A Vegetation Assessment of the Kearl Treatment Wetland following Exposure to Oil Sands Process-Affected Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Environmental and Water Quality Monitoring
2.3. Vegetation Assessment
2.4. Statistical Methods
3. Results and Discussion
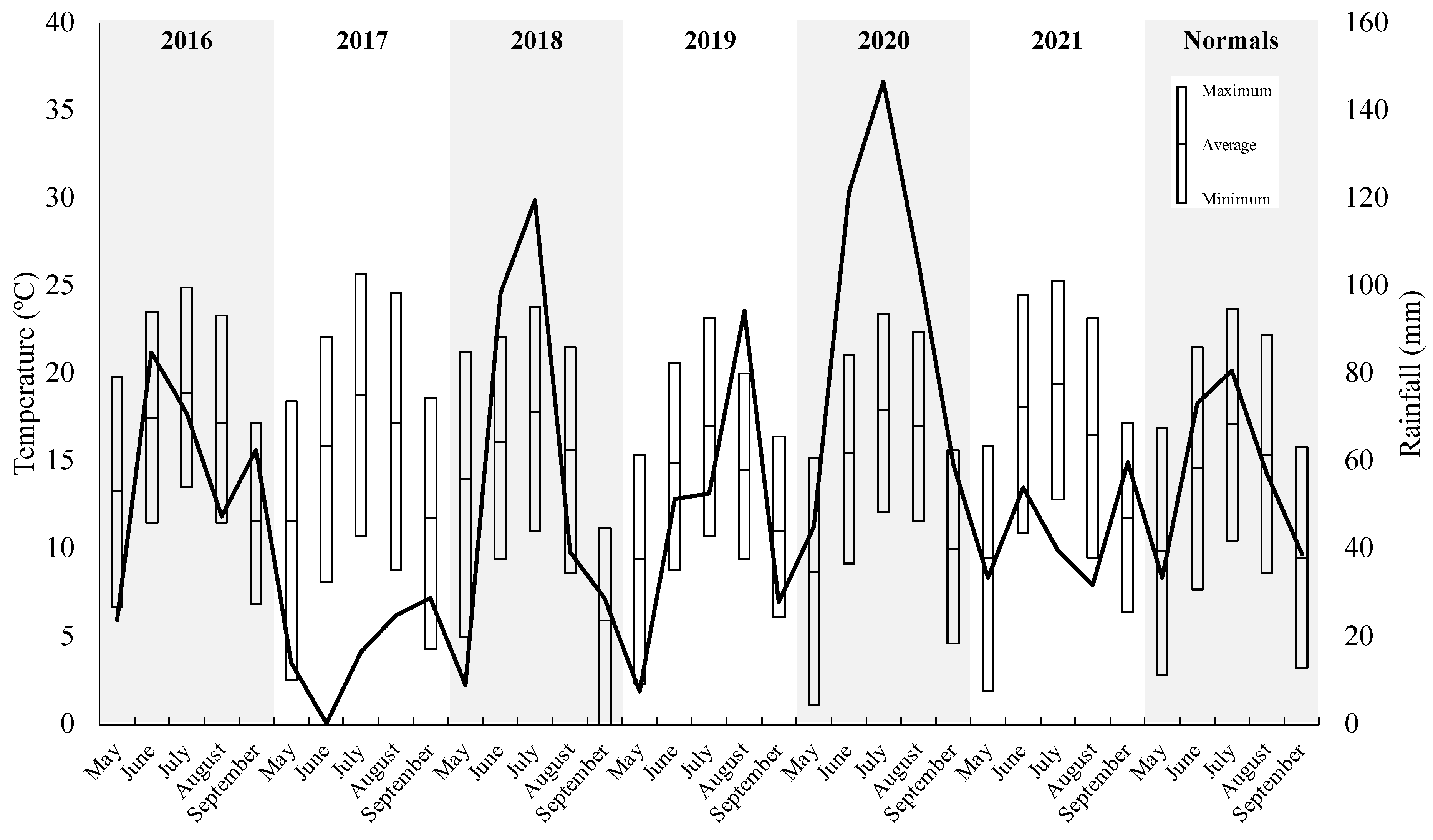
3.1. Water Quality and Environmental Data
3.2. Community Composition and Cover Types
3.3. Species Richness
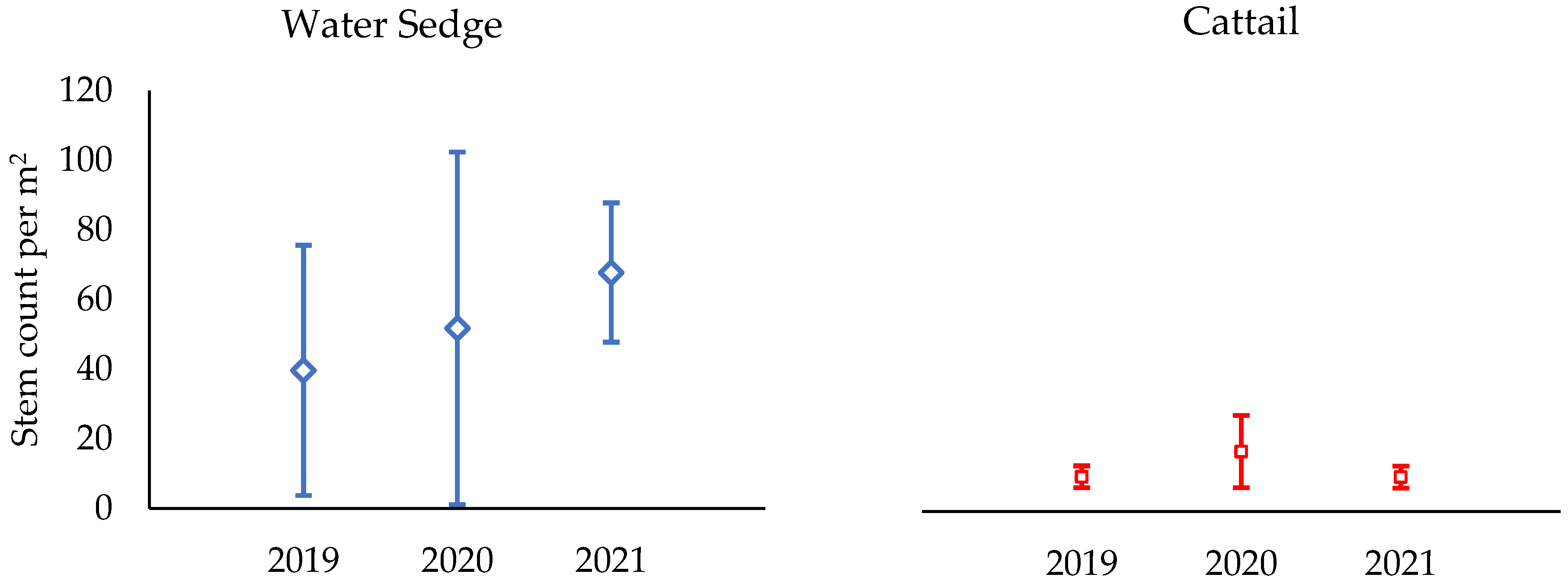
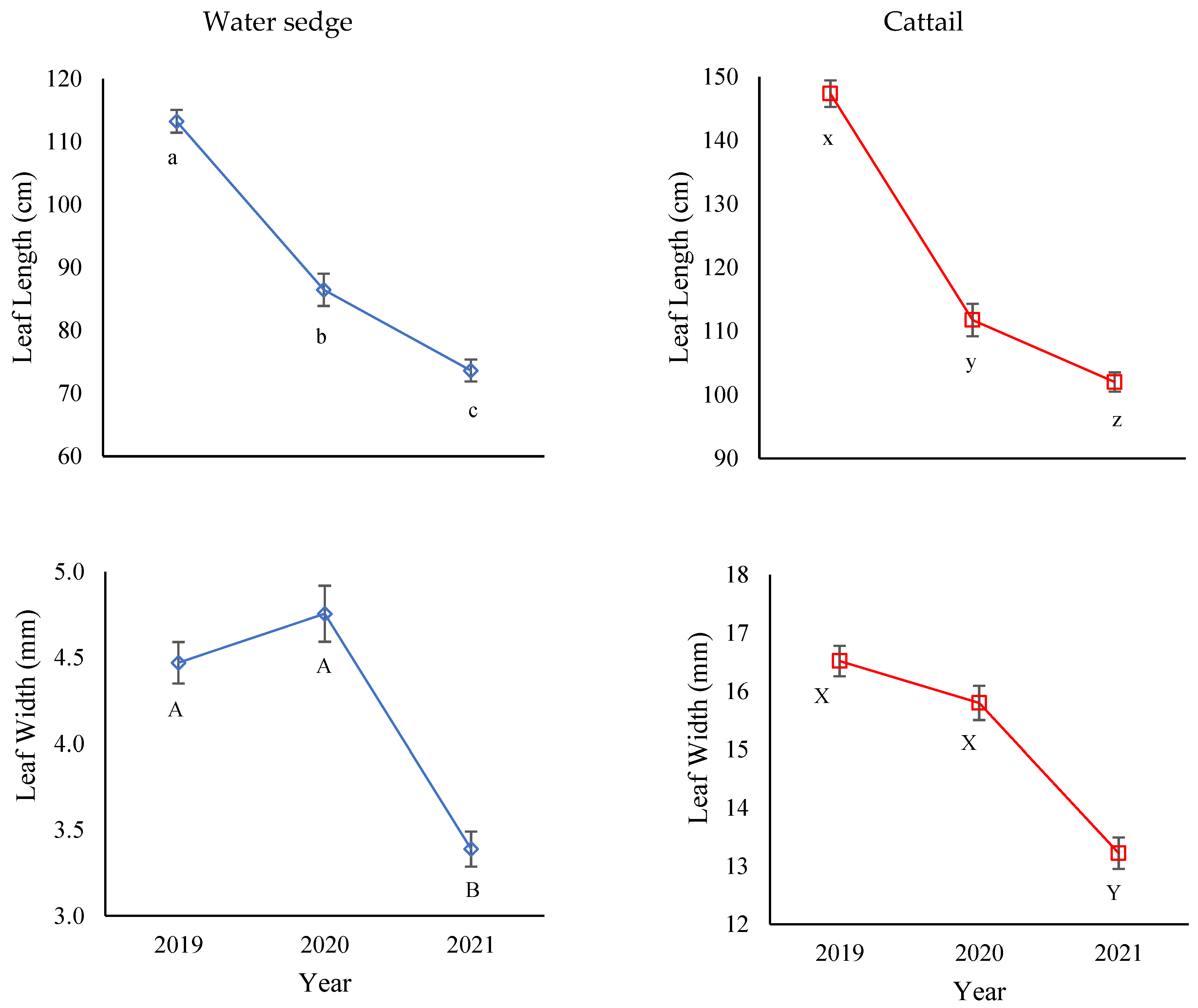
3.4. Morphology of Dominant Emergent Plants
3.5. Species Vigor
3.6. Effects of Water Level on Species Characteristics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miskimmin, B.; Fedorak, P.M.; Lauman, R.; Vinke, K. Oil Sands Water Toxicity: A Critical Review; Report No. 2010-089 (INT); Natural Resources Canada—CanmetENERGY: Devon, AB, Canada, 2010. [Google Scholar]
- Armstrong, S.A.; Headley, J.V.; Peru, K.M.; Germida, J.J. Phytotoxicity of oil sands naphthenic acids and dissipation from systems planted with emergent aquatic macrophytes. J. Environ. Sci. Health Part A 2007, 43, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, A.J.; Frank, R.A.; Gillis, P.L.; Parrott, J.L.; Marentette, J.R.; Brown, L.R.; Hewitt, L.M. Toxicity of naphthenic acids to invertebrates: Extracts from oil sands process-affected water versus commercial mixtures. Environ. Pollut. 2017, 227, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Marentette, J.R.; Frank, R.A.; Bartlett, A.J.; Gillis, P.L.; Hewitt, L.M.; Peru, K.M.; Parrott, J.L. Toxicity of naphthenic acid fraction components extracted from fresh and aged oil sands process-affected waters, and commercial naphthenic acid mixtures, to fathead minnow (Pimephales promelas) embryos. Aquat. Toxicol. 2015, 164, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Alberta Energy Regulator. Water Use Performance. 2020. Available online: https://www.aer.ca/protecting-what-matters/holding-industry-accountable/industry-performance/oil-sands-mining-water-use (accessed on 4 March 2020).
- Martin, J.W. The challenge: Safe release and reintegration of oil sands process-affected water. Environ. Toxicol. Chem. 2015, 34, 2682. [Google Scholar] [CrossRef] [Green Version]
