Phytoremediation Potential of Native Hyperaccumulator Plants Growing on Heavy Metal-Contaminated Soil of Khatunabad Copper Smelter and Refinery, Iran
Abstract
:1. Introduction
2. Materials and Methods
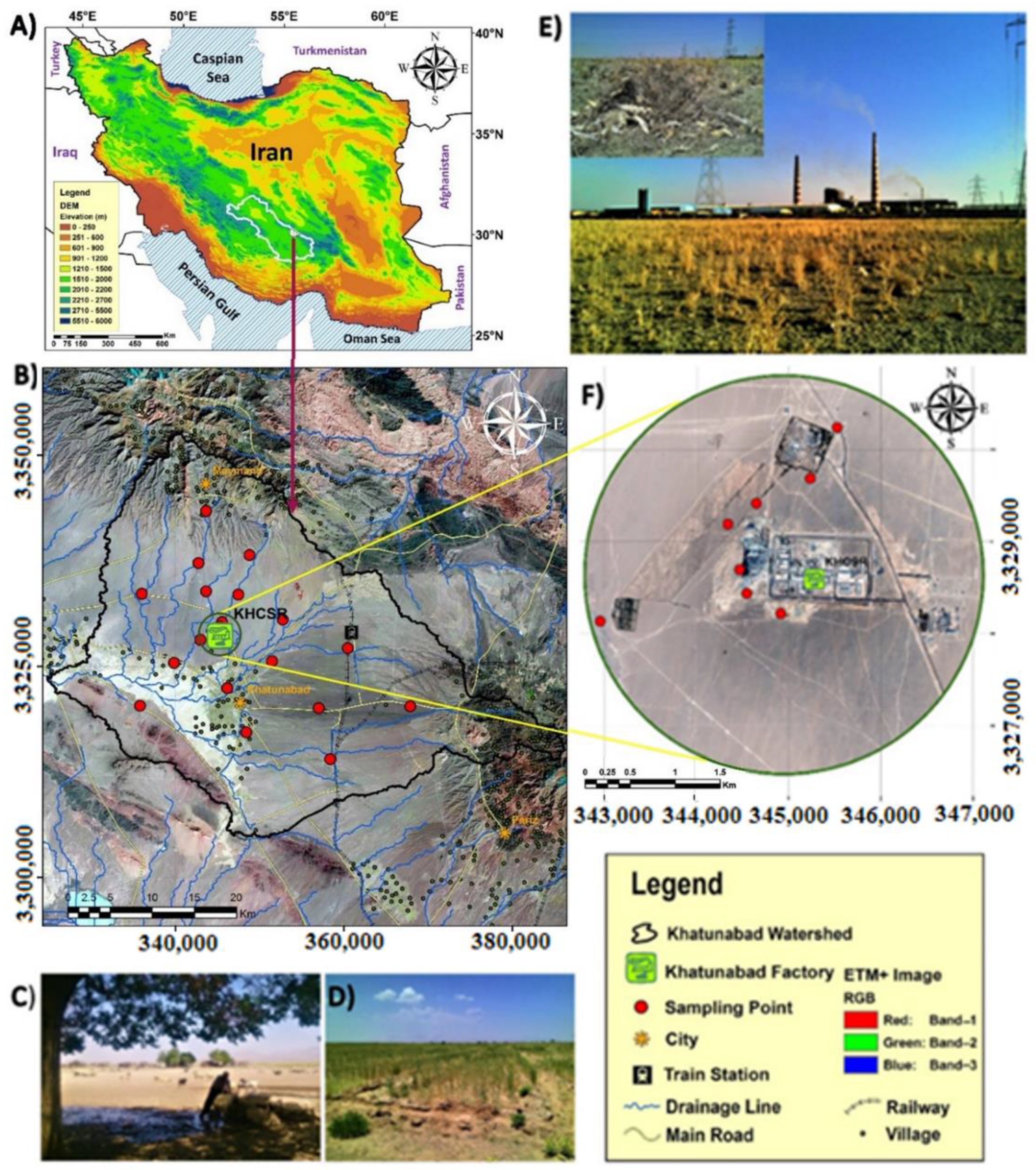
2.1. Study Area
2.2. Plants Species
2.3. Sampling and Methods of Analysis
2.4. Methods of Pollution Assessment of Heavy Metals
2.5. Metal Accumulation Index (MAI)
2.6. Bioconcentration Factor (BCF)
2.7. Translocation Factor (TF)
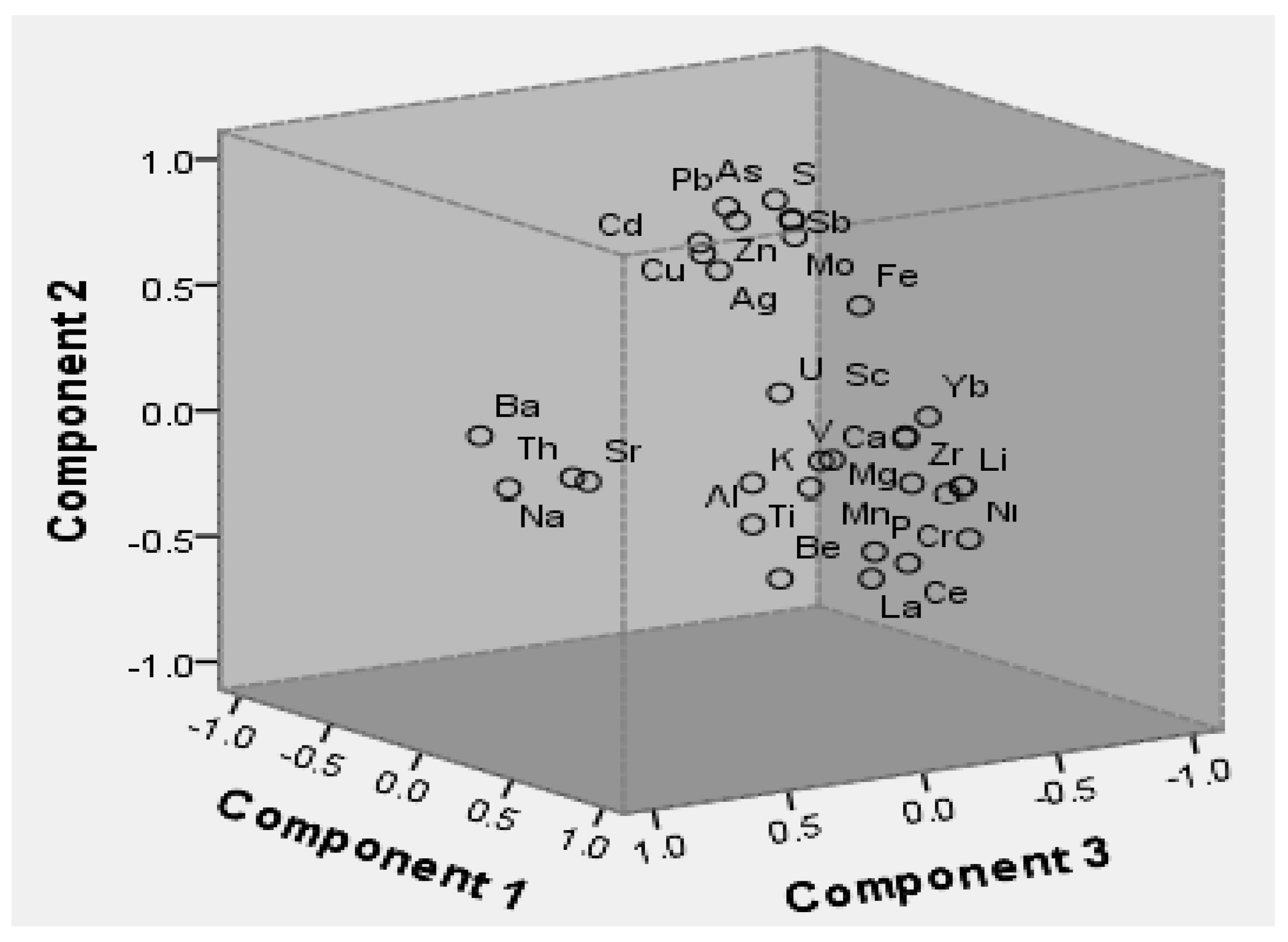
3. Results and Discussion
3.1. Soil Properties
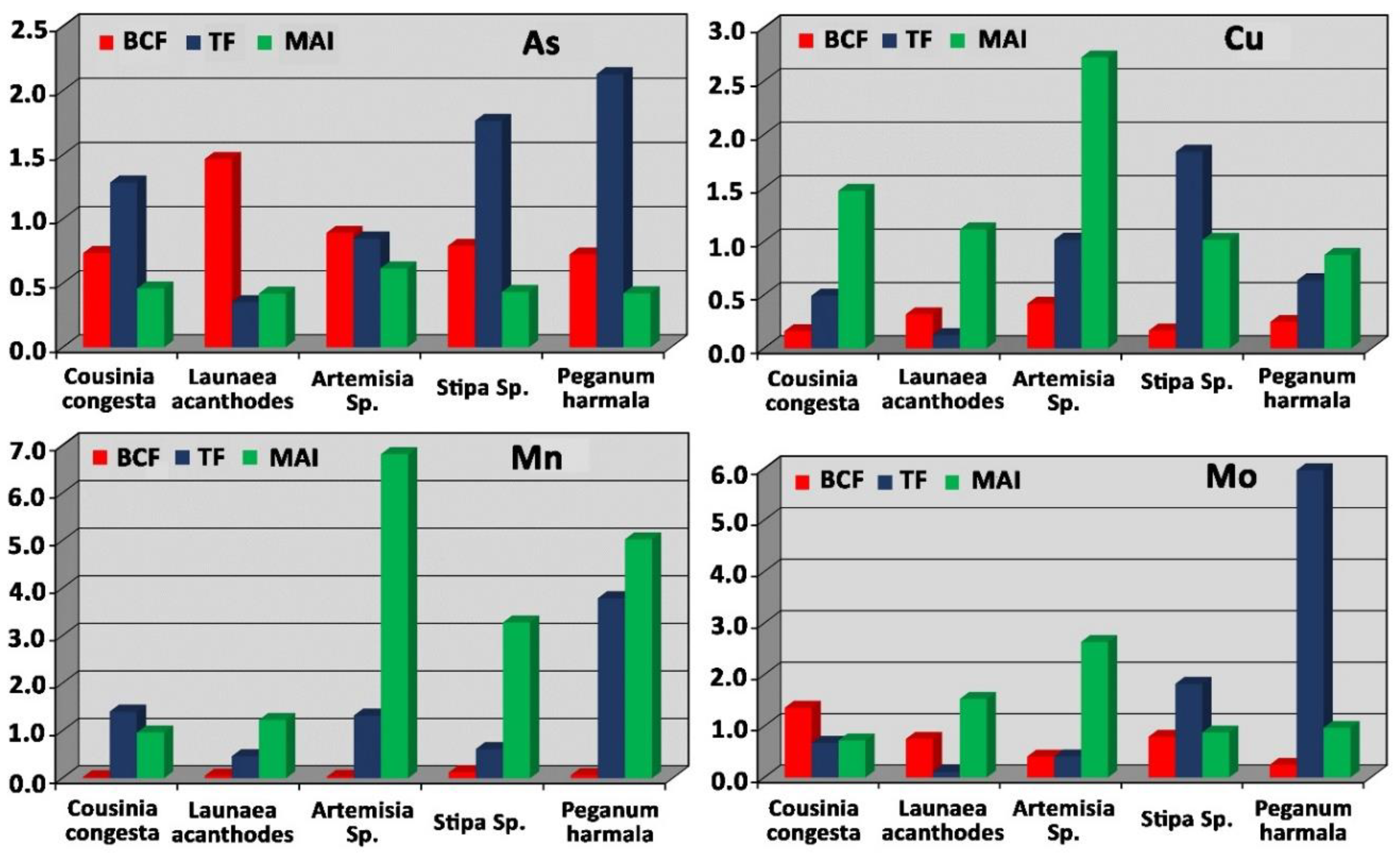
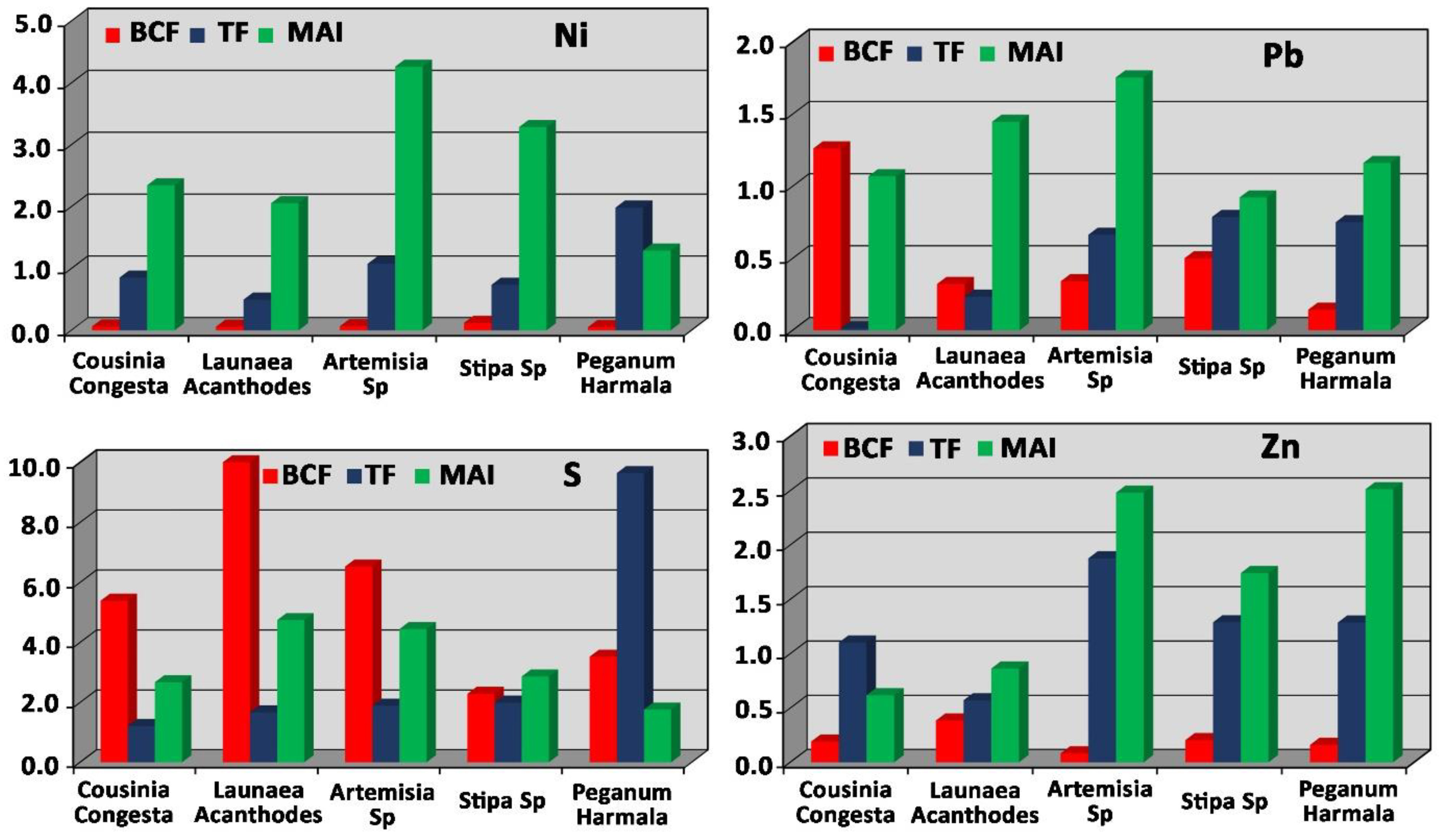
3.2. Metal Accumulation in Plant Tissue (MAI)
3.3. Bioconcentration Factor (BCF) in Plant Species
3.4. Translocation Factor (TF) in Plant Species
3.5. Metal Concentration in Plants
3.6. Arsenic Concentration
3.7. Copper Concentration
3.8. Molybdenum Concentration
3.9. Manganese Concentration
3.10. Nickel Concentration
3.11. Lead Concentration
3.12. Sulfur Concentration
3.13. Zinc Concentration
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khosravi, V.; Doulati Ardejani, F.; Yousefi, S.; Aryafar, A. Monitoring soil lead and zinc contents via combination of spectroscopy with extreme learning machine and other data mining methods. Geoderma 2018, 318, 29–41. [Google Scholar] [CrossRef]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Li, W.; Song, W.; Guo, M. Remediation techniques for heavy metal-contaminated soils: Principles and applicability. Sci. Total Environ. 2018, 633, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Xing, X.; Liang, J.; Peng, J.; Zhou, J. In situ phytoremediation of copper and cadmium in a co-contaminated soil and its biological and physical effects. RSC Adv. 2019, 9, 993–1003. [Google Scholar] [CrossRef] [Green Version]
- Rostami, S.; Azhdarpoor, A. The application of plant growth regulators to improve phytoremediation of contaminated soils: A review. Chemosphere 2019, 220, 818–827. [Google Scholar] [CrossRef] [PubMed]
- Al-Thani, R.F.; Yasseen, B.T. Phytoremediation of polluted soils and waters by native qatari plants: Future perspectives. Environ. Pollut. 2020, 259, 113694. [Google Scholar] [CrossRef]
- Mahar, A.; Wang, P.; Ali, A.; Awasthi, M.K.; Lahori, A.H.; Wang, Q.; Li, R.; Zhang, Z. Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: A review. Ecotoxicol. Environ. Saf. 2016, 126, 111–121. [Google Scholar] [CrossRef]
- Pajevic, S.; Borisev, M.; Nikolic, N.; Arsenov, D.D.; Orlovic, S.; Župunski, M. Phytoextraction of heavy metals by fast-growing trees: A review. Int. J. Phytoremediation 2016, 29–64. [Google Scholar] [CrossRef]
