Environmental Persistence of the Antidepressant Fluoxetine and Its Pharmaceutical Alternative: Kinetics of Oxidation and Mathematical Simulations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Procedures
2.1.1. Chemicals
2.1.2. Natural Water Sampling
2.1.3. Photolysis under Simulated Sunlight
2.1.4. Competition Kinetic Experiments under Simulated Sunlight
2.1.5. Analytical Methods
2.2. Modeling and Simulation
2.2.1. Modeling Photochemical Persistence
2.2.2. Simulation Conditions
3. Results and Discussions
3.1. Photodegradation of Antidepressants under Simulated Sunlight
3.1.1. Direct Photolysis
3.1.2. Indirect Photolysis
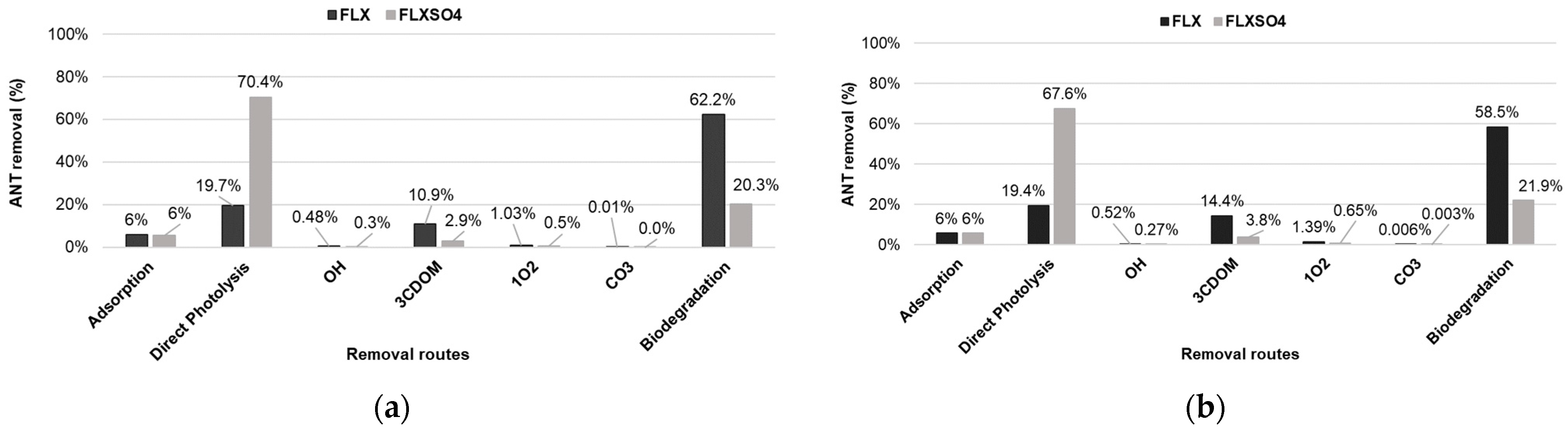
3.2. Photodegradation of Antidepressants in the Water of the Guarapiranga Reservoir
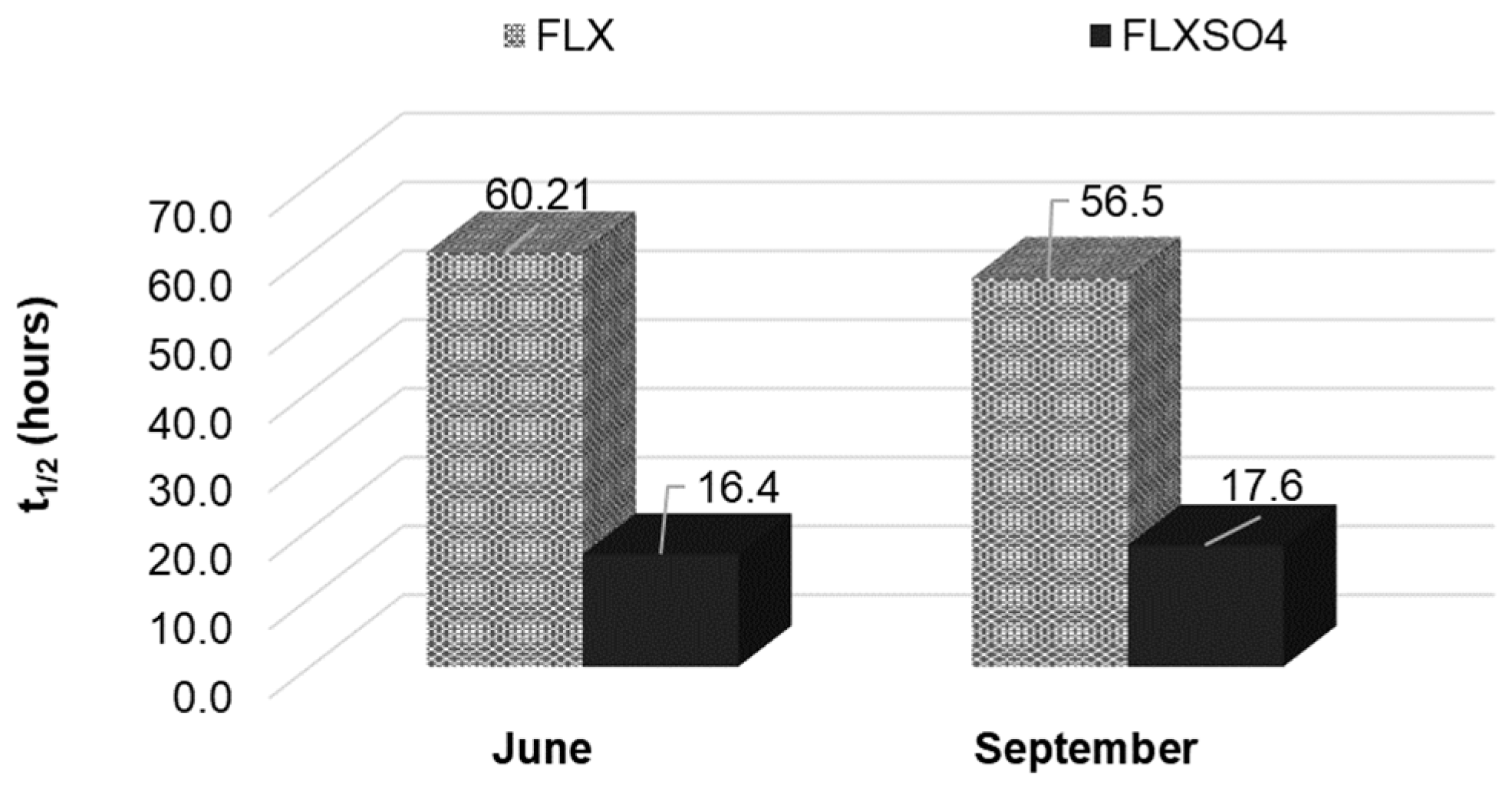
3.3. Photochemical Persistence of FLX versus FLXSO4 in the Guarapiranga Reservoir
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Castillo-Zacarías, C.; Barocio, M.E.; Hidalgo-Vázquez, E.; Sosa-Hernández, J.E.; Parra-Arroyo, L.; López-Pacheco, I.Y.; Barceló, D.; Iqbal, H.N.M.; Parra-Saldívar, R. Antidepressant drugs as emerging contaminants: Occurrence in urban and non-urban waters and analytical methods for their detection. Sci. Total Environ. 2021, 757, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Melchor-Martínez, E.M.; Jiménez-Rodríguez, M.G.; Martínez-Ruiz, M.; Peña-Benavides, S.A.; Iqbal, H.M.N.; Parra-Saldívar, R.; Sosa-Hernández, J.E. Antidepressants surveillance in wastewater: Overview extraction and detection. Case Stud. Chem. Environ. Eng. 2021, 3, 100074. [Google Scholar] [CrossRef]
- Fuentes, A.; Pineda, M.; Venkata, K. Comprehension of top 200 prescribed drugs in the US as a resource for pharmacy teaching, training and practice. Pharmacy 2018, 6, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lajeunesse, A.; Smyth, S.A.; Barclay, K.; Sauvé, S.; Gagnon, C. Distribution of antidepressant residues in wastewater and biosolids following different treatment processes by municipal wastewater treatment plants in Canada. Water Res. 2012, 46, 5600–5612. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Morales, D.; Masis-mora, M.; Montiel, J.R.; Cambronero-heinrichs, J.C.; Briceño, S.; Rojas-Sánchez, C.E.; Méndez-Rivera, M.; Arias-Mora, V.; Tormo-Budowski, R.; Brenes, L.; et al. Occurrence of pharmaceuticals, hazard assessment and ecotoxicological evaluation of WWTPs in Costa Rica. Sci. Total Environ. 2020, 746, 141200. [Google Scholar] [CrossRef] [PubMed]
- Boogaerts, T.; Degreef, M.; Covaci, A.; Van Nuijs, A.L. Development and validation of an analytical procedure to detect spatio-temporal differences in antidepressant use through a wastewater-based approach. Talanta 2019, 200, 340–349. [Google Scholar] [CrossRef]
