Quality Characterization of Groundwater for Drinking Purposes and Its Network Distribution to Assure Sustainability in Southern Region of Saudi Arabia
Abstract
:1. Introduction
2. Materials and Methods
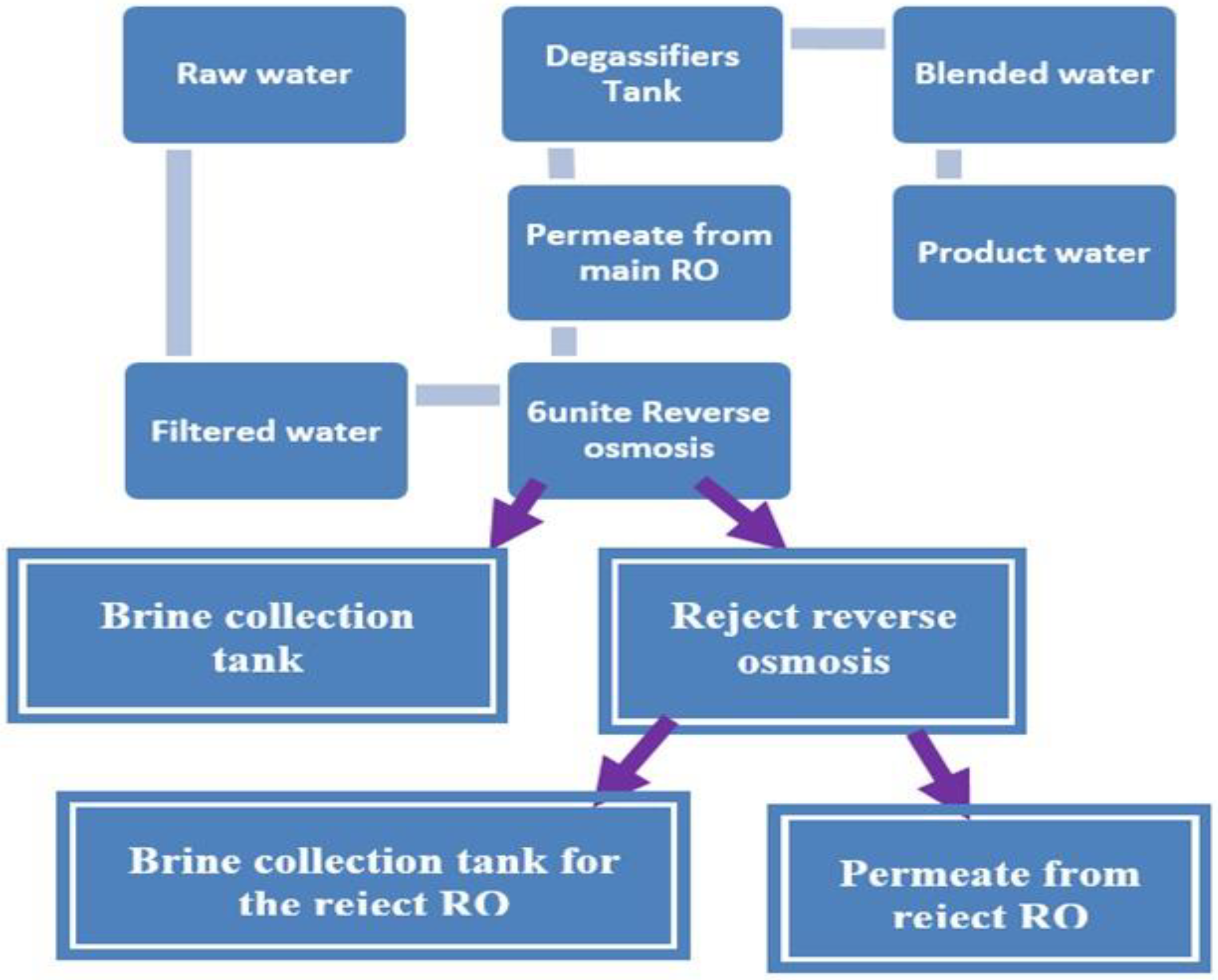
2.1. Al Wajeed Drinking Water Treatment System
2.1.1. The Wells Area
2.1.2. Raw Water Tanks
2.1.3. Raw Water Pumping Station
2.1.4. Primary Treatment with Chemicals (Addition of Coagulant Materials)
- Ferric chloride with a concentration of 5 ppm, is injected at the entrance of the raw water tanks, as coagulant materials to collect suspended particles.
- Sodium meta bisulfite is added as 0.1 mg/L to the water, to remove the residual chlorine before entering the membranes, as chlorine is the oxidizing agent for the membrane material.
- Then acid and an anti-scaling agent are added to prevent the scaling of salts inside the membranes.