- Prabakaran, K.; Li, J.; Anandkumar, A.; Leng, Z.; Zou, C.B.; Du, D. Managing environmental contamination through phytoremediation by invasive plants: A review. Ecol. Eng. 2019, 138, 28–37. [Google Scholar] [CrossRef]
- Fu, S.; Wei, C.; Xiao, Y.; Li, L.; Wu, D. Heavy metals uptake and transport by native wild plants: Implications for phytoremediation and restoration. Environ. Earth Sci. 2019, 78, 103. [Google Scholar] [CrossRef]
- Shu, W.S.; Ye, Z.H.; Lan, C.Y.; Zhang, Z.Q.; Wong, M.H. Lead, zinc and copper accumulation and tolerance in populations of Paspalum distichum and Cynodon dactylon. Environ. Pollut. 2002, 120, 445–453. [Google Scholar] [CrossRef]
- Cheng, S. Heavy metals in plants and phytoremediation. Environ. Sci. Pollut. Res. 2003, 10, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.C.; Gonzalez-chavez, M.C.A. Metal accumulation in wild plants surrounding mining wastes. Environ. Pollut. 2006, 144, 84–92. [Google Scholar] [CrossRef]
- Mc Grath, S.P.; Zhao, F.J. Phytoextraction of metals and metalloids from contaminated soils. Curr. Opin. Biotechnol. 2003, 14, 277–282. [Google Scholar] [CrossRef]
- Ginocchio, R.O.S.A.N.N.A.; Baker, A.J. Metallophytes in Latin America: A remarkable biological and genetic resource scarcely known and studied in the region. Rev. Chil. Hist. Nat. 2004, 77, 185–194. [Google Scholar] [CrossRef] [Green Version]
- Bech, J.; Duran, P.; Roca, N.; Poma, W.; Sanchez, I.; Barcelo, J.; Poschenrieder, C. Shoot accumulation of several trace elements in native plant species from contaminated soils in the Peruvian Andes. J. Geochem. Explor. 2012, 113, 106–111. [Google Scholar] [CrossRef]
- Chanu, L.B.; Gupta, A. Phytoremediation of lead using Ipomoea aquatica Forsk. in hydroponic solution. Chemosphere 2016, 156, 407–411. [Google Scholar] [CrossRef]
- Sekara, A.; Poniedzialeek, M.; Ciura, J.; Jedrszczyk, E. Zinc and copper accumulation and distribution in the tissues of nine crops: Implications for phytoremediation. Pol. J. Environ. Stud. 2005, 14, 829–835. [Google Scholar]
- Jeevanantham, S.; Saravanan, A.; Hemavathy, R.V.; Kumar, P.S.; Yaashikaa, P.R.; Yuvaraj, D. Removal of toxic pollutants from water environment by phytoremediation: A survey on application and future prospects. Environ. Technol. Innov. 2019, 13, 264–276. [Google Scholar] [CrossRef]
- Golda, S.; Korzeniowska, J. Comparison of phytoremediation potential of three grass species in soil contaminated with cadmium. Environ. Nat. Resour. J./Ochr. Sr. Zasobow Nat. 2016, 27, 8–14. [Google Scholar]
- Antonkiewicz, J.; Para, A. The use of dialdehyde starch derivatives in the phytoremediation of soils contaminated with heavy metals. Int. J. Phytoremediation 2016, 18, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Muthusaravanan, S.; Sivarajasekar, N.; Vivek, J.S.; Paramasivan, T.; Naushad, M.; Prakashmaran, J.; Gayathri, V.; Al-duaij, O.K. Phytoremediation of heavy metals: Mechanisms, methods and enhancements. Environ. Chem. Lett. 2018, 16, 1339–1359. [Google Scholar] [CrossRef]
