Research Progress of Microplastic Pollution in the Vadose Zone
Abstract
:1. Introduction
2. The Source, Types, and Properties of MPs in the Vadose Zone
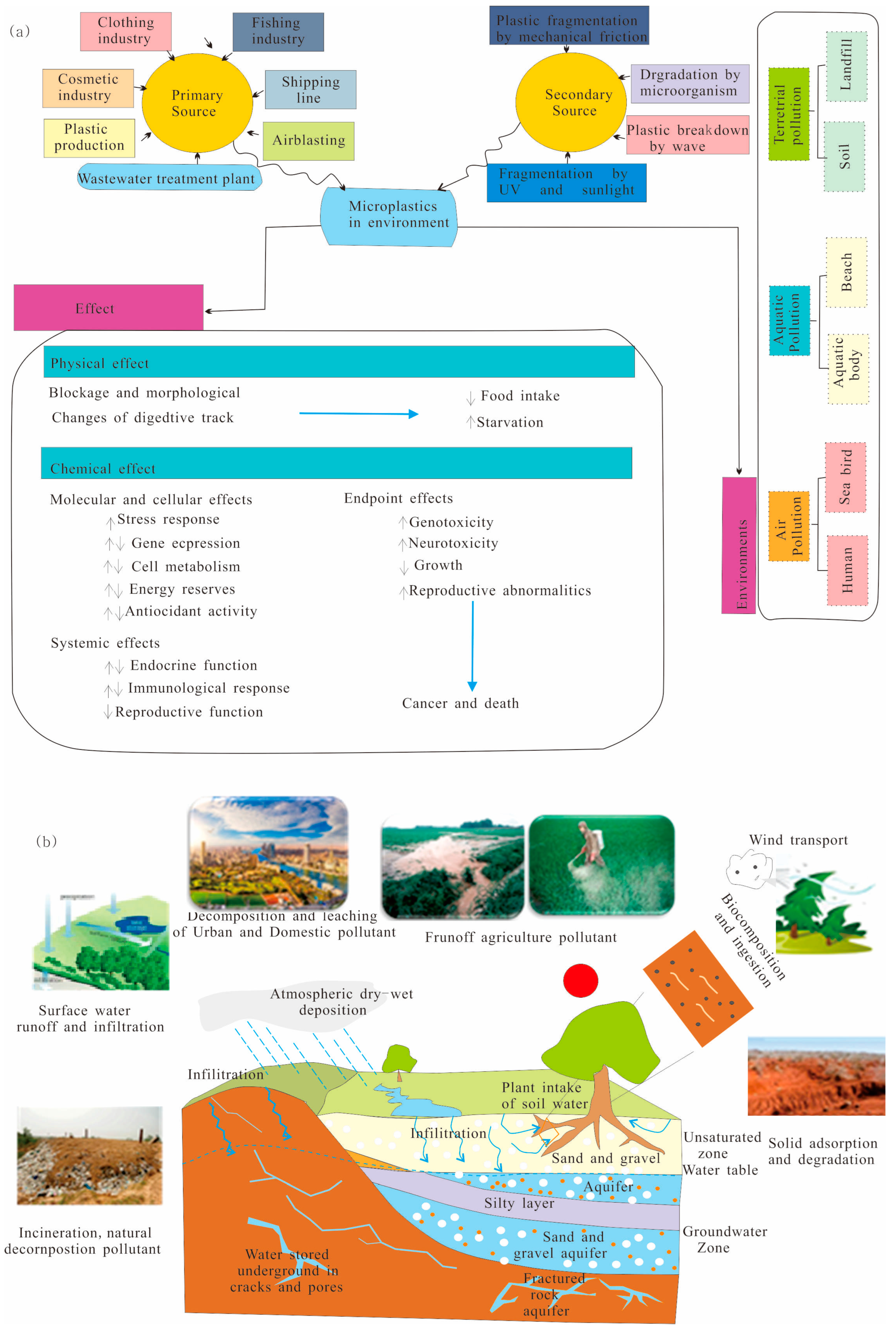
2.1. The Source of Microplastics in the Vadose Zone
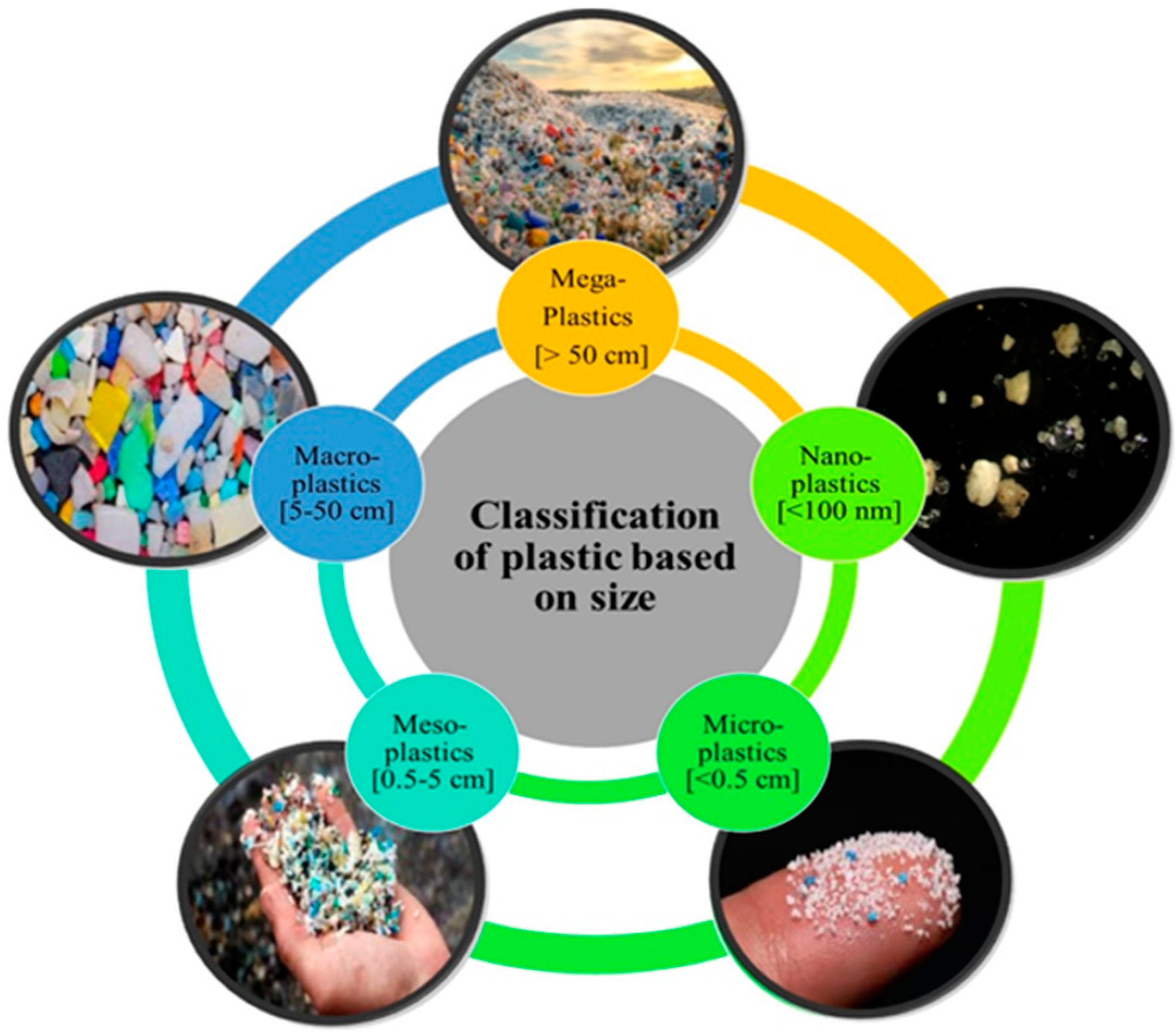
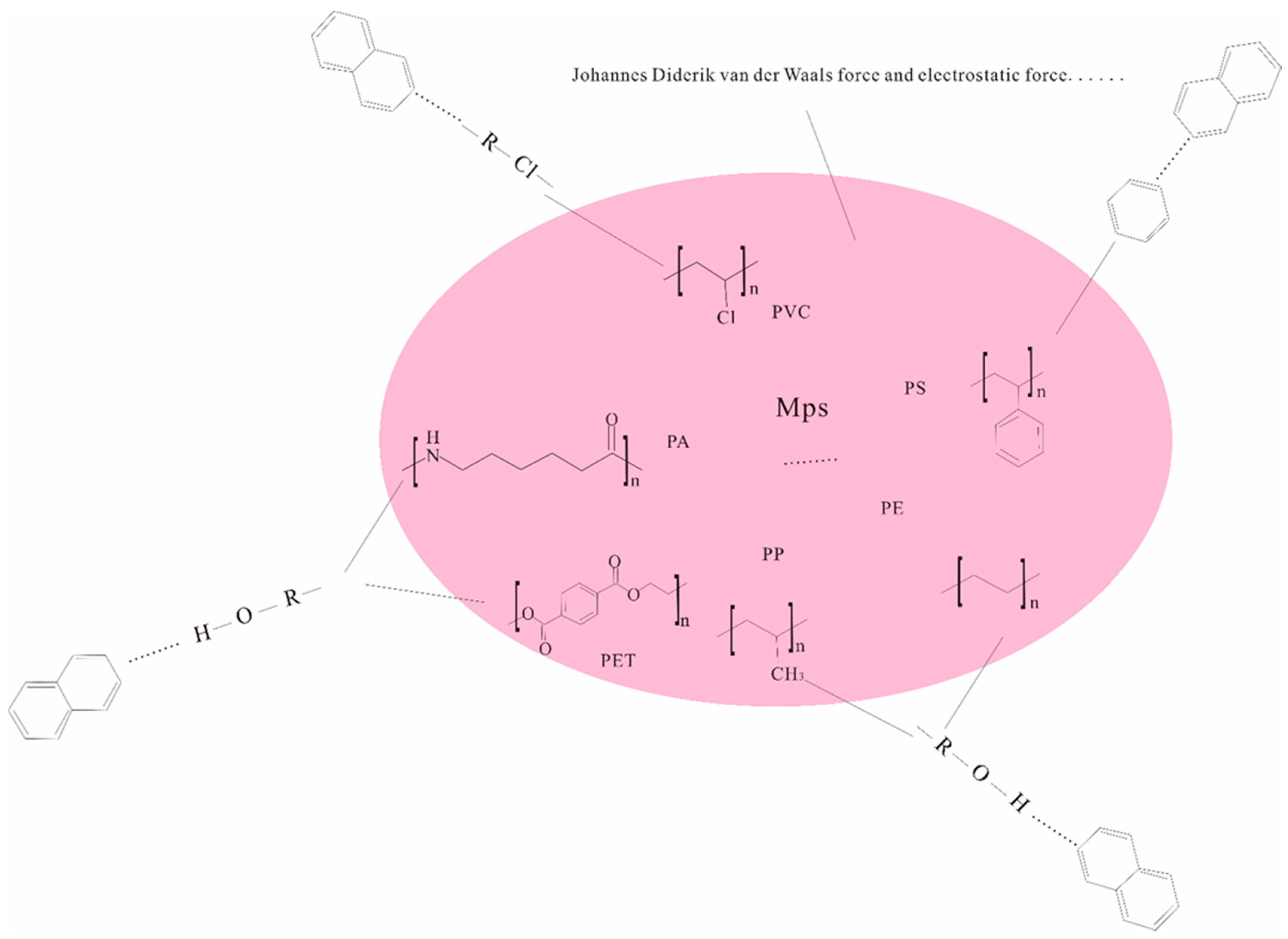
2.2. The Main Types and Properties of MPs in the Vadose Zone
3. Migration Mechanism
3.1. Characteristics of Microplastics in the Vadose Zone
3.2. Hydrometeorological Factors
3.3. Effect of Chemical Factors on the Migration of MPs in the Vadose Zone
3.4. Effect of Hydrogeological Factors on the Migration of MPs in the Vadose Zone
3.5. Effect of Biological Factors on the Migration of MPs in the Vadose Zone
| Factor | Changes in MPs with Influencing Factors | ||
|---|---|---|---|
| MPs | Characteristics of MPs | Particle size, shape, surface morphology, and density of MPs [27] | Smaller MPs and very small plastic fragments within the range of nanometer particles or colloids migrate easily, and large particles may cause blockage; the shape of MPs affects soil aggregate formation and organic decomposition in the soil system. |
| Hydrometeorological factors | Surface runoff, dry and wet deposition, wind and sun exposure | In the event of rain and snow, microplastics deposited in the soil are fragmented by gravity and can be transported further to the aquifer; the change in surface water level promotes the migration of microplastics. | |
| hydrogeology factor | Hydrodynamic condition | Flow;Topographic conditions [45] | MPs in the soils can be released and transported by water flow through the vadose zone to enter the groundwater under various conditions [19]. Microplastics migrate mainly through dominant channels, and this dominant effect is usually more pronounced with an increase in pore water flow velocities [46]. |
| Hydrogeological properties of the surrounding rock | Particle size; surface roughness; heterogeneity; saturation [47] | With increased pore water flow rate, the MPs’ capacity is enhanced; the smaller the medium particle size, the higher the medium roughness, and the smaller the medium saturation are, the lower the MPs’ capability is; MPs migrate mainly through dominant channels; frequent alternation of wetting and drying promotes vertical migration of MPs in the vadose zone [27]. | |
