Study of Organic Acid Pollutant Removal Efficient in Treatment of Industrial Wastewater with HDH Process Using ASPEN Modelling
Abstract
:1. Introduction
2. Materials and Methods
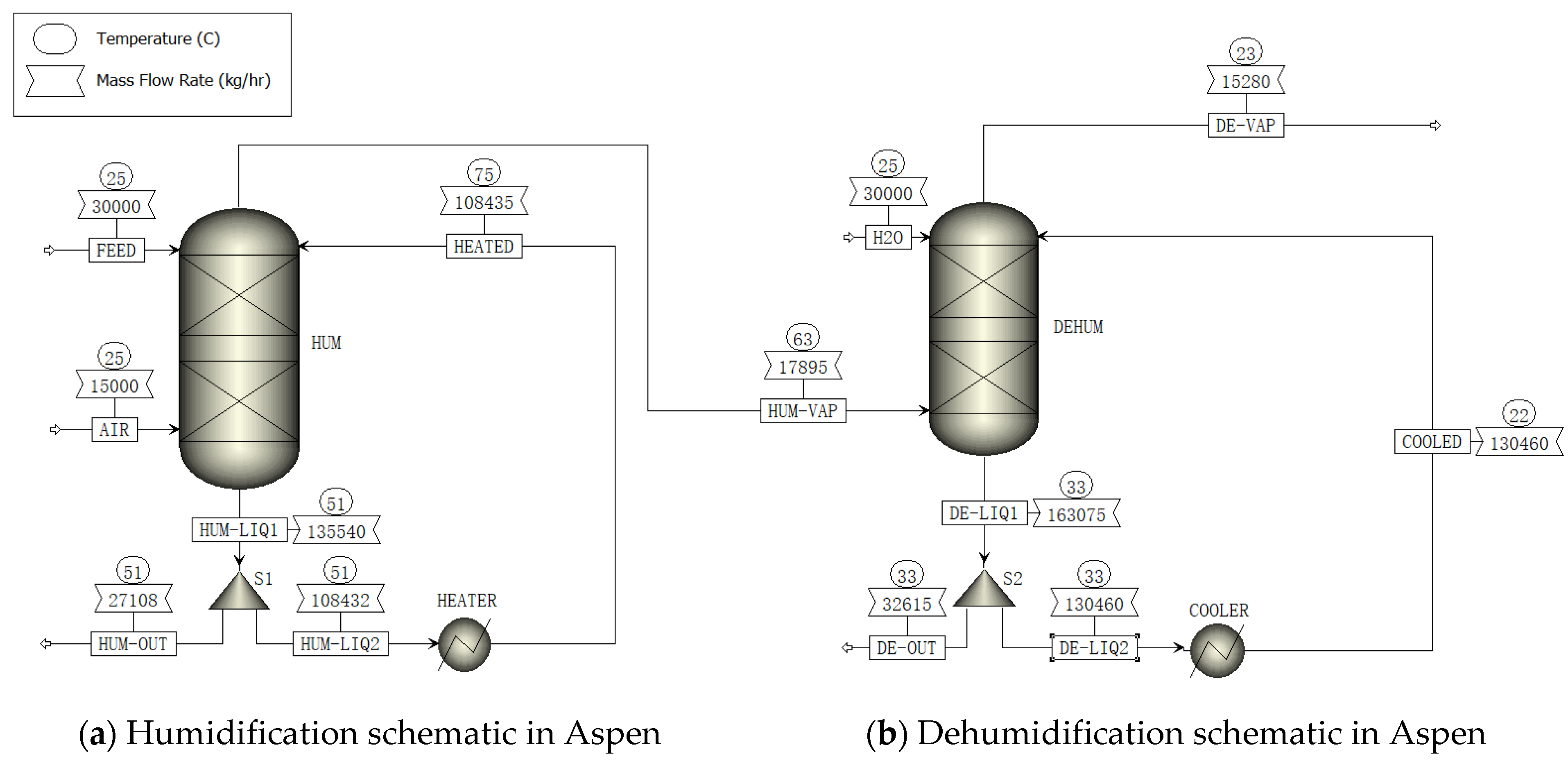
2.1. ASPEN Simulation
- (a)
- Humidification process
- (b)
- Dehumidification process
2.2. Thermodynamic Model
2.3. Input Acid Wastewater
2.4. Units in HDH System
3. Results and Discussion
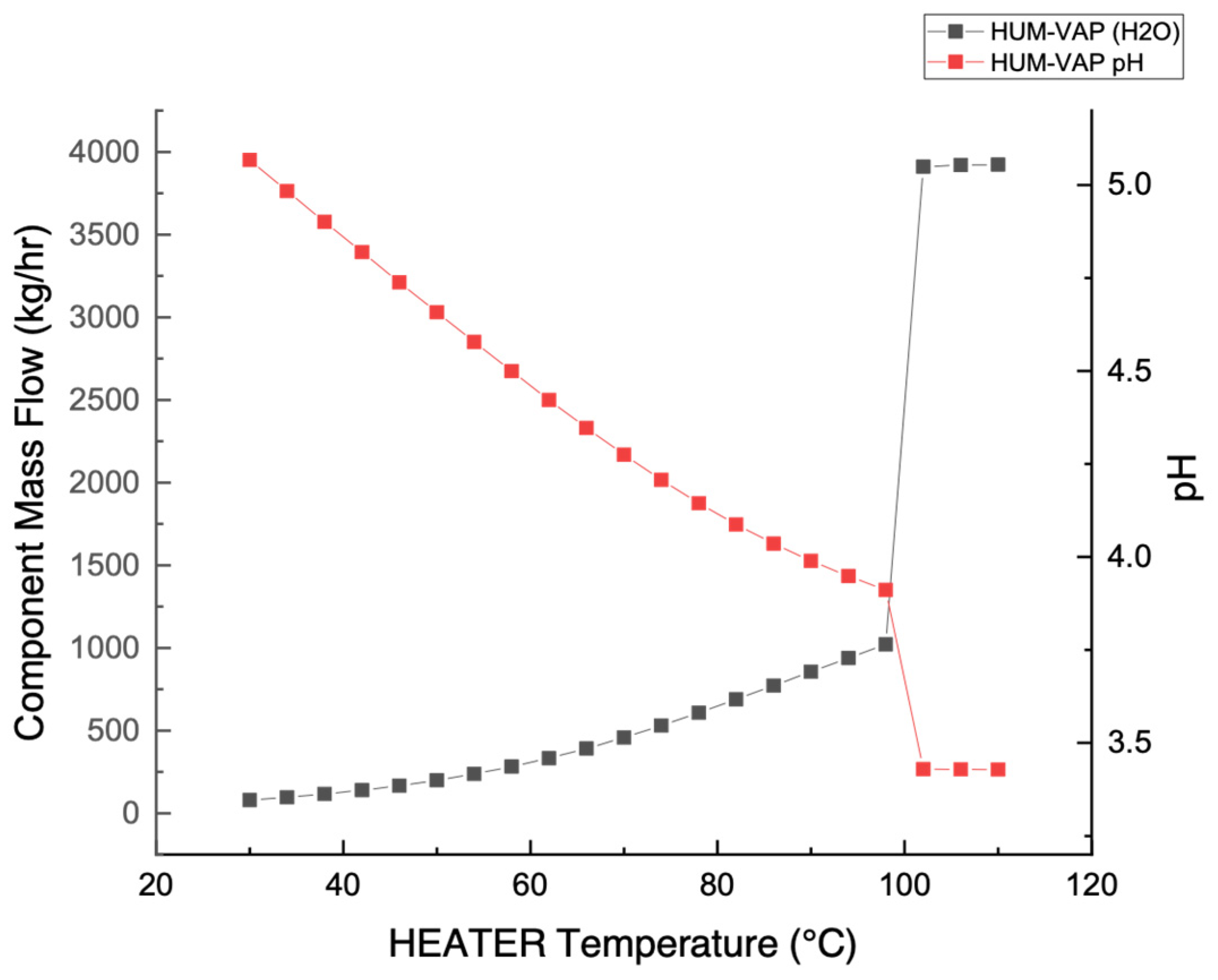
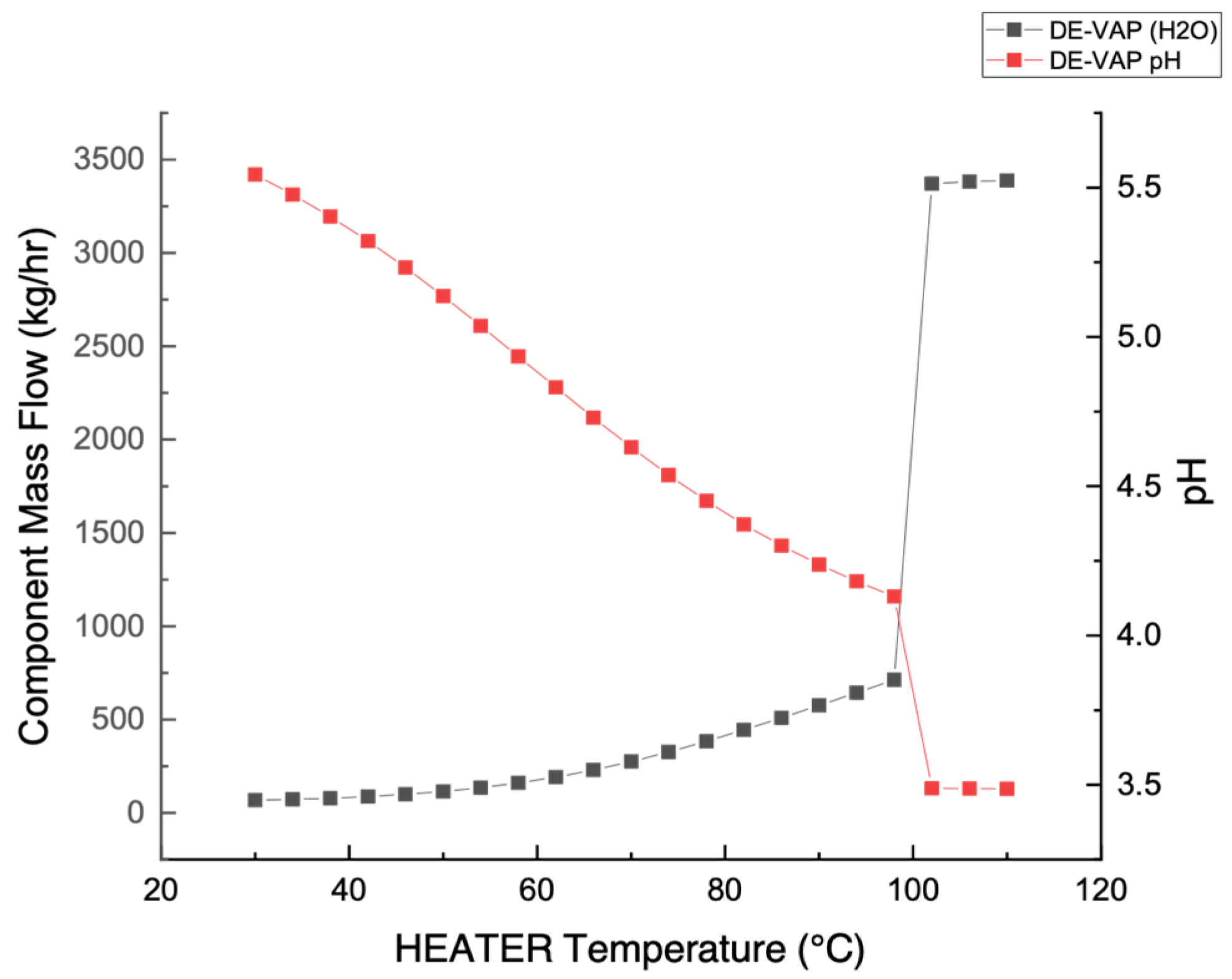
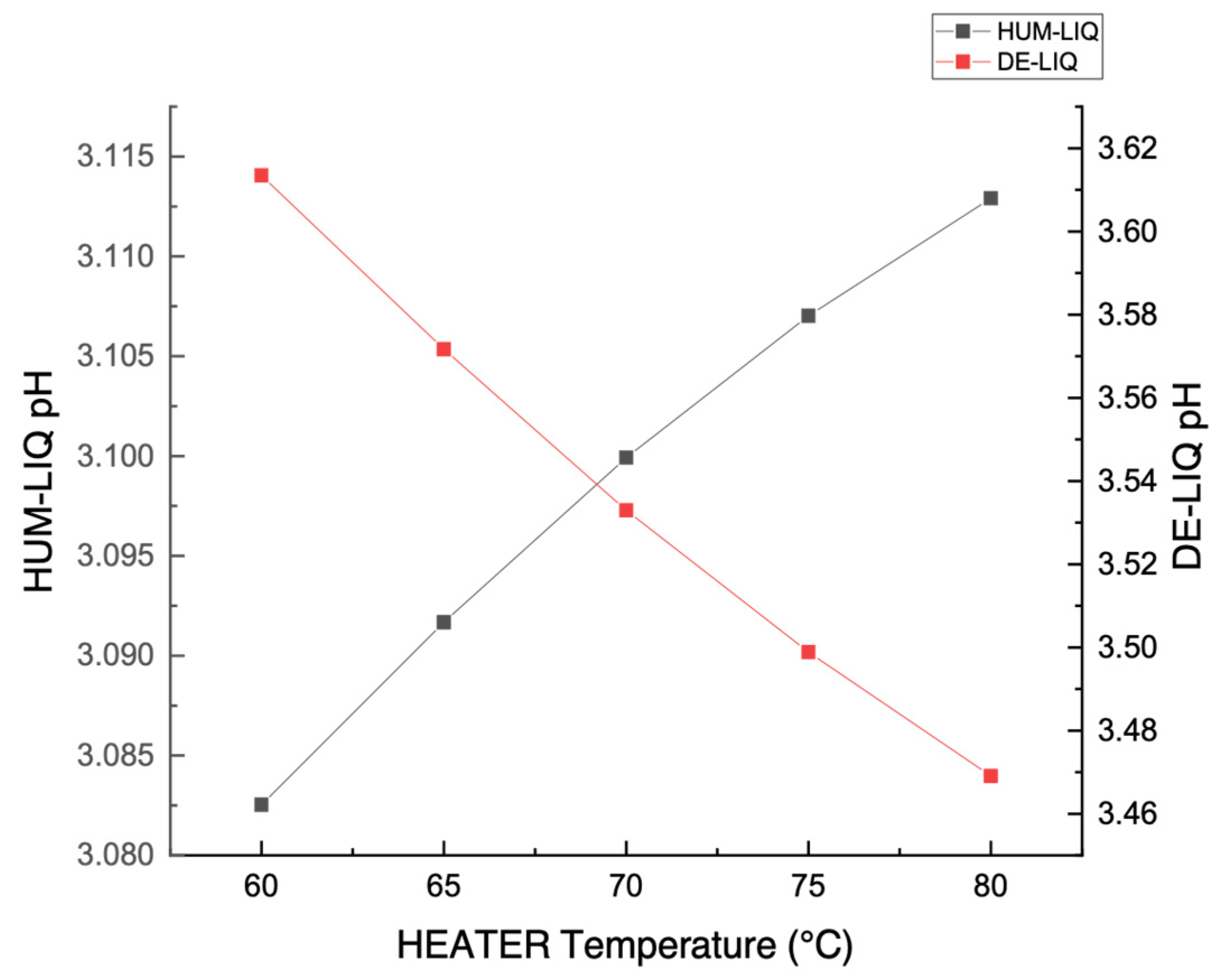
3.1. Stream Acidity (pH) Measurement
3.2. Organic Pollutant (COD) Measurement
- C = Number of carbon atoms
- H = Number of hydrogen atoms
- O = Number of oxygen atoms
- M = Molar Mass
3.3. Temperature Measurement
3.4. Yield Liquid Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dedecan, T.; Baylan, N.; Inci, I. Synthesis, characterization and application of calcium peroxide nanoparticles as a novel adsorbent for removal of malic acid from aqueous solutions. Chem. Phys. Lett. 2022, 797, 139581. [Google Scholar] [CrossRef]
