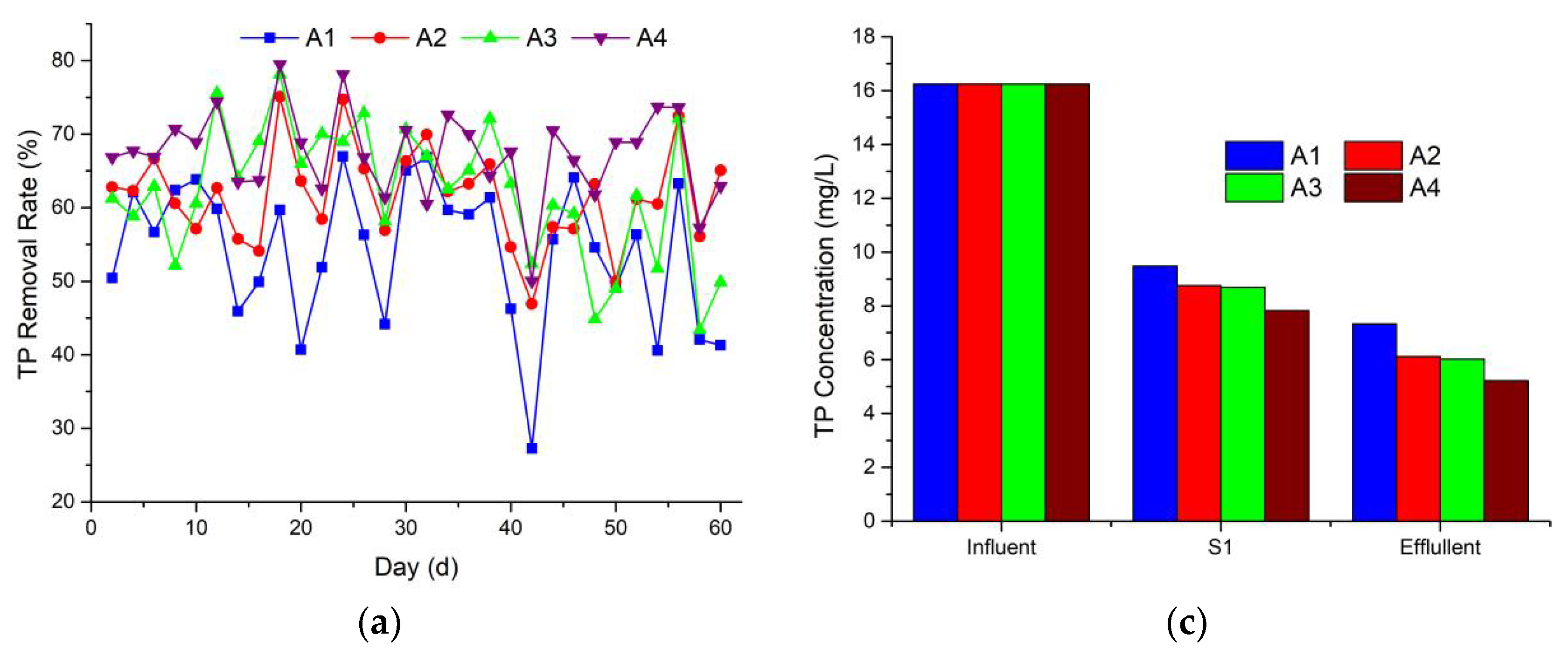
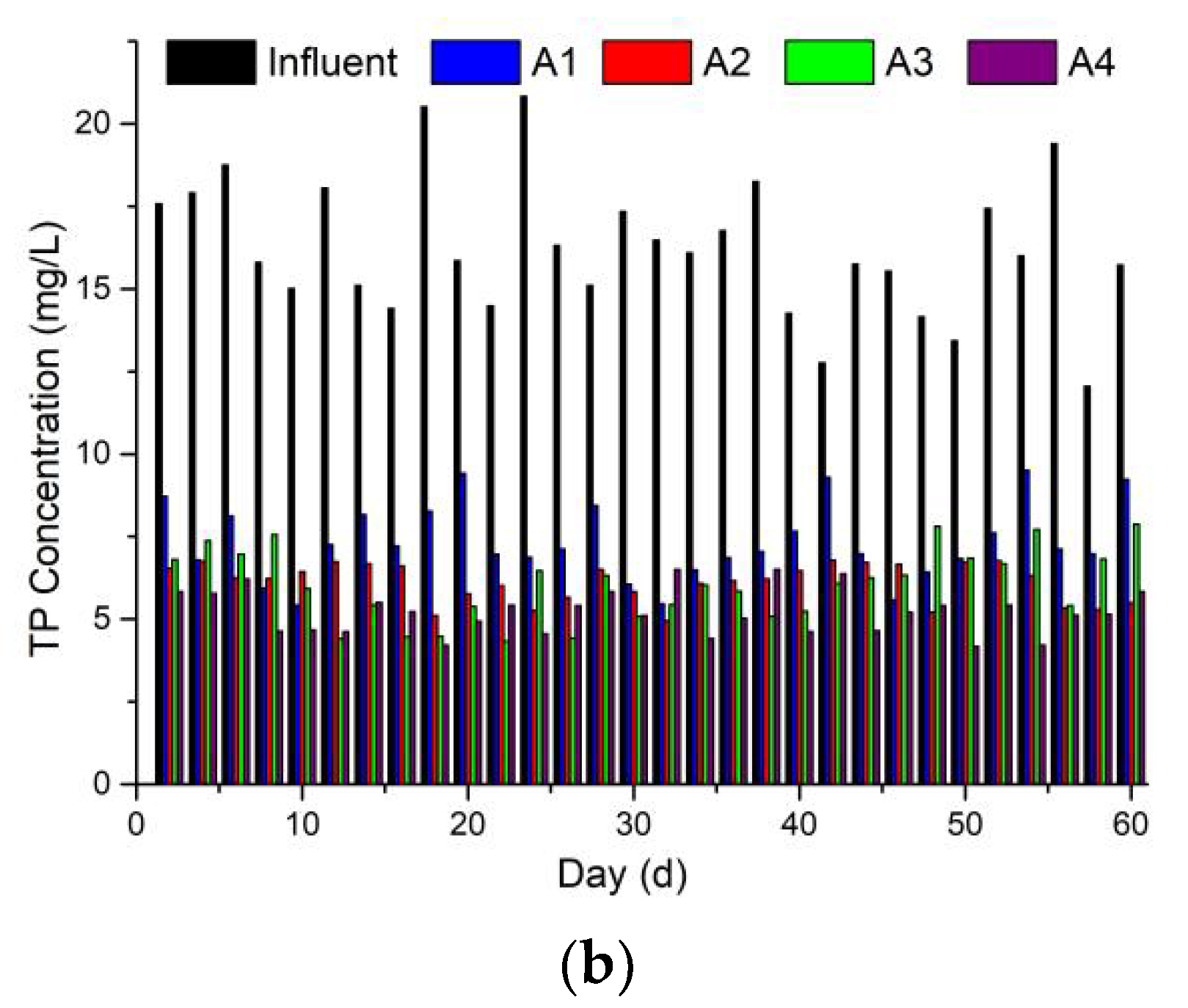
3.3. Phosphorus Removal
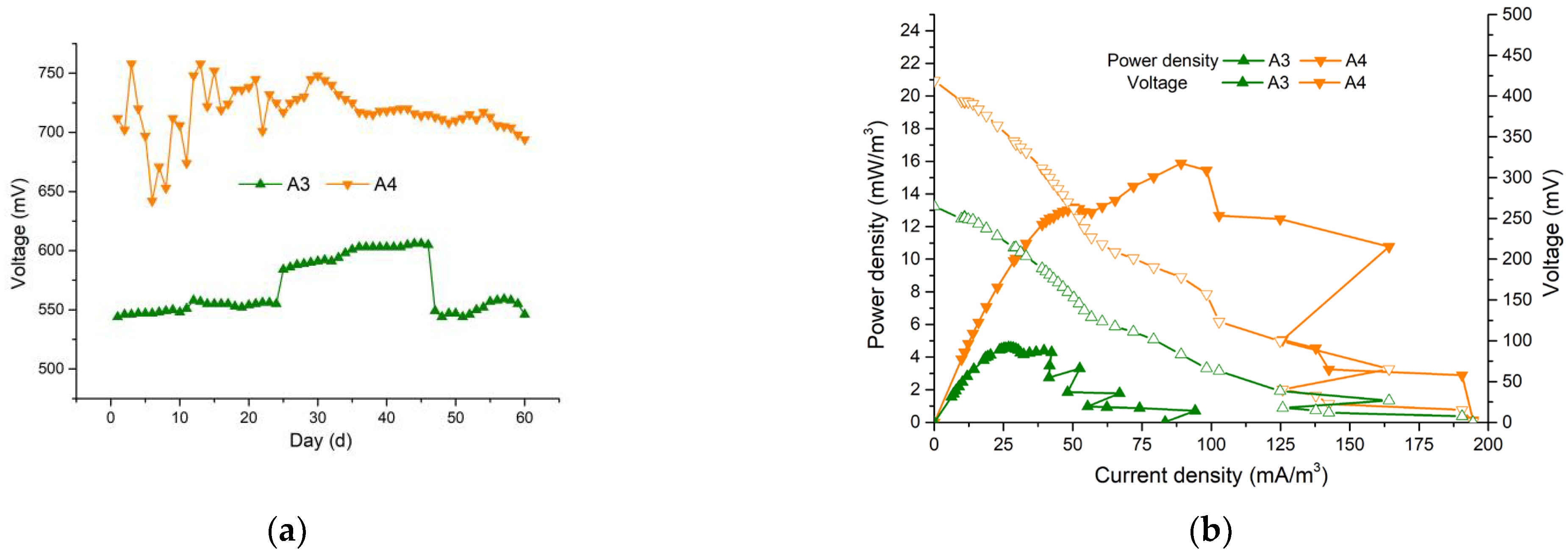
3.4. Electricity Generation
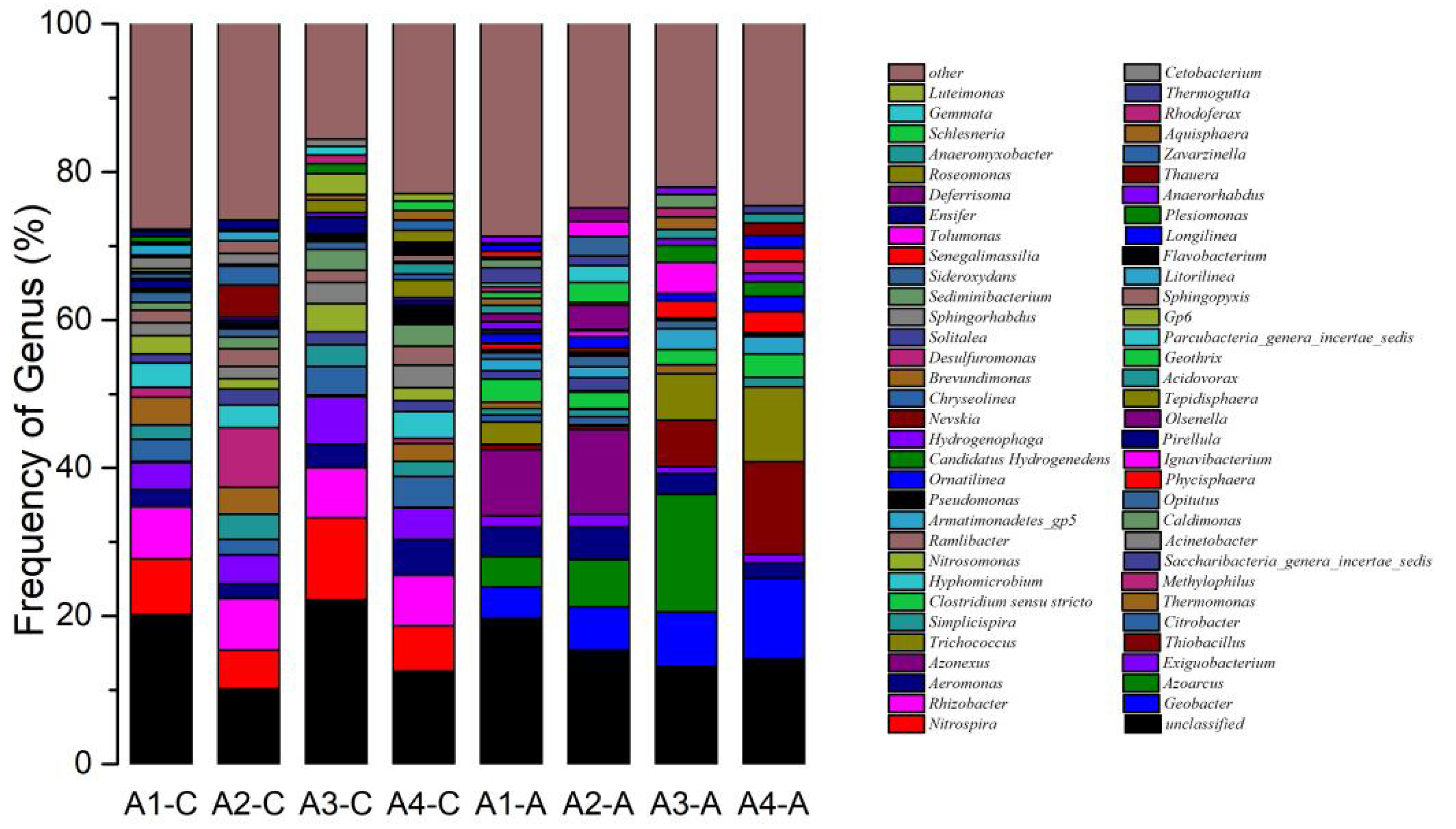
3.5. Biodiversity of Microbial Communities
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Soares, R.B.; Memelli, M.S.; Roque, R.P.; Gonçalves, R.F. Comparative analysis of the energy consumption of different wastewater treatment plants. Int. J. Archit. Arts Appl. 2017, 3, 79. [Google Scholar]
- Heidrich, E.S.; Curtis, T.P.; Dolfing, J. Determination of the internal chemical energy of wastewater. Environ. Sci. Technol. 2011, 45, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; He, Z. Integrating membrane filtration into bioelectrochemical systems as next generation energy-efficient wastewater treatment technologies for water reclamation: A review. Bioresour. Technol. 2015, 195, 202–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, J.; Yang, C.; Zeng, G. Treatment of swine wastewater using chemically modified zeolite and bioflocculant from activated sludge. Bioresour. Technol. 2013, 143, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Hu, Z.; Zhang, Y.; Zhuang, L.; Zhang, J.; Li, J.; Hu, H. Removal Processes of Carbamazepine in Constructed Wetlands Treating Secondary Effluent: A Review. Water 2018, 10, 1351. [Google Scholar] [CrossRef]
