Arsenate Removal from the Groundwater Employing Maghemite Nanoparticles
Abstract
:Highlights
- AsV removal in synthetic water representing elemental composition equivalent to contam-inated groundwater of Ballia;
- The prepared γ-Fe2O3 nanoparticles (MNPs) show efficient capabilities for AsV removal;
- Inter-parametric interactions revealed that adsorption also occurred through the formation of surface complexes;
- Surface complexation modeling (SCMs) shows the involvement of singlet (FeOH−0.5) and triplet (Fe3O−0.5) species in adsorption;
- Weak electrostatic interactions are responsible for the removal process.
Abstract
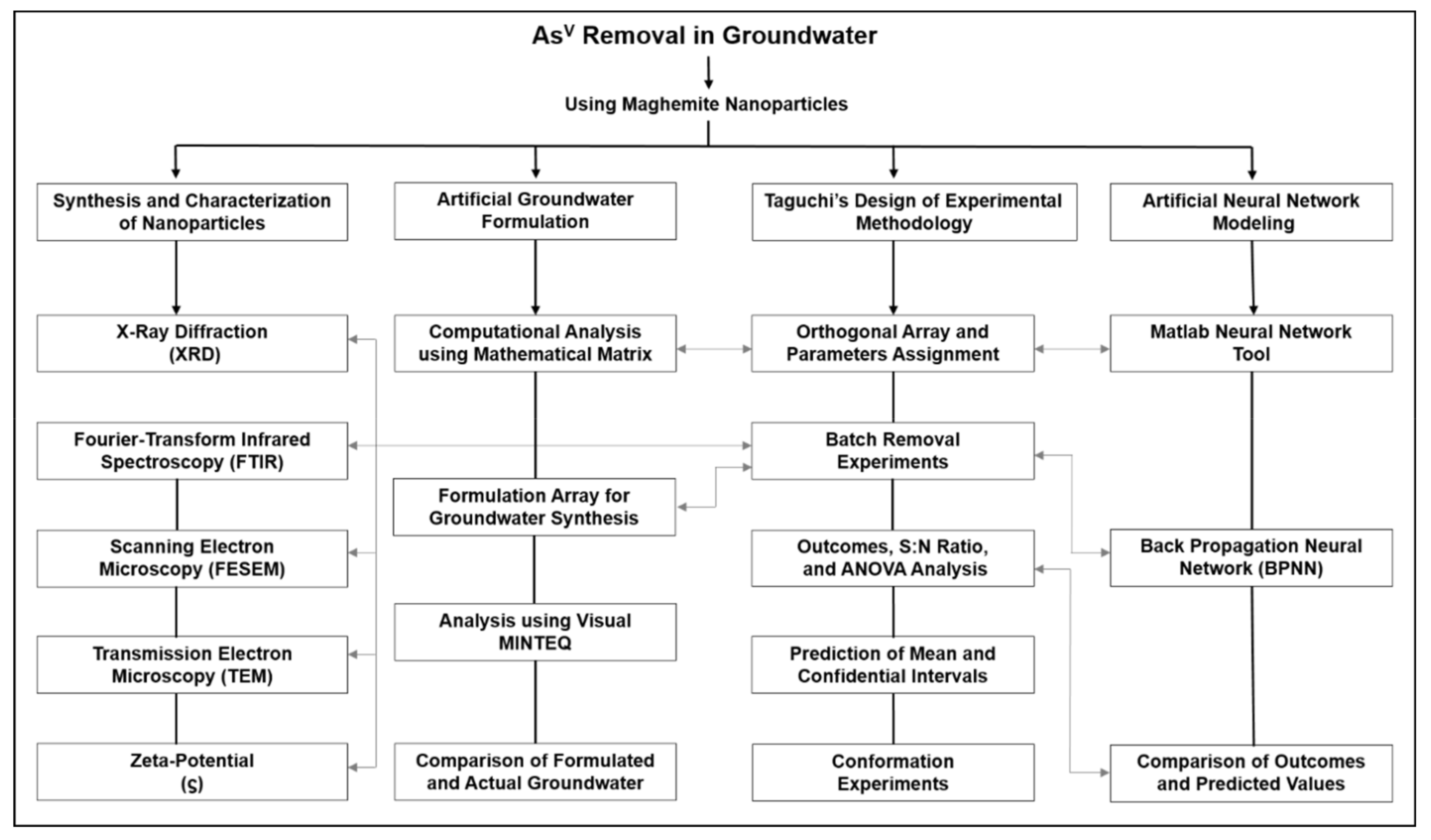
1. Introduction
2. Experimental Procedure
2.1. Materials and Reagents
2.2. Instrumentations and Equipment
2.3. Synthesis of Nanostructured Maghemite
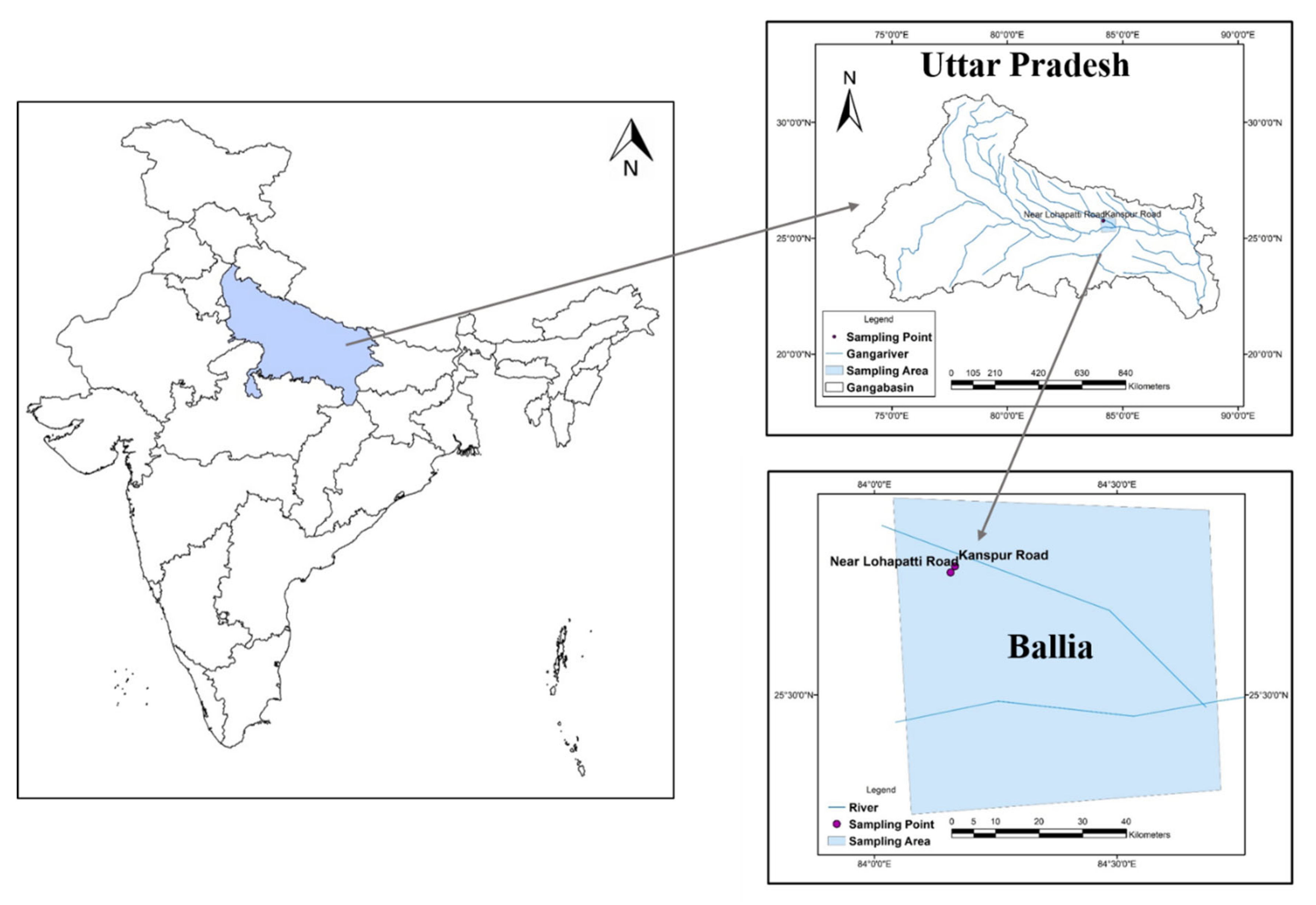
2.4. Sample Collection and Analysis
2.5. Formulation of Synthetic Water
2.6. Taguchi’s Design of Experimental Methodology
2.6.1. Design of Experiment (Phase 1)
2.6.2. Orthogonal Array (OA) and Assignment of Parameters
- Total DOF required for experiment ≤ total DOF of OA.
2.6.3. Batch Removal Experiments (Phase 2)
2.6.4. Evaluation of Outputs and Performance Assessment (Phase 3)
2.6.5. Signal to Noise Ratio (S/N)
2.6.6. Prediction of Average Adsorption Capacity
2.6.7. Determination of Confidential Interval
- (i)
- The CI lies around the estimated adsorption capacity of a treatment condition used in the conformation experiments to verify predictions. It is designated as (confidence interval for a group of sample);
- (ii)
- The CI lies around the estimated adsorption capacity of a treatment condition predicted from the experiment. It is designated as (population’s confidence interval).
