1. Introduction
Around 2 billion people worldwide do not have access to clean water. Of these, about 785 million people do not have even a basic drinking water supply [
1]. However, access to clean and safe water is essential for survival and development. It is also a human right and enshrined as Goal 6 “Ensure access to water and sanitation for all” in the United Nations (UN) Sustainable Development Goals (SDGs) [
2]. Clean water is not only important for health, but also essential for reducing poverty, ensuring peace, and respecting human rights. Many countries face challenges such as strong population growth and thus increasing demand for clean water, water scarcity due to overexploitation, water pollution, and advancing climate change [
3]. Approximately 114 million people rely on drinking untreated surface water [
4].
Flocculation is a treatment method for removing suspended and colloidally dissolved pollutants from water. It is used to coagulate suspended particles, including microbes, to improve their sedimentation [
5]. Chemical flocculants are usually used to purify turbid surface water. However, these are rarely available in rural areas and are often not affordable for local people [
6]. In addition, chemical flocculants can potentially have negative impacts on people’s health and the environment. They also generate a large amount of sludge that is costly to treat and dispose of [
7]. Natural flocculants are of great interest in this regard because they are natural and inexpensive products that are environmentally friendly and considered safe for human health [
8]. Natural flocculants are also suitable for household use. According to [
5], household water treatment approaches have the potential to have rapid and significant positive health effects, especially in situations where a piped water system is not available and people rely on other water sources that may be contaminated. Household water treatment can also serve to address the widespread problem of microbially unsafe piped water supplies [
5]. The most studied natural flocculant to date is the seed of the Moringa tree [
9]. In various studies [
9,
10,
11,
12,
13,
14,
15], Moringa seeds were found to remove turbidity by 90% to 99% and thus can be a possible substitute for conventional flocculants. However, none of the existing papers dealing with the use of
Moringa oleifera as a flocculant in drinking water treatment address the shear strength of the resulting flocs. Further literature searches also failed to find hardly any relevant publications dealing with the shear strength of natural flocculants, especially
Moringa oleifera.
Flocculation tests carried out with Moringa seeds because of this have shown that the flocs formed are very fragile, irrespective of the flocculation conditions and the water samples used, and disintegrate again even at low induced shear forces. This is particularly problematic for further processing steps such as filtration, sedimentation or flotation, as shear forces always act on the flocs due to the turbulence in the water. Flocculants from Moringa are therefore only suitable for water treatment without restrictions in individual cases.
In the course of further research work, other natural flocculants were also investigated and a high shear strength of the resulting flocs was observed in the case of flocculants from cactus; however, the flocculation activity was not satisfactory as residual turbidity remained too high in the supernatant. Based on these observations, two series of experiments were carried out to investigate whether a combined flocculant from Moringa and cactus could exhibit good flocculation activity and produce flocs that can withstand high shear forces. The first series of tests was carried out with artificially produced surface water in order to be able to exclude effects of ingredients of natural surface waters on the flocculation process. In the second series of tests, samples from a natural surface water were used to confirm the real use of the new flocculant on a laboratory scale.
2. Materials and Methods
2.1. Surface Waters Used
In the first series of experiments, the removal of turbidity by natural flocculants was investigated using an artificially prepared surface water. For this purpose, ten liters of the model suspension were prepared before the start of the experiment, the ingredients and concentration of which are shown in
Table 1. The suspension was then stirred on a magnetic stirring plate for at least 30 min to ensure that the solids dissolved as far as possible and the substances, such as the bolus contained, were sufficiently hydrated.
In the second series of experiments, samples were taken from a local stream called Mühlbach and used for the flocculation experiments in order to test the flocculants under conditions that were as real as possible. The water analyses can be viewed on the HLNUG data portal at the following link:
https://www.hlnug.de/messwerte/datenportal/messstelle/4/6/180/ (accessed on 20 May 2022).
2.2. Production of Flocculants
For the production of a flocculant from cactus, some shoots were cut off. The preparation of the powder was similar to that described in [
16,
17,
18]. First, the fresh cactus leaves were washed with distilled water to remove dirt. Then, the cactus shoots were crushed and dried in a drying oven at 80 °C for about 12 h. During this process, the cactus lost almost 44% of its weight. Subsequently, the cactus was crushed with a mill and finally sieved through a mesh size of 125 μm.
Figure 1 shows the process in pictures. The powder was then prepared with a solution of 0.5 mol/L NaCl and stirred for ten minutes.
For the combined flocculant from Moringa seeds and cactus, the dry Moringa seeds were first pulverized. The powder from the Moringa seeds and that from the cactus cladodes were then mixed together in different weight ratios and a 0.5 molar NaCl solution. The flocculant was then stirred on a stir plate for ten minutes and filtered through a filter cloth.