- Ng, K.T.; Rapp-Wright, H.; Egli, M.; Hartmann, A.; Steele, J.C.; Sosa-Hernández, J.E.; Parra-Saldivar, R. High-throughput multi-residue quantification of contaminants of emerging concern in wastewaters enabled using direct injection liquid chromatography-tandem mass spectrometry. J. Hazard. Mater. 2020, 398, 122933. [Google Scholar] [CrossRef]
- Lindim, C.; van Gils, J.; Georgieva, D.; Mekenyan, O.; Cousins, I.T. Evaluation of Human Pharmaceutical Emissions and Concentrations in Swedish River Basins. Sci. Total Environ. 2016, 572, 508–519. [Google Scholar] [CrossRef]
- Paíga, P.; Santos, L.H.; Ramos, S.; Jorge, S.; Silva, J.G.; Delerue-Matos, C. Presence of Pharmaceuticals in the Lis River (Portugal): Sources, Fate and Seasonal Variation. Sci. Total Environ. 2016, 573, 164–177. [Google Scholar] [CrossRef]
- Cortez, F.S.; Souza, L.S.; Guimarães, L.L.; Pusceddu, F.H.; Maranho, L.A.; Fontes, M.K.; Moreno, B.B.; Nobre, C.R.; Abessa, D.M.S.; Cesar, A.; et al. Marine contamination and cytogenotoxic effects of fluoxetine in the tropical brown mussel Perna perna. Mar. Pollut. Bull. 2019, 141, 366–372. [Google Scholar] [CrossRef]
- Glaser, C.; Zarfl, C.; Werneburg, M.; Böckmann, M.; Zwiener, C.; Schwientek, M. Temporal and spatial variable in-stream attenuation of selected pharmaceuticals. Sci. Total Environ. 2020, 741, 139514. [Google Scholar] [CrossRef] [PubMed]
- Telles-Correia, D.; Guerreiro, D.F.; Coentre, R.; Zuzarte, P.; Figueira, L. Psicofármaco na doença médica: Cardiologia, Nefrologia, Hepatologia. Acta Médica Portuguesa 2009, 22, 797–808. [Google Scholar] [PubMed]
- Fernandes, M.J.; Paíga, P.; Silva, A.; Llaguno, C.P.; Carvalho, M.; Vázquez, F.M.; Delerue-Matos, C. Antibiotics and antidepressants occurrence in surface waters and sediments collected in the north of Portugal. Chemosphere 2020, 239, 124729. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Kumar, P. Wastewater Stabilisation Ponds: Removal of emerging contaminants. J. Sustain. Dev. Energy Water Environ. Syst. 2020, 8, 344–359. [Google Scholar] [CrossRef]
- Mackul’ak, T.; Medvecká, E.; Vojs Staňová, A.; Brandeburová, P.; Grabic, R.; Golovko, O.; Marton, M.; Bodík, I.; Medvedòvá, A.; Gál, M.; et al. Boron doped diamond electrode—The elimination of psychoactive drugs and resistant bacteria from wastewater. Vacuum 2020, 171, 108957. [Google Scholar] [CrossRef]
- Giannakis, S.; Hendaoui, I.; Jovic, M.; Grandjean, D.; Alencastro, L.F.; Girault, H.; Pulgarin, C. Solar photo-Fenton and UV/H2O2 processes against the antidepressant venlafaxine in urban wastewaters and human urine. Intermediates formation and biodegradability assessment. Chem. Eng. J. 2017, 308, 492–504. [Google Scholar] [CrossRef] [Green Version]
- Gornik, T.; Carena, L.; Kosjek, T.; Vione, D. Phototransformation study of the antidepressant paroxetine in surface waters. Sci. Total Environ. 2021, 774, 145380. [Google Scholar] [CrossRef]
