Removal of Phosphorus by Ferric Ion-Rich Solutions Prepared Using Various Fe(III)-Containing Minerals
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Minerals
2.2. T-P Removal via Chemical Induction Using Cation Solutions
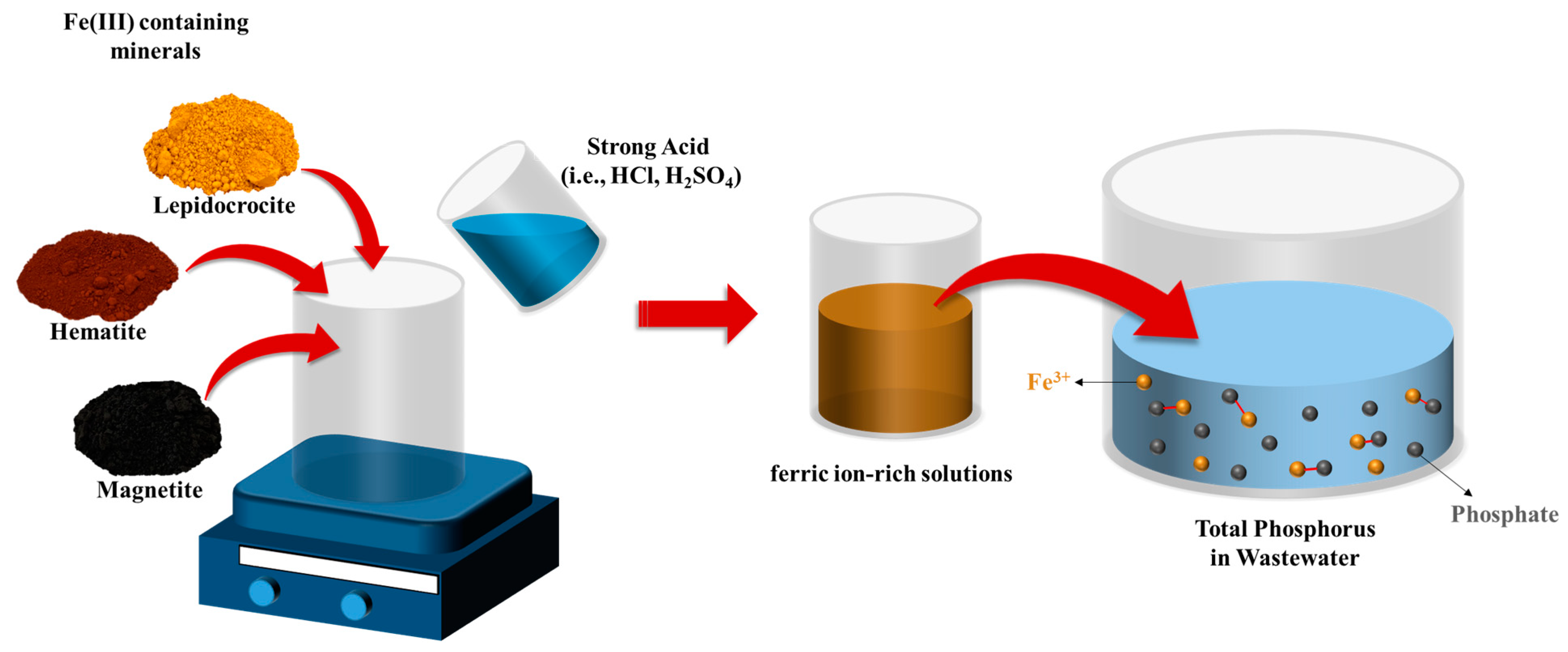
2.3. T-P Removal by Solutions Containing Dissolved Fe3+ That Were Prepared Using Fe(III)-Containing Minerals
2.4. Analytical Methods
3. Results and Discussion
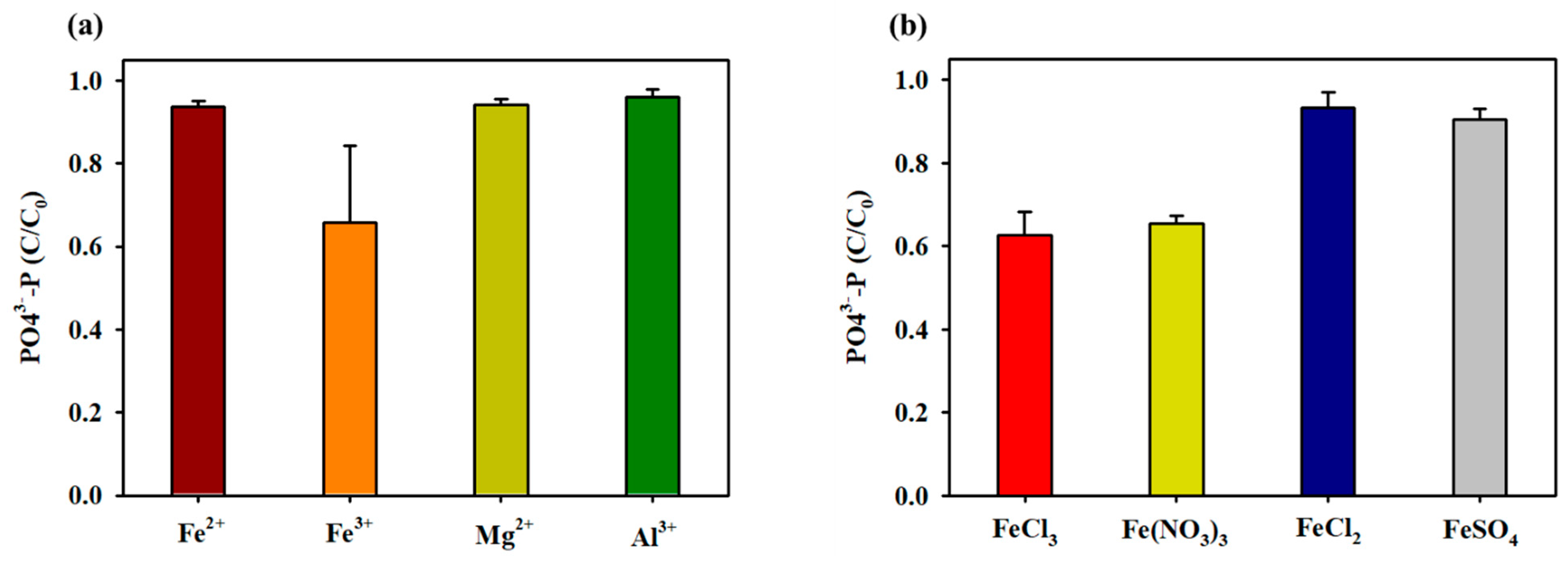
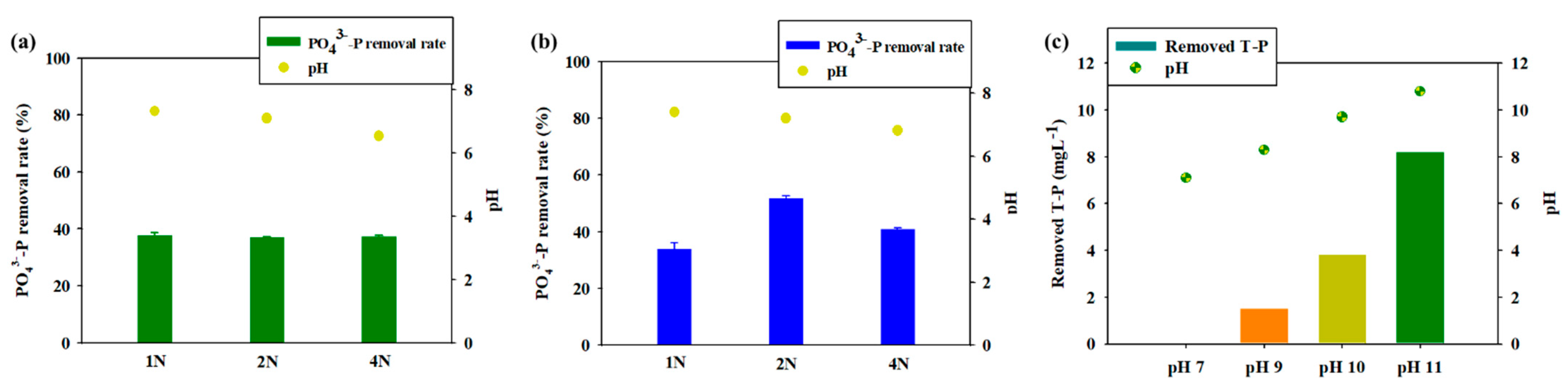
3.1. Removal of T-P via Chemical Induction Using Cation Solutions
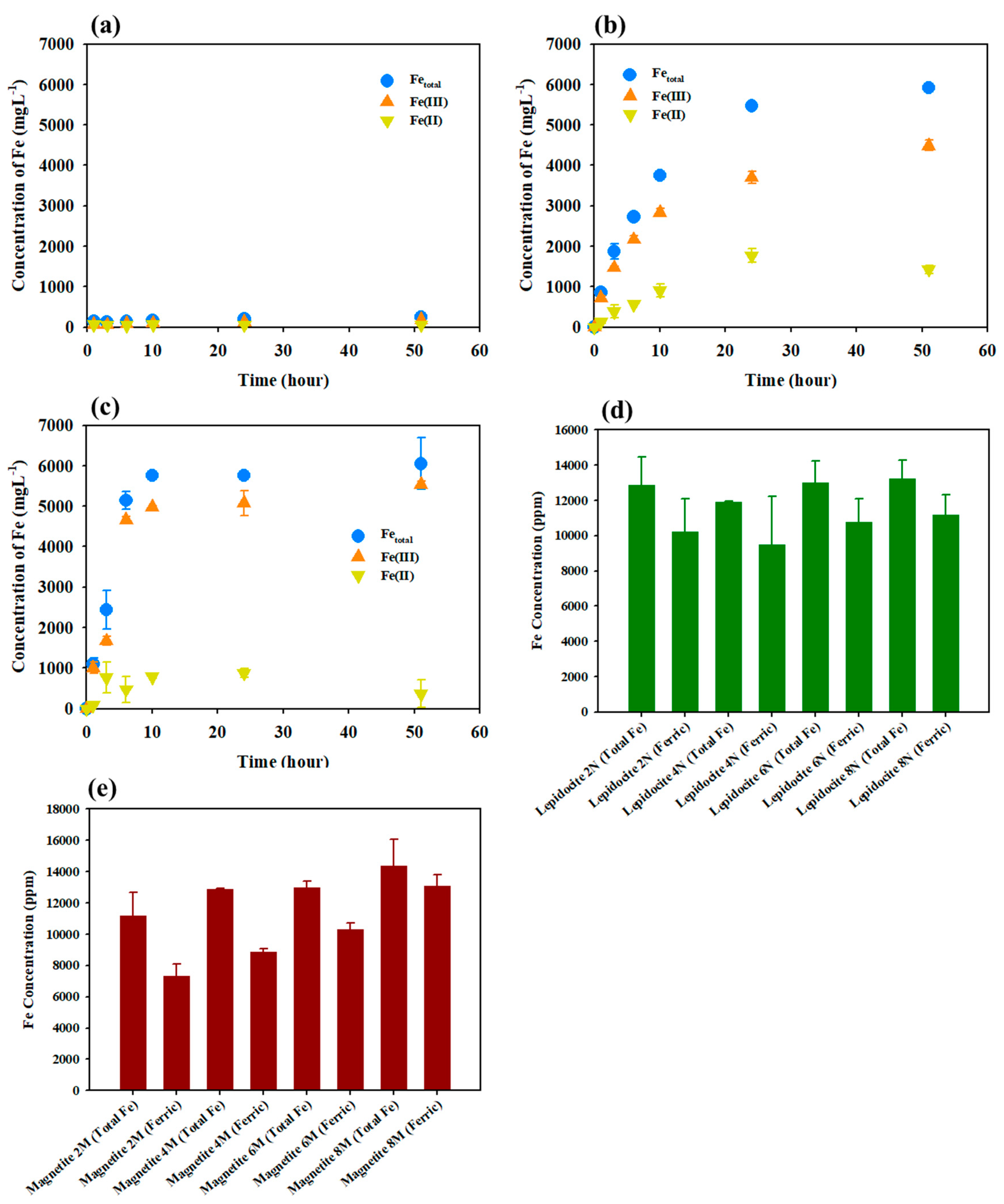
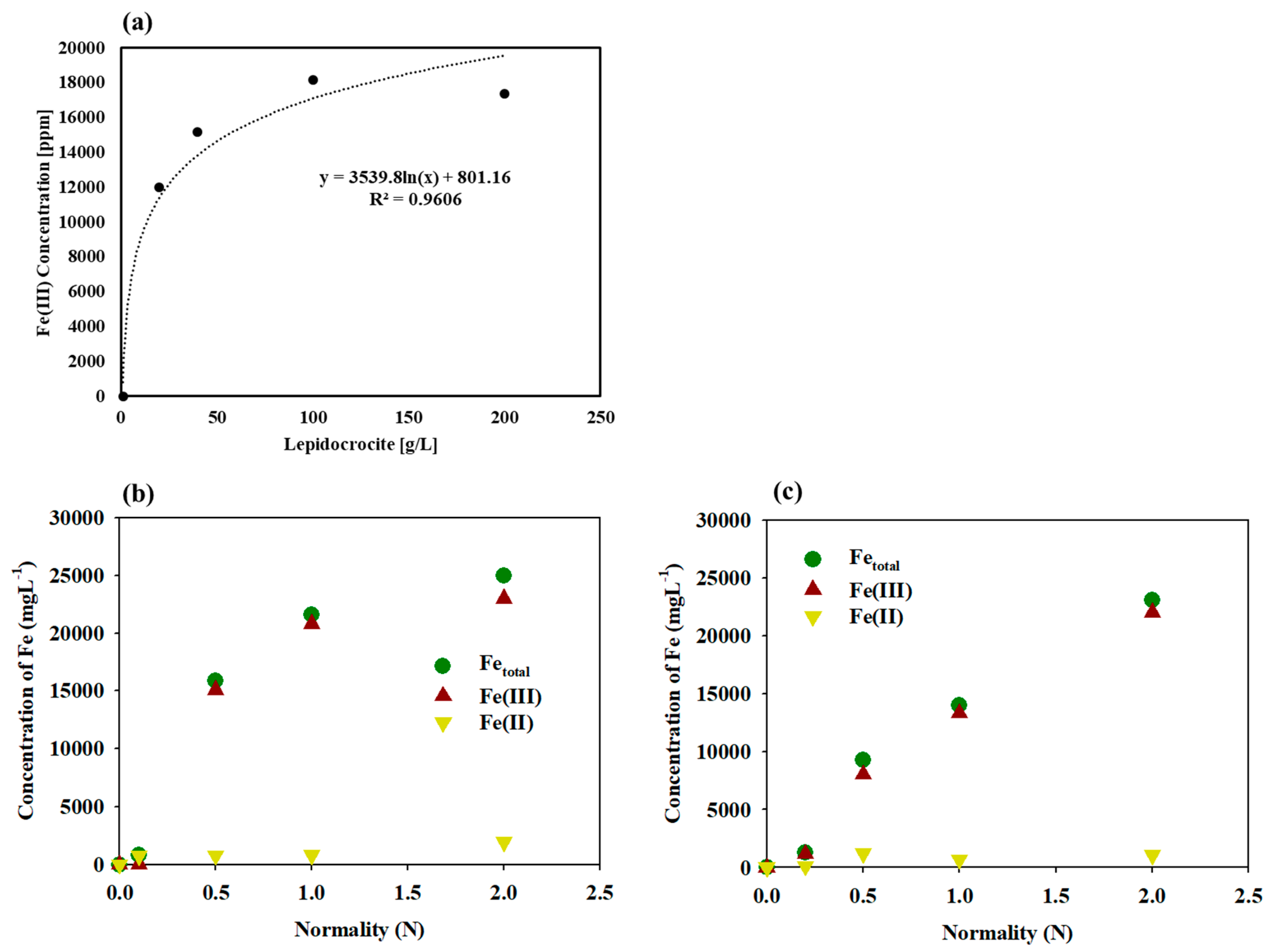
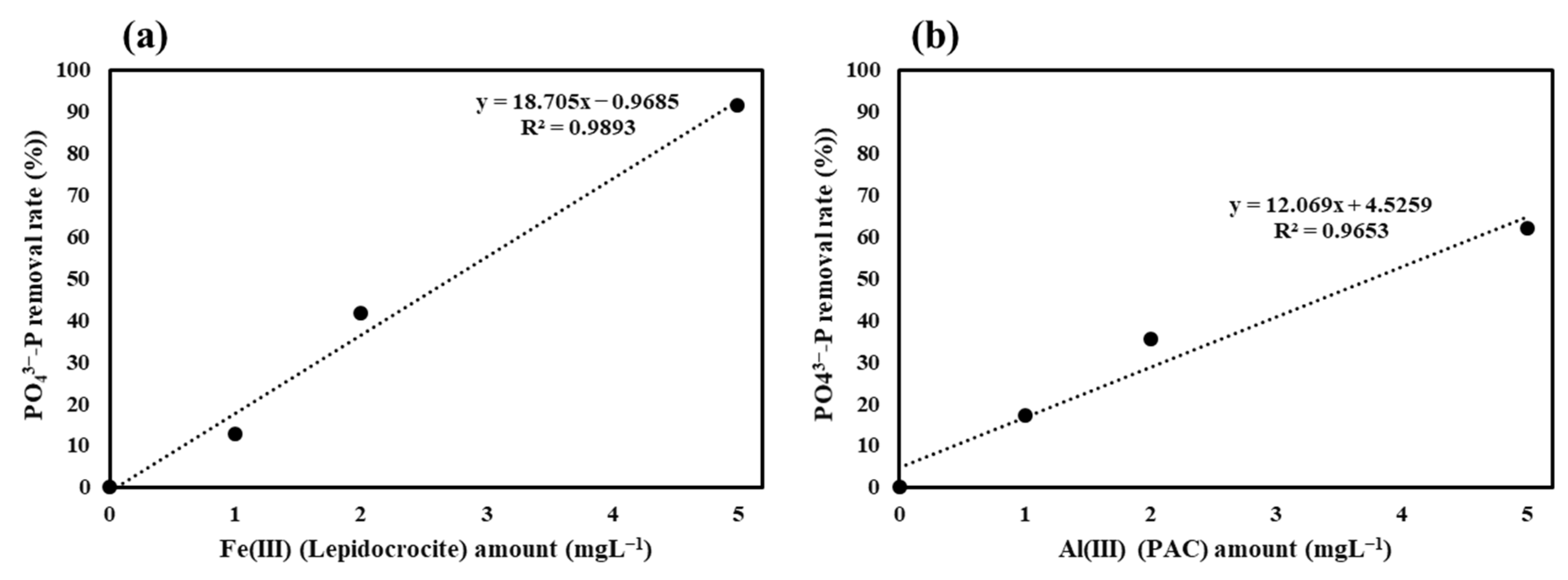
3.2. Monitoring Fe(III) Dissolution
3.3. Removal of T-P by L-Fe Dissolved Solution
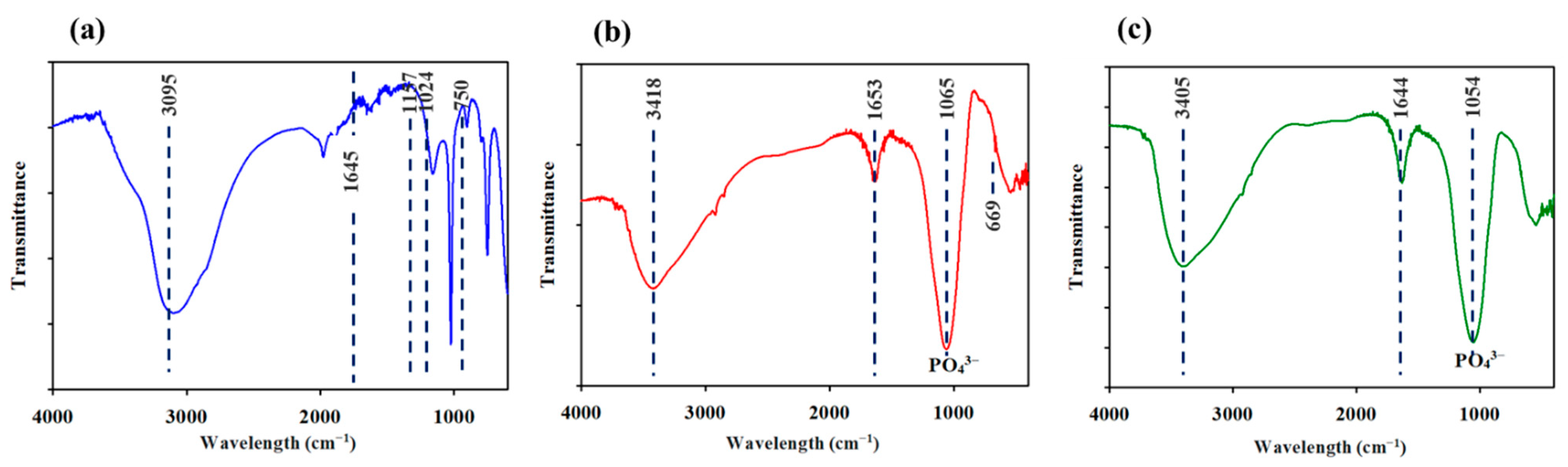
3.4. Analysis of the Precipitates
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kroiss, H.; Rechberger, H.; Egle, L. Phosphorus in water quality and waste management. Integr. Waste Manag. 2011, 2, 18482. [Google Scholar] [CrossRef] [Green Version]
- Holtan, H.; Kamp-Nielsen, L.; Stuanes, A.O. Phosphorus in soil, water and sediment: An overview. Phosphorus Freshw. Ecosyst. 1988, 34, 19–34. [Google Scholar] [CrossRef]