- Santoro, C.; Walter, X.A.; Soavi, F.; Greenman, J.; Ieropoulos, I. Self-stratified and self-powered micro-supercapacitor integrated into a microbial fuel cell operating in human urine. Electrochim. Acta. 2019, 307, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Goto, Y.; Yoshida, N. Scaling up Microbial Fuel Cells for Treating Swine Wastewater. Water 2019, 11, 1803. [Google Scholar] [CrossRef] [Green Version]
- Yadav, R.K.; Chiranjeevi, P.; Patil, S.A. Integrated drip hydroponics-microbial fuel cell system for wastewater treatment and resource recovery. Bioresour. Technol. Rep. 2020, 9, 100392. [Google Scholar] [CrossRef]
- Corbella, C.; Garfi, M.; Puigagut, J. Vertical redox profiles in treatment wetlands as function of hydraulic regime and macrophytes presence: Surveying the optimal scenario for microbial fuel cell implementation. Sci. Total Environ. 2014, 470, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; Verstraete, W. Microbial fuel cells: Novel biotechnology for energy generation. Trends Biotechnol. 2005, 23, 291–298. [Google Scholar] [CrossRef]
- Schroder, U. Anodic electron transfer mechanisms in microbial fuel cells and their energy efficiency. Phys. Chem. Chem. Phys. 2007, 9, 2619–2629. [Google Scholar] [CrossRef]
- Srivastava, P.; Abbassi, R.; Yadav, A.K.; Garaniya, V.; Asadnia, M. A review on the contribution of electron flow in electroactive wetlands: Electricity generation and enhanced wastewater treatment. Chemosphere. 2020, 254, 126926. [Google Scholar] [CrossRef] [PubMed]
- Saeed, T.; Miah, M.J. Organic matter and nutrient removal in tidal flow-based microbial fuel cell constructed wetlands: Media and flood-dry period ratio. Chem. Eng. J. 2021, 411, 128507. [Google Scholar] [CrossRef]