- Lastre-Acosta, A.M.; Barberato, B.; Parizi, M.P.S.; Teixeira, A.C.S.C. Direct and indirect photolysis of the antibiotic enoxacin: Kinetics of oxidation by reactive photo-induced species and simulations. Environ. Sci. Pollut. Res. 2019, 26, 4337–4347. [Google Scholar] [CrossRef] [Green Version]
- Bodrato, M.; Vione, D. APEX (Aqueous Photochemistry of Environmentally occurring Xenobiotics): A free software tool to predict the kinetics of photochemical processes in surface waters. Environ. Sci. Process. Impacts 2014, 16, 732–740. [Google Scholar] [CrossRef] [Green Version]
- Vione, D. A Critical view of the application of the APEX software (Aqueous Photochemistry of Environmentally-Occurring Xenobiotics) to predict photoreaction kinetics in surface freshwaters. Molecules 2020, 25, 9. [Google Scholar] [CrossRef]
- Vione, D.; Das, R.; Rubertelli, F.; Maurino, V.; Minero, C.; Barbati, S.; Chiron, S. Modelling the occurrence and reactivity of hydroxyl radicals in surface waters: Implications for the fate of selected pesticides. Int. J. Environ. Anal. Chem. 2010, 90, 260–275. [Google Scholar] [CrossRef]
- Bacilieri, F.; Vahatalo, A.V.; Carena, L.; Wang, M.; Gao, P.; Minella, M.; Vione, D. Wavelength trends of photoproduction of reactive transient species by chromophoric dissolved organic matter (CDOM), under steady-state polychromatic irradiation. Chemosphere 2022, 306, 135502. [Google Scholar] [CrossRef] [PubMed]
- Foote, C.S.; Valentine, J.S.; Greenberg, A.; Liebman, J.F. Active Oxygen in Chemistry, 1st ed.; Blackle Academic and Professional; Springer: Berlin/Heidelberg, Germany, 1995. [Google Scholar] [CrossRef]
- McNeill, K.; Canonica, S. Triplet state dissolved organic matter in aquatic photochemistry: Reaction mechanisms, substrate scope, and photophysical properties. Environ. Sci. Process. Impacts 2016, 18, 1381–1399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calza, P.; Jiménez-Holgado, C.; Coha, M.; Chrimatopoulos, C.; Dal Bello, F.; Medana, C.; Sakkas, V. Study of the photoinduced transformations of sertraline in aqueous media. Sci. Total Environ. 2021, 756, 143805. [Google Scholar] [CrossRef] [PubMed]
- Gornik, T.; Vozic, A.; Heath, E.; Trontelj, J.; Roskar, R.; Zigon, D.; Vione, D.; Kosjek, T. Determination and photodegradation of sertraline residues in aqueous environment. Environ. Pollut. 2020, 256, 113431. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.P.; Sanches-Neto, F.O.; Junior, G.M.Y.; Ramos, B.; Lastre-Acosta, A.M.; Carvalho-Silva, V.H.; Teixeira, A.C.S.C. Photochemical environmental persistence of venlafaxine in an urban water reservoir: A combined experimental and computational investigation. Process Saf. Environ. Prot. 2022, 166, 478–490. [Google Scholar] [CrossRef]
- Lam, M.W.; Young, C.J.; Mabury, S.A. Aqueous photochemical reaction kinetics and transformations of fluoxetine. Environ. Sci. Technol. 2005, 39, 513–522. [Google Scholar] [CrossRef]