- Ajaero, C.; Peru, K.M.; Simair, M.; Friesen, V.; O’Sullivan, G.; Hughes, S.A.; Headley, J.V. Fate and behavior of oil sands naphthenic acids in a pilot-scale treatment wetland as characterized by negative-ion electrospray ionization Orbitrap mass spectrometry. Sci. Total Environ. 2018, 631, 829–839. [Google Scholar] [CrossRef]
- Cancelli, A.M.; Gobas, F.A.P.C. Treatment of Polycyclic Aromatic Hydrocarbons in Oil Sands Process-Affected Water with a Surface Flow Treatment Wetland. Environments 2020, 7, 64. [Google Scholar] [CrossRef]
- Cancelli, A.M.; Gobas, F.A.P.C. Treatment of naphthenic acids in oil sands process-affected waters with a surface flow treatment wetland: Mass removal, half-life, and toxicity-reduction. Environ. Res. 2022, 213, 113755. [Google Scholar] [CrossRef]
- McQueen, A.D.; Hendrikse, M.; Gaspari, D.P.; Kinley, C.M.; Rodgers, J.H.; Castle, J.W. Performance of a hybrid pilot-scale constructed wetland system for treating oil sands process-affected water from the Athabasca oil sands. Ecol. Eng. 2017, 102, 152–165. [Google Scholar] [CrossRef]
- Hendrikse, M.; Gaspari, D.P.; McQueen, A.D.; Kinley, C.M.; Calomeni, A.J.; Geer, T.D.; Castle, J.W. Treatment of oil sands process-affected waters using a pilot-scale hybrid constructed wetland. Ecol. Eng. 2018, 115, 45–57. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Wallace, S.D. Treatment Wetlands; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Thullen, J.S.; Sartoris, J.J.; Nelson, S.M. Managing vegetation in surface-flow wastewater-treatment wetlands for optimal treatment performance. Ecol. Eng. 2005, 25, 583–593. [Google Scholar] [CrossRef]
- Marzocchi, U.; Benelli, S.; Larsen, M.; Bartoli, M.; Glud, R.N. Spatial heterogeneity and short-term oxygen dynamics in the rhizosphere of Vallisneria spiralis: Implications for nutrient cycling. Freshw. Biol. 2019, 64, 532–543. [Google Scholar] [CrossRef]
- Toscano, A.; Langergraber, G.; Consoli, S.; Cirelli, G.L. Modelling pollutant removal in a pilot-scale two-stage subsurface flow constructed wetlands. Ecol. Eng. 2009, 35, 281–289. [Google Scholar] [CrossRef]
- Zhao, R.; Cheng, J.; Yuan, Q.; Chen, Y.; Kim, Y. Contributions of emergent vegetation acting as a substrate for biofilms in a free water surface constructed wetland. Membr. Water Treat. 2019, 10, 57–65. [Google Scholar] [CrossRef]
- Karathanasis, A.D.; Potter, C.L.; Coyne, M.S. Vegetation effects on fecal bacteria, BOD, and suspended solid removal in constructed wetlands treating domestic wastewater. Ecol. Eng. 2003, 20, 157–169. [Google Scholar] [CrossRef]