- Liu, F.; Sun, L.; Wan, J.; Tang, A.; Deng, M.; Wu, R. Organic matter and ammonia removal by a novel integrated process of constructed wetland and microbial fuel cells. RSC Adv. 2019, 9, 5384–5393. [Google Scholar] [CrossRef] [Green Version]
- Doherty, L.; Zhao, Y.; Zhao, X.; Wang, W. Nutrient and organics removal from swine slurry with simultaneous electricity generation in an alum sludge-based constructed wetland incorporating microbial fuel cell technology. Chem. Eng. J. 2015, 266, 74–81. [Google Scholar] [CrossRef]
- Camacho, J.V.; Romero, L.R.; Marchante, C.M.F. The salinity effects on the performance of a constructed wetland microbial fuel cell. Ecolog. Eng. 2017, 107, 1–7. [Google Scholar] [CrossRef]
- Araneda, I.; Tapia, N.F.; Allende, K.L.; Vargas, I.T. Constructed Wetland-Microbial Fuel Cells for Sustainable Greywater Treatment. Water 2018, 10, 940. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Sun, L.; Wan, J.; Shen, L.; Yu, Y.; Hu, L.; Zhou, Y. Performance of Different Macrophytes in the Decontamination of and Electricity. J. Environ. Sci. 2020, 89, 252–263. [Google Scholar] [CrossRef]
- Patel, D.; Bapodra, S.L.; Madamwar, D.; Desai, C. Electroactive bacterial community augmentation enhances the performance of a pilot scale constructed wetland microbial fuel cell for treatment of textile dye wastewater. Bioresour. Technol. 2021, 332, 125088. [Google Scholar] [CrossRef]
- Wang, J.; Song, X.; Wang, Y.; Bai, J.; Li, M.; Dong, G.; Lin, F.; Lv, Y.; Yan, D. Bioenergy generation and rhizodegradation as affected by microbial community distribution in a coupled constructed wetland microbial fuel cell system associated with three macrophytes. Sci. Total Environ. 2017, 607, 53–62. [Google Scholar] [CrossRef]