2.7. Confirmation Experiments (Phase 4)
2.8. Modelling of Adsorption Processes
| Surface Species | ≡FeOH | ≡Fe3O | log K | Ref. | |||
|---|---|---|---|---|---|---|---|
| ≡FeOH−0.5 | 1 | 0 | 0 | 0 | 0 | 0 | [94] |
| ≡Fe3O−0.5 | 0 | 1 | 0 | 0 | 0 | 0 | [94] |
| ≡FeOH−0.5–Ca+ | 1 | 0 | 0.32 | 1.68 | 0 | 3.17 | [95,96] |
| ≡FeOH−0.5–CaCO3- | 1 | 0 | 0.6 | −1.6 | 0 | 15.55 | [97] |
| ≡FeOH−0.5–CaHCO3 | 1 | 0 | 0.6 | −0.6 | 0 | 24.15 | [97] |
| ≡FeOH−0.5–HNO3 | 1 | 0 | 1.00 | −1.00 | 0 | 7.42 | [96,98] |
| ≡Fe3O−0.5–HNO3 | 0 | 1 | 1.00 | −1.00 | 0 | 7.42 | [96] |
| 2(≡FeOH−0.5)–AsO2Ca | 2 | 0 | 0.60 | 0.40 | 0 | 36.04 | [99] |
| 2(≡FeOH−0.5)–AsO2HCa | 2 | 0 | 0.60 | 1.40 | 0 | 43.44 | [99] |
| 2(≡FeOH−0.5)–Mg2+ | 2 | 0 | 0.71 | 1.29 | 0 | 4.89 | [100] |
| 2(≡FeOH−0.5)–PO2Ca | 2 | 0 | 0.60 | 0.40 | 0 | 38.57 | [92] |
| 2(≡FeOH−0.5)–PO2HCa | 2 | 0 | 0.60 | 1.40 | 0 | 46.02 | [92] |
| 2(≡FeOH−0.5)–PO2− | 2 | 0 | 0.46 | −1.46 | 0 | 27.59 | [99] |
| 2(≡FeOH−0.5)–POOH | 2 | 0 | 0.63 | −0.63 | 0 | 32.89 | [99] |
| 2(≡FeOH−0.5)–Si(OH)2 | 2 | 0 | −0.29 | −0.29 | 0 | 5.85 | [95,100] |
| 2(≡FeOH−0.5)–SiOHOSi3O3(OH)9 | 2 | 0 | −0.29 | −0.29 | 0 | 13.98 | [100] |
| 2(≡FeOH−0.5)–SiO2HSi3O3+1(OH)9-1 | 2 | 0 | −0.29 | −1.29 | 0 | 7.47 | [100] |
| 2(≡FeOH−0.5)–ZnOH | 2 | 0 | 0.50 | 0.50 | 0 | −1.43 | [101] |
| Modeling parameters | |||||||
| Model Type | CD-MUSIC and 2-pk TPM | ||||||
| Number of site types | 2 | ||||||
| γ-Fe2O3 nanoparticle concentration (g L−1) | 0.03 | ||||||
| Specific surface area (m2 g−1) | 59.80 | This study | |||||
| Surface site density (sites nm−2) | 51.13 | This study | |||||
| Inner capacitance (F m−2) | 1.0 | [93] | |||||
| Outer capacitance (F m−2) | 0.2 | [93] | |||||
| pH range | 7–9 | ||||||
| Calculated qe (µg mg−1-Fe) | S:N Ratio (dB) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Runs | Trial 1, A | Trial 2, B | Trial 3, A.B | Trial 4, A.B | Trial 5, C | Trial 6, A.C | Trial 7, A.C | Trial 8, B.C | Trial 9, D | Trial 10, E | Trial 11, B.C | Trial 12, F | Trial 13, G | R1 | R2 | R3 | |
| 1. | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1.21 | 1.22 | 1.23 | 1.75 |
| 2. | 1 | 1 | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 0.45 | 0.46 | 0.45 | −6.89 |
| 3. | 1 | 1 | 1 | 1 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 0.38 | 0.35 | 0.44 | −8.31 |
| 4. | 1 | 2 | 2 | 2 | 1 | 1 | 1 | 2 | 2 | 2 | 3 | 3 | 3 | 0.18 | 0.17 | 0.19 | −14.89 |
| 5. | 1 | 2 | 2 | 2 | 2 | 2 | 2 | 3 | 3 | 3 | 1 | 1 | 1 | 0.74 | 0.79 | 0.85 | −2.03 |
| 6. | 1 | 2 | 2 | 2 | 3 | 3 | 3 | 1 | 1 | 1 | 2 | 2 | 2 | 0.69 | 0.63 | 0.69 | −3.49 |
| 7. | 1 | 3 | 3 | 3 | 1 | 1 | 1 | 3 | 3 | 3 | 2 | 2 | 2 | 0.28 | 0.26 | 0.29 | −11.09 |
| 8. | 1 | 3 | 3 | 3 | 2 | 2 | 2 | 1 | 1 | 1 | 3 | 3 | 3 | 0.38 | 0.38 | 0.40 | −8.26 |
| 9. | 1 | 3 | 3 | 3 | 3 | 3 | 3 | 2 | 2 | 2 | 1 | 1 | 1 | 0.59 | 0.57 | 0.59 | −4.71 |
| 10. | 2 | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 1.07 | 1.04 | 1.11 | 0.62 |
| 11. | 2 | 1 | 2 | 3 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 0.62 | 0.58 | 0.64 | −4.25 |
| 12. | 2 | 1 | 2 | 3 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 4.01 | 3.79 | 4.00 | 11.88 |
| 13. | 2 | 2 | 3 | 1 | 1 | 2 | 3 | 2 | 3 | 1 | 3 | 1 | 2 | 0.59 | 0.55 | 0.57 | −4.86 |
| 14. | 2 | 2 | 3 | 1 | 2 | 3 | 1 | 3 | 1 | 2 | 1 | 2 | 3 | 2.03 | 2.01 | 2.05 | 6.15 |
| 15. | 2 | 2 | 3 | 1 | 3 | 1 | 2 | 1 | 2 | 3 | 2 | 3 | 1 | 0.61 | 0.64 | 0.63 | −4.04 |
| 16. | 2 | 3 | 1 | 2 | 1 | 2 | 3 | 3 | 1 | 2 | 2 | 3 | 1 | 0.52 | 0.40 | 0.49 | −6.70 |
| 17. | 2 | 3 | 1 | 2 | 2 | 3 | 1 | 1 | 2 | 3 | 3 | 1 | 2 | 1.12 | 1.13 | 1.15 | 1.09 |
| 18. | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 2 | 3 | 1 | 1 | 2 | 3 | 0.84 | 0.93 | 0.80 | −1.40 |
| 19. | 3 | 1 | 3 | 2 | 1 | 3 | 2 | 1 | 3 | 2 | 1 | 3 | 2 | 1.38 | 1.40 | 1.43 | 2.94 |
| 20. | 3 | 1 | 3 | 2 | 2 | 1 | 3 | 2 | 1 | 3 | 2 | 1 | 3 | 5.93 | 5.52 | 6.12 | 15.33 |
| 21. | 3 | 1 | 3 | 2 | 3 | 2 | 1 | 3 | 2 | 1 | 3 | 2 | 1 | 1.38 | 1.36 | 1.41 | 2.83 |
| 22. | 3 | 2 | 1 | 3 | 1 | 3 | 2 | 2 | 1 | 3 | 3 | 2 | 1 | 3.08 | 2.93 | 3.03 | 9.57 |
| 23. | 3 | 2 | 1 | 3 | 2 | 1 | 3 | 3 | 2 | 1 | 1 | 3 | 2 | 0.99 | 1.06 | 0.96 | 0.02 |
| 24. | 3 | 2 | 1 | 3 | 3 | 2 | 1 | 1 | 3 | 2 | 2 | 1 | 3 | 0.37 | 0.30 | 0.33 | −9.70 |
| 25. | 3 | 3 | 2 | 1 | 1 | 3 | 2 | 3 | 2 | 1 | 2 | 1 | 3 | 1.46 | 1.74 | 1.52 | 3.87 |
| 26. | 3 | 3 | 2 | 1 | 2 | 1 | 3 | 1 | 3 | 2 | 3 | 2 | 1 | 1.61 | 1.36 | 1.48 | 3.38 |
| 27. | 3 | 3 | 2 | 1 | 3 | 2 | 1 | 2 | 1 | 3 | 1 | 3 | 2 | 2.10 | 2.03 | 2.33 | 6.62 |
| Total | 34.64 | 33.62 | 35.20 | ||||||||||||||
| Mean | 1.28 | ||||||||||||||||
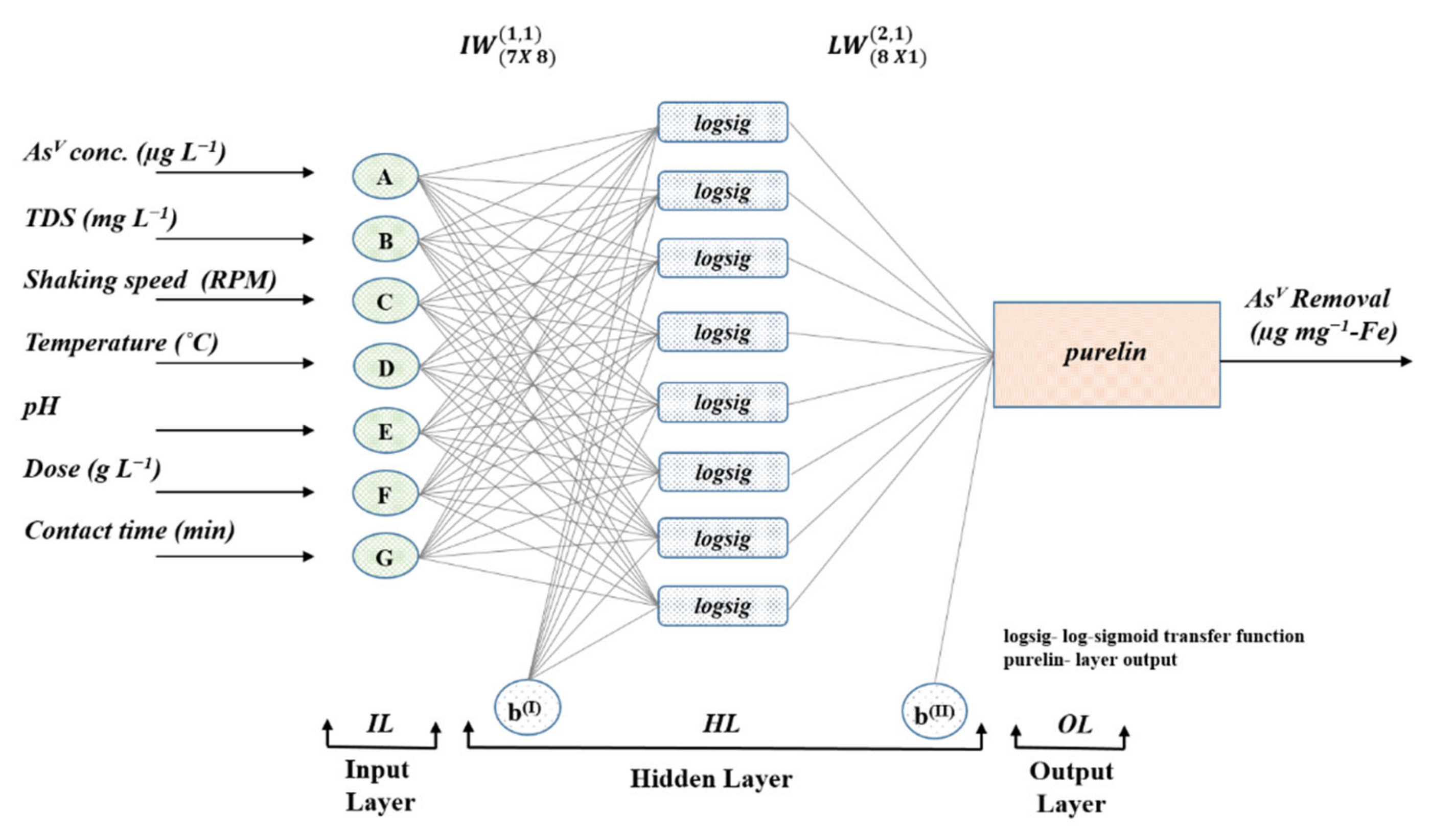
2.9. Artificial Neural Network (ANN) for Predictive Modeling
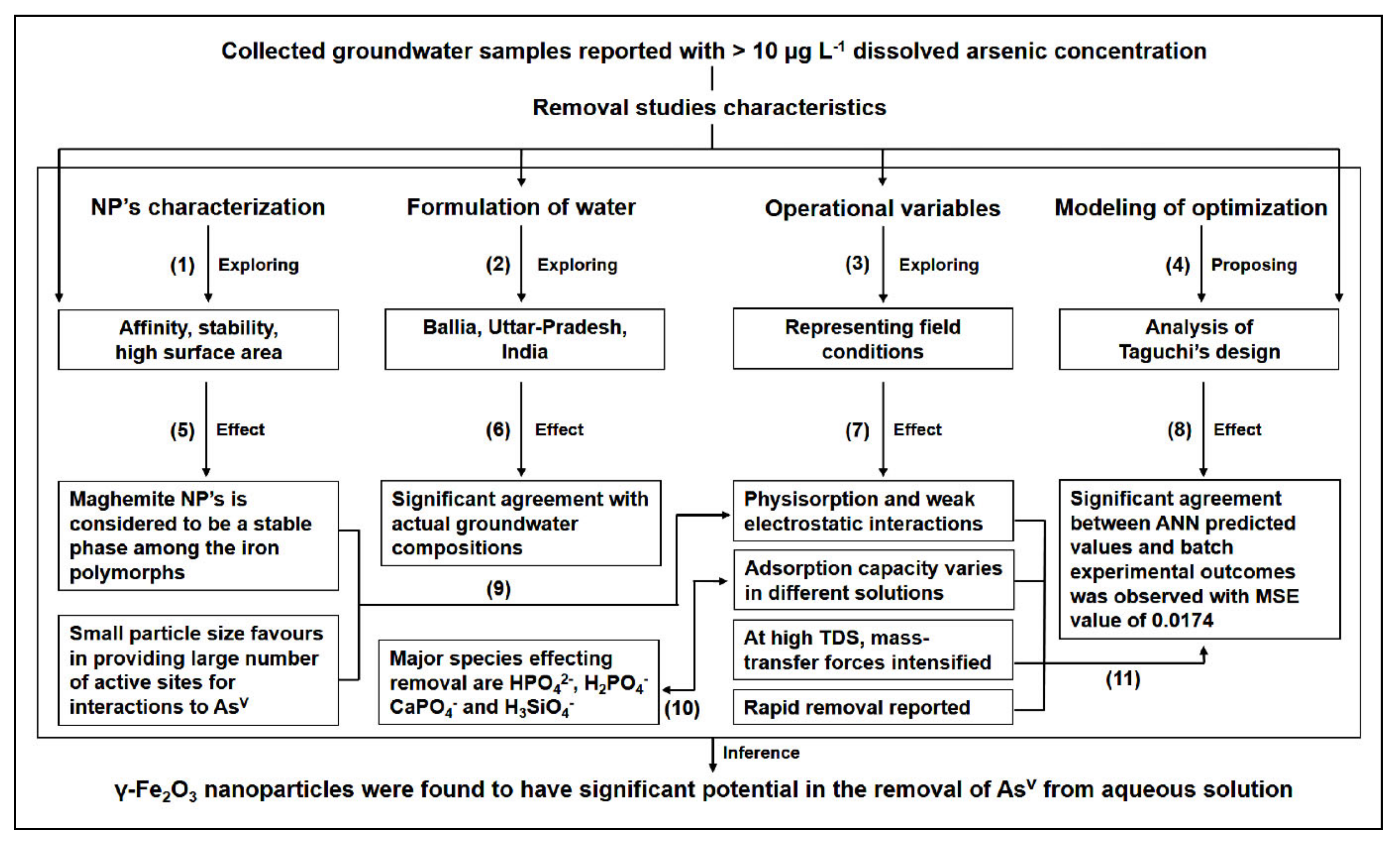
3. Results and Discussion
3.1. Characterization of Maghemite Nano-Particles
3.2. Elemental Characterization of Groundwater and Its Formulation
3.3. Characteristics of Arsenic Removal in the Multi-Ionic System
3.3.1. Effects of Process Parameters: Shaking Speed, Temperature, and Contact Time
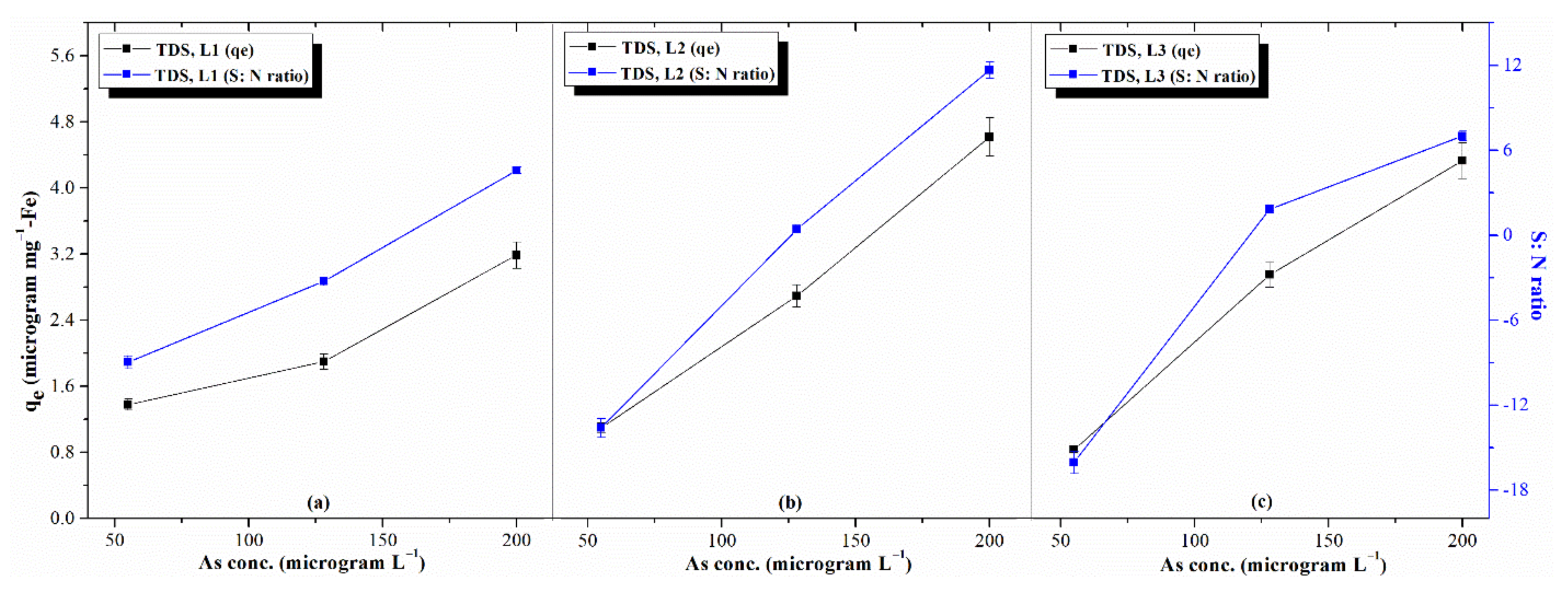
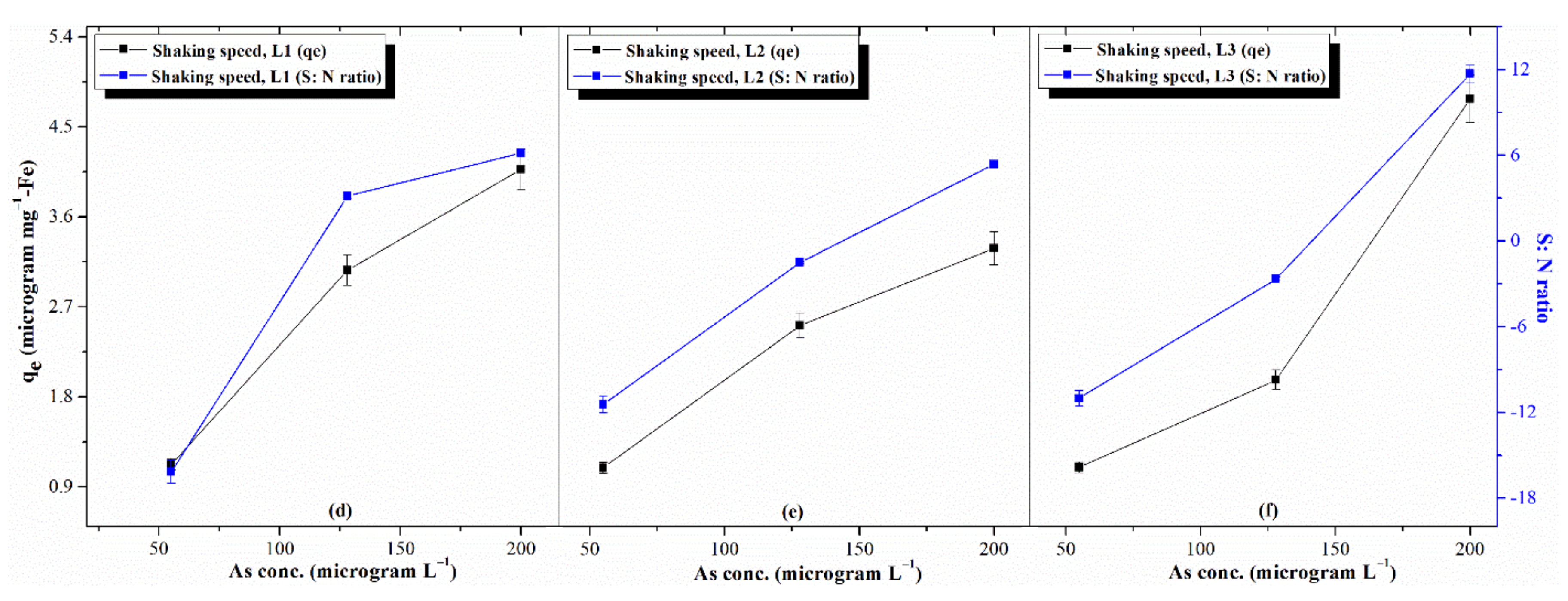