- Bouwman, L.; Goldewijk, K.K.; Van Der Hoek, K.W.; Beusen, A.H.W.; van Vuuren, D.P.; Willems, J.; Rufino, M.C.; Stehfest, E. Exploring global changes in nitrogen and phosphorus cycles in agriculture induced by livestock production over the 1900–2050 period. Proc. Natl. Acad. Sci. USA 2013, 110, 20882–20887. [Google Scholar] [CrossRef] [Green Version]
- Jarvie, H.P.; Neal, C.; Withers, P.J. Sewage-effluent phosphorus: A greater risk to river eutrophication than agricultural phosphorus? Sci. Total Environ. 2006, 360, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Kwak, J.; Kim, S.; Son, C.; Lee, Y.-G.; Kim, J.; Bae, S.; Park, Y.; Lee, S.-H.; Chon, K. Highly selective recovery of phosphate ions using a novel carbonaceous adsorbent synthesized via co-pyrolysis of spent coffee grounds and steel slags: A potential phosphatic fertilizer. Chem. Eng. J. 2023, 451, 138978. [Google Scholar] [CrossRef]
- Wang, H.; Xiao, K.; Yang, J.; Yu, Z.; Yu, W.; Xu, Q.; Wu, Q.; Liang, S.; Hu, J.; Hou, H.; et al. Phosphorus recovery from the liquid phase of anaerobic digestate using biochar derived from iron-rich sludge: A potential phosphorus fertilizer. Water Res. 2020, 174, 115629. [Google Scholar] [CrossRef]