| Chemical factor | Chemical Properties of MPs | Chemical composition; surface functional groups; hydrophobicity; surface charge | The particle surface electronegativity and hydrophilicity are enhanced, and the migration ability in the soil medium is significantly enhanced. Migration ability of MPs with different surface functional groups [36]. The transport of MP particles could be dramatically mitigated with increased hydrophobicity after they enter the vadose zone if they behaved as colloids. |
| Chemical properties of the medium | Medium mineral composition; surface charge; pH, and chemical bond between ions | The higher the Fe/Al oxide content, the lower the migration ability of MPs; the cations released on the medium surface may reduce the MPs’ capacity; the migration ability of MPs decreased with the increase in ionic strength or the presence of high valence cations. | |
| Chemical properties of water flow | pH; organic matter composition; colloid; coexistent pollutants [34] | Increased pH and dissolved organic matter can enhance MPs’ capacity. | |
| Biological factor | Plant response | Plant absorption, root fissure; root exudates [48] | Fractures formed by root development enhance the migration of MPs. |
| Animal effect | Animal feeding and sports activities [19] | Migration of MPs with feeding and movement of small animals. | |
| Microbial effect | The existence of microorganisms and biofilms [49] | Microorganisms exist, and MPs’ capacity decreases. | |
4. Study on Synergistic Migration and Transformation of MPs and Other Pollutants in Inflated Zone
5. Conclusions and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajmohan, K.V.; Ramya, C.; Sunita, V. Plastic pollutants: Effective waste management for pollution control and abatement. Curr. Opin. Environ. Sci. Health 2019, 12, 72–84. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.; Xie, Y.; Sun, X.; Du, H.; Wang, J. Identification and Quantification of Microplastics in Aquaculture Environment. Front. Mar. Sci. 2022, 8, 804208. [Google Scholar] [CrossRef]
- Liu, R.-P.; Dong, Y.; Quan, G.-C.; Zhu, H.; Xu, Y.-N.; Elwardany, R.M. Microplastic pollution in surface water and sediments of Qinghai-Tibet Plateau: Current status and causes. China Geol. 2021, 4, 1–7. [Google Scholar] [CrossRef]