- Shelef, O.; Gross, A.; Rachmilevitch, S. Role of Plants in a Constructed Wetland: Current and New Perspectives. Water 2013, 5, 405–419. [Google Scholar] [CrossRef]
- Alberts, M.E.; Wong, J.; Hindle, R.; Degenhardt, D.; Krygier, R.; Turner, R.J.; Muench, D.G. Detection of naphthenic acid uptake into root and shoot tissues indicates a direct role for plants in the remediation of oil sands process-affected water. Sci. Total Environ. 2021, 795, 148857. [Google Scholar] [CrossRef]
- Cancelli, A.M.; Gobas, F.A.P.C.; Wang, Q.; Kelly, B.C. Development and evaluation of a mechanistic model to assess the fate and removal efficiency of hydrophobic organic contaminants in horizontal subsurface flow treatment wetlands. Water Res. 2019, 151, 183–192. [Google Scholar] [CrossRef]
- Bendell-Young, L.I.; Bennett, K.E.; Crowe, A.; Kennedy, C.J.; Kermode, A.R.; Moore, M.M.; Plant, A.L.; Wood, A. Ecological characteristics of wetlands receiving an industrial effluent. Ecol. Appl. 2000, 10, 310–322. [Google Scholar] [CrossRef]
- Jivraj, M.N.; MacKinnon, M.; Fung, B. Naphthenic Acid Extraction and Quantitative Analysis with FT-IR Spectroscopy. In Syncrude Analytical Manuals; Syncrude Canada Ltd.: Edmonton, AB, USA, 1995. [Google Scholar]
- Holowenko, F.M.; MacKinnon, M.D.; Fedorak, P.M. Characterization of naphthenic acids in oil sands wastewaters by gas chromatography-mass spectrometry. Water Res. 2002, 36, 2843–2855. [Google Scholar] [CrossRef]
- Leung, S.S.; MacKinnon, M.D.; Smith, R.E.H. The ecological effects of naphthenic acids and salts on phytoplankton from the Athabasca oil sands region. Aquat. Toxicol. 2003, 62, 11–26. [Google Scholar] [CrossRef]
- Hughes, S.A.; Huang, R.; Mahaffey, A.; Chelme-Ayala, P.; Klamerth, N.; Meshref, M.N.A.; Ibrahim, M.D.; Brown, C.; Peru, K.M.; Headley, J.V.; et al. Comparison of methods for determination of total oil sands-derived naphthenic acids in water samples. Chemosphere 2017, 187, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Woudneh, M.B.; Coreen Hamilton, M.; Benskin, J.P.; Wang, G.; McEachern, P.; Cosgrove, J.R. A novel derivatization-based liquid chromatography tandem mass spectrometry method for quantitative characterization of naphthenic acid isomer profiles in environmental waters. J. Chromatogr. A 2013, 1293, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Collier, T.K.; Anulacion, B.F.; Arkoosh, M.R.; Dietrich, J.P.; Incardona, J.P.; Johnson, L.L.; Myers, M.S. Effects on Fish of Polycyclic Aromatic Hydrocarbons (PAHs) and Naphthenic Acid Exposures. Fish Physiol. 2013, 33, 195–255. [Google Scholar]