2.1.5. Sand Filters
2.1.6. Reverse Osmosis Unit
- Cartridge filters 5 µm to remove suspended particles.
- 6 reverse osmosis units which produce 400 m3/h of treated water for each unit (so, reject water reached to 70 m3/h from each unit). Therefore, the maximum possible amount of treated water produced by the plant is 2535 m3/day.
- Final treatment with chemicals: in this process, chemicals are added to the treated water to improve the quality of the water to ensure compliance with the Gulf water standards.
- Addition of sodium carbonate to elevate the pH value up to 7.5–8.5.
2.1.7. Chlorine Addition
2.1.8. Treated Water Tanks
2.1.9. Pumping Station
2.1.10. Booster Pumping Stations
2.1.11. Reservoir of Ashayab Tathleeth Station
2.1.12. Bishah Reservoir
2.2. Sampling
2.3. Analytical Measurements
2.4. Radioactive Materials Measurements
2.5. Statistical Data Analysis
3. Results and Discussion
3.1. Geology and Hydrology of Al Wajeed (Saudi Arabia) Wells Area
3.2. Wells Data of Al Wajeed Water Treatment Plant in Wadi Al Dawaser Governorate
3.3. Physicochemical Criteria
3.4. Microbiological Quality
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CARE. Leaving No One Behind. In The United Nations World Water Development Report; UNESCO—United Nations Educational, Scientific and Cultural Organization: Paris, France, 2019. [Google Scholar]
- UN. Substantive issues arising in the implementation of the International covenant on economic, social and cultural rights. General Comment No. 15, The right to water (arts. 11 and 12 of the International Covenant on Economic, Social and Cultural Rights). United Nations Digit. Libirary 2002, 11, 29. [Google Scholar]
- Hall, R.P.; Van Koppen, B.; Van, H.E. The human right to water: The importance of domestic and productive water rights. Sci. Eng. Ethics 2014, 20, 849–868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lande, L. Eliminating Discrimination and Inequalities in Access to Water and Sanitation; UN-Water, 2015; Available online: https://www.unwater.org/sites/default/files/app/uploads/2018/08/UN-Water_Policy_Brief_Anti-Discrimination_pdf (accessed on 25 September 2022).
- Grönwall, J.; Danert, K. Regarding groundwater and drinking water access through a human rights lens: Self-Supply as a norm. Water 2020, 12, 419. [Google Scholar] [CrossRef] [Green Version]
- Rossi, E.M.; Gerhard, M.I.; Zanella, M.S.; Bogo, M.; Scapin, D.; Oro, D. Assessment of microbiological quality of water wells in rural properties of the city of West of Santa Catarina, Brazil. Resour. Environ. 2012, 24, 164–168. [Google Scholar] [CrossRef]
- Mohsin, M.; Safdar, S.; Asghar, F.; Jamal, F. Assessment of drinking water quality and its impact on residents health in Bahawalpur city. Int. J. Humanit. Soc. Sci. 2013, 3, 114–128. [Google Scholar]
- Zektser, I.; Everett, L. Groundwaters of the World and Their Use; UNESCO Publishing: Paris, France, 2004. [Google Scholar]
- Amore, L. The United Nations World Water Development Report–N 4–Groundwater and Global Change: Trends, Opportunities and Challenges; UNESCO—United Nations Educational, Scientific and Cultural Organization: Paris, France, 2012; Volume 1. [Google Scholar]
- Sadeghi, A.; Galalizadeh, S.; Zehtabian, G.; Khosravi, H. Assessing the change of groundwater quality compared with land-use change and precipitation rate (Zrebar Lake’s Basin). Appl. Water Sci. 2021, 11, 170. [Google Scholar] [CrossRef]
- Sharma, S.; Bhattacharya, A. Drinking water contamination and treatment techniques. Appl. Water Sci. 2017, 7, 1043–1067. [Google Scholar] [CrossRef] [Green Version]
- Sarath Prasanth, S.V.; Magesh, N.S.; Jitheshlal, K.V.; Chandrasekar, N.; Gangadhar, K. Evaluation of groundwater quality and its suitability for drinking and agricultural use in the coastal stretch of Alappuzha District, Kerala, India. Appl. Water Sci. 2012, 2, 165–175. [Google Scholar] [CrossRef] [Green Version]
- Sadeghi, A.; Khaledi, J.; Nyman, P. Rainfall variability and land-use change in arid basins with traditional agricultural practices; insight from ground water monitoring in Northwestern Iran. J. Earth Sci. Clim. Chang. 2018, 9, 498:2. [Google Scholar] [CrossRef]
- Reyes, V.P.; Ventura, M.A.; Amarillo, P.B. Ecotoxicological assessment of water and sediment in areas of Taal Lake with heavy aquaculture practices using Allium cepa and Daphnia magna assay. Philipp. J. Sci. 2022, 151, 969–974. [Google Scholar] [CrossRef]
- Rajaei, F.; Dahmardeh, B.R.; Ahmadisharaf, E.; Galalizadeh, S.; Dudic, B.; Spalevic, V.; Novicevic, R. Application of Integrated Watershed Management Measures to Minimize the Land Use Change Impacts. Water 2021, 13, 2039. [Google Scholar] [CrossRef]
- DeNicola, E.; Aburizaiza, O.S.; Siddique, A.; Khwaja, H.; Carpenter, D.O. Climate change and water scarcity: The case of Saudi Arabia. Ann. Glob. Health 2015, 81, 342–353. [Google Scholar] [CrossRef] [PubMed]
- WHO/UNICEF. Joint Monitoring Program (JMP) Annual report 2007. Available online: https://www.unwater.org/publications/un-water-annual-report-2007 (accessed on 25 September 2022).