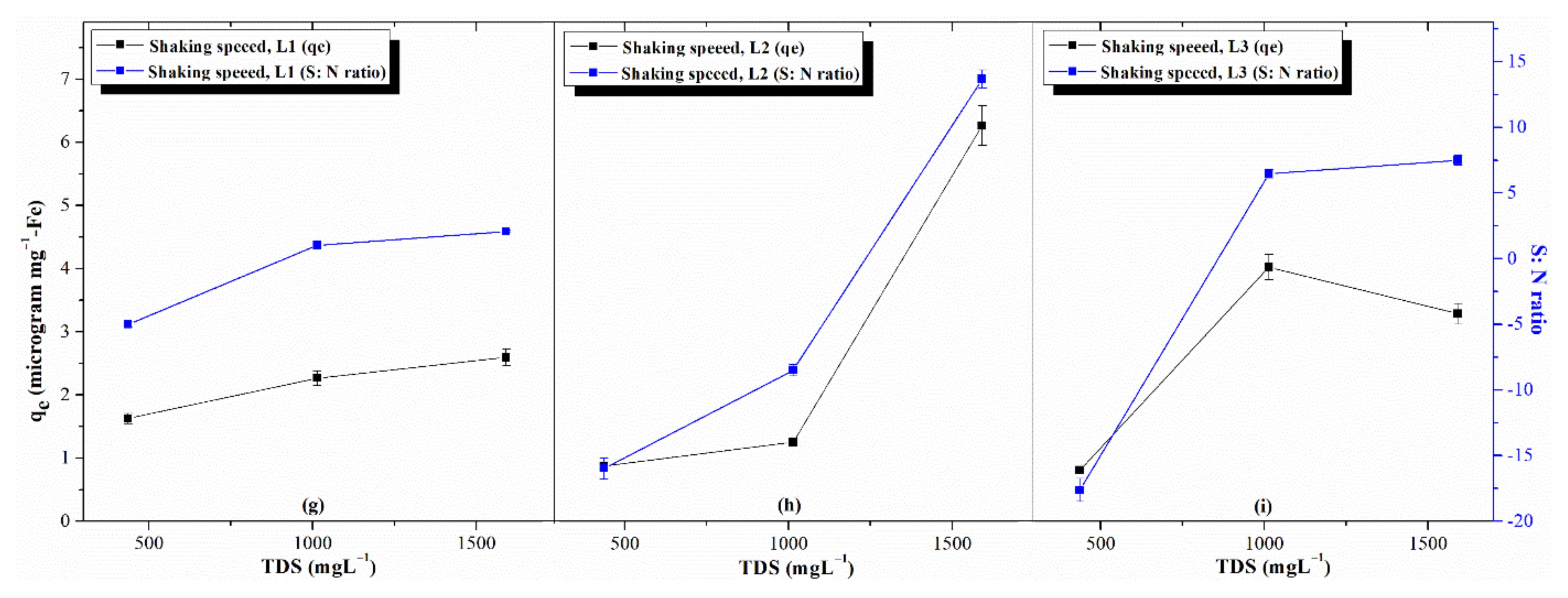
3.3.2. Effects of Process Parameters: AsV Concentration, TDS, and pH
3.4. Analysis of Inter-Parametric Interactions
3.5. Selection of Optimal Levels and Estimation of Response Characteristics
- The first level of concentration of AsV ions () = 0.6
- The third level of concentration of AsV ions () = 2.0
- The first level of concentration of TDS () = 1.8
- The third level of concentration of TDS () = 1.0
- A second level of shaking speed () = 1.5
- The first level of temperature () = 2.2
- The third level of pH () = 1.7
- The first level of dose concentration () = 1.8
- The third level of contact time () = 1.4
3.6. Confirmation Experiments
3.7. Analysis Using Visual MINTEQ
4. Surface Complexation Models (SCMs) for Adsorption Behavior
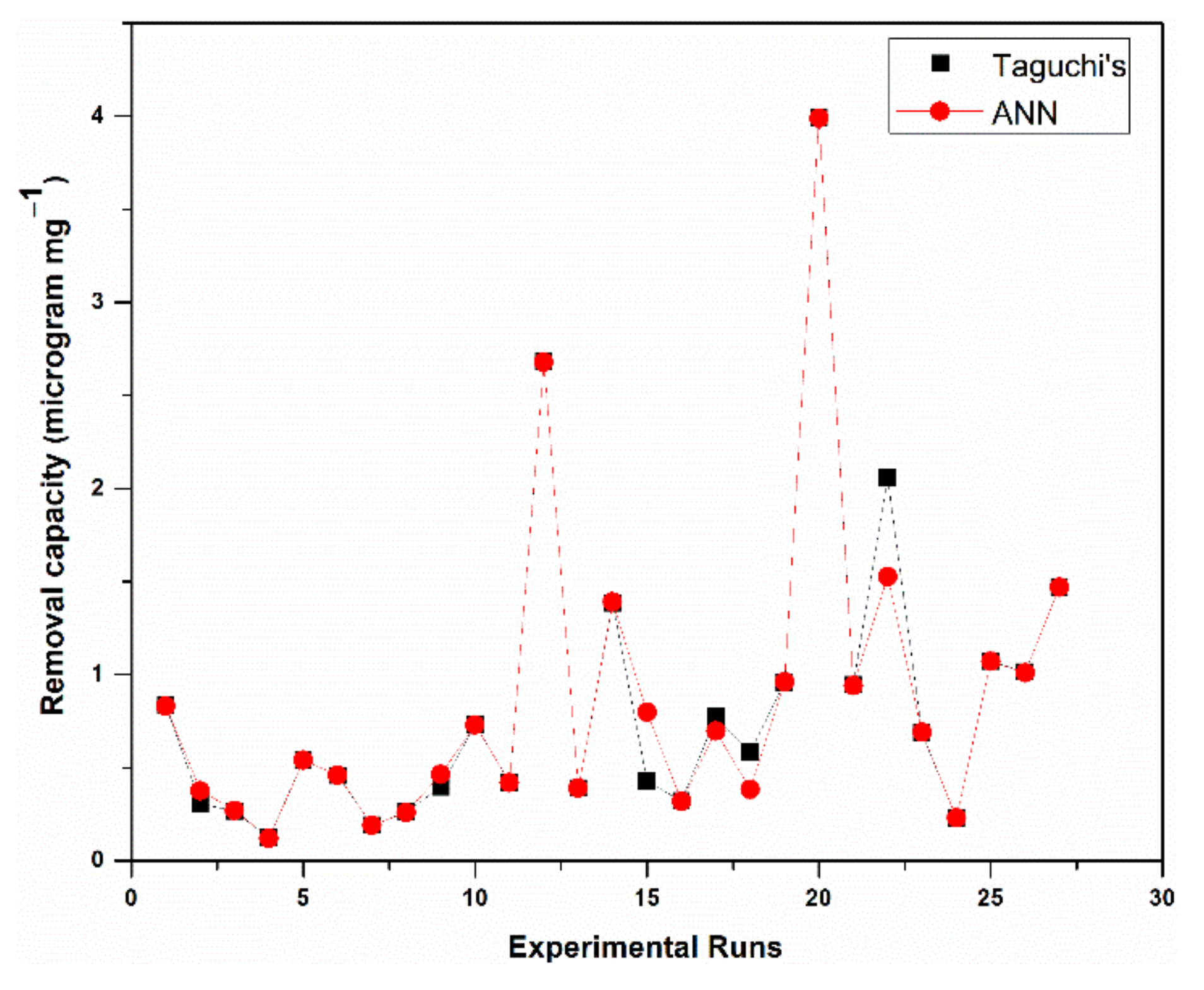
5. ANN Predictions
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Exposer to Arsenic: A Major Public Health Concern. Report World Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Raposo, J.C.; Sanz, J.; Zuloaga, O.; Olazabal, M.A.; Madariaga, J.M. Validation of the Thermodynamic Model of Inorganic Arsenic in Non Polluted River Waters of the Basque Country (Spain). Talanta 2004, 63, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Vahter, M. Health Effects of Early Life Exposure to Arsenic. Basic Clin. Pharmacol. Toxicol. 2008, 102, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Mohammed Abdul, K.S.; Jayasinghe, S.S.; Chandana, E.P.S.; Jayasumana, C.; De Silva, P.M.C.S. Arsenic and Human Health Effects: A Review. Environ. Toxicol. Pharmacol. 2015, 40, 828–846. [Google Scholar] [CrossRef]
- Smedley, P.L.; Kinniburgh, D.G.; Macdonald, D.M.J.; Nicolli, H.B.; Barros, A.J.; Tullio, J.O.; Pearce, J.M.; Alonso, M.S. Arsenic Associations in Sediments from the Loess Aquifer of La Pampa, Argentina. Appl. Geochemistry 2005, 20, 989–1016. [Google Scholar] [CrossRef]
- Ahmed, M.F. An Overview of Arsenic Removal Technologies in Bangladesh and India. In Proceedings of the BUET-UNU International Workshop on Technologies for Arsenic Removal from Drinking Water, Dhaka, Bangladesh, 5–7 May 2001. [Google Scholar]
- Ciminelli, V.S.T.; Gasparon, M.; Ng, J.C.; Silva, G.C.; Caldeira, C.L. Dietary Arsenic Exposure in Brazil: The Contribution of Rice and Beans. Chemosphere 2017, 168, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Bondu, R.; Cloutier, V.; Rosa, E.; Benzaazoua, M. Mobility and Speciation of Geogenic Arsenic in Bedrock Groundwater from the Canadian Shield in Western Quebec, Canada. Sci. Total Environ. 2017, 574, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Polya, D.; Gault, A.G.; Diebe, N.; Feldman, P.; Rosenboom, J.; Gilligan, E.; Cooke, D. Arsenic Hazard in Shallow Cambodian Groundwaters. Mineral. Mag. 2005, 69, 807–823. [Google Scholar] [CrossRef]
- Pino, P.; Iglesias, V.; Garreaud, R.; Cortés, S.; Canals, M.; Folch, W.; Burgos, S.; Levy, K.; Naeher, L.P.; Steenland, K. Chile Confronts Its Environmental Health Future After 25 Years of Accelerated Growth. Ann. Glob. Health 2015, 81, 354–367. [Google Scholar] [CrossRef]
- Adomako, E.E.; Williams, P.N.; Deacon, C.; Meharg, A.A. Inorganic Arsenic and Trace Elements in Ghanaian Grain Staples. Environ. Pollut. 2011, 159, 2435–2442. [Google Scholar] [CrossRef]
- Sugár, É.; Mihucz, V.G.; Záray, G. Determination of Arsenic in Drinking Water and Several Food Items. Elelmiszervizsgalati Kozlemenyek 2014, 60, 163–176. [Google Scholar]
- Navarro, O.; González, J.; Júnez-Ferreira, H.E.; Bautista, C.F.; Cardona, A. Correlation of Arsenic and Fluoride in the Groundwater for Human Consumption in a Semiarid Region of Mexico. Procedia Eng. 2017, 186, 333–340. [Google Scholar] [CrossRef]
- Berg, M.; Stengel, C.; Trang, P.; Hungviet, P.; Sampson, M.; Leng, M.; Samreth, S.; Fredericks, D. Magnitude of Arsenic Pollution in the Mekong and Red River Deltas—Cambodia and Vietnam. Sci. Total Environ. 2007, 372, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Bora, S.S.; Yadav, R.N.S.; Barooah, M. A Metagenomic Approach to Decipher the Indigenous Microbial Communities of Arsenic Contaminated Groundwater of Assam. Genomics Data 2017, 12, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Chakraborti, D.; Rahman, M.M.; Ahamed, S.; Dutta, R.N.; Pati, S.; Mukherjee, S.C. Arsenic Groundwater Contamination and Its Health Effects in Patna District (Capital of Bihar) in the Middle Ganga Plain, India. Chemosphere 2016, 152, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.S.; Sahu, B.L.; Dahariya, N.S.; Bhatia, A.; Patel, R.K.; Matini, L.; Sracek, O.; Bhattacharya, P. Groundwater Arsenic and Fluoride in Rajnandgaon District, Chhattisgarh, Northeastern India. Appl. Water Sci. 2017, 7, 1817–1826. [Google Scholar] [CrossRef] [Green Version]
- Rana, A.; Bhardwaj, S.K.; Thakur, M.; Verma, S. Assessment of Heavy Metals in Surface and Ground Water Sources under Different Land Uses in Mid-Hills of Himachal Pradesh. Int. J. Bioresour. Stress Manag. 2016, 7, 461–465. [Google Scholar] [CrossRef]