- Srivastava, P.; Yadav, A.K.; Garaniyai, K.; Lewis, T.; Abbassi, R.; Khan, S.J. Electrode dependent anaerobic ammonium oxidation in microbial fuel cell integrated hybrid constructed wetlands: A new process. Sci. Total Environ. 2020, 698, 134248. [Google Scholar] [CrossRef] [PubMed]
- Oon, Y.L.; Ong, S.A.; Ho, L.N.; Wong, Y.S.; Dahalan, F.A.; Oon, Y.S. Synergistic effect of up-flow constructed wetland and microbial fuel cell for simultaneous wastewater treatment and energy recovery. Bioresour. Technol. 2016, 203, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Yulistyorini, A.; Puspasari, A.K.; Mujiyono; Sari, A. Removal of BOD and TSS of Student Dormitory Greywater using Vertical Sub-Surface Flow Constructed Wetland of Ipomoea Aquatica. Mater. Sci. Eng. 2019, 515, 012056. [Google Scholar] [CrossRef]
- Ibekwe, A.M.; Lyon, S.R.; Leddy, M.; Jacobson-Meyers, M. Impact of plant density and microbial composition on water quality from a free water surface constructed wetland. J. Appl. Microbiol. 2007, 102, 921–936. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Srivastava, P.; Patil, S.A.; Yadav, A.K. A comprehensive review on emerging constructed wetland coupled microbial fuel cell technology: Potential applications and challenges. Bioresour. Technol. 2021, 320, 124376. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, Y.; Tang, C.; Xu, L.; Morgan, D.; Liu, R. Role of macrophyte species in constructed wetland-microbial fuel cell for simultaneous wastewater treatment and bioenergy generation. Chem. Eng. 2020, 392, 123708. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, D.; Xiao, E.; Xu, D.; Xu, P.; Zhang, X.; Zhou, Q.; He, F.; Wu, Z. Relationship between electrogenic performance and physiological change of four wetland plants in constructed wetland-microbial fuel cells during non-growing seasons. Int. J. Environ. Sci. 2018, 70, 54–62. [Google Scholar] [CrossRef]
- Domenico, E.G.D.; Petroni, G.; Mancini, D.; Geri, A.; Palma, L.D.; Ascenzioni1, F. Development of Electroactive and Anaerobic Ammonium-Oxidizing (Anammox) Biofilms from Digestate in Microbial Fuel Cells. BioMed Res. Intern. 2015, 2015, 351014. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.; Lv, T.; Lu, Q.; Ajmal, Z.; Dong, R. Treatment of anaerobic digestate supernatant in microbial fuel cell coupled constructed wetlands: Evaluation of nitrogen removal, electricity generation, and bacterial community response. Sci. Total Environ. 2017, 580, 339–346. [Google Scholar] [CrossRef]
- Muhammad, A.; Satoshi, O. Anammox-based technologies for nitrogen removal: Advances in process start-up and remaining issues. Chemosphere 2015, 141, 144–153. [Google Scholar]
- Lotti, T.; Kleerebezem, R.; Lubello, C.; Loosdrecht, M.C.M. Physiological and kinetic characterization of a suspended cell anammox culture. Water Res. 2014, 60, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lotti, T.; Kleerebezern, R.; Abelleira-Pereira, J.M.; Abbas, B.; van Loosdrecht, M.C.M. Faster through training: The anammox case. Water Res. 2009, 81, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Ni, B.