- No. 149/2000; GCC, Gulf Cooperation Council, Gulf Standardization Organization (GSO). Unbottled Drinking Water. GSO: Riyadh, Saudi Arabia, 2014.
- WHO. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2011; Volume 216, pp. 303–304. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; APHA, AWWA, WEF, Publication Office: Washington, DC, USA, 2017. [Google Scholar]
- Missimer, T.M.; Teaf, C.; Maliva, R.G.; Thomson, D.A.; Covert, D.; Hegy, M. Natural radiation in the rocks, soils, and groundwater of Southern Florida with a discussion on potential health impacts. Int. J. Environ. Res. Public Health 2019, 16, 1793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- SAS. Statistical Analysis System (SAS) User’s Guide: Statistics; SAS Institute Inc.: Cary, NC, USA, 2004. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Wickham, H. “Package ggplot2.” Create Elegant Data Visualisations Using the Grammar of Graphics; Version 2.1; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 1–189. [Google Scholar]
- Mallick, J.; Singh, C.K.; AlMesfer, M.K.; Singh, V.P.; Alsubih, M. Groundwater Quality Studies in the Kingdom of Saudi Arabia: Prevalent Research and Management Dimensions. Water 2021, 13, 1266. [Google Scholar] [CrossRef]
- Rehman, F.; Cheema, T. Effects of sewage waste disposal on the groundwater quality and agricultural potential of a flood plain near Jeddah, Saudi Arabia. Arab. J. Geosci. 2016, 9, 307. [Google Scholar] [CrossRef]
- Saleem, H.M.; Subyani, A.M.; Elfeki, A. Environmental Impacts on Groundwater of Wadi Bani Malik Jeddah Saudi Arabia. Int. J. Sci. Eng. Res. 2017, 8, 1658–1662. Available online: https://www.ijser.org/onlineResearchPaperViewer.aspx?Environmental-Impacts-on-Groundwater-of-Wadi-Bani-Malik-Jeddah-Saudi-Arabia.pdf (accessed on 8 April 2021).
- Water Atlas of Saudi Arabia (1984 Edition)|Open Library. Available online: https://openlibrary.org/works/OL10946619W/Water_atlas_of_Saudi_Arabia (accessed on 6 March 2021).
- Lloyd, J.W.; Pim, R.H. The hydrogeology and groundwater resources development of the Cambro-Ordovician sandstone aquifer in Saudi Arabia and Jordan. J. Hydrol. 1990, 121, 1–20. [Google Scholar] [CrossRef]
- Alsharhan, A.S.; Rizk, Z.A.; Nairn, A.E.M.; Bakhit, D.W.; Alhajari, S. Hydrogeology of an Arid Region: The Arabian Gulf and Adjoining Areas; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Powers, R.W.; Ramirez, L.F.; Redmond, C.D.; Elberg, E.L. Geology of the Arabian Peninsula Sedimentary Geology of Saudi Arabia; United States Geological Survey: Reston, VA, USA, 1966.