2.3. Jar Test
Flocculation tests were carried out in order to investigate the test objectives mentioned at the beginning. The tests were conducted as jar tests in accordance with DVGW Code of Practice W 218 “Coagulation and flocculation in water treatment—Part 2: Test methods”. The raw water was initially stirred at 100 rpm for about five minutes to homogeneously distribute any settled solids. The flocculant was then added and stirred at 100 rpm for one minute. In this coagulation phase, care must be taken to ensure that the flocculant is distributed quickly and homogeneously in the water, since coagulation and thus the formation of microflocs begins directly with the addition of the flocculant. For the subsequent flocculation phase, during which the microflocs can agglomerate into macroflocs, the stirring speed was reduced to 20 rpm. After 15 min, the agitators were switched off. During the subsequent sedimentation phase, the stirrers were removed from the beaker so as not to hinder the sinking of the flocs and prevent their destruction.
2.4. Turbidity Measurement
Turbidity is used as the most important indicator for checking the effectiveness of the flocculants. A turbidimeter, which measures the scattered light component, was used to measure turbidity. A detailed description of the operation of a turbidimeter can be found in DIN EN ISO 7027-1:2016.
Samples were taken below the water surface using a pipette and mixed again before measurement to avoid sedimentation processes distorting the measurement. The reduction of turbidity was calculated according to the following equation:
From the measured turbidity values, the flocculation activity could also be calculated using the following formula:
The value of the flocculation activity, in contrast to the turbidity reduction, allows conclusions to be drawn about the actual additional turbidity reduction by the flocculant, since it includes the final turbidity in the control beaker. That is, the turbidity reduction that occurs by sedimentation alone is also considered and the reduction by the flocculant is assessed in relation to it. For this reason, this value is considered more meaningful than that of the turbidity reduction alone.
2.5. Shear Strength Test
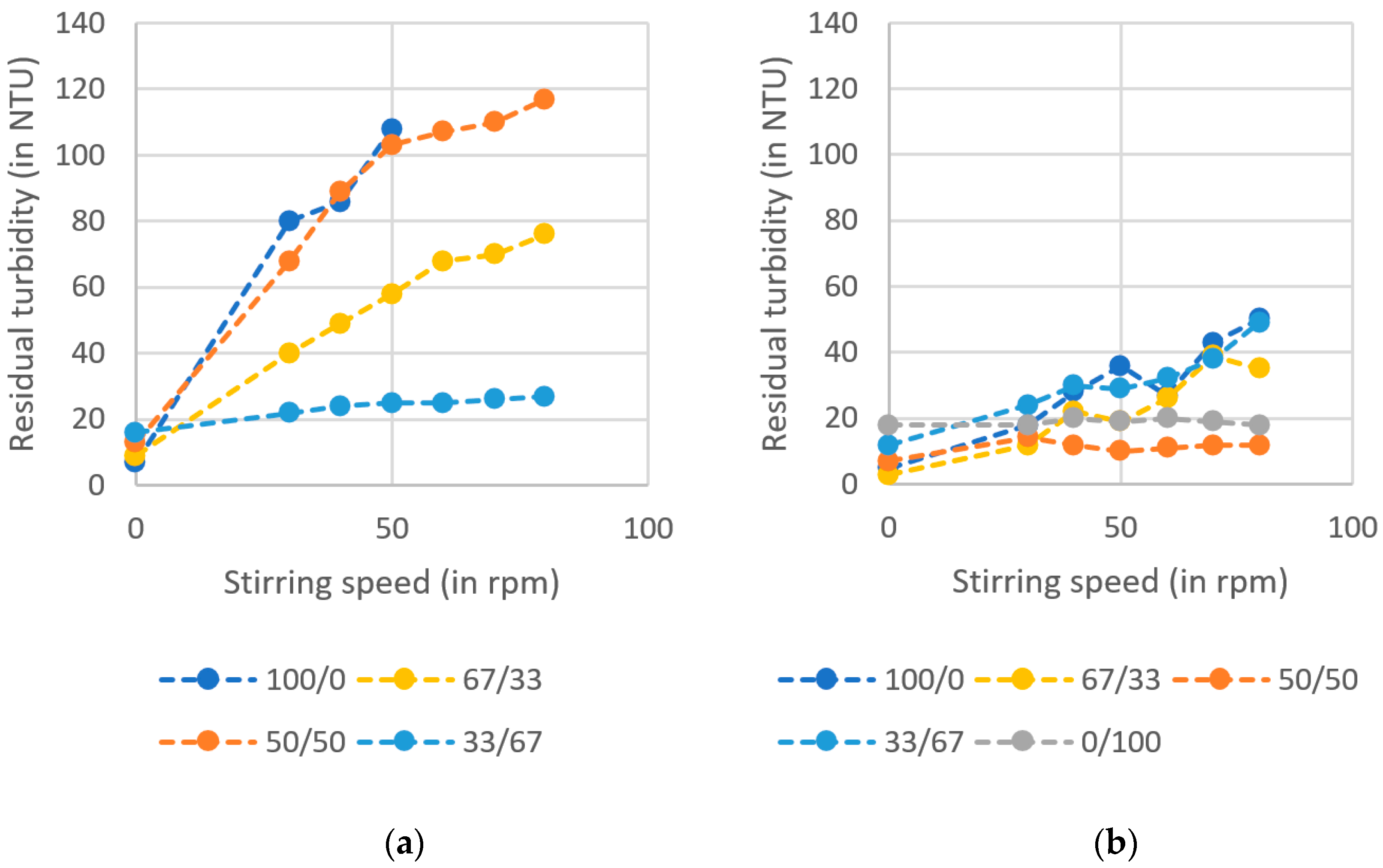
After completion of the previously described test procedure, additional shear tests were carried out. For this purpose, the beaker with the highest flocculation activity was selected for each experiment. In this process, the stirrer was turned on again for one minute and then sedimentation was allowed to resume. The sedimentation time selected was based on the sedimentation time measured in the jar test, during which most of the flocs had settled. After this time, the turbidity in the beaker was measured again. If flocs were mechanically damaged by the renewed energy input, increased residual turbidity resulted. The residual turbidity readings were plotted against the set stirring speeds, giving a visual representation of the shear strength of the flocs. The following stirring speeds were selected for the shear strength tests: 30 rpm, 40 rpm, 50 rpm, 60 rpm, 70 rpm, and 80 rpm.
4. Discussion
In contrast to the experiments with surface water, the flocculant of cactus showed very low or no flocculation activity in the model suspension (see
Figure 2b). According to the results of [