- McQueen, A.D.; Kinley, C.M.; Hendrikse, M.; Gaspari, D.P.; Calomeni, A.J.; Iwinski, K.J.; Castle, J.W.; Haakensen, M.C.; Peru, K.M.; Headley, J.V.; et al. A risk-based approach for identifying constituents of concern in oil sands process-affected water from the Athabasca Oil Sands region. Chemosphere 2017, 173, 340–350. [Google Scholar] [CrossRef]
- Shotyk, W.; Bicalho, B.; Cuss, C.; Donner, M.; Grant-Weaver, I.; Javed, M.B.; Noernberg, T. Trace elements in the Athabasca Bituminous Sands: A geochemical explanation for the paucity of environmental contamination by chalcophile elements. Chem. Geol. 2021, 581, 120392. [Google Scholar] [CrossRef]
- Government of Alberta. Canadian Climate Normals 1981–2010 Station Data. Fort McMurray, Alberta Station. 2020. Available online: https://climate.weather.gc.ca/climate_normals/index_e.html (accessed on 4 March 2020).
- Alberta Climate Information Service. Current and Historical Alberta Weather Station Data Viewer. Mildred Lake Weather Station. Data Provided by Alberta Agriculture and Forestry, Alberta Environment and Parks and Environment Canada. Government of Alberta. 2020. Available online: https://acis.alberta.ca/weather-data-viewer.jsp (accessed on 1 November 2016).
- Alberta Environment and Sustainable Resources Development. Ecological Land Survey Site Description Manual, 2nd ed.; Resource Data Branch, Strategic Corporate Services Division; Alberta Sustainable Resource Development: Edmonton, AB, USA, 2003. [Google Scholar]
- Stewart, R.E.; Kantrud, H.A. Classification of Natural Ponds and Lakes in the Glaciated Prairie Region; Resource Publication; US Bureau of Sport Fisheries and Wildlife: Washington, DC, USA, 1971; pp. 57–92.
- JMP®, Version 16; SAS Institute Inc.: Cary, NC, USA, 1989–2021.
- Hummel, R.M.; Claassen, E.A.; Wolfinger, R.D. JMP for Mixed Models; SAS Institute: Cary, NC, USA, 2021. [Google Scholar]
- Government of Alberta. Environmental Quality Guidelines for Alberta Surface Waters; Water Policy Branch, Alberta Environment and Parks: Edmonton, AB, USA, 2018.