- Alam, M.O.; Shaikh, W.A.; Chakraborty, S.; Avishek, K.; Bhattacharya, T. Groundwater Arsenic Contamination and Potential Health Risk Assessment of Gangetic Plains of Jharkhand, India. Expo. Health 2016, 8, 125–142. [Google Scholar] [CrossRef]
- Sharma, S.; Kaur, J.; Nagpal, A.K.; Kaur, I. Quantitative Assessment of Possible Human Health Risk Associated with Consumption of Arsenic Contaminated Groundwater and Wheat Grains from Ropar Wetand and Its Environs. Environ. Monit. Assess. 2016, 188, 506. [Google Scholar] [CrossRef]
- Chandrashekhar, A.K.; Chandrasekharam, D.; Farooq, S.H. Contamination and Mobilization of Arsenic in the Soil and Groundwater and Its Influence on the Irrigated Crops, Manipur Valley, India. Environ. Earth Sci. 2016, 75, 1–15. [Google Scholar] [CrossRef]
- Olea, R.A.; Raju, N.J.; Egozcue, J.J.; Pawlowsky-Glahn, V.; Singh, S. Advancements in Hydrochemistry Mapping: Methods and Application to Groundwater Arsenic and Iron Concentrations in Varanasi, Uttar Pradesh, India. Stoch. Environ. Res. Risk Assess. 2018, 32, 241–259. [Google Scholar] [CrossRef] [Green Version]
- Bhowmick, S.; Pramanik, S.; Singh, P.; Mondal, P.; Chatterjee, D.; Nriagu, J. Arsenic in Groundwater of West Bengal, India: A Review of Human Health Risks and Assessment of Possible Intervention Options. Sci. Total Environ. 2018, 612, 148–169. [Google Scholar] [CrossRef] [PubMed]
- Podgorski, J.; Wu, R.; Chakravorty, B.; Polya, D.A. Groundwater Arsenic Distribution in India by Machine Learning Geospatial Modeling. Int. J. Environ. Res. Public Heal. 2020, 17, 7119. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Sarkar, S.; Chakraborty, M.; Duttagupta, S.; Bhattacharya, A.; Saha, D.; Bhattacharya, P.; Mitra, A.; Gupta, S. Occurrence, Predictors and Hazards of Elevated Groundwater Arsenic across India through Field Observations and Regional-Scale AI-Based Modeling. Sci. Total Environ. 2021, 759, 143511. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum. WHO Library Cataloguing-in-Publication Data. 2017. Available online: http://apps.who.int/iris/bitstream/handle/10665/254637/9789241549950\eng.pdf;jsessionid=2E1B7E42B16F03AB88E253B119976D63?sequence=1 (accessed on 21 June 2021).
- Office of Water, U.S. Environmental Protection Agency. Edition of the Drinking Water Standards and Health Advisories Tables. EPA 822-F-18-001 Off. 2018. Available online: https://www.epa.gov/system/files/documents/2022-01/dwtable2018.pdf (accessed on 12 March 2022).
- Meng, X.; Korfiatis, G.P.; Christodoulatos, C.; Bang, S. Treatment of Arsenic in Bangladesh Well Water Using a Household Co-Precipitation and Filtration System. Water Res. 2001, 35, 2805–2810. [Google Scholar] [CrossRef]
- Leupin, O.X.; Hug, S.J. Oxidation and Removal of Arsenic (III) from Aerated Groundwater by Filtration through Sand and Zero-Valent Iron. Water Res. 2005, 39, 1729–1740. [Google Scholar] [CrossRef]
- Greenleaf, J.E.; Lin, J.C.; Sengupta, A.K. Two Novel Applications of Ion Exchange Fibers: Arsenic Removal and Chemical-Free Softening of Hard Water. Environ. Prog. 2006, 25, 300–311. [Google Scholar] [CrossRef]
- Ning, R. Arsenic Removal by Reverse Osmosis. Desalination 2002, 143, 237–241. [Google Scholar] [CrossRef]
- Danish, M.I.; Qazi, I.A.; Zeb, A.; Habib, A.; Awan, M.A.; Khan, Z. Arsenic Removal from Aqueous Solution Using Pure and Metal-Doped Titania Nanoparticles Coated on Glass Beads: Adsorption and Column Studies. J. Nanomater. 2013, 2013, 1–17. [Google Scholar] [CrossRef]
- Jain, H.; Kumar, A.; Verma, A.K.; Wadhwa, S.; Rajput, V.D.; Minkina, T.; Garg, M.C. Treatment of Textile Industry Wastewater by Using High-Performance Forward Osmosis Membrane Tailored with Alpha-Manganese Dioxide Nanoparticles for Fertigation. Environ. Sci. Pollut. Res. 2022, 29, 80032–80043. [Google Scholar] [CrossRef]
- Özlem Kocabaş-Atakli, Z.; Yürüm, Y. Synthesis and Characterization of Anatase Nanoadsorbent and Application in Removal of Lead, Copper and Arsenic from Water. Chem. Eng. J. 2013, 225, 625–635. [Google Scholar] [CrossRef]
- Ungureanu, G.; Santos, S.; Boaventura, R.; Botelho, C. Arsenic and Antimony in Water and Wastewater: Overview of Removal Techniques with Special Reference to Latest Advances in Adsorption. J. Environ. Manage. 2015, 151, 326–342. [Google Scholar] [CrossRef] [PubMed]
- Fierro, V.; Muñiz, G.; Gonzalez-Sánchez, G.; Ballinas, M.L.; Celzard, A. Arsenic Removal by Iron-Doped Activated Carbons Prepared by Ferric Chloride Forced Hydrolysis. J. Hazard. Mater. 2009, 168, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, W.; Chen, L.; Huang, X.; Liu, J. Preparation and Evaluation of Magnetic Nanoparticles Impregnated Chitosan Beads for Arsenic Removal from Water. Chem. Eng. J. 2014, 251, 25–34. [Google Scholar] [CrossRef]
- Yürüm, A.; Kocabaş-Atakli, Z.Ö.; Sezen, M.; Semiat, R.; Yürüm, Y. Fast Deposition of Porous Iron Oxide on Activated Carbon by Microwave Heating and Arsenic (V) Removal from Water. Chem. Eng. J. 2014, 242, 321–332. [Google Scholar] [CrossRef]
- Xu, Z.; Li, Q.; Gao, S.; Shang, J.K. As(III) Removal by Hydrous Titanium Dioxide Prepared from One-Step Hydrolysis of Aqueous TiCl4 Solution. Water Res. 2010, 44, 5713–5721. [Google Scholar] [CrossRef]