- Loaiza, J.G.; Bustos-Terrones, Y.; Bustos-Terrones, V.; Monjardín-Armenta, S.A.; Quevedo-Castro, A.; Estrada-Vazquez, R.; Rangel-Peraza, J.G. Evaluation of the Hydrochemical and Water Quality Characteristics of an Aquifer Located in an Urbanized Area. Appl. Sci. 2022, 12, 6879. [Google Scholar]
- Guo, Y.; Zhai, Y.; Wu, Q.; Teng, Y.; Jiang, G.; Wang, J.; Liu, S. Proposed APLIE method for ground water vulnerability assessment in karst-phreatic aquifer, Shandong Province, China: A case study. Environ. Earth Sci. 2016, 75, 112. [Google Scholar]
- Odey, M.O.; Ibor, O.R.; Andem, A.B.; Ettah, I.; Chukwuka, A.V. Drinking water quality and risk implications for community health: A case study of shallow water wells and boreholes in three major communities in Northern Cross-River, Southern Nigeria. Hum. Ecol. Risk Assess. Int. J. 2018, 24, 427–444. [Google Scholar] [CrossRef]
- Valko, M.; Morris, H.; Cronin, M.T. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Omar, W.A.; Khalid, H.; Zaghloul, A.A.; Abo-Hegab, A.S. Risk assessment and toxic effects of metal pollution in two cultured and wild fish species from highly degraded aquatic habitats. Arch. Environ. Contam. Toxicol. 2013, 65, 753–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jerome, F.C.; Hassan, A.; Omoniyi-Esan, G.O.; Odujoko, O.O.; Chukwuka, A.V. Metal uptake, oxidativestress and histopathological alterations in gills and hepatopancreas of Callinectesamnicola exposed to industrial effluent. Ecotoxicol. Environ. Saf. 2017, 139, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar, M.; Chandola, V.K.; Kumar, V.; Saini, R.K.; Pant, N.; Kumari, N.; Srivastava, A.; Singh, S.; Singh, R.; et al. Groundwater Quality Issues and Challenges for Drinking and Irrigation Uses in Central Ganga Basin Dominated with Rice-Wheat Cropping System. Water 2021, 13, 2344. [Google Scholar] [CrossRef]
- Kehagia, K.; Koukouliou, V.; Bratakos, S.; Seferlis, S.; Tzoumerkas, F.; Potiriadis, C. Radioactivity monitoring in drinking water of Attika, Greece. Desalination 2007, 213, 98–110. [Google Scholar] [CrossRef]
- Kozlowska, B.; Walencik, A.; Dorda, J.; Przylibski, T.A. Uranium, radium and 40K isotopes in bottled mineral waters from Outer Carpathians, Poland. Radiat. Meas. 2007, 42, 1380–1386. [Google Scholar] [CrossRef]
- Kallithrakas-Kontos, N.G.; Xarchoulakos, D.C.; Boultadaki, P.; Potiriadis, C.; Kehagia, K. Selective membrane complexation and uranium isotopes analysis in tap water and seawater samples. Anal. Chem. 2018, 90, 4611–4615. [Google Scholar] [CrossRef]
- Asadi, E.; Isazadeh, M.; Samadianfard, S.; Ramli, M.F.; Mosavi, A.; Nabipour, N.; Shamshirband, S.; Hajnal, E.; Chau, K.W. Groundwater quality assessment for sustainable drinking and irrigation. Sustainability 2020, 12, 177. [Google Scholar] [CrossRef] [Green Version]