- Hauptvogl, M.; Kotrla, M.; Prcik, M.; Paukova, Ž.; Kovacik, M.; Losak, T. Phytoremediation potential of fast-growing energy plants: Challenges and perspectives–a review. Pol. J. Environ. Stud. 2019, 29, 505–516. [Google Scholar] [CrossRef] [Green Version]
- Korzeniowska, J.; Stanislawska-glubiak, E. Phytoremediation potential of Phalaris arundinacea, Salix viminalis and Zea mays for nickel-contaminated soils. Int J Environ Sci Technol. 2019, 16, 1999–2008. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Song, Y.; He, X.; Yi, L. Enhancing phytoremediation efficiency using regulated deficit irrigation. Pol. J. Environ. Stud. 2019, 28, 2399–2405. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Dong, F.; Yang, G.; Zhang, W.; Zong, M.; Nie, X.; Zhou, L.; Babar, A.; Liu, J.; Ram, B.K.; et al. Characterization of Arsenic and Uranium Pollution Surrounding a Uranium Mine in Southwestern China and Phytoremediation Potential. Pol. J. Environ. Stud. 2020, 29, 173–185. [Google Scholar] [CrossRef]
- Devi, P.; Kumar, P. Concept and application of phytoremediation in the fight of heavy metal toxicity. J. Pharm. Sci. Res. 2020, 12, 795–804. [Google Scholar]
- Akin, B. In vitro Germination and Phytoremediation Potential of Endemic Plant Species Verbascum phrygium Bornm. Growing under Zinc Stress. Pol. J. Environ. Stud. 2021, 30, 1513–1520. [Google Scholar] [CrossRef]
- Ouatiki, E.; Tounsi, A.; Amir, S.; Midhat, L.; Radi, M.; Ouahmane, L. Inoculation of Pinus halepensis with the Ectomycorrhizal Fungi Scleroderma Helps in Phytoremediation of Soil Polymetallic Pollution. Pol. J. Environ. Stud. 2021, 30, 5669–5680. [Google Scholar] [CrossRef]
- Durante-Yanez, E.V.; Martinez-Macea, M.A.; Enamorado-Montes, G.; Combatt Caballero, E.; Marrugo-Negrete, J. Phytoremediation of soils contaminated with heavy metals from gold mining activities using Clidemia sericea D. Don. Plants 2022, 11, 597. [Google Scholar] [CrossRef]
- Bhat, S.A.; Bashir, O.; Hag, S.A.U.; Amin, T.; Rafiq, A.; Ali, M.; Americo-Pinheiro, J.H.P.; Sher, F. Phytoremediation of heavy metals in soil and water: An eco-friendly, sustainable and multidisciplinary approach. Chemosphere 2022, 303, 134788. [Google Scholar] [CrossRef] [PubMed]
- Laghlimi, M.; Baghdad, B.; El Hadi, H.; Bouabdli, A. Phytoremediation mechanisms of heavy metal contaminated soils: A review. Open J. Ecol. 2015, 5, 375. [Google Scholar] [CrossRef] [Green Version]
- Nadgorska-Socha, A.; Kandziora-Ciupa, M.; Ciepal, R. Element accumulation, distribution, and phytoremediation potential in selected metallophytes growing in a contaminated area. Environ. Monit. Assess. 2015, 187, 441. [Google Scholar] [CrossRef]