- Selvam, S.; Jesuraja, K.; Venkatramanan, S.; Roy, P.D.; Kumari, V.J. Hazardous microplastic characteristics and its role as a vector of heavy metal in groundwater and surface water of coastal south India. J. Hazard. Mater. 2020, 402, 123786. [Google Scholar] [CrossRef]
- Wu, P.; Huang, J.; Zheng, Y.; Yang, Y.; Zhang, Y.; He, F.; Chen, H.; Quan, G.; Yan, J.; Li, T.; et al. Environmental occurrences, fate, and impacts of microplastics. Ecotoxicol. Environ. Saf. 2019, 184, 109612. [Google Scholar] [CrossRef]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.G.; McGonigle, D.; Russell, A.E. Lost at Sea: Where Is All the Plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef]
- Palmas, S.; Annalisa, V.; Laura, M. Bibliometric Analysis on the Papers Dedicated to Microplastics in Wastewater Treatments. Catalysts 2021, 11, 913. [Google Scholar] [CrossRef]
- Andrady, A.L. The plastic in microplastics: A review. Mar. Pollut. Bull. 2017, 119, 12–22. [Google Scholar] [CrossRef]
- Bouwmeester, H.; Hollman, P.C.H.; Peters, R.J.B. Potential Health Impact of Environmentally Released Micro- and Nanoplastics in the Human Food Production Chain: Experiences from Nanotoxicology. Environ. Sci. Technol. 2015, 49, 8932–8947. [Google Scholar] [CrossRef]
- Devriese, L.; De Witte, B.; Vethaak, A.D.; Hostens, K.; Leslie, H.A. Bioaccumulation of PCBs from Microplastics in Norway Lobster Nephrops N: An Experimental Study. Chemosphere 2017, 186, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Fen, Q.; Du, J.; Gao, J.; Liu, G.Y.; Song, Y.Q.; Yang, A.F.; Wang, H.; Ding, Y.; Wang, Q. Bibliometric Profile of Global Microplastics Research from 2004 to 2019. Int. J. Env. Res. Public Health 2020, 17, 5639. [Google Scholar] [CrossRef]
- Panno, S.V.; Kelly, W.R.; Scott, J.; Zheng, W.; McNeish, R.E.; Holm, N.; Hoellein, T.J.; Baranski, E.L. Microplastic contamination in Karst groundwater systems. Groundwater 2019, 572, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pu, S.; Lv, X.; Gao, Y.; Ge, L. Global trends and prospects in microplastics research: A bibliometric analysis. J. Hazard. Mater. 2020, 5, 123110. [Google Scholar] [CrossRef]
- Dey, T.K.; Uddin, E.; Jamal, M. Detection and removal of microplastics in wastewater: Evolution and impact. Environ. Sci. Pollut. Res. 2021, 28, 16925–16947. [Google Scholar] [CrossRef]