-J.; Ruscalleda, M.; Smets, B.F. Evaluation on the microbial interactions of anaerobic ammonium oxidizers and heterotrophs in Anammox biofilm. Water Res. 2012, 46, 4645–4652. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.L.; Jiang, Y.; Xie, J.X.; Cao, J.; Zhang, Q.; Mabruk, A.; Chen, C. Bamboo charcoal addition enhanced the nitrogen removal of anammox granular sludge with COD: Performance, physicochemical characteristics and microbial community. J. of Environ. Sci. 2022, 115, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Hou, D.; Zhang, S.; Cao, X.; Wang, H.; Li, X. Aerobic Denitrification Is Enhanced Using Biocathode of SMFC in Low-Organic Matter Wastewater. Water 2021, 13, 3512. [Google Scholar] [CrossRef]
- Wang, Q.; Lv, R.; Rene, E.R.; Qi, X.; Hao, Q.; Du, Y.; Zhao, C.; Xu, F.; Kong, Q. Characterization of microbial community and resistance gene (CzcA) shifts in upflow constructed wetlands-microbial fuel cell treating Zn (II) contaminated wastewater. Bioresour. Technol. 2020, 302, 122867. [Google Scholar] [CrossRef]
- Zhao, Y.; Collum, S.; Phelan, M.; Goodbody, T.; Doherty, L.; Hu, Y. Preliminary investigation of constructed wetland incorporating microbial fuel cell: Batch and continuous flow trials. Chem. Eng. J. 2013, 229, 364–370. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Shang, D.; Zou, Y.; Du, Y.; Wang, Q.; Xu, F.; Ren, L.; Kong, Q. Changes in electricity production and microbial community evolution in constructed wetland-microbial fuel cell exposed to wastewater containing Pb(II). Sci. Total Environ. 2020, 732, 139127. [Google Scholar] [CrossRef]