- Bozdogan Sert, E.; Turkmen, M.; Cetin, M. Heavy metal accumulation in rosemary leaves and stems exposed to traffic-related pollution near Adana-İskenderun Highway (Hatay, Turkey). Environ. Monit. Assess. 2019, 191, 553. [Google Scholar] [CrossRef] [PubMed]
- Sevik, H.; Cetin, M.; Ucun Ozel, H.; Ozel, H.B.; Mossi, M.M.M.; Zeren Cetin, I. Determination of Pb and Mg accumulation in some of the landscape plants in shrub forms. Environ. Sci. Pollut. Res. 2020, 27, 2423–2431. [Google Scholar] [CrossRef]
- Siyar, R.; Doulati Ardejani, F.; Farahbakhsh, M.; Yavarzadeh, M.; Maghsoudy, S. Application of phytoremediation to reduce environmental pollution of copper smelting and refining factories: A review. J. Min. Environ. 2020, 11, 517–537. [Google Scholar]
- Siyar, R.; Doulati Ardejani, F.; Farahbakhsh, M.; Norouzi, P.; Yavarzadeh, M.; Maghsoudy, S. Potential of Vetiver grass for the phytoremediation of a real multi-contaminated soil, assisted by electrokinetic. Chemosphere 2020, 246, 125802. [Google Scholar] [CrossRef]
- Sevik, H.; Cetin, M.; Ozel, H.B.; Akarsu, H.; Zeren Cetin, I. Analyzing of usability of tree-rings as biomonitors for monitoring heavy metal accumulation in the atmosphere in urban area: A case study of cedar tree (Cedrus sp.). Environ. Monit. Assess. 2020, 192, 23. [Google Scholar] [CrossRef]
- Cetin, M.; Sevik, H.; Cobanoglu, O. Ca, Cu, and Li in washed and unwashed specimens of needles, bark, and branches of the blue spruce (Picea pungens) in the city of Ankara. Environ. Sci. Pollut. Res. 2020, 27, 21816–21825. [Google Scholar] [CrossRef]
- Alaqouri, H.A.A.; Genc, C.O.; Aricak, B.; Kuzmina, N.; Menshikov, S.; Cetin, M. The possibility of using Scots pine needles as biomonitor in determination of heavy metal accumulation. Environ. Sci. Pollut. Res. 2020, 27, 20273–20280. [Google Scholar] [CrossRef]
- Sevik, H.; Cetin, M.; Ozel, H.B.; Ozel, S.; Zeren Cetin, I. Changes in heavy metal accumulation in some edible landscape plants depending on traffic density. Environ. Monit. Assess. 2020, 192, 78. [Google Scholar] [CrossRef] [PubMed]
- Rosatto, S.; Mariotti, M.; Romeo, S.; Roccotiello, E. Root and shoot response to nickel in hyperaccumulator and non-hyperaccumulator species. Plants 2021, 10, 508. [Google Scholar] [CrossRef] [PubMed]
- Cesur, A.; Zeren Cetin, I.; Abo Aisha, A.E.S.; Alrabiti, O.B.M.; Aljama, A.M.O.; Jawed, A.A.; Cetin, M.; Sevik, H.; Ozel, H.B. The usability of Cupressus arizonica annual rings in monitoring the changes in heavy metal concentration in air. Environ. Sci. Pollut. Res. 2021, 28, 35642–35648. [Google Scholar] [CrossRef] [PubMed]