- Moon, S.Y.; Hong, S.H.; Kim, T.Y.; Lee, S.Y. Metabolic engineering of Escherichia coli for the production of malic acid. Biochem. Eng. J. 2008, 40, 312–320. [Google Scholar] [CrossRef]
- Zou, X.; Zhou, Y.; Yang, S.T. Production of polymalic acid and malic acid by Aureo basidium pullulans fermentation and acid hydrolysis. Biotechnol. Bioeng. 2013, 110, 2105–2113. [Google Scholar] [CrossRef] [PubMed]
- Nada, S.A.; Elattar, H.F.; Fouda, A. Experimental study for hybrid humidification–dehumidification water desalination and air conditioning system. Desalination 2015, 363, 112–125. [Google Scholar] [CrossRef]
- Giwa, A. Salty groundwater treatment: Recovery of magnetic nano-particles. In Proceedings of the International Conference on Hydrology & Groundwater Expo, Raleigh, NC, USA, 26–27 August 2013. [Google Scholar]
- Narayan, G.P.; Sharqawy, M.H.; Summers, E.K.; Lienhard, J.H.; Zubair, S.M.; Antar, M.A. The potential of solar-driven humidification-dehumidification desalination for small-scale decentralized water production. Renew. Sustain. Energy Rev. 2010, 14, 1187–1201. [Google Scholar] [CrossRef]