- Correll, D.L. The role of phosphorus in the eutrophication of receiving waters: A review. J. Environ. Qual. 1998, 27, 261–266. [Google Scholar] [CrossRef] [Green Version]
- Mkilima, T.; Bazarbayeva, T.; Assel, K.; Nurmukhanbetova, N.N.; Ostretsova, I.B.; Khamitova, A.S.; Makhanova, S.; Sergazina, S. Pore size in the removal of phosphorus and nitrogen from poultry slaughterhouse wastewater using polymeric nanofiltration membranes. Water 2022, 14, 2929. [Google Scholar] [CrossRef]
- Bashar, R.; Gungor, K.; Karthikeyan, K.; Barak, P. Cost effectiveness of phosphorus removal processes in municipal wastewater treatment. Chemosphere 2018, 197, 280–290. [Google Scholar] [CrossRef]
- Peng, Y.; He, S.; Wu, F. Biochemical processes mediated by iron-based materials in water treatement: Enhancing nitrogen and phosphorus removal in low C/N ratio wastewater. Sci. Total Environ. 2021, 775, 145137. [Google Scholar] [CrossRef]
- Jia, Y.; Zeng, W.; Fan, Z.; Meng, Q.; Liu, H.; Peng, Y. An effective titanium salt dosing strategy for phosphorus removal from wastewater: Synergistic enhancement of chemical and biological treatment. Sci. Total Environ. 2022, 842, 156960. [Google Scholar] [CrossRef]