- Önnby, L.; Kumar, P.S.; Sigfridsson, K.G.V.; Wendt, O.F.; Carlson, S.; Kirsebom, H. Improved Arsenic(III) Adsorption by Al2O3 Nanoparticles and H2O2: Evidence of Oxidation to Arsenic(V) from X-Ray Absorption Spectroscopy. Chemosphere 2014, 113, 151–157. [Google Scholar] [CrossRef]
- Önnby, L.; Svensson, C.; Mbundi, L.; Busquets, R.; Cundy, A.; Kirsebom, H. γ-Al2O3-Based Nanocomposite Adsorbents for Arsenic(V) Removal: Assessing Performance, Toxicity and Particle Leakage. Sci. Total Environ. 2014, 473–474, 207–214. [Google Scholar] [CrossRef]
- Reddy, K.J.; McDonald, K.J.; King, H. A Novel Arsenic Removal Process for Water Using Cupric Oxide Nanoparticles. J. Colloid Interface Sci. 2013, 397, 96–102. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Li, Q.; Gao, S.; Shang, J.K. High Efficient As(III) Removal by Self-Assembled Zinc Oxide Micro-Tubes Synthesized by a Simple Precipitation Process. J. Mater. Sci. 2011, 46, 5851–5858. [Google Scholar] [CrossRef]
- Piquette, A.; Cannon, C.; Apblett, A.W. Remediation of Arsenic and Lead with Nanocrystalline Zinc Sulfide. Nanotechnology 2012, 23, 294014. [Google Scholar] [CrossRef]
- Cui, H.; Li, Q.; Gao, S.; Shang, J.K. Strong Adsorption of Arsenic Species by Amorphous Zirconium Oxide Nanoparticles. J. Ind. Eng. Chem. 2012, 18, 1418–1427. [Google Scholar] [CrossRef]
- Luo, X.; Wang, C.; Wang, L.; Deng, F.; Luo, S.; Tu, X.; Au, C. Nanocomposites of Graphene Oxide-Hydrated Zirconium Oxide for Simultaneous Removal of As(III) and As(V) from Water. Chem. Eng. J. 2013, 220, 98–106. [Google Scholar] [CrossRef]
- Deng, S.; Li, Z.; Huang, J.; Yu, G. Preparation, Characterization and Application of a Ce-Ti Oxide Adsorbent for Enhanced Removal of Arsenate from Water. J. Hazard. Mater. 2010, 179, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Li, Q.; Gao, S.; Shang, J.K. Exceptional Arsenic Adsorption Performance of Hydrous Cerium Oxide Nanoparticles: Part B. Integration with Silica Monoliths and Dynamic Treatment. Chem. Eng. J. 2012, 185–186, 136–143. [Google Scholar] [CrossRef]
- Kanel, S.R.; Grenèche, J.-M.; Choi, H. Arsenic(V) Removal from Groundwater Using Nano Scale Zero-Valent Iron as a Colloidal Reactive Barrier Material. Environ. Sci. Technol. 2006, 40, 2045–2050. [Google Scholar] [CrossRef]
- Morgada, M.E.; Levy, I.K.; Salomone, V.N.; Farías, S.S.; López, G.; Litter, M.I. Arsenic (V) Removal with Nanoparticulate Zerovalent Iron: Effect of UV Light and Humic Acids. Catal. Today 2009, 143, 261–268. [Google Scholar] [CrossRef]
- Jiang, W.; Chen, X.; Niu, Y.; Pan, B. Spherical Polystyrene-Supported Nano-Fe3O4 of High Capacity and Low-Field Separation for Arsenate Removal from Water. J. Hazard. Mater. 2012, 243, 319–325. [Google Scholar] [CrossRef]
- Vitela-Rodriguez, A.V.; Rangel-Mendez, J.R. Arsenic Removal by Modified Activated Carbons with Iron Hydro(Oxide) Nanoparticles. J. Environ. Manage. 2013, 114, 225–231. [Google Scholar] [CrossRef]
- Vaughan, R.L.; Reed, B.E. Modeling As(V) Removal by a Iron Oxide Impregnated Activated Carbon Using the Surface Complexation Approach. Water Res. 2005, 39, 1005–1014. [Google Scholar] [CrossRef]
- Sun, X.; Hu, C.; Hu, X.; Qu, J.; Yang, M. Characterization and Adsorption Performance of Zr-Doped Akaganéite for Efficient Arsenic Removal. J. Chem. Technol. Biotechnol. 2013, 88, 629–635. [Google Scholar] [CrossRef]
- Saiz, J.; Bringas, E.; Ortiz, I. Functionalized Magnetic Nanoparticles as New Adsorption Materials for Arsenic Removal from Polluted Waters. J. Chem. Technol. Biotechnol. 2014, 89, 909–918. [Google Scholar] [CrossRef]
- Jin, Y.; Liu, F.; Tong, M.; Hou, Y. Removal of Arsenate by Cetyltrimethylammonium Bromide Modified Magnetic Nanoparticles. J. Hazard. Mater. 2012, 227–228, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Morin, G.; Ona-Nguema, G.; Juillot, F.; Calas, G.; Brown, G.E. Distinctive Arsenic(V) Trapping Modes by Magnetite Nanoparticles Induced by Different Sorption Processes. Environ. Sci. Technol. 2011, 45, 7258–7266. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Xu, H.; Xie, Q.; Chen, J.; Ji, J.; Lu, H. Characteristics and Genesis of Maghemite in Chinese Loess and Paleosols: Mechanism for Magnetic Susceptibility Enhancement in Paleosols. Earth Planet. Sci. Lett. 2005, 240, 790–802. [Google Scholar] [CrossRef]
- Alijani, H.; Shariatinia, Z. Effective Aqueous Arsenic Removal Using Zero Valent Iron Doped MWCNT Synthesized by in Situ CVD Method Using Natural α-Fe2O3 as a Precursor. Chemosphere 2017, 171, 502–511. [Google Scholar] [CrossRef]
- Liu, T.; Yang, Y.; Wang, Z.L.; Sun, Y. Remediation of Arsenic(III) from Aqueous Solutions Using Improved Nanoscale Zero-Valent Iron on Pumice. Chem. Eng. J. 2016, 288, 739–744. [Google Scholar] [CrossRef]
- Wu, X.; Tan, X.; Yang, S.; Wen, T.; Guo, H.; Wang, X.; Xu, A. Coexistence of Adsorption and Coagulation Processes of Both Arsenate and NOM from Contaminated Groundwater by Nanocrystallined Mg/Al Layered Double Hydroxides. Water Res. 2013, 47, 4159–4168. [Google Scholar] [CrossRef]