- Lawal, D.U.; Antar, M.A.; Khalifa, A.E. Integration of a MSF desalination system with a HDH system for brine recovery. Sustainability 2021, 13, 3506. [Google Scholar] [CrossRef]
- Hamed, M.H.; Kabeel, A.E.; Omara, Z.M.; Sharshir, S.W. Mathematical and experimental investigation of a solar humidification–dehumidification desalination unit. Desalination 2015, 358, 9–17. [Google Scholar] [CrossRef]
- Abdel Dayem, A.M. Effificient solar desalination system using humidification-dehumidification process. J. Sol. Energy Eng. 2014, 136, 041014. [Google Scholar] [CrossRef]
- Abdullah, A.S.; Essa, F.A.; Omara, Z.M.; Bek, M.A. Performance evaluation of a humidification–dehumidification unit integrated with wick solar stills under different operating conditions. Desalination 2018, 441, 52–61. [Google Scholar] [CrossRef]
- Kappagantula, R.V.; Ingram, G.D.; Vuthaluru, H.B. Application of Aspen Plus fluidized bed reactor model for chemical Looping of synthesis gas. Fuel 2022, 324, 124698. [Google Scholar] [CrossRef]
- Emami, M.; Hejazi, B.; Karimi, M.; Mousavi, S.A. Quantitative risk assessment and risk reduction of integrated acid gas enrichment and amine regeneration process using Aspen Plus dynamic simulation. Results Eng. 2022, 15, 100566. [Google Scholar] [CrossRef]
- Liu, G.; Sun, J.; Zhang, J.; Tu, Y.; Bao, J. High titer l-lactic acid production from corn stover with minimum wastewater generation and techno-economic evaluation based on Aspen plus modeling. Bioresour. Technol. 2015, 198, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Rahman, N.A.; Jol, C.J.; Albania, A. Fischer Tropsch water composition study from distillation process in gas to liquid technology with ASPEN simulation. Case Stud. Chem. Environ. Eng. 2021, 3, 100106. [Google Scholar] [CrossRef]
- Taqvi, S.A.; Tufa, L.D.; Muhadizir, S. Optimization and dynamics of distillation column using aspen Plus. Procedia Eng. 2016, 148, 978–984. [Google Scholar] [CrossRef] [Green Version]
- Puentes, C.; Joulia, X.; Athès, V.; Esteban-Decloux, M. Review and thermodynamic modeling with nrtl model of vapor–liquid equilibria (VLE) of aroma compounds highly diluted in ethanol–water mixtures at 101.3 kPa. Ind. Eng. Chem. Res. 2018, 57, 3443–3470. [Google Scholar] [CrossRef] [Green Version]