- Cetin, M.; Jawed, A.A. Variation of Ba concentrations in some plants grown in Pakistan depending on traffic density. Biomass Convers. Biorefin. 2022, 1–7. [Google Scholar] [CrossRef]
- Larner, B.L.; Seen, A.J.; Townsend, A.T. Comparative study of optimised BCR sequential extraction scheme and acid leaching of elements in the certified reference material NIST 2711. Anal. Chim. Acta 2006, 556, 444–449. [Google Scholar] [CrossRef] [Green Version]
- Kabala, C.; Singh, B.R. Distribution and forms of cadmium in soils near a copper smelter. Pol. J. Environ. Stud. 2006, 15, 90–97. [Google Scholar]
- Welp, G.; Brummer, G.W. Adsorption and solubility of ten metals in soil samples of different composition. J. Plant. Nutr. Soil Sci. 1999, 162, 155–161. [Google Scholar] [CrossRef]
- Chibuike, G.U.; Obiora, S.C. Heavy metal polluted soils: Effect on plants and bioremediation methods. Appl. Environ. Soil Sci. 2014, 2014, 752708. [Google Scholar] [CrossRef] [Green Version]
- Avangbenro, A.S.; Babalola, O.O. A new strategy for heavy metal polluted environments: A review of microbial biosorbents. Int. J. Environ. Res. Public Health 2017, 14, 94. [Google Scholar] [CrossRef]
- Yang, J.S.; Kwon, M.J.; Choi, J.; Baek, K.; O’loughlin, E.J. The transport behavior of As, Cu, Pb and Zn during electrokinetic remediation of a contaminated soil using electrolyte conditioning. Chemosphere 2014, 117, 79–86. [Google Scholar] [CrossRef]
- Einollahi, F.; Pakzad, S. Survey of Cu concentration in some grassland plants (Lactuca serriola, Artemisia sieberi and Astragalus bisulcatus) around the Khatoon Abad melting Copper mine in Shahr Babak. Hum. Environ. 2012, 10, 55–63. [Google Scholar]
- Salmabadi, H.; Saeedi, M. Determination of the transport routes of and the areas potentially affected by SO2 emanating from Khatoonabad Copper Smelter (KCS), Kerman province, Iran using HYSPLIT. Atmos. Pollut. Res. 2019, 10, 321–333. [Google Scholar] [CrossRef]
- NICICO. Environmental and Social Face of Shahrebabak, Iranian National Copper Company (NICICO); Annual Report (In Persian). Iranian National Copper Company (NICICO): Tehran, Iran, 2015. [Google Scholar]
- Sakhaee, E.; Behzadi, M.J.; Shahrad, E. Subclinical copper poisoning in asymptomatic people in residential area near copper smelting complex. Asian Pac J Trop Dis. 2012, 2, 475–477. [Google Scholar] [CrossRef]
- Mozaffari, A.A.; Derakhshanfar, A.; Amoli, J.S. Industrial copper intoxication of Iranian fat-tailed sheep in Kerman province, Iran. Turk. J. Vet. Anim. Sci. 2009, 33, 113–119. [Google Scholar] [CrossRef]
- Kabata-Pendias, A.; Mukherjee, A.B. Trace Elements from Soil to Human; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; 550p. [Google Scholar]