- Wagner, M.; Lambert, S. Microplastics Are Contaminants of Emerging Concern in Freshwater En-vironments: An Overview.in Freshwater Microplastics. Freshw. Microplastics 2018, 58, 1–23. [Google Scholar] [CrossRef] [Green Version]
- Monkul, M.M.; Özhan, H.O. Microplastic Contamination in Soils: A Review from Geotechnical Engineering View. Polymers 2021, 13, 4129. [Google Scholar] [CrossRef]
- Zhang, K.; Su, J.; Xiong, X.; Wu, X.; Wu, C.; Liu, J. Microplastic pollution of lakeshore sediments from remote lakes in Tibet plateau, China. Environ. Pollut. 2016, 219, 450–455. [Google Scholar] [CrossRef]
- Wu, X.; Lyu, X.; Li, Z.; Gao, B.; Zeng, X.; Wu, J.; Sun, Y. Transport of polystyrene nanoplastics in natural soils: Effect of soil properties, ionic strength and cation type. Sci. Total Environ. 2020, 707, 136065. [Google Scholar] [CrossRef]
- Blair, R.M.; Waldron, S.; Gauchotte-Lindsay, C. Average daily flow of microplastics through a tertiary wastewater treatment plant over a ten-month period. Water Res. 2019, 163, 114909. [Google Scholar] [CrossRef]
- He, P.; Chen, L.; Shao, L.; Zhang, H.; Lü, F. Municipal solid waste (MSW) landfill: A source of microplastics? -Evidence of microplastics in landfill leachate. Water Res. 2019, 159, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, E.; Nowack, B.; Mitrano, D.M. Polyester Textiles as a Source of Microplastics from Households: A Mechanistic Study to Understand Microfiber Release during Washing. Environ. Sci. Technol. 2017, 51, 7036–7046. [Google Scholar] [CrossRef]
- Yao, Z.; Li, X.; Xiao, J. Characteristics of daily extreme wind gusts on the Qinghai-Tibet Plateau, China. J. Arid Land 2018, 10, 673–685. [Google Scholar] [CrossRef] [Green Version]
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef] [Green Version]
- Junhua, H.; Wen-Bing, T.; Hong, Y. Microplastics in soil ecosystem: A review on sources, fate, and ecological impact. Environ. Eng. 2020, 38, 16–27. [Google Scholar]
- Mei, W.; Chen, G.; Bao, J.; Song, M.; Li, Y.; Luo, C. Interactions between microplastics and organic compounds in aquatic environments: A mini review. Sci. Total Environ. 2020, 736, 139472. [Google Scholar] [CrossRef]