- Kelly, B.D.; de Klerk, A. Modelling vapor–liquid–liquid phase equilibria in fischer–tropsch syncrude. Ind. Eng. Chem. Res. 2015, 54, 9857–9869. [Google Scholar] [CrossRef]
- Mehrgoo, M.; Amidpour, M. Constructable design and optimization of a direct contact humidification-dehumidification desalination unit. Desalination 2012, 293, 69–77. [Google Scholar] [CrossRef]
- Muthusamy, C.; Srithar, K. Energy and exergy analysis for a humidification–dehumidification desalination system integrated with multiple inserts. Desalination 2015, 367, 49–59. [Google Scholar] [CrossRef]
- Nikooei, E.; Yeung, N.A.; Zhang, X.; Goulas, K.; Abbasi, B.; Dyall, A.; Abbasi, B. Controlled dehumidification to extract clean water from a multicomponent gaseous mixture of organic contaminants. J. Water Process Eng. 2021, 43, 102229. [Google Scholar] [CrossRef]
- Aspen Plus: Chemical Engineering Applications, 1st ed.; Wiley: Hoboken, NJ, USA, 2016; ISBN 978-1-119-13123-6. Available online: http://eu.wiley.com/WileyCDA/WileyTitle/productCd-1119131235.html (accessed on 7 November 2022).