- Zhang, W.; Fu, J.; Zhang, G.; Zhang, X. Enhanced Arsenate Removal by Novel Fe-La Composite (Hydr)Oxides Synthesized via Coprecipitation. Chem. Eng. J. 2014, 251, 69–79. [Google Scholar] [CrossRef]
- Li, Z.; Deng, S.; Yu, G.; Huang, J.; Lim, V.C. As(V) and As(III) Removal from Water by a Ce-Ti Oxide Adsorbent: Behavior and Mechanism. Chem. Eng. J. 2010, 161, 106–113. [Google Scholar] [CrossRef]
- He, J.; Matsuura, T.; Chen, J.P. A Novel Zr-Based Nanoparticle-Embedded PSF Blend Hollow Fiber Membrane for Treatment of Arsenate Contaminated Water: Material Development, Adsorption and Filtration Studies, and Characterization. J. Memb. Sci. 2014, 452, 433–445. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, H.; Yu, M.; Emmanuelawati, I.; Zou, J.; Yuan, Z.; Yu, C. High-Content, Well-Dispersed γ-Fe2O3 Nanoparticles Encapsulated in Macroporous Silica with Superior Arsenic Removal Performance. Adv. Funct. Mater. 2014, 24, 1354–1363. [Google Scholar] [CrossRef]
- Poguberović, S.S.; Krčmar, D.M.; Maletić, S.P.; Kónya, Z.; Pilipović, D.D.T.; Kerkez, D.V.; Rončević, S.D. Removal of As(III) and Cr(VI) from Aqueous Solutions Using “Green” Zero-Valent Iron Nanoparticles Produced by Oak, Mulberry and Cherry Leaf Extracts. Ecol. Eng. 2016, 90, 42–49. [Google Scholar] [CrossRef]
- Kumar, A.; Joshi, H.; Kumar, A. Remediation of Arsenic by Metal/ Metal Oxide Based Nanocomposites/ Nanohybrids: Contamination Scenario in Groundwater, Practical Challenges, and Future Perspectives. Sep. Purif. Rev. 2021, 50, 283–314. [Google Scholar] [CrossRef]
- McBride, M.B. Chemisoption and Precipitation Reactions. In Handbook of Soil Science; Sumner, M.E., Ed.; CRC Press: Boca Raton, FL, USA, 1999; pp. B285–B286. [Google Scholar]
- Lin, S.; Lu, D.; Liu, Z. Removal of Arsenic Contaminants with Magnetic γ-Fe2O3 Nanoparticles. Chem. Eng. J. 2012, 211–212, 46–52. [Google Scholar] [CrossRef]
- Terlecka, E. Arsenic Speciation Analysis in Water Samples: A Review of the Hyphenated Techniques. Environ. Monit. Assess. 2005, 107, 259–284. [Google Scholar] [CrossRef]
- Smedley, P.L.; Kinniburgh, D.G. A Review of the Source, Behaviour and Distribution of Arsenic in Natural Waters. Appl. Geochemistry 2002, 17, 517–568. [Google Scholar] [CrossRef] [Green Version]
- Ghaedi, A.M.; Ghaedi, M.; Vafaei, A.; Iravani, N.; Keshavarz, M.; Rad, M.; Tyagi, I.; Agarwal, S.; Gupta, V.K. Adsorption of Copper (II) Using Modified Activated Carbon Prepared from Pomegranate Wood: Optimization by Bee Algorithm and Response Surface Methodology. J. Mol. Liq. 2015, 206, 195–206. [Google Scholar] [CrossRef]
- Asfaram, A.; Ghaedi, M.; Hajati, S.; Goudarzi, A.; Bazrafshan, A.A. Simultaneous Ultrasound-Assisted Ternary Adsorption of Dyes onto Copper-Doped Zinc Sulfide Nanoparticles Loaded on Activated Carbon: Optimization by Response Surface Methodology. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 145, 203–212. [Google Scholar] [CrossRef]
- Asfaram, A.; Ghaedi, M.; Agarwal, S.; Tyagi, I.; Gupta, V.K. Removal of Basic Dye Auramine-O by ZnS:Cu Nanoparticles Loaded on Activated Carbon: Optimization of Parameters Using Response Surface Methodology with Central Composite Design. RSC Adv. 2015, 5, 18438–18450. [Google Scholar] [CrossRef]
- Ghaedi, M.; Mazaheri, H.; Khodadoust, S.; Hajati, S.; Purkait, M.K. Application of Central Composite Design for Simultaneous Removal of Methylene Blue and Pb2+ions by Walnut Wood Activated Carbon. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 135, 479–490. [Google Scholar] [CrossRef]
- Fakhri, A. Application of Response Surface Methodology to Optimize the Process Variables for Fluoride Ion Removal Using Maghemite Nanoparticles. J. Saudi Chem. Soc. 2014, 18, 340–347. [Google Scholar] [CrossRef]
- Ahmadi, A.; Heidarzadeh, S.; Mokhtari, A.R.; Darezereshki, E.; Harouni, H.A. Optimization of Heavy Metal Removal from Aqueous Solutions by Maghemite (γ-Fe2O3) Nanoparticles Using Response Surface Methodology. J. Geochemical Explor. 2014, 147, 151–158. [Google Scholar] [CrossRef]
- Rezaei, S.; Rahpeima, S.; Esmaili, J.; Javanbakht, V. Optimization by Response Surface Methodology of the Adsorption of Anionic Dye on Superparamagnetic Clay/Maghemite Nanocomposite. Russ. J. Appl. Chem. 2021, 94, 533–548. [Google Scholar] [CrossRef]
- Kumar, A.; Joshi, H.; Kumar, A. An Approach of Multi-Variate Statistical Design (Taguchi) and Numerical Tool (COMSOL) in Exploring the Arsenic Sequestration Potential of γ-Fe2O3NPs in Groundwater of Ballia District, Uttar-Pradesh, India. In Proceedings of the AGU Fall Meeting Abstracts, San Francisco, CA, USA, 9–13 December 2019; p. GH23B-1227. [Google Scholar]
- Kaloti, M.; Kumar, A.; Navani, N.K. Synthesis of Glucose-Mediated Ag–γ-Fe2O3 Multifunctional Nanocomposites in Aqueous Medium—A Kinetic Analysis of Their Catalytic Activity for 4-Nitrophenol Reduction. Green Chem. 2015, 17, 4786–4799. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater Part 1000; APHA: Washington, DC, USA; AWWA: Denver, CO, USA; WEF: Alexandria, WV, USA, 1999. [Google Scholar]
- Part 4000 Inorganic Nonmetallic Constituents. In Standard Methods for the Examination of Water and Wastewater; Water Environment Federation: Alexandria, VA, USA, 1999.