- Hashim, M.; Nor, S.S.M.; Nayan, N.; Mahat, H.; Saleh, Y.; See, K.L.; Norkhaidi, S.B. Analysis of Well Water Quality in the District of Pasir Puteh, Kelantan, Malaysia. Earth Environ. Sci. 2019, 286, 012021. [Google Scholar] [CrossRef]
- Chaves, R.S.; Salvador, D.; Paulo Nogueira, P.; Santos, M.M.; Aprisco, P.; Célia Neto, C.; Cardoso, V.; Benoliel, M.J.; Rodrigues, J.E.; Carneiro, R.N. Assessment of Water Quality Parameters and their Seasonal Behavior in a Portuguese Water Supply System: A 6-year Monitoring Study. Environ. Manag. 2022, 69, 111–127. [Google Scholar] [CrossRef]
- Ober, J.; Karwot, J.; Rusakov, S. TapWater Quality and Habits of Its Use: A Comparative Analysis in Poland and Ukraine. Energies 2022, 15, 981. [Google Scholar] [CrossRef]
- Ji, Y.; Wu, J.; Wang, Y.; Elumalia, V.; Subramani, T. Seasonal Variation of Drinking Water Quality and Human Health Risk Assessment in Hancheng City of Guanzhong Plain, China. Expo. Health 2020, 12, 469–485. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, L.; Huang, T.; Yan, M.; Liu, K.; Miao, Y.; He, H.; Li, S.; Sekar, R. Combined effects of seasonality and stagnation on tap water quality: Changes in chemical parameters, metabolic activity and co-existence in bacterial community. J. Hazard. Mater. 2021, 403, 124018. [Google Scholar] [CrossRef]
- Zabed, H.; Suely, A.; Faruq, G.; Sahu, J.N. Water quality assessment of an unusual ritual well in Bangladesh and impact of mass bathing on this quality. Sci. Total Environ. 2014, 472, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Fadaei, A.; Sadeghi, M. Evaluation and assessment of drinking water quality in Shahrekord, Iran. Res. Environ. 2014, 4, 168–172. [Google Scholar]
- Ibrahim, M.N. Assessing Groundwater Quality for Drinking Purpose inJordan: Application of Water Quality Index. J. Ecolog. Engine 2019, 20, 101–111. [Google Scholar] [CrossRef]
- Zafar, M.M.; Sulaiman, M.A.; Prabhakar, R.; Kumari, A. Evaluation of the suitability of groundwater for irrigational purposes using irrigation water quality indices and geographical information systems (GIS) at Patna (Bihar), India. Int. J. Energy Water Resour. 2022, 1–14. [Google Scholar] [CrossRef]
- Agwu, A.; Avoaja, A.G.; Kalu, A.U. The assessment of drinking water sources in Aba Metropolis, Abia State, Nigeria. Resour. Environ. 2013, 3, 72–76. [Google Scholar]
- Mohamed, A.K.; Dan, L.; Kai, S.; Eldaw, E.; Abualela, S. Evaluating the suitability of groundwater for drinking pur-poses in the North Chengdu Plain, China. E3S Web Conf. 2019, 81, 01006. [Google Scholar] [CrossRef]
- Khan, S.; Shahnaz, M.; Jehan, N.; Rehman, S.; Tahir Shah, M.; Din, I. Drinking water quality and human health risk in Charsadda district, Pakistan. J. Clean. Produc. 2013, 60, 93–101. [Google Scholar] [CrossRef]