- Nguyen, T.A.H.; Ngo, H.H.; Guo, W.S.; Nguyen, T.T.; Vu, N.D.; Soda, S.; Nguyen, T.H.H.; Nguyen, M.K.; Tran, T.V.H.; Dang, T.T.; et al. White hard clam (Meretrix lyrata) shells as novel filter media to augment the phosphorus removal from wastewater. Sci. Total Environ. 2020, 741, 140483. [Google Scholar] [CrossRef]
- Wang, H.; Dai, J.; Chen, H.; Wang, F.; Zhu, Y.; Liu, J.; Zhou, B.; Yuan, R. Adsorption of phosphate by Mg/Fe-doped wheat straw biochars optimized using response surface methodology: Mechanisms and application in domestic sewage. Environ. Eng. Res. 2022, 28, 210602. [Google Scholar] [CrossRef]
- Naz, A.; Masood, H.; Ehsan, S.; Tahir, T. Removal of acid black 1 by acacia concinna; adsorption kinetics, isotherm and thermodynamic study. Membr. Water Treat. 2020, 11, 407–416. [Google Scholar] [CrossRef]
- Nielsen, P.H.; McIlroy, S.J.; Albertsen, M.; Nierychlo, M. Re-evaluating the microbiology of the enhanced biological phosphorus removal process. Curr. Opin. Biotechnol. 2019, 57, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Bellier, N.; Chazarenc, F.; Comeau, Y. Phosphorus removal from wastewater by mineral apatite. Water Res. 2006, 40, 2965–2971. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Pratt, C.; Parsons, S.A.; Soares, A.; Martin, B.D. Biologically and chemically mediated adsorption and precipitation of phosphorus from wastewater. Curr. Opin. Biotechnol. 