- CCREM (Canadian Council of Resource and Environment Ministers). Canadian Water Quality Guidelines; Prepared by the Task Force on Water Quality Guidelines; CCREM: Winnipeg, MB, Canada, 1987.
- Canadian Council of Ministers of the Environment. Canadian water quality guidelines for the protection of aquatic life, Molybdenum. In Canadian Environmental Quality Guidelines; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 1999. [Google Scholar]
- Aihemaiti, A.; Jiang, J.; Gao, Y.; Meng, Y.; Zou, Q.; Yang, M.; Xu, Y.; Han, S.; Yan, W.; Tuerhong, T. The effect of vanadium on essential element uptake of Setaria viridis seedlings. J. Environ. Manag. 2019, 237, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Toor, N.S.; Franz, E.D.; Fedorak, P.M.; MacKinnon, M.D.; Liber, K. Degradation and aquatic toxicity of naphthenic acids in oil sands process-affected waters using simulated wetlands. Chemosphere 2013, 90, 449–458. [Google Scholar] [CrossRef]
- Mahaffey, A.; Dubé, M. Review of the composition and toxicity of oil sands process-affected water. Environ. Rev. 2017, 25, 97–114. [Google Scholar] [CrossRef]
- Galinato, M.I.; Van Der Valk, A.G. Seed germination traits of annuals and emergents recruited during drawdowns in the Delta Marsh, Manitoba, Canada. Aquat. Bot. 1986, 26, 89–102. [Google Scholar] [CrossRef]
- Trites, M.; Bayley, S.E. Vegetation communities in continental boreal wetlands along a salinity gradient: Implications for oil sands mining reclamation. Aquat. Bot. 2009, 91, 27–39. [Google Scholar] [CrossRef]
- Headley, J.V.; Crosley, B.; Conly, F.M.; Quagraine, E.K. The Characterization and Distribution of Inorganic Chemicals in Tributary Waters of the Lower Athabasca River, Oilsands Region, Canada. J. Environ. Sci. Health Part A 2005, 40, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Klomjek, P.; Nitisoravut, S. Constructed treatment wetland: A study of eight plant species under saline conditions. Chemosphere 2005, 58, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.W.S.; Pinto, L.J.; Kiehlmann, E.; Bendell-Young, L.I.; Moore, M.M. Factors that affect the degradation of naphthenic acids in oil sands wastewater by indigenous microbial communities. Environ. Toxicol. Chem. 1996, 15, 1482–1491. [Google Scholar] [CrossRef]
- Hicks, K.; Scrimgeour, G. Summary of Enhanced Monitoring of the Lower Athabasca River; Government of Alberta, Ministry of Environment and Parks, 2018. Available online: open.alberta.ca/publications/9781460145371 (accessed on 10 April 2022)ISBN 978-1-4601-4537-1.
- Allen, E.W. Process water treatment in Canada’s oil sands industry: II. A review of emerging technologies. J. Environ. Eng. Sci. 2008, 7, 499–524. [Google Scholar] [CrossRef]
- Regional Aquatics Monitoring Program. Query Water Quality Data. RAMP Water Quality Data Report for all Sites from 1997 to 2009. 2022. Available online: http://www.ramp-alberta.org/data/Water/waterquality/water.aspx (accessed on 15 March 2022).
- Bhatia, M.; Goyal, D. Analyzing Remediation Potential of Wastewater Through Wetland Plants: A Review. Environ. Prog. Sustain. Energy 2013, 33, 9–27. [Google Scholar] [CrossRef]
- Leguizamo, M.A.; Gomez, W.D.F.; Sarmiento, M. Native herbaceous plant species with potential use in phytoremediation of heavy metals, spotlight on wetlands—A review. Chemosphere 2017, 168, 1230–1247. [Google Scholar] [CrossRef]
- Smith, S.G. Schoenoplectus. Flora of North America Association. Available online: http://floranorthamerica.org/Schoenoplectus (accessed on 10 April 2022).
- Allen, L.; Kershaw, L. Vascular Flora of Alberta: An Illustrated Guide; Amazon Digital Services LLC-KDP Print US. 2020. Available online: https://www.amazon.com/Vascular-Flora-Alberta-Illustrated-Guide/dp/1777244102 (accessed on 10 April 2022).