- Vione, D.; Scozzaro, A. Photochemistry of surface fresh waters in the framework of climate change. Environ. Sci. Technol. 2019, 53, 7945–7963. [Google Scholar] [CrossRef]
- CETESB. InfoÁGUAS Portal. Environmental Company of the State of São Paulo (Portal InfoÁGUAS. Companhia Ambiental do Estado de São Paulo). Available online: https://sistemainfoaguas.cetesb.sp.gov.br/ (accessed on 30 May 2022).
- Shemer, H.; Sharpless, C.M.; Elovitz, M.S.; Linden, K.G. Relative rate constants of contaminant candidate list pesticides with hydroxyl radicals. Environ. Sci. Technol. 2006, 40, 4460–4466. [Google Scholar] [CrossRef]
- Mostafa, S.; Rosario-Ortiz, F.L. Singlet oxygen formation from wastewater organic matter. Environ. Sci. Technol. 2013, 47, 8179–8186. [Google Scholar] [CrossRef]
- Elovitz, M.S.; Gunten, V.U. Hydroxyl radical ozone ratios during ozonation processes. I. the Rct concept. Ozone Sci. Eng. 1999, 21, 239–260. [Google Scholar] [CrossRef]
- Al Housari, F.; Vione, D.; Chiron, S.; Barbati, S. Reactive photoinduced species in estuarine waters. Characterization of hydroxyl radical, singlet oxygen and dissolved organic matter triplet state in natural oxidation processes. Photochem. Photobiol. Sci. 2010, 9, 78–86. [Google Scholar] [CrossRef]
- Lastre-Acosta, A.M.; Cristofoli, B.S.; Parizi, M.P.S.; Nascimento, C.A.O.; Teixeira, A.C.S.C. Photochemical persistence of sulfa drugs in aqueous medium: Kinetic study and mathematical simulations. Environ. Sci. Pollut. Res. 2021, 28, 23887–23895. [Google Scholar] [CrossRef]
- Gueymard, C.A. SMARTS2: A Simple Model of the Atmospheric Radiative Transfer of Sunshine: Algorithms and Performance Assessment; Rep No. FSEC-PF-270–95; Florida Solar Energy Center: Cocoa, FL, USA, 1995; pp. 1–84. [Google Scholar]
- Šúri, M.; Huld, T.A.; Dunlop, E.D. PV-GIS: A web-based solar radiation database for the calculation of PV potential in Europe. Int. J. Sustain. Energy 2005, 24, 55–67. [Google Scholar] [CrossRef]
- Brown, R. Relationships between suspended solids, turbidity, light attenuation, and algal productivity. Lake Reserv. Manag. 1984, 1, 198–205. [Google Scholar] [CrossRef]
- Pompêo, M.; Padial, P.R.; Mariani, C.F.; Cardoso-Silva, S.; Moschini-Carlos, V.; Silva, D.C.V.R.D.; de Paiva, T.C.B.; Brandimarte, A.L. Biodisponibilidade de metais no sedimento de um reservatório tropical urbano (reservatório Guarapiranga—São Paulo (SP), Brasil): Há toxicidade potencial e heterogeneidade espacial? Geochim. Bras. 2013, 27, 104. Available online: https://www.geobrasiliensis.org.br/geobrasiliensis/article/view/364 (accessed on 30 August 2022). [CrossRef]
- CETESB. Environmental Company of the State of São Paulo (Atlas de Cianobactérias da Bacia do Alto Tietê). Available online: https://www.cetesb.sp.gov.br (accessed on 8 March 2022).