- Singh, B.; Chauhan, J.S.; Mohan, A. A construction of water quality index considering physicochemical properties for drinking purposes in a rural settlement: A case study of Gajraula region, Ganga River Basin (North India). Water Sci. Technol. Water Supp. J. 2012, 126, 818–828. [Google Scholar] [CrossRef]
- Al-Zarah, A.I. Evaluation of household drinking water quality in Al-Ahsa city, Saudi Arabia. Rese. J. Environ. Sci. 2014, 8, 62–77. [Google Scholar] [CrossRef] [Green Version]
- U.S.EPA. Fecal Indicator Organism Behavior in Ambient Waters and Alternative Indicators for Tropical Regions; U.S. Environmental Protection Agency, Office of Science and Technology: Washington, DC, USA, 2009.
- Gofti, L.; Zmirou, D.; Murandi, F.S.; Hartemann, P.; Poleton, J.L. Waterborne microbiological risk assessment: A state of the art and perspectives. Rev. Epidemiol. Sante Publique. 1999, 47, 61–73. [Google Scholar] [PubMed]
- Emmanuel, E.; Pierre, M.G.; Perrodin, Y. Groundwater contamination by microbiological and chemical substances released from hospital wastewater and health risk assessment for drinking water consumers. Environ. Intern. J. 2009, 35, 718–726. [Google Scholar] [CrossRef]
- Gala, G.H. Chlorine in water disinfection. Pure Appl. Chem. 1996, 68, 1731–1735. [Google Scholar]
- Badr, E.-S.A.; Al-Naeem, A.A. Assessment of Drinking Water Purification Plant Efficiency in Al-Hassa, Eastern Region of Saudi Arabia. Sustainability 2021, 13, 6122. [Google Scholar] [CrossRef]
- Fazel, H.K.; Abdo, S.M.; Elthagafi, A.M.; Eldosari, S.H.; Baoku, Z.; Safaa, H.M. View of Saudi Arabia strategy for water resources management at Bishah, Aseer Southern Region Water Assessment. Sustainability 2022, 14, 4198. [Google Scholar] [CrossRef]

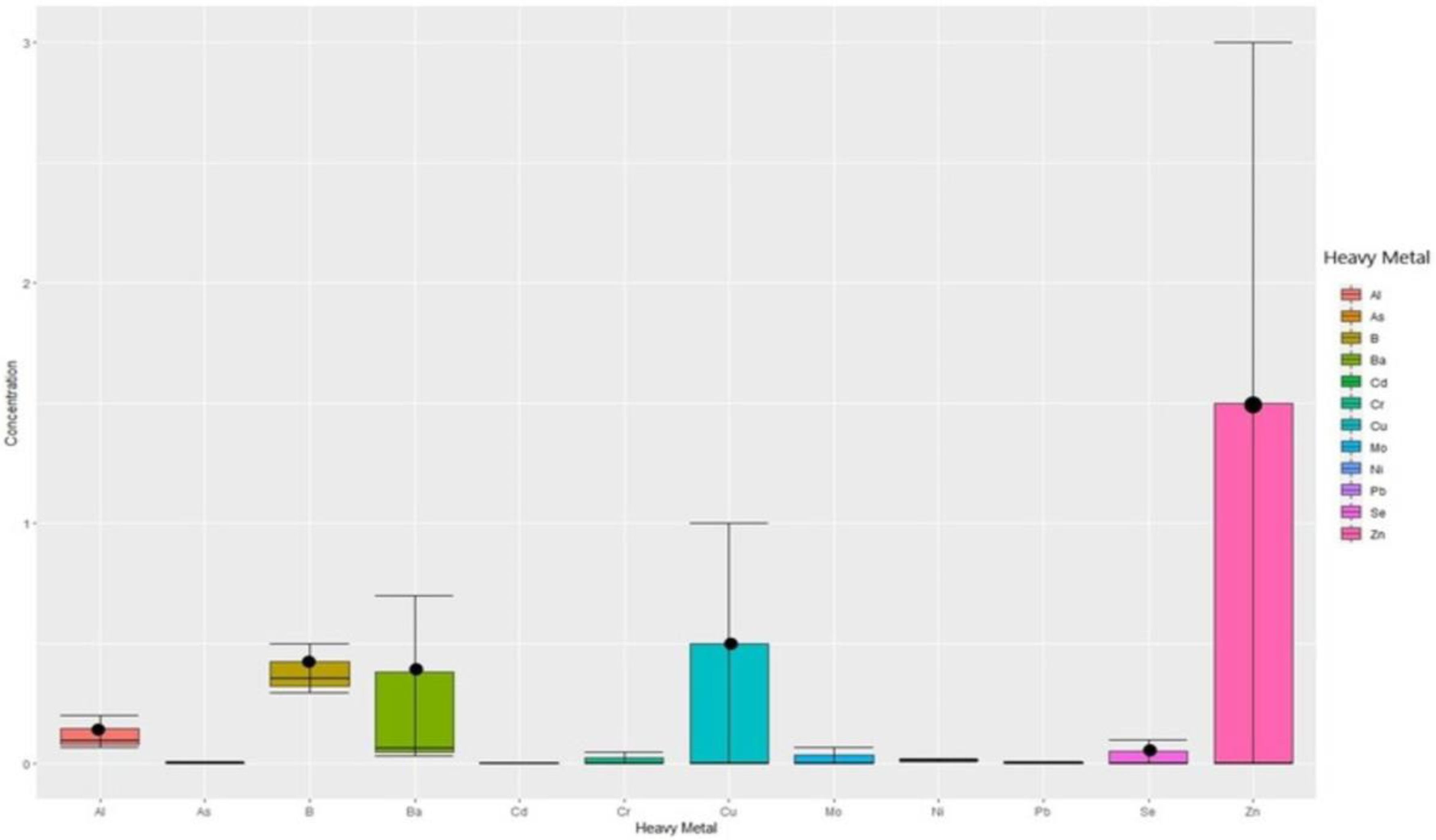



| Item | Mean + SD | WHO (2011) | GCC (2014) |
|---|---|---|---|
| Radium-226 (piC/L) | 11.1 ± 1.80 | ||
| Radium-228 (piC/L) | 12.3 ± 0.81 | ||
| Uranium-234 (mBq/L) | 74.6 ± 3.91 | ||
| Uranium-238 (mBq/L) | 53.6 ± 3.02 | ||
| Total uranium (ppb) | 4.3 ± 0.24 | 300 | 300 |
| Parameter | Unit | 600 2PS | 600 Stage Line | 800 2PS | 2nd Stage Line | WHO (2011) | GCC (2014) |
|---|---|---|---|---|---|---|---|
| pH | 6.75 | 6.70 | 6.95 | 6.80 | 6.5–8.5 | 6.5–8.5 | |
| Color | 10.5 | 10.0 | 8.5 | 6.5 | 15 | 15 | |
| Turbidity | NTU | 0.55 | 0.70 | 0.80 | 0.75 | 5 | 5 |
| TDS | mg/L | 437 | 435 | 439.5 | 445 | 1000 | 1000 |
| Total alkalinity (CaCO3) | mg/L | 77 | 74 | 86 | 58 | ||
| Total hardness (CaCO3) | mg/L | 216 | 225 | 226 | 219 | 500 | 500 |
| Calcium hardness (CaCO3) | mg/L | 179 | 188 | 187 | 186 | ||
| Magnesium hardness (CaCO3) | mg/L | 37 | 37 | 39 | 33 | ||
| Calcium | mg/L | 71.6 | 75.2 | 74.8 | 74.2 | ||
| Magnesium | mg/L | 8.8 | 8.9 | 9.4 | 7.9 | ||
| Chloride | mg/L | 87 | 83 | 82 | 95 | 250 | 250 |
| Sulphate | mg/L | 94 | 93 | 86 | 114 | 250 | 250 |