- Weisner, S.E.; Thiere, G. Effects of vegetation state on biodiversity and nitrogen retention in created wetlands: A test of the biodiversity–ecosystem functioning hypothesis. Freshw. Biol. 2010, 55, 387–396. [Google Scholar] [CrossRef]
- Hadad, H.R.; Maine, M.A.; Bonetto, C.A. Macrophyte growth in a pilot-scale constructed wetland for industrial wastewater treatment. Chemosphere 2006, 63, 1744–1753. [Google Scholar] [CrossRef]
- Wang, Q.; Kelly, B.C. Occurrence, distribution and bioaccumulation behaviour of hydrophobic organic contaminants in a large-scale constructed wetland in Singapore. Chemosphere 2017, 183, 257–265. [Google Scholar] [CrossRef]
- Mollard, F.P.O.; Roy, M.C.; Frederick, K.; Foote, L. Growth of the dominant macrophyte Carex aquatilis is inhibited in oil sands affected wetlands in Northern Alberta, Canada. Ecol. Eng. 2012, 38, 11–19. [Google Scholar] [CrossRef]
- AU Crowe, B Han, AR Kermode, LI Bendell-Young, and AL Plant, Effects of oil sands effluent on cattail and clover: Photosynthesis and the level of stress proteins. Environ. Pollut. 2001, 113, 311–322. [CrossRef]
- Simair, M.C.; Parrott, J.L.; le Roux, M.; Gupta, V.; Frank, R.A.; Peru, K.M.; Headley, J.V. Treatment of oil sands process affected waters by constructed wetlands: Evaluation of designs and plant types. Sci. Total Environ. 2021, 772, 145508. [Google Scholar] [CrossRef] [PubMed]
- Crowe, A.U.; Plant, A.L.; Kermode, A.R. Effects of an industrial effluent on plant colonization and on the germination and post-germinative growth of seeds of terrestrial and aquatic plant species. Environ. Pollut. 2002, 117, 179–189. [Google Scholar] [CrossRef]
- Vretare, V.; Weisner, S.E.; Strand, J.A.; Granéli, W. Phenotypic plasticity in Phragmites australis as a functional response to water depth. Aquat. Bot. 2001, 69, 127–145. [Google Scholar] [CrossRef]
- Kemp, W.M.; Batleson, R.; Bergstrom, P.; Carter, V.; Gallegos, C.L.; Hunley, W.; Karrh, L.; Koch, E.W.; Landwehr, J.M.; Moore, K.A.; et al. Habitat requirements for submerged aquatic vegetation in Chesapeake Bay: Water quality, light regime, and physical-chemical factors. Estuaries 2004, 27, 363–377. [Google Scholar] [CrossRef]
- Rooney, R.C.; Bayley, S.E. Setting reclamation targets and evaluating progress: Submersed aquatic vegetation in natural and post-oil sands mining wetlands in Alberta, Canada. Ecol. Eng. 2011, 37, 569–579. [Google Scholar] [CrossRef]