19], one explanation for this is the absence of organic material. The authors concluded that the flocculation process was positively influenced or even enabled by organic material. Flocculation in water with purely mineral sediments would at least result in smaller diameters of the flocs. Since organic material was completely absent in the model suspension, this is most likely the reason why the cactus flocculant did not form flocs and thus showed hardly any flocculation activity in the model suspension. The already low flocculation activity at dosages between 150 and 250 mg/L reduced again to 0% at flocculant dosages of 300 mg/L, which was most likely due to restabilization of the particles. This effect can also be seen in the right part of
Figure 2. With cactus, as with combined flocculants, the flocculation activity decreased again from a dosage of 200 mg/L, indicating that the optimum or maximum flocculant quantity had been exceeded and that a further increase would only have detrimental effects. With Moringa flocculant, there was no increase in flocculation activity above 200 mg/L. It can be assumed that at higher dosages than those investigated here, the activity also decreased again for the same reasons described.
The reason why the Moringa flocculant nevertheless exhibited flocculation activity in the model suspension was, in turn, the positive charge of the polyelectrolytes in the flocculant. In contrast to the cactus flocculant, the Moringa flocculant has compounds with cationic groups according to [
13,
14,
15]. Due to the mostly negative surface charge of the particles contained in the water, the flocculation effect is enhanced by charge neutralization.
The resulting long sedimentation time could be significantly shortened by the addition of cactus. The shortened time indicates more compact, denser flocs with larger diameters, which could also be confirmed by photographic images. Cactus flocculants or flocculants containing cactus produce denser flocs because the flocculation mechanism of bridging is favored by the long-chain polyelectrolytes [
16,
20]. However, contrary to expectations, no linear dependence of the shear strength of the flocs on the cactus content in the flocculant could be conclusively demonstrated. Due to the use of natural flocculants, it is possible that sometimes more, sometimes less flocculant compound of the materials used was present in the flocculants prepared. Thus, contrary to the expected trend, outliers may occur. This can only be eliminated by further extensive investigations in order to compensate as far as possible for this systematic error by the statistical certainty thus gained.
Based on the findings of the test series shown here in conjunction with the results of [
21], it can be concluded that the flocculating active substances of both raw materials used (Moringa and cactus) are cationic polyelectrolytes. Although Moringa forms smaller, weaker flocs, a high flocculation activity is nevertheless achieved. If sufficient organic matter is present in the water sample used, flocculants from cactus also show high activity and form compact, shear-resistant flocs.
In conclusion, no general rule can be derived from this as to which combination of Moringa and cactus is the best, but it can be said that a combination of Moringa and cactus can offer great advantages over Moringa in terms of shear strength and sedimentation time, and over cactus in terms of turbidity reduction. For example, shear strength was improved by about 76% (at 80 rpm) and sedimentation time was reduced by up to 98%. Turbidity reduction was improved by up to 83% compared to cactus (max 54%). Another advantage over the flocculant from Moringa is the size of the flocs that form. Since they are much larger, they not only sink faster, but are also easier to separate. Nevertheless, there is no universal mixing ratio. For each waterway, preliminary tests must be conducted to determine the optimal mixing ratio of Moringa oleifera and cactus.