- Alimi, O.S.; Farner Budarz, J.; Hernandez, L.M.; Tufenkji, N. Microplastics and Nanoplastics in Aquatic Environments: Aggregation, Deposition, and Enhanced Contaminant Transport. Environ. Sci. Technol. 2018, 52, 1704–1724. [Google Scholar] [CrossRef]
- Anika, L.; Eva, F.L.; Maurice, G.; Matthias, C.R. Microplastics have shape and polymer-dependent effects on soil processes. Ecol. Environ. Conserv. 2020, 530, 130054. [Google Scholar] [CrossRef]
- Zhao, X.L.; Bi, E.P. Effects of dissolved organic matter on the sorption of organic pollutants to soils. Environ. Chem. 2014, 33, 256–261. [Google Scholar]
- Lin, X.M.; Su, C.Y.; Wu, S.M. Effects of PES and 2,4-DPC the on the Extracellular Polymeric Substances and microbial community of anaerobic granular sludge. Environ. Sci. 2020, 42, 1946–1955. [Google Scholar] [CrossRef]
- Wang, J.; Coffin, S.; Sun, C.; Schlenk, D.; Gan, J. Negligible effects of microplastics on animal fitness and HOC bioaccumulation in earthworm Eisenia fetida in soil. Environ. Pollut. 2019, 249, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Pu, S.Y.; Zhang, Y.; Lv, X. Review on the environmental behavior and ecotoxicity of microplastics in soil-groundwater. Asian J. Ecotoxicol. 2020, 15, 44–55. (In Chinese) [Google Scholar] [CrossRef]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bläsing, M.; Amelung, W. Plastics in soil: Analytical methods and possible sources. Sci. Total Environ. 2018, 612, 422–435. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.-P.; Xu, Y.-N.; Rui, H.-C.; Rm, E.-W.; Dong, Y. Migration and speciation transformation mechanisms of mercury in undercurrent zones of the Tongguan gold mining area, Shannxi and impact on the environment. China Geol. 2021, 4, 311–328. [Google Scholar] [CrossRef]
- Gao, J.; Pan, S.; Li, P.; Wang, L.; Hou, R.; Wu, W.-M.; Luo, J.; Hou, D. Vertical migration of microplastics in porous media: Multiple controlling factors under wet-dry cycling. J. Hazard. Mater. 2021, 419, 126413. [Google Scholar] [CrossRef]
- Colpaert, R.; Petit dit Grézériat, L.; Louzon, M.; de Vaufleury, A.; Gimbert, F. Polyethylene microplastic toxicity to the terrestrial snail Cantareus aspersus: Size matters. Environ. Sci. Pollut. Res. 2022, 29, 29258–29267. [Google Scholar] [CrossRef]