- Wood, K.R.; Liu, Y.A.; Yu, Y. Design, Simulation and Optimization of Adsorptive and Chromatographic Separations: A Hands-On Approach; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2018; ISBN 978-3-527-81501-2. [Google Scholar]
- Amer, E.H.; Kotb, H.; Mostafa, G.H.; El-Ghalban, A.R. Theoretical and experimental investigation of humidification–dehumidification desalination unit. Desalination 2009, 249, 949–959. [Google Scholar] [CrossRef]



| Stream | Temperature | Pressure | Component | Mass Fraction (%) | Mass Flow (kg/h) |
|---|---|---|---|---|---|
| FEED (Wastewater) | 25 °C | 1 bar | H2O (H2O) | 98.3 | 30,000 |
| Maleic Anhydride (MA) | 0.07 | ||||
| Acetic Acid (ACETIC) | 0.15 | ||||
| Acrylic Acid (ACRYLIC) | 0.15 | ||||
| Dibutyl Phthalate (DBP) | 0.07 | ||||
| Phthalic Anhydride (PHTHALIC) | 0.02 | ||||
| Phthalic Acid (PHTHA-AC) | 0.25 | ||||
| Maleic Acid (MALEI) | 0.8 | ||||
| Fumaric Acid (FUMAR) | 0.08 | ||||
| N-butanol (BUTANOL) | 0.11 | ||||
| AIR | 25 °C | 1 bar | Nitrogen (N2) | 79 (Noble gases included) | 15,000 |
| Oxygen (O2) | 21 | ||||
| H2O (Freshwater) | 25 °C | 1 bar | H2O (H2O) | 100 | 30,000 |
| Humidifier (HUM) | |
|---|---|
| Number of stages | 5 |
| Feed streams convention | FEED: Stage 1 (Above-Stage) |
| HEATED: Stage 1 (Above-Stage) | |
| AIR: Stage 5 (On-Stage) | |
| Pressure | 1 bar |
| Packed Height | 15 m |
| Column Diameter | 1.2 m |
| Dehumidifier (DEHUM) | |
| Number of stages | 10 |
| Feed streams convention | H2O: Stage 1 (Above-Stage) |
| COOLED: Stage 1 (Above-Stage) | |
| HUM-VAP: Stage 10 (On-Stage) | |
| Pressure | 1 bar |
| Packed Height | 15 m |
| Column Diameter | 1.2 m |
| Packing | Material | Dimension | Specific Surface Area (m2/m3) |
|---|---|---|---|
| Pall Ring | Plastic | 0.625 in/16 mm | 364 |
| Temperature | Pressure | Utility Input | ||
|---|---|---|---|---|
| HEATER | 75 °C | 1 bar | Medium pressure steam (MS) | 4 MPa |
| COOLER | 22 °C | 1 bar | Cooling water (CW) | Inlet Temperature 7 °C |
| Outlet Temperature 12 °C | ||||
| 1 bar | ||||
| Stream | FEED | HUM-VAP | DE-VAP |
|---|---|---|---|
| pH | 3.02 | 4.15 | 5.74 |
| Stream | FEED (kg/h) | FEED (cum/h) | FEED (kg/cum) | DE-LIQ (kg/h) | DE-LIQ (cum/h) | DE-LIQ (kg/cum) |
|---|---|---|---|---|---|---|
| / | 30,000 | 29.98 | 1000.70 | 163,075.17 | 163.96 | 994.63 |
| Component | Chemical Formula | C | H | O | Molar Mass | Conversion Factor |
|---|---|---|---|---|---|---|