- Strain, H.H.; Thomas, M.R.; Katz, J.J. Spectral absorption properties of ordinary and fully deuteriated chlorophylls-a and b. Biochim. Biophys. Acta 1963, 75, 306–311. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry, 2nd ed.; John Wiley & Sons, Inc.: New Hoboken, NJ, USA, 2003. [Google Scholar]
- Carena, L.; Puscasu, C.G.; Comis, S.; Sarakha, M.; Vione, D. Environmental photodegradation of emerging contaminants: A re-examination of the importance of triplet-sensitised processes, based on the use of 4-carboxybenzophenone as proxy for the chromophoric dissolved organic matter. Chemosphere 2019, 237, 124476. [Google Scholar] [CrossRef]
- Nunes, R.F.; Metolina, P.; Teixeira, A.C.S.C. Dodecylpyridinium chloride removal by persulfate activation using UVA radiation or temperature: Experimental design and kinetic modeling. Environ. Sci. Pollut. Res. 2021, 28, 68229–68243. [Google Scholar] [CrossRef]
- Hora, P.I.; Novak, P.J.; William, A.A. Photodegradation of pharmaceutical compounds in partially nitritated wastewater during UV irradiation. Environ. Sci. Water Res. Technol. 2019, 5, 897–909. [Google Scholar] [CrossRef]
- Liu, W.C.; Hsu, M.H.; Chen, S.Y.; Wu, C.R.; Kuo, A.Y. Water column light attenuation in Danshuei river estuary, Taiwan. J. Am. Water Resour. Assoc. 2005, 41, 425–436. [Google Scholar] [CrossRef]
- Magazinovic, R.S.; Nicholson, B.C.; Mulcahy, D.E.; Davey, D.E. Bromide levels in natural waters: Its relationship to levels of both chloride and total dissolved solids and the implications for water treatment. Chemosphere 2004, 57, 329–335. [Google Scholar] [CrossRef]