- Han, Y.; Li, Q.; Liu, N. Heavy metal accumulation of 13 native plant species around a coal gangue dump and their potentials for phytoremediation. Nat. Environ. Pollut. Technol. 2020, 19, 191–199. [Google Scholar]
- Claveria, R.J.R.; Perez, T.R.; Navarrete, I.A.; Perez, R.E.C.; Lim, B.C.C. The identification of heavy metal accumulator ferns in abandoned mines in the Philippines with applications to mine rehabilitation and metal recovery. J. Sustain. Min. 2020, 19, 46–57. [Google Scholar] [CrossRef]
- Haiyan, W.; Stuanes, A.O. Heavy metal pollution in air-water-soil-plant system of Zhuzhou City, Hunan Province, China. Water Air Soil Pollut. 2003, 147, 79–107. [Google Scholar] [CrossRef]
- Alahabadi, A.; Ehrampoush, M.H.; Miri, M.; Ebrahimi Aval, H.; Yousefzadeh, S.; Ghaffari, H.R.; Ahmadi, E.; Talebi, P.; Abaszadeh Fathabadi, Z.; Babai, F.; et al. A comparative study on capability of different tree species in accumulating heavy metals from soil and ambient air. Chemosphere 2017, 172, 459–467. [Google Scholar] [CrossRef]
- Chamba, I.; Gazquez, M.J.; Selvaraj, T.; Calva, J.; Toledo, J.J.; Armijos, C. Selection of a suitable plant for phytoremediation in mining artisanal zones. Int. J. Phytoremediation 2016, 18, 853–860. [Google Scholar] [CrossRef]
- Alaboudi, K.A.; Ahmed, B.; Brodie, G. Phytoremediation of Pb and Cd contaminated soils by using sunflower (Helianthus annuus) plant. Ann. Agric. Sci. 2018, 63, 123–127. [Google Scholar] [CrossRef]
- Marrugo-Negrete, J.; Marrugo-Madrid, S.; Pinedo-Hernandez, J.; Durango-Hernandez, J.; Diez, S. Screening of native plant species for phytoremediation potential at a Hg-contaminated mining site. Sci. Total Environ. 2016, 542, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Van Der Ent, A.; Baker, A.J.; Reeves, R.D.; Pollard, A.J.; Schat, H. Hyperaccumulators of metal and metalloid trace elements: Facts and fiction. Plant Soil 2013, 362, 319–334. [Google Scholar] [CrossRef]
- Liu, B.; Shiwei, A.; Zhang, W.; Huang, D.; Zhang, Y. Assessment of the bioavailability, bioaccessibility and transfer of heavy metals in the soil-grain-human systems near a mining and smelting area in NW China. Sci. Total Environ. 2017, 609, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Aubert, H.; Pinta, M. Trace elements in soils. Soil Sci. 1978, 125, 334. [Google Scholar] [CrossRef]
- EPA US. Supplemental guidance for developing soil screening levels for superfund sites. Peer Rev. Draft. OSWER 2001, 9355, 4–24. [Google Scholar]
- Kabata-pendias, A.; Pendias, H. Trace Elements in Soils and Plants, 4th ed.; CRC Press: Boca Raton, FL, USA, 2010; 548p. [Google Scholar]
- Zhao, X.; Liu, J.; Xia, X.; Chu, J.; Wei, Y.; Shi, S.; Chang, E.; Yin, W.; Jiang, Z. The evaluation of heavy metal accumulation and application of a comprehensive bio-concentration index for woody species on contaminated sites in Hunan, China. Environ. Sci. Pollut. Res. 2014, 21, 5076–5085. [Google Scholar] [CrossRef]