| Ammonia | mg/L | 0.04 | 0.01 | 0.01 | 0.23 | 1.5 | 1.5 |
| Nitrite | mg/L | 0.16 | 0.03 | 0.02 | 0.03 | 3 | 3 |
| Nitrate | mg/L | 4.0 | 3.0 | 6.5 | 3.5 | 50 | 50 |
| Fluoride | mg/L | 0.42 | 0.27 | 0.29 | 0.27 | 1.5 | 1.5 |
| Iron | mg/L | 0.14 | 0.09 | 0.13 | 0.10 | 0.3 | 0.3 |
| Residual chlorine | mg/L | 0.10 | 0.10 | 0.60 | 0.03 | ||
| Total coliform | MPN/100 mL | −ve | −ve | −ve | −ve | −ve | −ve |
| E. coli | MPN/100 mL | −ve | −ve | −ve | −ve | −ve | −ve |
| Parameter | Unit | King Fahd Station Bishah | Wadi Hergab | Bisha-Net Tank | South Bisha Tank | Bisha Running (400 km) |
|---|---|---|---|---|---|---|
| pH | 7.0 | 7.9 | 7.0 | 7.6 | 8.5 | |
| Color | Co-Pt unit | 4 | 5 | 3 | 4 | 4 |
| Turbidity | NTU | 0.6 | 15.3 | 0.9 | 0.5 | 2.7 |
| TDS | mg/L | 535 | 140 | 543 | 368 | 774 |
| Total alkalinity (CaCO3) | mg/L | 124 | 64 | 122 | 138 | 178 |
| Total hardness (CaCO3) | mg/L | 218 | 102 | 219 | 224 | 206 |
| Calcium hardness (CaCO3) | mg/L | 164 | 88 | 168 | 156 | 167 |
| Magnesium hardness (CaCO3) | mg/L | 54 | 14 | 51 | 61 | 39 |
| Calcium | mg/L | 65.6 | 35.2 | 67.2 | 62.3 | 66.8 |
| Magnesium | mg/L | 13.0 | 3.4 | 12.3 | 14.7 | 9.4 |
| Chloride | mg/L | 102 | 15 | 104 | 77 | 157 |
| Sulphate | mg/L | 90 | 22 | 92 | 67 | 115 |
| Ammonia | mg/L | 0.01 | 0.20 | 0.01 | 0.01 | 0.03 |
| Nitrite | mg/L | 0.02 | 0.01 | 0.02 | 0.01 | 0.05 |
| Nitrate | mg/L | 0.33 | 0.80 | 4.00 | 0.98 | 7.00 |
| Fluoride | mg/L | 3.00 | 0.13 | 0.36 | 0.31 | 0.38 |
| Iron | mg/L | 0.07 | 0.17 | 0.06 | 0.04 | 0.05 |
| Residual chlorine | mg/L | 0.24 | 0.05 | 0.95 | 0.57 | 0.20 |
| Total coliform | MPN/100 mL | −ve | −ve | −ve | −ve | −ve |
| E. coli | MPN/100 mL | −ve | −ve | −ve | −ve | −ve |
| Parameter | Unit | Elsharf Tank | Elmaared Tank | Ashyab King Fahd Dam | Bis-02 | Bis-01 | 800 Stage Line | 800 End-line |
|---|---|---|---|---|---|---|---|---|
| pH | 6.95 | 7.05 | 8.11 | 7.80 | 8.00 | 6.60 | 6.95 | |
| Color | Co/Pt unite | 5.0 | 3.5 | 7.0 | 4.0 | 9.0 | 11.0 | 9.5 |
| Turbidity | NTU | 0.9 | 0.7 | 1.0 | 0.4 | 1.3 | 0.8 | 0.6 |
| TDS | mg/L | 152 | 154 | 469 | 951 | 502 | 440 | 433 |
| Total alkalinity (CaCO3) | mg/L | 50 | 51 | 155 | 174 | 143 | 70 | 88 |
| Total hardness (CaCO3) | mg/L | 88 | 76 | 272 | 417 | 214 | 218 | 225 |
| Calcium hardness (CaCO3) | mg/L | 75 | 68 | 228 | 309 | 163 | 182 | 182 |
| Magnesium hardness (CaCO3) | mg/L | 13 | 8 | 44 | 108 | 51 | 36 | 43 |
| Calcium | mg/L | 30.0 | 27.2 | 91.2 | 123.6 | 65.2 | 72.8 | 72.8 |
| Magnesium | mg/L | 3.0 | 1.9 | 10.6 | 26.0 | 12.2 | 8.6 | 10.2 |
| Chloride | mg/L | 23 | 23 | 124 | 203 | 102 | 88 | 81 |
| Sulphate | mg/L | 28 | 27 | 132 | 197 | 87 | 86 | 85 |
| Ammonia | mg/L | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| Nitrite | mg/L | 0.02 | 0.02 | 0.01 | 0.03 | 0.02 | 0.01 | 0.03 |