| MA | C4H2O3 | 4 | 2 | 3 | 98.06 | 0.979 |
| ACETIC | CH3COOH | 2 | 4 | 2 | 60.052 | 1.066 |
| ACRYLIC | C3H4O2 | 3 | 4 | 2 | 72.063 | 1.332 |
| DBP | C16H22O4 | 16 | 22 | 4 | 278.344 | 2.242 |
| PHTHALIC | C8H4O3 | 8 | 4 | 3 | 148.12 | 1.620 |
| PHTHA-AC | C8H6O4 | 8 | 6 | 4 | 166.13 | 1.445 |
| MALEI | C4H4O4 | 4 | 4 | 4 | 116.07 | 0.827 |
| FUMAR | C4H4O4 | 4 | 4 | 4 | 116.07 | 0.827 |
| BUTANOL | C4H10O | 4 | 10 | 1 | 74.14 | 2.590 |
| FEED | ||||
|---|---|---|---|---|
| Component | Mass Flow | COD Emission | ||
| kg/h | kg/d | t/a | mg/L | |
| MA | 21.00 | 420.00 | 126.00 | 686 |
| ACETIC | 43.22 | 864.34 | 259.30 | 1536 |
| ACRYLIC | 45.00 | 900.00 | 270.00 | 2000 |
| DBP | 21.00 | 420.00 | 126.00 | 1570 |
| PHTHALIC | 6.00 | 120.00 | 36.00 | 324 |
| PHTHA-AC | 75.00 | 1500.00 | 450.00 | 3614 |
| MALEI | 240.00 | 4800.00 | 1440.00 | 6621 |
| FUMAR | 24.00 | 480.00 | 144.00 | 662 |
| BUTANOL | 33.00 | 660.00 | 198.00 | 2851 |
| Total | 508.22 | 1404.34 | 421.30 | 19,864.72 |
| DE-LIQ | ||||
|---|---|---|---|---|
| Component | Mass Flow | COD Emission | ||
| kg/h | kg/d | t/a | mg/L | |
| MA | 0.28 | 5.55 | 1.66 | 2 |
| ACETIC | 28.00 | 560.01 | 168.00 | 182 |
| ACRYLIC | 22.16 | 443.19 | 132.96 | 180 |
| DBP | 1.60 | 31.97 | 9.59 | 22 |
| PHTHALIC | N/A | N/A | N/A | N/A |
| PHTHA-AC | N/A | N/A | N/A | N/A |
| MALEI | 7.59 | 151.73 | 45.52 | 38 |
| FUMAR | N/A | N/A | N/A | N/A |
| BUTANOL | 109.58 | 2191.50 | 657.45 | 1,731 |
| Total | 169.20 | 3383.94 | 1015.18 | 2154.59 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, Y.; Ma, L.; Bai, P. Study of Organic Acid Pollutant Removal Efficient in Treatment of Industrial Wastewater with HDH Process Using ASPEN Modelling. Water 2022, 14, 3681. https://doi.org/10.3390/w14223681
Zeng Y, Ma L, Bai P. Study of Organic Acid Pollutant Removal Efficient in Treatment of Industrial Wastewater with HDH Process Using ASPEN Modelling. Water. 2022; 14(22):3681. https://doi.org/10.3390/w14223681
Chicago/Turabian StyleZeng, Ying, Limin Ma, and Peng Bai. 2022. "Study of Organic Acid Pollutant Removal Efficient in Treatment of Industrial Wastewater with HDH Process Using ASPEN Modelling" Water 14, no. 22: 3681. https://doi.org/10.3390/w14223681
APA StyleZeng, Y., Ma, L., & Bai, P. (2022). Study of Organic Acid Pollutant Removal Efficient in Treatment of Industrial Wastewater with HDH Process Using ASPEN Modelling. Water, 14(22), 3681. https://doi.org/10.3390/w14223681