- Whitehouse, H.E.; Bayley, S.E. Vegetation patterns and biodiversity of peatland plant communities surrounding mid-boreal wetland ponds in Alberta, Canada. Can. J. Bot. 2005, 83, 621–637. [Google Scholar] [CrossRef]
- Narayan, D.; Agrawal, M.; Pandey, J.; Singh, J. Changes in vegetation characteristics downwind of an aluminum factory in India. Ann. Bot. 1994, 73, 557–565. [Google Scholar] [CrossRef]
- Brisson, J.; Chazarenc, F. Maximizing pollutant removal in constructed wetlands: Should we pay more attention to macrophyte species selection? Sci. Total Environ. 2009, 407, 3923–3930. [Google Scholar] [CrossRef]
- Taylor, C.R.; Hook, P.B.; Stein, O.R.; Zabinski, C.A. Seasonal effects of 19 plant species on COD removal in subsurface treatment wetland microcosms. Ecol. Eng. 2011, 37, 703–710. [Google Scholar] [CrossRef] [Green Version]
- Terry, N.; Banuelos, G.S. Phytoremediation of Contaminated Soil and Water, 1st ed.; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Geng, Y.; Ge, Y.; Luo, B.; Chen, Z.; Min, Y.; Schmid, B.; Chang, J. Plant diversity increases N removal in constructed wetlands when multiple rather than single N processes are considered. Ecol. Appl. 2019, 29, e01965. [Google Scholar] [CrossRef]
- Griffiths, L.N.; Haupt, T.N.; Zhang, L.; Mitsch, W.J. Role of emergent and submerged vegetation and algal communities on nutrient retention and management in a subtropical urban stormwater treatment wetland. Wetl. Ecol. Manag. 2021, 29, 245–264. [Google Scholar] [CrossRef]
- Haakensen, M.; Pittet, V.; Spacil, M.M.; Castle, J.W.; Rodgers, J.H., Jr. Key aspects for successful design and implementation of passive water treatment systems. J. Environ. Solut. Oil Gas Min. 2015, 1, 59–81. [Google Scholar] [CrossRef]
- Kamaluddin, M.; Zwiazek, J.J. Naphthenic acids inhibit root water transport, gas exchange and leaf growth in aspen (Populus tremuloides) seedlings. Tree Physiol. 2002, 22, 1265–1270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, S.A.; Headley, J.V.; Peru, K.M.; Mikula, R.J.; Germida, J. Phytotoxicity and naphthenic acid dissipation from oil sands fine tailings treatments planted with the emergent macrophyte Phragmites australis. J. Environ. Sci. Health Part A 2010, 45, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Pouliot, R.; Rochefort, L.; Graf, M.D. Impacts of oil sands process water on fen plants: Implications for plant selection in required reclamation projects. Environ. Pollut. 2012, 167, 132–137. [Google Scholar] [CrossRef]