- He, L.; Rong, H.; Li, M.; Zhang, M.; Liu, S.; Yang, M.; Tong, M. Bacteria have different effects on the transport behaviors of positively and negatively charged microplastics in porous media. J. Hazard. Mater. 2021, 415, 125550. [Google Scholar] [CrossRef]
- Wu, P.; Cai, Z.; Jin, H.; Tang, Y. Adsorption mechanisms of five bisphenol analogues on PVC microplastics. Sci. Total Environ. 2018, 650, 671–678. [Google Scholar] [CrossRef]
- Li, M.; Zhang, X.; Yi, K.; He, L.; Han, P.; Tong, M. Transport and deposition of microplastic particles in saturated porous media: Co-effects of clay particles and natural organic matter. Environ. Pollut. 2021, 287, 117585. [Google Scholar] [CrossRef]
- Dong, S.; Sun, Y.; Gao, B.; Shi, X.; Xu, H.; Wu, J.; Wu, J. Retention and transport of graphene oxide in water-saturated limestone media. Chemosphere 2017, 180, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.-P.; Zhu, H.; Liu, F.; Dong, Y.; El-Wardany, R.M. Current situation and human health risk assessment of fluoride enrichment in groundwater in the Loess Plateau: A case study of Dali County, Shaanxi Province, China. China Geol. 2021, 4, 492–502. [Google Scholar] [CrossRef]
- Liu, R.-P.; Li, Z.-Z.; Liu, F.; Dong, Y.; Jiao, J.-G.; Sun, P.-P.; Rm, E.-W. Microplastic pollution in Yellow River: Current status and research progress of biotoxicological effects. China Geol. 2021, 4, 1–8. [Google Scholar] [CrossRef]
- Liu, R.P.; Xu, Y.N.; Zhang, J.H.; Wang, W.K.; El-Wardany, R.M. The effect of heavy metal pollution on soil and crops in farmland: An example from the Xiaoqinling Gold Belt, China. China Geol. 2020, 3, 403–411. [Google Scholar] [CrossRef]
- Zhu, H.H.; Jia, C.; Xu, Y.L.; Xu, Y.L.; Yu, Z.M.; Yu, W.J. Study on numerical simulation of organic pollutant transport in ground-water northwest of Laixi. J. Groundw. Sci. Eng. 2018, 6, 293–305. [Google Scholar] [CrossRef]
- Morales, V.L.; Parlange, J.Y.; Steenhuis, T.S. Are preferentialflow paths perpetuated by microbial activity in the soil matrix: A review. J. Hydrol. 2010, 393, 29–36. [Google Scholar] [CrossRef]
- Tong, M.; Johnson, W.P. Excess Colloid Retention in Porous Media as a Function of Colloid Size, Fluid Ve-locity, and Grain Angularity. Environ. Sci. Technol. 2006, 40, 7725–7731. [Google Scholar] [CrossRef]
- Hou, J.; Xu, X.; Lan, L.; Miao, L.; Xu, Y.; You, G.; Liu, Z. Transport behavior of micro polyethylene particles in saturated quartz sand: Impacts of input concentration and physicochemical factors. Environ. Pollut. 2020, 263, 114499. [Google Scholar] [CrossRef]