- Xu, F.; Cao, F.; Kong, Q.; Zhou, L.; Yuan, Q.; Zhu, Y.; Wang, Q.; Du, Y.; Wang, Z. Electricity production and evolution of microbial community in the constructed wetland-microbial fuel cell. Chem. Eng. J. 2018, 339, 479–486. [Google Scholar] [CrossRef]
- Zhao, W.; Bi, X.; Peng, Y.; Bai, M. Research advances of the phosphorus-accumulating organisms of Candidatus Accumulibacter, Dechloromonas and Tetrasphaera: Metabolic mechanisms, applications and influencing factors. Chemosphere 2022, 307, 135675. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, M.; Zhou, S. Conversion of nitrogen and carbon in enriched paddy soil by denitrification coupled with anammox in a bioelectrochemical system. J. Environ. Sci. 2022, 111, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Song, X.; Wang, Y.; Abayneh, B.; Ding, Y.; Yan, D.; Bai, J. Microbial community structure of different electrode materials in constructed wetland incorporating microbial fuel cell. Bioresour. Technol. 2016, 221, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Singh, L.; Zularisam, A.W. Exoelectrogens Recent advances in molecule drivers involved in extracellular electron transfer and strategies used to improve it for microbial fuel cell applications. Renew. Sustain. Energy Rev. 2016, 56, 1322–1336. [Google Scholar] [CrossRef] [Green Version]
- Gemma, R.; Mccarthy, K.D.; Teena, M.; Nicoll, J.S.; Tuominen, M.T.; Lovley, D.R. Extracellular electron transfer via microbial nanowires. Nature 2005, 435, 1098–1101. [Google Scholar]