2012, 23, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Kyllönen, H.; Heikkinen, J.; Ceras, J.; Fernandez, C.; Porc, O.; Grönroos, A. Membrane-based conceptual design of reuse water production from candy factory wastewater. Water Sci. Technol. 2021, 84, 1389–1402. [Google Scholar] [CrossRef]
- Yin, Z.; Chen, Q.; Zhao, C.; Fu, Y.; Li, J.; Feng, Y.; Li, L. A new approach to removing and recovering phosphorus from livestock wastewater using dolomite. Chemosphere 2020, 255, 127005. [Google Scholar] [CrossRef]
- Li, R.; Kelly, C.; Keegan, R.; Xiao, L.; Morrison, L.; Zhan, X. Phosphorus removal from wastewater using natural pyrrhotite. Colloids Surfaces A Physicochem. Eng. Asp. 2013, 427, 13–18. [Google Scholar] [CrossRef]
- Paul, E.; Laval, M.L.; Sperandio, M. Excess sludge production and costs due to phosphorus removal. Environ. Technol. 2001, 22, 1363–1371. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Kim, J.; Yoon, S.; Reddy, P.A.K.; Hwang, Y.; Bae, S. The role of Fe dissolution in olivine-hydroxylamine-induced Fenton reaction for enhanced oxidative degradation of organic pollutant. Chemosphere 2022, 306, 135557. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Lee, W. Enhanced reductive degradation of carbon tetrachloride by biogenic vivianite and Fe(II). Geochim. Et Cosmochim. Acta 2012, 85, 170–186. [Google Scholar] [CrossRef]
- Yoon, S.; Bae, S. Novel synthesis of nanoscale zerovalent iron from coal fly ash and its application in oxidative degradation of methyl orange by fenton reaction. J. Hazard. Mater. 2018, 365, 751–758. [Google Scholar] [CrossRef]
- Wu, H.-F.; Wang, J.-P.; Duan, E.-G.; Hu, W.-H.; Dong, Y.-B.; Zhang, G.-Q. Phosphorus removal by adsorbent based on poly-aluminum chloride sludge. Water Sci. Eng. 2020, 13, 193–201. [Google Scholar] [CrossRef]
- Caravelli, A.H.; Contreras, E.M.; Zaritzky, N.E. Phosphorous removal in batch systems using ferric chloride in the presence of activated sludges. J. Hazard. Mater. 2010, 177, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xing, X.-H.; Liu, Z.; Cui, L.; Yu, A.; Feng, Q.; Yang, H. Enhanced coagulation of ferric chloride aided by tannic acid for phosphorus removal from wastewater. Chemosphere 2008, 72, 290–298. [Google Scholar] [CrossRef]
- Cui, H.; Ren, W.; Lin, P.; Liu, Y. Structure control synthesis of iron oxide polymorph nanoparticles through an epoxide precipitation route. J. Exp. Nanosci. 2012, 8, 869–875. [Google Scholar] [CrossRef]