- Smičiklas, I.; Onjia, A.; Raičević, S.; Janaćković, D. Authors’ Response to Comments on “Factors Influencing the Removal of Divalent Cations by Hydroxyapatite”. J. Hazard. Mater. 2009, 168, 560–562. [Google Scholar] [CrossRef]
- Tang, W.; Su, Y.; Li, Q.; Gao, S.; Shang, J.K. Superparamagnetic Magnesium Ferrite Nanoadsorbent for Effective Arsenic (III, V) Removal and Easy Magnetic Separation. Water Res. 2013, 47, 3624–3634. [Google Scholar] [CrossRef]
- Kilianová, M.; Prucek, R.; Filip, J.; Kolařík, J.; Kvítek, L.; Panáček, A.; Tuček, J.; Zbořil, R. Remarkable Efficiency of Ultrafine Superparamagnetic Iron(III) Oxide Nanoparticles toward Arsenate Removal from Aqueous Environment. Chemosphere 2013, 93, 2690–2697. [Google Scholar] [CrossRef]
- Goswami, A.; Raul, P.K.; Purkait, M.K. Arsenic Adsorption Using Copper (II) Oxide Nanoparticles. Chem. Eng. Res. Des. 2012, 90, 1387–1396. [Google Scholar] [CrossRef]
- Engin, A.B.; Özdemir, Ö.; Turan, M.; Turan, A.Z. Color Removal from Textile Dyebath Effluents in a Zeolite Fixed Bed Reactor: Determination of Optimum Process Conditions Using Taguchi Method. J. Hazard. Mater. 2008, 159, 348–353. [Google Scholar] [CrossRef]
- Srivastava, V.C.; Mall, I.D.; Mishra, I.M. Multicomponent Adsorption Study of Metal Ions onto Bagasse Fly Ash Using Taguchi’s Design of Experimental Methodology. Ind. Eng. Chem. Res. 2007, 46, 5697–5706. [Google Scholar] [CrossRef]
- Googerdchian, F.; Moheb, A.; Emadi, R.; Asgari, M. Optimization of Pb(II) Ions Adsorption on Nanohydroxyapatite Adsorbents by Applying Taguchi Method. J. Hazard. Mater. 2018, 349, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Varala, S.; Kumari, A.; Dharanija, B.; Bhargava, S.K.; Parthasarathy, R.; Satyavathi, B. Removal of Thorium (IV) from Aqueous Solutions by Deoiled Karanja Seed Cake: Optimization Using Taguchi Method, Equilibrium, Kinetic and Thermodynamic Studies. J. Environ. Chem. Eng. 2016, 4, 405–417. [Google Scholar] [CrossRef]
- Gustafsson, J.P. Visual MINTEQ 3.1; KTH Sweden: Stockholm, Sweden, 2016; Available online: https://vminteq.lwr.kth.se/visual-minteq-ver-3-1/ (accessed on 18 December 2021).
- Hiemstra, T.; Venema, P.; Van Riemsdijk, W.H. Intrinsic Proton Affinity of Reactive Surface Groups of Metal (Hydr)Oxides: The Bond Valence Principle. J. Colloid Interface Sci. 1996, 184, 680–692. [Google Scholar] [CrossRef] [PubMed]
- Garcell, L.; Morales, M.P.; Andres-Vergés, M.; Tartaj, P.; Serna, C.J. Interfacial and Rheological Characteristics of Maghemite Aqueous Suspensions. J. Colloid Interface Sci. 1998, 205, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Schröder, D.; Shaik, S.; Schwarz, H. Two-State Reactivity as a New Concept in Organometallic Chemistry. Acc. Chem. Res. 2000, 33, 139–145. [Google Scholar] [CrossRef]
- Smith, S.D.; Edwards, M. The Influence of Silica and Calcium on Arsenate Sorption to Oxide Surfaces. J. Water Supply Res. Technol. AQUA 2005, 54, 201–211. [Google Scholar] [CrossRef]
- Restrepo, A.; Ibarguen, C.; Flórez, E.; Acelas, N.Y.; Hadad, C. Adsorption of Nitrate and Bicarbonate on Fe-(Hydr)Oxide. Inorg. Chem. 2017, 56, 5455–5464. [Google Scholar] [CrossRef]
- Saalfield, S.L.; Bostick, B.C. Synergistic Effect of Calcium and Bicarbonate in Enhancing Arsenate Release from Ferrihydrite. Geochim. Cosmochim. Acta 2010, 74, 5171–5186. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A. ScienceDirect—Water Research: Removal of Arsenic from Contaminated Water Sources by Sorption onto Iron-Oxide-Coated Polymeric Materials. Water Res. 2002, 36, 5141–5155. [Google Scholar] [CrossRef]
- Deng, Y.; Li, Y.; Li, X.; Sun, Y.; Ma, J.; Lei, M.; Weng, L. Influence of Calcium and Phosphate on PH Dependency of Arsenite and Arsenate Adsorption to Goethite. Chemosphere 2018, 199, 617–624. [Google Scholar] [CrossRef]
- Jolsterå, R.; Gunneriusson, L.; Forsling, W. Adsorption and Surface Complex Modeling of Silicates on Maghemite in Aqueous Suspensions. J. Colloid Interface Sci. 2010, 342, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Bhattacharya, J. Removal of Cu ( II ), Zn ( II ) and Pb ( II ) from Water Using Microwave-Assisted Synthesized Maghemite Nanotubes. Chem. Eng. J. 2012, 211–212, 493–500. [Google Scholar] [CrossRef]
- Mandal, S.; Mahapatra, S.S.; Sahu, M.K.; Patel, R.K. Artificial Neural Network Modelling of As(III) Removal from Water by Novel Hybrid Material. Process Saf. Environ. Prot. 2014, 93, 1–16. [Google Scholar] [CrossRef]
- Roy, P.; Mondal, N.K.; Das, K. Modeling of the Adsorptive Removal of Arsenic: A Statistical Approach. J. Environ. Chem. Eng. 2014, 2, 585–597. [Google Scholar] [CrossRef]
- Kumar, A.; Agarwal, S.; Garg, M.C.; Joshi, H. Use of Artificial Intelligence for Optimizing Biosorption of Textile Wastewater Using Agricultural Waste. Environ. Technol. 2021, 1–35. [Google Scholar] [CrossRef]
- Roca, A.G.; Marco, J.F.; Del Puerto Morales, M.; Serna, C.J. Effect of Nature and Particle Size on Properties of Uniform Magnetite and Maghemite Nanoparticles. J. Phys. Chem. C 2007, 111, 18577–18584. [Google Scholar] [CrossRef]
- Nadeem, M.; Mahmood, A.; Shahid, S.A.; Shah, S.S.; Khalid, A.M.; McKay, G. Sorption of Lead from Aqueous Solution by Chemically Modified Carbon Adsorbents. J. Hazard. Mater. 2006, 138, 604–613. [Google Scholar] [CrossRef]
- Zhou, J.; Yang, S.; Yu, J.; Shu, Z. Novel Hollow Microspheres of Hierarchical Zinc-Aluminum Layered Double Hydroxides and Their Enhanced Adsorption Capacity for Phosphate in Water. J. Hazard. Mater. 2011, 192, 1114–1121. [Google Scholar] [CrossRef]
- Shan, C.; Tong, M. Efficient Removal of Trace Arsenite through Oxidation and Adsorption by Magnetic Nanoparticles Modified with Fe-Mn Binary Oxide. Water Res. 2013, 47, 3411–3421. [Google Scholar] [CrossRef]
- Trainor, T.P.; Chaka, A.M.; Eng, P.J.; Newville, M.; Waychunas, G.A.; Catalano, J.G.; Brown, G.E. Structure and Reactivity of the Hydrated Hematite (0 0 0 1) Surface. Surf. Sci. 2004, 573, 204–224. [Google Scholar] [CrossRef]
- Bargar, J.R.; Towle, S.N.; Brown, G.E.; Parks, G.A. XAFS and Bond-Valence Determination of the Structures and Compositions of Surface Functional Groups and Pb(II) and Co(II) Sorption Products on Single-Crystal α-Al2O3. J. Colloid Interface Sci. 1997, 185, 473–492. [Google Scholar] [CrossRef] [PubMed]



| Location I | Location II | |||||||
|---|---|---|---|---|---|---|---|---|
| Element | Actual Groundwater Conc. (mg L−1) | Calculated Values from the Matrix | Measured Synthetic Water Conc. (mg L−1) | Diff. (%) | Actual Groundwater Conc. (mg L−1) | Calculated Values from the Matrix | Artificial Water Conc. (mg L−1) | Diff. (%) |
| TDS (gravimetric) | 435.9 | - | 411.5 | 5.6 | 1590.6 | - | 1405.7 | 11.6 |
| TDS (calculated by ions) | 337.4 | 261.4 | 310.6 | 7.9 | 1446 | 1171.1 | 1321.5 | 8.6 |
| Chloride | 132 | 67.1 | 112.0 | 15.2 | 435 | 222.7 | 346 | 20.5 |
| Sodium | 33 | 23.1 | 28.0 | 15.2 | 169 | 111.2 | 141 | 16.6 |
| Bicarbonate | 27 | 27.0 | 28.0 | −3.7 | 115 | 114.8 | 113 | 1.7 |
| Potassium | 15 | 13.8 | 13.3 | 11.3 | 95 | 91.6 | 93 | 2.1 |
| Fluoride | 1.5 | 1.5 | 1.5 | 1.3 | 3 | 2.94 | 3.16 | −5.3 |