- Mansouri, K.; Grulke, C.M.; Judson, R.S.; Williams, A.J. OPERA models for predicting physicochemical properties and environmental fate endpoints. J. Cheminformatics 2018, 10, 10. [Google Scholar] [CrossRef]
 ) Sampling station from which the data used in the analysis were gathered.
) Sampling station from which the data used in the analysis were gathered.
 ) Sampling station from which the data used in the analysis were gathered.
) Sampling station from which the data used in the analysis were gathered.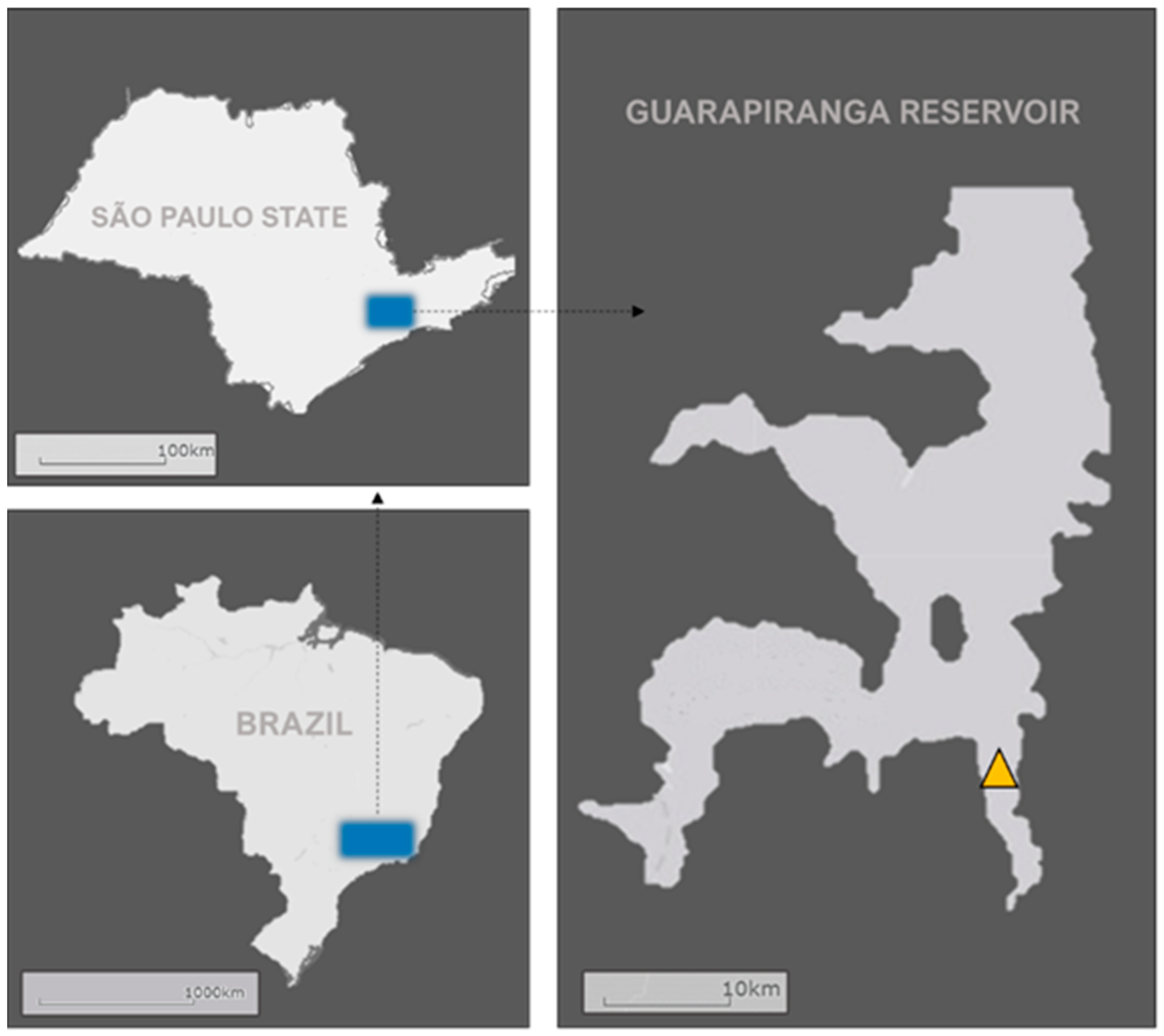
 ) FLXSO4 under simulated solar radiation; (b) Spectral irradiance of the solar simulator (left vertical axis) and spectral decadic molar absorption coefficient of ANT (FLX and FLXSO4) measured in this work (right vertical axis). Conditions: [ANT]0 = (10.33 ± 0.53) mg L−1; natural pH (FLX: ~6.7; FLXSO4: ~6.3); pure water.
) FLXSO4 under simulated solar radiation; (b) Spectral irradiance of the solar simulator (left vertical axis) and spectral decadic molar absorption coefficient of ANT (FLX and FLXSO4) measured in this work (right vertical axis). Conditions: [ANT]0 = (10.33 ± 0.53) mg L−1; natural pH (FLX: ~6.7; FLXSO4: ~6.3); pure water.
 ) FLXSO4 under simulated solar radiation; (b) Spectral irradiance of the solar simulator (left vertical axis) and spectral decadic molar absorption coefficient of ANT (FLX and FLXSO4) measured in this work (right vertical axis). Conditions: [ANT]0 = (10.33 ± 0.53) mg L−1; natural pH (FLX: ~6.7; FLXSO4: ~6.3); pure water.