- Sudirjo, E.; Buisman, C.J.N.; Strik, D.P.B.T.B. Activated Carbon Mixed with Marine Sediment is Suitable as Bioanode Material for Spartina anglica Sediment/Plant Microbial Fuel Cell: Plant Growth, Electricity Generation, and Spatial Microbial Community Diversity. Water 2019, 11, 1810. [Google Scholar] [CrossRef] [Green Version]
- Pham, C.A.; Jung, S.J.; Phung, N.T.; Lee, J.; Chang, I.S.; Kim, B.H.; Yi, H.; Chun, J. A novel electrochemically active and Fe(III) reducing bacterium Phylogenetically related to Aeromonas hydrophila isolated from a microbial fuel cell. FEMS Microbiol. Lett. 2003, 223, 129–134. [Google Scholar] [CrossRef]
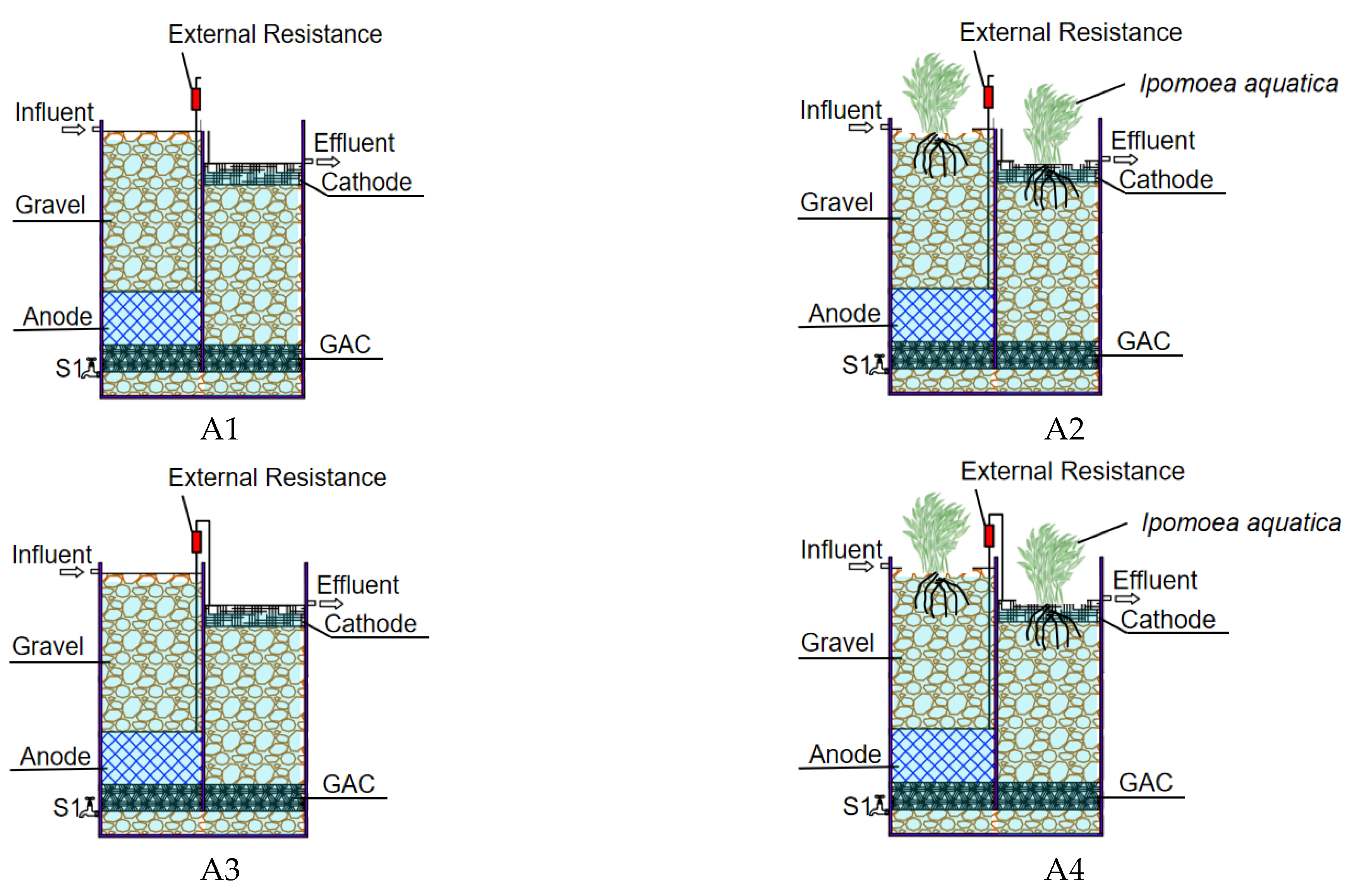
| Systems | No Plants | Planting Ipomoea aquatica |
|---|---|---|
| The open circuit | A1 | A2 |
| The closed circuit | A3 | A4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Liu, F.; Lin, Y.; Sun, L.; Guo, X.; Yang, S.; He, J. Enhanced Swine Wastewater Treatment by Constructed Wetland—Microbial Fuel Cell Systems. Water 2022, 14, 3930. https://doi.org/10.3390/w14233930
Zhang Y, Liu F, Lin Y, Sun L, Guo X, Yang S, He J. Enhanced Swine Wastewater Treatment by Constructed Wetland—Microbial Fuel Cell Systems. Water. 2022; 14(23):3930. https://doi.org/10.3390/w14233930
Chicago/Turabian StyleZhang, Yun, Feng Liu, Yidong Lin, Lei Sun, Xinru Guo, Shuai Yang, and Jinlong He. 2022. "Enhanced Swine Wastewater Treatment by Constructed Wetland—Microbial Fuel Cell Systems" Water 14, no. 23: 3930. https://doi.org/10.3390/w14233930