| Nitrate | 20 | 20.0 | 21.2 | −6.0 | 140 | 139.8 | 139 | 0.7 |
| Calcium | 29 | 28.9 | 26.4 | 9.0 | 185 | 185.3 | 181.5 | 1.9 |
| Zinc | 2 | 1.9 | 1.9 | 5.0 | 4 | 4.43 | 4.1 | −2.5 |
| Magnesium | 27 | 27.0 | 24.5 | 9.3 | 33 | 33.2 | 32.4 | 1.8 |
| Sulfate | 30 | 30.1 | 32.4 | −8.0 | 220 | 219.4 | 221.1 | −0.5 |
| Silica | 18 | 18.1 | 18.6 | −3.3 | 42 | 41.4 | 42.3 | −0.7 |
| Manganese | 2 | 1.9 | 1.9 | 7.5 | 3 | 3.4 | 3.2 | −6.7 |
| Phosphates | 0.91 | 1.1 | 1.0 | −6.6 | 2 | 1.06 | 1.75 | 12.5 |
| Arsenate * | 0.055 | - | 0.057 | −3.6 | 0.2 | - | 0.201 | −0.5 |
| Parameters | Level 1 | Level 2 | Level 3 | |
|---|---|---|---|---|
| A | Arsenate concentration (µg L−1) | 55 | 128 | 200 |
| B | TDS (mg L−1) | 436 | 1014 | 1591 |
| C | Shaking speed (rpm) | 100 | 170 | 240 |
| D | Temperature (°C) | 10 | 20 | 30 |
| E | pH (pH unit) | 7 | 8 | 9 |
| F | Dose (g L−1) | 0.05 | 0.10 | 0.15 |
| G | Contact time (min) | 2 | 53 | 104 |
| Parameter | Unit | Method of Test | DL * | PL * | Location I | Location II |
|---|---|---|---|---|---|---|
| pH | pH unit | pH probe | 6.5–8.5 | NR | 7.45 ± 0.1 | 7.34 ± 0.1 |
| Electrical conductivity | µS cm−1 | EC probe | - | - | 341 ± 6 | 1524 ± 7 |
| TDS | mg L−1 | TDS probe | 500 | 2000 | 435.9 ± 4 | 1590.6 ± 4 |
| Total hardness | mg L−1 | Titrimetric | 300 | 600 | 160 ± 4 | 424 ± 4 |
| Alkalinity | mg L−1 | Titrimetric | 200 | 600 | 86 ± 3 | 222 ± 4 |
| Bicarbonate (as CaCO3) | mg L−1 | Titrimetric | - | - | 27 ± 2 | 115.3 ± 1.5 |
| Phosphate (as PO43−) | mg L−1 | Spectrophotometric | 0.91 ± 0.1 | 2.02 ± 0.1 | ||
| Silica (as H4SiO4) | mg L−1 | Spectrophotometric | 18.4 ± 2 | 42.4 ± 1 | ||
| Sodium (as Na+) | mg L−1 | IC | - | - | 32.4 ± 1 | 169.4 ± 1 |
| Potassium (as K+) | mg L−1 | IC | - | - | 15.3 ± 0.2 | 94.7 ± 1 |
| Fluoride (as F−) | mg L−1 | IC | 1.0 | 1.5 | 1.54 ± 0.1 | 3.04 ± 0.1 |
| Chloride (as Cl−) | mg L−1 | IC | 250 | 1000 | 132.4 ± 1 | 435.2 ± 1 |
| Calcium (as Ca2+) | mg L−1 | IC | 75 | 200 | 28.9 ± 0.4 | 185.1 ± 1 |
| Magnesium (as Mg2+) | mg L−1 | IC | 30 | 100 | 26.8 ± 1 | 32.4 ± 1 |
| Sulphate (as SO42−) | mg L−1 | IC | 200 | 400 | 30.4 ± 1.2 | 220.4 ± 2 |
| Nitrate | mg L−1 | IC | 45 | NR | 20.0 ± 0.4 | 140.0 ± 1 |
| Iron | mg L−1 | ICP-MS | 0.3 | 1.0 | 0.53 ± 0.02 | 0.37 ± 0.03 |
| Manganese | mg L−1 | ICP-MS | 0.1 | 0.3 | 2 ± 0.1 | 3.03 ± 0.1 |
| Copper | mg L−1 | ICP-MS | 0.05 | 1.5 | ND | ND |
| Lead | mg L−1 | ICP-MS | 0.01 | NR | ND | ND |
| Cadmium | mg L−1 | ICP-MS | 0.003 | NR | ND | ND |
| Zinc | mg L−1 | ICP-MS | 5 | 15 | 2 ± 0.1 | 3.96 ± 0.3 |
| Arsenic (as Total) | µg L−1 | MP-AES | 10 | 50 | 54 ± 2 | 200 ± 1 |
| Raw Data, Average Value | Main Effects, Raw Data | S:N Ratio (dB), Average Value | S:N Ratio (dB), Main Effects | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Factor | L1 | L2 | L3 | L2–L1 | L3–L2 | L1 | L2 | L3 | L2–L1 | L3–L2 |
| A | 0.6 | 1.3 | 2.0 | 0.7 | 0.8 | −6.4 | −0.2 | 3.9 | 6.3 | 4.0 |
| B | 1.8 | 1.0 | 1.0 | −0.8 | Nil | 1.8 | −2.6 | −1.9 | −4.4 | 0.7 |
| C | 1.1 | 1.5 | 1.2 | 0.4 | −0.3 | −2.1 | 0.5 | −1.2 | 2.6 | −1.7 |
| D | 2.2 | 0.9 | 0.8 | −1.3 | −0.2 | 3.7 | −2.5 | −3.9 | −6.1 | −1.5 |
| E | 0.9 | 1.2 | 1.7 | 0.3 | 0.5 | −1.5 | −2.1 | 0.9 | −0.5 | 2.9 |
| F | 1.8 | 1.3 | 0.8 | −0.5 | −0.5 | 1.4 | Nil | −4.1 | −1.4 | −4.1 |
| G | 1.1 | 1.3 | 1.4 | 0.2 | 0.1 | −0.5 | −0.4 | −1.8 | 0.1 | −1.4 |
| A.B | 2.2 | 2.8 | 2.7 | 0.7 | −0.1 | −2.5 | −0.5 | −2.4 | 2.0 | −1.9 |
| A.C | 2.8 | 2.3 | 2.6 | −0.5 | 0.3 | −2.3 | −2.5 | −0.7 | −0.2 | 1.9 |
| B.C | 2.2 | 2.8 | 2.7 | 0.6 | −0.1 | −0.6 | −3.6 | −1.2 | −3.0 | 2.4 |
| Raw Data | S:N ratio | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | Sum of Square | DOF a | Mean Square | F Value | % Contrib. | Sum of Square | DOF a | Mean Square | F Value | % Contrib. |
| A | 29.3 | 2 | 14.6 | 292.7 | 23.66 | 485.67 | 2 | 242.84 | 933.98 | 39.7 |
| B | 11.7 | 2 | 5.9 | 117.4 | 9.41 | 98.66 | 2 | 49.33 | 189.73 | 8.1 |
| C | 2.8 | 2 | 1.4 | 27.9 | 2.23 | 31 | 2 | 15.5 | 59.62 | 2.5 |
| D | 34.3 | 2 | 17.2 | 343.4 | 27.65 | 290.58 | 2 | 145.09 | 558.81 | 23.7 |
| E | 8.5 | 2 | 4.3 | 85.3 | 6.90 | 43.69 | 2 | 21.85 | 84.02 | 3.6 |
| F | 12.9 | 2 | 6.4 | 128.5 | 10.32 | 146.46 | 2 | 73.23 | 281.65 | 12.0 |
| G | 1.0 | 2 | 0.5 | 10.4 | 0.83 | 11.77 | 2 | 5.89 | 22.63 | 1.0 |
| A x B | 8.8 | 4 | 2.2 | 43.8 | 3.48 | 39.64 | 4 | 9.91 | 38.12 | 1.6 |
| A x C | 25.6 | 4 | 6.4 | 127.4 | 10.25 | 108.93 | 4 | 27.23 | 104.74 | 4.5 |
| B x C | 13.2 | 4 | 3.3 | 65 | 5.27 | 85.48 | 4 | 21.37 | 82.19 | 3.5 |
| Residual | 0.08 | 54 | 0.06 | 0.52 | 2 | 0.26 | ||||
| Model | 124.10 | 26 | 62.09 | 1341.08 | 24 | 612.69 | 2356.5 | |||
| Corr. Total | 124.18 | 80 | 62.15 | 1341.60 | 26 | 612.43 | ||||
| Optimal Levels of Parameters | Predicted Values (µg mg−1-Fe) | Confidence Intervals (95%) | Average (µg mg−1-Fe) | % Variation | |
|---|---|---|---|---|---|
| Loc. I | A1, B1, C2, D1, E3, F1, G3 | 3.2 | CICE: 2.7 < < 3.7 CIPOP: 2.9 < < 3.5 | 3.4 ± 0.04 | 5.9 |
| Loc. II | A3, B3, C2, D1, E3, F1, G3 | 3.8 | CICE: 4.3 < < 3.3 CIPOP: 4.1 < < 3.5 | 3.9 ± 0.09 | 2.6 |
| qe, max. | A3, B1, C2, D1, E3, F1, G3 | 4.6 | CICE: 5.1 < < 4.1 CIPOP: 4.9 < < 4.3 | 4.8 ± 0.02 | 4.2 |
| Location I | Location II | ||||||
|---|---|---|---|---|---|---|---|
| Ionic Species | Molecular Formula | pH 7 | pH 8 | pH 9 | pH 7 | pH 8 | pH 9 |
| Bicarbonate | HCO3- | 81.8 | 94.6 | 85.0 | 81.1 | 91.7 | 76.6 |
| Carbonic acid | H2CO3 * (aq.) | 16.8 | 2.0 | 0.2 | 15.5 | 1.8 | 0.2 |
| Calcium sulfate | CaSO4 (aq.) | 6.5 | 6.5 | 6.5 | 19.2 | 9.1 | 18.7 |
| Tri-hydrogen ortho-silicate | H3SiO4− | 0.2 | 1.6 | 13.7 | 0.2 | 1.7 | 14.5 |
| Silicic acid | H4SiO4 | 99.8 | 98.4 | 86.3 | 99.8 | 98.3 | 85.4 |
| Hydrogen phosphate | HPO4−2 | 35.8 | 57.9 | 50.4 | 34.1 | 47.2 | 29.6 |
| Dihydrogen phosphate | H2PO4− | 43.1 | 7.0 | 0.6 | 33.6 | 4.7 | 0.3 |
| Magnesium phosphate | MgHPO4 (aq.) | 11.7 | 18.9 | 16.3 | 7.6 | 10.4 | 6.4 |
| Calcium hydrogen phosphate | CaHPO4 (aq.) | 5.5 | 8.9 | 7.6 | 17.8 | 24.5 | 14.8 |
| Calcium phosphate | CaPO4− | 0.2 | 2.9 | 22.6 | 0.7 | 7.7 | 46.4 |
| Magnesium phosphate | MgPO4− | - | 0.1 | 0.5 | - | 0.1 | 0.2 |
| Hydrogen arsenate | HAsO4−2 | 57.3 | 93.0 | 99.0 | 62.1 | 94.2 | 99.0 |
| Dihydrogen arsenate | H2AsO4− | 42.7 | 6.9 | 0.7 | 37.9 | 5.8 | 0.6 |
| Arsenate ion | AsO4−3 | - | - | 0.3 | - | - | 0.3 |
| Zinc hydroxide | Zn(OH)2 (aq.) | 0.1 | 6.9 | 78.0 | 0.1 | 4.5 | 64.9 |
| Zinc tri-hydroxide | Zn(OH)3− | - | - | - | - | - | 0.3 |
| Magnesium sulfate | MgSO4 (aq.) | 2.2 | 2.2 | 2.2 | 7.7 | 7.7 | 7.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, A.; Joshi, H.; Kumar, A. Arsenate Removal from the Groundwater Employing Maghemite Nanoparticles. Water 2022, 14, 3617. https://doi.org/10.3390/w14223617
Kumar A, Joshi H, Kumar A. Arsenate Removal from the Groundwater Employing Maghemite Nanoparticles. Water. 2022; 14(22):3617. https://doi.org/10.3390/w14223617
Chicago/Turabian StyleKumar, Ajay, Himanshu Joshi, and Anil Kumar. 2022. "Arsenate Removal from the Groundwater Employing Maghemite Nanoparticles" Water 14, no. 22: 3617. https://doi.org/10.3390/w14223617