) FLXSO4 under simulated solar radiation; (b) Spectral irradiance of the solar simulator (left vertical axis) and spectral decadic molar absorption coefficient of ANT (FLX and FLXSO4) measured in this work (right vertical axis). Conditions: [ANT]0 = (10.33 ± 0.53) mg L−1; natural pH (FLX: ~6.7; FLXSO4: ~6.3); pure water.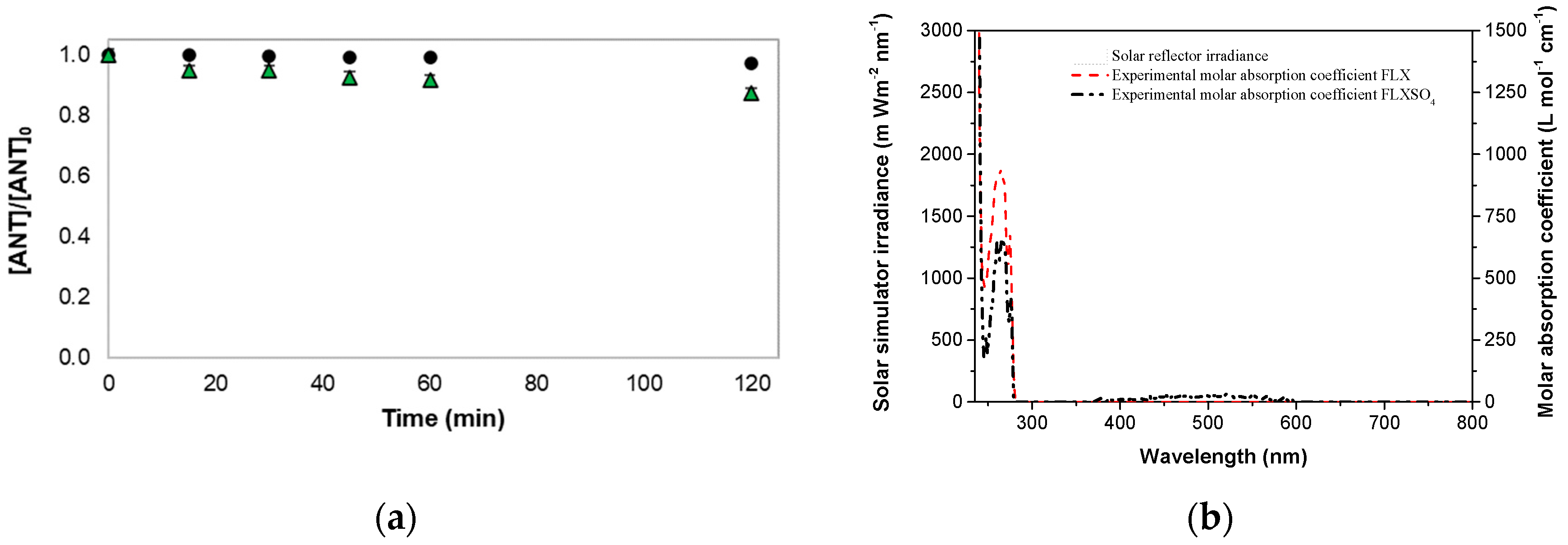
 ) FLX (
) FLX ( ) FLXSO4; Pure water matrix (natural pH: FLX: ~6.7; FLXSO4: ~6.3): (●) FLX (
) FLXSO4; Pure water matrix (natural pH: FLX: ~6.7; FLXSO4: ~6.3): (●) FLX ( ) FLXSO4. [ANT]0 = (10.67 ± 0.49) mg L−1. Experiments were performed in duplicate.
) FLXSO4. [ANT]0 = (10.67 ± 0.49) mg L−1. Experiments were performed in duplicate.
 ) FLX (
) FLX ( ) FLXSO4; Pure water matrix (natural pH: FLX: ~6.7; FLXSO4: ~6.3): (●) FLX (
) FLXSO4; Pure water matrix (natural pH: FLX: ~6.7; FLXSO4: ~6.3): (●) FLX ( ) FLXSO4. [ANT]0 = (10.67 ± 0.49) mg L−1. Experiments were performed in duplicate.
) FLXSO4. [ANT]0 = (10.67 ± 0.49) mg L−1. Experiments were performed in duplicate.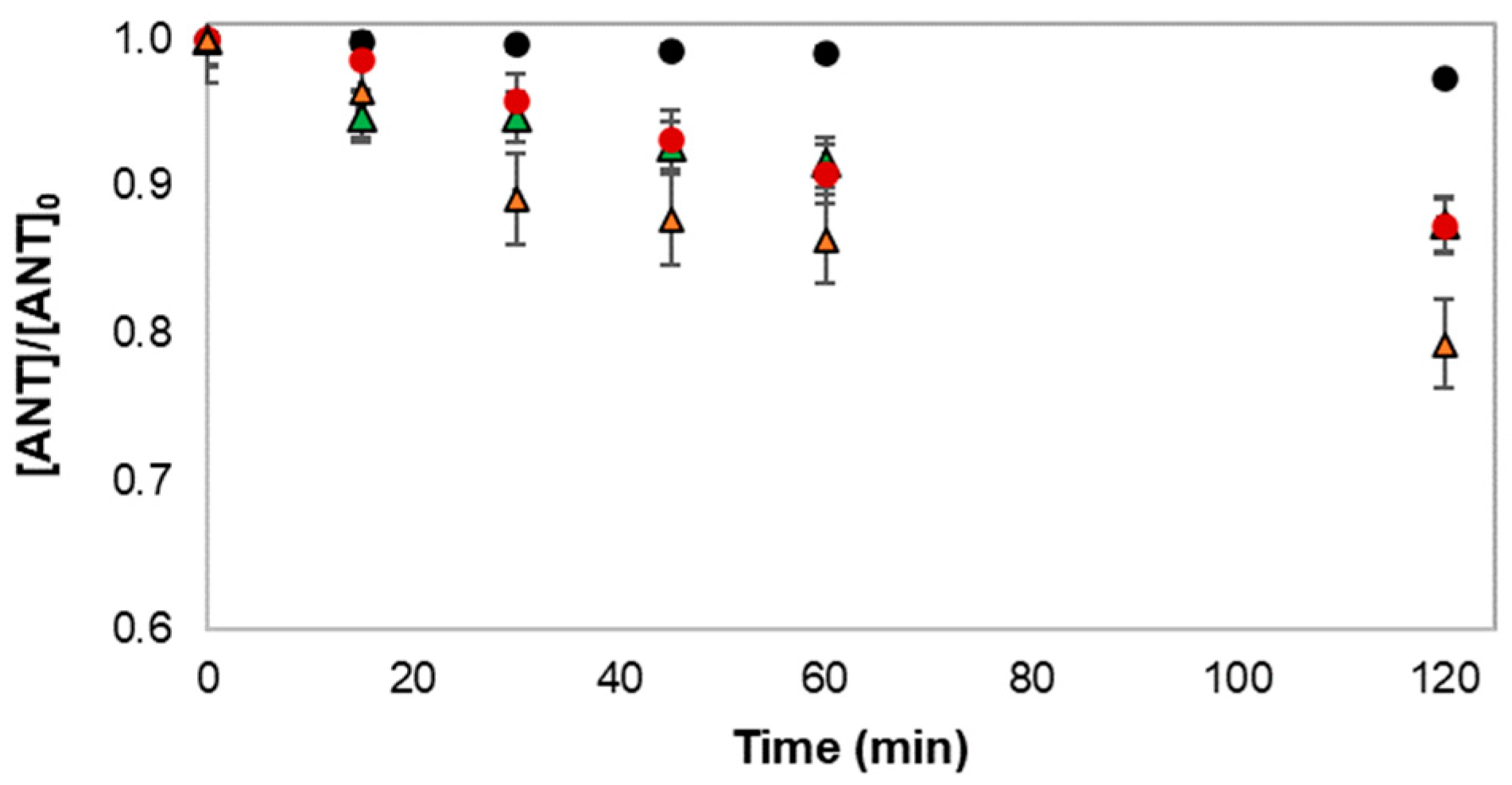
| Parameter | June | September |
|---|---|---|
| Mean depth (m) 1 | 5.7 | 5.7 |
| Turbidity (NTU) 2 | 5.2 | 11 |
| pH2 | 6.5 | 6.8 |
| Total suspended solids (TSS) | 100 | 100 |
| Water temperature (°C) 2 | 18.5 | 23.4 |
| Hardness (mg CaCO3) 2 | 43.1 | 41.2 |
| Nitrates (mg L−1) 2 | 0.32 | 0.72 |
| Nitrites (mg L−1) 2 | 0.12 | 0.11 |
| TOC (mg L−1) 2 | 5.28 | 6.51 |
| Total chlorine (mg L−1) 2 | 16 | 19.7 |
| Chlorophyll-a (µg L−1) 2 | 61.4 | 74.84 |
| Dissolved oxygen (mg L−1) 2 | 6.47 | 6.7 |