- Zhu, D.; Chen, Q.L.; An, X.L.; Yang, X.R.; Christie, P.; Ke, X.; Wu, L.-H.; Zhu, Y.G. Exposure of soil collembolans to microplastics perturbs their gut microbiota and alters their isotopic composition. Soil Biol. Biochem. 2018, 116, 302–310. [Google Scholar] [CrossRef]
- Liu, R.; Xu, Y.; Zhang, J.; Qiao, G.; Ke, H.; Chen, H.; He, F. Safe Technology of Crops in Reclaimed Farmland of Heavy Metals Tail Slag Field. Northwestern Geol. 2019, 52, 236–247. [Google Scholar] [CrossRef]
- Rong, H.; Li, M.; He, L.; Zhang, M.; Hsieh, L.; Wang, S.; Han, P.; Tong, M. Transport and deposition behaviors of microplastics in porous media: Co-impacts of N fertilizers and humic acid. J. Hazard. Mater. 2021, 426, 127787. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Xia, J.; Sheng, L.; Wang, W.; Liu, H.; Gao, B. Transport characteristics of fragmental polyethylene glycol terephthalate (PET) microplastics in porous media under various chemical conditions. Chemosphere 2021, 276, 130214. [Google Scholar] [CrossRef] [PubMed]
- Poritosh, R.; Amar, K.M.; Manjusri, M. Microplastics in ecosystems: Their implications and mitigation pathways. Environ. Sci. Adv. 2022, 1, 9–29. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, Z.; Zhang, Y.; Fan, P.; Xi, B.; Tan, W. Metal type and aggregate microenvironment govern the response sequence of speciation transformation of different heavy metals to microplastics in soil. Sci. Total Environ. 2020, 752, 141956. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, R.-P.; Liu, F.; Sun, P.-P.; R.M., E.-W.; Dong, Y.; Zhang, Y.-B.; Chen, H.-Q.; Jiao, J.-G. Research Progress of Microplastic Pollution in the Vadose Zone. Water 2022, 14, 3586. https://doi.org/10.3390/w14213586
Liu R-P, Liu F, Sun P-P, R.M. E-W, Dong Y, Zhang Y-B, Chen H-Q, Jiao J-G. Research Progress of Microplastic Pollution in the Vadose Zone. Water. 2022; 14(21):3586. https://doi.org/10.3390/w14213586
Chicago/Turabian StyleLiu, Rui-Ping, Fei Liu, Ping-Ping Sun, El-Wardany R.M., Ying Dong, Yi-Bing Zhang, Hua-Qing Chen, and Jian-Gang Jiao. 2022. "Research Progress of Microplastic Pollution in the Vadose Zone" Water 14, no. 21: 3586. https://doi.org/10.3390/w14213586