- Moreno, E.M.; Zayat, M.; Morales, M.P.; Serna, C.J.; Roig, A.; Levy, D. Preparation of narrow size distribution superparamagnetic γ-Fe2O3 nanoparticles in a sol-gel transparent SiO2 matrix. Langmuir 2002, 18, 4972–4978. [Google Scholar] [CrossRef]
- Burstein, G.T. The iron oxides: Structure, properties, reactions, occurrence and uses. Corros. Sci. 1997, 39, 1499–1500. [Google Scholar] [CrossRef]
- Yousefi, T.; Davarkhah, R.; Golikand, A.N.; Mashhadizadeh, M.H.; Abhari, A. Facile cathodic electrosynthesis and characterization of iron oxide nano-particles. Prog. Nat. Sci. 2013, 23, 51–54. [Google Scholar] [CrossRef] [Green Version]
- Hou, J.; Luo, J.; Song, S.; Li, Y.; Li, Q. The remarkable effect of the coexisting arsenite and arsenate species ratios on arsenic removal by manganese oxide. Chem. Eng. J. 2017, 315, 159–166. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Z.; You, Y.; Zheng, X.; Wen, J. Synthesis of different structured FePO4 for the enhanced conversion of methyl cellulose to 5-hydroxymethylfurfural. RSC Adv. 2017, 7, 51281–51289. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.E.; Teng, T.T.; Morad, N.; Poh, B.T.; Mahalingam, M. Flocculation activity of novel ferric chloride–polyacrylamide (FeCl3-PAM) hybrid polymer. Desalination 2011, 266, 108–113. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Y.; Zhang, J.; Zhang, S.; Xu, S. The transformation from amorphous iron phosphate to sodium iron phosphate in sodium-ion batteries. Phys. Chem. Chem. Phys. 2015, 17, 22144–22151. [Google Scholar] [CrossRef]

| Fe (wt%) | O (wt%) | P (wt%) | S (wt%) | |
|---|---|---|---|---|
| Before reaction | 55.8 | 43.3 | 0 | 0 |
| After reaction | 24.6 | 60.2 | 11.7 | 2.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, J.; Choi, M.; Police, A.K.R.; Lee, J.; Bae, S. Removal of Phosphorus by Ferric Ion-Rich Solutions Prepared Using Various Fe(III)-Containing Minerals. Water 2022, 14, 3765. https://doi.org/10.3390/w14223765
Jung J, Choi M, Police AKR, Lee J, Bae S. Removal of Phosphorus by Ferric Ion-Rich Solutions Prepared Using Various Fe(III)-Containing Minerals. Water. 2022; 14(22):3765. https://doi.org/10.3390/w14223765
Chicago/Turabian StyleJung, Jueun, Minhee Choi, Anil Kumar Reddy Police, Jungho Lee, and Sungjun Bae. 2022. "Removal of Phosphorus by Ferric Ion-Rich Solutions Prepared Using Various Fe(III)-Containing Minerals" Water 14, no. 22: 3765. https://doi.org/10.3390/w14223765