| Oxides (%) | Anions (ppm) | ||||
|---|---|---|---|---|---|
| SiO2 | 69.05 | MnO | 0.06 | HCO3(1−) | 9.0 |
| Al2O3 | 13.48 | Na2O | 3.18 | CO3(2−) | 2.0 |
| BaO | 0.07 | P2O5 | 0.10 | F(−) | 2.6 |
| CaO | 4.30 | SO3 | 0.07 | Cl(−) | 26.7 |
| Fe2O3 | 3.37 | TiO2 | 0.40 | Br(−) | < |
| FeO | 0.63 | Cr2O3 | 0.01 | SO4(2−) | 119.9 |
| K2O | 2.19 | Sr | 0.06 | NO2(−) | < |
| MgO | 1.18 | LOI | 2.48 | NO3(−) | < |
| Soil pH | 9.03 | Soil Ec | 164.5 ms | PO4(3−) | < |
| HMs (Water-Soluble and Exchangeable Fraction) | Concentration (ppm) | ||||||
|---|---|---|---|---|---|---|---|
| As | Cd | Cu | Mo | Pb | Sb | Zn | |
| The average concentration in the study area | 276 | 7.1 | 5444 | 26.6 | 253.7 | 14.1 | 421.5 |
| The average concentration in soil [66] | 0.1–15 | 0.01–2 | 15–40 | 1–2 | 15–30 | * | 50–100 |
| The maximum concentration in extractable resources | * | * | 250 | 101 | 1200 | * | 900 |
| Metal concentration threshold in soil [67] | 15 | 1 | 75 | 2 | 100 | * | 200 |
| Threshold for uncontaminated soil [68] | <3 | * | <25 | * | <40 | * | <90 |
| Threshold for contaminated soil [68] | 3–8 | * | 25–50 | * | 40–60 | * | 90–200 |
| Threshold for highly contaminated soil [68] | >8 | >6 | >50 | * | >60 | * | >90 |
| Mineral Name | Chemical Equation | Percentage (Semi-Quantitative) |
|---|---|---|
| Quartz | SiO2 | 41 |
| Labradorite | (Na0.4Ca0.6)Al1.6Si2.4O8 | 23.3 |
| Albite (calcian-ordered) | (Na, Ca)(Si, Al)4O8 | 17.1 |
| Calcite, syn | CaCO3 | 3.6 |
| Chlorite | (Mg, Fe)5(Al, Si)5O10(OH)8 | 3.2 |
| Illite | K(AlFe)2AlSi3O10(OH)2·H2O | 2.8 |
| Magnetite | (Fe, Mg)(Al, Cr, Fe, Ti)2O4 | 2.7 |
| Montmorillonite | (Al(OH)2)0.33Al2(Si3.67Al0.33O10)(OH)2 | 2.4 |
| Magnesio-hornblende | (Ca, Na)2.26(Mg, Fe, Al)5.15(Si, Al)8O22(OH)2 | 1.8 |
| Kaolinite | Al2Si2O5(OH)4 | 1.5 |
| Hematite, syn | Fe2O3 | 0.6 |
| Kaolinite | Al2Si2O5(OH)4/Al2O3·2SiO22H2O | 0 |
| MAI | Mean/Standard Deviation (mg/kg) | Species Name | |||||||
|---|---|---|---|---|---|---|---|---|---|
| As | Cu | Mn | Mo | Ni | Pb | S | Zn | ||
| 1.29 | 4.3 ± 9.4 | 28.2 ± 19.2 | 47 ± 48.9 | 1.0 ± 1.4 | 2.3 ± 1.0 | 36.1 ± 33.8 | 2847 ± 1067 | 54.7 ± 88.7 | Cousinia congesta |
| 0.46 | 1.47 | 0.96 | 0.72 | 2.34 | 1.07 | 2.67 | 0.62 | ||
| 1.67 | 3.9 ± 9.3 | 36.6 ± 32.9 | 49.7 ± 40.5 | 0.6 ± 0.4 | 1.5 ± 0.7 | 24.7 ± 17.1 | 7045 ± 1486 | 45.4 ± 52.7 | Launaea acanthodes |
| 0.42 | 1.11 | 1.23 | 1.52 | 2.05 | 1.45 | 4.74 | 0.86 | ||
| 3.21 | 2.2 ± 3.6 | 32 ± 11.8 | 37.3 ± 5.5 | 0.5 ± 0.2 | 1.6 ± 0.4 | 12.3 ± 7.0 | 1930 ± 435 | 18.9 ± 7.6 | Artemisia sp. |
| 0.61 | 2.72 | 6.82 | 2.63 | 4.26 | 1.75 | 4.44 | 2.48 | ||