| Nitrate | mg/L | 3.0 | 4.5 | 0.8 | 1.0 | 10.0 | 4.0 | 4.0 |
| Fluoride | mg/L | 0.12 | 0.25 | 0.26 | 0.50 | 0.29 | 0.44 | 0.33 |
| Iron | mg/L | 0.12 | 0.11 | 0.02 | 0.02 | 0.03 | 0.10 | 0.11 |
| Residual chlorine | mg/L | 0.02 | 0.01 | 0.04 | 0.01 | 0.05 | 0.24 | 0.10 |
| Total coliform | MPN/100 mL | −ve | −ve | −ve | −ve | −ve | −ve | −ve |
| E. coli | MPN/100 mL | −ve | −ve | −ve | −ve | −ve | −ve | −ve |
| Parameter | Site Effect | SD | p-Value | ||
|---|---|---|---|---|---|
| I | III | III | |||
| N = 15 | N = 12 | N = 21 | |||
| pH | 6.8 | 7.6 | 7.35 | 0.594 | 0.002 |
| Color | 8.9 | 4 | 7.0 | 2.81 | 0.001 |
| Turbidity | 0.70 | 4.0 | 0.8 | 3.64 | 0.022 |
| TDS | 439 | 472 | 443 | 209.0 | 0.909 |
| Total alkalinity (CaCO3) | 74 | 125 | 104 | 44.1 | 0.012 |
| Total hardness (CaCO3) | 222 | 194 | 216 | 78.5 | 0.648 |
| Calcium hardness (CaCO3) | 185 | 149 | 172 | 57.9 | 0.281 |
| Magnesium hardness (CaCO3) | 37 | 44 | 43 | 23.1 | 0.692 |
| Calcium | 74.0 | 59.4 | 69.0 | 23.2 | 0.281 |
| Magnesium | 8.8 | 10.6 | 10.4 | 5.57 | 0.694 |
| Chloride | 87 | 91 | 92 | 47.5 | 0.958 |
| Sulphate | 97 | 77 | 92 | 42.8 | 0.494 |
| Ammonia | 0.07 | 0.05 | 0.01 | 0.070 | 0.036 |
| Nitrite | 0.06 | 0.02 | 0.02 | 0.036 | 0.005 |
| Nitrate | 4.3 | 2.62 | 3.9 | 2.622 | 0.249 |
| Fluoride | 0.31 | 0.84 | 0.31 | 0.680 | 0.058 |
| Iron | 0.12 | 0.08 | 0.07 | 0.045 | 0.035 |
| Residual chlorine | 0.21 | 0.40 | 0.07 | 0.270 | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alramthi, S.M.; Ali, G.H.; Shaban, A.M.; Abdou, T.A.; Elthagafi, A.M.; Eldosari, S.H.; Zhu, B.-K.; Safaa, H.M. Quality Characterization of Groundwater for Drinking Purposes and Its Network Distribution to Assure Sustainability in Southern Region of Saudi Arabia. Water 2022, 14, 3565. https://doi.org/10.3390/w14213565
Alramthi SM, Ali GH, Shaban AM, Abdou TA, Elthagafi AM, Eldosari SH, Zhu B-K, Safaa HM. Quality Characterization of Groundwater for Drinking Purposes and Its Network Distribution to Assure Sustainability in Southern Region of Saudi Arabia. Water. 2022; 14(21):3565. https://doi.org/10.3390/w14213565
Chicago/Turabian StyleAlramthi, Saad M., Gamila H. Ali, Ahmed M. Shaban, Tarek A. Abdou, Atiah M. Elthagafi, Saad H. Eldosari, Bao-Ku Zhu, and Hosam M. Safaa. 2022. "Quality Characterization of Groundwater for Drinking Purposes and Its Network Distribution to Assure Sustainability in Southern Region of Saudi Arabia" Water 14, no. 21: 3565. https://doi.org/10.3390/w14213565
APA StyleAlramthi, S. M., Ali, G. H., Shaban, A. M., Abdou, T. A., Elthagafi, A. M., Eldosari, S. H., Zhu, B.-K., & Safaa, H. M. (2022). Quality Characterization of Groundwater for Drinking Purposes and Its Network Distribution to Assure Sustainability in Southern Region of Saudi Arabia. Water, 14(21), 3565. https://doi.org/10.3390/w14213565