| Fraction of organic carbon in solids (fOC) 3 | 0.152 | 0.152 |
| ANT | kANT,HO• (109 L mol−1 s−1) | kANT,1O2 (107 L mol−1 s−1) | kANT,3CBBP* (109 L mol−1 s−1) 1 |
|---|---|---|---|
| FLX | 2.54 ± 0.06 | 1.37 ± 0.07 | 2.67 ± 0.05 |
| FLXSO4 | 3.07 ± 0.03 | 1.63 ± 0.33 | 1.48 ± 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souza, L.P.; Carneiro, J.G.M.; Lastre-Acosta, A.M.; Ramos, B.; Teixeira, A.C.S.C. Environmental Persistence of the Antidepressant Fluoxetine and Its Pharmaceutical Alternative: Kinetics of Oxidation and Mathematical Simulations. Water 2022, 14, 3536. https://doi.org/10.3390/w14213536
Souza LP, Carneiro JGM, Lastre-Acosta AM, Ramos B, Teixeira ACSC. Environmental Persistence of the Antidepressant Fluoxetine and Its Pharmaceutical Alternative: Kinetics of Oxidation and Mathematical Simulations. Water. 2022; 14(21):3536. https://doi.org/10.3390/w14213536
Chicago/Turabian StyleSouza, Larissa P., João G. M. Carneiro, Arlen M. Lastre-Acosta, Bruno Ramos, and Antonio C. S. C. Teixeira. 2022. "Environmental Persistence of the Antidepressant Fluoxetine and Its Pharmaceutical Alternative: Kinetics of Oxidation and Mathematical Simulations" Water 14, no. 21: 3536. https://doi.org/10.3390/w14213536
APA StyleSouza, L. P., Carneiro, J. G. M., Lastre-Acosta, A. M., Ramos, B., & Teixeira, A. C. S. C. (2022). Environmental Persistence of the Antidepressant Fluoxetine and Its Pharmaceutical Alternative: Kinetics of Oxidation and Mathematical Simulations. Water, 14(21), 3536. https://doi.org/10.3390/w14213536