| 1.8 | 4.7 ± 10.8 | 42.3 ± 41.7 | 76.5 ± 23.4 | 1 ± 1.1 | 2.9 ± 0.9 | 29.3 ± 31.8 | 1267 ± 443 | 27.7 ± 15.9 | Stipa sp. |
| 0.43 | 1.01 | 3.28 | 0.87 | 3.29 | 0.92 | 2.86 | 1.74 | ||
| 1.75 | 5.4 ± 12.9 | 33.1 ± 37.9 | 106 ± 21 | 1.0 ± 1.0 | 2.4 ± 1.8 | 19.3 ± 16.7 | 5798 ± 3296 | 19.2 ± 7.6 | Peganum harmala |
| 0.42 | 0.87 | 5.03 | 0.96 | 1.29 | 1.16 | 1.76 | 2.51 | ||
| 3.3 | 13.3 ± 7.8 | 66.4 ± 69.0 | 609.8 ± 168.7 | 1.2 ± 0.2 | 29.1 ± 9.1 | 23.0 ± 9.0 | 297.3 ± 103.7 | 70.0 ± 16.9 | Soil |
| 1.7 | 1 | 3.6 | 7.1 | 3.2 | 2.6 | 2.9 | 4.1 | ||
| Plants/Elements | Bioconcentration Factor (BCF) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| As | Cu | Mn | Mo | Ni | Pb | S | Zn | Average | |
| Cousinia congesta | 0.73 | 0.16 | 0.02 | 1.35 | 0.06 | 1.26 | 5.39 | 0.19 | 1.15 |
| Launaea acanthodes | 1.47 | 0.32 | 0.05 | 0.75 | 0.06 | 0.32 | 10.02 | 0.38 | 1.67 |
| Artemisia sp. | 0.89 | 0.42 | 0.03 | 0.41 | 0.07 | 0.34 | 6.52 | 0.08 | 1.09 |
| Stipa sp. | 0.79 | 0.17 | 0.12 | 0.79 | 0.12 | 0.50 | 2.28 | 0.20 | 0.62 |
| Peganum harmala | 0.72 | 0.25 | 0.06 | 0.25 | 0.05 | 0.14 | 3.52 | 0.16 | 0.64 |
| Plants/Elements | Translocation Factor (TF) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| As | Cu | Mn | Mo | Ni | Pb | S | Zn | Average | |
| Cousinia congesta | 1.28 | 0.49 | 1.40 | 0.67 | 0.85 | 0.01 | 1.21 | 1.10 | 0.88 |
| Launaea acanthodes | 0.35 | 0.12 | 0.46 | 0.11 | 0.50 | 0.23 | 1.66 | 0.57 | 0.50 |
| Artemisia sp. | 0.85 | 1.02 | 1.32 | 0.41 | 1.07 | 0.66 | 1.88 | 1.88 | 1.14 |
| Stipa sp. | 1.76 | 1.84 | 0.62 | 1.82 | 0.74 | 0.79 | 1.99 | 1.29 | 1.35 |
| Peganum harmala | 2.13 | 0.64 | 3.79 | 5.97 | 1.98 | 0.75 | 9.64 | 1.28 | 3.27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siyar, R.; Doulati Ardejani, F.; Norouzi, P.; Maghsoudy, S.; Yavarzadeh, M.; Taherdangkoo, R.; Butscher, C. Phytoremediation Potential of Native Hyperaccumulator Plants Growing on Heavy Metal-Contaminated Soil of Khatunabad Copper Smelter and Refinery, Iran. Water 2022, 14, 3597. https://doi.org/10.3390/w14223597
Siyar R, Doulati Ardejani F, Norouzi P, Maghsoudy S, Yavarzadeh M, Taherdangkoo R, Butscher C. Phytoremediation Potential of Native Hyperaccumulator Plants Growing on Heavy Metal-Contaminated Soil of Khatunabad Copper Smelter and Refinery, Iran. Water. 2022; 14(22):3597. https://doi.org/10.3390/w14223597
Chicago/Turabian StyleSiyar, Raheleh, Faramarz Doulati Ardejani, Parviz Norouzi, Soroush Maghsoudy, Mohammad Yavarzadeh, Reza Taherdangkoo, and Christoph Butscher. 2022. "Phytoremediation Potential of Native Hyperaccumulator Plants Growing on Heavy Metal-Contaminated Soil of Khatunabad Copper Smelter and Refinery, Iran" Water 14, no. 22: 3597. https://doi.org/10.3390/w14223597