- Vitt, D.H.; Glaeser, L.C.; House, M.; Kitchen, S.P. Structural and functional responses of Carex aquatilis to increasing sodium concentrations. Wetl. Ecol. Manag. 2020, 28, 753–763. [Google Scholar] [CrossRef]
- Grace, J.B. Effects of water depth on Typha latifolia and Typha domingensis. Am. J. Bot. 1989, 76, 762–768. [Google Scholar] [CrossRef]
- Grace, J.B.; Wetzel, R.G. Niche differentiation between two rhizomatous plant species: Typha latifolia and Typha angustifolia. Can. J. Bot. 1982, 60, 46–57. [Google Scholar] [CrossRef]
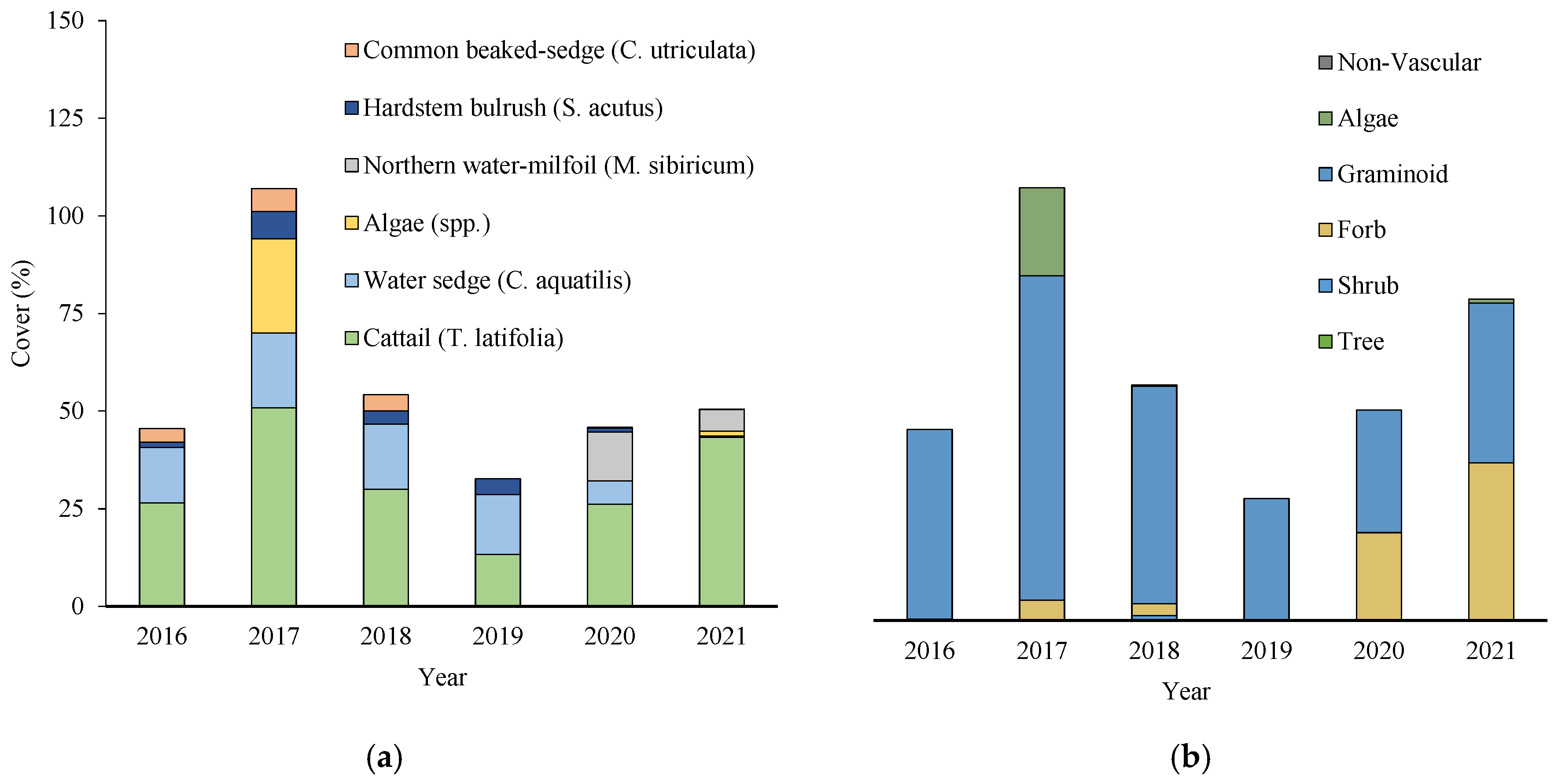

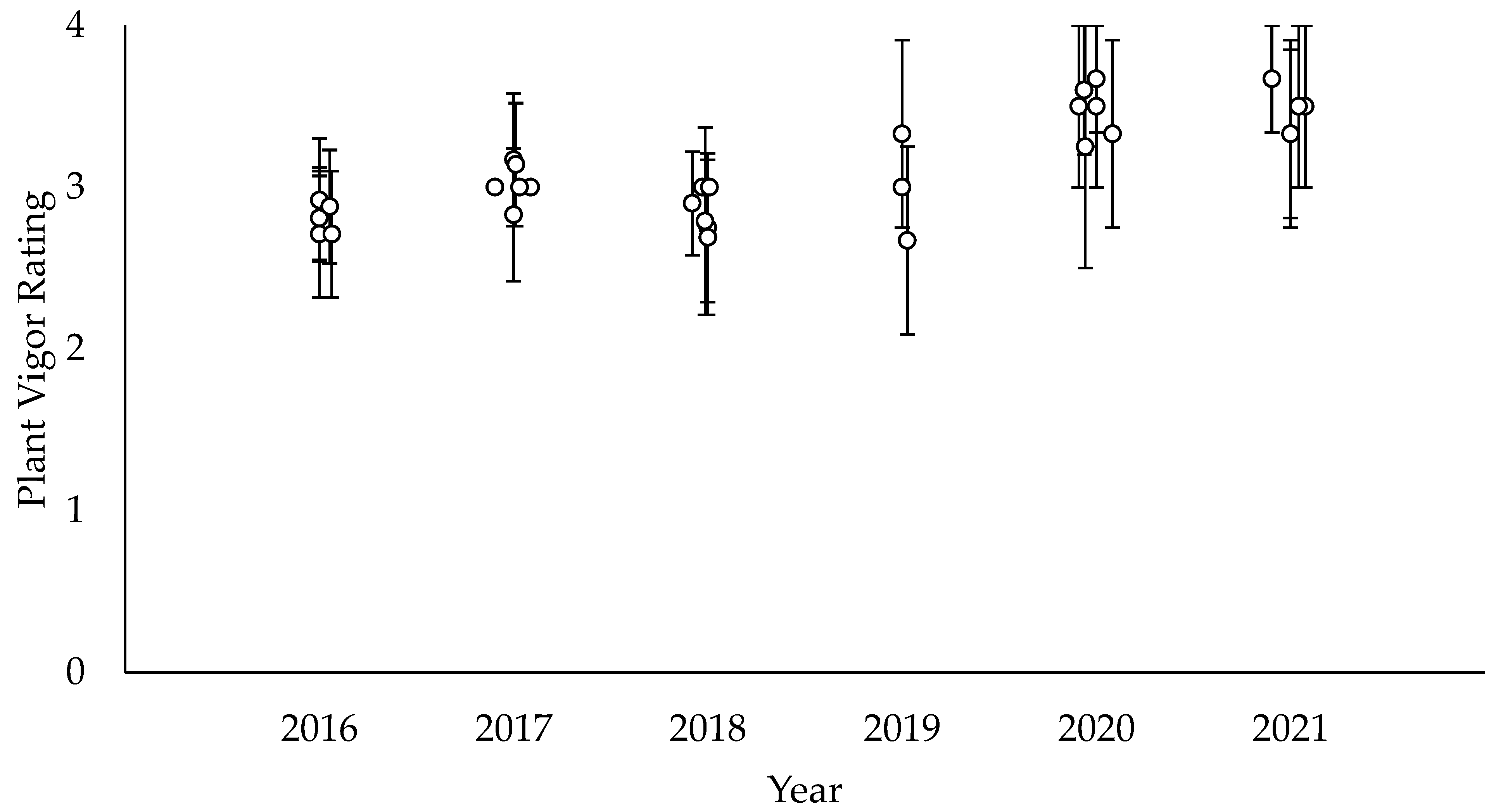
| Laboratory Measurements | Field Measurements | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year | OSPW Source | ΣTM | ΣTDM | PHCs | NAs | TDS | TKN | TP | Ortho-P | pH | SC | Turbidity | DO | ||
| ug/L | ug/L | ug/L | mg/L | mg/L | mg/L | mg/L | mg/L | n | -- | µS/cm | NTU | mg/L | n | ||
| 2016 | Runoff | 4.65 | 3.83 | <500 | <2.0 a | 520 | 0.93 | 0.029 | -- | 1 | 8.2 | 750 | 11 | -- | 0 * |
| 2017 | Runoff | 6.67 (2.54) | 6.55 (2.53) | 55 | 3.1 a (0.87) | 610 (99) | 0.47 | 0.008 (0.002) | 0.006 (0.006) | 2 | 7.9 (0.04) | 883 (101) | 0.84 (0.14) | 4.83 (0.37) | 2 |
| 2018 | DPW | 5.4 | 4.4 | 150 | 19.7 b | 560 | 0.66 | 0.013 | 0.003 | 1 | 7.0 | 1343 | 65.6 | 8.02 | 1 |
| 2019 | DPW | 17.7 (19.0) | 17.4 (19.9) | 45 | 20.7 b (0.28) | 1250 (58) | 0.59 | 0.007 (0.005) | 0.004 (0.002) | 4 | 7.5 (0.4) | 1515 (280) | 19.6 (6.8) | 7.76 (1.76) | 10 |
| 2020 | † | 5.9 | 5.1 | <500 | <2.0 a | 1000 | 0.71 | 0.004 | -- | 1 | 7.8 | 1118 | 1.0 | 7.46 | 1 |
| 2021 | TPW | 46 (37) | 40.3 (38) | 4328 (3307) | 47.6 b (8.2) | 858 (36) | 0.94 | 0.022 (0.005) | 0.005 (0.003) | 4 | 8.0 (0.2) | 1226 (72) | 10.2 (1.5) | 7.37 (1.8) | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cancelli, A.M.; Borkenhagen, A.K.; Bekele, A. A Vegetation Assessment of the Kearl Treatment Wetland following Exposure to Oil Sands Process-Affected Water. Water 2022, 14, 3686. https://doi.org/10.3390/w14223686
Cancelli AM, Borkenhagen AK, Bekele A. A Vegetation Assessment of the Kearl Treatment Wetland following Exposure to Oil Sands Process-Affected Water. Water. 2022; 14(22):3686. https://doi.org/10.3390/w14223686
Chicago/Turabian StyleCancelli, Alexander M., Andrea K. Borkenhagen, and Asfaw Bekele. 2022. "A Vegetation Assessment of the Kearl Treatment Wetland following Exposure to Oil Sands Process-Affected Water" Water 14, no. 22: 3686. https://doi.org/10.3390/w14223686





